Submitted:
04 November 2025
Posted:
06 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
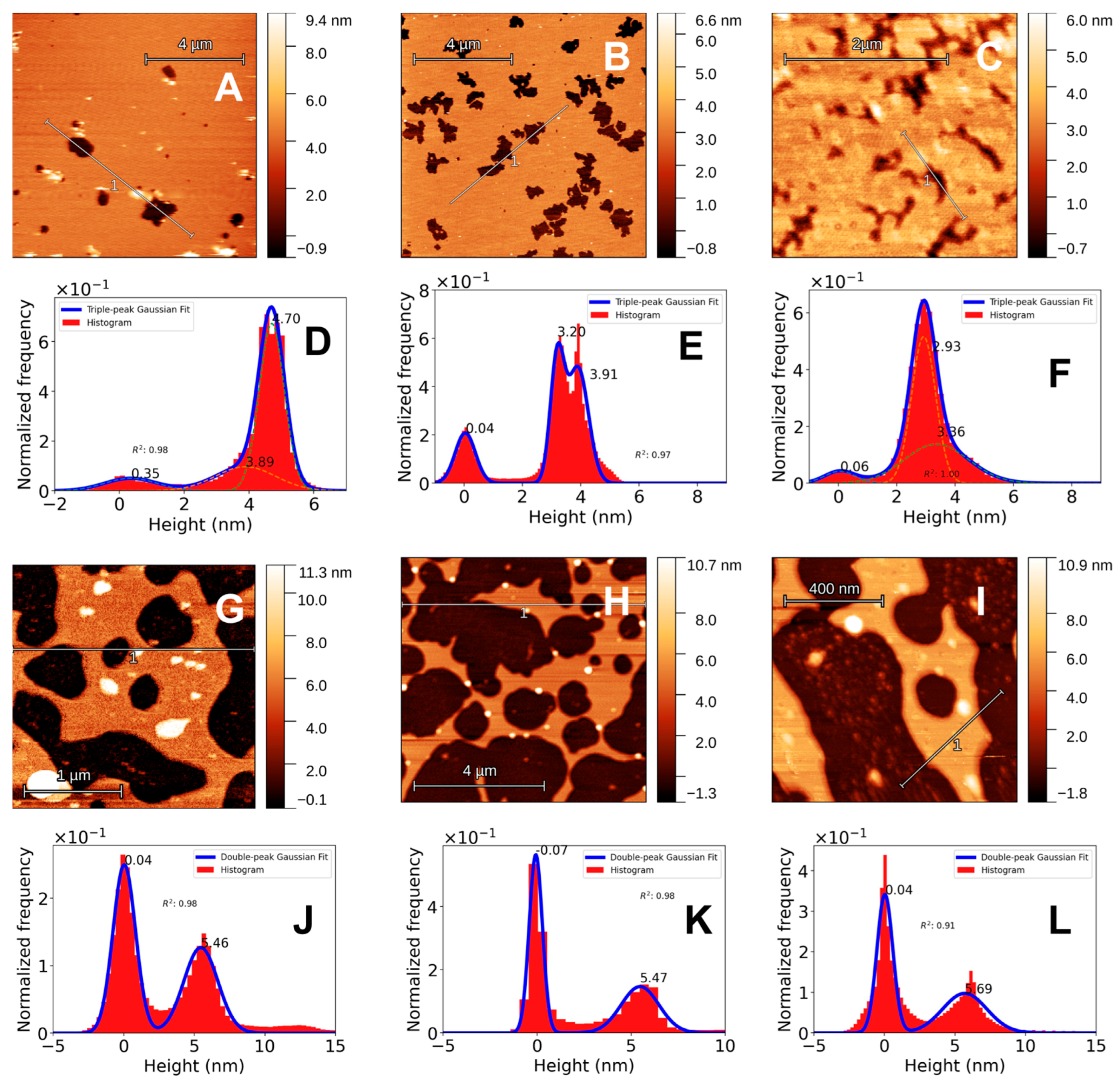
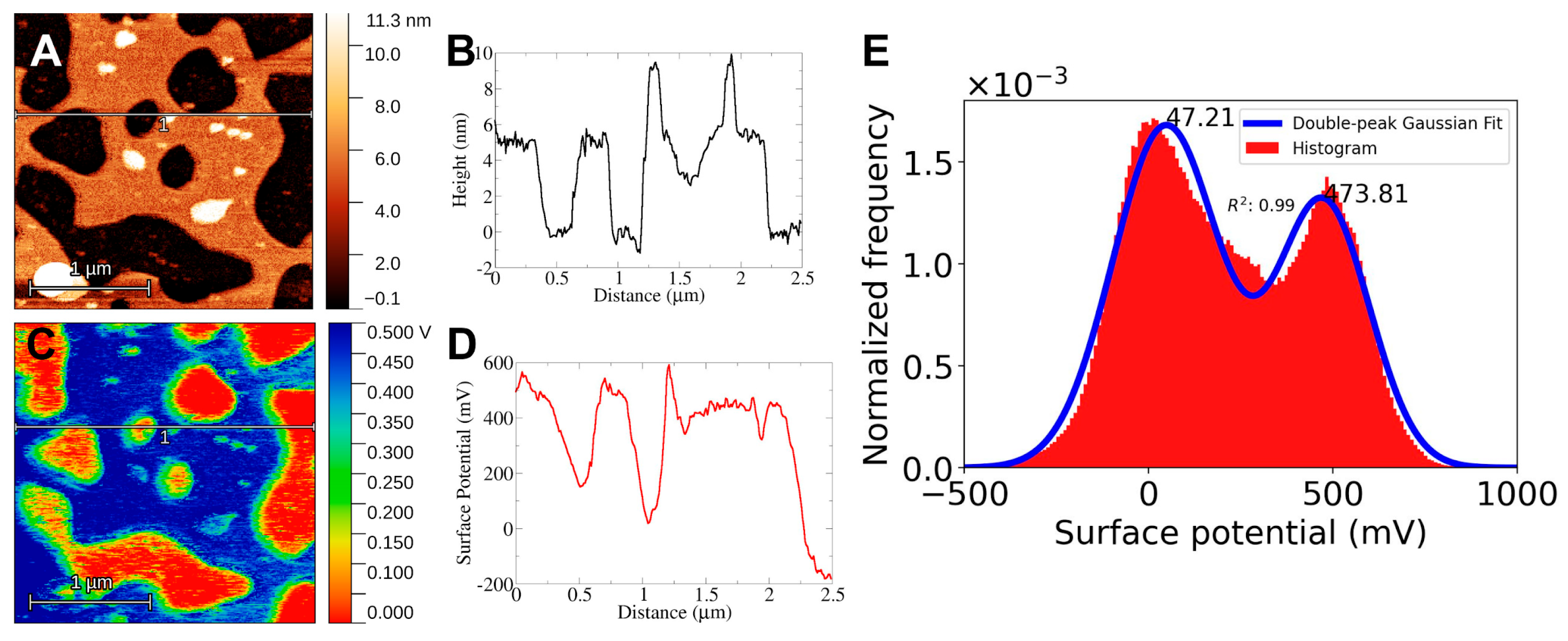
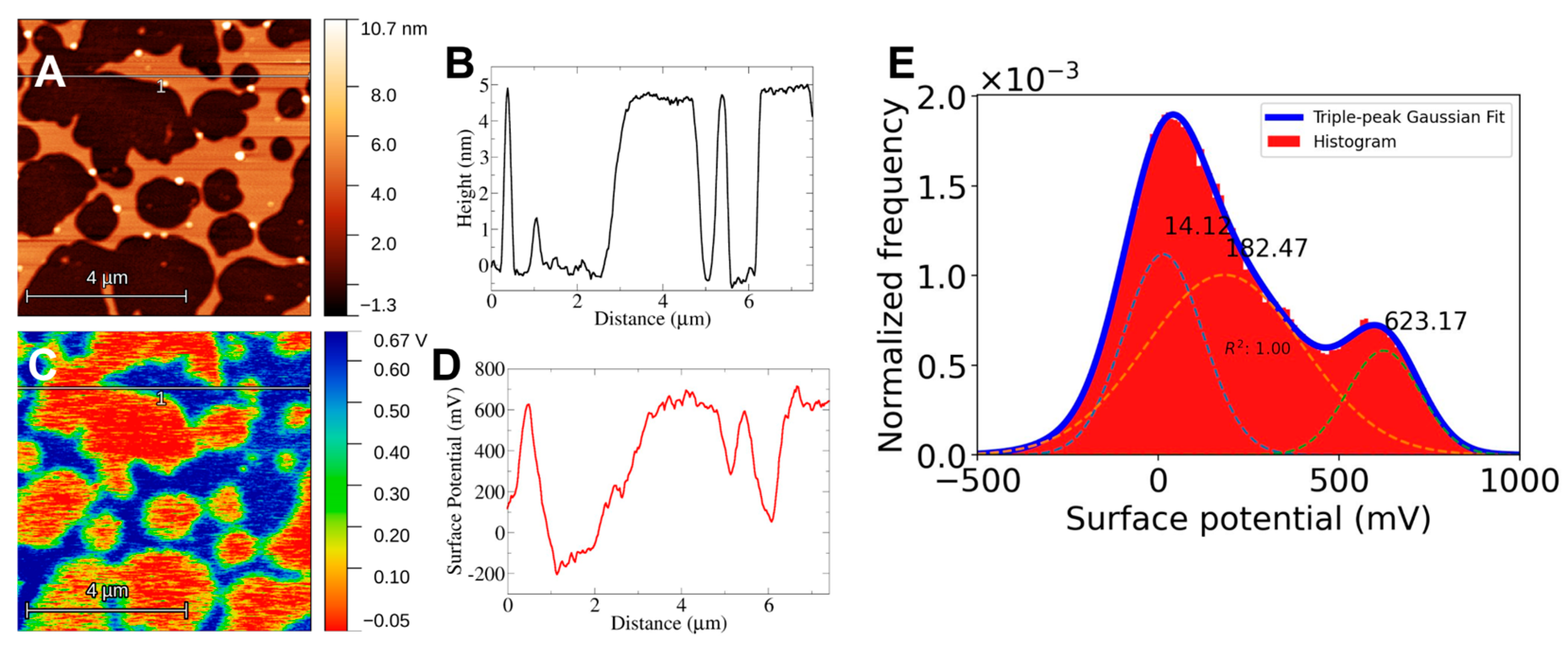
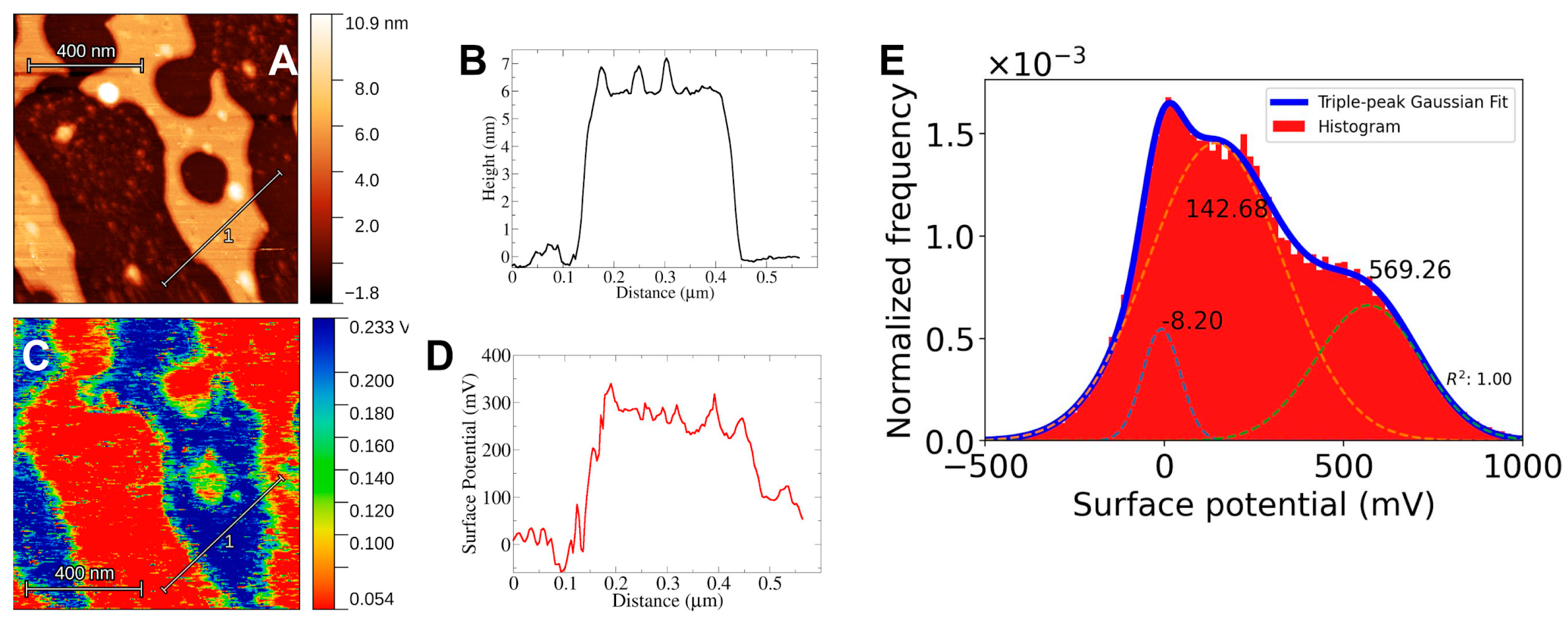
3. Results
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | Atomic Force Microscopy |
| KPFM | Kelvin Probe Force Microscopy |
| SLB | Supported Lipid Bilayer |
| F-SLB | Freeze-dried Supported Lipid Bilayer |
| DPPC | 1,2-dipalmitoyl-sn-glycero-3-phosphocholine |
| Chol | Cholesterol |
| Ergosterol | Ergosterol |
References
- Fang, R.H.; Gao, W.; Zhang, L. Targeting drugs to tumours using cell membrane-coated nanoparticles. Nat Rev Clin Oncol. 2023, 20, 33–48. [Google Scholar] [CrossRef] [PubMed]
- Zalba, S.; Ten Hagen, T.L.M. Cell membrane modulation as adjuvant in cancer therapy. Cancer Treat Rev. 2017, 52, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Prinz, W.A.; Toulmay, A.; Balla, T. The functional universe of membrane contact sites. Nat Rev Mol Cell Biol. 2020, 21, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Martinac, B.; Nikolaev, Y.A.; Silvani, G.; Bavi, N.; Romanov, V.; Nakayama, Y.; et al. Cell membrane mechanics and mechanosensory transduction. Curr Top Membr. 2020, 86, 83–141. [Google Scholar] [CrossRef]
- Schaefer, K.G.; Pittman, A.E.; Barrera, F.N.; King, G.M. Atomic force microscopy for quantitative understanding of peptide-induced lipid bilayer remodeling. Methods. 2022, 197, 20–29. [Google Scholar] [CrossRef]
- Endo, M. Surface Assembly of DNA Origami on a Lipid Bilayer Observed Using High-Speed Atomic Force Microscopy. Molecules. 2022, 27. [Google Scholar] [CrossRef]
- Henderson, R.D.E.; Filice, C.T.; Wettig, S.; Leonenko, Z. Kelvin probe force microscopy to study electrostatic interactions of DNA with lipid-gemini surfactant monolayers for gene delivery. Soft Matter. 2021, 17, 826–833. [Google Scholar] [CrossRef]
- Gumí-Audenis, B.; Costa, L.; Carlá, F.; Comin, F.; Sanz, F.; Giannotti, M.I. Structure and Nanomechanics of Model Membranes by Atomic Force Microscopy and Spectroscopy: Insights into the Role of Cholesterol and Sphingolipids. Membranes 2016, 6. [Google Scholar] [CrossRef]
- Franz, C.M.; Puech, P.-H. Atomic Force Microscopy: A Versatile Tool for Studying Cell Morphology, Adhesion and Mechanics. Cellular and Molecular Bioengineering. 2008, 1, 289–300. [Google Scholar] [CrossRef]
- Wilhelm, M.; Shen, J.; Kummel, A.C.; Lee, S. Kelvin probe force microscopy and its application. Surface Science Reports. 2011, 66, 1–27. [Google Scholar] [CrossRef]
- Collins, L.; Jesse, S.; Kilpatrick, J.I.; Tselev, A.; Okatan, M.B.; Kalinin, S.V.; et al. Kelvin probe force microscopy in liquid using electrochemical force microscopy. Beilstein J Nanotechnol. 2015, 6, 201–214. [Google Scholar] [CrossRef]
- Kilpatrick, J.I.; Kargin, E.; Rodriguez, B.J. Comparing the performance of single and multifrequency Kelvin probe force microscopy techniques in air and water. Beilstein J Nanotechnol. 2022, 13, 922–943. [Google Scholar] [CrossRef]
- Salerno, M.; Dante, S. Scanning Kelvin Probe Microscopy: Challenges and Perspectives towards Increased Application on Biomaterials and Biological Samples. Materials. 2018, 11, 951. [Google Scholar] [CrossRef]
- Klausen, L.H.; Fuhs, T.; Dong, M. Mapping surface charge density of lipid bilayers by quantitative surface conductivity microscopy. Nature Communications. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Povilaitis, S.C.; Hector, J.A.; Mantsch, M.E.; Webb, L.J. Direct Measurement of the Effect of Cholesterol and 7-Dehydrocholesterol on Membrane Dipole Electric Field in Single and Mixed Sterol Vesicles Using Vibrational Stark Effect Spectroscopy. The Journal of Physical Chemistry B. 2025. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Tao, Y.; Zou, W.; Chen, X.; Chen, X.; Freindorf, M.; et al. A Critical Evaluation of Vibrational Stark Effect (VSE) Probes with the Local Vibrational Mode Theory. Sensors 2020, 20. [Google Scholar] [CrossRef]
- Nanoscale Electrostatic Domains in Cholesterol-Laden Lipid Membranes Create a Target for Amyloid Binding. Biophysical Journal. 2012, 103, L27–L29. [CrossRef] [PubMed]
- Schafer, E.A.; Davis, E.; Manzer, Z.; Daniel, S.; Rivnay, J. Hybrid Supported Lipid Bilayers for Bioinspired Bioelectronics with Enhanced Stability. ACS Appl Mater Interfaces. 2023, 15, 24638–24647. [Google Scholar] [CrossRef]
- Jing, H.; Wang, Y.; Desai, P.R.; Ramamurthi, K.S.; Das, S. Lipid flip-flop and desorption from supported lipid bilayers is independent of curvature. PLoS ONE. 2020, 15, e0244460. [Google Scholar] [CrossRef]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J Mol Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Lind, T.K.; Cárdenas, M. Understanding the formation of supported lipid bilayers via vesicle fusion-A case that exemplifies the need for the complementary method approach (Review). Biointerphases. 2016, 11, 020801. [Google Scholar] [CrossRef]
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Open Phys. 2012, 10. [Google Scholar] [CrossRef]
- Orlikowska-Rzeznik, H.; Krok, E.; Chattopadhyay, M.; Lester, A.; Piatkowski, L. Laurdan Discerns Lipid Membrane Hydration and Cholesterol Content. The Journal of Physical Chemistry B. 2023. [Google Scholar] [CrossRef]
- García-Arribas, A.B.; Busto, J.V.; Alonso, A.; Goñi, F.M. Atomic force microscopy characterization of palmitoylceramide and cholesterol effects on phospholipid bilayers: A topographic and nanomechanical study. Langmuir. 2015, 31, 3135–3145. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, H.; Fukuma, T. The molecular-scale arrangement and mechanical strength of phospholipid/cholesterol mixed bilayers investigated by frequency modulation atomic force microscopy in liquid. Nanotechnology. 2009, 20, 264008. [Google Scholar] [CrossRef] [PubMed]
- Almeida, P.F. A simple thermodynamic model of the liquid-ordered state and the interactions between phospholipids and cholesterol. Biophys J. 2011, 100, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Galván-Hernández, A.; Kobayashi, N.; Hernández-Cobos, J.; Antillón, A.; Nakabayashi, S.; Ortega-Blake, I. Morphology and dynamics of domains in ergosterol or cholesterol containing membranes. Biochim Biophys Acta Biomembr. 2020, 1862, 183101. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



