Submitted:
03 November 2025
Posted:
04 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Isolation of the Spinal Cord
2.3. Spinal Cord Slice Preparation
2.4. Culture of Spinal Astrocytes from WT Mice
2.5. Human Tissue Samples
2.6. Generation of hiAstrocytes from Skin Fibroblasts
2.7. Induction of SMN Deficiency in Cultured Spinal Astrocytes
2.8. Immunostaining
2.9. Western Blots
2.10. Real-Time Quantitative Polymerase Chain Reactions
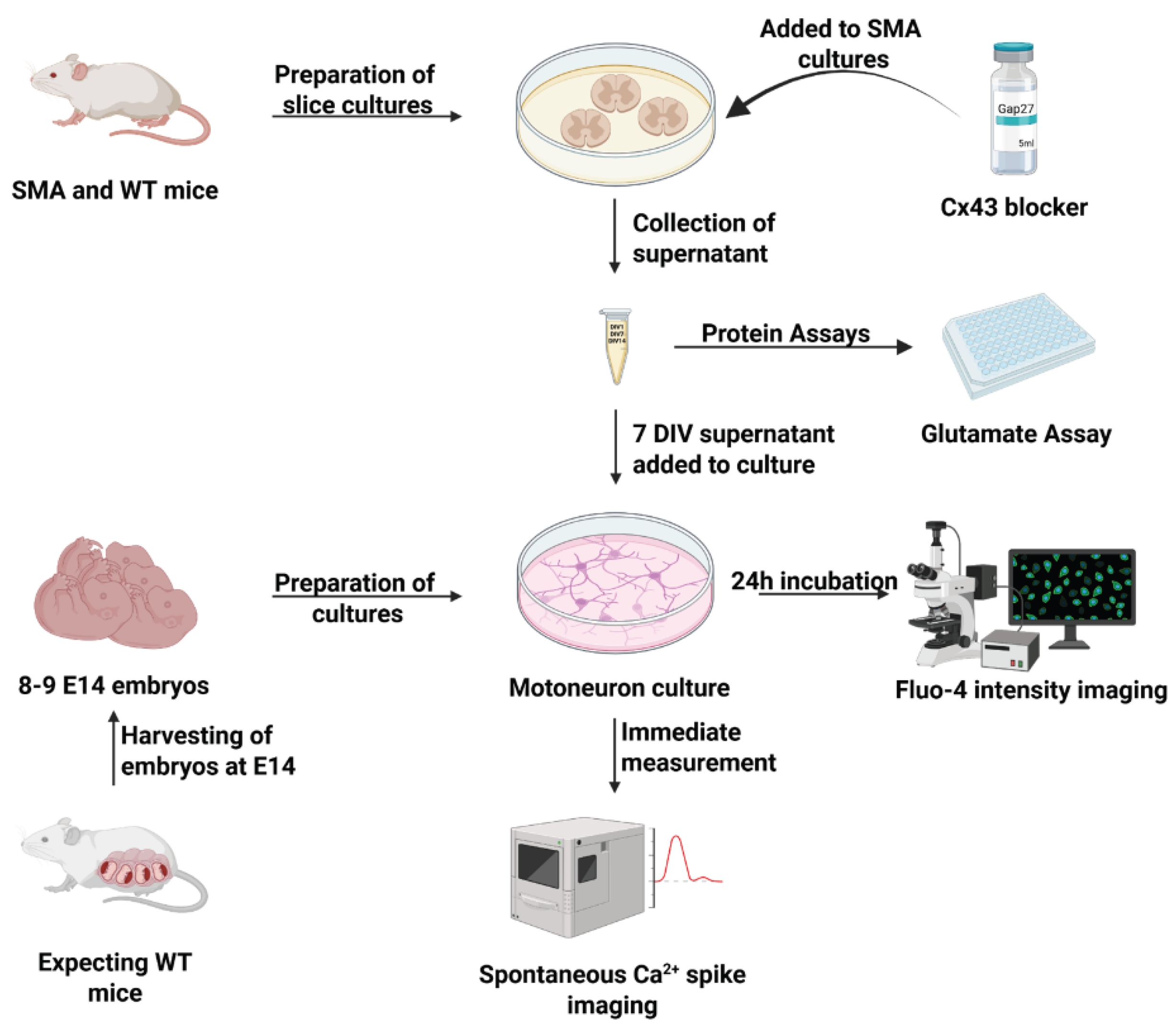
2.11. Isolation and Culture of Organotypic Spinal Cord Slice Cultures from WT and SMA-Mice
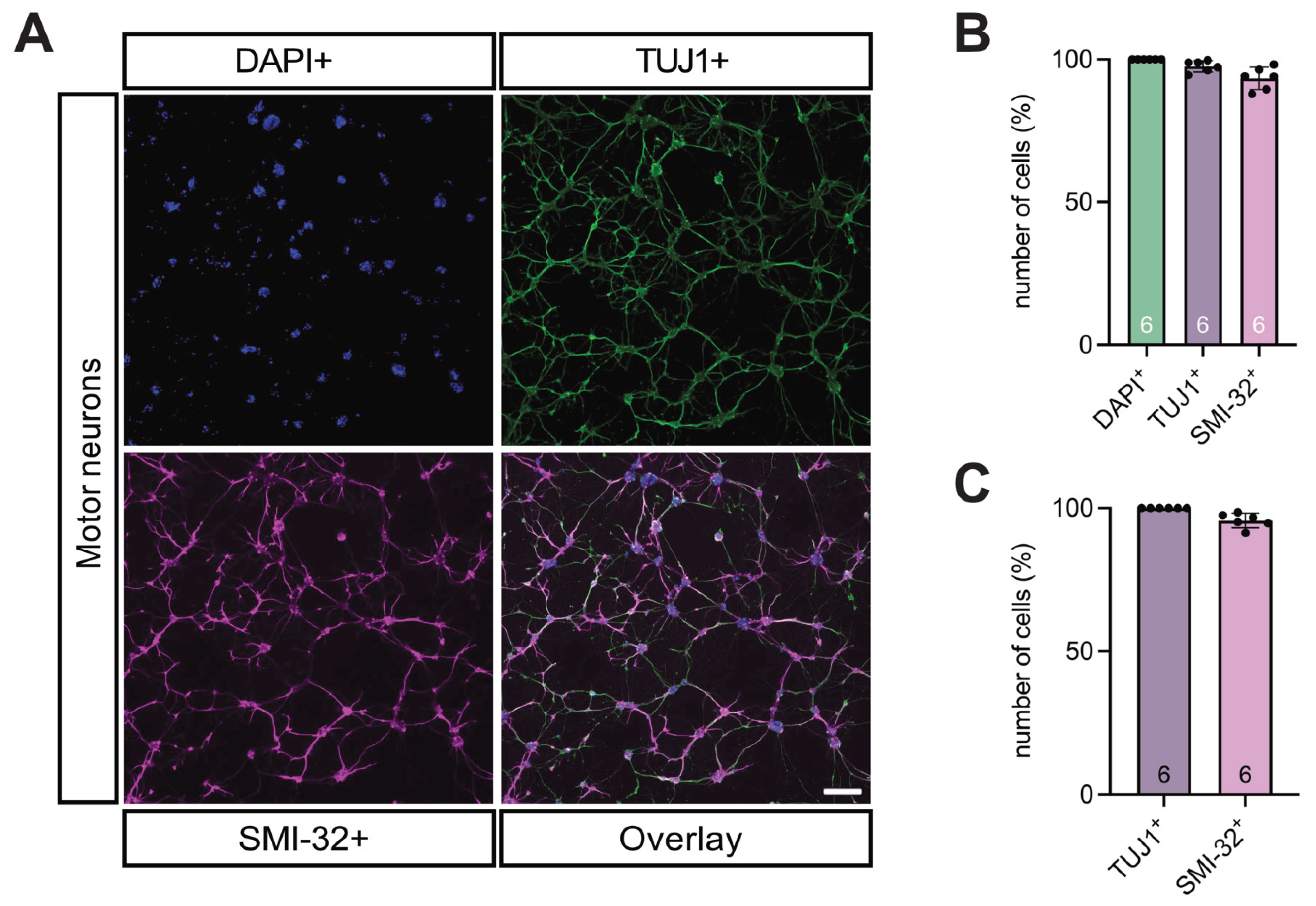
2.12. Isolation and Culture of Spinal Motor Neurons from Embryonic WT Mice
2.13. Calcium Imaging
2.14. Glutamate Assay
2.15. Statistical Analysis
3. Results
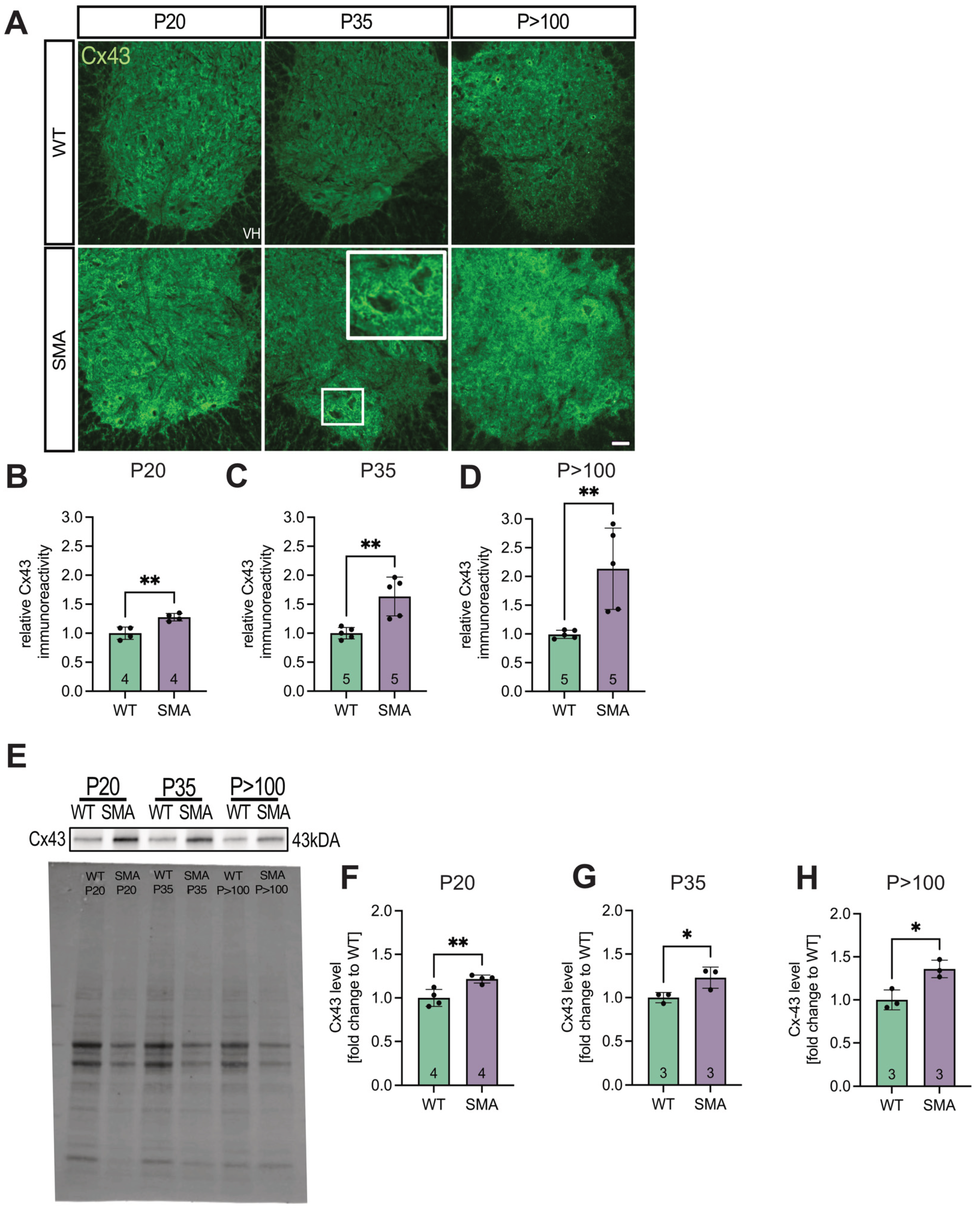
3.1. Spinal Cord Tissue Exhibited Increased Expression of Cx43 in Late-Onset SMA Mice
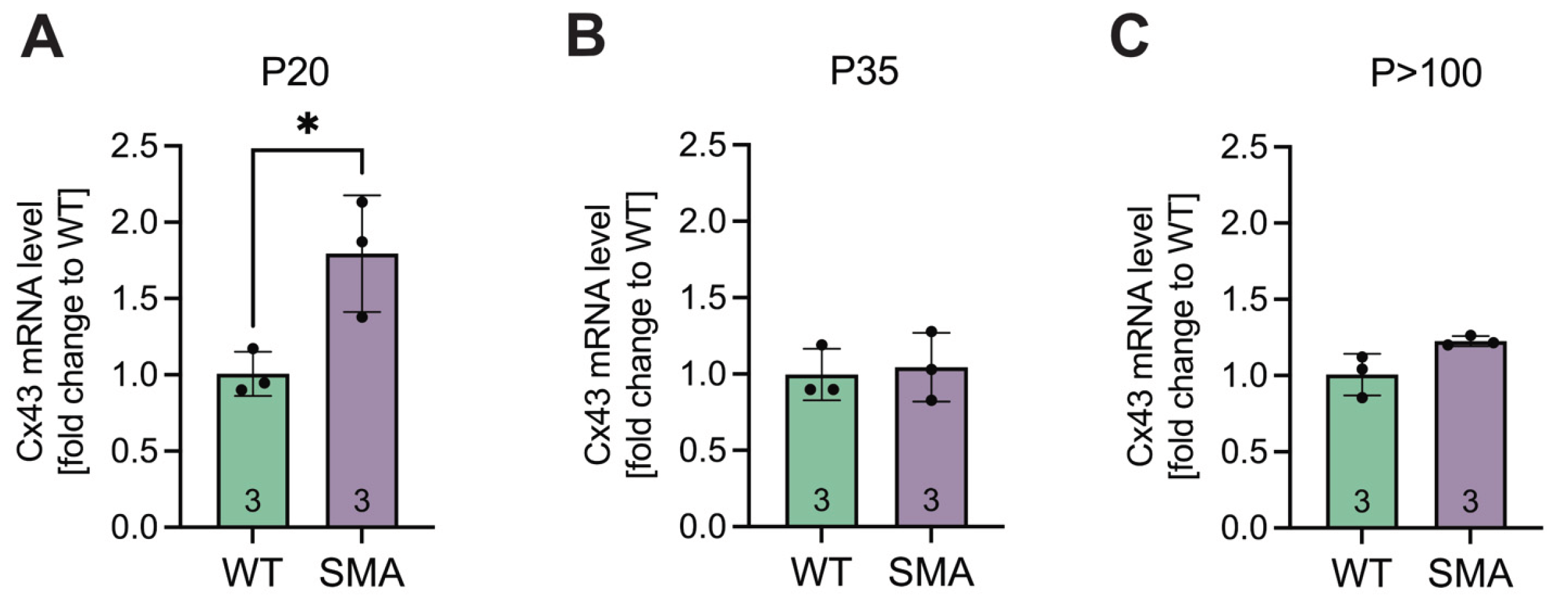
3.2. Cx43 mRNA Expression Is Post-Transcriptionally Modulated
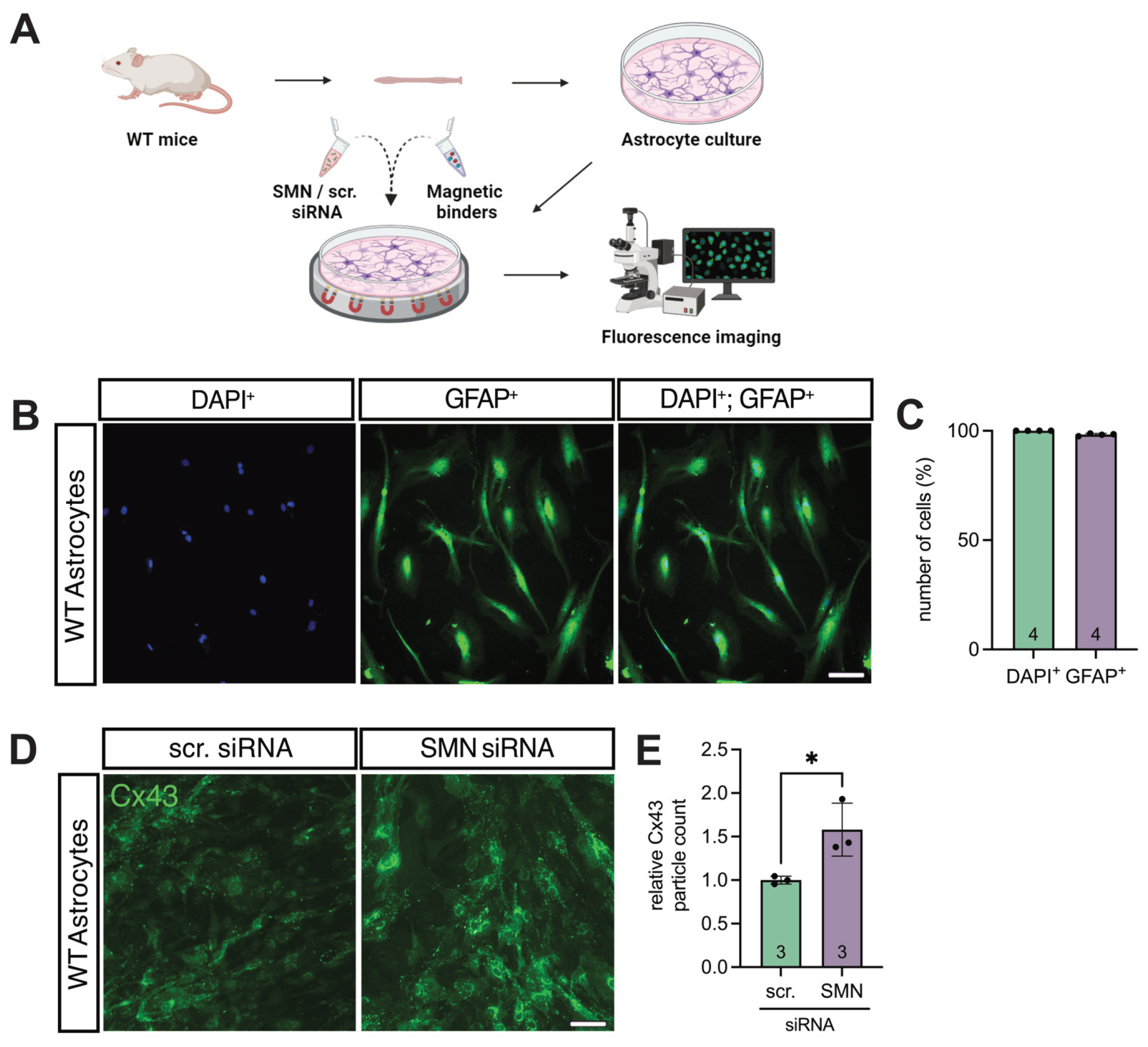
3.3. SMN Deficiency Increased Astrocytic Cx43 Expression
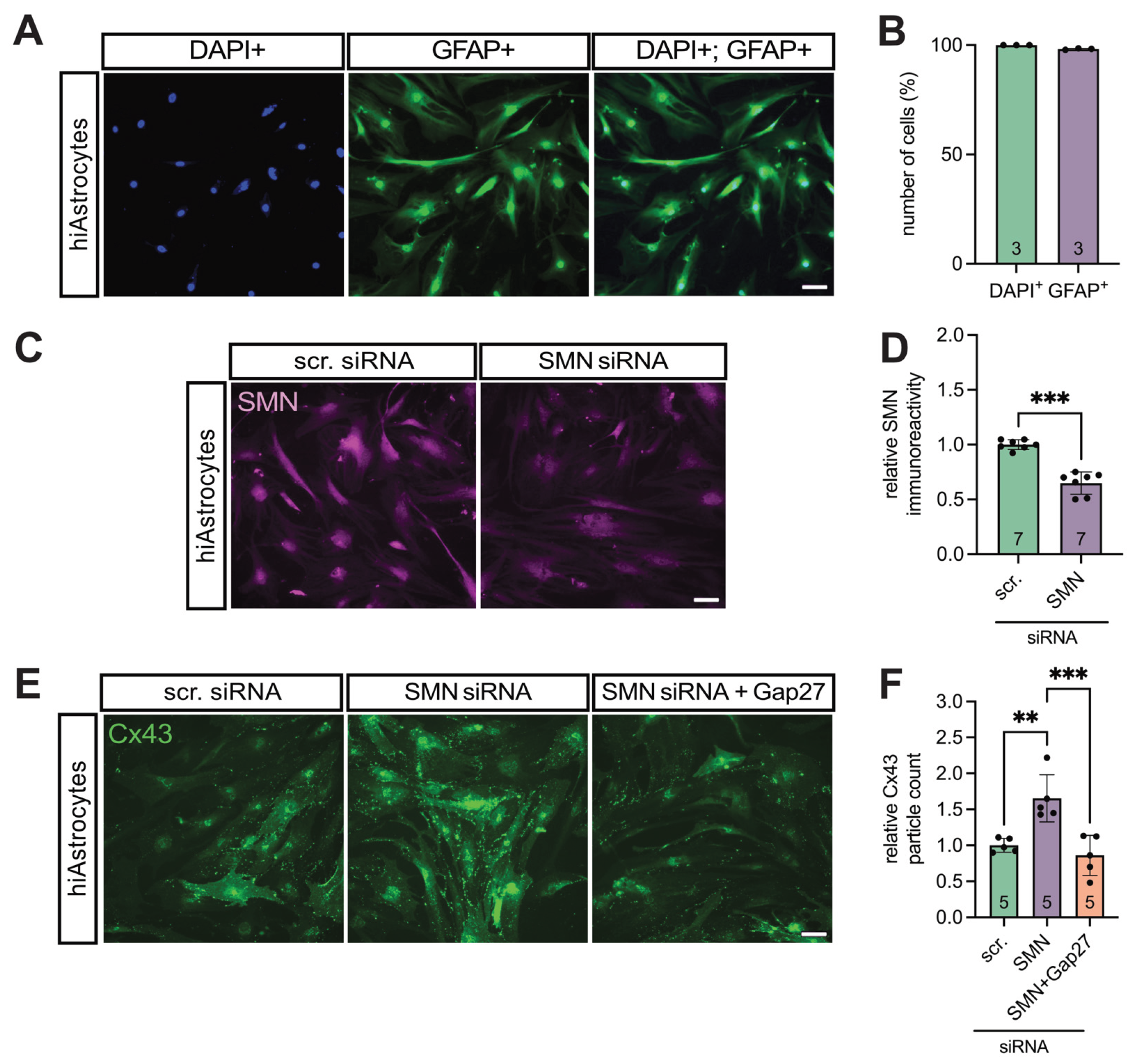
3.4. Translational Validation of Cx43 Dysregulation Using a Human iPSC-Based Model
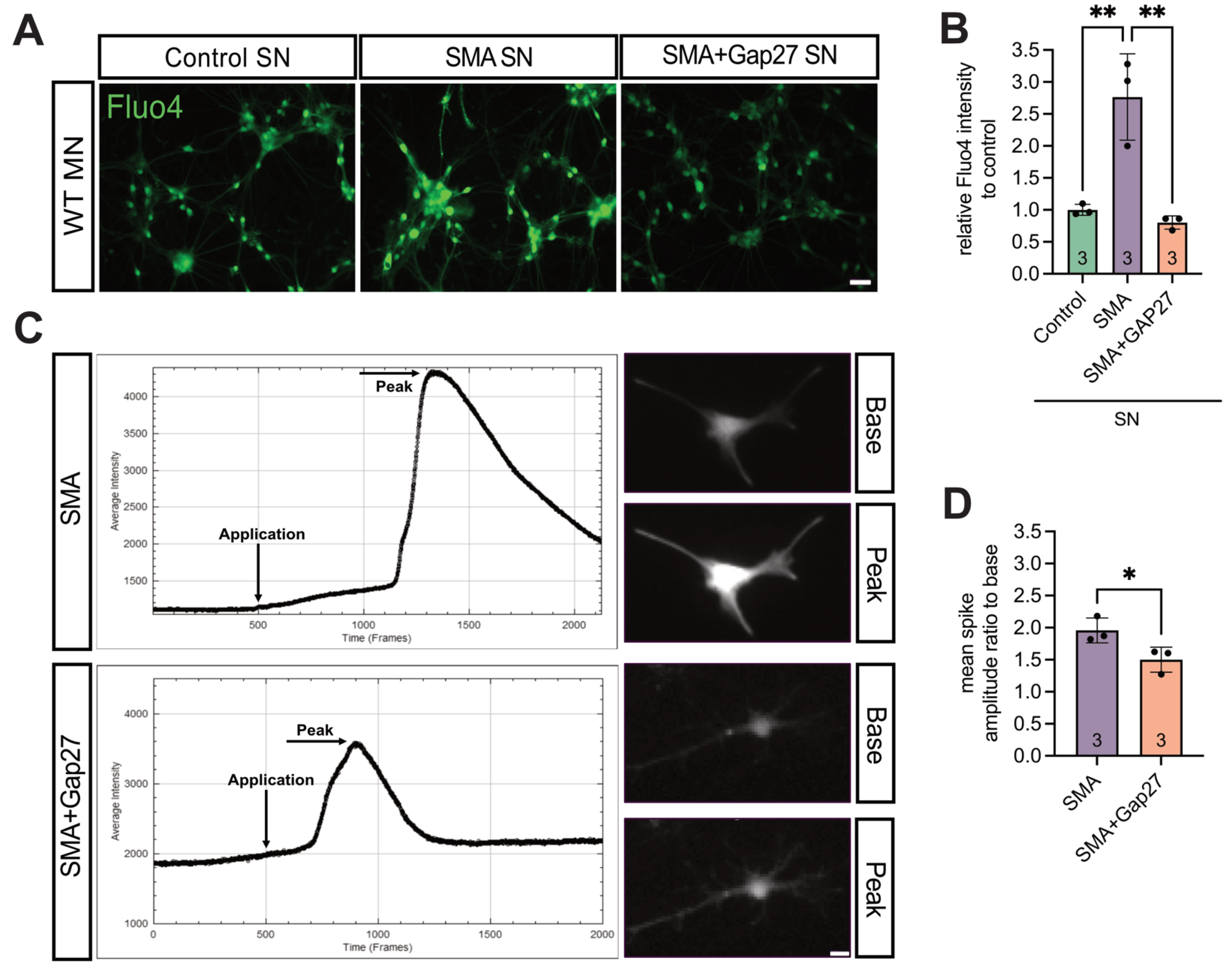
3.5. Cx43 Inhibition Reduces SMA-Associated Calcium Levels in MNs
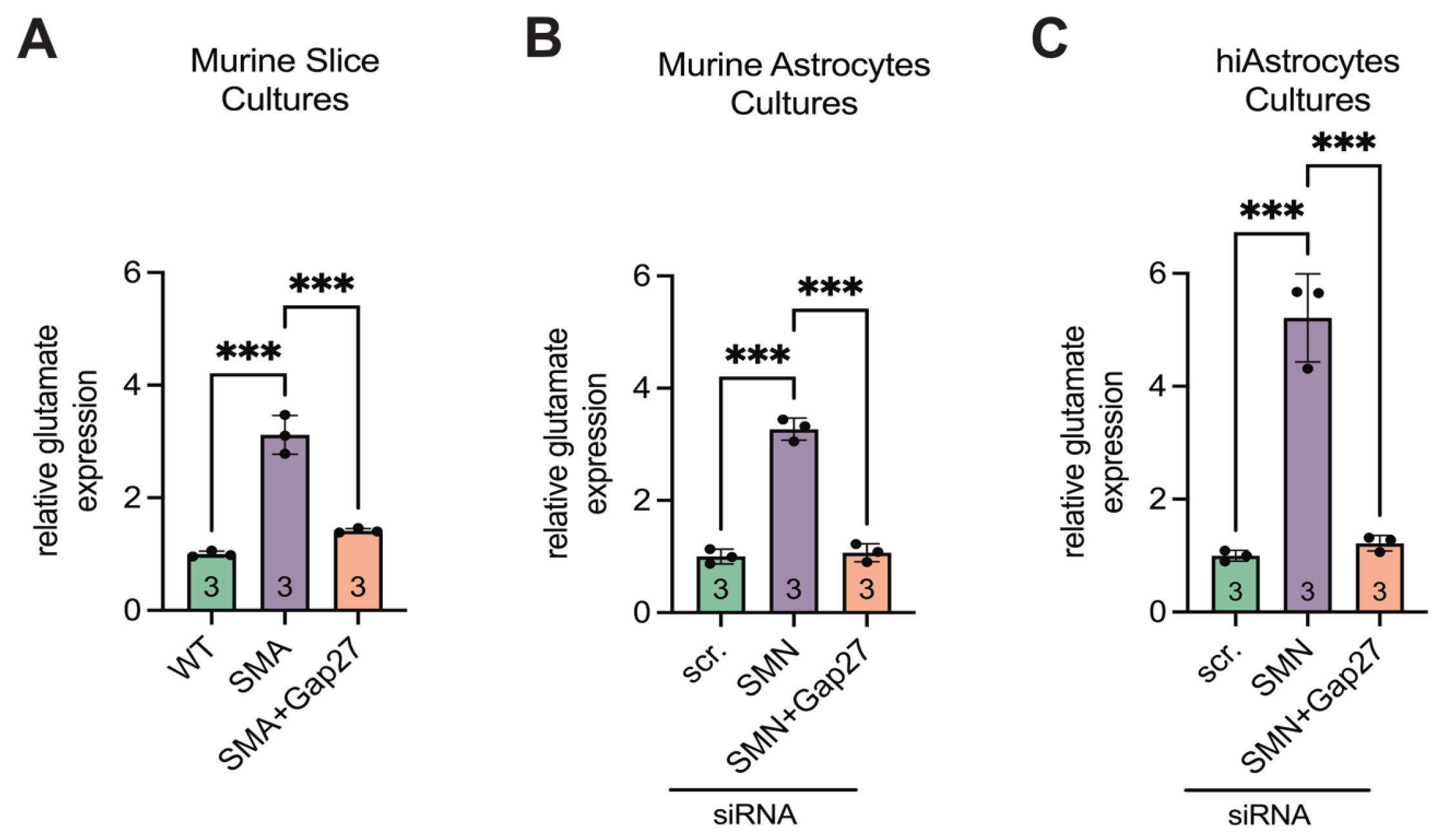
3.6. SMA Cultures Showed Elevated Glutamate Levels, Reversed by Inhibiting Cx43
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALS | amyotrophic lateral sclerosis |
| ATP | adenosine triphosphate |
| BCA | bicinchoninic acid protein assay |
| Ca2+ | calcium ion |
| Cx43 | connexin 43 |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DIV | days in vitro |
| DMD | Duchenne muscular dystrophy |
| E | embryonal day |
| EGF | epidermal growth factor |
| ELISA | enzyme-linked immunosorbent assay |
| FBS | fetal bovine serum |
| FGF | fibroblast growth factor |
| iPSC | induced pluripotent stem cells |
| GFAP | glial fibrillary acidic protein |
| GJA1 | gap junction alpha-1 |
| hiAstrocytes | human induced astrocytes |
| late-onset SMA | late-onset spinal muscular atrophy |
| P | postnatal day |
| PBS | phosphate-buffered saline |
| PDL | poly-d-lysine |
| qPCR | quantitative polymerase chain reactions |
| RT | room temperature |
| scr | scrambled |
| SMA | spinal muscular atrophy |
| SMI-32 | non-phosphorylated neurofilament H |
| SMN1 | survival of motor neuron-1 |
| SMN2 | survival of motor neuron-2 |
| SMN | survival of motor neuron |
| TUJ1 | βIII-tubulin |
| WB | Western blot |
| WT | wild-type |
References
- Crawford, T. O. and C. A. Pardo. “The neurobiology of childhood spinal muscular atrophy.” Neurobiology of Disease 3 (1996): 97-110. [CrossRef]
- Lefebvre, S., L. Bürglen, S. Reboullet, O. Clermont, P. Burlet, L. Viollet, B. Benichou, C. Cruaud, P. Millasseau, M. Zeviani, et al. “Identification and characterization of a spinal muscular atrophy-determining gene.” Cell 80 (1995): 155-65. [CrossRef]
- Monani, U. R. “The human centromeric survival motor neuron gene (smn2) rescues embryonic lethality in smn-/- mice and results in a mouse with spinal muscular atrophy.” Human Molecular Genetics 9 (2000): 333-39. [CrossRef]
- Feldkötter, M., V. Schwarzer, R. Wirth, T. F. Wienker and B. Wirth. “Quantitative analyses of smn1 and smn2 based on real-time lightcycler pcr: Fast and highly reliable carrier testing and prediction of severity of spinal muscular atrophy.” The American Journal of Human Genetics 70 (2002): 358-68. [CrossRef]
- McAndrew, P. E., D. W. Parsons, L. R. Simard, C. Rochette, P. N. Ray, J. R. Mendell, T. W. Prior and A. H. M. Burghes. “Identification of proximal spinal muscular atrophy carriers and patients by analysis of smnt and smnc gene copy number.” The American Journal of Human Genetics 60 (1997): 1411-22. [CrossRef]
- Chaytow, H., K. M. E. Faller, Y.-T. Huang and T. H. Gillingwater. “Spinal muscular atrophy: From approved therapies to future therapeutic targets for personalized medicine.” Cell Reports Medicine 2 (2021): 10.1016/j.xcrm.2021.100346.
- Finkel, R. S., E. Mercuri, B. T. Darras, A. M. Connolly, N. L. Kuntz, J. Kirschner, C. A. Chiriboga, K. Saito, L. Servais, E. Tizzano, et al. “Nusinersen versus sham control in infantile-onset spinal muscular atrophy.” N Engl J Med 377 (2017): 1723-32. https://www.ncbi.nlm.nih.gov/pubmed/29091570. [CrossRef]
- Hagenacker, T., C. D. Wurster, R. Günther, O. Schreiber-Katz, A. Osmanovic, S. Petri, M. Weiler, A. Ziegler, J. Kuttler, J. C. Koch, et al. “Nusinersen in adults with 5q spinal muscular atrophy: A non-interventional, multicentre, observational cohort study.” The Lancet Neurology 19 (2020): 317-25. [CrossRef]
- Chiriboga, C. A., K. J. Swoboda, B. T. Darras, S. T. Iannaccone, J. Montes, D. C. De Vivo, D. A. Norris, C. F. Bennett and K. M. Bishop. “Results from a phase 1 study of nusinersen (isis-smn rx) in children with spinal muscular atrophy.” Neurology 86 (2016): 890-97. [CrossRef]
- Mercuri, E., B. T. Darras, C. A. Chiriboga, J. W. Day, C. Campbell, A. M. Connolly, S. T. Iannaccone, J. Kirschner, N. L. Kuntz, K. Saito, et al. “Nusinersen versus sham control in later-onset spinal muscular atrophy.” New England Journal of Medicine 378 (2018): 625-35. [CrossRef]
- Mercuri, E., F. Muntoni, G. Baranello, R. Masson, O. Boespflug-Tanguy, C. Bruno, S. Corti, A. Daron, N. Deconinck, L. Servais, et al. “Onasemnogene abeparvovec gene therapy for symptomatic infantile-onset spinal muscular atrophy type 1 (str1ve-eu): An open-label, single-arm, multicentre, phase 3 trial.” The Lancet Neurology 20 (2021): 832-41. [CrossRef]
- Mercuri, E., N. Deconinck, E. S. Mazzone, A. Nascimento, M. Oskoui, K. Saito, C. Vuillerot, G. Baranello, O. Boespflug-Tanguy, N. Goemans, et al. “Safety and efficacy of once-daily risdiplam in type 2 and non-ambulant type 3 spinal muscular atrophy (sunfish part 2): A phase 3, double-blind, randomised, placebo-controlled trial.” The Lancet Neurology 21 (2022): 42-52. [CrossRef]
- Ponath, G., S. Ramanan, M. Mubarak, W. Housley, S. Lee, F. R. Sahinkaya, A. Vortmeyer, C. S. Raine and D. Pitt. “Myelin phagocytosis by astrocytes after myelin damage promotes lesion pathology.” Brain 140 (2017): 399-413. [CrossRef]
- Yamanaka, K., S. J. Chun, S. Boillee, N. Fujimori-Tonou, H. Yamashita, D. H. Gutmann, R. Takahashi, H. Misawa and D. W. Cleveland. “Astrocytes as determinants of disease progression in inherited amyotrophic lateral sclerosis.” Nature Neuroscience 11 (2008): 251-53. [CrossRef]
- McGivern, J. V., T. N. Patitucci, J. A. Nord, M. A. Barabas, C. L. Stucky and A. D. Ebert. “Spinal muscular atrophy astrocytes exhibit abnormal calcium regulation and reduced growth factor production.” Glia 61 (2013): 1418-28. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3941074/pdf/nihms-561874.pdf. [CrossRef]
- Schmitt, L. I., C. David, R. Steffen, S. Hezel, A. Roos, U. Schara-Schmidt, C. Kleinschnitz, M. Leo and T. Hagenacker. “Spinal astrocyte dysfunction drives motor neuron loss in late-onset spinal muscular atrophy.” Acta Neuropathologica 145 (2023): 611-35. https://www.ncbi.nlm.nih.gov/pubmed/36930296. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10119066/pdf/401_2023_Article_2554.pdf. [CrossRef]
- Leo, M., L. I. Schmitt, M. Fleischer, R. Steffen, C. Osswald, C. Kleinschnitz and T. Hagenacker. “Induction of survival of motor neuron (smn) protein deficiency in spinal astrocytes by small interfering rna as an in vitro model of spinal muscular atrophy.” Cells 11 (2022): https://mdpi-res.com/d_attachment/cells/cells-11-00558/article_deploy/cells-11-00558-v2.pdf?version=1644496536. [CrossRef]
- Konietzko, U. and C. M. Müller. “Astrocytic dye coupling in rat hippocampus: Topography, developmental onset, and modulation by protein kinase c.” Hippocampus 4 (2004): 297-306. [CrossRef]
- Musil, L. S. and D. A. Goodenough. “Biochemical analysis of connexin43 intracellular transport, phosphorylation, and assembly into gap junctional plaques.” The Journal of cell biology 115 (1991): 1357-74. [CrossRef]
- Soares, A. R., T. Martins-Marques, T. Ribeiro-Rodrigues, J. V. Ferreira, S. Catarino, M. J. Pinho, M. Zuzarte, S. Isabel Anjo, B. Manadas, J. P.G. Sluijter, et al. “Gap junctional protein cx43 is involved in the communication between extracellular vesicles and mammalian cells.” Scientific Reports 5 (2015): 10.1038/srep13243.
- Wang, X., M. L. Veruki, N. V. Bukoreshtliev, E. Hartveit and H.-H. Gerdes. “Animal cells connected by nanotubes can be electrically coupled through interposed gap-junction channels.” Proceedings of the National Academy of Sciences 107 (2010): 17194-99. [CrossRef]
- Giaume, C. and X. Liu. “From a glial syncytium to a more restricted and specific glial networking.” Journal of Physiology-Paris 106 (2012): 34-39. [CrossRef]
- Cheung, G., O. Chever, A. Rollenhagen, N. Quenech’du, P. Ezan, J. H. R. Lübke and N. Rouach. “Astroglial connexin 43 regulates synaptic vesicle release at hippocampal synapses.” Cells 12 (2023): 10.3390/cells12081133.
- Jiang, S., H. Yuan, L. Duan, R. Cao, B. Gao, Y.-F. Xiong and Z.-R. Rao. “Glutamate release through connexin 43 by cultured astrocytes in a stimulated hypertonicity model.” Brain Research 1392 (2011): 8-15. [CrossRef]
- Almad, A. A., A. Doreswamy, S. K. Gross, J. P. Richard, Y. Huo, N. Haughey and N. J. Maragakis. “Connexin 43 in astrocytes contributes to motor neuron toxicity in amyotrophic lateral sclerosis.” Glia 64 (2016): 1154-69. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5635605/pdf/nihms908168.pdf. [CrossRef]
- Hsieh-Li, H. M., J. G. Chang, Y. J. Jong, M. H. Wu, N. M. Wang, C. H. Tsai and H. Li. “A mouse model for spinal muscular atrophy.” Nature Genetics 24 (2000): 66-70. https://www.ncbi.nlm.nih.gov/pubmed/10615130. https://www.nature.com/articles/ng0100_66. [CrossRef]
- Meyer, K., Y. Rodriguez, F. S. Roussel, A. M. Hartlaub, S. S. Ray, J. A. Sierra-Delgado and C. N. Dennys. “In vitro modeling for neurological diseases using direct conversion from fibroblasts to neuronal progenitor cells and differentiation into astrocytes.” Journal of Visualized Experiments (2021): 10.3791/62016.
- Orellana, J. A., D. E. Hernández, P. Ezan, V. Velarde, M. V. L. Bennett, C. Giaume and J. C. Sáez. “Hypoxia in high glucose followed by reoxygenation in normal glucose reduces the viability of cortical astrocytes through increased permeability of connexin 43 hemichannels.” Glia 58 (2009): 329-43. [CrossRef]
- Baker, K. E. and J. Coller. “The many routes to regulating mrna translation.” Genome Biology 7 (2006): 10.1186/gb-2006-7-12-332.
- Pellizzoni, L., N. Kataoka, B. Charroux and G. Dreyfuss. “A novel function for smn, the spinal muscular atrophy disease gene product, in pre-mrna splicing.” Cell 95 (1998): 615-24. [CrossRef]
- Markoullis, K., I. Sargiannidou, N. Schiza, A. Hadjisavvas, F. Roncaroli, R. Reynolds and K. A. Kleopa. “Gap junction pathology in multiple sclerosis lesions and normal-appearing white matter.” Acta Neuropathologica 123 (2012): 873-86. [CrossRef]
- Mercuri, E., M. C. Pera, M. Scoto, R. Finkel and F. Muntoni. “Spinal muscular atrophy — insights and challenges in the treatment era.” Nature Reviews Neurology 16 (2020): 706-15. [CrossRef]
- Abudara, V. n., J. Bechberger, M. Freitas-Andrade, M. De Bock, N. Wang, G. Bultynck, C. C. Naus, L. Leybaert and C. Giaume. “The connexin43 mimetic peptide gap19 inhibits hemichannels without altering gap junctional communication in astrocytes.” Frontiers in Cellular Neuroscience 8 (2014): 10.3389/fncel.2014.00306.
- Grek, C. L., G. M. Prasad, V. Viswanathan, D. G. Armstrong, R. G. Gourdie and G. S. Ghatnekar. “Topical administration of a connexin43--based peptide augments healing of chronic neuropathic diabetic foot ulcers: A multicenter, randomized trial.” Wound Repair and Regeneration 23 (2015): 203-12. [CrossRef]
- Ghatnekar, G. S., C. L. Grek, D. G. Armstrong, S. C. Desai and R. G. Gourdie. “The effect of a connexin43-based peptide on the healing of chronic venous leg ulcers: A multicenter, randomized trial.” Journal of Investigative Dermatology 135 (2015): 289-98. [CrossRef]
- Kwakowsky, A., B. Chawdhary, A. de Souza, E. Meyer, A. H. Kaye, C. R. Green, S. S. Stylli and H. Danesh-Meyer. “Tonabersat significantly reduces disease progression in an experimental mouse model of multiple sclerosis.” International Journal of Molecular Sciences 24 (2023): 10.3390/ijms242417454. https://mdpi-res.com/d_attachment/ijms/ijms-24-17454/article_deploy/ijms-24-17454-v2.pdf?version=1702716205.
- Wei, H., F. Deng, Y. Chen, Y. Qin, Y. Hao and X. Guo. “Ultrafine carbon black induces glutamate and atp release by activating connexin and pannexin hemichannels in cultured astrocytes.” Toxicology 323 (2014): 32-41. [CrossRef]
- Mugisho, O. O., C. R. Green, D. T. Kho, J. Zhang, E. S. Graham, M. L. Acosta and I. D. Rupenthal. “The inflammasome pathway is amplified and perpetuated in an autocrine manner through connexin43 hemichannel mediated atp release.” Biochimica et Biophysica Acta (BBA) - General Subjects 1862 (2018): 385-93. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



