Figure 1.
Postnatal bone remodeling and healing. In contrast to prenatal bone formation, via an endochondral template (Anlage) that mineralizes over time or via direct intramembranous bone formation, remodeling and postnatal healing of bone depends on formation of space where new infilling can commence, enabling a reweaving of the bone without formation of mechanically inferior scar tissue. (A,B) Laser confocal imaging of bone remodeling in an adult long (cortical bone) demonstrates an osteoclastic cutting cone in the plane of focus (upper half, left to right), where osteoclasts cut through the dense cortex and osteoblasts (white arrows, B) are in the process of infilling the cone, from the outside inwards. Tissue neogenesis is evident, as the osteoblasts produce new bone matrix, layer by layer, filling inwards like tree rings (see infilled osteon in orthogonal plane, B); the unmineralized bone matrix fluoresces under laser light excitation (A). [6,7] (C) In a one stage bone transport surgical model of a critical sized defect of the femur [8,9], intramembranous bone formation starts nearly immediately in haematoma filled defects. This process is retarded if the defect is packed with cancellous bone graft, because the additional osteoclastic resorption step must take place before osteoblastic infilling can commence. (C, inset) Within three weeks, cambium cells from the periosteum migrate inwards (arrow) from the periosteum, toward the intramedullary nail that stabilizes the bone. First, a disorganized matrix is laid down (red), after which osteoblasts fill in layer by layer (first green, then blue), filling the defect within 16 weeks. [8,9,10] Scale bars indicate 50 microns.
Figure 1.
Postnatal bone remodeling and healing. In contrast to prenatal bone formation, via an endochondral template (Anlage) that mineralizes over time or via direct intramembranous bone formation, remodeling and postnatal healing of bone depends on formation of space where new infilling can commence, enabling a reweaving of the bone without formation of mechanically inferior scar tissue. (A,B) Laser confocal imaging of bone remodeling in an adult long (cortical bone) demonstrates an osteoclastic cutting cone in the plane of focus (upper half, left to right), where osteoclasts cut through the dense cortex and osteoblasts (white arrows, B) are in the process of infilling the cone, from the outside inwards. Tissue neogenesis is evident, as the osteoblasts produce new bone matrix, layer by layer, filling inwards like tree rings (see infilled osteon in orthogonal plane, B); the unmineralized bone matrix fluoresces under laser light excitation (A). [6,7] (C) In a one stage bone transport surgical model of a critical sized defect of the femur [8,9], intramembranous bone formation starts nearly immediately in haematoma filled defects. This process is retarded if the defect is packed with cancellous bone graft, because the additional osteoclastic resorption step must take place before osteoblastic infilling can commence. (C, inset) Within three weeks, cambium cells from the periosteum migrate inwards (arrow) from the periosteum, toward the intramedullary nail that stabilizes the bone. First, a disorganized matrix is laid down (red), after which osteoblasts fill in layer by layer (first green, then blue), filling the defect within 16 weeks. [8,9,10] Scale bars indicate 50 microns.
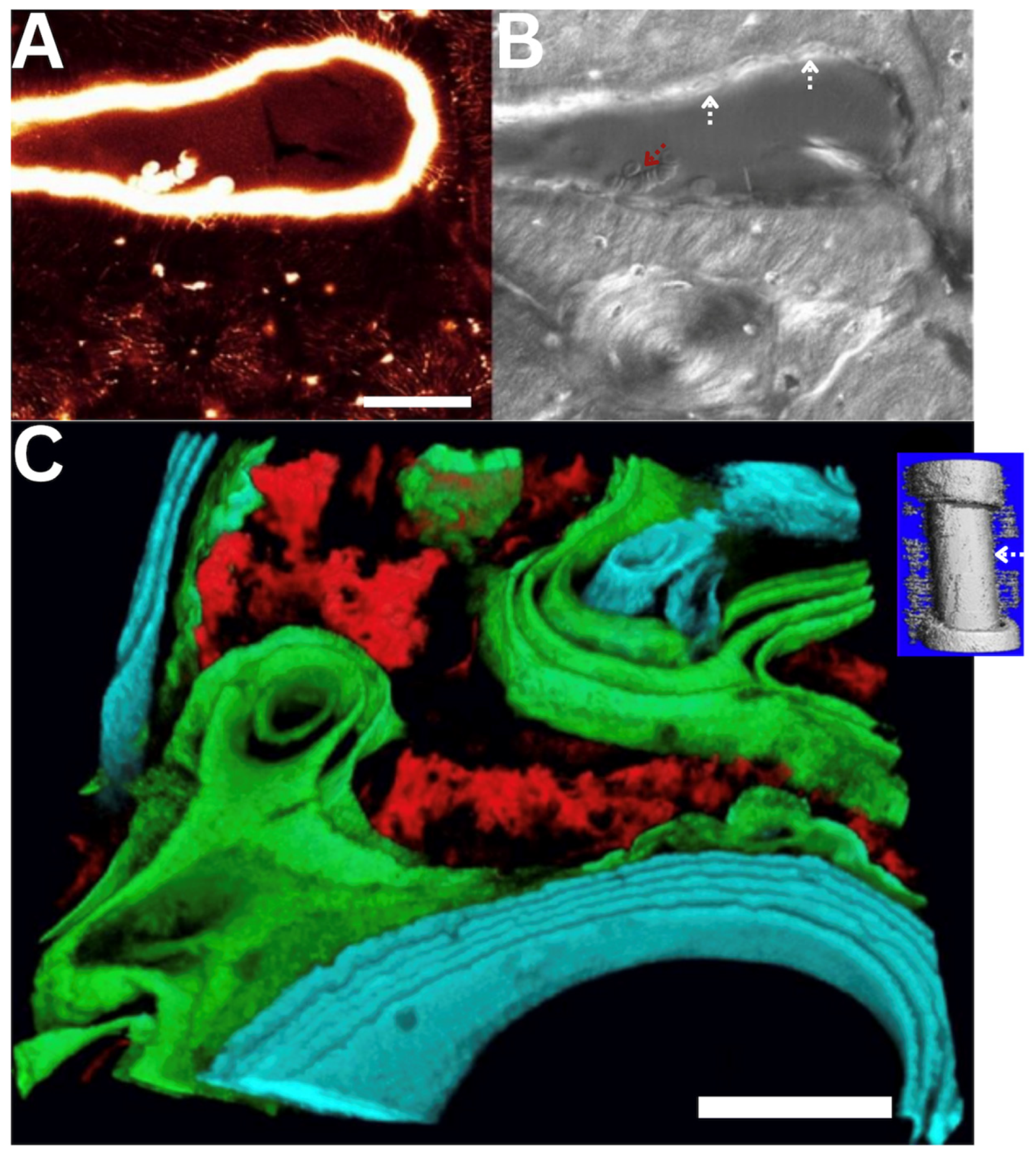
Figure 2.
At different physiological length and time scales of the life cycle, starting with human conception, followed by in utero development, birth and postnatal healing until end of life, work and energy can be considered from the outside—in and the inside—out, from system to organ to tissue to cell. Similarly, novel epidemiological approaches are being applied to predict emergence of disease at earliest stages, in cellular populations within the ecosystems of the organs and tissues making up e.g., skeletal to brain-nervous systems. These interdisciplinary approaches cross length and time scales seamlessly to predict emergent behavior and to probe tissue as a physical rendering of cellular experience throughout the life cycle. [1] (A) Figure adapted from collaboration with Dr. Joanna Ng [11], at University of New South Wales. Epidemiology of cellular health in the osteoarthritic hip of human patients, in collaboration with Dr. Dirk Zeidler at Zeiss Microscopy, Oberkochen, Germany [12,13,14].
Figure 2.
At different physiological length and time scales of the life cycle, starting with human conception, followed by in utero development, birth and postnatal healing until end of life, work and energy can be considered from the outside—in and the inside—out, from system to organ to tissue to cell. Similarly, novel epidemiological approaches are being applied to predict emergence of disease at earliest stages, in cellular populations within the ecosystems of the organs and tissues making up e.g., skeletal to brain-nervous systems. These interdisciplinary approaches cross length and time scales seamlessly to predict emergent behavior and to probe tissue as a physical rendering of cellular experience throughout the life cycle. [1] (A) Figure adapted from collaboration with Dr. Joanna Ng [11], at University of New South Wales. Epidemiology of cellular health in the osteoarthritic hip of human patients, in collaboration with Dr. Dirk Zeidler at Zeiss Microscopy, Oberkochen, Germany [12,13,14].
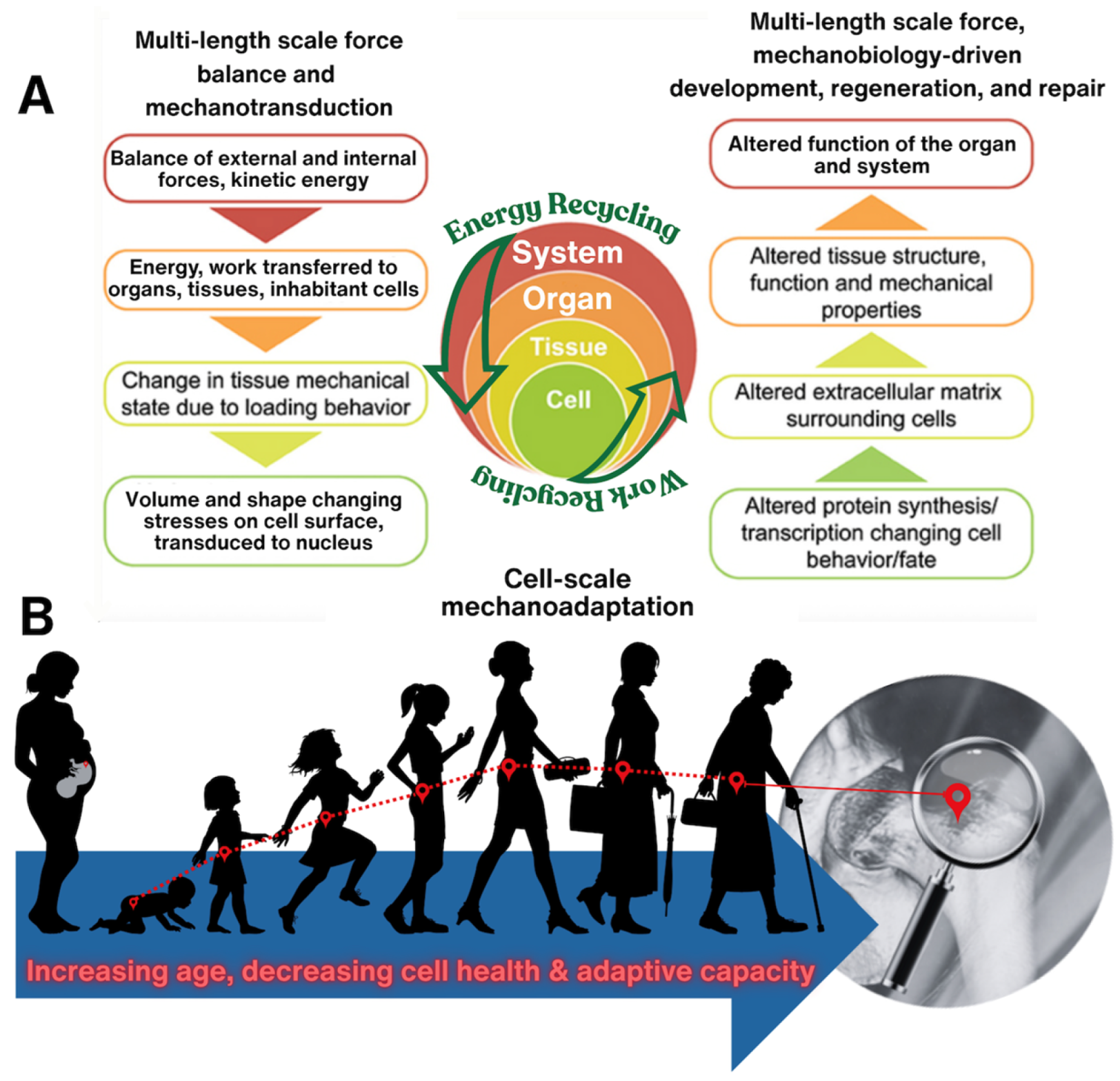
Figure 3.
Tying together mechanical loading effects on tissue deformation to load-induced fluid flow through tissues. (A) Through surgical placement of strain gauges, e.g., on the anterior aspect of the sheep metacarpus, in vivo measurements of strains (D1-D3) induced during normal gait can be measured with minimal confounding factors (in collaboration with Dr. Roland Steck). (B,C) Ex vivo perfusion models enable close replication of in vivo loading condition while allowing for control of perfusion rates and post hoc monitoring of tracer transport at organ to subcellular length scales. Such intertwined approaches have been applied for human transplant medicine as well as to elucidate mechanotransduction and load-induced transport (convection) across length scales. (B) In a compression model designed to study mechanotransduction and transport in the middiaphysis of the metacarpus (compact bone), loading is applied via Schanz screws inserted through the proximal and distal entheses of the metacarpus while perfusion is controlled using microsurgical preparation of a closed loop perfusion system (in collaboration with Dr. Ulf Knothe). (C) In a compression model designed to study mechanotransduction and transport through the synovial joint (radius—metacarpus), load is applied mimicking physiological conditions, via the radius, with the hoof fixed in place. Perfusion is controlled in a manner analogous to the previous preparation (B). (D) The in vivo model provides “near real world” measures of strains (principal strain [D1] and directions [D2] over the gait cycle (time). In general, compressive strain increaases with increasing body mass and rate of walking (D3, negative strain indicative of compression). All experiments depicted in this figure were conducted at the AO Research Institute in Davos, Switzerland, with IACUC approvals under the guidance of the Canton of Grisons, 1989–2009.
Figure 3.
Tying together mechanical loading effects on tissue deformation to load-induced fluid flow through tissues. (A) Through surgical placement of strain gauges, e.g., on the anterior aspect of the sheep metacarpus, in vivo measurements of strains (D1-D3) induced during normal gait can be measured with minimal confounding factors (in collaboration with Dr. Roland Steck). (B,C) Ex vivo perfusion models enable close replication of in vivo loading condition while allowing for control of perfusion rates and post hoc monitoring of tracer transport at organ to subcellular length scales. Such intertwined approaches have been applied for human transplant medicine as well as to elucidate mechanotransduction and load-induced transport (convection) across length scales. (B) In a compression model designed to study mechanotransduction and transport in the middiaphysis of the metacarpus (compact bone), loading is applied via Schanz screws inserted through the proximal and distal entheses of the metacarpus while perfusion is controlled using microsurgical preparation of a closed loop perfusion system (in collaboration with Dr. Ulf Knothe). (C) In a compression model designed to study mechanotransduction and transport through the synovial joint (radius—metacarpus), load is applied mimicking physiological conditions, via the radius, with the hoof fixed in place. Perfusion is controlled in a manner analogous to the previous preparation (B). (D) The in vivo model provides “near real world” measures of strains (principal strain [D1] and directions [D2] over the gait cycle (time). In general, compressive strain increaases with increasing body mass and rate of walking (D3, negative strain indicative of compression). All experiments depicted in this figure were conducted at the AO Research Institute in Davos, Switzerland, with IACUC approvals under the guidance of the Canton of Grisons, 1989–2009.
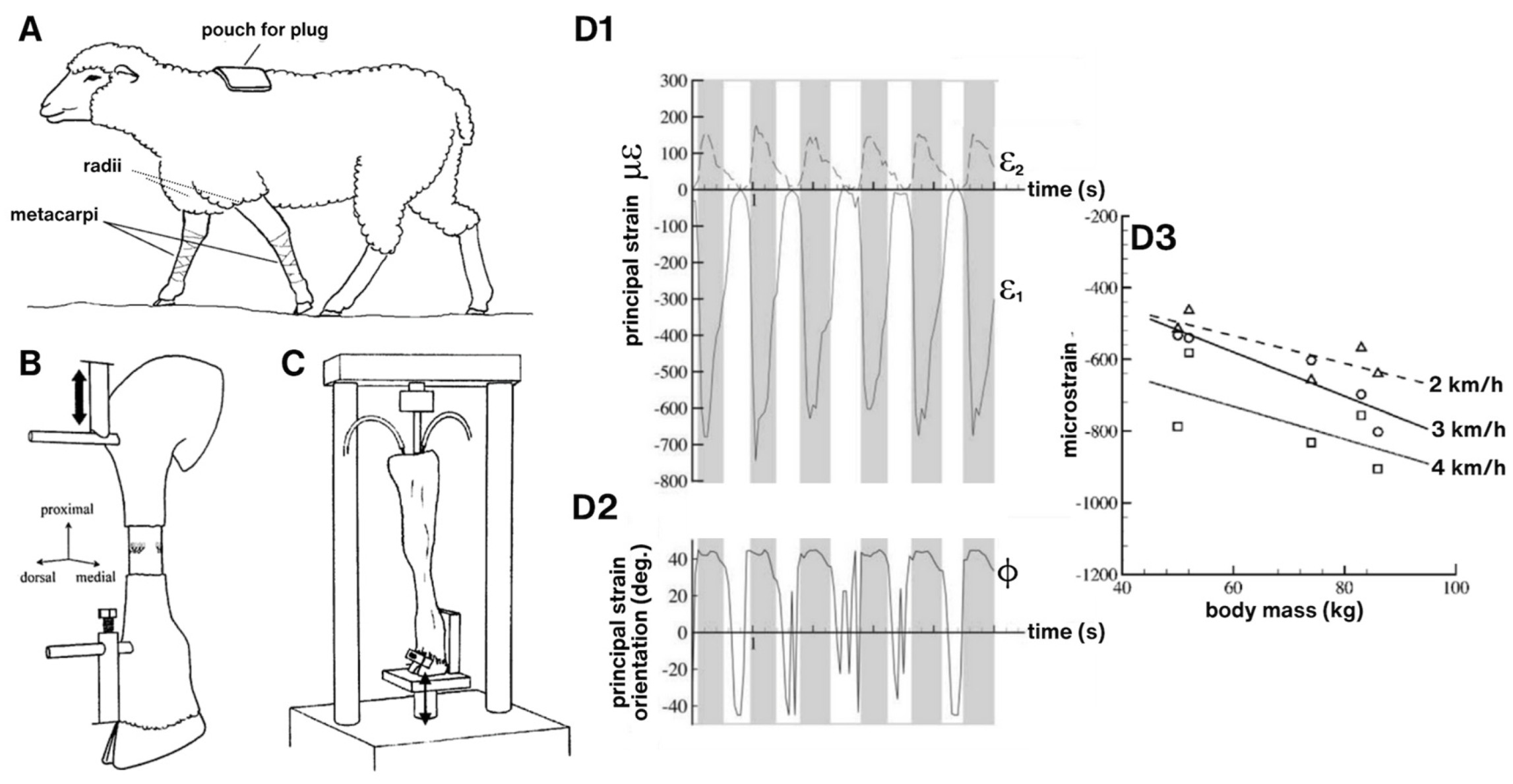
Figure 4.
Implant design and implementation with microscope imaging in situ of cell-scale remodeling within a threaded ring titanium implant on the surface of the sheep metacpus. (A) Finite Element (FE) modeling prediction of loading within the ring shows a reduction in strain in tissues offloaded by the titanium implant, i.e., within the ring. Bone surface strains are displayed for the entire model of the metacarpus without and with the implant, for the region of interest, and for a cut through the metacarpus at the location of the implant (top). Physiological loading induces compressive strains on the dorsal surface of the metacarpus in physiological loading conditions. Maximal compressive strains (dark blue) up to approximately 900 microstrain (0.09% strain) are found near the distal diaphysis. At the region of interest, the surface strains are reduced from 500-600 microstrain to about 200-400 microstrain, due to the presence of the implant. (B-C) Correlative imaging in reflection and using laser scanning confocal microscopy enabled concomitant imaging of bone resorption and local perfusion of a fluorescent tracer. (B) Collage of histological micrographs showing resorption spaces (black arrows) and blood vessels (appear as vertically orientation striations, referred to as Haversian canals, indicated by arrowheads) in tissue within the implant ring. (C1) Raw (no post-processing or other image modification/enhancement) confocal image taken in vivo and in situ. Red blood cells moving through capillaries (double arrows) as well as individual osteocytes and their nuclei (single arrows) are observable. (C2) Osteocyte viewed in situ (left), and (right) magnified 100% to visualize the nucleus and pericellular domain. (C3) Osteoclastic cutting cone in vivo and in situ outlined for visual clarity. This IACUC approved study was carried out at the Cleveland Clinic Lerner Research Institute and involved moving an anaesthetized sheep into the imaging facility, for in vivo imaging, which was unprecedented at the time.
Figure 4.
Implant design and implementation with microscope imaging in situ of cell-scale remodeling within a threaded ring titanium implant on the surface of the sheep metacpus. (A) Finite Element (FE) modeling prediction of loading within the ring shows a reduction in strain in tissues offloaded by the titanium implant, i.e., within the ring. Bone surface strains are displayed for the entire model of the metacarpus without and with the implant, for the region of interest, and for a cut through the metacarpus at the location of the implant (top). Physiological loading induces compressive strains on the dorsal surface of the metacarpus in physiological loading conditions. Maximal compressive strains (dark blue) up to approximately 900 microstrain (0.09% strain) are found near the distal diaphysis. At the region of interest, the surface strains are reduced from 500-600 microstrain to about 200-400 microstrain, due to the presence of the implant. (B-C) Correlative imaging in reflection and using laser scanning confocal microscopy enabled concomitant imaging of bone resorption and local perfusion of a fluorescent tracer. (B) Collage of histological micrographs showing resorption spaces (black arrows) and blood vessels (appear as vertically orientation striations, referred to as Haversian canals, indicated by arrowheads) in tissue within the implant ring. (C1) Raw (no post-processing or other image modification/enhancement) confocal image taken in vivo and in situ. Red blood cells moving through capillaries (double arrows) as well as individual osteocytes and their nuclei (single arrows) are observable. (C2) Osteocyte viewed in situ (left), and (right) magnified 100% to visualize the nucleus and pericellular domain. (C3) Osteoclastic cutting cone in vivo and in situ outlined for visual clarity. This IACUC approved study was carried out at the Cleveland Clinic Lerner Research Institute and involved moving an anaesthetized sheep into the imaging facility, for in vivo imaging, which was unprecedented at the time.
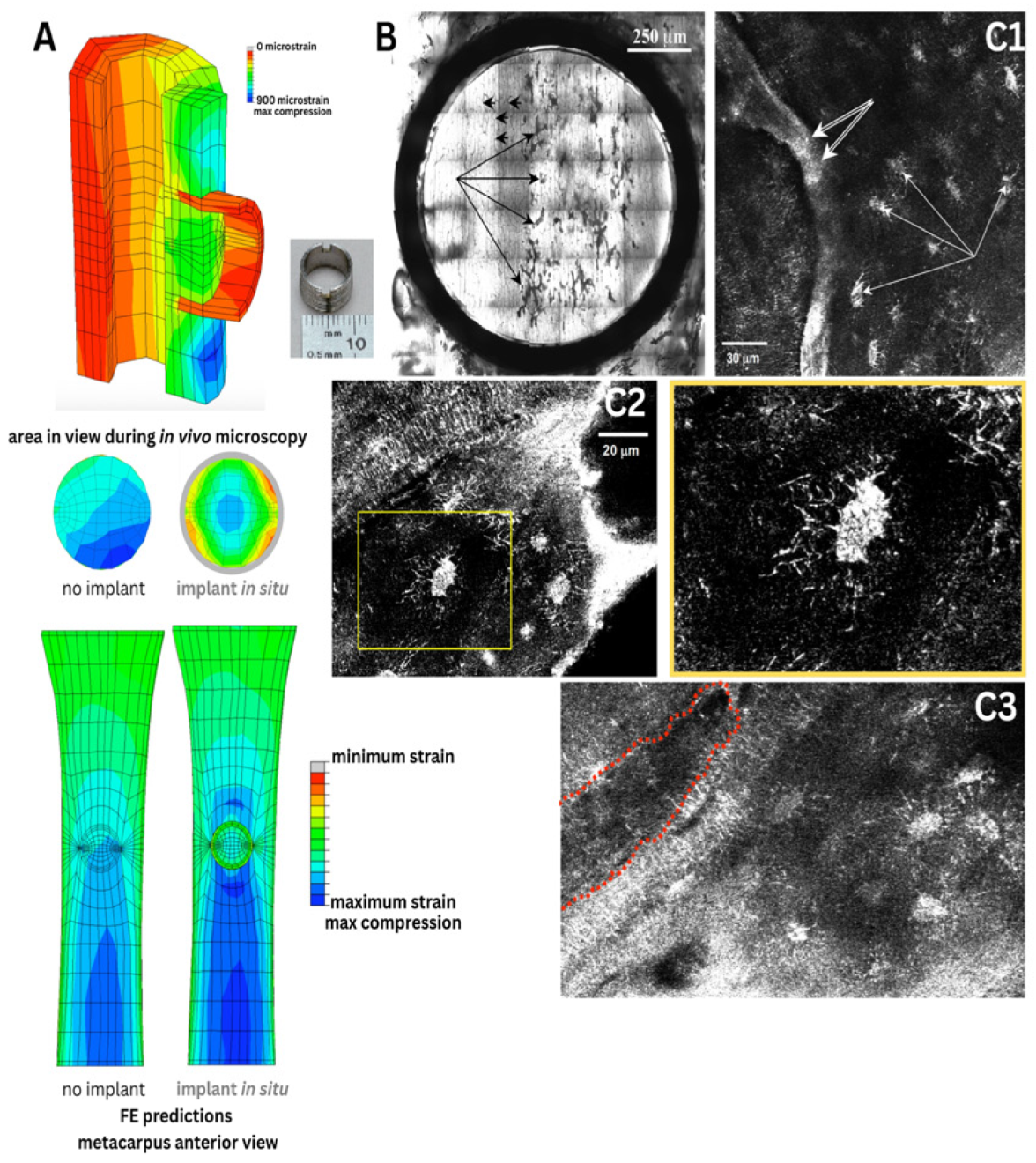
Figure 5.
(A) We used novel physical and in silico models to understand nano-microscale flow fields around osteocytes in situ. Based on high resolution confocal image stacks (volumes) of osteocyte networks from healthy, osteoporotic and osteoarthritic human bone biopsies (A2), we 3D-printed inverse physical models, where the osteocyte network created voids in the resin blocks (A1) which were scaled up by 1000x. Using similitude theory, we could then measure the permeability of the blocks with a fluid of viscosity 1000x that of bone fluid. We predicted signal transmission efficiency through the respective cellular networks using stochastic models (A3,B) and virtual parametric models of cells and their networks (A4), where we could probe the respective effects of live cells in situ as well as pericellular matrix on signaling efficiency around cells. (A5) Using nano-microfluidics computational models, we could import actual 3D geometries from electron microscopy to simulate and predict flow around osteocytes, showing that natural constrictions in pericellular spaces would be predicted to amplify drag on osteocytes, like narrowings in a stream. (B) These could then be tied to in situ measures of cell viability in preserved slices of bone (B1), where effects of cell death on signal transmission efficiency could be predicted (B2,B3). (A-B) In collaboration with Professor Eric J. Anderson [25,26]. (C) State-of-the-art episcopic correlative imaging studies enable cell to organ scale high resolution imaging of tracer transport from the heart to cells of the different tissues comprising the synovial joint, in healthy and osteoarthritic animals, and subjected to spikes in cytokines delivered as a bolus to the heart. This work was carried out in collaboration with Dr. Lucy Ngo [27,28,29].
Figure 5.
(A) We used novel physical and in silico models to understand nano-microscale flow fields around osteocytes in situ. Based on high resolution confocal image stacks (volumes) of osteocyte networks from healthy, osteoporotic and osteoarthritic human bone biopsies (A2), we 3D-printed inverse physical models, where the osteocyte network created voids in the resin blocks (A1) which were scaled up by 1000x. Using similitude theory, we could then measure the permeability of the blocks with a fluid of viscosity 1000x that of bone fluid. We predicted signal transmission efficiency through the respective cellular networks using stochastic models (A3,B) and virtual parametric models of cells and their networks (A4), where we could probe the respective effects of live cells in situ as well as pericellular matrix on signaling efficiency around cells. (A5) Using nano-microfluidics computational models, we could import actual 3D geometries from electron microscopy to simulate and predict flow around osteocytes, showing that natural constrictions in pericellular spaces would be predicted to amplify drag on osteocytes, like narrowings in a stream. (B) These could then be tied to in situ measures of cell viability in preserved slices of bone (B1), where effects of cell death on signal transmission efficiency could be predicted (B2,B3). (A-B) In collaboration with Professor Eric J. Anderson [25,26]. (C) State-of-the-art episcopic correlative imaging studies enable cell to organ scale high resolution imaging of tracer transport from the heart to cells of the different tissues comprising the synovial joint, in healthy and osteoarthritic animals, and subjected to spikes in cytokines delivered as a bolus to the heart. This work was carried out in collaboration with Dr. Lucy Ngo [27,28,29].
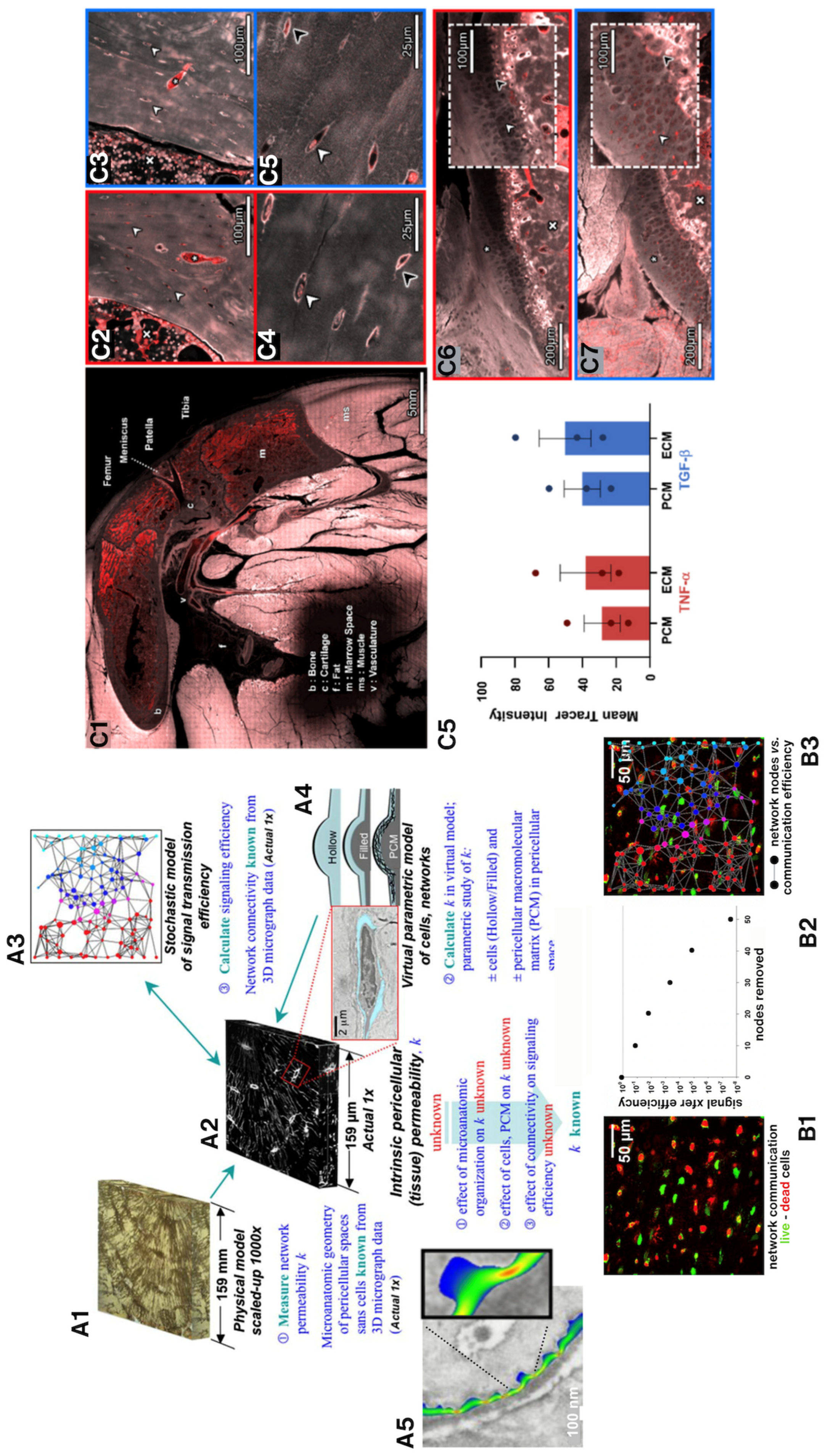
Figure 6.
To understand the role of biophysical factors in the surprising rate and volume of new osteogenesis observed in the one-stage bone transport procedure (developed by Dr. U. Knothe), we needed to elucidate of the mechanical milieux of mesenchymal stem cells residing in the periosteum via ex vivo (A,B—in collaboration with Professor Sara McBride-Gagy [30]), in vitro (C—in collaboration with Dr. Nicole Yu and Dr. Renee Whan [35]; D—in collaboration with Professor Sara McBride-Gagy [31]), and in silico (not shown—in collaboration with Dr. Shannon Moore [34]) models. (A) After surgical preparation for the one-stage bone transport procedure, the femur was resected and placed in a loading rig to mimic stance shift loading in vivo, the condition under which in vivo studies demonstrated robust intramembranous infilling of the defect within 16 weeks of surgery. To measure cell-scale microstrains at high resolution during stance shift loading, we videotaped the surface of the periosteum from different angles using ultra-high-definition television lenses (A, upper left) and (B) carried out digital image correlation (DIC) to calculate strains in the vicinity of quiescent stem cells during loading. Statistical analysis of strains to osteogenesis patterns revealed that change in baseline strains showed greater correlation to tissue genesis than strain magnitudes per se or particular thresholds [30]. (C) To determine whether periosteal lifting, which attaches the periosteum in a pre-stressed state to the surfaces of bone via collagenous Sharpey’s fibers, we carried out live imaging protocols on ex vivo periosteal preparations to measure the change in volume and shape of periosteal cells with (C1) and without (C3) pre-stress in situ. Imaging showed that cutting of Sharpey’s fibers to lift the periosteum increases crimping of the collagen to which periosteal cells attach, and subsequent rounding of quiescent stem cell nuclei; hence, cutting of the Sharpey’ fibers acts as a mechanical trigger for the cells to migrate to sites of injury and initiate tissue genesis [35]. (D) Mechanical testing and shrinkage (not shown) studies of ovine periosteum enable measurement of periosteum’s anisotropy and prestress [31,32,33]. Experiments depicted in this figure were conducted at the AO Research Institute in Davos, Switzerland (A,B), the Biomedical Imaging Facility of the University of New South Wales (C) and the Experimental and Computational Mechanobiology Labs of Case Western Reserve University (D), with IACUC approvals under the guidance of the relevant local committees.
Figure 6.
To understand the role of biophysical factors in the surprising rate and volume of new osteogenesis observed in the one-stage bone transport procedure (developed by Dr. U. Knothe), we needed to elucidate of the mechanical milieux of mesenchymal stem cells residing in the periosteum via ex vivo (A,B—in collaboration with Professor Sara McBride-Gagy [30]), in vitro (C—in collaboration with Dr. Nicole Yu and Dr. Renee Whan [35]; D—in collaboration with Professor Sara McBride-Gagy [31]), and in silico (not shown—in collaboration with Dr. Shannon Moore [34]) models. (A) After surgical preparation for the one-stage bone transport procedure, the femur was resected and placed in a loading rig to mimic stance shift loading in vivo, the condition under which in vivo studies demonstrated robust intramembranous infilling of the defect within 16 weeks of surgery. To measure cell-scale microstrains at high resolution during stance shift loading, we videotaped the surface of the periosteum from different angles using ultra-high-definition television lenses (A, upper left) and (B) carried out digital image correlation (DIC) to calculate strains in the vicinity of quiescent stem cells during loading. Statistical analysis of strains to osteogenesis patterns revealed that change in baseline strains showed greater correlation to tissue genesis than strain magnitudes per se or particular thresholds [30]. (C) To determine whether periosteal lifting, which attaches the periosteum in a pre-stressed state to the surfaces of bone via collagenous Sharpey’s fibers, we carried out live imaging protocols on ex vivo periosteal preparations to measure the change in volume and shape of periosteal cells with (C1) and without (C3) pre-stress in situ. Imaging showed that cutting of Sharpey’s fibers to lift the periosteum increases crimping of the collagen to which periosteal cells attach, and subsequent rounding of quiescent stem cell nuclei; hence, cutting of the Sharpey’ fibers acts as a mechanical trigger for the cells to migrate to sites of injury and initiate tissue genesis [35]. (D) Mechanical testing and shrinkage (not shown) studies of ovine periosteum enable measurement of periosteum’s anisotropy and prestress [31,32,33]. Experiments depicted in this figure were conducted at the AO Research Institute in Davos, Switzerland (A,B), the Biomedical Imaging Facility of the University of New South Wales (C) and the Experimental and Computational Mechanobiology Labs of Case Western Reserve University (D), with IACUC approvals under the guidance of the relevant local committees.
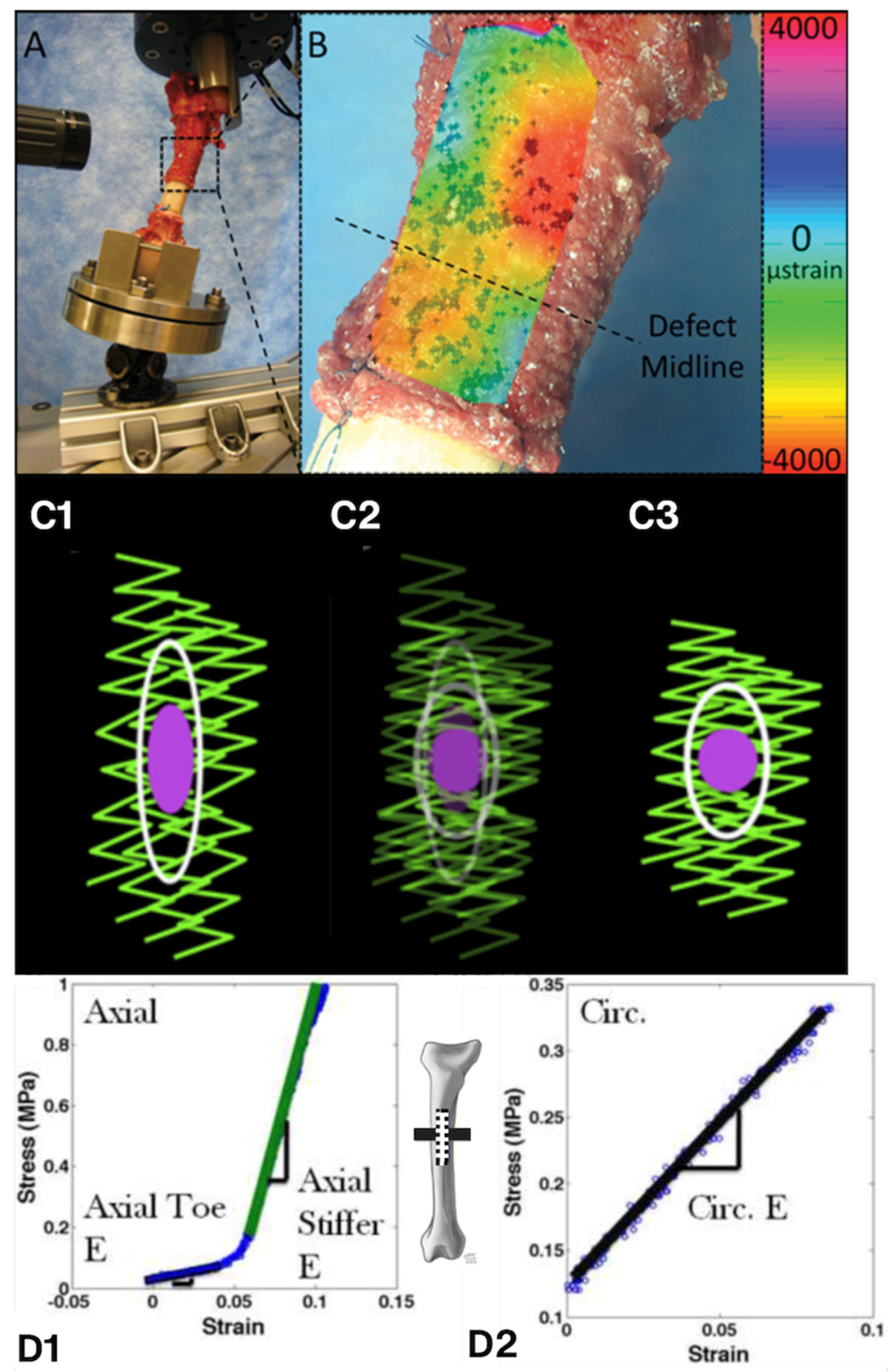
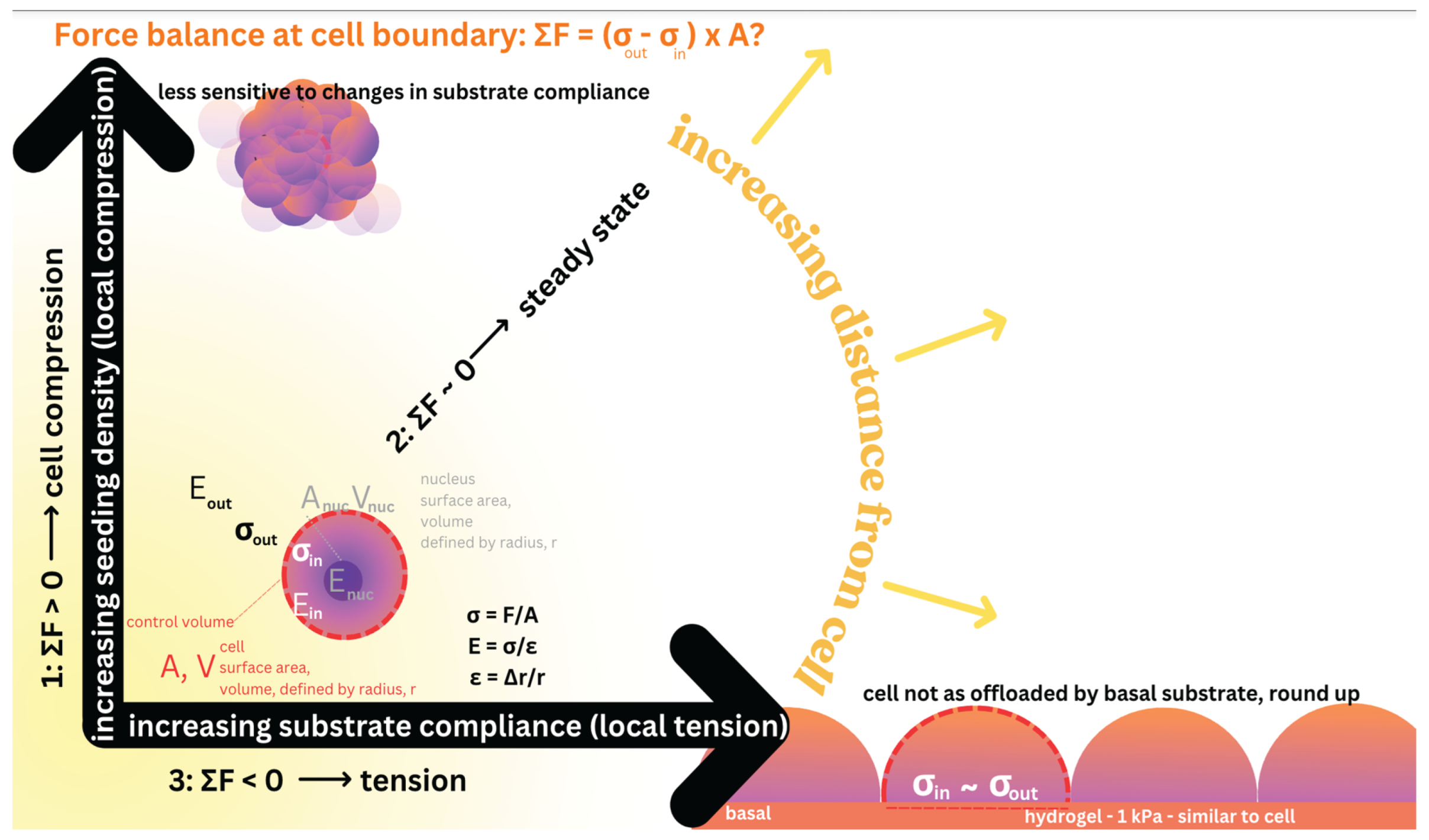
Figure 7.
At cell boundaries force balances modulate stem cells’ mechanoadaptation to their dynamic, local mechanical environment. The force balance at the cell boundary is defined by the control volume (indicated by red dashed circle, with A—Area, and V—volume, and surface area of the cell) delineating the border between the outside of the cell (matrix and/or substrate in cultured cells, “out”), including the nucleus. The stress (σ) and strain (ε) relationships are delineated by theory of elasticity and plasticity, and theory of virtual work [2]. If the force balance (ΣF) is greater than zero (1), the cell experiences local compression. If the force balance (ΣF) is around zero (2), the cell is at steady state. If the balance (ΣF) is less than zero (3), the cell experiences local tension. Animated version of figure [36]: https://figshare.com/articles/media/Animation_1/27936261.
Figure 7.
At cell boundaries force balances modulate stem cells’ mechanoadaptation to their dynamic, local mechanical environment. The force balance at the cell boundary is defined by the control volume (indicated by red dashed circle, with A—Area, and V—volume, and surface area of the cell) delineating the border between the outside of the cell (matrix and/or substrate in cultured cells, “out”), including the nucleus. The stress (σ) and strain (ε) relationships are delineated by theory of elasticity and plasticity, and theory of virtual work [2]. If the force balance (ΣF) is greater than zero (1), the cell experiences local compression. If the force balance (ΣF) is around zero (2), the cell is at steady state. If the balance (ΣF) is less than zero (3), the cell experiences local tension. Animated version of figure [36]: https://figshare.com/articles/media/Animation_1/27936261.