Submitted:
07 November 2025
Posted:
12 November 2025
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
2.1. Materials
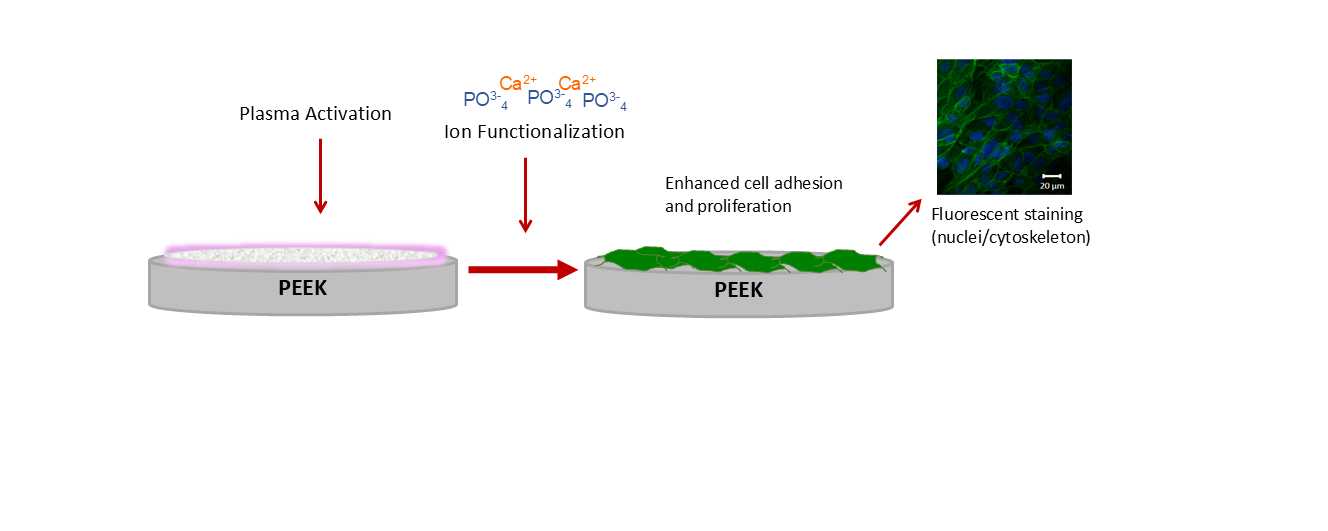
2.2. Sample Fabrication and Surface Modification
2.2.1. Fabrication and Polishing
2.2.2. Surface Activation
2.2.3. Surface Functionalization
2.3. Surface Evaluation
2.3.1. Hydrophilicity
2.3.2. Crystallinity
2.3.3. Topography
2.3.4. Chemical Composition
2.4. Cell Culture Assays
2.4.1. Cell Viability
2.4.2. Cell Proliferation
2.4.3. Cell Adhesion and Spreading
2.5. Statistical Evaluation
3. Results
3.1. Hydrophilicity
3.2. Crystallinity
3.3. Topography
3.4. Chemical Composition
3.5. Cell Culture Assays
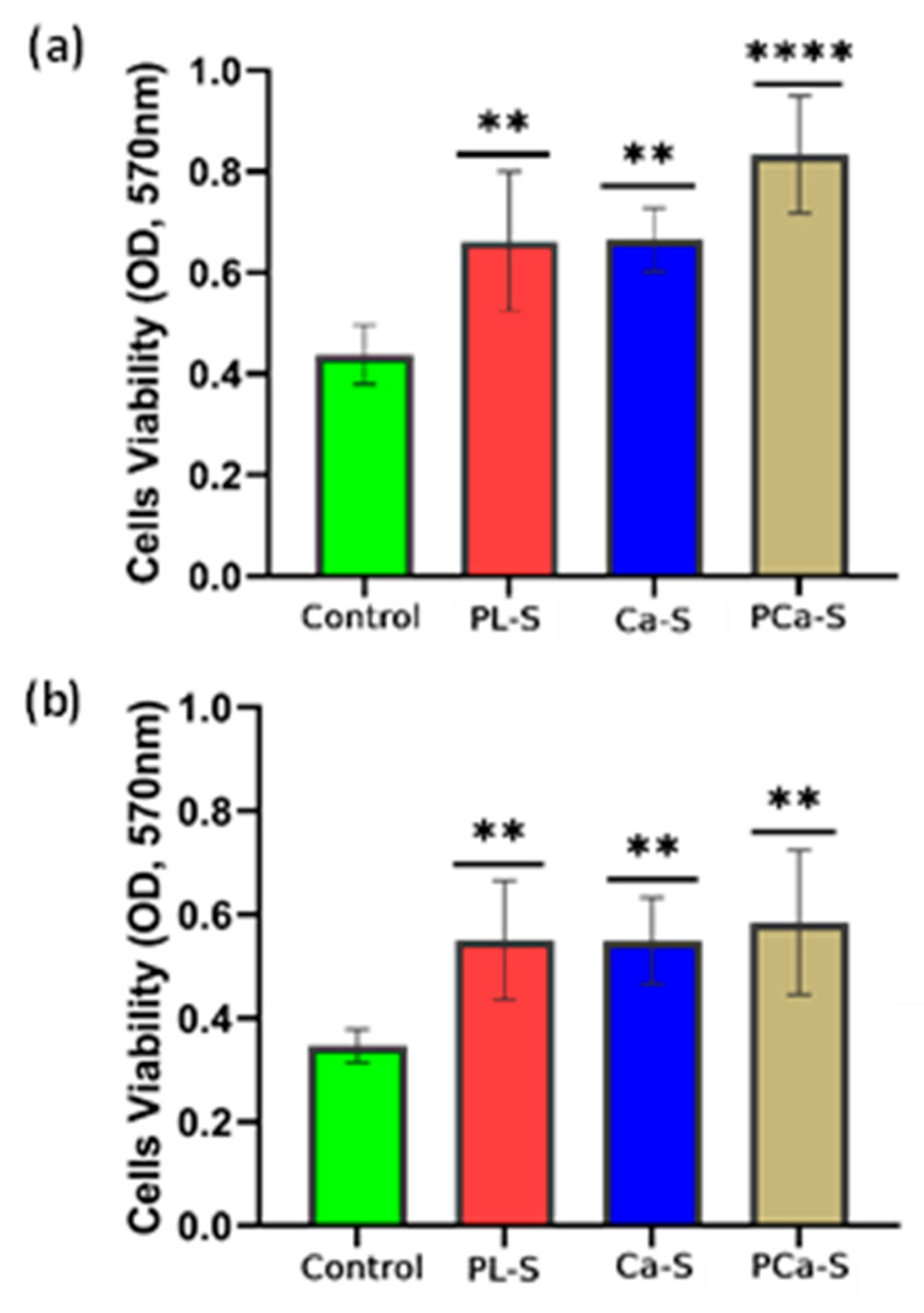
3.5.1. Cell Viability
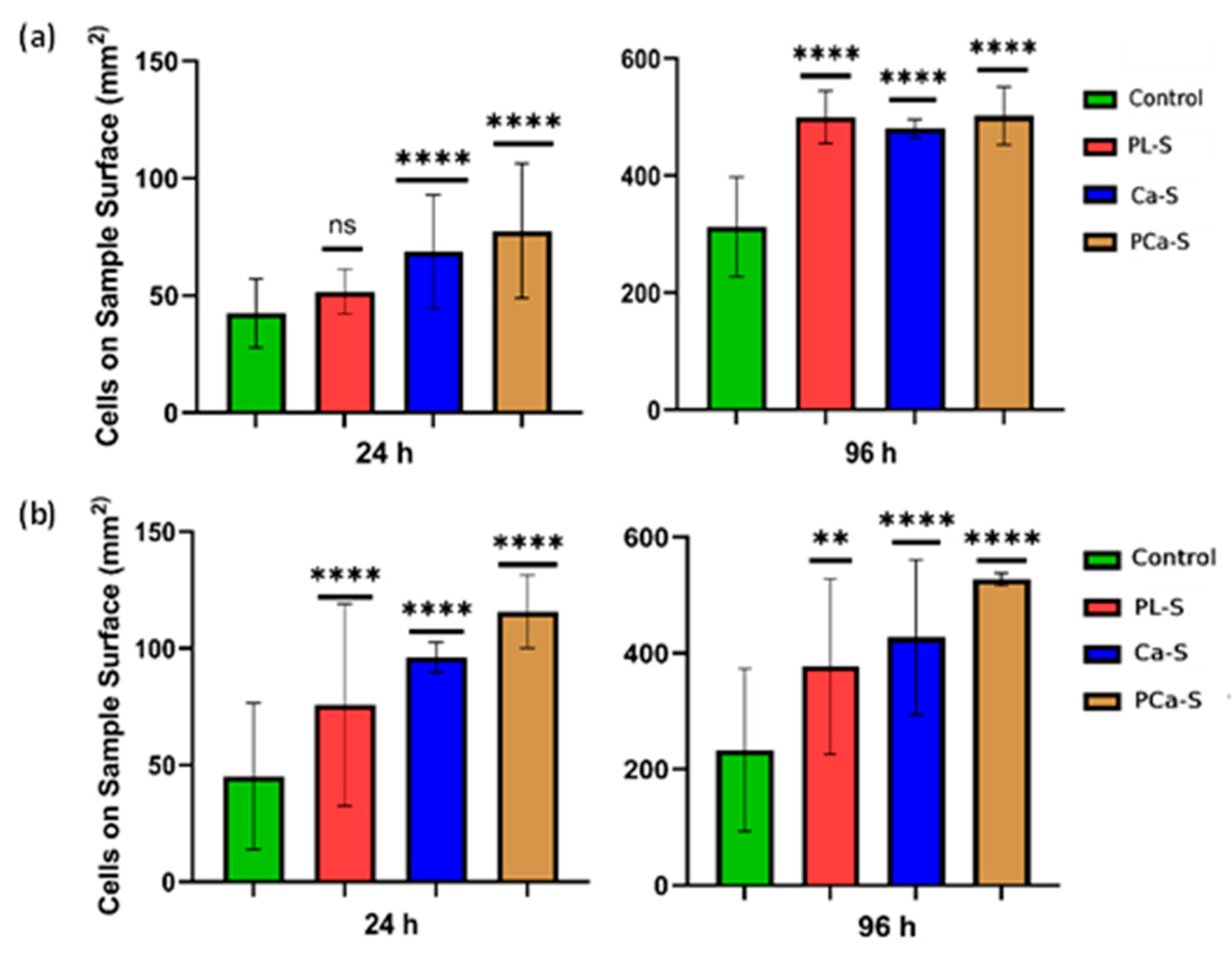
3.5.2. Cell Proliferation
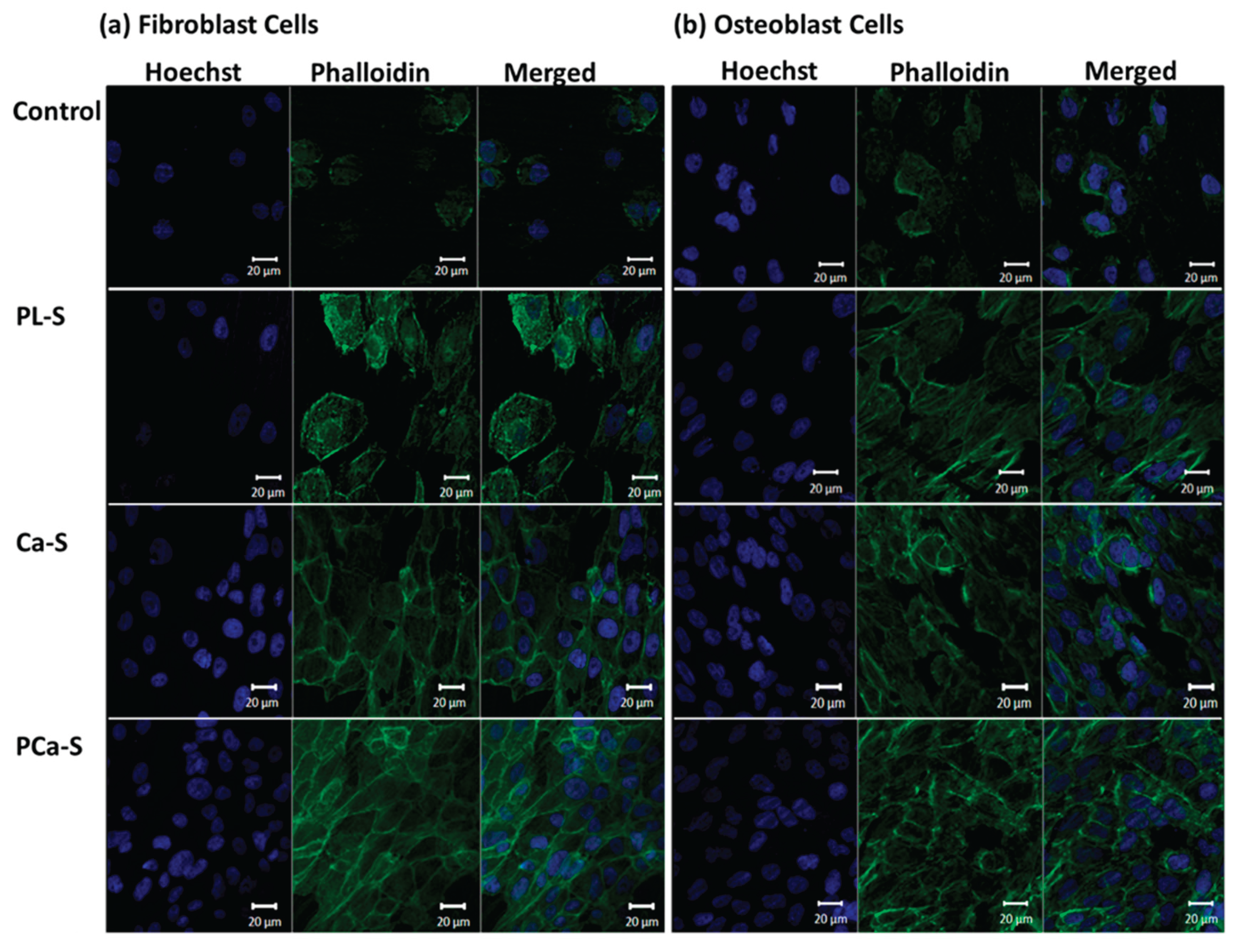
3.5.3. Cell Adhesion and Spreading
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PEEK | Poly ether ether ketone |
| SEM | Scanning Electron Microscopy |
| XPS | X-ray Photoelectron Spectroscopy |
| FT-IR | Fourier Transform Infrared |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide |
| DMEM-F12 | Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 |
References
- A. Haleem, M. Javaid, A. Vaish, R. Vaishya, Three-dimensional-printed polyether ether ketone implants for orthopedics, Indian J Orthop 53 (2019) 377–379. [CrossRef]
- P. Honigmann, N. Sharma, B. Okolo, U. Popp, B. Msallem, F.M. Thieringer, Patient-specific surgical implants made of 3D printed PEEK: Material, technology, and scope of surgical application, Biomed Res Int 2018 (2018). [CrossRef]
- A. Haleem, M. Javaid, Polyether ether ketone (PEEK) and its 3D printed implants applications in medical field: An overview, Clin Epidemiol Glob Health 7 (2019) 571–577. [CrossRef]
- H. Ma, A. Suonan, J. Zhou, Q. Yuan, L. Liu, X. Zhao, X. Lou, C. Yang, D. Li, Y. gang Zhang, PEEK (Polyether-ether-ketone) and its composite materials in orthopedic implantation, Arabian Journal of Chemistry 14 (2021). [CrossRef]
- M. Mbogori, A. Vaish, R. Vaishya, A. Haleem, M. Javaid, Poly-Ether-Ether-Ketone (PEEK) in orthopaedic practice- A current concept review, Journal of Orthopaedic Reports 1 (2022) 3–7. [CrossRef]
- S. Hussain, L. Rutledge, J.G. Acheson, A.R. Boyd, B.J. Meenan, The surface characterisation of polyetheretherketone (Peek) modified via the direct sputter deposition of calcium phosphate thin films, Coatings 10 (2020) 1–26. [CrossRef]
- P. Rendas, L. Figueiredo, M. Geraldo, C. Vidal, B.A. Soares, Improvement of tensile and flexural properties of 3D printed PEEK through the increase of interfacial adhesion, J Manuf Process 93 (2023) 260–274. [CrossRef]
- Q. Fu, M. Gabriel, F. Schmidt, W.D. Müller, A.D. Schwitalla, The impact of different low-pressure plasma types on the physical, chemical and biological surface properties of PEEK, Dental Materials 37 (2021) e15–e22. [CrossRef]
- S. Moharil, A. Reche, K. Durge, Polyetheretherketone (PEEK) as a Biomaterial: An Overview, Cureus (2023). [CrossRef]
- S. Najeeb, M.S. Zafar, Z. Khurshid, F. Siddiqui, Applications of polyetheretherketone (PEEK) in oral implantology and prosthodontics, J Prosthodont Res 60 (2016) 12–19. [CrossRef]
- M. Zhao, M. An, Q. Wang, X. Liu, W. Lai, X. Zhao, S. Wei, J. Ji, Quantitative proteomic analysis of human osteoblast-like MG-63 cells in response to bioinert implant material titanium and polyetheretherketone, J Proteomics 75 (2012) 3560–3573. [CrossRef]
- M. He, Y. Huang, H. Xu, G. Feng, L. Liu, Y. Li, D. Sun, L. Zhang, Modification of polyetheretherketone implants: From enhancing bone integration to enabling multi-modal therapeutics, Acta Biomater 129 (2021) 18–32. [CrossRef]
- F.S.F. Dos Santos, M. Vieira, H.N. da Silva, H. Tomás, M.V.L. Fook, Surface bioactivation of polyether ether ketone (Peek) by sulfuric acid and piranha solution: Influence of the modification route in capacity for inducing cell growth, Biomolecules 11 (2021). [CrossRef]
- S. Cai, C. Wu, W. Yang, W. Liang, H. Yu, L. Liu, Recent advance in surface modification for regulating cell adhesion and behaviors, (2020). [CrossRef]
- A. Obilor, W. Sainsbury, M. Pacella, A. Wilson, V. V. Silberschmidt, Laser Processing of Polymers for Surface Energy Control of Biomedical Implants, in: Procedia CIRP, Elsevier B.V., 2022: pp. 558–563. [CrossRef]
- G. Nageswaran, L. Jothi, S. Jagannathan, Plasma Assisted Polymer Modifications, in: Non-Thermal Plasma Technology for Polymeric Materials: Applications in Composites, Nanostructured Materials, and Biomedical Fields, Elsevier, 2018: pp. 95–127. [CrossRef]
- Sunarso, R. Toita, K. Tsuru, K. Ishikawa, Immobilization of calcium and phosphate ions improves the osteoconductivity of titanium implants, Materials Science and Engineering C 68 (2016) 291–298. [CrossRef]
- L. V. Tapia-Lopez, H.E. Esparza-Ponce, A. Luna-Velasco, P.E. Garcia-Casillas, H. Castro-Carmona, J.S. Castro, Bioactivation of zirconia surface with laminin protein coating via plasma etching and chemical modification, Surf Coat Technol 402 (2020). [CrossRef]
- L. V. Tapia-Lopez, M.A. Luna-Velasco, E.K. Beaven, A.S. Conejo-Dávila, M. Nurunnabi, J.S. Castro, RGD Peptide-Functionalized Polyether Ether Ketone Surface Improves Biocompatibility and Cell Response, ACS Biomater Sci Eng 9 (2023) 5270–5278. [CrossRef]
- J. Jeong, J.H. Kim, J.H. Shim, N.S. Hwang, C.Y. Heo, Bioactive calcium phosphate materials and applications in bone regeneration, Biomater Res 23 (2019). [CrossRef]
- R. Shi, J. Zhang, W. Li, Y. Zhang, Z. Ma, C. Wu, An effective surface modification strategy to boost PEEK osteogenesis using porous CaP generated in well-tuned collagen matrix, Appl Surf Sci 555 (2021). [CrossRef]
- R. Narayanan, S.K. Seshadri, T.Y. Kwon, K.H. Kim, Calcium phosphate-based coatings on titanium and its alloys, J Biomed Mater Res B Appl Biomater 85 (2008) 279–299. [CrossRef]
- H. Mahjoubi, E. Buck, P. Manimunda, R. Farivar, R. Chromik, M. Murshed, M. Cerruti, Surface phosphonation enhances hydroxyapatite coating adhesion on polyetheretherketone and its osseointegration potential, Acta Biomater 47 (2017) 149–158. [CrossRef]
- Sunarso, A. Tsuchiya, R. Toita, K. Tsuru, K. Ishikawa, Enhanced osseointegration capability of poly(Ether ether ketone) via combined phosphate and calcium surface-functionalization, Int J Mol Sci 21 (2020). [CrossRef]
- R. Teng, Y. Meng, X. Zhao, J. Liu, R. Ding, Y. Cheng, Y. Zhang, Y. Zhang, D. Pei, A. Li, Combination of Polydopamine Coating and Plasma Pretreatment to Improve Bond Ability Between PEEK and Primary Teeth, Front Bioeng Biotechnol 8 (2021). [CrossRef]
- T. Jacobs, R. Morent, N. De Geyter, P. Dubruel, C. Leys, Plasma surface modification of biomedical polymers: Influence on cell-material interaction, Plasma Chemistry and Plasma Processing 32 (2012) 1039–1073. [CrossRef]
- A.H.C. Poulsson, R.G. Richards, Surface Modification Techniques of Polyetheretherketone, Including Plasma Surface Treatment, in: PEEK Biomaterials Handbook, Elsevier, 2011: pp. 145–161. [CrossRef]
- P. Chytrosz-Wrobel, M. Golda-Cepa, E. Stodolak-Zych, J. Rysz, A. Kotarba, Effect of oxygen plasma-treatment on surface functional groups, wettability, and nanotopography features of medically relevant polymers with various crystallinities, Applied Surface Science Advances 18 (2023). [CrossRef]
- M.F. Azooz, M.A. Zaghloul, M.M. Sayed, H.M. Soliman, N.M. Mounir, N. Kamal, W.H. El-Dabae, S.E. Ali, Efficiency of MTT and Trypan Blue Assays for Detection of Viability and Recovery of Different Frozen Cell Lines, Egyptian Journal of Veterinary Science (Egypt) 55 (2024) 1649–1657. [CrossRef]
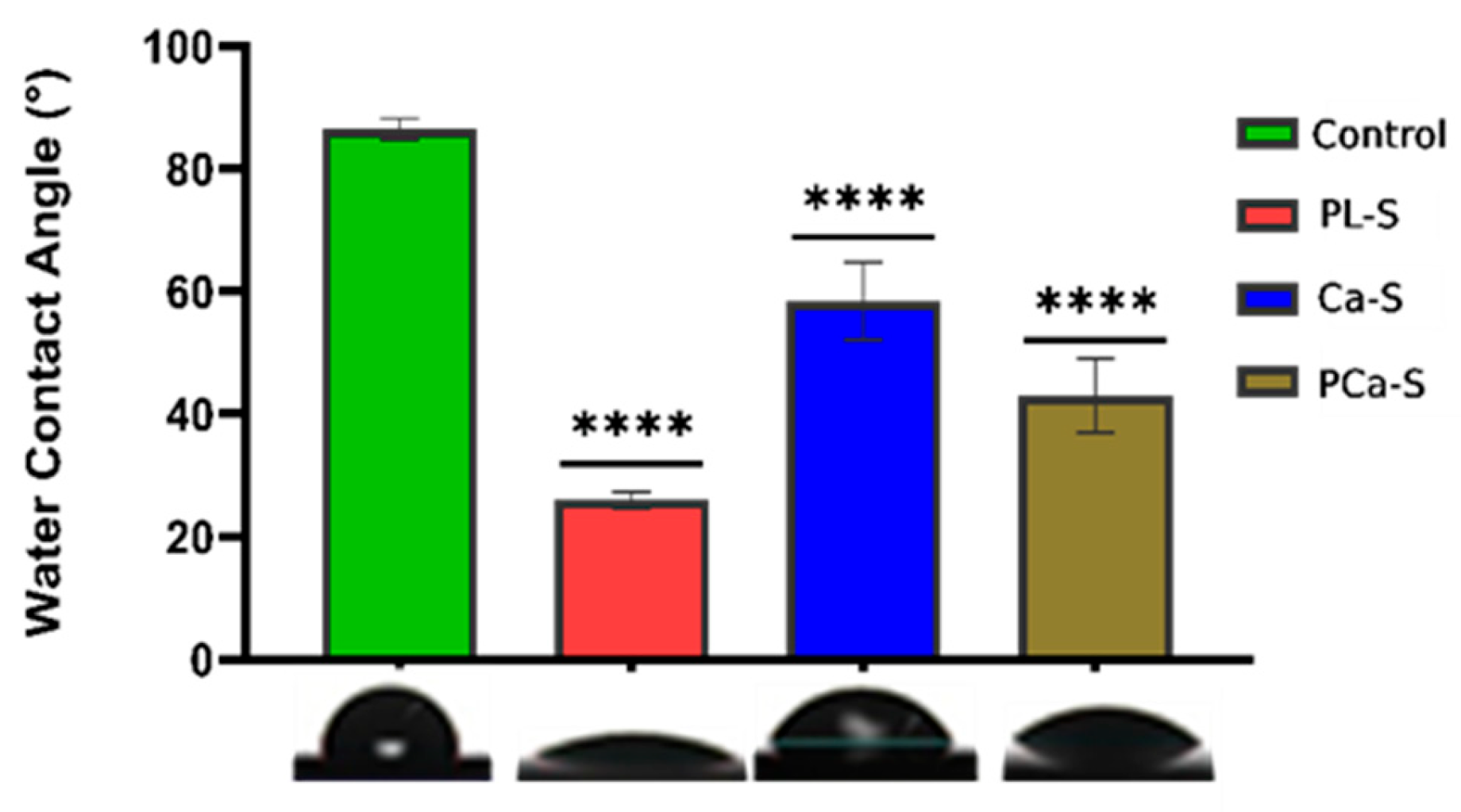
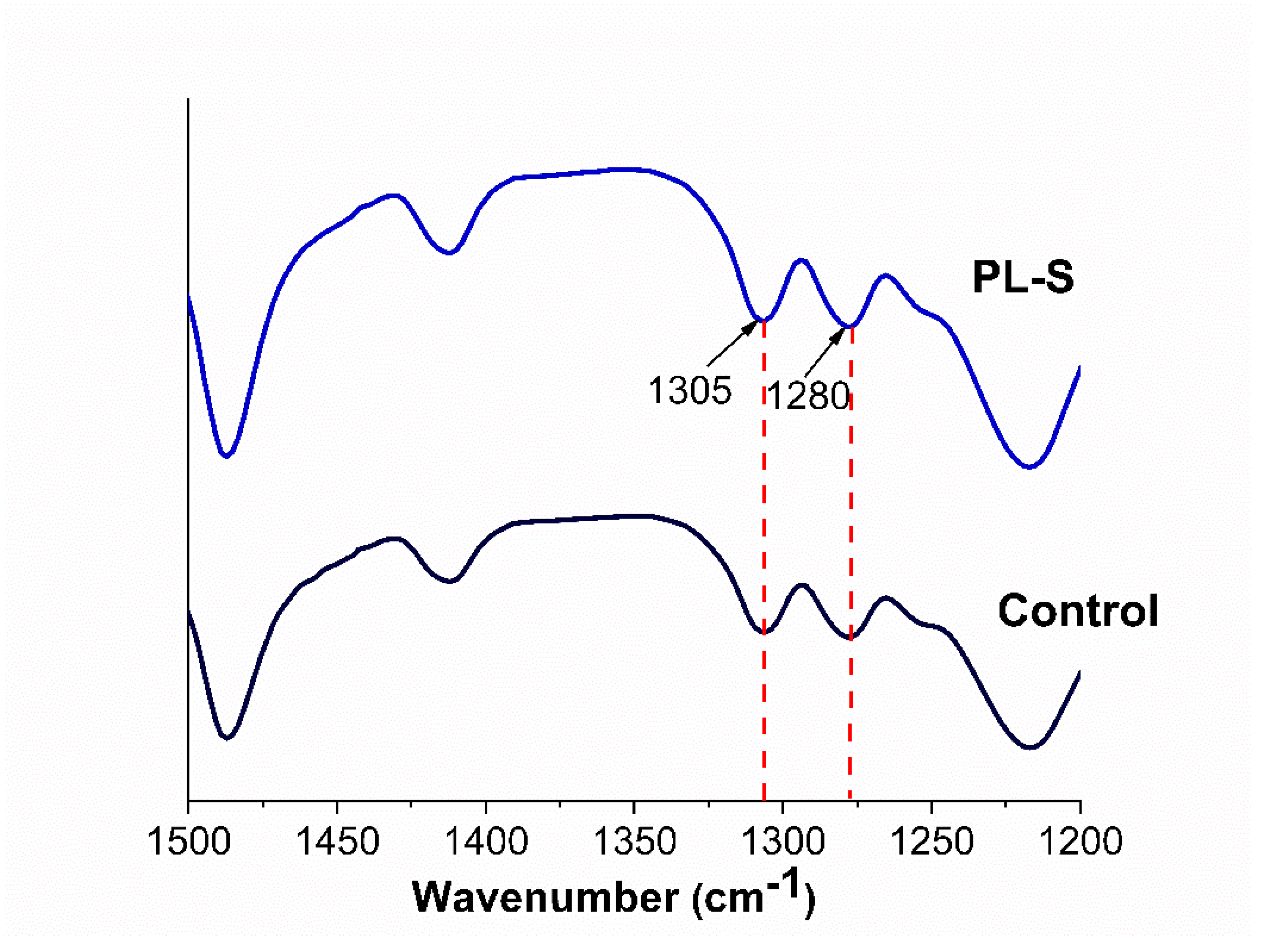

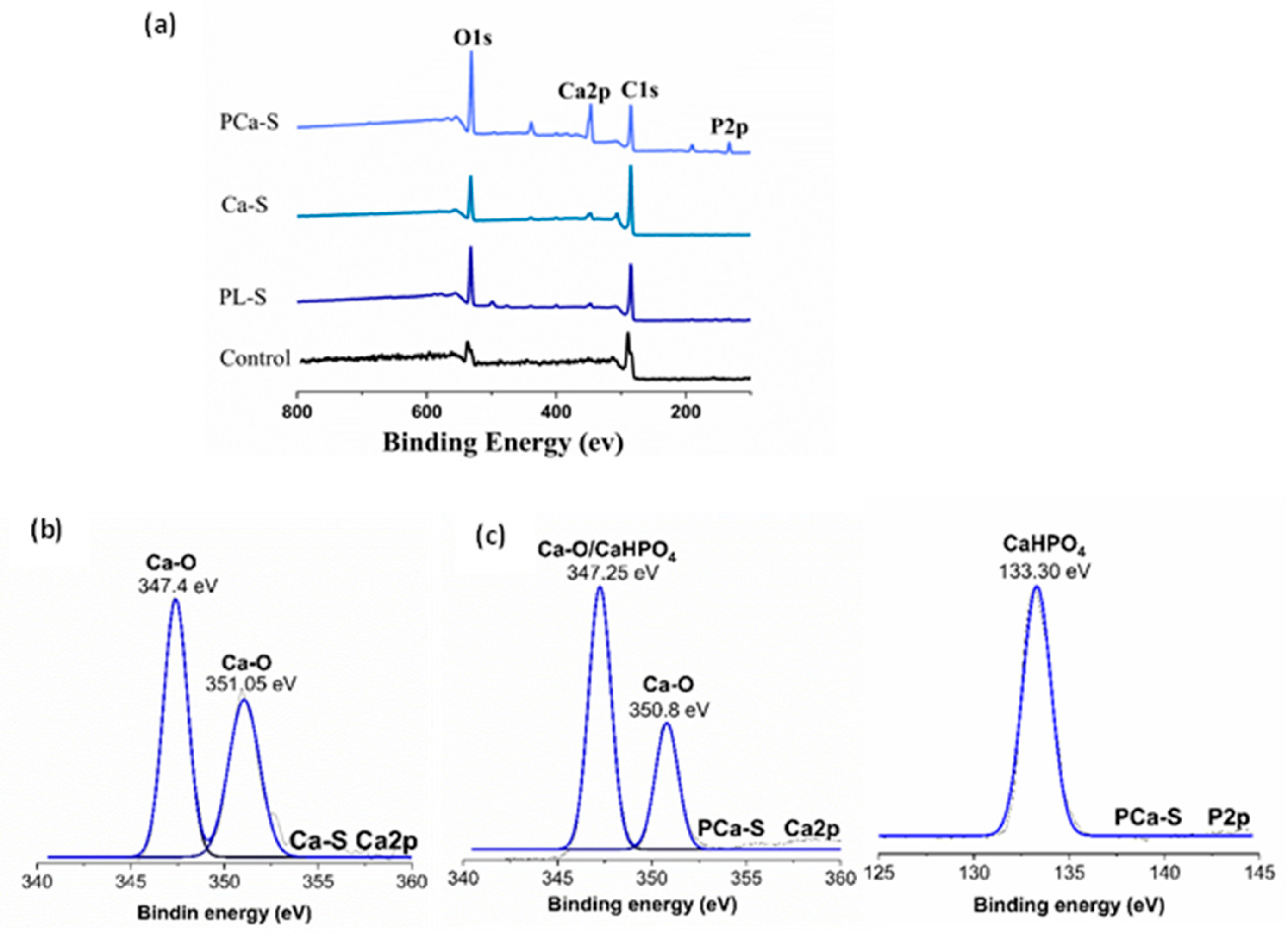
| Label | Name | Surface treatment |
|---|---|---|
| Control | Control sample | No surface treatment |
| PL-S | Plasma sample | Plasma activated surface |
| Ca-S | Calcium sample | Calcium functionalized surface |
| PCa-S | Phosphate and calcium sample | Phosphate and calcium functionalized surface |
| Sample | C1s | O1s | P2p | Ca2p | Ca/P |
|---|---|---|---|---|---|
| P-P | 79.47±0.11 | 20.54±0.11 | |||
| PL-P | 73.27±3.62 | 26.73±3.62 | |||
| Ca-P | 72.69±4.69 | 24.92±3.63 | 2.38±1.12 | ||
| PCa-P | 50.5±3.03 | 34.83±1.52 | 6.47±0.56 | 8.20±1.07 | 1.26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




