Submitted:
04 November 2025
Posted:
05 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Funding
Conflicts of Interest
References
- Tsuiki E, Kusano M, Kitaoka T: Complication associated with intravitreal injection of tissue plasminogen activator for treatment of submacular hemorrhage due to rupture of retinal arterial macroaneurysm. Am J Ophthalmol Case Rep. 2019, 16:. 10.1016/j.ajoc.2019. 1005.
- Mohammed TK, Simon CL, Gorman EF, Taubenslag KJ: Management of Submacular Hemorrhage. Curr Surg Rep. 2022, 10:. 10. 1007.
- Iannetta D, De Maria M, Bolletta E, Mastrofilippo V, Moramarco A, Fontana L: Subretinal injection of recombinant tissue plasminogen activator and gas tamponade to displace acute submacular haemorrhages secondary to age-related macular degeneration. Clinical Ophthalmology. 2021, 15:. 10.2147/OPTH. 3240.
- Toth CA, Morse LS, Hjelmeland LM, Landers MB: Fibrin Directs Early Retinal Damage After Experimental Subretinal Hemorrhage. Archives of Ophthalmology. 1991, 109:. 10.1001/archopht.1991. 0108.
- Daniel E, Toth CA, Grunwald JE, et al.: Risk of scar in the comparison of Age-related Macular Degeneration Treatments Trials. Ophthalmology. 2014, 121:. 10.1016/j.ophtha.2013.10.
- Stanescu-Segall D, Balta F, Jackson TL: Submacular hemorrhage in neovascular age-related macular degeneration: A synthesis of the literature. Surv Ophthalmol. 2016, 61:. 10.1016/j.survophthal.2015.04.
- Jackson TL, Bunce C, Desai R, et al.: Vitrectomy, subretinal Tissue plasminogen activator and Intravitreal Gas for submacular haemorrhage secondary to Exudative Age-Related macular degeneration (TIGER): study protocol for a phase 3, pan-European, two-group, non-commercial, active-control, observer-masked, superiority, randomised controlled surgical trial. Trials. 2022, 23:99. 10. 1186.
- Iannetta D, De Maria M, Bolletta E, Mastrofilippo V, Moramarco A, Fontana L: Subretinal Injection of Recombinant Tissue Plasminogen Activator and Gas Tamponade to Displace Acute Submacular Haemorrhages Secondary to Age-Related Macular Degeneration. Clinical Ophthalmology. 2021, Volume 15:3649–59. 10.2147/OPTH. 3240.
- Veritti D, Sarao V, Martinuzzi D, Menzio S, Lanzetta P: Submacular Hemorrhage during Neovascular Age-Related Macular Degeneration: A Meta-Analysis and Meta-Regression on the Use of tPA and Anti-VEGFs. Ophthalmologica. 2024, 247:191–202. 10. 1159.
- Wolfrum P, Böhm EW, König S, Lorenz K, Stoffelns B, Korb CA: Safety and Long-Term Efficacy of Intravitreal rtPA, Bevacizumab and SF6 Injection in Patients with Submacular Hemorrhage Secondary to Age-Related Macular Degeneration. J Clin Med. 2025, 14:. 10. 3390.
- Szeto SKH, Tsang CW, Mohamed S, et al.: Displacement of Submacular Hemorrhage Using Subretinal Cocktail Injection versus Pneumatic Displacement: A Real-World Comparative Study. Ophthalmologica. 2024, 247:118–31. 10. 1159.
- De Silva SR, Bindra MS: Early treatment of acute submacular haemorrhage secondary to wet AMD using intravitreal tissue plasminogen activator, C 3F8, and an anti-VEGF agent. Eye (Basingstoke). 2016, 30:. 10.1038/eye.2016.
- Lim JH, Han YS, Lee SJ, Nam KY: Risk factors for breakthrough vitreous hemorrhage after intravitreal tissue plasminogen activator and gas injection for submacular hemorrhage associated with age related macular degeneration. PLoS One. 2020, 15:e0243201. 10.1371/journal.pone. 0243.
- NCT04663750: Vitrectomy, Subretinal Tissue Plasminogen Activator (TPA) and Intravitreal Gas for Submacular Haemorrhage Secondary to Exudative (Wet) Age-related Macular Degeneration (TIGER). https://clinicaltrials.gov/show/NCT04663750. 2020.
- Lee CN, Desai R, Ramazzotto L, et al.: Vitrectomy, subretinal Tissue plasminogen activator and Intravitreal Gas for submacular haemorrhage secondary to Exudative Age-Related macular degeneration (TIGER): update to study protocol and addition of a statistical analysis plan and health economic analysis plan for a randomised controlled surgical trial. Trials. 2025, 26:. 10. 1186.
- Bae K, Cho GE, Yoon JM, Kang SW: Optical coherence tomographic features and prognosis of pneumatic displacement for submacular hemorrhage. PLoS One. 2016, 11:. 10.1371/journal.pone. 0168.
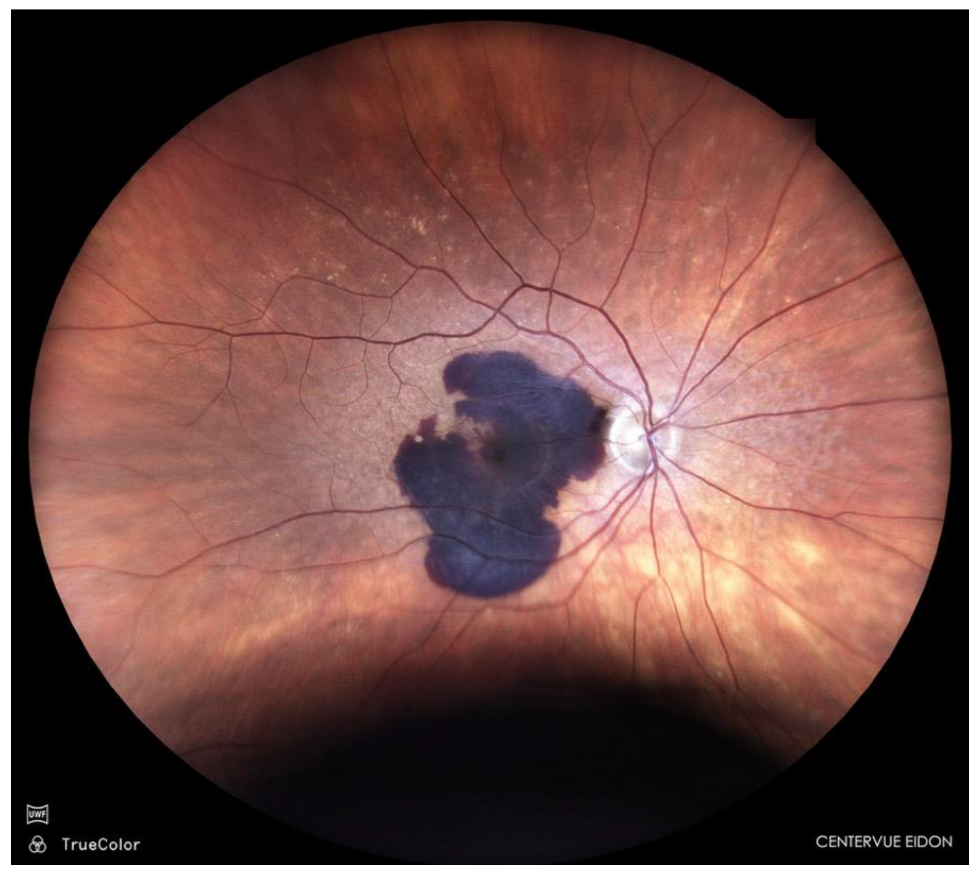
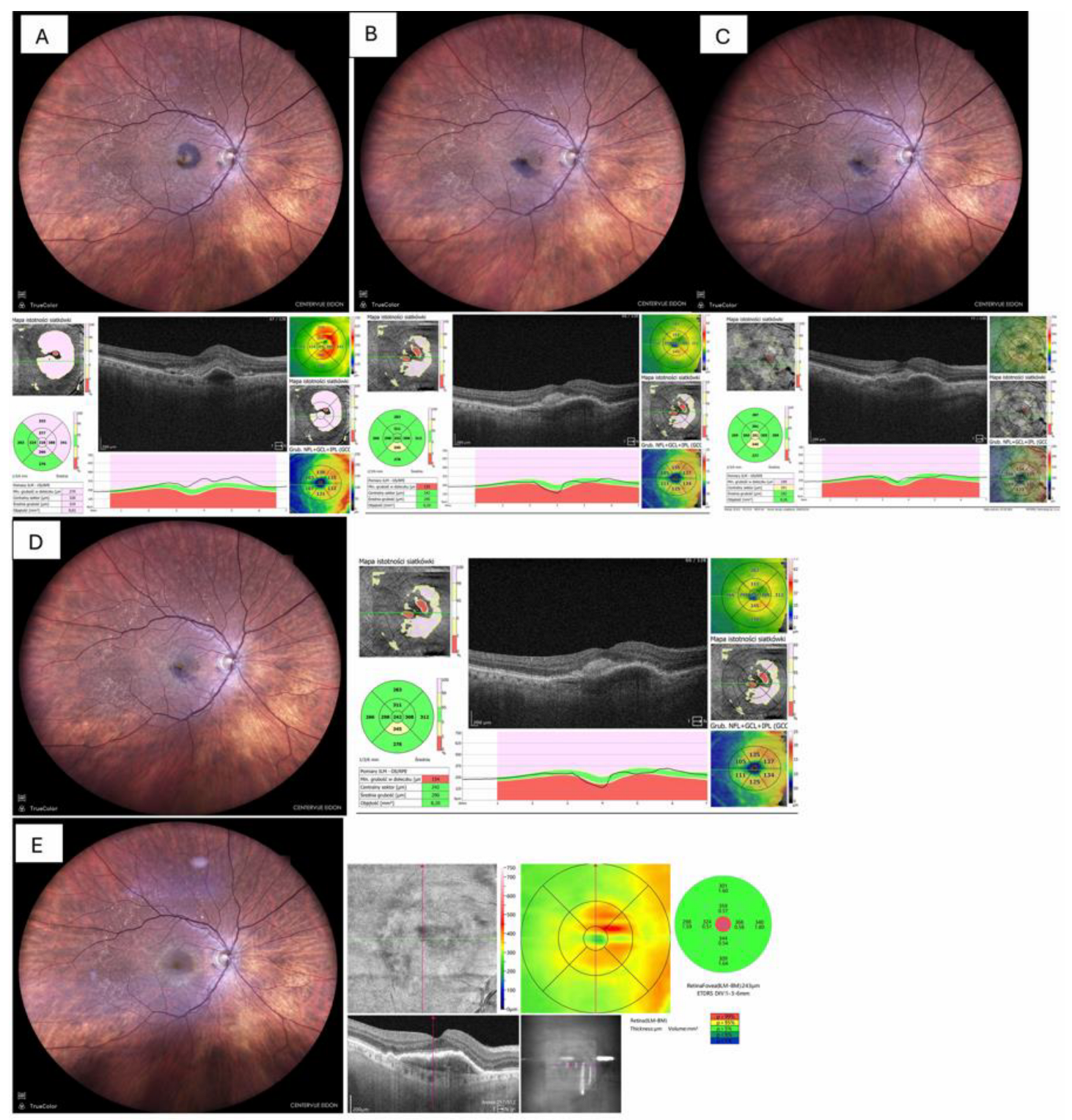
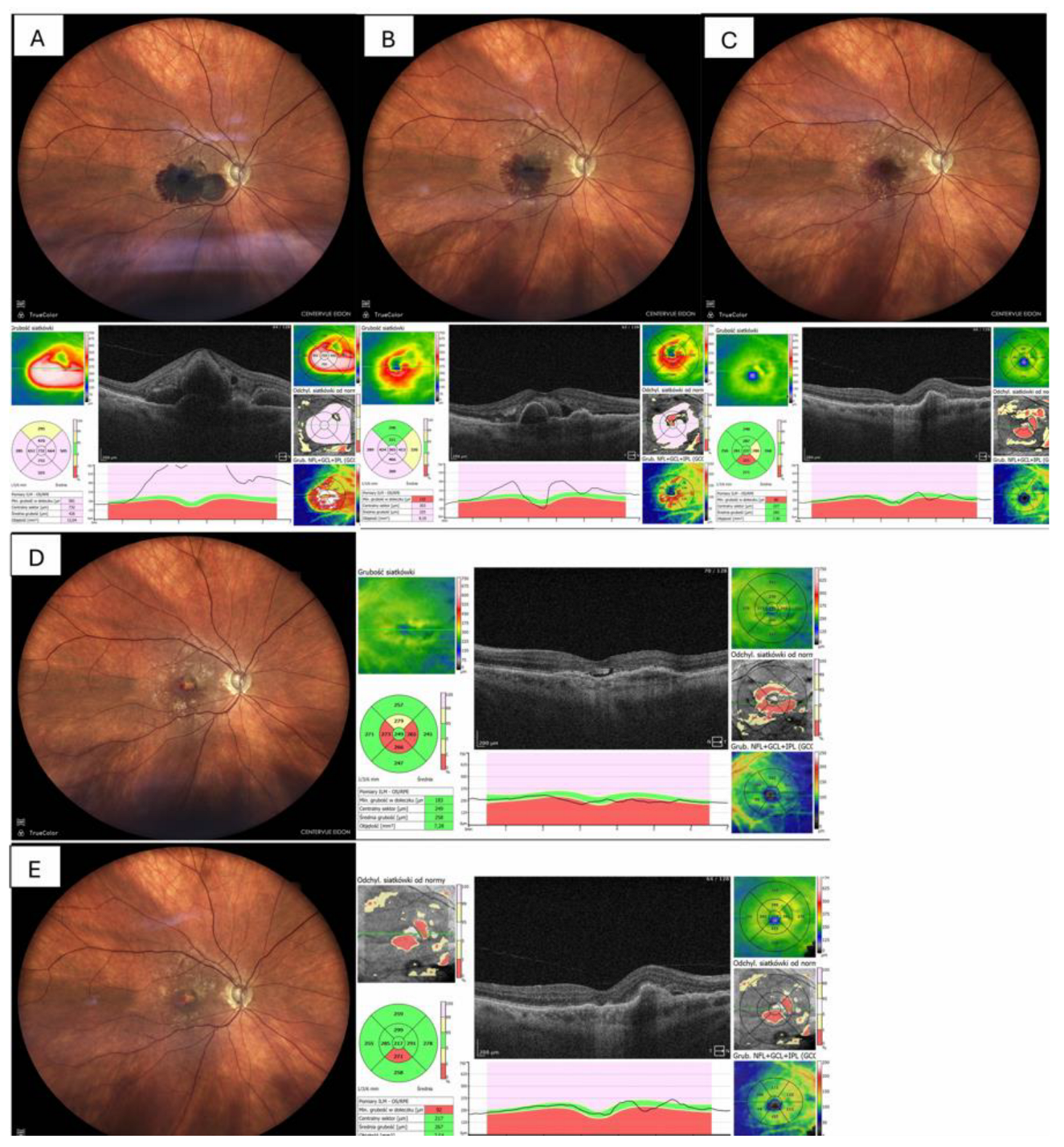
| Case No | Age (y) | Sex | Initial diagnosis | Duration of symptoms (days) |
|---|---|---|---|---|
| 1. | 90 | F | Sub-macular haemorrhage, AMD | 60 |
| 2. | 85 | F | Sub-macular haemorrhage, AMD | 7 |
| 3. | 74 | M | Sub-macular haemorrhage, AMD | 21 |
| 4. | 78 | M | Sub-macular haemorrhage, AMD | 10 |
| 5. | 78 | M | Sub-macular haemorrhage, AMD | 7 |
| 6. | 68 | M | Sub-macular haemorrhage, AMD | 14 |
| 7. | 73 | M | Sub-macular haemorrhage, AMD | 7 |
| 8. | 77 | F | Sub-macular haemorrhage, AMD | 56 |
| 9. | 60 | F | Sub-macular haemorrhage, AMD | 60 |
| 10. | 77 | F | Sub-macular haemorrhage, AMD | 8 |
| mean | 76 | 24 | ||
| median | 77 | 12 |
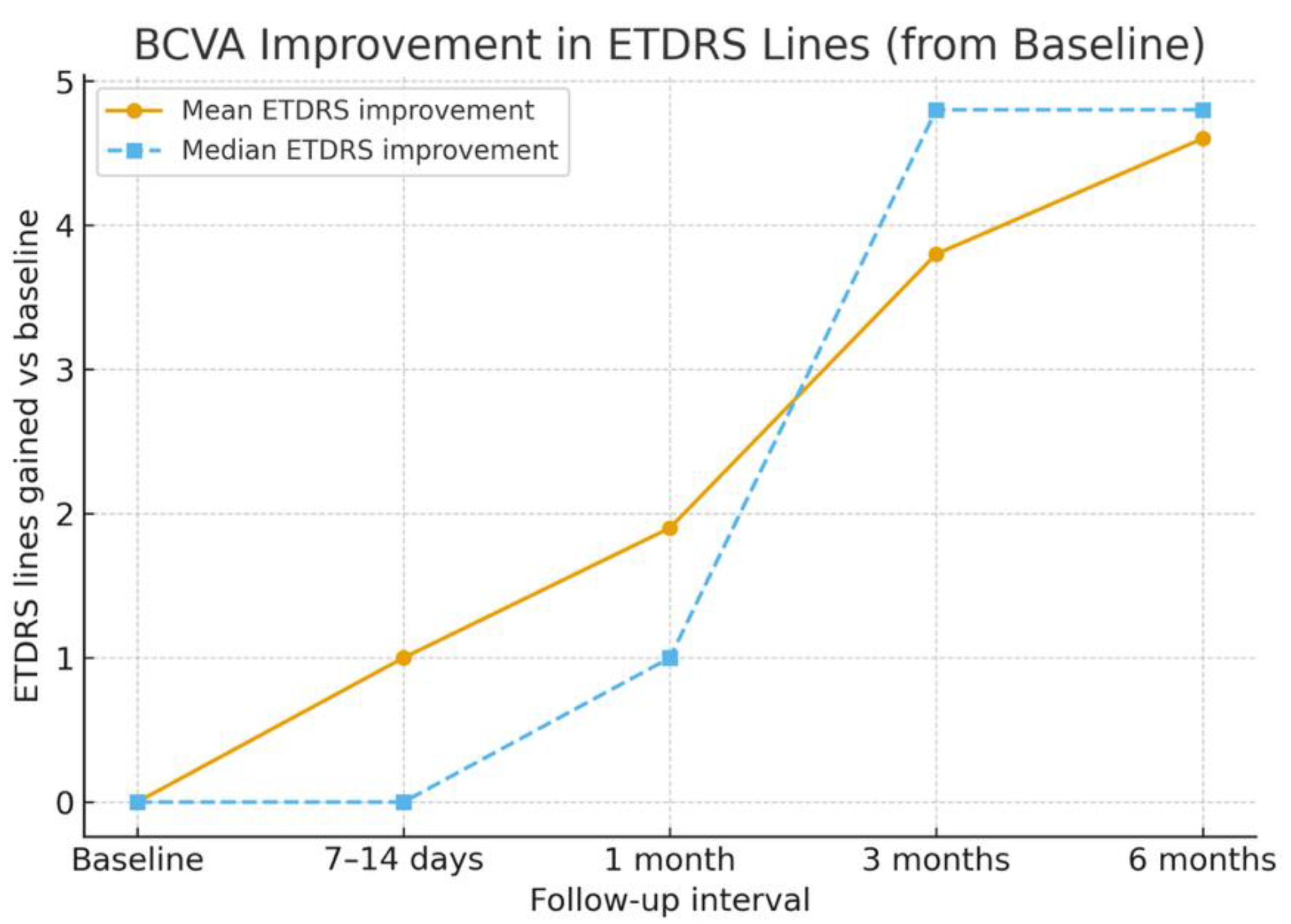
| Case No | BCVA Before | 7–14 days postop. | 1 month postop. | 3 months postop. | 6 months postop. | Complications |
|---|---|---|---|---|---|---|
| 1 | 1.00 | 0.70 | 1.00 | 0.52 | 0.52 | none |
| 2 | 1.30 | 0.70 | 0.70 | 0.52 | 0.40 | none |
| 3 | 2.00 | 2.00 | 2.00 | 1.40 | 1.40 | MNV scar |
| 4 | 1.30 | 1.30 | 0.52 | 0.40 | 0.30 | none |
| 5 | 0.30 | 0.52 | 0.52 | 0.40 | 0.40 | none |
| 6 | 0.60 | 0.60 | 0.60 | 0.52 | 0.22 | none |
| 7 | 0.40 | 0.40 | 0.52 | 0.52 | 0.52 | cataract |
| 8 | 1.00 | 1.00 | 0.70 | 0.52 | 0.52 | cataract |
| 9 | 0.70 | 0.40 | 0.30 | 0.22 | 0.10 | IOP rise |
| 10 | 1.30 | 1.30 | 1.10 | 1.10 | 1.10 | MNV scar |
| Mean | 0.99 | 0.89 | 0.8 | 0.6 | 0.53 | |
| Median | 1.0 | 0.7 | 0.65 | 0.52 | 0.4 |
| Finding | Reported | Case No |
|---|---|---|
| IOP rise | 1 patient | Case 9 |
| Cataract | 2 cases | Cases 7 & 8 |
| MNV scar | 2 cases | Cases 3 & 10 |
| Endophthalmitis/retinal detachment/other complications | None | None |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




