Submitted:
09 November 2025
Posted:
10 November 2025
You are already at the latest version
Abstract
Keywords:
Introduction
1. Nutrient Flow in Mycorrhizal-Plant-Belowground Herbivore Interactions
1.1. Mechanisms of AMF-Mediated Nutrient Uptake
1.2. Disturbance of Root Systems by Belowground Herbivores
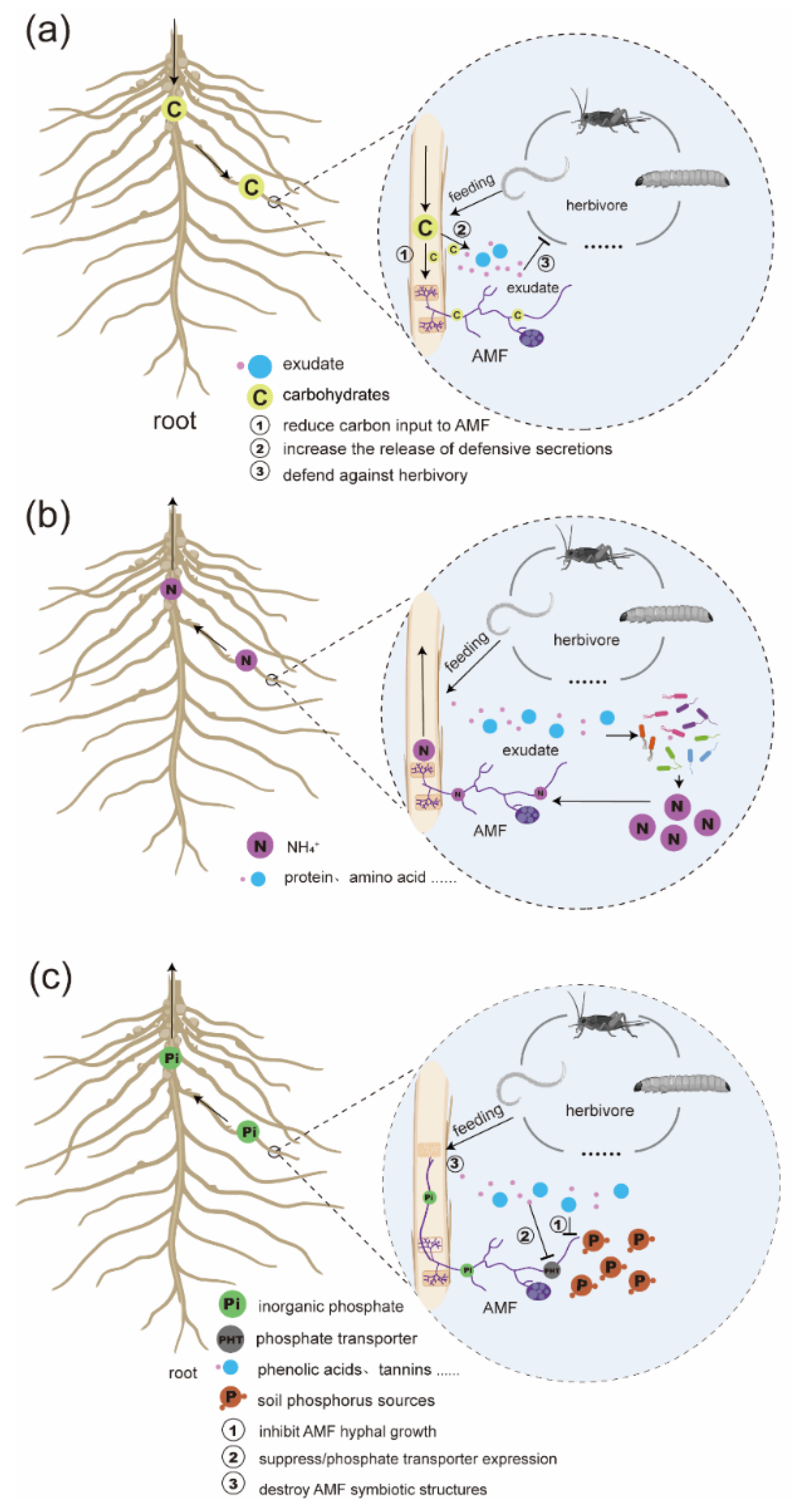
1.3. Carbon Flow in Mycorrhizal-Plant-Belowground Herbivore Interactions
1.4. Nitrogen Flow in Mycorrhizal-Plant-Belowground Herbivore Interactions
1.5. Phosphorus Flow in Mycorrhizal-Plant-Belowground Herbivore Systems
2. Nutrient Flow in Mycorrhizal-Plant-Aboveground Herbivore Interactions
2.1. Mechanisms of Bi-directional Nutrient Flow
2.2. Nutrient-mediated Plant Defense Strategy
2.3. The Impact of Aboveground Herbivory on Belowground Communities
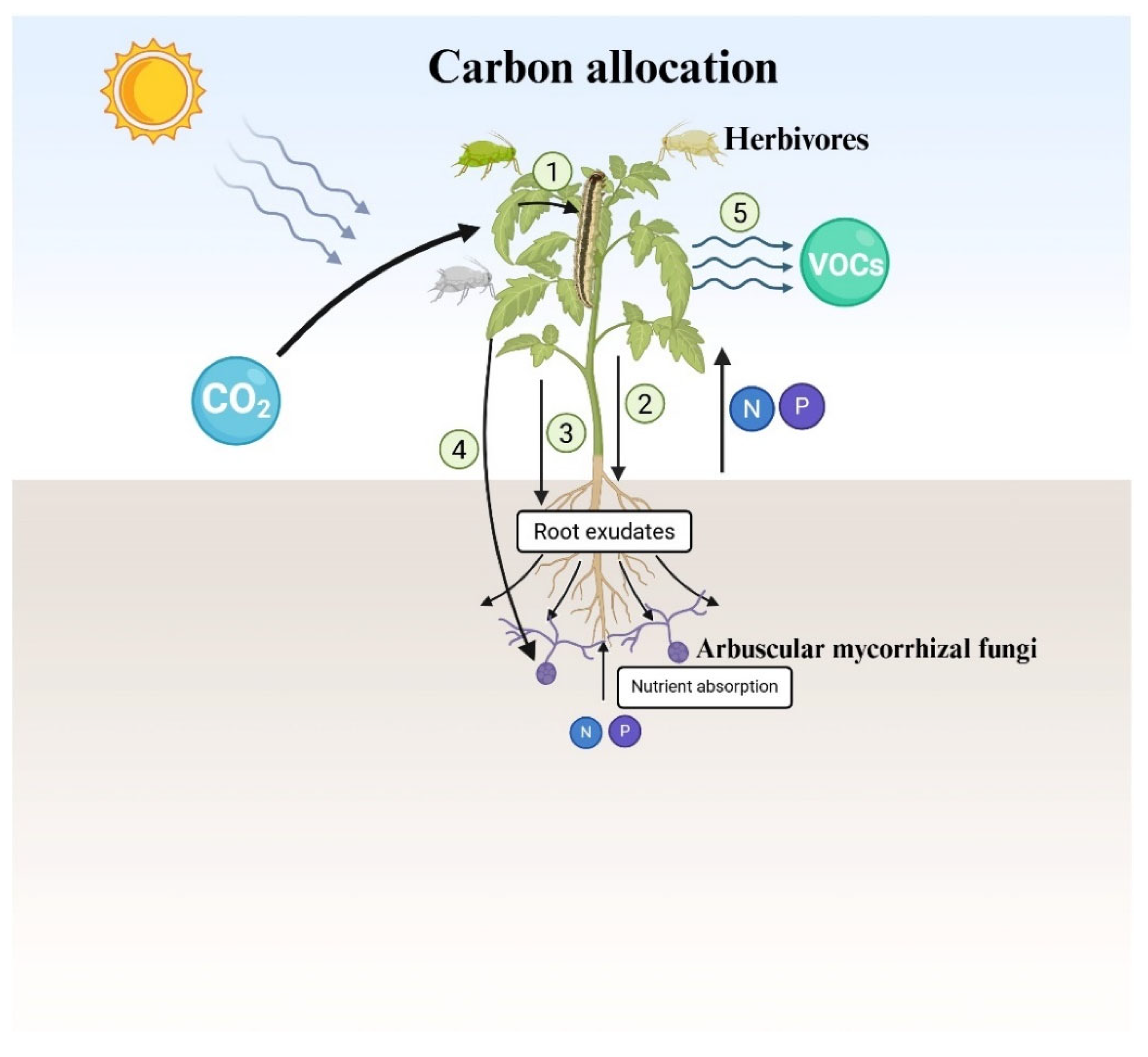
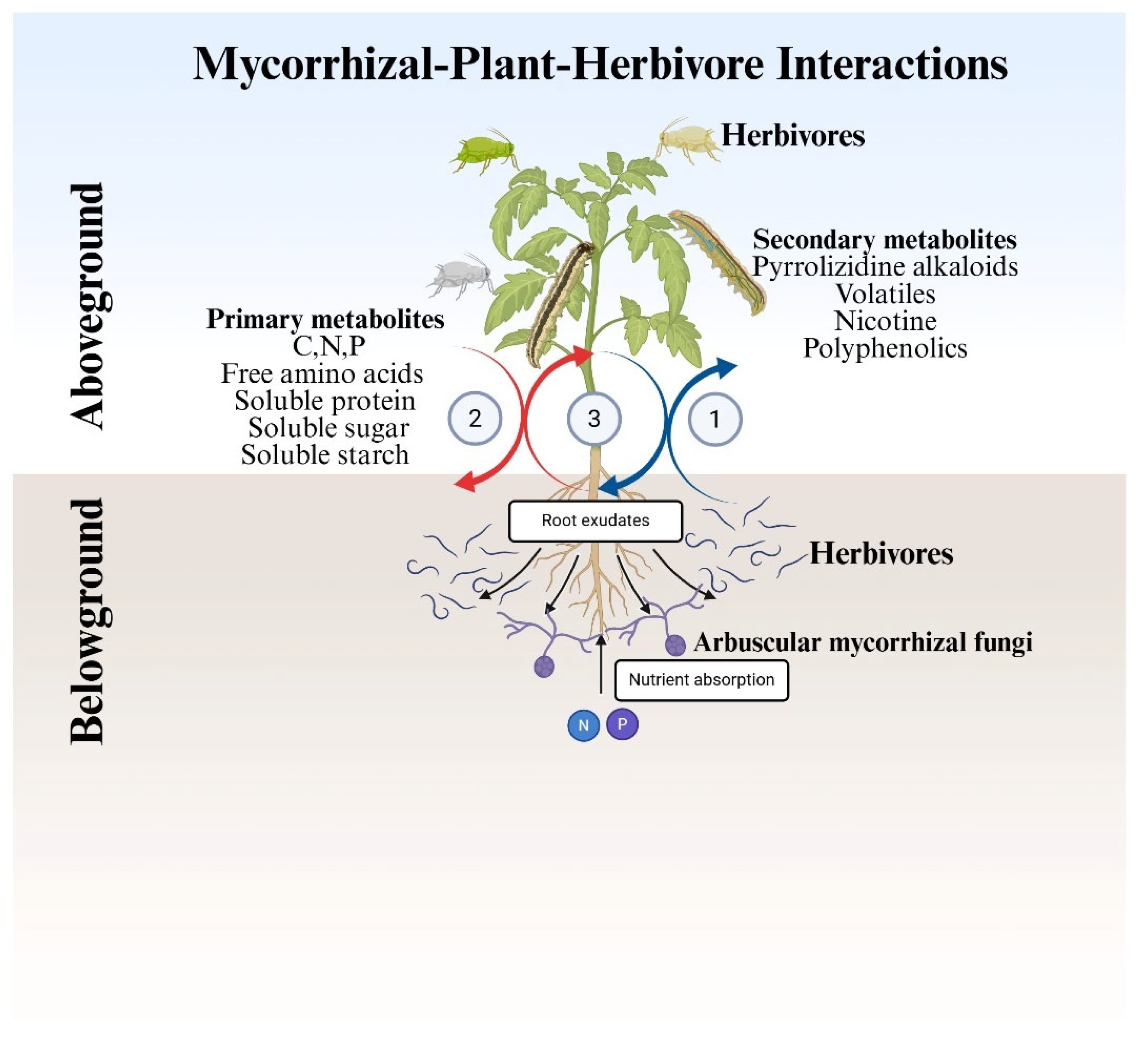
3. Nutrient Flow in Mycorrhizal-Plant-Above-and Below-ground Herbivore Interactions
3.1. Mechanisms in Mycorrhizal-Plant-Above-and Below-ground Herbivore Interactions
3.2. Primary and Secondary Metabolites in Multitrophic Interactions
3.3. Carbon, Nitrogen, and Phosphorus Flow
3.4. It Matters Who Comes First and How about the Amount and the Intensity of Damage
Author Contributions
Acknowledgments
References
- Achat, D.L., Pousse, N., Nicolas, M., Brédoire, F., Augusto, L., 2016. Soil properties controlling inorganic phosphorus availability: general results from a national forest network and a global compilation of the literature. Biogeochemistry 127, 255–272.
- Andrino, A., Guggenberger, G., Sauheitl, L., Burkart, S., Boy, J., 2021. Carbon investment into mobilization of mineral and organic phosphorus by arbuscular mycorrhiza. Biol. Fertil. Soils 57, 47–64.
- Aqueel, M.A., Collins, C.M., Raza, A. bakar M., Ahmad, S., Tariq, M., Leather, S.R., 2014. Effect of plant nutrition on aphid size, prey consumption, and life history characteristics of green lacewing. Insect Sci. 21, 74–82. [CrossRef]
- Ba, L., Ning, J., Wang, D., Facelli, E., Facelli, J.M., Yang, Y., Zhang, L., 2012. The relationship between the diversity of arbuscular mycorrhizal fungi and grazing in a meadow steppe. Plant Soil 352, 143–156. [CrossRef]
- Babikova, Z., Gilbert, L., Bruce, T., Dewhirst, S.Y., Pickett, J.A., Johnson, D., 2014. Arbuscular mycorrhizal fungi and aphids interact by changing host plant quality and volatile emission. Funct. Ecol. 28, 375–385. [CrossRef]
- Bakker, E.S., Olff, H., Boekhoff, M., Gleichman, J.M., Berendse, F., 2004. Impact of herbivores on nitrogen cycling: contrasting effects of small and large species. Oecologia 138, 91–101.
- Barber, N.A., Adler, L.S., Theis, N., Hazzard, R. V., Kiers, E.T., 2012. Herbivory reduces plant interactions with above- and belowground antagonists and mutualists. Ecology 93, 1560–1570. [CrossRef]
- Barto, E.K., Rillig, M.C., 2010. Does herbivory really suppress mycorrhiza? A meta-analysis. J. Ecol. 98, 745–753.
- Bell, C.A., Magkourilou, E., Urwin, P.E., Field, K.J., 2022. Disruption of carbon for nutrient exchange between potato and arbuscular mycorrhizal fungi enhanced cyst nematode fitness and host pest tolerance. New Phytol. 234, 269–279. [CrossRef]
- Belovsky, G.E., Slade, J.B., 2000. Insect herbivory accelerates nutrient cycling and increases plant production. Proc. Natl. Acad. Sci. U. S. A. 97, 14412–14417. [CrossRef]
- Bezemer, T.M., Van Dam, N.M., 2005. Linking aboveground and belowground interactions via induced plant defenses. Trends Ecol. Evol. 20, 617–624. [CrossRef]
- Biere, A., Goverse, A., 2016. Plant-Mediated Systemic Interactions Between Pathogens, Parasitic Nematodes, and Herbivores Above- and Belowground. Annu. Rev. Phytopathol. 54, 499–527. [CrossRef]
- Bonkowski, M., Villenave, C., Griffiths, B., 2009. Rhizosphere fauna: the functional and structural diversity of intimate interactions of soil fauna with plant roots. Plant Soil 321, 213–233. [CrossRef]
- Bryant, J.P., Chapin, F.S., Klein, D.R., Carbon, D.R., 1983. Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40, 357–368.
- Burghardt, K.T., 2016. Nutrient supply alters goldenrod’s induced response to herbivory. Funct. Ecol. 30, 1769–1778. [CrossRef]
- Charters, M.D., Sait, S.M., Field, K.J., 2020. Aphid Herbivory Drives Asymmetry in Carbon for Nutrient Exchange between Plants and an Arbuscular Mycorrhizal Fungus. Curr. Biol. 30, 1801-1808.e5. [CrossRef]
- Cibils-Stewart, X., Vandegeer, R.K., Mace, W.J., Hartley, S.E., Powell, J.R., Popay, A.J., Johnson, S.N., 2025. Mycorrhizal fungi compromise production of endophytic alkaloids, increasing plant susceptibility to an aphid herbivore. J. Ecol. 113, 1368–1381. [CrossRef]
- Coley, P.D., Bryant, J.P., Chapin, F.S., 1985. Resource availability and plant antiherbivore defense. Science (80-. ). 230, 895–899. [CrossRef]
- Contreras-Cornejo, H.A., Macias-Rodriguez, L., Real-Santillan, R.O., Lopez-Carmona, D., Garcia-Gomez, G., Galicia-Gallardo, A.P., Alfaro-Cuevas, R., Gonzalez-Esquivel, C.E., Najera-Rincon, M.B., Adame-Garnica, S.G., Rebollar-Alviter, A., Alvarez-Navarrete, M., Larsen, J., 2021. In a belowground multitrophic interaction, Trichoderma harzianum induces maize root herbivore tolerance against Phyllophaga vetula. PEST Manag. Sci. 77, 3952–3963. [CrossRef]
- Cosme, M., Stout, M.J., Wurst, S., 2011. Effect of arbuscular mycorrhizal fungi (Glomus intraradices) on the oviposition of rice water weevil (Lissorhoptrus oryzophilus). Mycorrhiza 21, 651–658. [CrossRef]
- Delavaux, C.S., Burrill, H., Menning, R., Duell, E.B., Bryant, R.L., Lubin, T., Bever, J.D., 2025. Origin matters: mycorrhizal growth response and induced resistance to pathogens depend on mycorrhizal and pathogen source. New Phytol. [CrossRef]
- Duell, E.B., Todd, T.C., Wilson, G.W.T., 2025. Mycorrhizal-herbivore interactions and the competitive release of subdominant tallgrass prairie species. J. Ecol. 113, 1409–1421. [CrossRef]
- Durant, E., Hoysted, G.A., Howard, N., Sait, S.M., Childs, D.Z., Johnson, D., Field, K.J., 2023. Herbivore-driven disruption of arbuscular mycorrhizal carbon-for-nutrient exchange is ameliorated by neighboring plants. Curr. Biol. 33, 2566–2573. [CrossRef]
- Elliott, A.J., Daniell, T.J., Cameron, D.D., Field, K.J., 2021. A commercial arbuscular mycorrhizal inoculum increases root colonization across wheat cultivars but does not increase assimilation of mycorrhiza-acquired nutrients. Plants People Planet 3, 588–599. [CrossRef]
- Eskelinen, A., Jessen, M.T., Bahamonde, H.A., Bakker, J.D., Borer, E.T., Caldeira, M.C., Harpole, W.S., Jia, M., Lannes, L.S., Nogueira, C., Olde Venterink, H., Peri, P.L., Porath-Krause, A.J., Seabloom, E.W., Schroeder, K., Tognetti, P.M., Yasui, S.L.E., Virtanen, R., Sullivan, L.L., 2023. Herbivory and nutrients shape grassland soil seed banks. Nat. Commun. 14, 3949. [CrossRef]
- Ferlian, O., Goldmann, K., Eisenhauer, N., Tarkka, M.T., Buscot, F., Heintz-Buschart, A., 2021. Distinct effects of host and neighbour tree identity on arbuscular and ectomycorrhizal fungi along a tree diversity gradient. ISME Commun. 1, 40. [CrossRef]
- Fernandez-Conradi, P., Jactel, H., Robin, C., Tack, A.J.M., Castagneyrol, B., 2018. Fungi reduce preference and performance of insect herbivores on challenged plants. Ecology 99, 300–311. [CrossRef]
- Frew, A., 2021. Aboveground herbivory suppresses the arbuscular mycorrhizal symbiosis, reducing plant phosphorus uptake. Appl. SOIL Ecol. 168. [CrossRef]
- Frew, A., Antunes, P.M., Cameron, D.D., Hartley, S.E., Johnson, S.N., Rillig, M.C., Bennett, A.E., 2022. Plant herbivore protection by arbuscular mycorrhizas: a role for fungal diversity? New Phytol. 233, 1022–1031. [CrossRef]
- Frew, A., Powell, J.R., Allsopp, P.G., Sallam, N., Johnson, S.N., 2017. Arbuscular mycorrhizal fungi promote silicon accumulation in plant roots, reducing the impacts of root herbivory. Plant Soil 419, 423–433. [CrossRef]
- Frew, A., Weinberger, N., Powell, J.R., Watts-Williams, S.J., Aguilar-Trigueros, C.A., 2024. Community assembly of root-colonizing arbuscular mycorrhizal fungi: beyond carbon and into defence? ISME J. 18. [CrossRef]
- Frew, A., Wilson, B.A.L., 2021. Different mycorrhizal fungal communities differentially affect plant phenolic-based resistance to insect herbivory. Rhizosphere 19, 100365. [CrossRef]
- Gange, A.C., Bower, E., Brown, V.K., 2002. Differential effects of insect herbivory on arbuscular mycorrhizal colonization. Oecologia 131, 103–112. [CrossRef]
- Gange, A.C., Brown, V.K., 2002. Soil food web components affect plant community structure during early succession. Ecol. Res. 17, 217–227. [CrossRef]
- Govindarajulu, M., Pfeffer, P.E., Jin, H., Abubaker, J., Douds, D.D., Allen, J.W., Lammers, P.J., Shachar-hill, Y., 2005. Nitrogen transfer in the arbuscular mycorrhizal symbiosis. Nature 435, 819–823. [CrossRef]
- Grman, E., 2012. Plant species differ in their ability to reduce allocation to non-beneficial arbuscular mycorrhizal fungi. Ecology 93, 711–718.
- Hamilton III, E.W., Frank, D.A., 2001. Can plants stimulate soil microbes and their own nutrient supply? Evidence from a grazing tolerant grass. Ecology 82, 2397–2402.
- Harrison, M.J., Dewbre, G.R., Liu, J.Y., 2002. A phosphate transporter from Medicago truncatula involved in the acquisiton of phosphate released by arbuscular mycorrhizal fungi. Plant Cell 14, 2413–2429. [CrossRef]
- Helfenstein, J., Ringeval, B., Tamburini, F., Mulder, V.L., Goll, D.S., He, X., Alblas, E., Wang, Y., Mollier, A., Frossard, E., 2024. Understanding soil phosphorus cycling for sustainable development: A review. One Earth 7, 1727–1740.
- Hodge, A., Fitter, A.H., 2010. Substantial nitrogen acquisition by arbuscular mycorrhizal fungi from organic material has implications for N cycling. Proc. Natl. Acad. Sci. 107, 31. [CrossRef]
- Hol, W.H.G., Cook, R., 2005. An overview of arbuscular mycorrhizal fungi–nematode interactions. Basic Appl. Ecol. 6, 489–503.
- Huang, W., Robert, C.A.M., Hervé, M.R., Hu, L., Bont, Z., Erb, M., 2017. A mechanism for sequence specificity in plant-mediated interactions between herbivores. New Phytol. 214, 169–179. [CrossRef]
- Huang, W., Siemann, E., Xiao, L., Yang, X., Ding, J., 2014. Species-specific defence responses facilitate conspecifics and inhibit heterospecifics in above-belowground herbivore interactions. Nat. Commun. 5, 4851. [CrossRef]
- Ishida, T., Suzuki, R., Nakagami, S., Kuroha, T., Sakamoto, S., Nakata, M.T., Yokoyama, R., Kimura, S., Mitsuda, N., Nishitani, K., 2020. Root-knot nematodes modulate cell walls during root-knot formation in Arabidopsis roots. J. Plant Res. 133, 419–428.
- Johnson, N.C., Wilson, G.W.T., Bowker, M.A., Wilson, J.A., Miller, R.M., 2010. Resource limitation is a driver of local adaptation in mycorrhizal symbioses. Proc. Natl. Acad. Sci. 107, 2093–2098. [CrossRef]
- Johnson, S.N., Clark, K.E., Hartley, S.E., Jones, T.H., McKenzie, S.W., Koricheva, J., 2012. Aboveground – belowground herbivore interactions : a meta-analysis. Ecology 93, 2208–2215.
- Johnson, S.N., Mitchell, C., Mcnicol, J.W., Thompson, J., Karley, A.J., 2013. Downstairs drivers - root herbivores shape communities of above-ground herbivores and natural enemies via changes in plant nutrients. J. Anim. Ecol. 82, 1021–1030. [CrossRef]
- Kafle, D., Haenel, A., Lortzing, T., Steppuhn, A., Wurst, S., 2017. Sequential above-and belowground herbivory modifies plant responses depending on herbivore identity. BMC Ecol. 17, 5. [CrossRef]
- Kaplan, I., Halitschke, R., Kessler, A., Rehill, B.J., Sardanelli, S., Denno, R.F., 2008a. Physiological integration of roots and shoots in plant defense strategies links above- and belowground herbivory. Ecol. Lett. 11, 841–851. [CrossRef]
- Kaplan, I., Halitschke, R., Kessler, A., Sardanelli, S., Denno, R.F., 2008b. Constitutive and induced defenses to herbivory in above- and belowground plant tissues. Ecology 89, 392–406. [CrossRef]
- Katayama, N., Silva, A.O., Kishida, O., Ushio, M., Kita, S., Ohgushi, T., 2014. Herbivorous insect decreases plant nutrient uptake: The role of soil nutrient availability and association of below-ground symbionts. Ecol. Entomol. 39, 511–518. [CrossRef]
- Khan, G.A., Vogiatzaki, E., Glauser, G., Poirier, Y., 2016. Phosphate deficiency induces the jasmonate pathway and enhances resistance to insect herbivory. Plant Physiol. 171, 632–644. [CrossRef]
- Kiers, E.T., Duhamel, M., Beesetty, Y., Mensah, J.A., Franken, O., Verbruggen, E., Fellbaum, C.R., Kowalchuk, G.A., Hart, M.M., Bago, A., 2011. Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science (80-. ). 333, 880–882.
- Konvalinková, T., Püschel, D., Řezáčová, V., Gryndlerová, H., Jansa, J., 2017. Carbon flow from plant to arbuscular mycorrhizal fungi is reduced under phosphorus fertilization. Plant Soil 419, 319–333.
- Kostenko, O., van de Voorde, T.F.J., Mulder, P.P.J., van der Putten, W.H., Martijn Bezemer, T., 2012. Legacy effects of aboveground-belowground interactions. Ecol. Lett. 15, 813–821. [CrossRef]
- Kristensen, J. a., Rousk, J., Metcalfe, D.B., 2020. Below-ground responses to insect herbivory in ecosystems with woody plant canopies: A meta-analysis. J. Ecol. 108, 917–930. [CrossRef]
- Lambers, H., Mougel, C., Jaillard, B., Hinsinger, P., 2009. Plant-microbe-soil interactions in the rhizosphere: an evolutionary perspective. Plant Soil 18, 253–264.
- Lanoue, A., Burlat, V., Schurr, U., Röse, U.S.R., 2010. Induced root-secreted phenolic compounds as a belowground plant defense. Plant Signal. Behav. 5, 1037–1038.
- Li, X., Guo, W., Siemann, E., Wen, Y., Huang, W., Ding, J., 2016. Plant genotypes affect aboveground and belowground herbivore interactions by changing chemical defense. Oecologia 182, 1107–1115. [CrossRef]
- Li, Y., Gao, Y., van Kleunen, M., Liu, Y., 2022. Herbivory may mediate the effects of nutrients on the dominance of alien plants. Funct. Ecol. 36, 1292–1302. [CrossRef]
- Machado, R.A.R., Arce, C.C.M., McClure, M.A., Baldwin, I.T., Erb, M., 2018. Aboveground herbivory induced jasmonates disproportionately reduce plant reproductive potential by facilitating root nematode infestation. Plant Cell Environ. 41, 797–808. [CrossRef]
- Magkourilou, E., Durant, E.K., Bell, C.A., Daniell, T.J., Urwin, P.E., Field, K.J., 2025. Plant pests influence the movement of plant-fixed carbon and fungal-acquired nutrients through arbuscular mycorrhizal networks. Funct. Ecol. 39, 1400–1410. [CrossRef]
- Martijn Bezemer, T., van der Putten, W.H., Martens, H., van de Voorde, T.F.J., Mulder, P.P.J., Kostenko, O., 2013. Above- and below-ground herbivory effects on below-ground plant-fungus interactions and plant-soil feedback responses. J. Ecol. 101, 325–333. [CrossRef]
- Martin, F.M., Uroz, S., Barker, D.G., 2017. Ancestral alliances: Plant mutualistic symbioses with fungi and bacteria. Science (80-. ). 356, eaad4501. [CrossRef]
- Martínez-Medina, A., Fernandez, I., Lok, G.B., Pozo, M.J., Pieterse, C.M.J., Van Wees, S.C.M., 2017. Shifting from priming of salicylic acid-to jasmonic acid-regulated defences by Trichoderma protects tomato against the root knot nematode Meloidogyne incognita. New Phytol. 213, 1363–1377.
- Masters, A.G.J., Brown, V.K., Gange, A.C., 1993. Plant mediated interactions between above-and below-ground insect herbivores. Oikos 66, 148–151.
- Meesters, C., Cialdella, L., Ingels, R., Jacquemyn, H., 2023. Cultivar - dependent effects of plant - beneficial fungi on plant nutrient composition and feeding damage by Nesidiocoris tenuis. Plant Soil 492, 177–190. [CrossRef]
- Meier, A.R., Hunter, M.D., 2018. Arbuscular mycorrhizal fungi mediate herbivore-induction of plant defenses differently above and belowground. Oikos 127, 1759–1775. [CrossRef]
- Morris, W.F., Hufbauer, R.A., Agrawal, A.A., Bever, J.D., Borowicz, V.A., Gilbert, G.S., Maron, J.L., Mitchell, C.E., Parker, I.M., Power, A.G., Torchin, M.E., Vaźquez, D.P., 2007. Direct and interactive effects of enemies and mutualists on plant performance: A meta-analysis. Ecology 88, 1021–1029. [CrossRef]
- Ng, A., Wilson, B.A.L., Frew, A., 2023. Belowground crop responses to root herbivory are associated with the community structure of native arbuscular mycorrhizal fungi. Appl. Soil Ecol. 185, 104797. [CrossRef]
- Orians, C.M., Thorn, A., Gómez, S., 2011. Herbivore-induced resource sequestration in plants: Why bother? Oecologia 167, 1–9. [CrossRef]
- Ourry, M., Lebreton, L., Chaminade, V., Guillerm-Erckelboudt, A.-Y., Hervé, M., Linglin, J., Marnet, N., Ourry, A., Paty, C., Poinsot, D., 2018. Influence of belowground herbivory on the dynamics of root and rhizosphere microbial communities. Front. Ecol. Evol. 6, 91.
- Parniske, M., 2008. Arbuscular mycorrhiza: the mother of plant root endosymbioses. Nat. Rev. Microbiol. 6, 763–775.
- Paszkowski, U., Kroken, S., Roux, C., Briggs, S.P., 2002. Rice phosphate transporters include an evolutionarily divergent gene specifically activated in arbuscular mycorrhizal symbiosis. Proc. Natl. Acad. Sci. 99, 13324–13329.
- Piippo, S., Markkola, A., Harma, E., Tuomi, J., 2011. Do compensatory shoot growth and mycorrhizal symbionts act as competing above- and below-ground sinks after simulated grazing? PLANT Ecol. 212, 33–42. [CrossRef]
- Piotrowski, J.S., Morford, S.L., Rillig, M.C., 2008. Inhibition of colonization by a native arbuscular mycorrhizal fungal community via Populus trichocarpa litter, litter extract, and soluble phenolic compounds. Soil Biol. Biochem. 40, 709–717.
- Prescott, C.E., Grayston, S.J., Helmisaari, H.S., Kaštovská, E., Körner, C., Lambers, H., Meier, I.C., Millard, P., Ostonen, I., 2020. Surplus Carbon Drives Allocation and Plant–Soil Interactions. Trends Ecol. Evol. 35, 1110–1118. [CrossRef]
- Real-Santillán, R.O., del-Val, E., Cruz-Ortega, R., Contreras-Cornejo, H.Á., González-Esquivel, C.E., Larsen, J., 2019. Increased maize growth and P uptake promoted by arbuscular mycorrhizal fungi coincide with higher foliar herbivory and larval biomass of the Fall Armyworm Spodoptera frugiperda. Mycorrhiza 29, 615–622. [CrossRef]
- Rutter, W.B., Franco, J., Gleason, C., 2022. Rooting out the mechanisms of root-knot nematode–plant interactions. Annu. Rev. Phytopathol. 60, 43–76.
- Schreiner, R.P., Pinkerton, J.N., 2008. Ring nematodes (Mesocriconema xenoplax) alter root colonization and function of arbuscular mycorrhizal fungi in grape roots in a low P soil. Soil Biol. Biochem. 40, 1870–1877.
- Shan, L., Oduor, A.M.O., Huang, W., Liu, Y., 2024. Nutrient enrichment promotes invasion success of alien plants via increased growth and suppression of chemical defenses. Ecol. Appl. 34, e2791. [CrossRef]
- Sharma, E., Anand, G., Kapoor, R., 2017. Terpenoids in plant and arbuscular mycorrhiza-reinforced defence against herbivorous insects. Ann. Bot. 119, 791–801. [CrossRef]
- Smith, S.E., Read, D.J., 2010. Mycorrhizal symbiosis. Academic press.
- Tao, L., Ahmad, A., de Roode, J.C., Hunter, M.D., 2016. Arbuscular mycorrhizal fungi affect plant tolerance andchemical defences to herbivory through different mechanisms. J. Ecol. 104, 561–571. [CrossRef]
- Thompson, M.N., Arriaga, J., Bradford, B.J., Kurian, R., Strozier, G., Helms, A.M., 2024. Belowground insect herbivory induces systemic volatile emissions that strengthen neighbouring plant resistance aboveground. Plant Cell Environ. 47, 714–725. [CrossRef]
- Tronson, E., Enders, L., 2025. Root microbes can improve plant tolerance to insect damage: A systematic review and meta-analysis. Ecology 106, e4502. [CrossRef]
- Van Dam, N.M., Raaijmakers, C.E., Van Der Putten, W.H., 2005. Root herbivory reduces growth and survival of the shoot feeding specialist Pieris rapae on Brassica nigra. Entomol. Exp. Appl. 115, 161–170. [CrossRef]
- Vance, C.P., Uhde-Stone, C., Allan, D.L., 2003. Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol. 157, 423–447.
- Vannette, R.L., Hunter, M.D., 2013. Mycorrhizal abundance affects the expression of plant resistance traits and herbivore performance. J. Ecol. 101, 1019–1029. [CrossRef]
- Wang, M., Biere, A., Van der Putten, W.H., Bezemer, T.M., 2014. Sequential effects of root and foliar herbivory on aboveground and belowground induced plant defense responses and insect performance. Oecologia 175, 187–198. [CrossRef]
- Wang, M., Wang, Z., Guo, M., Qu, L., Biere, A., 2023. Effects of arbuscular mycorrhizal fungi on plant growth and herbivore infestation depend on availability of soil water and nutrients. Front. Plant Sci. 14, 1101932. [CrossRef]
- Wang, W., Shi, J., Xie, Q., Jiang, Y., Yu, N., Wang, E., 2017. Nutrient Exchange and Regulation in Arbuscular Mycorrhizal Symbiosis. Mol. Plant 10, 1147–1158. [CrossRef]
- Wieczorek, K., Bell, C.A., 2025. Exploited mutualism: the reciprocal effects of plant parasitic nematodes on the mechanisms underpinning plant–mutualist interactions. New Phytol. 246, 2435–2439. [CrossRef]
- Xing, Z., Ma, T., Wu, L., Zhang, Z., Ding, J., Siemann, E., 2024a. Foliar herbivory modifies arbuscular mycorrhizal fungal colonization likely through altering root flavonoids. Funct. Ecol. 38, 259–271. [CrossRef]
- Xing, Z., Zhang, Z., Zhao, Y., Biere, A., Liu, S., Shi, Y., Ding, J., 2024b. Foliar herbivory-enhanced mycorrhization is associated with increased levels of lipids in root and root exudates. J. Ecol. 112, 701–716. [CrossRef]
- Yu, L., Zhang, W., Geng, Y., Liu, K., Shao, X., 2022. Cooperation With Arbuscular Mycorrhizal Fungi Increases Plant Nutrient Uptake and Improves Defenses Against Insects. Front. Ecol. Evol. 10, 833389. [CrossRef]
- Zeng, M., Hause, B., van Dam, N.M., Uthe, H., Hoffmann, P., Krajinski, F., Martínez-Medina, A., 2022. The mycorrhizal symbiosis alters the plant defence strategy in a model legume plant. Plant Cell Environ. 45, 3412–3428. [CrossRef]
- Zhao, R., Wang, C., Koorem, K., Song, X., Siemann, E., Ding, J., Yang, Q., 2024. Aboveground antagonists mitigate belowground plant–antagonist interactions but not affect plant–mutualist interactions. Eur. J. Soil Biol. 120, 103577. [CrossRef]
- Zhou, J., Jia, F., Shao, S., Zhang, H., Li, G., Xia, X., Zhou, Y., Yu, J., Shi, K., 2015. Involvement of nitric oxide in the jasmonate-dependent basal defense against root-knot nematode in tomato plants. Front. Plant Sci. 6, 193.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



