Submitted:
08 November 2025
Posted:
12 November 2025
You are already at the latest version
Abstract
Keywords:
Background
Methods
Study Design and Data Sources
Study Participants
Outcome Measures
Demographic and Behavioural Variables
Statistical Analysis
Descriptive Statistics and Exploratory Analysis
Propensity Score Matching (PSM)
Primary Effectiveness Analysis
Longitudinal Trajectory Analyses
Predictors of Success Analysis
Ethical Considerations
Results
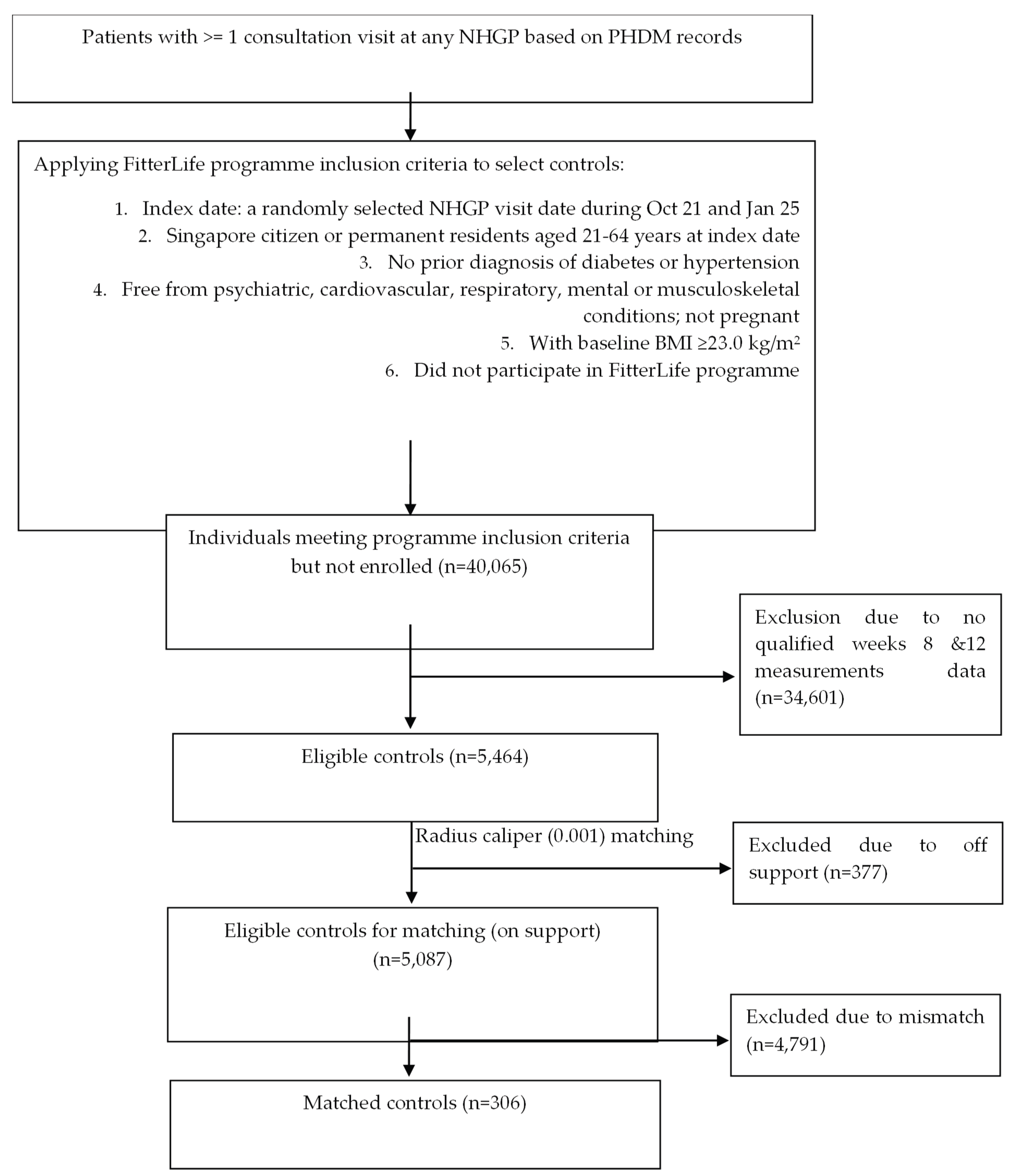
Participant Characteristics and Propensity Score Matching
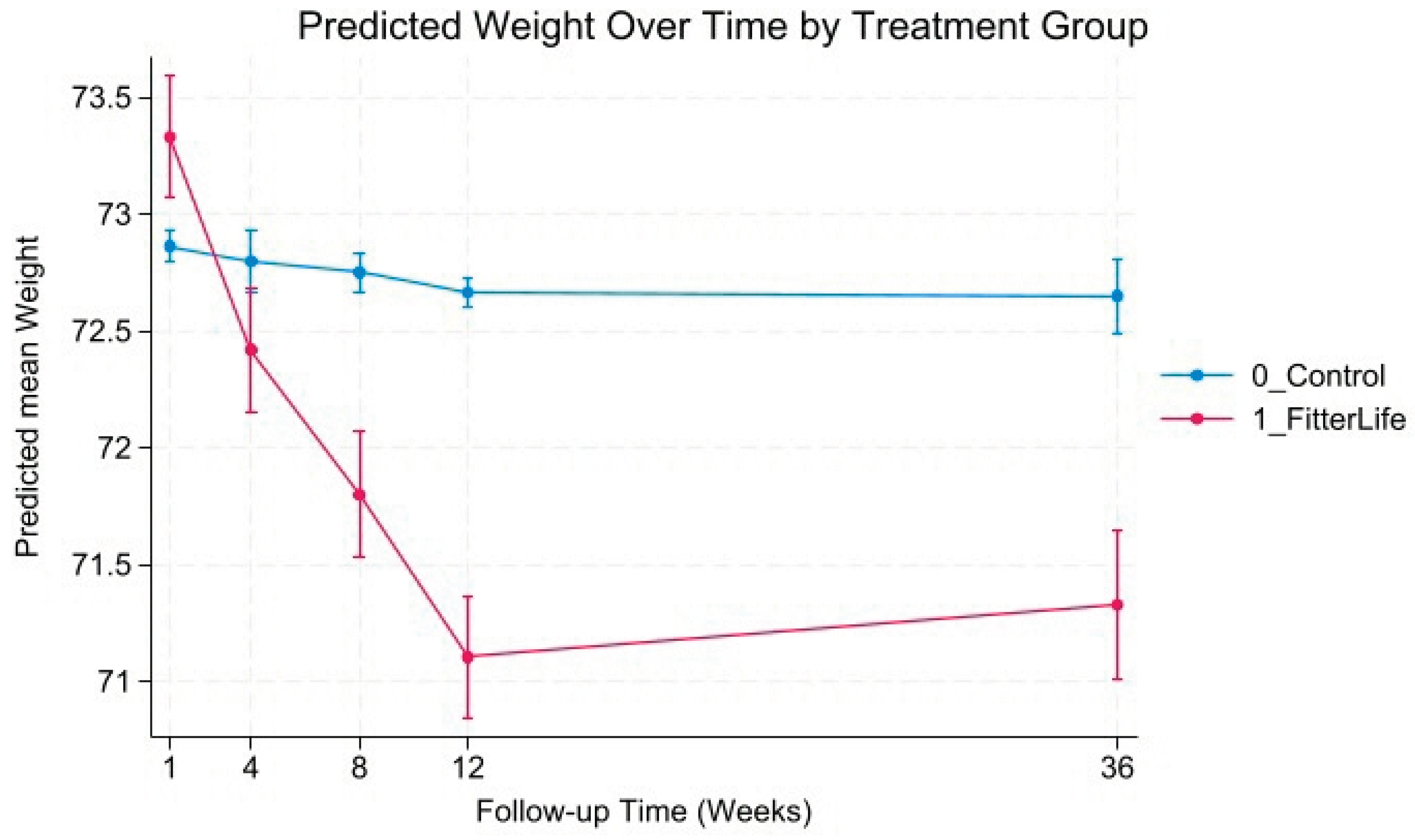
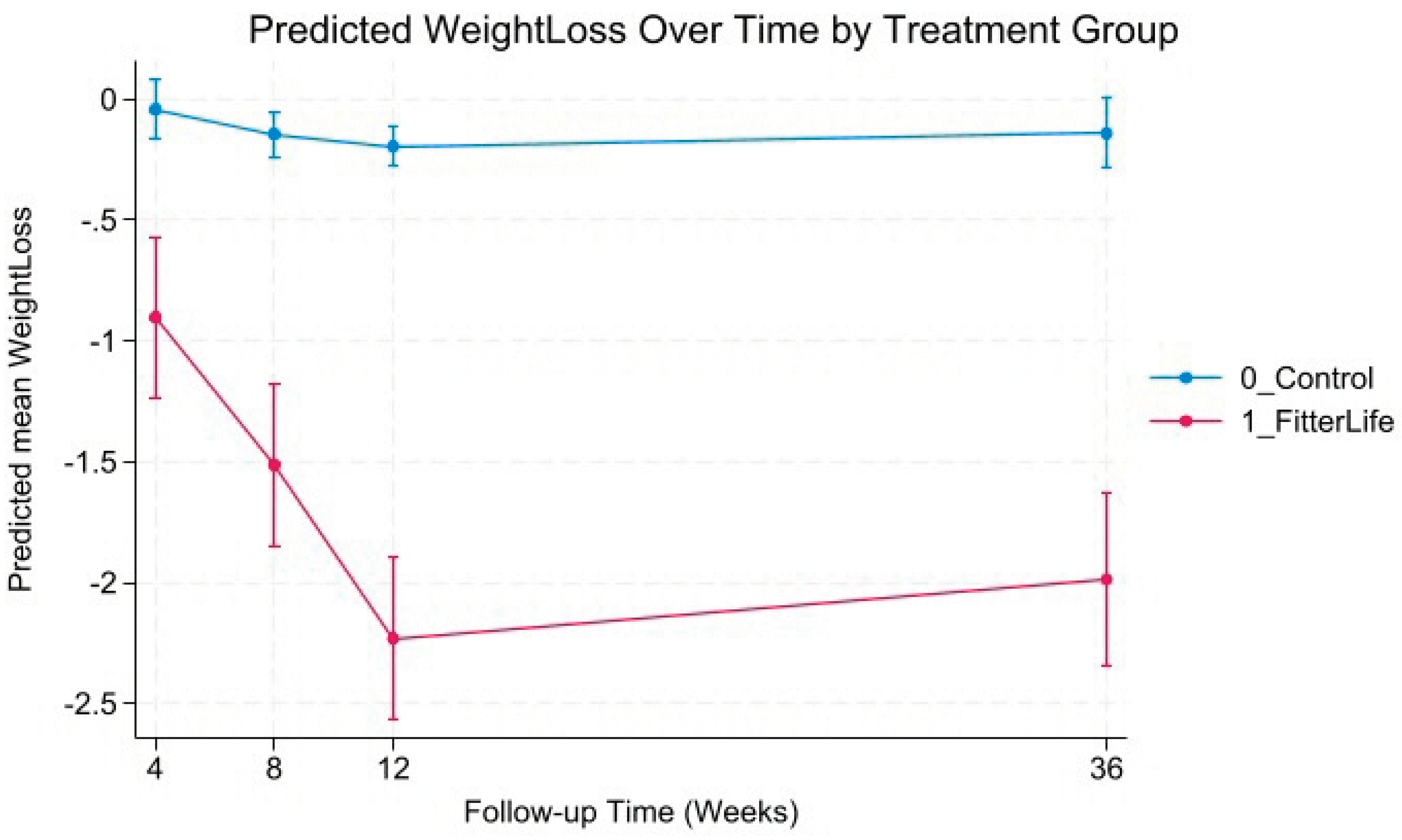
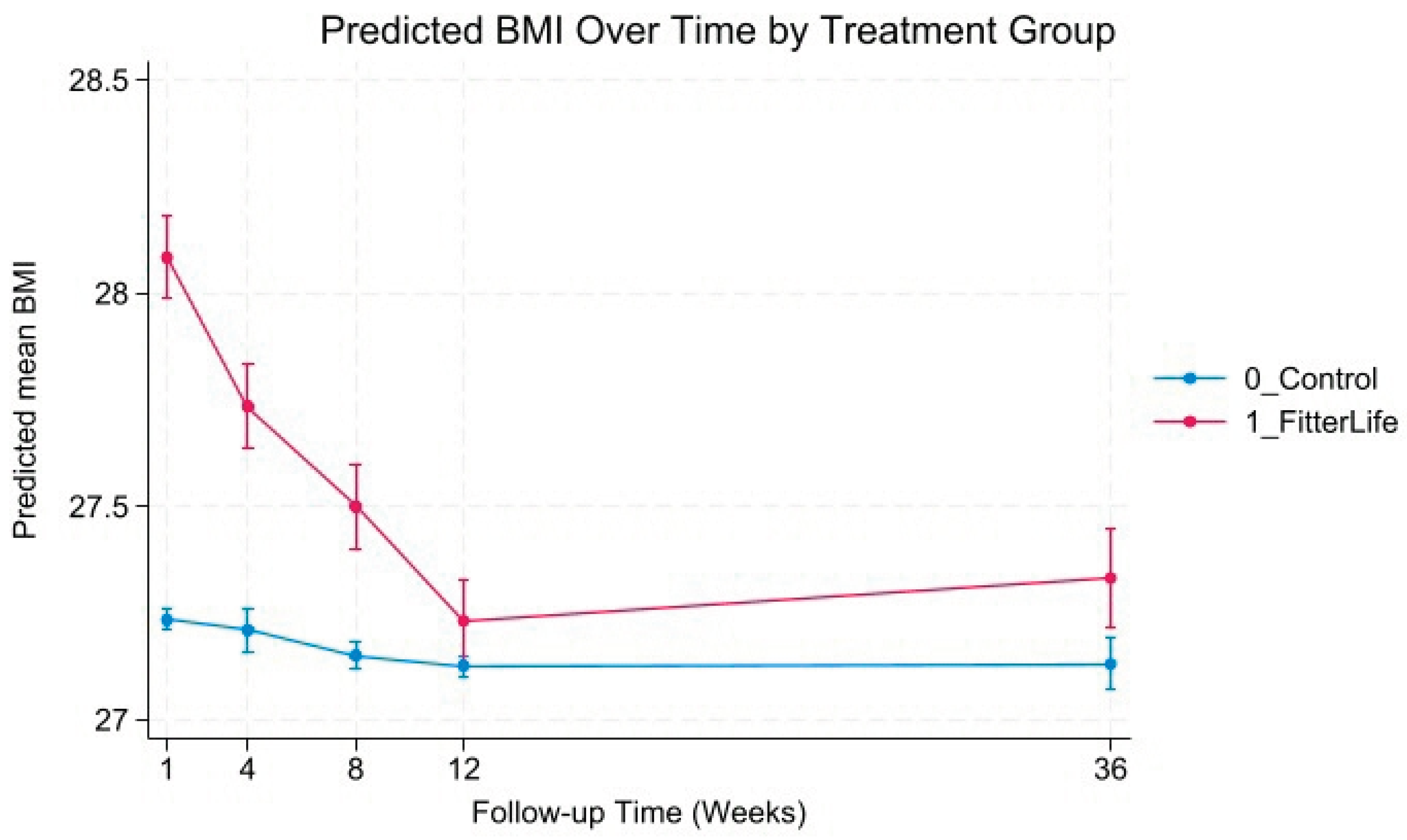
Effectiveness on Weight Loss Target
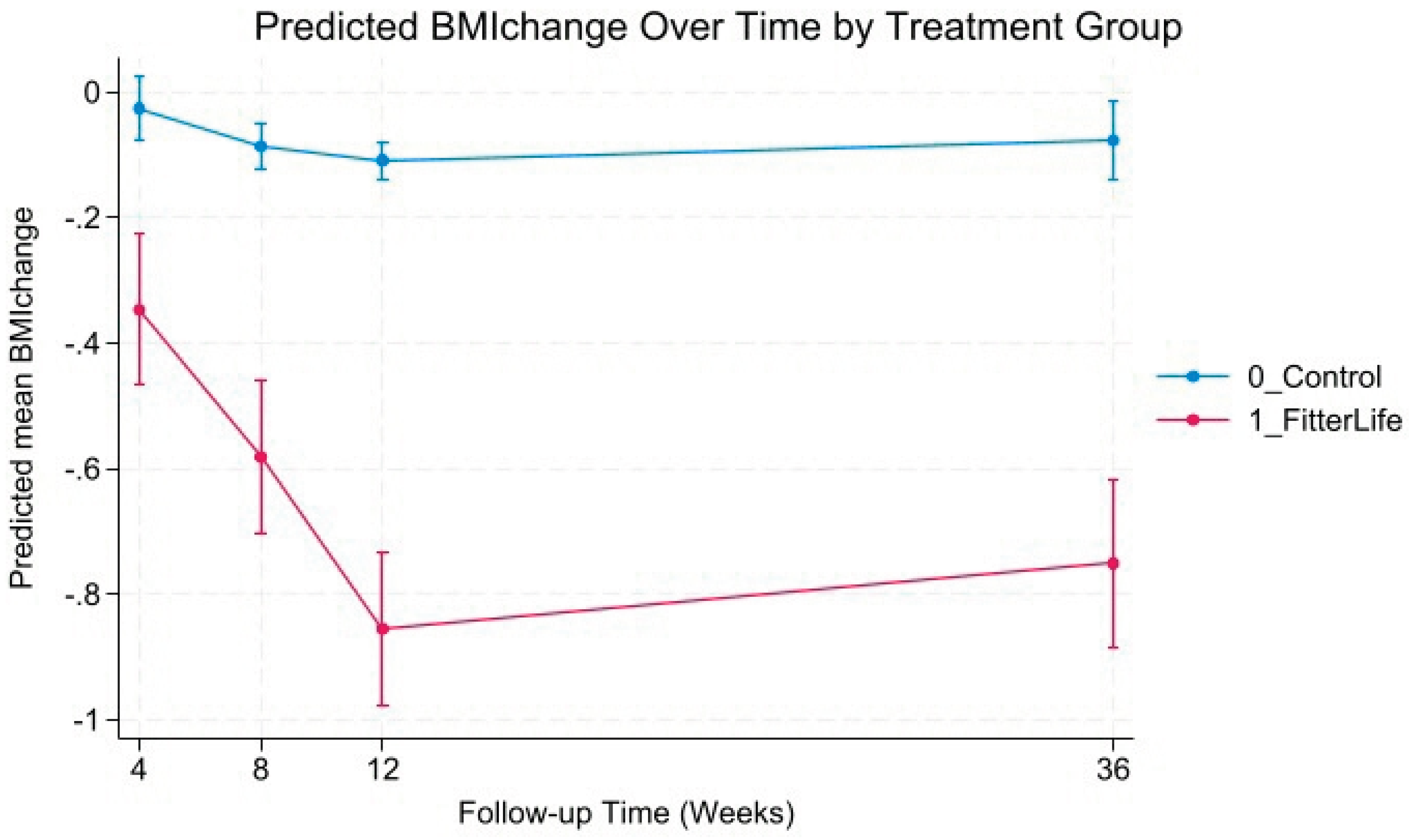
Trajectories of Weight and BMI Change
Behavioural factors Associated with Weight Loss Success
Discussion
Strengths and Limitations
Programme Implications and Future Directions
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interests
Abbreviations
| aIRR | Adjusted incidence rate ratio; |
| ATET | average treatment effect on the treated; |
| BMI | Body Mass Index; |
| IPWRA | inverse probability weighted regression adjustment; |
| MVPA | moderate-to-vigorous physical activity; |
| NHG | National Healthcare Group; |
| NHGP | National Healthcare Group Polyclinic; |
| PHDM | NHG Population Health Data Mart; |
| PSM | propensity score matching; |
| SD | standard deviations; |
| VIFs | variance inflation factors |
References
- Lam BCC, Lim AYL, Chan SL, Yum MPS, Koh NSY, Finkelstein EA. The impact of obesity: a narrative review. Singapore Med J. 2023;64:163–71. [CrossRef]
- Chandrasekaran P, Weiskirchen R. The Role of Obesity in Type 2 Diabetes Mellitus-An Overview. Int J Mol Sci. 2024;25:1882. [CrossRef]
- Blüher, M. An overview of obesity-related complications: The epidemiological evidence linking body weight and other markers of obesity to adverse health outcomes. Diabetes Obes Metab. 2025;27 Suppl 2 Suppl 2:3–19. [CrossRef]
- WHO WHO. Obesity and overweight. Fact sheets. 2025. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. Accessed 19 Sep 2025.
- Fontaine KR, Redden DT, Wang C, Westfall AO, Allison DB. Years of life lost due to obesity. JAMA. 2003;289:187–93. [CrossRef]
- Bhaskaran K, Dos-Santos-Silva I, Leon DA, Douglas IJ, Smeeth L. Association of BMI with overall and cause-specific mortality: a population-based cohort study of 3·6 million adults in the UK. Lancet Diabetes Endocrinol. 2018;6:944–53. [CrossRef]
- Ahmed SK, Mohammed RA. Obesity: Prevalence, causes, consequences, management, preventive strategies and future research directions. Metabolism Open. 2025;27:100375. [CrossRef]
- Ministry of Health, Health Promotion Board. National Population Health Survey 2022. Singapore; 2023.
- Ng M, Gakidou E, Lo J, Abate YH, Abbafati C, Abbas N, et al. Global, regional, and national prevalence of adult overweight and obesity, 1990–2021, with forecasts to 2050: a forecasting study for the Global Burden of Disease Study 2021. The Lancet. 2025;405:813–38. [CrossRef]
- Ryan DH, Yockey SR. Weight Loss and Improvement in Comorbidity: Differences at 5%, 10%, 15%, and Over. Curr Obes Rep. 2017;6:187–94. [CrossRef]
- Wing RR, Lang W, Wadden TA, Safford M, Knowler WC, Bertoni AG, et al. Benefits of Modest Weight Loss in Improving Cardiovascular Risk Factors in Overweight and Obese Individuals With Type 2 Diabetes. Diabetes Care. 2011;34:1481–6. [CrossRef]
- Johns DJ, Hartmann-Boyce J, Jebb SA, Aveyard P, Behavioural Weight Management Review Group. Diet or exercise interventions vs combined behavioural weight management programs: a systematic review and meta-analysis of direct comparisons. J Acad Nutr Diet. 2014;114:1557–68. [CrossRef]
- LeBlanc ES, Patnode CD, Webber EM, Redmond N, Rushkin M, O’Connor EA. Behavioural and Pharmacotherapy Weight Loss Interventions to Prevent Obesity-Related Morbidity and Mortality in Adults: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2018;320:1172–91. [CrossRef]
- Madigan CD, Graham HE, Sturgiss E, Kettle VE, Gokal K, Biddle G, et al. Effectiveness of weight management interventions for adults delivered in primary care: systematic review and meta-analysis of randomised controlled trials. BMJ. 2022;377:e069719. [CrossRef]
- Franz MJ, VanWormer JJ, Crain AL, Boucher JL, Histon T, Caplan W, et al. Weight-loss outcomes: a systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J Am Diet Assoc. 2007;107:1755–67. [CrossRef]
- Rotunda, W. Weight Loss in Short-Term Interventions for Physical Activity and Nutrition Among Adults With Overweight or Obesity: A Systematic Review and Meta-Analysis. Prev Chronic Dis. 2024;21. [CrossRef]
- Borek AJ, Abraham C, Greaves CJ, Tarrant M. Group-Based Diet and Physical Activity Weight-Loss Interventions: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Applied Psychology: Health and Well-Being. 2018;10:62–86. [CrossRef]
- Kupila SKE, Joki A, Suojanen L-U, Pietiläinen KH. The Effectiveness of eHealth Interventions for Weight Loss and Weight Loss Maintenance in Adults with Overweight or Obesity: A Systematic Review of Systematic Reviews. Curr Obes Rep. 2023;12:371–94. [CrossRef]
- Myers-Ingram R, Sampford J, Milton-Cole R, Jones GD. Effectiveness of eHealth weight management interventions in overweight and obese adults from low socioeconomic groups: a systematic review. Systematic Reviews. 2023;12:59. [CrossRef]
- Cavero-Redondo I, Martinez-Vizcaino V, Fernandez-Rodriguez R, Saz-Lara A, Pascual-Morena C, Álvarez-Bueno C. Effect of Behavioural Weight Management Interventions Using Lifestyle mHealth Self-Monitoring on Weight Loss: A Systematic Review and Meta-Analysis. Nutrients. 2020;12:1977. [CrossRef]
- Booth, M. Assessment of physical activity: an international perspective. Res Q Exerc Sport. 2000;71 Suppl 2:114–20. [CrossRef]
- Fat and fibre behaviour questionnaire: Reliability, relative validity and responsiveness to change in Australian adults with type 2 diabetes and/or hypertension - Reeves - 2015 - Nutrition & Dietetics - Wiley Online Library. https://onlinelibrary.wiley.com/doi/10.1111/1747-0080.12160. Accessed 30 Sep 2025.
- Caliendo M, Kopeinig S. Some practical guidance for the implementation of propensity score matching. Journal of Economic Surveys. 2008;22:31–72. [CrossRef]
- Kim, JH. Multicollinearity and misleading statistical results. Korean J Anesthesiol. 2019;72:558–69. [CrossRef]
- Alolayan RA, Aldisi DA, Hussain DS, Alafif N, Abulmeaty MMA. The Efficacy of Telehealth Versus In-Person Management Delivery in Adult Patients with Obesity. Healthcare (Basel). 2024;12:2190. [CrossRef]
- Cleo G, Glasziou P, Beller E, Isenring E, Thomas R. Habit-based interventions for weight loss maintenance in adults with overweight and obesity: a randomized controlled trial. Int J Obes. 2019;43:374–83. [CrossRef]
- Morgan PJ, Callister R, Collins CE, Plotnikoff RC, Young MD, Berry N, et al. The SHED-IT Community Trial: A Randomized Controlled Trial of Internet- and Paper-Based Weight Loss Programs Tailored for Overweight and Obese Men. Ann Behav Med. 2013;45:139–52. [CrossRef]
- Adebile TV, Adebile TM, Oloyede TF, Asifat OA, Biswas P, Sejoro S, et al. Telemedicine for obesity management among United States adults: A systematic and meta-analysis of intervention studies. J Telemed Telecare. 2025;31:1078–90. [CrossRef]
- Whitehall JM, Cook EJ, Vseteckova J, Jones K, Pappas Y, Donald L, et al. A systematic review of influences on engagement with remote health interventions targeting weight management for individuals living with excess weight. Int J Obes. 2025;49:1427–68. [CrossRef]
- Poupakis S, Kolotourou M, MacMillan HJ, Chadwick PM. Attendance, Weight Loss, and Participation in a Behavioural Diabetes Prevention Programme. Int J Behav Med. 2023;30:904–13. [CrossRef]
- Piernas C, MacLean F, Aveyard P, Ahern AL, Woolston J, Boyland EJ, et al. Greater Attendance at a Community Weight Loss Programme over the First 12 Weeks Predicts Weight Loss at 2 Years. Obes Facts. 2020;13:349–60. [CrossRef]
- Guarneiri LL, Kirkpatrick CF, Maki KC. Protein, fiber, and exercise: a narrative review of their roles in weight management and cardiometabolic health. Lipids Health Dis. 2025;24:237. [CrossRef]
- Kelly RK, Calhoun J, Hanus A, Payne-Foster P, Stout R, Sherman BW. Increased dietary fiber is associated with weight loss among Full Plate Living program participants. Front Nutr. 2023;10:1110748. [CrossRef]
- Howarth NC, Saltzman E, Roberts SB. Dietary fiber and weight regulation. Nutr Rev. 2001;59:129–39. [CrossRef]
- Ow Yong LM, Koe LWP. War on Diabetes in Singapore: a policy analysis. Health Research Policy and Systems. 2021;19:15. [CrossRef]
- Hu L, Illiano P, Pompeii ML, Popp CJ, Kharmats AY, Curran M, et al. Challenges of conducting a remote behavioural weight loss study: Lessons learned and a practical guide. Contemp Clin Trials. 2021;108:106522. [CrossRef]
| Baseline characteristics | Before matching | p-value | After matching | p-value | ||
|---|---|---|---|---|---|---|
| FitterLife | Control | FitterLife | Control | |||
| n=306 | n=5,087 | n=306 | n=306 | |||
| Age in years, mean ± SD | 47.8 ± 10.7 | 47.5± 11.9 | 0.676 | 47.8 ± 10.7 | 48.1 ± 11.6 | 0.805 |
| Gender, n (%) | <0.001 | 0.876 | ||||
| Male | 68 (22.2) | 2265 (44.5) | 68 (22.2) | 66 (21.6) | ||
| Female | 238 (77.8) | 2822 (55.5) | 238 (77.8) | 240 (78.4) | ||
| Chinese ethnicity, n (%) | 255 (83.3) | 3563 (70.0) | <0.001 | 255 (83.3) | 257 (83.9) | 0.853 |
| Weight in kg, mean ± SD | 73.6 ± 12.1 | 72.9 ± 12.2 | 0.325 | 73.6 ± 12.1 | 72.7 ± 12.0 | 0.125 |
| BMI in kg/m2, mean ± SD | 28.1 ± 3.6 | 27.2 ± 3.2 | <0.001 | 28.1 ± 3.6 | 28.0 ± 3.5 | 0.605 |
| Statistical methods | Outcome: Achieved weight loss target at week 12 (≥5% reduction in weight or ≥1 kg/m2 reduction in BMI) | ||||
|---|---|---|---|---|---|
| Control | FitterLife | P-value | Coefficient /Adjusted incidence rate ratio (ref: Control) | 95% confidence interval | |
| IPWRA (ATET) | 13.7% | 45.7% | <0.001 | 0.32 | 0.26, 0.38 |
| Modified Poisson regression on unmatched sample* | 644 (12.7%) | 140 (45.8%) | <0.001 | 3.32 | 2.85, 3.86 |
| Modified Poisson regression on matched sample* | 42 (13.6%) | 140 (45.8%) | <0.001 | 3.37 | 2.87, 3.93 |
| Behavioural factor | Outcome: Achieved weight loss target at week 12 (≥5% reduction in weight or ≥1 kg/m2 reduction in BMI) | ||||
|---|---|---|---|---|---|
| n (%) | p-value | Adjusted IRR (95% CI) | p-value | ||
| Sessions attended | 0.001 | 0.011 | |||
| 2 to 6 sessions (n=68) | 20 (29.4%) | Ref. | |||
| 7 to 9 sessions (n=222) | 115 (51.8%) | 1.63 (1.12, 2.37) | |||
| Change in fat intake score | 0.057 | ||||
| No change or increase (n=61) | 21 (34.4%) | Ref. | |||
| 0 to <20% decrease (n=166) | 79 (47.6%) | 1.46 (0.99, 2.16) | 0.057 | ||
| ≥20% decrease (n=63) | 35 (55.6%) | 1.66 (1.14, 2.42) | 0.041 | ||
| Change in fibre intake score | 0.001 | ||||
| No change or decrease (n=68) | 20 (29.4%) | Ref. | |||
| Increase (n=222) | 115 (51.8%) | 2.58 (1.30, 5.14) | 0.007 | ||
| Change in weekly MVPA | 0.013 | ||||
| No change or decrease (n=136) | 56 (41.2%) | Ref. | |||
| 0 to <1h increase (n=69) | 28 (41.6%) | 1.26(0.88, 1.81) | 0.202 | ||
| ≥1h increase (n=85) | 51 (60.0%) | 1.66 (1.24, 2.23) | 0.001 | ||
| Change in daily walking time | 0.079 | ||||
| No change or decrease (n=118) | 48 (40.7%) | Ref. | |||
| 0 to <16mins increase (n=79) | 35 (44.3%) | 1.03 (0.73, 1.47) | 0.859 | ||
| ≥16mins increase (n=93) | 52 (55.9%) | 1.77 (0.95, 1.70) | 0.104 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




