Submitted:
10 November 2025
Posted:
10 November 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Epidemiology
Timing of Symptom Presentation
- a preclinical phase, supported by molecular or imaging markers, but without clinical signs or symptoms of PD
- a premotor phase (or prodromal phase), characterized by NMS such as hyposmia and sleep behavior disorder
- and the motor phase, often including NMS such as pain, fatigue, and dementia
What Causes NMS in PD?
Dopamine Pathways Affected in PD:
Serotonergic Pathways Affected in PD:
Noradrenergic Pathways Affected in PD:
Glutamatergic Pathways Affected in PD:
Cholinergic Pathways Affected in PD:
GABAergic Dysfunction Affected in PD:
The Burden of NMS
A Focus on PD Nurses
- ✓ providing information, education, and instruction
- ✓ supporting the patient and caregiver in the promotion of self-management
- ✓ supporting psychosocial care questions
- ✓ specializing in diagnostic strategies and therapeutic nursing interventions
- ✓ promoting multidisciplinary collaboration.
Tools for Assessing NMS
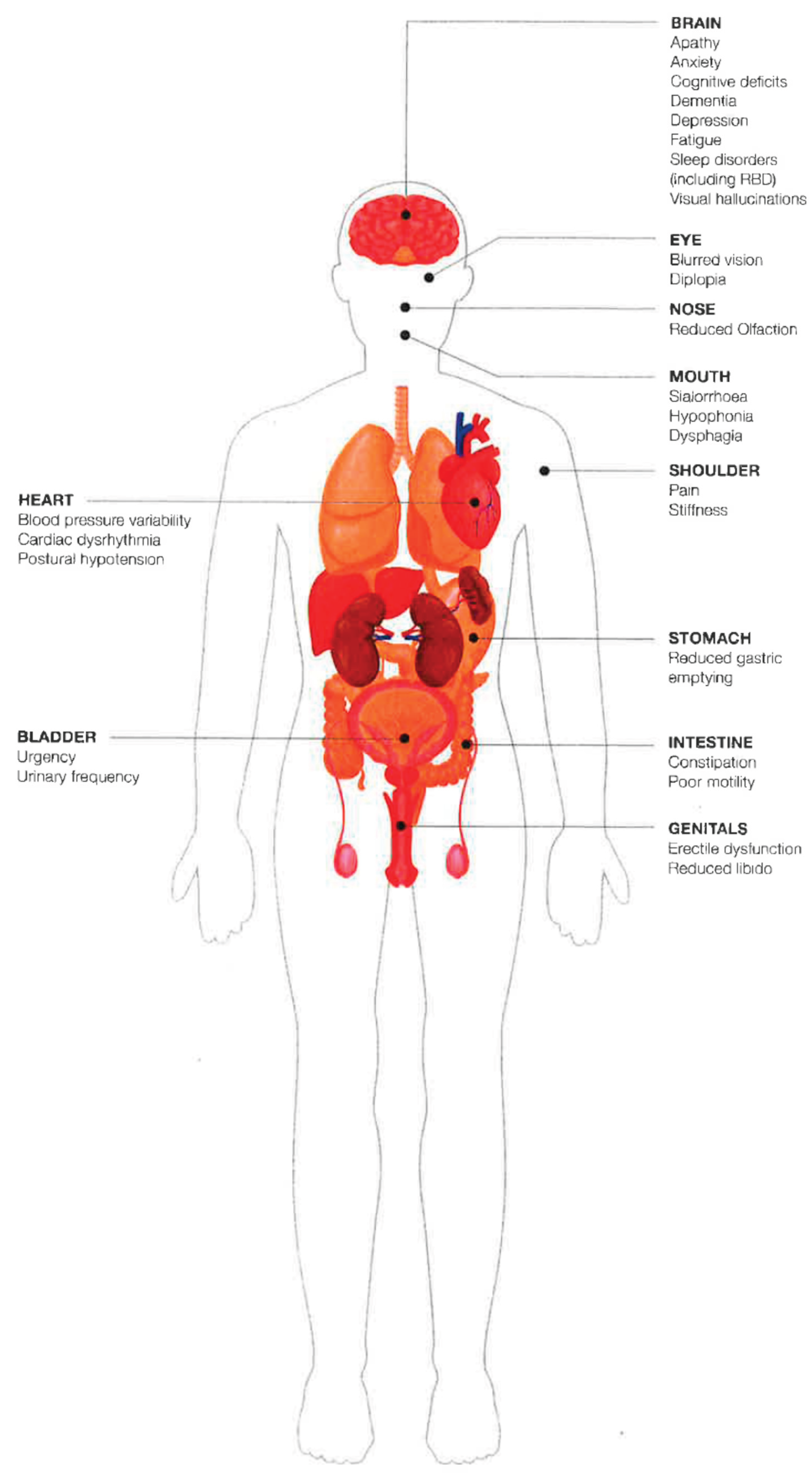
The Different NMS in PD
- Pain and other sensory symptoms (olfactory disfunction; changes in visual function)
- Neuropsychiatric symptoms (depression; anxiety; apathy; cognitive impairment and dementia; psychotic symptoms, hallucinations and delusions; compulsive behaviors)
- Sleep disorders (rapid eye movement sleep behavior disorder; insomnia; restless legs syndrome and periodic limb movements; excessive daytime sleepiness)
- Autonomic symptoms (bladder dysfunction; gastrointestinal symptoms; neurogenic orthostatic hypotension; sexual dysfunction)
- Fatigue
Pain and Other Sensory Symptoms
Pain
Olfactory Disfunction
Changes in Visual Function
Neuropsychiatric Symptoms
Depression
Anxiety
Apathy
Cognitive Impairment and Dementia
Psychotic Symptoms, Hallucinations and Delusions
Compulsive Behaviors
Sleep Disorders
Rapid Eye Movement Sleep Behavior Disorder (RBD)
Insomnia
Restless Legs Syndrome and Periodic Limb Movements
Excessive Daytime Sleepiness (EDS)
Autonomic Symptoms
Bladder Dysfunction
Gastrointestinal Symptoms
Neurogenic Orthostatic Hypotension
Sexual Dysfunction
Fatigue
Conclusions
Authors Contribution
Ethical Approval
Funding
<i>Acknowledgments</i>
Conflicts of Interest
References
- Tanner CM, Ostrem JL. Parkinson’s disease. N Engl J Med 2024; 391(5): 442-452.
- Su D, Cui Y, He C et al. Projections for prevalence of Parkinson’s disease and its driving factor in 195 countries and territories to 2050: modelling study of Global Burden of Disease Study 2021; BMJ 2025; 388: e080952.
- Dahodwala N, Li P, Jahnke J et al. Burden of Parkinson’s disease by severity: health care costs in the US Medicare population. Mov Disord 2021; 36(1): 133-142.
- Goldman, JG. Non-motor symptoms and treatments in Parkinson’s disease. Neurol Clin 2025; 43(2): 291-317.
- Rietdijk CD, Perez-Pardo P, Garssen J et al. Exploring Braak’s hypothesis of Parkinson’s disease. Front Neurol 2017; 8: 37.
- Van der Meer F, Jorgensen H, Hiligsmann M. Burden of non-motor symptoms of Parkinson’s disease: cost-of-illness and quality-of-life estimates through a scoping review. Expert Rev Pharmacoecon Outcomes Res 2025; 25(1): 17-27.
- Rukavina K, Batzu L, Boogers A et al. Non-motor complications in late-stage Parkinson’s disease: recognition, management and unmet needs. Expert Rev Neurother 2021; 21(3): 335-352.
- Lewitt PA, Chaudhury KR. Unmet needs in Parkinsons disease: motor and non-motor. Parkinsonism Relat Disord 2020; 80(Suppl 1): S7-S12.
- Hurt CS, Rixon L, Chaudhuri KR et al. Barriers to reporting non-motor symptoms to health-care providers in people with Parkinson’s. Parkinsonism Relat Disord 2019; 64: 220–225.
- Crosiers D, Pickut B, Theuns J et al. Non-motor symptoms in a Flanders-Belgian population of 215 Parkinson’s disease patients as assessed by the non-motor symptoms questionnaire. American Journal of Neurodegenerative Disease 2012; 1(2): 160-167.
- Cattaneo C, Pagonabarraga J. Sex differences in Parkinson’s disease: a narrative review. Neurol Ther 2025; 14(1): 57-70.
- Brun L, Lefaucheur R, Fetter D et al. Non-motor fluctuations in Parkinson’s disease: prevalence, characteristics and management in a large cohort of parkinsonian outpatients. Clin Neurol Neurosurg 2014; 127: 93–96.
- Batzu L, Podlewska A, Gibson L et al. A general clinical overview of the non-motor symptoms in Parkinson’s disease: neuropsychiatric symptoms. Int Rev Neurobiol 2024; 174: 59-97.
- Sauerbier A, Lenka A, Aris A, Pal PK. Nonmotor symptoms in Parkinson's disease: gender and ethnic differences. Int Rev Neurobiol 2017; 133: 417-446.
- Jercic KG, Blazekovic A, Borovecki S, Borovecki F. Non-motor symptoms of Parkinson’s disease - insights from genetics. J Neural Transm 2024; 131(11): 1277-1284.
- Song T, Zhou X, Wang C et al. Clinical features and progression of Parkinson’s disease with LLRK2 variants: a prospective study. Ann Clin Transl Neurol 2025; 12(1): 34-42.
- Brockmann K, Srulijes K, Pflederer S et al. GBA-associated Parkinsons’ disease: reduced survival and more rapid progression in a prospective longitudinal study. Mov Disord 2015; 30(3): 407-411.
- Li Q, Jing Y, Lun P et al. Association of gender and age at onset with glucocerebrosidase associated Parkinson’s disease: a systematic review and meta-analysis. Neurol Sci 2021; 42(6): 2261–71.
- Chen H, Burton EA, Ross GW et al., Research on the premotor symptoms of Parkinson’s disease: clinical and etiological implications. Environ Health Perspect 2013; 121: 1245–1252.
- Hely MA, Reid WG, Adena M, et al. The Sydney multicenter study of Parkinson’s disease: the inevitability of dementia at 20 years. Mov Disord 2008; 23: 837–844.
- Morris HR, Spillantini MG, Sue CM, Williams-Gray CH. The pathogenesis of Parkinson’s disease. Lancet 2024; 403(10423): 293–304.
- Berg D, Postuma RB, Bloem B et al. Time to redefine PD? Introductory statement of the MDS Task Force on the definition of Parkinson’s disease. Mov Disord 2014; 29: 454–62.
- Schapira AHV, Chaudhuri KR and Jenner P. Non-motor features of Parkinson disease. Nat Rev Neurosci 2017; 18: 435–450.
- Park A and Stacy, M. Dopamine-induced nonmotor symptoms of Parkinson’s disease. Parkinsons Dis 2011: 485063.
- Politis M and Loane, C. Serotonergic dysfunction in Parkinson’s disease and its relevance to disability. ScientificWorldJournal 2011; 11: 1726–1734.
- Delaville C, Deurwaerdère PD and Benazzouz A. Noradrenaline and Parkinson’s disease. Front Syst Neurosci 2011; 5: 31.
- Remy P, Doder M, Lees A et al. Depression in Parkinson’s disease: loss of dopamine and noradrenaline innervation in the limbic system. Brain 2005; 128: 1314–1322.
- Jenner P and Caccia, C. The role of glutamate in the healthy brain and in the pathophysiology of Parkinson’s disease. Eur Neurol Rev 2019; 14: 2–12.
- Müller MLTM, Bohnen NI. Cholinergic dysfunction in Parkinson’s disease. Curr Neurol Neurosci Rep 2013; 13(9): 377.
- Pasquini J, Brooks DJ, Pavese N. The involvement of the cholinergic system in Parkinson disease. Handb Clin Neurol 2025; 211: 215-229.
- Alharbi B, Al-Kuraishy HM, Al-Gareeb AL et al. Role of GABA pathway in motor and non-motor symptoms in Parkinson’s disease: a bidirectional circuit. Eur J Med Res 2024; 29(1): 205.
- Mehra S, Sahay S, Maji SK. Alpha-synuclein misfolding and aggregation: implications in Parkinson’s disease pathogenesis. Biochim Biophys Acta Proteins Proteom 2019; 1867(10): 890-908.
- Del Tredici K, Braak H. Idiopathic Parkinson’s disease: staging an α -synucleinopathy with a predictable pathoanatomy. In: Kahle P, Haass C, eds. Molecular mechanisms in Parkinson’s disease. Georgetown, TX: Landes Bioscience, 2004; 1–32.
- Braak H, Del Tredici K, Rüb U et al. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 2003; 24: 197–211.
- Halliday G, Lees A, Stern M. Milestones in Parkinson’s disease – clinical and pathologic features. Mov Disord 2011; 26(6): 1015–1021.
- Kulcsarova K, Skorvanek M, Postuma RB, Berg D. Defining Parkinson’s disease: past and future. J Parkinsons Dis 2024; 14(s2): S257-S271.
- Global Parkinson’s Disease Survey Steering Committee. Factors impacting on quality of life in Parkinson’s disease: results from an international survey. Mov Disord 2022; 17: 60-67.
- Wu DD; Su W, He J et al. Nonmotor symptoms and quality of life in Parkinson’s disease with different motor subtypes. Z Gerontol Geriatr 2022; 55(6): 496-501.
- Candel-Parra E, Corcoles-Jimenez MP, Delicado-Useros V et al. Evolution of quality of life in persons with Parkinson’s disease: a prospective cohort study. J Clin Med 2021; 10(9): 1824.
- Martinez-Martin P, Macaulay D, Jalundhwala YJ et al. The long- term direct and indirect economic burden among Parkinson’s disease caregivers in the United States. Mov Disord 2019; 34(2): 236–245.
- Hermanowicz N, Jones SA and Hauser RA. Impact of non-motor symptoms in Parkinson’s disease: a PMDAlliance survey. Neuropsychi atr Dis Treat 2019; 15: 2205–2212.
- Gumber A, Ramaswamy B, Thongchundee O. Effects of Parkinson’s on employment, cost of care, and quality of life of people with condition and family caregivers in the UK: a systematic literature review. Patient Relat Outcome Meas 2019; 10: 321–333.
- Yang W, Hamilton JL, Kopil C et al. Current and projected future economic burden of Parkinson’s disease in the U.S. NPJ Parkinsons Dis 2020; 6: 15.
- Findley L, Aujla M, Bain PG et al. Direct economic impact of Parkinson’s disease: a research survey in the United Kingdom. Mov Disord 2003; 18: 1139-1145.
- Mudiyanselage SB, Watts JJ, Abimanyi-Ochom J et al. Cost of living with Parkinson’s disease over 12 months in Australia: a prospective cohort study. Parkinsons Dis 2017; 2017: 5932675.
- Gustafsson A, Hjalte F, Norlin J et al. The association between non-motor symptoms and cost in Parkinson’s disease. J Neurol 2025; 272(4): 297.
- Weise D, Claus I, Dresel C et al. Multidisciplinary care in Parkinson’s disease. J Neural Transm 2024; 131(10): 1217–1227.
- Radder DLM, Lennaerts HH, Vermeulen H et al. The cost-effectiveness of specialized nursing interventions for people with Parkinson’s disease: the NICE-PD study protocol for a randomized controlled clinical trial. Trials 2020; 21(1): 88.
- Hellqvist C, Berterö C. Support supplied by Parkinson’s disease specialist nurses to Parkinson’s disease patients and their spouses. Appl Nurs Res 2015; 28(2): 86–91.
- Fujita T, Iwaki M, Hatono Y. The role of nurses for patients with Parkinson’s disease at home: a scoping review. BMS Nurs 2024; 23(1): 318.
- Chaudhuri KR, Martinez-Martin P, Schapira AH et al. International multicenter pilot study of the first comprehensive self-completed nonmotor symptoms questionnaire for Parkinson’s disease: the NMSQuest study. Mov Disord 2006; 21: 916-923.
- Chaudhuri KR, Martinez-Martin P, Brown RG et al. The metric properties of a novel non-motor symptoms scale for Parkinson’s disease: Results from an international pilot study. Mov Disord 2007; 22: 1901–1911.
- Marinus J, Visser M, Stiggelbout AM et al. A short scale for the assessment of motor impairments and disabilities in Parkinson's disease: the SPES/SCOPA. J Neurol Neurosurg Psychiatry 2004; 75(3): 388-95.
- Goetz CG, Tilley BC, Shaftman SR et al. Movement Disorder Society -sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 2008; 23: 2129-2170.
- Peto V, Jenkinson C, Fitzpatrick R et al. The development and validation of a short measure of functioning and well-being for individuals with Parkinson’s disease. Qual Lif Res 1995; 4: 241-248.
- Chaudhuri KR, Rizos A, Trenkwalder C et al. King´s Parkinson´s disease pain scale, the first scale for pain in PD: an international validation. Mov Disord 2015; 30: 1623–163.
- Trenkwalder C, Kohnen R, Högl B et al. Parkinson’s disease sleep scale validation of the revised version PDSS-2. Mov. Disord 2011; 26(4): 644-652.
- Johns, MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991; 14(6): 540-545.
- Hamilton, M. A rating scale for depression. J Neurol Neurosurg Psychiatry 1960: 23: 56–62.
- Beck AJ, Ward CH, Mendelson M et al. An inventory for measuring depression. Arch Gen Psychiatry 1961; 4: 561–71.
- Krupp LB, LaRocca NG, Muir-Nash J et al. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 1989; 46(10): 1121-1123.
- Brown RG, Dittner A, Findley L et al. The Parkinson fatigue scale. Parkinsonism Relat Disord 2005; 11(1): 49–55.
- Antonini A, Tinazzi M, Abbruzzese G et al. Pain in Parkinson’s disease: facts and uncertainties. Eur J Neurol 2018; 25: 917-e69.
- Ford, B. Pain in Parkinson’s disease. Clin Neurosci 1998; 5(2): 63–72.
- Mylius V, Perez Lloret S, Cury RG et al. The Parkinson disease pain classification system: results from an international mechanism-based classification approach. Pain 2021; 162: 1201–1210.
- Cattaneo C, Jost WH. Pain in Parkinson’s disease: pathophysiology, classification and treatment. J Integr Neurosci 2023; 22(5): 132.
- Storch A, Bremer A, Gandor F et al. Pain fluctuations in Parkinson’s disease and their association with motor and non-motor fluctuations. J Parkinsons Dis 2024; 14(7): 1451-1468.
- Tinazzi M, Gandolfi M, Artusi CA et al. Advances in diagnosis, classification and management of pain in Parkinson’s disease. Lancet Neurol 2025; 24(4): 331-347.
- Pagonabarraga J, Tinazzi M, Caccia C, Jost WH. The role of glutamatergic neurotransmission in the motor and non-motor symptoms in Parkinson’s disease: clinical cases and a review of the literature. J Clin Neurosci 2021; 90: 178–183.
- Gao L, Yang Y, Cai L, Xiong Y. Gender differences in pain subtypes among patients with Parkinson’s disease. J Integr Neurosci 2022; 21(4): 120.
- Mehanna R, Jankovic J. Young-onset Parkinson’s disease: Its unique features and their impact on quality of life. Parkinsonism Relat Disord 2019; 65: 39–48.
- Eryilmaz IE, Erer S, Zarifoglu M et al. Contribution of functional dopamine D2 and D3 receptor variants to motor and non-motor symptoms of early onset Parkinson’s disease. Clin Neurol Neurosurg 2020; 199: 106257.
- Zhang LM, Zhang XP, Chen YQ, Ye W. Association of CHRNA4 gene rs1044396 and rs1044397 polymorphisms with Parkinson’s disease symptoms and smoking. Genet Mol Res 2015; 14(2): 5112–5122.
- Osikowicz M, Mika J, Przewlocka B. The glutamatergic system as a target for neuropathic pain relief. Exp Physiol 2013; 98(2): 372–384.
- Qureshi AR, Rana AQ, Malik SH et al. Comprehensive examination of therapies for pain in Parkinson’s disease: a systematic review and meta-analysis. Neuroepidemiology 2018; 51(3-4): 190–206.
- Santos García D, Yáñez Baña R, Labandeira Guerra C et al. Pain improvement in Parkinson’s disease patients treated with safinamide: results from the SAFINONMOTOR study. J Pers Med 2021; 11(8): 798.
- Edinoff A, Sathivadivel N, McBride T et al. Chronic pain treatment strategies in Parkinsons’ disease. Neurol Int 2020; 12(3): 61-76.
- Brefel-Courbon C, Grolleau S, Thalamas C et al. Comparison of chronic analgesic drugs prevalence in Parkinson’s disease, other chronic diseases and the general population. Pain 2009; 141(1-2): 14-18.
- Li J, Mi TM, Zhu BF et al. High-frequency repetitive transcranial magnetic stimulation over the primary motor cortex relieves musculoskeletal pain in patients with Parkinson’s disease: a randomized controlled trial. Parkinsonism Relat Disord 2020; 80: 113–119.
- Huissoud M, Boussac M, Joineau K et al. The effectiveness and safety of non-pharmacological intervention for pain management in Parkinson’s disease: a systematic review. Rev Neurol 2024; 180(8): 715–735.
- Ponsen MM, Stoffers D, Booij J et al. Idiopathic hyposmia as a preclinical sign of Parkinson’s disease. Ann Neurol 2004; 56: 173–181.
- Haehner A, Hummel T and Reichmann H. Olfactory loss in Parkinson’s disease. Parkinson’s Disease 2011; 2011: 450939.
- Mitchell E, Mattjie C, Bestwick JP et al. Hyposmia in Parkinson’s disease; exploring selective odour loss. NPJ Parkinsons Dis 2025; 11(1): 67.
- Marek K, Russel DS, Concha-Marambio L et al. Evidence of alpha-synuclein aggregation in older individuals with hyposmia: a cross-sectional study. EBioMedicine 2025; 112: 105567.
- Hawkes, C. Olfaction in neurodegenerative disorders. Mov Disord 2003; 18(4): 364–372.
- Ubeda-Bañon I, Saiz-Sanchez D, de la Rosa-Prieto C et al. Alpha-synucle inopathy in the human olfactory system in Parkinson’s disease: involvement of calcium-binding protein- and substance P-positive cells. Acta Neuropathol 2010; 119: 723–735.
- Nieto-Esamez F, Obrero-Gaitan E, Cortés-Pérez I. Visual dysfunction in Parkinson’s disease. Brain Sci 2023; 13(8): 1173.
- Gryc W, Roberts KA, Zabetian CP et al. Hallucinations and development of dementia in Parkinson’s disease. J Parkinsons Dis 2020; 10(4): 1643-1648.
- Tran KKN, Lee PY, Finkelstein DI et al. Altered outer retinal structure, electrophysiology and visual perception in Parkinson’s disease. J Parkinsons Dis 2024; 14(1): 167-180.
- Borm CDJM, Bloem BR, Hoyng C et al. The many faces of blurry vision in Parkinson’s disease: an illustrative case series. Case Rep Neurol 2022; 14(1): 173-178.
- Van der Lijn I, de Haan GA, Huizinga F et al. Self-reported visual complaints in people with Parkinson’s disease: a systematic review. J Parkinsons Dis 2022; 12(3): 785-806.
- Kedar, S. Symptomatic treatment of neuro-ophthalmic visual disturbances. Continuum 2025; 31(2): 566-582.
- Cong S, Xiang C, Zhang S et al. Prevalence and clinical aspects of depression in Parkinson’s disease: a systematic review and meta-analysis of 129 studies. Neurosci Biobehav Rev. 2022; 141: 104749.
- Dujardin K, Sgambato V. Neuropsychiatric disorders in Parkinson’s disease: what do we know about the role of dopaminergic and non-dopaminergic systems? Front Neurosci 2020; 14: 25.
- Mai AS, Chao Y, Xiao B et al. Risk of suicidal ideation and behaviour in individuals with Parkinson disease: a systematic review and meta-analysis. JAMA Neurol 2024; 81(1): 10-18.
- Mills KA; Greene MC, Dezube R et al. Efficacy and tolerability of antidepressants in Parkinson’s disease: a systematic review and network meta-analysis. Int J Geriatr Psychiatry 2018; 33(4): 642-651.
- Barone P, Poewe W, Albrecht S et al. Pramipexole for the treatment of depressive symptoms in patients with Parkinson’s disease: a randomized, double-bind, placebo-controlled trial. Lancet Neurol 2010; 22: 573-580.
- Barone P, Santangelo G, Morgante L et al. A randomized clinical trial to evaluate the effects of rasagiline on depressive symptoms in non-demented Parkinson’s disease patients. Eur J Neurol 2015; 22: 1184-1191.
- Miladinovic T, Nashed MG, Singh G. Overview of glutamatergic dysregulation in central pathologies. Biomolecules 2015; 5: 3122-3141.
- Peña E, Borrué C, Mata M et al. Impact of SAfinamide on Depressive symptoms in Parkinson’s Disease patients (SADness-PD study): a multicenter retrospective study. Brain Sci 2021; 11(2): 232.
- Wamelen DJV, Rukavina K, Podlewska AM, Chaudhuri KR. Advances in the pharmacological and non-pharmacological management of non-motor symptoms in Parkinson’s disease: an update since 2017. Cur Neuropharmacol 2023; 21(8): 1786-1805.
- Dobkin RD, Mann SL, Interian A et al. Cognitive behavioral therapy improves diverse profiles of depressive symptoms in Parkinson’s disease. Int J Geriatr Psychiatry 2019; 34(5): 722–729.
- Takamiya A, Seki M, Kudo S et al. Electroconvulsive therapy for Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 2021; 36(1): 50-58.
- Dissanayaka NN, Forbes EJ Perepezko K et al. Phenomenology of atypical anxiety disorders in Parkinson’s disease: a systematic review. Am J Geriatr Psychiatry. 2022; 30: 1026–1050.
- Wang Z, Wei H, Xin Y, Qin W. Advances in the study of depression and anxiety in Parkinson’s disease: a review. Medicine 2025; 104(10): e41674.
- Lintel H, Corpuz T, Paracha SU, Grossberg GT. Mood disorders and anxiety in Parkinson’s disease: current concepts. J Geriatr Psychiatry Neurol 2021; 34:280–288.
- Perepezko K, Naaz F, Wagandt C et al. Anxiety in Parkinson’s disease: a systematic review of neuroimaging studies. J Neuropsychiatry Clin Neurosci 2021; 33(4): 280-294.
- Pontone GM, Mills KA. Optimal treatment of depression and anxiety in Parkinson’s disease. Am J Geriatr Psychiatry 2021; 29(6): 530-540.
- Moonen AJH, Mulders AEP, Defebvre L et al. Cognitive behavioral therapy for anxiety in Parkinson’s disease: a randomized controlled trial. Mov Disord 2021; 36(11): 2539-2548.
- Bereau M, Van Waes V, Servant M et al. Apathy in Parkinson’s disease: clinical patterns and neurobiological basis. Cells 2023; 12(12): 1599.
- Miller DS, Robert P, Ereshefsky L et al. Diagnostic criteria for apathy in neurocognitive disorders. Alzheimers Dement 2021; 17(12): 1892-1904.
- Yang C, Hu Y, Talishinsky AD et al. Medial prefrontal cortex and anteromedial thalamus interaction regulates goal-directed behavior and dopaminergic neuron activity. Nat Commun 2022; 13: 1386.
- Maggi G, Loayza F, Vitale C et al. Anatomical correlates of apathy and impulsivity co-occurrence in early Parkinson’s disease. J Neurol 2024; 271(5): 2798-2809.
- Vachez YM, Bahout M, Magnard R et al. Unilateral and Bilateral Subthalamic Deep Brain Stimulation Differently Favour Apathy in Parkinson's Disease. Eur J Neurosci 2025; 61(4): e70019.
- Mele B, Van S, Holroyd-Leduc J et al. Diagnosis, treatment and management of apathy in Parkinson’s disease: a scoping review. BMJ Open 2020; 10(9): e037632.
- Strasser A, Luksys G, Xin L et al. Glutamine-to-Glutamate ratio in the nucleus accumbens predicts effort based motivated performance in humans. Neuropsychopharmacology 2020; 45: 2048–57.
- Kulisevsky J, Martínez-Horta S, Campolongo A et al. A randomized clinical trial to evaluate the effects of safinamide on apathetic non-demented patients with Parkinson’s disease. Front Neurol 2022; 13:866502.
- Devos D, Moreau C, Maltete D et al. Rivastigmine in apathetic but dementia and depression-free patients with Parkinson’s disease: a double-blind, placebo-controlled, randomized clinical trial. J Neurol Neurosurg Psychiatry 2014; 85(6): 668–674.
- Mele B, Ismail Z, Goodarzi Z et al. Non-pharmacologic interventions to treat apathy in Parkinson's disease: a realist review. Clin Park Relat Disord 2021; 4: 100096.
- Aarsland D, Batzu L, Halliday GM et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers 2021; 7(1): 47.
- Goldman JG, Sieg E. Cognitive impairment and dementia in Parkinson’s disease. Clin Geriatr Med 2020; 36(2): 365-377.
- Guo Y, Xu W, Liu FT et al. Modifiable risk factors for cognitive impairment in Parkinson’s disease: a systematic review and meta-analysis of prospective cohort studies. Mov Disord 2019; 34: 876–883.
- Pourzinal D, Yang J, Lawson RA et al. Systematic review of data-driven cognitive subtypes in Parkinson disease. Eur J Neurol 2022; 29(11): 3395-3417.
- Singh, S. Noradrenergic pathways of locus coeruleus in Parkinson's and Alzheimer's pathology. Int J Neurosci 2020; 130(3): 251-261.
- Sun C, Armstrong MJ. Treatment of Parkinson's disease with cognitive impairment: current approaches and future directions. Behav Sci 2021; 11(4): 54.
- Ghosh A, Das S, Behera SK et al. Atomoxetine does not improve complex attention in idiopathic Parkinson’s disease patients with cognitive deficits: a meta-analysis. Parkinsons Dis 2020; 2020: 4853590.
- Zhang Y, Liu S, Xu K et al. Non-pharmacological therapies for treating non-motor symptoms in patients with Parkinson’s disease: a systematic review and meta-analysis. Front Aging Neurosci 2024; 16: 1363115.
- Chendo I, Silva C, Duarte GS et al. Frequency and characteristics of psychosis in Parkinson's disease: a systematic review and meta-analysis. J Parkinsons Dis. 2022;12(1):85-94.
- Pagonabarraga J, Bejr-Kasem H, Martinez-Horta S, Kulisevsky J. Parkinson disease psychosis: from phenomenology to neurobiological mechanisms. Nat Rev Neurol 2024; 20(3): 135-150.
- Brown P, Freeman D, Loe BS et al. Paranoid and unusual sensory experiences in Parkinson’s disease. Aging Ment Health 2025; 29(5): 935-950.
- Pisani S, Gunasekera B, Lu Y et al. Functional and connectivity correlates associated with Parkinson’s disease psychosis: a systematic review. Brain Commun 2024; 6(6): fcae358.
- Komagamine T, Suzuki K, Kokubun N et al. Sleep-related hallucinations in patients with Parkinson’s disease. PLoS One 2022; 17(10): e0276736.
- Goldman JG, Goetz CG, Berry-Kravis E et al. Genetic polymorphisms in Parkinson’s disease subjects with and without hallucinations: an analysis of the cholecystokinin system. Arch Neurol 2004; 61(8): 1280-1284.
- Segal GS, Xie SJ, Paracha SU, Grossberg GT. Psychosis in Parkinson's disease: current treatment options and impact on patients and caregivers. J Geriatr Psychiatry Neurol 2021; 34(4): 274-279.
- Tampi RR, Tampi DJ, Young JJ et al. Evidence for using pimavanserin for the treatment of Parkinson’s disease psychosis. World J Psychiatry 2019; 9(3): 47-54.
- Rose O, Huber S, Trinka E et al. Treatment of Parkinson’s disease psychosis – a systematic review and multi-methods approach. Biomedicines 2024; 12(10) :2317.
- Jellinger, KA. Behavioral disorders in Parkinson’s disease: current view. J Neural Transm 2025; 132(2): 169-201.
- Zhang JF, Wang XX, Feng Y et al. Impulse control disorders in Parkinson’s disease: epidemiology, pathogenesis and therapeutic strategies. Front Psychiatry 2021; 12: 635494.
- Ansari MF, Prasad S, Bhardwaj S et al. Morphometric alterations of the mesocorticolimbic network in Parkinson’s disease with impulse control disorders. J Neural Transm 2024; 131: 229–237.
- Gros P, Videnovic A. Overview of sleep and circadian rhythm disorders in Parkinson disease. Clin Geriatr Med 2020; 36(1): 119–130.
- Liguori C, De Franco V, Cerroni R et al. Sleep problems affect quality of life in Parkinson's disease along disease progression. Sleep Med 2021; 81: 307-311.
- Jones BM, McCarter SJ. Rapid eye movement sleep behavior disorder: clinical presentation and diagnostic criteria. Sleep Med Clin 2024; 19(1): 71-81.
- Gnarra O, Wulf MA, Schäfer C et al. Rapid eye movement sleep behavior disorder: a narrative review from a technological perspective. Sleep 2023; 46(6): zsad030.
- Kunz D, Oster H, Rawashdeh O et al. Sleep and circadian rhythms in α-synucleinopathies-Perspectives for disease modification. Acta Physiol 2023; 238(1): e13966.
- Howell M, Avidan AY, Foldvary-Schaefer N et al. Management of REM sleep behavior disorder: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2023; 19(4): 759-768.
- Bovenzi R, Conti M, Pierantozzi M et al. Safinamide effect on sleep architecture of motor fluctuating Parkinson’s disease patients: a polysomnographic rasagiline-controlled study. Parkinsonism Relat Disord 2024; 127: 107103.
- Arias-Carrion O, Ortega-Robles E, Ortuno-Sahagun D et al. Sleep-related disorders in Parkinson's disease: mechanisms, diagnosis, and therapeutic approaches. CNS Neurol Disord Drug Targets 2025; 24(2): 132-143.
- Suzuki K, Fujita H, Kobayashi S. Managing sleep issues in Parkinson's disease: an up-to-date review. Expert Rev Neurother 2025; 25(2): 211-226.
- Bliwise DL, Karroum EG, Greer SA et al. Restless legs symptoms and periodic leg movements in sleep among patients with Parkinson's disease. J Parkinsons Dis 2022; 12(4): 1339-1344.
- Maggi G, Barone A, Mastromarino C et al. Prevalence and clinical profile of patients with restless legs syndrome in Parkinson's disease: a meta-analysis. Sleep Med 2024; 121: 275-286.
- Cochen De Cock, V. Therapies for restless legs in Parkinson’s disease. Curr Treat Options Neurol 2019; 21(11): 56.
- Maggi G, Vitale C, Cerciello F, Santangelo G. Sleep and wakefulness disturbances in Parkinson's disease: a meta-analysis on prevalence and clinical aspects of REM sleep behavior disorder, excessive daytime sleepiness and insomnia. Sleep Med Rev 2023; 68: 101759.
- Arias-Carrion O, Ortega-Robles E, Ortuno-Sahagun D et al. Sleep-related disorders in Parkinson's disease: mechanisms, diagnosis, and therapeutic approaches. CNS Neurol Disord Drug Targets 2025; 24(2): 132-143.
- Haq IZ, Naidu Y, Reddy P, Chaudhuri KR. Narcolepsy in Parkinson’s disease. Expert Rev Neurother 2010; 10(6): 879-884.
- Saper CB, Fuller PM, Pedersen NP et al. Sleep state switching. Neuron 2010; 68(6):1023-1042.
- Zheng JH, Ma JJ, Sun WH et al. Excessive daytime sleepiness in Parkinson's disease is related to functional abnormalities in the left angular gyrus. 1: Clin Neuroradiol 2023; 33(1), 2023.
- Fujita H, Ogaki K, Shiina T et al. Impact of autonomic symptoms on the clinical course of Parkinson's disease. Neurol Sci 2024; 45(8): 3799-3807.
- Li FF, Cui YS, Yan R et al. Prevalence of lower urinary tract symptoms, urinary incontinence and retention in Parkinson's disease: a systematic review and meta-analysis. Front Aging Neurosci 2022; 14: 977572).
- Barone P, Antonini A, Colosimo C et al. The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson's disease. Mov Disord 2009; 24(11): 1641-1649.
- Seki S, Igawa Y, Kaidoh K. et al. Role of dopamine D1 and D2 receptors in the micturition reflex in conscious rats. Neurourol Urodyn 2001; 20(1): 105-113.
- Chen Z, Li G, Liu J. Autonomic dysfunction in Parkinson's disease: implications for pathophysiology, diagnosis, and treatment. Neurobiol Dis 2020; 134: 104700.
- Hu JC, Hsu LN, Lee WC et al. Role of urological botulinum toxin-A injection for overactive bladder and voiding dysfunction in patients with Parkinson’s disease or post-stroke. Toxins 2023; 15(2): 166.
- Gómez-López A, Sánchez-Sánchez A, Natera-Villalba E et al. SURINPARK: safinamide for urinary symptoms in Parkinson's disease. Brain Sci 2021 6; 11(1): 57.
- Palma JA, Thijs RD. Non-pharmacological treatment of autonomic dysfunction in Parkinson's disease and other synucleinopathies. J Parkinsons Dis 2024; 14(s1): S81-S92.
- Safarpour D, Sharzehi K, Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Drugs 2022; 82(2): 169-197.
- Dogra N, Mani RJ, Katare DP. The gut-brain axis: two ways signaling in Parkinson's disease. Cell Mol Neurobiol 2022; 42(2): 315-332.
- Isaacson J, Patel S, Torres-Yaghi Y, Pagán F. Sialorrhea in Parkinson’s disease. Toxins 2020; 12(11): 691.
- Santos Junior LC, Santos JR, Reis A et al. Effectiveness of the pharmacological treatments for sialorrhea in patients with Parkinson's disease: a systematic review and network meta-analysis. Clin Oral Investig 2023; 27(6): 2449-2463.
- Umemoto G, Furuya H. Management of dysphagia in patients with Parkinson's disease and related disorders. Intern Med. 2020; 59(1): 7-14.
- Schindler A, Pizzorni N, Cereda E et al. Consensus on the treatment of dysphagia in Parkinson's disease. J Neurol Sci 2021; 430: 120008.
- Pfeiffer RF, Isaacson SH, Pahwa R. Clinical implications of gastric complications on levodopa treatment in Parkinson's disease. Parkinsonism Relat Disord 2020; 76: 63-71.
- Soliman H, Coffin B, Gourcerol G. Gastroparesis in Parkinson disease: pathophysiology, and clinical management. Brain Sci 2021; 11(7): 831.
- Yao L, Liang W, Chen J, Wang Q, Huang X. Constipation in Parkinson's disease: a systematic review and meta-analysis. Eur Neurol 2023; 86(1): 34-44.
- Cheesman M, Ho H, Bishop K, Sin MK. Constipation management in Parkinson disease. J Neurosci Nurs 2021; 53(6): 262-266.
- Ramu SK, Oblizajek NR, Savica R et al. Defecatory disorders are a common cause of chronic constipation in Parkinson disease. Neurogastroenterol Motil 2024; 36(5): e14767.
- Palma JA, Kaufmann H. Orthostatic hypotension in Parkinson disease. Clin Geriatr Med 2020; 36(1): 53-67.
- Grosu C, Noea O, Maștaleru A et al. Neurogenic orthostatic hypotension in Parkinson disease-a narrative review of diagnosis and management. J Clin Med 2025; 14(2): 630.
- Kaufmann H, Norcliffe-Kaufmann L, Palma JA. Baroreflex dysfunction. N Engl J Med. 2020; 382(2): 163-178.
- Fanciulli A, Leys F, Falup-Pecurariu C et al. Management of orthostatic hypotension in Parkinson’s disease. J Parkinsons Dis 2020; 10(s1): S57-S64.
- Buhmann, C. Prevalence, clinical presentations and impact on relationship of sexual dysfunction in Parkinson's Disease. Int Rev Neurobiol 2022; 162: 1-19.
- Batzu L, Titova N, Bhattacharyya KB, Chaudhuri KR. The pathophysiology of sexual dysfunction in Parkinson's disease: an overview. Int Rev Neurobiol 2022; 162: 21-34.
- Urso D, Leta V, Rukavina K. Management strategies of sexual dysfunctions in Parkinson's disease. Int Rev Neurobiol 2022; 162: 97-116.
- Bernard BA, Metman LV, Levine L et al. Sildenafil in the treatment of erectile dysfunction in Parkinson's Disease. Mov Disord Clin Pract 2016; 4(3): 412-415.
- Pitton Rissardo J, Jayasinghe M, Rashidi M et al. Exploring fatigue in Parkinson's Disease: a comprehensive literature review. Cureus 2025; 17(3): e81129.
- Lou, JS. Physical and mental fatigue in Parkinson’s disease: epidemiology, pathophysiology and treatment. Drugs Aging 2009; 26(3): 195-208.
- Kang SY, Bang M, Hong JY et al. Neural and dopaminergic correlates of fatigue in Parkinson's disease. J Neural Transm 2020; 127(3): 301-309.
- Di Vico IA, Moretto M, Tamanti A et al. Molecular-informed network analysis unveils fatigue-related functional connectivity in Parkinson's disease. 2025. [CrossRef]
- Pavese N, Metta V, Bose SK et al. Fatigue in Parkinson’s disease is linked to striatal and limbic serotonergic dysfunction. Brain 2010; 133(11): 3434–3443.
- Wang H, Liu Y, Zhao J, Guo X et al. Possible inflammatory mechanisms and predictors of Parkinson's disease patients with fatigue (brief review). Clin Neurol Neurosurg 2021; 208: 106844.
- Wang L, Yi H, Liang X et al.: Plasma TNF-α and phosphorylated α-syn are associated with fatigue in patients with Parkinson's disease. J Neuroimmunol 2023; 385: 578222.
- Versace V, Sebastianelli L, Ferrazzoli D et al. Intracortical GABAergic dysfunction in patients with fatigue and dysexecutive syndrome after COVID-19. Clin Neurophysiol 2021, 132(5): 1138-1143.
- McNeill A, Duran R, Hughes DA et al. A clinical and family history study of Parkinson's disease in heterozygous glucocerebrosidase mutation carriers. J Neurol Neurosurg Psychiatry 2012; 83(8): 853-854.
- Fu R, Cui SS, Du JJ et al. Fatigue correlates with LRRK2 G2385R variant in Chinese Parkinson's disease patients. Parkinsonism Relat Disord 2017; 44: 101-105.
- [194] Panigrahi B, Pillai KS, Radhakrishnan DM, Rajan R, Srivastava AK: Fatigue in Parkinson’s disease—a narrative review. Ann Mov Disord 2024, 7:157-71.
- Tinazzi M, Geroin C, Siciliano M et al. Pain and fatigue in Parkinson's disease: advances in diagnosis and management. Neurol Sci 2025; 46(6): 2437-2454.
- Pauletti C, Locuratolo N, Mannarelli D et al. Fatigue in fluctuating Parkinson’s disease patients: possible impact of safinamide. J Neural Transm 2023; 130(7): 915-923.
- Kluger BM, Parra V, Jacobson C et al. The prevalence of fatigue following deep brain stimulation surgery in Parkinson's disease and association with quality of life. Parkinsons Dis 2012; 2012:769506.
- Folkerts AK, Nielsen J, Gollan R et al. Physical exercise as a potential treatment for fatigue in Parkinson's disease? A systematic review and meta-analysis of pharmacological and non-pharmacological interventions. J Parkinsons Dis 2023; 13(5): 659-679.
- Lawrie S, Coe S, Mansoubi M et al. Dietary patterns and nonmotor symptoms in Parkinson's disease: a cross-sectional analysis. J Am Nutr Assoc 2023; 42(4): 393-402.
| Non-motor symptoms | Mean (%) |
|---|---|
| Urinary problems | 74% |
| Pain | 65% |
| Fatigue | 64% |
| Sleep problems | 62% |
| Constipation | 48% |
| Cognitive impairment | 46% |
| Depression/apathy | 43% |
| Excessive drooling | 35% |
| Related to the disease process or pathophysiology | Dopaminergic |
|---|---|
| Non-dopaminergic (cholinergic, serotonergic, noradrenergic, glutamatergic, and mixed) | |
| Related to non-motor fluctuations | Present only during OFF periods |
| Present during ON periods and worse in OFF | |
| Related to drug therapy for PD | e.g. hallucinations, impulse control disorders, excessive daytime sleepiness |
| Genetically determined | Dementia or MCI in patients with glucocerebrosidase mutation |
| Depression and sleep disorders in patients with LRRK-2 mutation |
| NMS | Brain region | Neurotransmitter | |||||
|---|---|---|---|---|---|---|---|
| DA | SE | NA | GLU | ACE | GABA | ||
| Hyposmia | Olfactory bulb & amigdala | √ | √ | ||||
| Hallucinations | Occipital cortex | √ | √ | √ | √ | ||
| Pain | Basal ganglia, locus coeruleus, raphe nucleus, amygdala, thalamus | √ | √ | ||||
| Anxiety | Limbic area including amygdala | √ | √ | √ | √ | √ | |
| Depression | Limbic & cortical areas | √ | √ | √ | √ | ||
| Early cognitive dysfunction | Frontal cortex | √ | √ | √ | √ | ||
| Dementia | Temporal, parietal & occipital lobes | √ | √ | ||||
| Sleep disturbances | Hypothalamus & reticular formation | √ | √ | √ | √ | √ | |
| Bladder hyperreflexia | Basal ganglia | √ | √ | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




