Submitted:
07 November 2025
Posted:
11 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
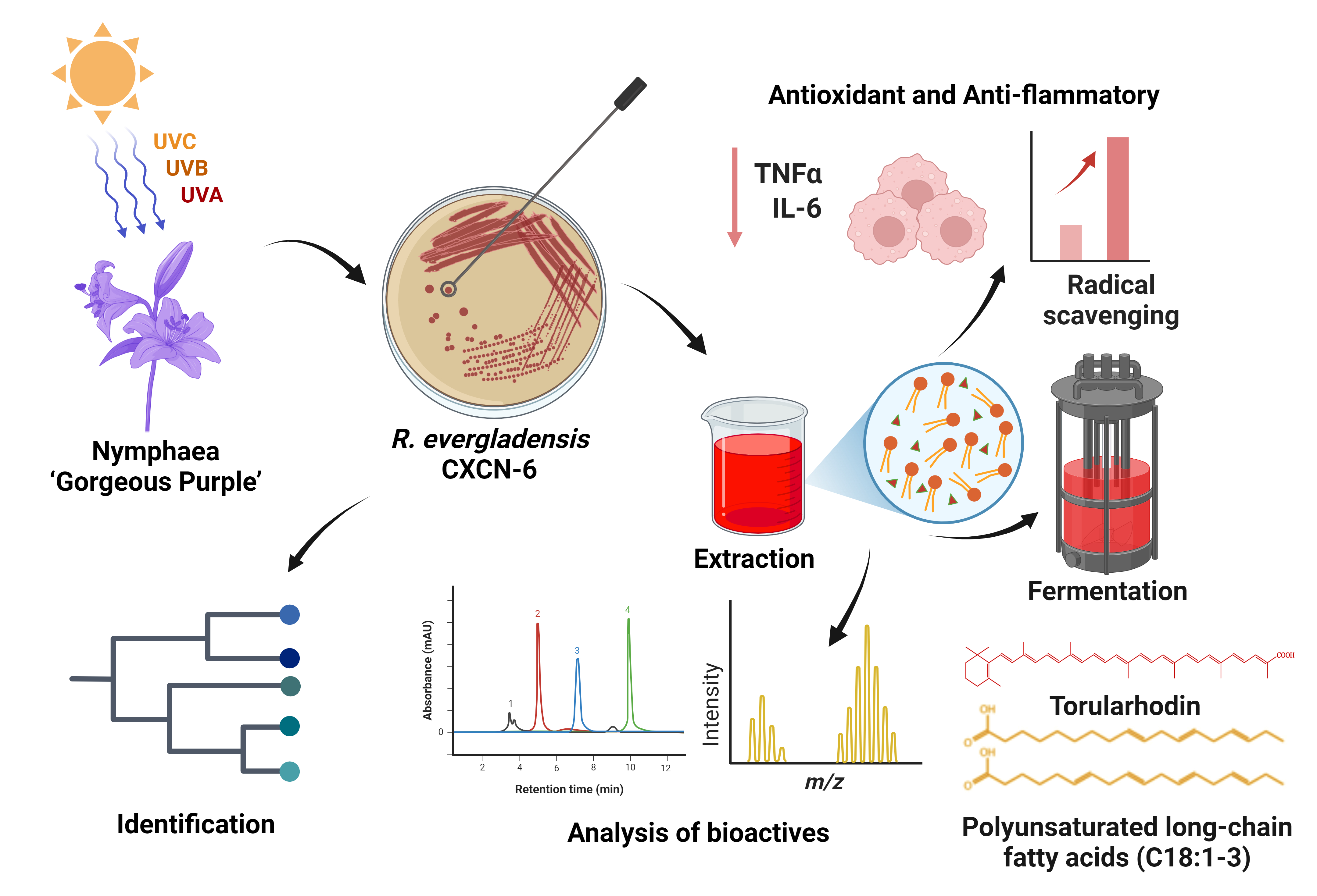
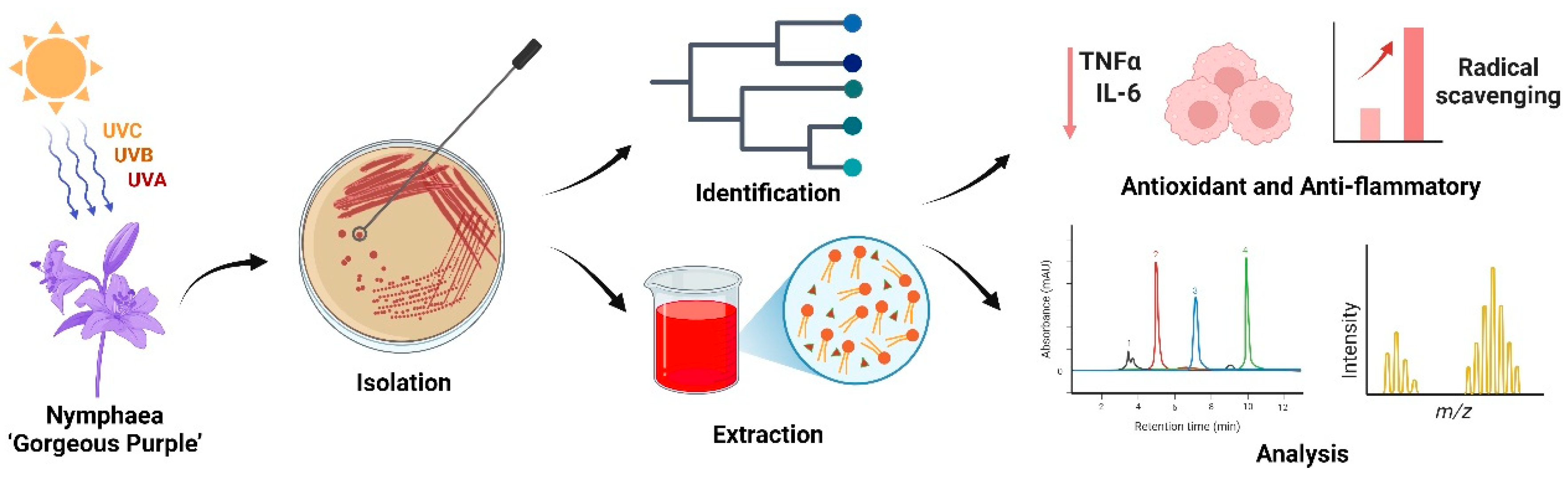
2.1. Sample Collection and Strain Isolation
2.2. Strain Identification
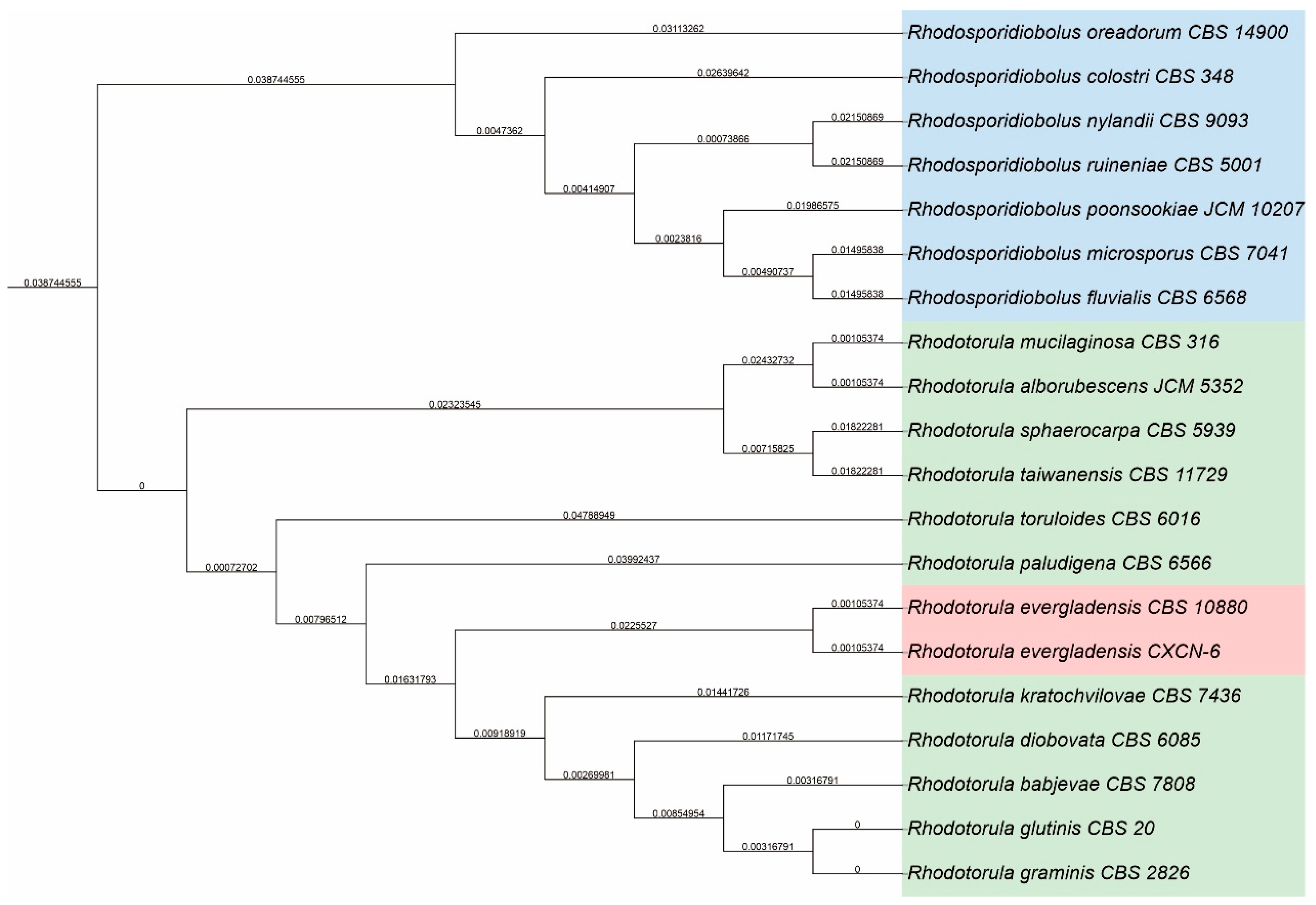
2.3. Phylogenetic Analysis
2.4. Culture Medium and Extract Preparation
2.5. Carotenoid Analysis of R. evergladensis CXCN-6 Extract
2.6. Lipid Analysis of R. evergladensis CXCN-6 Extract
2.8. Antioxidant Activity Assays
2.8.1. DPPH Radical Scavenging Activity
2.8.2. ABTS Radical Cation Scavenging Activity
2.8.3. Intracellular ROS Assay
2.9. Anti-inflammatory Activity Assay
3. Results
3.1. Isolation and Identification of R. evergladensis CXCN-6
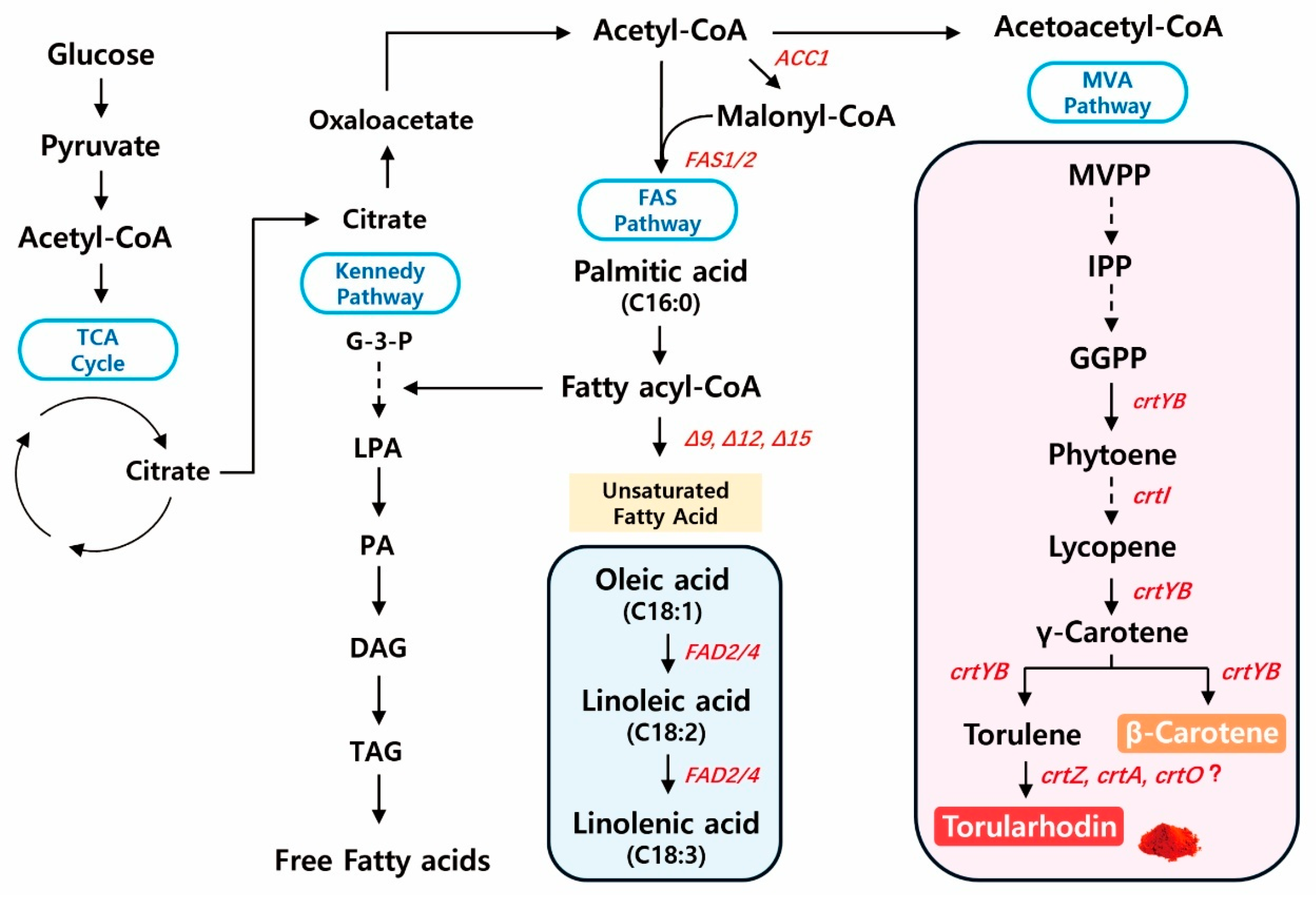
3.2. Whole-Genome Sequencing and Functional Annotation of R. evergladensis CXCN-6
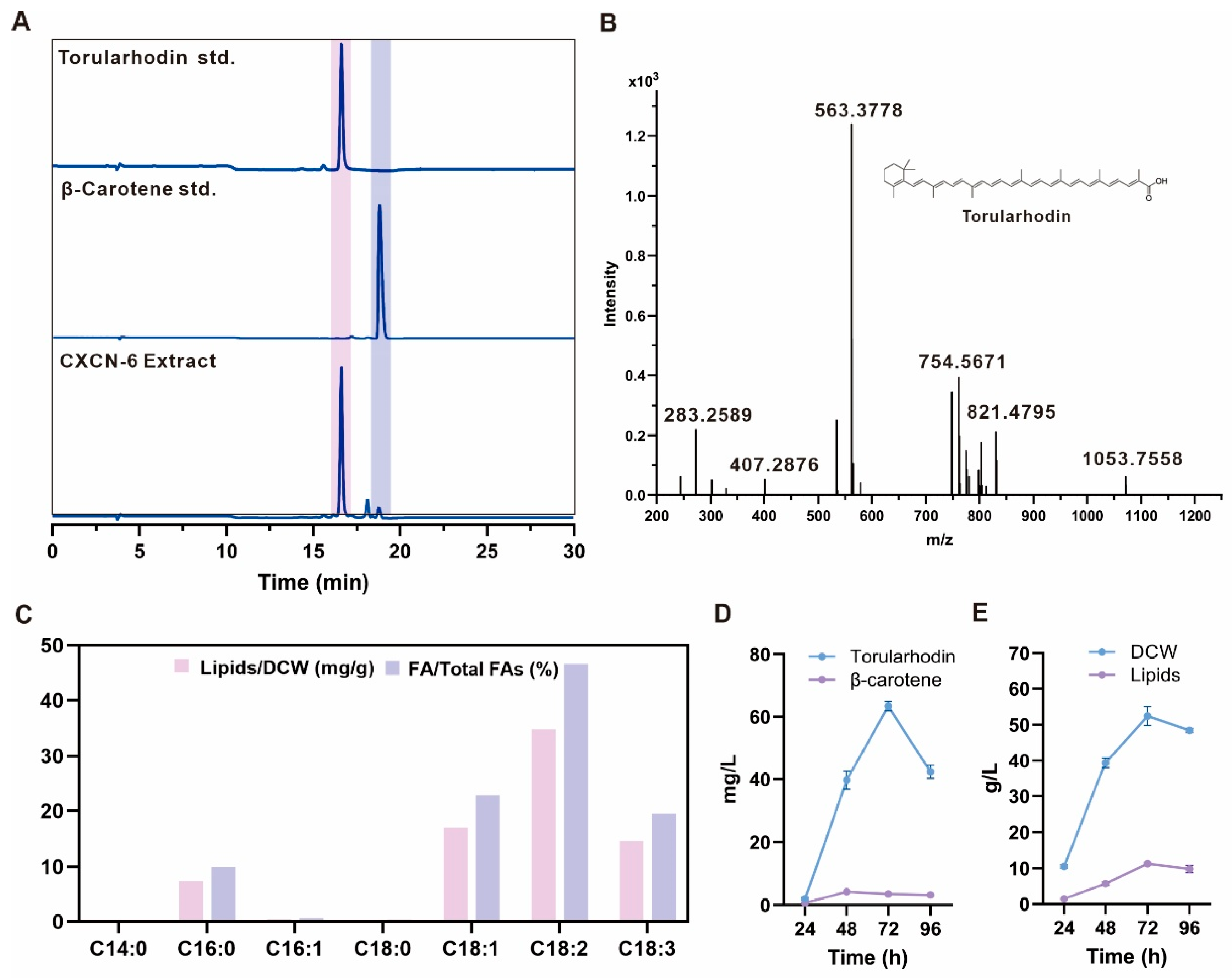
3.3. Torularhodin Production and Spectral Characterization in R. evergladensis CXCN-6
3.4. Fatty Acid Profile of R. evergladensis CXCN-6
3.5. Fermentation Performance of R. evergladensis CXCN-6
3.6. Stability of R. evergladensis CXCN-6 Extracts
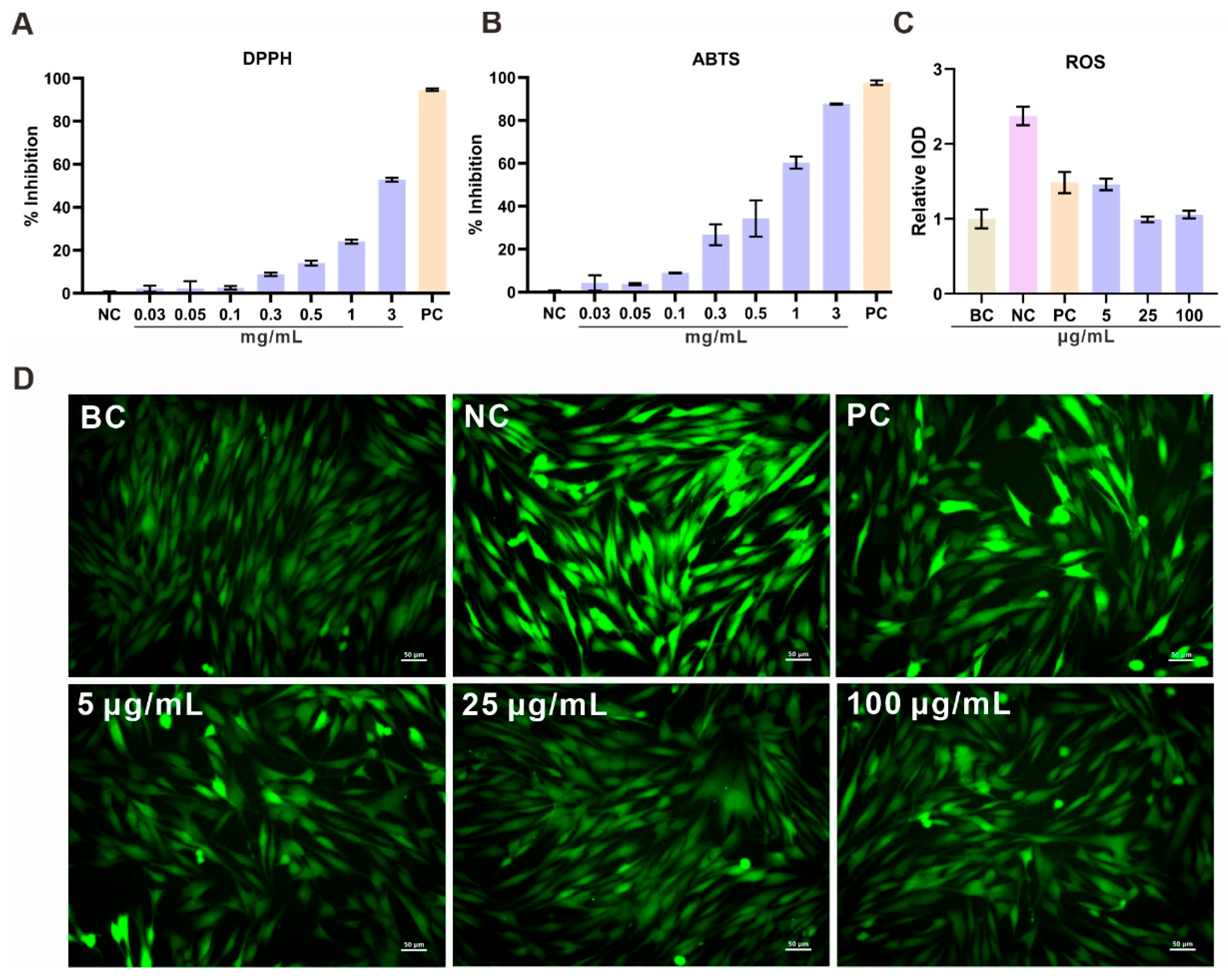
3.7. Antioxidant Activity of R. evergladensis CXCN-6 Extracts
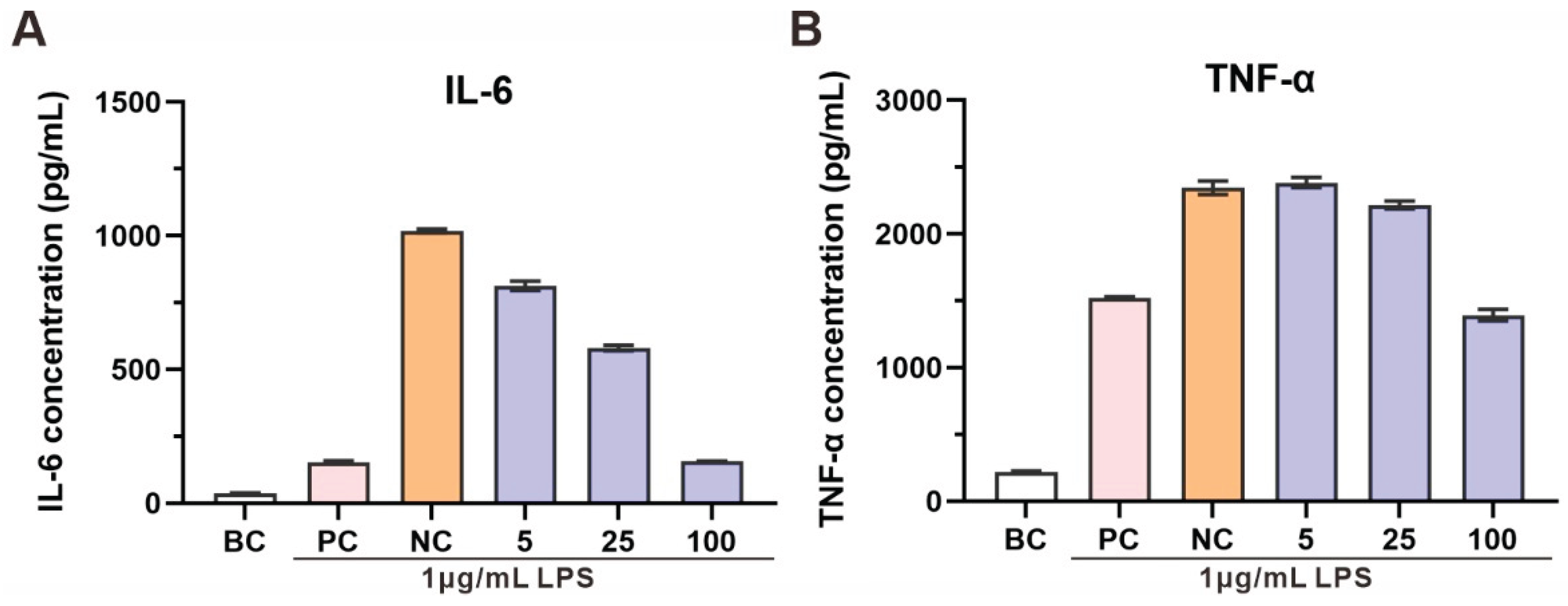
3.8. Anti-inflammatory Activity of R. evergladensis CXCN-6 Extracts
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Maoka, T. Carotenoids as natural functional pigments. J Nat Med 2020, 74, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol Aspects Med 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Frengova, G.I.; Beshkova, D.M. Carotenoids from Rhodotorula and Phaffia: yeasts of biotechnological importance. J Ind Microbiol Biotechnol 2009, 36, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Kot, A.M.; et al. Torulene and torularhodin: “new” fungal carotenoids for industry? Microb Cell Fact 2018, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- Moliné, M.; Libkind, D.; van Broock, M. Production of torularhodin, torulene, and β-carotene by Rhodotorula yeasts. Methods Mol Biol 2012, 898, 275–283. [Google Scholar]
- Libkind, D.; van Broock, M. Biomass and carotenoid pigment production by patagonian native yeasts. World Journal of Microbiology and Biotechnology 2006, 22, 687–692. [Google Scholar] [CrossRef]
- Libkind, D.; Brizzio, S.; van Broock, M. Rhodotorula mucilaginosa, a carotenoid producing yeast strain from a Patagonian high-altitude lake. Folia Microbiol (Praha) 2004, 49, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; et al. Biosynthetic Pathway of Carotenoids in Rhodotorula and Strategies for Enhanced Their Production. J Microbiol Biotechnol 2019, 29, 507–517. [Google Scholar] [CrossRef]
- Ochsenreither, K.; et al. Production Strategies and Applications of Microbial Single Cell Oils. Front Microbiol 2016, 7, 1539. [Google Scholar] [CrossRef]
- Ringel, M.; et al. Sustainable Lipid Production with Cutaneotrichosporon oleaginosus: Insights into Metabolism, Feedstock Valorization and Bioprocess Development. 2025, 13, 1988. [Google Scholar] [CrossRef]
- Ageitos, J.M.; et al. Oily yeasts as oleaginous cell factories. Appl Microbiol Biotechnol 2011, 90, 1219–1227. [Google Scholar] [CrossRef]
- Sitepu, I.R.; et al. Oleaginous yeasts for biodiesel: current and future trends in biology and production. Biotechnol Adv 2014, 32, 1336–1360. [Google Scholar] [CrossRef]
- Budilarto, E.S.; Kamal-Eldin, A. The supramolecular chemistry of lipid oxidation and antioxidation in bulk oils. Eur J Lipid Sci Technol 2015, 117, 1095–1137. [Google Scholar] [CrossRef] [PubMed]
- Boonlao, N.; Ruktanonchai, U.R.; Anal, A.K. Enhancing bioaccessibility and bioavailability of carotenoids using emulsion-based delivery systems. Colloids and Surfaces B: Biointerfaces 2022, 209, 112211. [Google Scholar] [CrossRef]
- Sereti, F.; et al. Green extraction of carotenoids and oil produced by Rhodosporidium paludigenum using supercritical CO2 extraction: Evaluation of cell disruption methods and extraction kinetics. Food Chemistry 2025, 483, 144261. [Google Scholar] [CrossRef]
- Mussagy, C.U.; et al. Selective recovery and purification of carotenoids and fatty acids from Rhodotorula glutinis using mixtures of biosolvents. Separation and Purification Technology 2021, 266, 118548. [Google Scholar] [CrossRef]
- Adadi, P.; Barakova, N.V.; Krivoshapkina, E.F. Selected Methods of Extracting Carotenoids, Characterization, and Health Concerns: A Review. J Agric Food Chem 2018, 66, 5925–5947. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.; et al. Enhanced biomass and lipid production by light exposure with mixed culture of Rhodotorula glutinis and Chlorella vulgaris using acetate as sole carbon source. Bioresource Technology 2022, 364, 128139. [Google Scholar] [CrossRef]
- Lin, P.; et al. Engineered microbial production of carotenoids and their cleavage products: Recent advances and prospects. Biotechnol Adv 2025, 85, 108708. [Google Scholar] [CrossRef]
- Mata-Gómez, L.C.; et al. Biotechnological production of carotenoids by yeasts: an overview. Microb Cell Fact 2014, 13, 12. [Google Scholar] [CrossRef]
- Joshi, K.; Kumar, P.; Kataria, R. Microbial carotenoid production and their potential applications as antioxidants: A current update. Process Biochemistry 2023, 128, 190–205. [Google Scholar] [CrossRef]
- Fonseca, R.S.K.; et al. Exploration of carotenoid-producing Rhodotorula yeasts from amazonian substrates for sustainable biotechnology applications. Current Research in Microbial Sciences 2025, 8, 100373. [Google Scholar] [CrossRef]
- Kot, A.M.; et al. Rhodotorula glutinis-potential source of lipids, carotenoids, and enzymes for use in industries. Appl Microbiol Biotechnol 2016, 100, 6103–6117. [Google Scholar] [CrossRef]
- Park, Y.K.; Nicaud, J.M.; Ledesma-Amaro, R. The Engineering Potential of Rhodosporidium toruloides as a Workhorse for Biotechnological Applications. Trends Biotechnol 2018, 36, 304–317. [Google Scholar] [CrossRef]
- Kumar, S.; et al. MEGA: A biologist-centric software for evolutionary analysis of DNA and protein sequences. Briefings in Bioinformatics 2008, 9, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v6: recent updates to the phylogenetic tree display and annotation tool. Nucleic Acids Res 2024, 52, W78–W82. [Google Scholar] [CrossRef]
- Elfeky, N.; et al. Lipid and Carotenoid Production by Rhodotorula glutinis with a Combined Cultivation Mode of Nitrogen, Sulfur, and Aluminium Stress. 2019. 9 2444. [CrossRef]
- Mishra, S.K.; et al. Rapid quantification of microalgal lipids in aqueous medium by a simple colorimetric method. Bioresource Technology 2014, 155, 330–333. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Re, R.; et al. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-J.; et al. Whole genome sequencing and comparative genomic analysis of oleaginous red yeast Sporobolomyces pararoseus NGR identifies candidate genes for biotechnological potential and ballistospores-shooting. BMC Genomics 2020, 21, 181. [Google Scholar] [CrossRef]
- Kot, A.M.; et al. Effect of exogenous stress factors on the biosynthesis of carotenoids and lipids by Rhodotorula yeast strains in media containing agro-industrial waste. World J Microbiol Biotechnol 2019, 35, 157. [Google Scholar] [CrossRef]
- Mosqueda-Martínez, E.; et al. In Rhodotorula mucilaginosa, active oxidative metabolism increases carotenoids to inactivate excess reactive oxygen species. Front Fungal Biol 2024, 5, 1378590. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.-T.; et al. Developing Rhodotorula as microbial cell factories for the production of lipids and carotenoids. Green Carbon 2024, 2, 409–420. [Google Scholar] [CrossRef]
- Patel, A.; et al. Lipids detection and quantification in oleaginous microorganisms: an overview of the current state of the art. BMC Chemical Engineering 2019, 1, 13. [Google Scholar] [CrossRef]
- Bufka, J.; et al. Exploring carotenoids: Metabolism, antioxidants, and impacts on human health. Journal of Functional Foods 2024, 118, 106284. [Google Scholar] [CrossRef]
- Aiello, A.; et al. Polyunsaturated fatty acid status and markers of oxidative stress and inflammation across the lifespan: A cross-sectional study in a cohort with long-lived individuals. Experimental Gerontology 2024, 195, 112531. [Google Scholar] [CrossRef]
- Zhang, N.; et al. Engineering a manganese superoxide dismutase with enhanced thermostability and activity via protein language Models: Toward antioxidant and anti-inflammatory applications in biomedicine and skincare. Free Radic Biol Med 2025, 239, 565–578. [Google Scholar] [CrossRef]
- Sebastian, S.; et al. Production kinetics and characterization of natural food color (torularhodin) with antimicrobial potential. Bioresource Technology Reports 2023, 24, 101652. [Google Scholar] [CrossRef]
- Li, Z.; et al. Rhodotorula mucilaginosa-alternative sources of natural carotenoids, lipids, and enzymes for industrial use. Heliyon 2022, 8, e11505. [Google Scholar] [CrossRef]
- Ochoa-Viñals, N.; et al. Current Advances in Carotenoid Production by Rhodotorula sp. 2024, 10, 190. [Google Scholar] [CrossRef]
- Sperstad, S.; et al. Torularhodin and torulene are the major contributors to the carotenoid pool of marine Rhodosporidium babjevae (Golubev). J Ind Microbiol Biotechnol 2006, 33, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Zoz, L.; et al. Torularhodin and torulene: bioproduction, properties and prospective applications in food and cosmetics-a review. 2015, 58, 278–288. [Google Scholar] [CrossRef]
- Acevedo-Fani, A.; et al. Chapter 20 - Aspects of food structure in digestion and bioavailability of LCn-3PUFA-rich lipids, in Omega-3 Delivery Systems, P.J. García-Moreno; et al. Editors. 2021, Academic Press. p. 427-448.
- Danielli, M.; et al. Lipid droplets and polyunsaturated fatty acid trafficking: Balancing life and death. Front Cell Dev Biol 2023, 11, 1104725. [Google Scholar] [CrossRef]
- Tkáčová, J.; et al. Correlation between lipid and carotenoid synthesis in torularhodin-producing Rhodotorula glutinis. Annals of Microbiology 2017, 67, 541–551. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, X.; Tan, T. Lipid and carotenoid production by Rhodotorula glutinis under irradiation/high-temperature and dark/low-temperature cultivation. Bioresource Technology 2014, 157, 149–153. [Google Scholar] [CrossRef]
- Liu, C.; et al. Torularhodin Ameliorates Oxidative Activity in Vitro and d-Galactose-Induced Liver Injury via the Nrf2/HO-1 Signaling Pathway in Vivo. Journal of Agricultural and Food Chemistry 2019, 67, 10059–10068. [Google Scholar] [CrossRef]
- Rani, A.; et al. Microorganisms: A Potential Source of Bioactive Molecules for Antioxidant Applications. 2021. 26 1142. [CrossRef]
- Zhang, W.; et al. Torularhodin from Sporidiobolus pararoseus Attenuates d-galactose/AlCl3-Induced Cognitive Impairment, Oxidative Stress, and Neuroinflammation via the Nrf2/NF-κB Pathway. Journal of Agricultural and Food Chemistry 2020, 68, 6604–6614. [Google Scholar] [CrossRef] [PubMed]
- Firsov, A.M.; et al. Deuterated polyunsaturated fatty acids inhibit photoirradiation-induced lipid peroxidation in lipid bilayers. J Photochem Photobiol B 2022, 229, 112425. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, C.C.C.R.; Caramujo, M.J. The Various Roles of Fatty Acids. 2018, 23, 2583. [Google Scholar] [CrossRef] [PubMed]
- Varmira, K.; et al. Statistical optimization of airlift photobioreactor for high concentration production of torularhodin pigment. Biocatalysis and Agricultural Biotechnology 2016, 8, 197–203. [Google Scholar] [CrossRef]
- Sakaki, H.; et al. Activation of torularhodin production by Rhodotorula glutinis using weak white light irradiation. Journal of Bioscience and Bioengineering 2001, 92, 294–297. [Google Scholar] [CrossRef]
- Bao, R.; et al. Enhancement of Torularhodin Production in Rhodosporidium toruloides by Agrobacterium tumefaciens-Mediated Transformation and Culture Condition Optimization. J Agric Food Chem 2019, 67, 1156–1164. [Google Scholar] [CrossRef]
- Lee, J.J.; et al. Metabolomic profiling of Rhodosporidium toruloides grown on glycerol for carotenoid production during different growth phases. J Agric Food Chem 2014, 62, 10203–10209. [Google Scholar] [CrossRef]
- Zheng, X.; et al. Lipid and carotenoid production by the Rhodosporidium toruloides mutant in cane molasses. Bioresour Technol 2021, 326, 124816. [Google Scholar] [CrossRef]
- Zhu, L.; et al. Strategies for optimizing acetyl-CoA formation from glucose in bacteria. Trends in Biotechnology 2022, 40, 149–165. [Google Scholar] [CrossRef]
- Sun, J.; et al. Advances and trends for astaxanthin synthesis in Phaffia rhodozyma. Microb Cell Fact 2025, 24, 100. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; et al. Lignocellulosic biomass as promising substrate for polyhydroxyalkanoate production: Advances and perspectives. Biotechnology Advances 2025, 79, 108512. [Google Scholar] [CrossRef]
- Sulis, D.B.; et al. Advances in lignocellulosic feedstocks for bioenergy and bioproducts. Nature Communications 2025, 16, 1244. [Google Scholar] [CrossRef] [PubMed]
| CXCN-6 Extract | Production (mg/L) | Production/DCW (mg/g) | ||
|---|---|---|---|---|
| Before optimization | After optimization | Before optimization | After optimization | |
| DCW | 20 | 50 | / | / |
| Total lipids | 733.3 | 7126.7 | 36.7 | 142.5 |
| Torularhodin | 15.3 | 63.6 | 0.77 | 1.3 |
| β-Carotene | 1.8 | 2.9 | 0.08989 | 0.058 |
| Yeast strain | Carbon source | Nitrogen source | Torularhodin | β-Carotene | Reference |
|---|---|---|---|---|---|
| R. evergladensis CXCN-6 | Glucose | Yeast extract, Peptone | 63.6 mg/L | 2.9 mg/L | This study |
| R. rubra PTCC 5255 | Glucose | Ammonium sulfate | 35.6 mg/L | 1.0 mg/L | [53] |
| R. glutinis JMT 21978 | Glucose | Yeast extract | 6.6 mg/L | 2.0 mg/L | [46] |
| R. glutinis ZHK | Dextrose | Yeast extract, Peptone | 1.4 mg/L | 1.7 mg/L | [54] |
| R. toruloides A1-15-BRQ | Glucose | Ammonium sulfate | 21.3 mg/L | 1.2 mg/L | [55] |
| R. toruloides CBS 5490 | Glycerol | Yeast extract, Peptone | 19.7 mg/L | 6.8 mg/L | [56] |
| R. toruloides M18 | Glucose | Yeast extract, Peptone | 8.95 mg/L | 285.5 mg/L | [57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




