Submitted:
01 November 2025
Posted:
04 November 2025
Read the latest preprint version here
Abstract
Keywords:
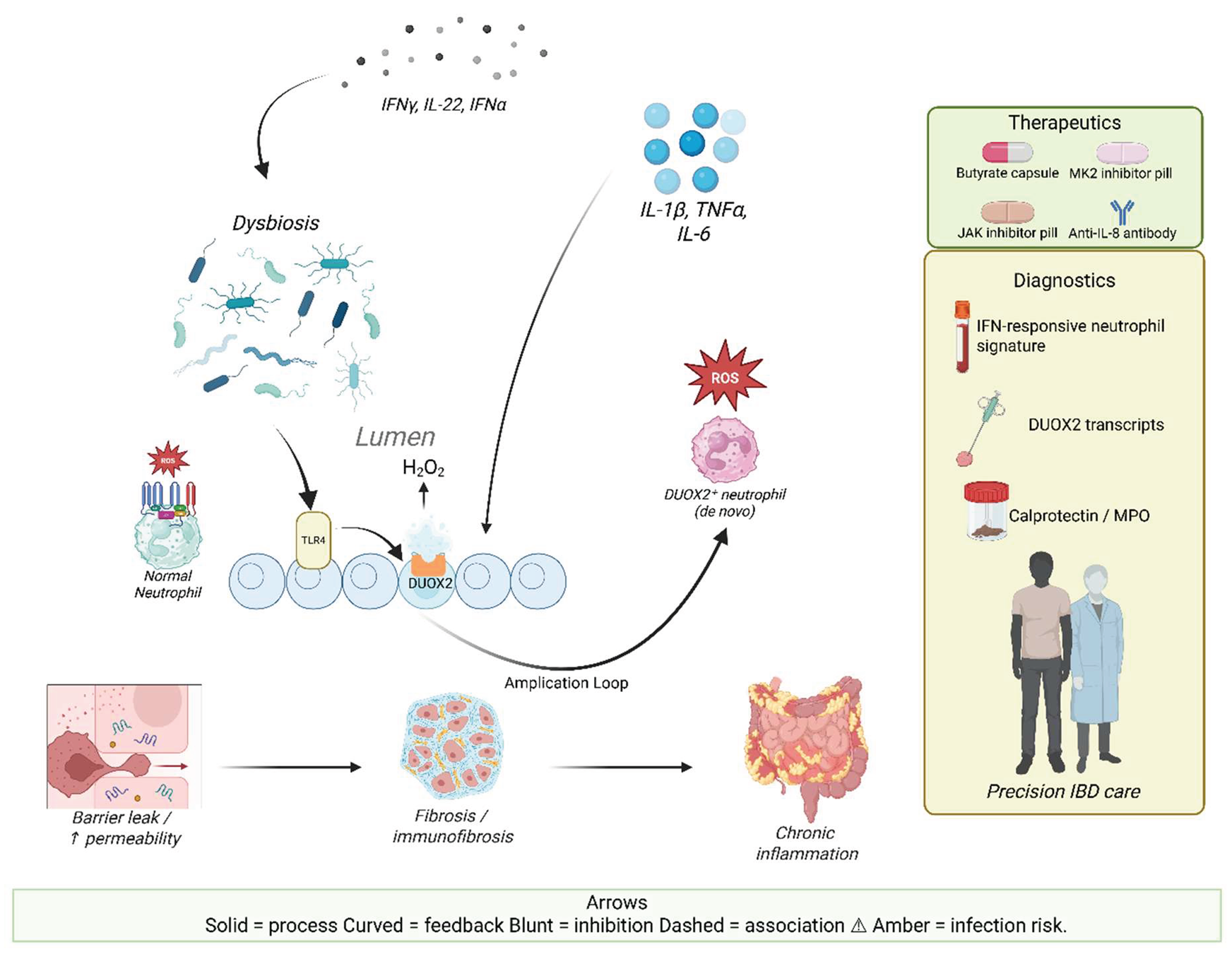
1. Introduction

2. Neutrophil plasticity in IBD and tissue contexts
2.1. Evidence for neutrophil heterogeneity and plasticity in IBD
| Neutrophil Subset | Key Markers / Traits | Context / Location | Functional Roles | References |
| Resting circulating neutrophils | CD16^+, CD62L^+, CXCR2^+ | Blood (homeostasis) | Baseline antimicrobial defense; short-lived; enter tissues upon chemokine signaling. | [24] |
| CD177^+ neutrophils | CD177^hi, CD66b^+, FcγRIIIb^+ | Inflamed intestinal mucosa in IBD | Enhanced chemotaxis and bactericidal activity; support barrier defense; also release pro-inflammatory mediators; correlate with IBD severity. | [25,26] |
| CD177^- neutrophils | CD177^-; otherwise phenotypically similar to CD177^+ | Blood, IBD mucosa | Lower recruitment compared to CD177^+; may have less effector potency; role still under investigation. | [27] |
| Low-density neutrophils (LDNs) | Low buoyant density; often immature; heterogeneous; markers vary | Autoimmunity, cancer, severe inflammation, IBD flares | Immunosuppressive (MDSC-like) or proinflammatory; can release ROS and NETs; expansion reported in active IBD. | [28,29] |
| Inflammation-primed neutrophils | Upregulated CXCR1/2, CD11b; downregulated CXCR4; increased activation markers | Inflamed tissue niches (gut mucosa in IBD) | Heightened effector functions (ROS, degranulation, NETs); strong recruitment cascades; IFN-priming can drive fibrotic plasticity in Crohn’s. | [30] |
| DUOX2^+ neutrophils | De novo DUOX2 expression (with NOX2); upregulated chemokines (CXCL1/2), cytokines (IL-1β, TNFα, IL-6) | Inflamed intestine (murine colitis, human IBD) | Expanded oxidative capacity; extracellular H₂O₂ production; amplify cytokine loops; sustain chronic mucosal inflammation and fibrosis. | [31] |
2.2. Microenvironmental cues that drive tissue reprogramming
2.3. Dichotomous effector programs: protection versus pathology
| Effector Mechanism | Protective Role in IBD | Pathogenic Role in IBD | References |
| Reactive Oxygen Species (ROS) | Potent bactericidal activity that limits microbial dissemination; essential for early host defense. | Excessive ROS disrupts epithelial tight junctions, damages DNA/proteins, and induces apoptosis of intestinal epithelial cells. | [36,37] |
| Neutrophil Extracellular Traps (NETs) | Trap and neutralize pathogens extracellularly, preventing microbial spread; can support wound repair and resolution. | Dysregulated NETosis increases gut permeability, induces IEC apoptosis, and destroys tight junctions, perpetuating inflammation. | [38,39] |
| Proteases (MMPs, elastase) | Release of lytic enzymes clears invading microbes and assists in matrix remodeling during repair. | Overproduction degrades epithelial adherens junctions, weakens barrier integrity, and contributes to mucosal injury. | [40,41] |
| Immune Cell Recruitment | Release of chemokines ensures rapid recruitment of immune cells, enabling pathogen clearance and resolution. | Excessive or chronic recruitment drives uncontrolled inflammation, amplifies cytokine cascades, and worsens epithelial damage. | [30,42] |
2.4. Disease-relevant subsets in the gut niche (including DUOX2+ and CD177+ states)
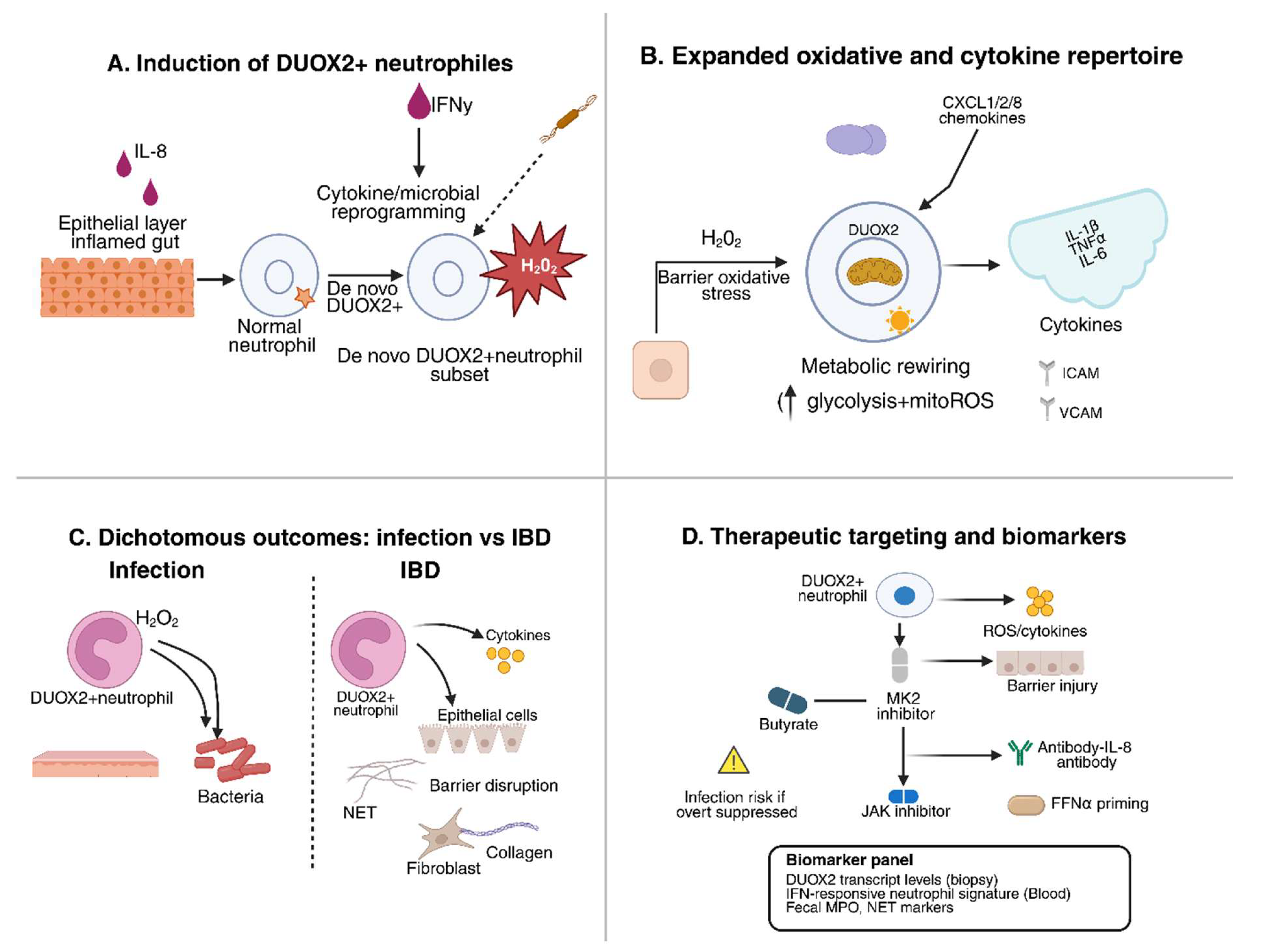
3. DUOX2 in IBD Pathogenesis
3.1. Regulation of epithelial DUOX2 expression
3.2. DUOX2 as a driver of dysbiosis and barrier dysfunction
3.3. Crosstalk between epithelial DUOX2 and immune-mediated pathology
4. De novo DUOX2 expression in neutrophils
4.1. Functional consequences of neutrophil DUOX2
4.2. Mechanistic insights: redox diversification
4.3. Therapeutic implications
5. Targeting neutrophils in IBD: therapeutic and diagnostic frontiers
5.1. Why current therapies leave a neutrophil-shaped gap
5.2. Drugging neutrophil pathways: from tractable targets to rational combinations
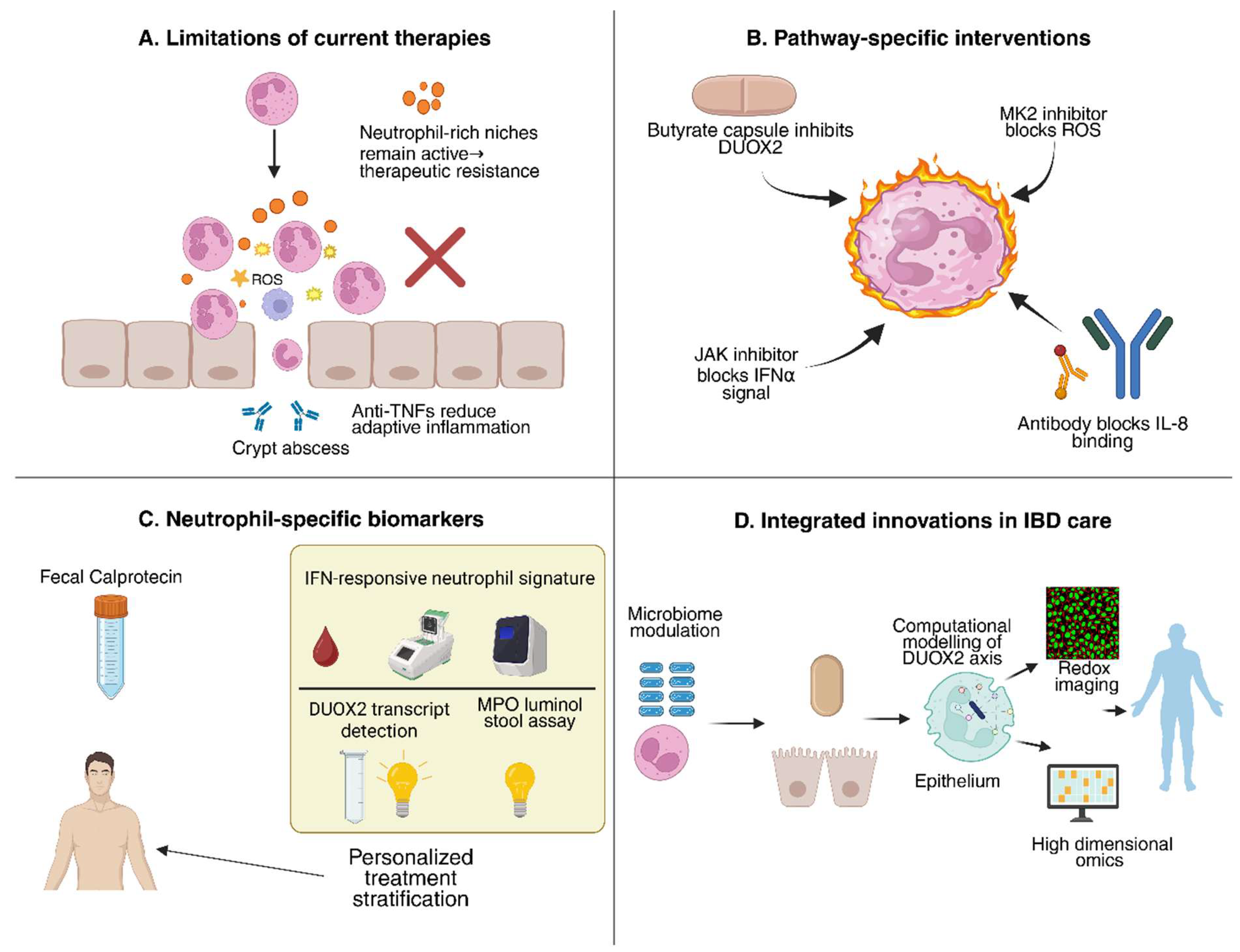
5.3. Diagnostics and monitoring: beyond calprotectin toward functional neutrophil readouts
5.4. Translational roadmap
6. Future Directions and Research Gaps
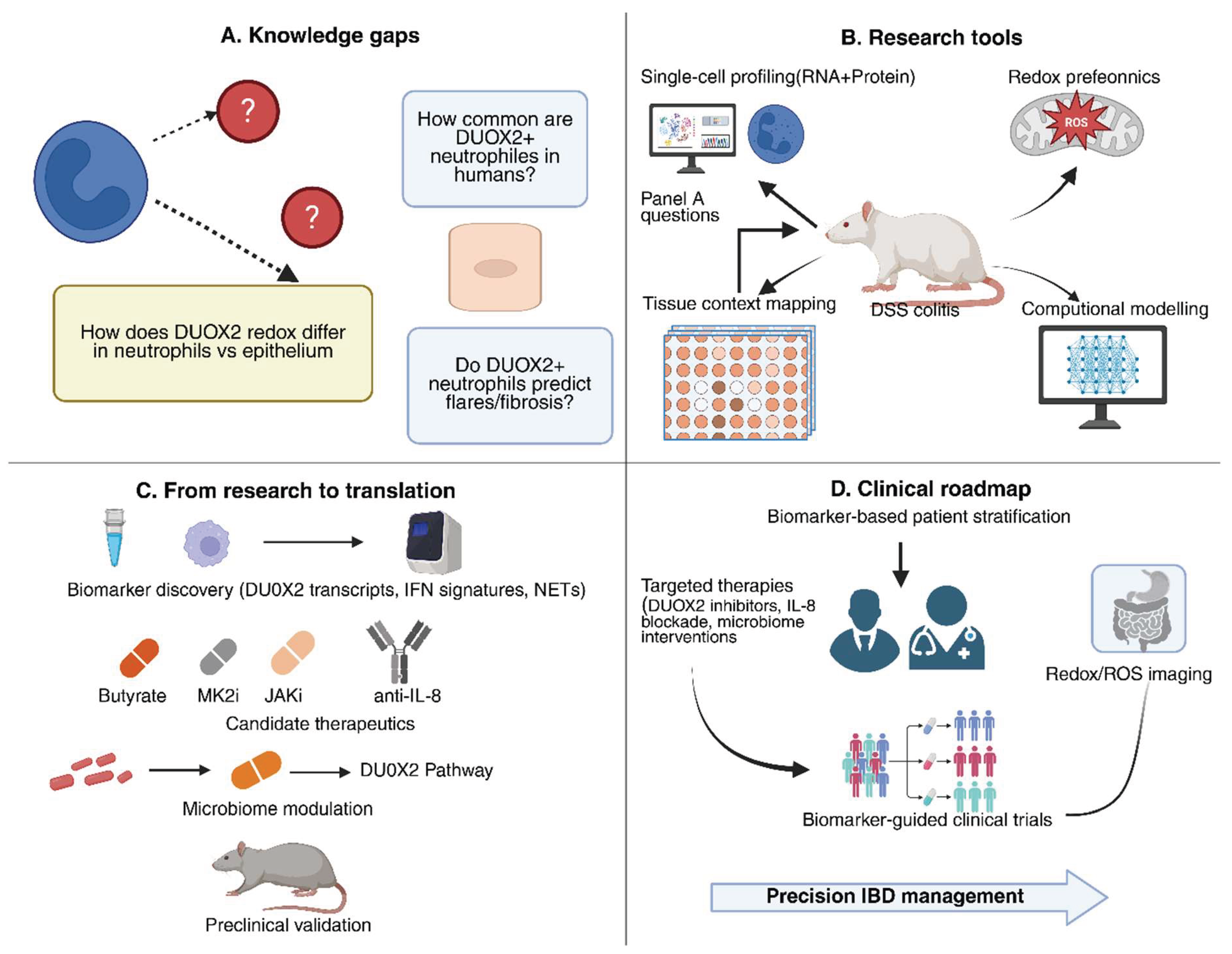
7. Conclusions
Funding
Biotechnology
Industrial Research
Authorship Contribution Statement
Conflicts of Interest
Declaration of generative AI and AI-assisted technologies in the writing process
Data and Materials Availability
References
- Kaplan GG, Windsor JW. The four epidemiological stages in the global evolution of inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2021 Jan;18(1):56–66.
- Neurath MF. Targeting immune cell circuits and trafficking in inflammatory bowel disease. Nat Immunol. 2019 Aug;20(8):970–9.
- Ordás I, Eckmann L, Talamini M, Baumgart DC, Sandborn WJ. Ulcerative colitis. Lancet. 2012 Nov 3;380(9853):1606–19.
- Liu JZ, van Sommeren S, Huang H, Ng SC, Alberts R, Takahashi A, Ripke S, Lee JC, Jostins L, Shah T, Abedian S, Cheon JH, Cho J, Dayani NE, Franke L, Fuyuno Y, Hart A, Juyal RC, Juyal G, Kim WH, Morris AP, Poustchi H, Newman WG, Midha V, Orchard TR, Vahedi H, Sood A, Sung JY, Malekzadeh R, Westra HJ, Yamazaki K, Yang SK, International Multiple Sclerosis Genetics Consortium, International IBD Genetics Consortium, Barrett JC, Alizadeh BZ, Parkes M, Bk T, Daly MJ, Kubo M, Anderson CA, Weersma RK. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat Genet. 2015 Sep;47(9):979–86.
- Jostins L, Ripke S, Weersma RK, Duerr RH, McGovern DP, Hui KY, Lee JC, Schumm LP, Sharma Y, Anderson CA, Essers J, Mitrovic M, Ning K, Cleynen I, Theatre E, Spain SL, Raychaudhuri S, Goyette P, Wei Z, Abraham C, Achkar JP, Ahmad T, Amininejad L, Ananthakrishnan AN, Andersen V, Andrews JM, Baidoo L, Balschun T, Bampton PA, Bitton A, Boucher G, Brand S, Büning C, Cohain A, Cichon S, D’Amato M, De Jong D, Devaney KL, Dubinsky M, Edwards C, Ellinghaus D, Ferguson LR, Franchimont D, Fransen K, Gearry R, Georges M, Gieger C, Glas J, Haritunians T, Hart A, Hawkey C, Hedl M, Hu X, Karlsen TH, Kupcinskas L, Kugathasan S, Latiano A, Laukens D, Lawrance IC, Lees CW, Louis E, Mahy G, Mansfield J, Morgan AR, Mowat C, Newman W, Palmieri O, Ponsioen CY, Potocnik U, Prescott NJ, Regueiro M, Rotter JI, Russell RK, Sanderson JD, Sans M, Satsangi J, Schreiber S, Simms LA, Sventoraityte J, Targan SR, Taylor KD, Tremelling M, Verspaget HW, De Vos M, Wijmenga C, Wilson DC, Winkelmann J, Xavier RJ, Zeissig S, Zhang B, Zhang CK, Zhao H, International IBD Genetics Consortium (IIBDGC), Silverberg MS, Annese V, Hakonarson H, Brant SR, Radford-Smith G, Mathew CG, Rioux JD, Schadt EE, Daly MJ, Franke A, Parkes M, Vermeire S, Barrett JC, Cho JH. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature. 2012 Nov 1;491(7422):119–24.
- Nishida A, Inoue R, Inatomi O, Bamba S, Naito Y, Andoh A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin J Gastroenterol. 2018 Feb;11(1):1–10.
- Mitsialis V, Wall S, Liu P, Ordovas-Montanes J, Parmet T, Vukovic M, Spencer D, Field M, McCourt C, Toothaker J, Bousvaros A, Boston Children’s Hospital Inflammatory Bowel Disease Center, Brigham and Women’s Hospital Crohn’s and Colitis Center, Shalek AK, Kean L, Horwitz B, Goldsmith J, Tseng G, Snapper SB, Konnikova L. Single-Cell Analyses of Colon and Blood Reveal Distinct Immune Cell Signatures of Ulcerative Colitis and Crohn’s Disease. Gastroenterology. 2020 Aug;159(2):591-608.e10.
- Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013 Mar;13(3):159–75.
- Sidler MA, Leach ST, Day AS. Fecal S100A12 and fecal calprotectin as noninvasive markers for inflammatory bowel disease in children. Inflamm Bowel Dis. 2008 Mar;14(3):359–66.
- Ishida N, Onoue S, Takebe T, Takahashi K, Asai Y, Tamura S, Matsuura T, Yamade M, Iwaizumi M, Hamaya Y, Yamada T, Osawa S, Sugimoto K. Fecal Calprotectin as a Biomarker of Crohn’s Disease in Patients With Short Disease Durations: A Prospective, Single-Center, Cross-Sectional Study. [cited 2025 Sep 4]; Available from: https://onlinelibrary.wiley.com/doi/10.1155/grp/9984055.
- Zhou G, Yu L, Fang L, Yang W, Yu T, Miao Y, Chen M, Wu K, Chen F, Cong Y, Liu Z. CD177+ neutrophils as functionally activated neutrophils negatively regulate IBD. Gut. 2018 Jun;67(6):1052–63.
- Xie X, Shi Q, Wu P, Zhang X, Kambara H, Su J, Yu H, Park SY, Guo R, Ren Q, Zhang S, Xu Y, Silberstein LE, Cheng T, Ma F, Li C, Luo HR. Single-cell transcriptome profiling reveals neutrophil heterogeneity in homeostasis and infection. Nat Immunol. 2020 Sep;21(9):1119–33.
- Smillie CS, Biton M, Ordovas-Montanes J, Sullivan KM, Burgin G, Graham DB, Herbst RH, Rogel N, Slyper M, Waldman J, Sud M, Andrews E, Velonias G, Haber AL, Jagadeesh K, Vickovic S, Yao J, Stevens C, Dionne D, Nguyen LT, Villani AC, Hofree M, Creasey EA, Huang H, Rozenblatt-Rosen O, Garber JJ, Khalili H, Desch AN, Daly MJ, Ananthakrishnan AN, Shalek AK, Xavier RJ, Regev A. Intra- and Inter-cellular Rewiring of the Human Colon during Ulcerative Colitis. Cell. 2019 Jul 25;178(3):714-730.e22.
- Donkó A, Péterfi Z, Sum A, Leto T, Geiszt M. Dual oxidases. Philos Trans R Soc Lond B Biol Sci. 2005 Dec 29;360(1464):2301–8.
- Grasberger H, Gao J, Nagao-Kitamoto H, Kitamoto S, Zhang M, Kamada N, Eaton KA, El-Zaatari M, Shreiner AB, Merchant JL, Owyang C, Kao JY. Increased Expression of DUOX2 Is an Epithelial Response to Mucosal Dysbiosis Required for Immune Homeostasis in Mouse Intestine. Gastroenterology. 2015 Dec;149(7):1849–59.
- Aguilar EC, Santos LCD, Leonel AJ, de Oliveira JS, Santos EA, Navia-Pelaez JM, da Silva JF, Mendes BP, Capettini LSA, Teixeira LG, Lemos VS, Alvarez-Leite JI. Oral butyrate reduces oxidative stress in atherosclerotic lesion sites by a mechanism involving NADPH oxidase down-regulation in endothelial cells. J Nutr Biochem. 2016 Aug;34:99–105.
- Wen X, Tang L, Zhong R, Liu L, Chen L, Zhang H. Role of Mitophagy in Regulating Intestinal Oxidative Damage. Antioxidants (Basel). 2023 Feb 14;12(2):480.
- Haque PS, Kapur N, Barrett TA, Theiss AL. Mitochondrial function and gastrointestinal diseases. Nat Rev Gastroenterol Hepatol. 2024 Aug;21(8):537–55.
- Filep JG. Targeting Neutrophils for Promoting the Resolution of Inflammation. Front Immunol [Internet]. 2022 Mar 16 [cited 2025 Sep 13];13. Available from: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.866747/full.
- Byun MJ, Nakasone ES, Shin HE, Lee H, Park JC, Lee W, Park W, Park CG, Park J, Kim SN. Advanced Nanoparticle Therapeutics for Targeting Neutrophils in Inflammatory Diseases. Adv Healthc Mater. 2025 Jul 22;e2502092.
- Wigerblad G, Cao Q, Brooks S, Naz F, Gadkari M, Jiang K, Gupta S, O’Neil L, Dell’Orso S, Kaplan MJ, Franco LM. Single-Cell Analysis Reveals the Range of Transcriptional States of Circulating Human Neutrophils. J Immunol. 2022 Aug 15;209(4):772–82.
- Garrido-Trigo A, Corraliza AM, Veny M, Dotti I, Melón-Ardanaz E, Rill A, Crowell HL, Corbí Á, Gudiño V, Esteller M, Álvarez-Teubel I, Aguilar D, Masamunt MC, Killingbeck E, Kim Y, Leon M, Visvanathan S, Marchese D, Caratù G, Martin-Cardona A, Esteve M, Ordás I, Panés J, Ricart E, Mereu E, Heyn H, Salas A. Macrophage and neutrophil heterogeneity at single-cell spatial resolution in human inflammatory bowel disease. Nat Commun. 2023 Jul 26;14(1):4506.
- Friedrich M, Pohin M, Jackson MA, Korsunsky I, Bullers SJ, Rue-Albrecht K, Christoforidou Z, Sathananthan D, Thomas T, Ravindran R, Tandon R, Peres RS, Sharpe H, Wei K, Watts GFM, Mann EH, Geremia A, Attar M, Oxford IBD Cohort Investigators, Roche Fibroblast Network Consortium, McCuaig S, Thomas L, Collantes E, Uhlig HH, Sansom SN, Easton A, Raychaudhuri S, Travis SP, Powrie FM. IL-1-driven stromal-neutrophil interactions define a subset of patients with inflammatory bowel disease that does not respond to therapies. Nat Med. 2021 Nov;27(11):1970–81.
- Summers C, Rankin SM, Condliffe AM, Singh N, Peters AM, Chilvers ER. Neutrophil kinetics in health and disease. Trends Immunol. 2010 Aug;31(8):318–24.
- Xu H, Zhan M, Wu Z, Chen J, Zhao Y, Feng F, Wang F, Li Y, Zhang S, Liu Y. Aberrant expansion of CD177+ neutrophils promotes endothelial dysfunction in systemic lupus erythematosus via neutrophil extracellular traps. J Autoimmun. 2025 Mar;152:103399.
- Bai M, Grieshaber-Bouyer R, Wang J, Schmider AB, Wilson ZS, Zeng L, Halyabar O, Godin MD, Nguyen HN, Levescot A, Cunin P, Lefort CT, Soberman RJ, Nigrovic PA. CD177 modulates human neutrophil migration through activation-mediated integrin and chemoreceptor regulation. Blood. 2017 Nov 9;130(19):2092–100.
- von Vietinghoff S, Tunnemann G, Eulenberg C, Wellner M, Cristina Cardoso M, Luft FC, Kettritz R. NB1 mediates surface expression of the ANCA antigen proteinase 3 on human neutrophils. Blood. 2007 May 15;109(10):4487–9.
- Hacbarth E, Kajdacsy-Balla A. Low density neutrophils in patients with systemic lupus erythematosus, rheumatoid arthritis, and acute rheumatic fever. Arthritis Rheum. 1986 Nov;29(11):1334–42.
- Blanco-Camarillo C, Alemán OR, Rosales C. Low-Density Neutrophils in Healthy Individuals Display a Mature Primed Phenotype. Front Immunol. 2021;12:672520.
- Neutrophils in cancer: heterogeneous and multifaceted | Request PDF. ResearchGate [Internet]. [cited 2025 Sep 14]; Available from: https://www.researchgate.net/publication/353047575_Neutrophils_in_cancer_heterogeneous_and_multifaceted.
- Singh AK, Ainciburu M, Wynne K, Bhat SA, Blanco A, Tzani I, Akiba Y, Lalor SJ, Kaunitz J, Bourke B, Kelly VP, Doherty GA, Zerbe CS, Clarke C, Hussey S, Knaus UG. De novo DUOX2 expression in neutrophil subsets shapes the pathogenesis of intestinal disease. Proc Natl Acad Sci U S A. 2025 May 13;122(19):e2421747122.
- Gavriilidis E, Divolis G, Natsi AM, Kafalis N, Kogias D, Antoniadou C, Synolaki E, Pavlos E, Koutsi MA, Didaskalou S, Papadimitriou E, Tsironidou V, Gavriil A, Papadopoulos V, Agelopoulos M, Tsilingiris D, Koffa M, Giatromanolaki A, Kouklakis G, Ritis K, Skendros P. Neutrophil-fibroblast crosstalk drives immunofibrosis in Crohn’s disease through IFNα pathway. Front Immunol. 2024 Sep 13;15:1447608.
- Li G, Lin J, Zhang C, Gao H, Lu H, Gao X, Zhu R, Li Z, Li M, Liu Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes. 2021;13(1):1968257.
- De Filippo K, Rankin SM. CXCR4, the master regulator of neutrophil trafficking in homeostasis and disease. Eur J Clin Invest. 2018 Nov;48 Suppl 2(Suppl Suppl 2):e12949.
- Chen F, Liu Y, Shi Y, Zhang J, Liu X, Liu Z, Lv J, Leng Y. The emerging role of neutrophilic extracellular traps in intestinal disease. Gut Pathog. 2022 Jun 22;14(1):27.
- Segal, AW. How neutrophils kill microbes. Annu Rev Immunol. 2005;23:197–223.
- Aviello G, Knaus UG. NADPH oxidases and ROS signaling in the gastrointestinal tract. Mucosal Immunol. 2018 Jul;11(4):1011–23.
- Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, Weinrauch Y, Zychlinsky A. Neutrophil extracellular traps kill bacteria. Science. 2004 Mar 5;303(5663):1532–5.
- Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol. 2012 Sep 15;189(6):2689–95.
- Pham CTN. Neutrophil serine proteases: specific regulators of inflammation. Nat Rev Immunol. 2006 Jul;6(7):541–50.
- Kabouridis PS, Lasrado R, McCallum S, Chng SH, Snippert HJ, Clevers H, Pettersson S, Pachnis V. The gut microbiota keeps enteric glial cells on the move; prospective roles of the gut epithelium and immune system. Gut Microbes. 2015 Nov 11;6(6):398–403.
- Sadik CD, Kim ND, Luster AD. Neutrophils cascading their way to inflammation. Trends Immunol. 2011 Oct;32(10):452–60.
- Zhu N, Zhu J, Mei Q. Low-density Granulocytes as a Novel Biomarkers of Disease Activity in IBD. Inflammatory Bowel Diseases. 2023 Aug 1;29(8):e31–e31.
- Delaveris C, Wilk A, Riley N, Stark J, Yang S, Rogers A, Ranganath T, Nadeau K, Blish C, Bertozzi C. Synthetic Siglec-9 Agonists Inhibit Neutrophil Activation Associated with COVID-19 [Internet]. Chemistry; 2020 [cited 2025 Sep 14]. Available from: https://chemrxiv.org/engage/chemrxiv/article-details/60c75305469df463f7f44ca1.
- Sonnenberg GF, Fouser LA, Artis D. Functional biology of the IL-22-IL-22R pathway in regulating immunity and inflammation at barrier surfaces. Adv Immunol. 2010;107:1–29.
- Schroder K, Hertzog PJ, Ravasi T, Hume DA. Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol. 2004 Feb;75(2):163–89.
- Ha EM, Oh CT, Bae YS, Lee WJ. A direct role for dual oxidase in Drosophila gut immunity. Science. 2005 Nov 4;310(5749):847–50.
- Grasberger H, Magis AT, Sheng E, Conomos MP, Zhang M, Garzotto LS, Hou G, Bishu S, Nagao-Kitamoto H, El-Zaatari M, Kitamoto S, Kamada N, Stidham RW, Akiba Y, Kaunitz J, Haberman Y, Kugathasan S, Denson LA, Omenn GS, Kao JY. DUOX2 variants associate with preclinical disturbances in microbiota-immune homeostasis and increased inflammatory bowel disease risk. J Clin Invest. 2021 May 3;131(9):e141676, 141676.
- Smith PD, Smythies LE, Shen R, Greenwell-Wild T, Gliozzi M, Wahl SM. Intestinal macrophages and response to microbial encroachment. Mucosal Immunol. 2011 Jan;4(1):31–42.
- Sun X, Chalmers L, Fu X, Zhao M. A Molecular Link Between Interleukin 22 and Intestinal Mucosal Wound Healing. Adv Wound Care (New Rochelle). 2012 Dec 1;1(6):231–7.
- Lamas B, Natividad JM, Sokol H. Aryl hydrocarbon receptor and intestinal immunity. Mucosal Immunol. 2018 Jul;11(4):1024–38.
- Small CL, Xing L, McPhee JB, Law HT, Coombes BK. Acute Infectious Gastroenteritis Potentiates a Crohn’s Disease Pathobiont to Fuel Ongoing Inflammation in the Post-Infectious Period. PLOS Pathogens. 2016 Oct 6;12(10):e1005907.
- Ragland SA, Criss AK. From bacterial killing to immune modulation: Recent insights into the functions of lysozyme. PLoS Pathog. 2017 Sep;13(9):e1006512.
- Bots S, Nylund K, Löwenberg M, Gecse K, D’Haens G. Intestinal Ultrasound to Assess Disease Activity in Ulcerative Colitis: Development of a novel UC-Ultrasound Index. J Crohns Colitis. 2021 Aug 2;15(8):1264–71.
- Shang J, Ding M, Zhou X. Recent advances in S-palmitoylation and its emerging roles in human diseases. J Hematol Oncol. 2025 Sep 1;18:83.
- Canani RB, Costanzo MD, Leone L, Pedata M, Meli R, Calignano A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J Gastroenterol. 2011 Mar 28;17(12):1519–28.
- Davie JR. Inhibition of histone deacetylase activity by butyrate. J Nutr. 2003 Jul;133(7 Suppl):2485S-2493S.
- Jarade A, Garcia Z, Marie S, Demera A, Prinz I, Bousso P, Di Santo JP, Serafini N. Inflammation triggers ILC3 patrolling of the intestinal barrier. Nat Immunol. 2022;23(9):1317–23.
- Ostrowski J, Dabrowska M, Lazowska I, Paziewska A, Balabas A, Kluska A, Kulecka M, Karczmarski J, Ambrozkiewicz F, Piatkowska M, Goryca K, Zeber-Lubecka N, Kierkus J, Socha P, Lodyga M, Klopocka M, Iwanczak B, Bak-Drabik K, Walkowiak J, Radwan P, Grzybowska-Chlebowczyk U, Korczowski B, Starzynska T, Mikula M. Redefining the Practical Utility of Blood Transcriptome Biomarkers in Inflammatory Bowel Diseases. J Crohns Colitis. 2019 Apr 26;13(5):626–33.
- Hegazy AN, West NR, Stubbington MJT, Wendt E, Suijker KIM, Datsi A, This S, Danne C, Campion S, Duncan SH, Owens BMJ, Uhlig HH, McMichael A, Oxford IBD Cohort Investigators, Bergthaler A, Teichmann SA, Keshav S, Powrie F. Circulating and Tissue-Resident CD4+ T Cells With Reactivity to Intestinal Microbiota Are Abundant in Healthy Individuals and Function Is Altered During Inflammation. Gastroenterology. 2017 Nov;153(5):1320-1337.e16.
- Li JY, Xiao J, Gao M, Zhou HF, Fan H, Sun F, Cui DD. IRF/Type I IFN signaling serves as a valuable therapeutic target in the pathogenesis of inflammatory bowel disease. International Immunopharmacology. 2021 Mar 1;92:107350.
- Westbrook AM, Szakmary A, Schiestl RH. Mechanisms of intestinal inflammation and development of associated cancers: lessons learned from mouse models. Mutat Res. 2010;705(1):40–59.
- Parlato M, Charbit-Henrion F, Hayes P, Tiberti A, Aloi M, Cucchiara S, Bègue B, Bras M, Pouliet A, Rakotobe S, Ruemmele F, Knaus UG, Cerf-Bensussan N. First Identification of Biallelic Inherited DUOX2 Inactivating Mutations as a Cause of Very Early Onset Inflammatory Bowel Disease. Gastroenterology. 2017 Aug;153(2):609-611.e3.
- Altenhöfer S, Radermacher KA, Kleikers PWM, Wingler K, Schmidt HHHW. Evolution of NADPH Oxidase Inhibitors: Selectivity and Mechanisms for Target Engagement. Antioxid Redox Signal. 2015 Aug 10;23(5):406–27.
- Donkó A, Péterfi Z, Sum A, Leto T, Geiszt M. Dual oxidases. Philos Trans R Soc Lond B Biol Sci. 2005 Dec 29;360(1464):2301–8.
- Geiszt M, Witta J, Baffi J, Lekstrom K, Leto TL. Dual oxidases represent novel hydrogen peroxide sources supporting mucosal surface host defense. FASEB J. 2003 Aug;17(11):1502–4.
- Staicu I. Neutrophil subsets. Nat Immunol. 2024 Apr;25(4):583–583.
- DUOX2 activation drives bacterial translocation and subclinical inflammation in IBD-associated dysbiosis | Gut [Internet]. [cited 2025 Sep 13]. Available from: https://gut.bmj.com/content/74/10/1589.
- Ortega-Zapero M, Gomez-Bris R, Pascual-Laguna I, Saez A, Gonzalez-Granado JM. Neutrophils and NETs in Pathophysiology and Treatment of Inflammatory Bowel Disease. Int J Mol Sci. 2025 Jul 23;26(15):7098.
- Zaiatz Bittencourt V, Jones F, Doherty G, Ryan EJ. Targeting Immune Cell Metabolism in the Treatment of Inflammatory Bowel Disease. Inflamm Bowel Dis. 2021 Mar 10;27(10):1684–93.
- Taman H, Fenton CG, Hensel IV, Anderssen E, Florholmen J, Paulssen RH. Transcriptomic Landscape of Treatment-Naïve Ulcerative Colitis. J Crohns Colitis. 2018 Feb 28;12(3):327–36.
- Nowak JK, Adams AT, Kalla R, Lindstrøm JC, Vatn S, Bergemalm D, Keita ÅV, Gomollón F, Jahnsen J, Vatn MH, Ricanek P, Ostrowski J, Walkowiak J, Halfvarson J, Satsangi J, IBD Character Consortium. Characterisation of the Circulating Transcriptomic Landscape in Inflammatory Bowel Disease Provides Evidence for Dysregulation of Multiple Transcription Factors Including NFE2, SPI1, CEBPB, and IRF2. J Crohns Colitis. 2022 Aug 1;16(8):1255–68.
- Grasberger H, Magis AT, Sheng E, Conomos MP, Zhang M, Garzotto LS, Hou G, Bishu S, Nagao-Kitamoto H, El-Zaatari M, Kitamoto S, Kamada N, Stidham RW, Akiba Y, Kaunitz J, Haberman Y, Kugathasan S, Denson LA, Omenn GS, Kao JY. DUOX2 variants associate with preclinical disturbances in microbiota-immune homeostasis and increased inflammatory bowel disease risk. J Clin Invest. 2021 May 3;131(9):e141676, 141676.
- Ota S, Sakuraba H. Uptake and Advanced Therapy of Butyrate in Inflammatory Bowel Disease. Immuno. 2022 Dec;2(4):692–702.
- Shin Y, Han S, Kwon J, Ju S, Choi TG, Kang I, Kim SS. Roles of Short-Chain Fatty Acids in Inflammatory Bowel Disease. Nutrients. 2023 Oct 21;15(20):4466.
- Conder E, Shay HC, Vekaria H, Erinkitola I, Bhogoju S, Goretsky T, Sullivan P, Barrett T, Kapur N. BUTYRATE-INDUCED MITOCHONDRIAL FUNCTION IMPROVES BARRIER FUNCTION IN INFLAMMATORY BOWEL DISEASE (IBD). Inflamm Bowel Dis. 2023 Feb 1;29(Supplement_1):S71–2.
- Sandborn, WJ. Are short-chain fatty acid enemas effective for left-sided ulcerative colitis? Gastroenterology. 1998 Jan;114(1):218–9.
- Sandborn WJ, Su C, Sands BE, D’Haens GR, Vermeire S, Schreiber S, Danese S, Feagan BG, Reinisch W, Niezychowski W, Friedman G, Lawendy N, Yu D, Woodworth D, Mukherjee A, Zhang H, Healey P, Panés J, OCTAVE Induction 1, OCTAVE Induction 2, and OCTAVE Sustain Investigators. Tofacitinib as Induction and Maintenance Therapy for Ulcerative Colitis. N Engl J Med. 2017 May 4;376(18):1723–36.
- Zhang X, Guo R, Kambara H, Ma F, Luo HR. The role of CXCR2 in acute inflammatory responses and its antagonists as anti-inflammatory therapeutics. Curr Opin Hematol. 2019 Jan;26(1):28–33.
- Sitaru S, Budke A, Bertini R, Sperandio M. Therapeutic inhibition of CXCR1/2: where do we stand? Intern Emerg Med. 2023 May 30;1–18.
- Sun L, Wu Q, Nie Y, Cheng N, Wang R, Wang G, Zhang D, He H, Ye RD, Qian F. A Role for MK2 in Enhancing Neutrophil-Derived ROS Production and Aggravating Liver Ischemia/Reperfusion Injury. Front Immunol. 2018;9:2610.
- Zhang T, Jiang J, Liu J, Xu L, Duan S, Sun L, Zhao W, Qian F. MK2 Is Required for Neutrophil-Derived ROS Production and Inflammatory Bowel Disease. Front Med (Lausanne). 2020;7:207.
- Tam JSY, Coller JK, Hughes PA, Prestidge CA, Bowen JM. Toll-like receptor 4 (TLR4) antagonists as potential therapeutics for intestinal inflammation. Indian J Gastroenterol. 2021 Feb 1;40(1):5–21.
- Kuzmich NN, Sivak KV, Chubarev VN, Porozov YB, Savateeva-Lyubimova TN, Peri F. TLR4 Signaling Pathway Modulators as Potential Therapeutics in Inflammation and Sepsis. Vaccines (Basel). 2017 Oct 4;5(4):34.
- Vaure C, Liu Y. A comparative review of toll-like receptor 4 expression and functionality in different animal species. Front Immunol. 2014;5:316.
- Paramsothy S, Kamm MA, Kaakoush NO, Walsh AJ, van den Bogaerde J, Samuel D, Leong RWL, Connor S, Ng W, Paramsothy R, Xuan W, Lin E, Mitchell HM, Borody TJ. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: a randomised placebo-controlled trial. Lancet. 2017 Mar 25;389(10075):1218–28.
- Nguyen GT, Green ER, Mecsas J. Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance. Front Cell Infect Microbiol. 2017;7:373.
- Castrillón-Betancur JC, López-Agudelo VA, Sommer N, Cleeves S, Bernardes JP, Weber-Stiehl S, Rosenstiel P, Sommer F. Epithelial Dual Oxidase 2 Shapes the Mucosal Microbiome and Contributes to Inflammatory Susceptibility. Antioxidants (Basel). 2023 Oct 21;12(10):1889.
- Sommer F, Bäckhed F. The gut microbiota engages different signaling pathways to induce Duox2 expression in the ileum and colon epithelium. Mucosal Immunol. 2015 Mar;8(2):372–9.
- Bento AF, Leite DFP, Claudino RF, Hara DB, Leal PC, Calixto JB. The selective nonpeptide CXCR2 antagonist SB225002 ameliorates acute experimental colitis in mice. J Leukoc Biol. 2008 Oct;84(4):1213–21.
- Zhu F, He H, Fan L, Ma C, Xu Z, Xue Y, Wang Y, Zhang C, Zhou G. Blockade of CXCR2 suppresses proinflammatory activities of neutrophils in ulcerative colitis. Am J Transl Res. 2020;12(9):5237–51.
- Xie Y, Kuang W, Wang D, Yuan K, Yang P. Expanding role of CXCR2 and therapeutic potential of CXCR2 antagonists in inflammatory diseases and cancers. European Journal of Medicinal Chemistry. 2023 Mar 15;250:115175.
- Li JY, Xiao J, Gao M, Zhou HF, Fan H, Sun F, Cui DD. IRF/Type I IFN signaling serves as a valuable therapeutic target in the pathogenesis of inflammatory bowel disease. International Immunopharmacology. 2021 Mar 1;92:107350.
- Loftus EV, Panés J, Lacerda AP, Peyrin-Biroulet L, D’Haens G, Panaccione R, Reinisch W, Louis E, Chen M, Nakase H, Begun J, Boland BS, Phillips C, Mohamed MEF, Liu J, Geng Z, Feng T, Dubcenco E, Colombel JF. Upadacitinib Induction and Maintenance Therapy for Crohn’s Disease. N Engl J Med. 2023 May 25;388(21):1966–80.
- A E, M F, F P. IL-1-mediated remodelling of the stromal-neutrophil landscape is associated with non-response to therapy in a subset of inflammatory bowel disease patients. Cell [Internet]. 2020 Jul 3 [cited 2025 Sep 13]; Available from: https://www.oncology.ox.ac.uk/publications/1116098.
- (PDF) IL-1-driven stromal-neutrophil interaction in deep ulcers defines a pathotype of therapy non-responsive inflammatory bowel disease [Internet]. ResearchGate. [cited 2025 Sep 13]. Available from: https://www.researchgate.net/publication/349100219_IL-1-driven_stromal-neutrophil_interaction_in_deep_ulcers_defines_a_pathotype_of_therapy_non-responsive_inflammatory_bowel_disease.
- Ahmad G, Chami B, Liu Y, Schroder AL, San Gabriel PT, Gao A, Fong G, Wang X, Witting PK. The Synthetic Myeloperoxidase Inhibitor AZD3241 Ameliorates Dextran Sodium Sulfate Stimulated Experimental Colitis. Front Pharmacol. 2020;11:556020.
- Xie K, Hunter J, Lee A, Ahmad G, Witting PK, Ortiz-Cerda T. The PAD4 inhibitor GSK484 diminishes neutrophil extracellular trap in the colon mucosa but fails to improve inflammatory biomarkers in experimental colitis. Biosci Rep. 2025 May 20;45(6):375–97.
- Mehandru S, Colombel JF, Juarez J, Bugni J, Lindsay JO. Understanding the molecular mechanisms of anti-trafficking therapies and their clinical relevance in inflammatory bowel disease. Mucosal Immunology. 2023 Dec;16(6):859–70.
- Soler D, Chapman T, Yang LL, Wyant T, Egan R, Fedyk ER. The binding specificity and selective antagonism of vedolizumab, an anti-alpha4beta7 integrin therapeutic antibody in development for inflammatory bowel diseases. J Pharmacol Exp Ther. 2009 Sep;330(3):864–75.
- Roosenboom B, Lochem EG van, Meijer J, Smids C, Nierkens S, Brand EC, Erp LW van, Kemperman LGJM, Groenen MJM, Horje CSHT, Wahab PJ. Development of Mucosal PNAd+ and MAdCAM-1+ Venules during Disease Course in Ulcerative Colitis. Cells. 2020 Apr 6;9(4):891.
- Facchin S, Vitulo N, Calgaro M, Buda A, Romualdi C, Pohl D, Perini B, Lorenzon G, Marinelli C, D’Incà R, Sturniolo GC, Savarino EV. Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel disease. Neurogastroenterol Motil. 2020 Oct;32(10):e13914.
- Hodgkinson K, El Abbar F, Dobranowski P, Manoogian J, Butcher J, Figeys D, Mack D, Stintzi A. Butyrate’s role in human health and the current progress towards its clinical application to treat gastrointestinal disease. Clin Nutr. 2023 Feb;42(2):61–75.
- Chiang HS. PAD4-dependent neutrophil extracellular traps dagame intestinal barrier integrity through histone protein components. J Immunol. 2023 May 1;210(Supplement_1):61.13.
- Wang P, Liu D, Zhou Z, Liu F, Shen Y, You Q, Lu S, Wu J. The role of protein arginine deiminase 4-dependent neutrophil extracellular traps formation in ulcerative colitis. Front Immunol. 2023;14:1144976.
- Long D, Mao C, Xu Y, Zhu Y. The emerging role of neutrophil extracellular traps in ulcerative colitis. Front Immunol [Internet]. 2024 Aug 7 [cited 2025 Sep 13];15. Available from: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1425251/full.
- Lutz M, Horst S, Caldera F. Applying Biomarkers in Treat-to-target Approach for IBD. Curr Gastroenterol Rep. 2025;27(1):41.
- Liu F, Lee SA, Riordan SM, Zhang L, Zhu L. Global Studies of Using Fecal Biomarkers in Predicting Relapse in Inflammatory Bowel Disease. Front Med (Lausanne). 2020;7:580803.
- Sun T, Wang P, Zhai X, Wang Z, Miao X, Yang Y, Wu J. Neutrophil extracellular traps induce barrier dysfunction in DSS-induced ulcerative colitis via the cGAS-STING pathway. International Immunopharmacology. 2024 Dec 25;143:113358.
- Dmochowska N, Tieu W, Keller MD, Wardill HR, Mavrangelos C, Campaniello MA, Takhar P, Hughes PA. Immuno-PET of Innate Immune Markers CD11b and IL-1β Detects Inflammation in Murine Colitis. J Nucl Med. 2019 Jun;60(6):858–63.
- Bettenworth D, Reuter S, Hermann S, Weckesser M, Kerstiens L, Stratis A, Nowacki TM, Ross M, Lenze F, Edemir B, Maaser C, Pap T, Koschmieder S, Heidemann J, Schäfers M, Lügering A. Translational 18F-FDG PET/CT imaging to monitor lesion activity in intestinal inflammation. J Nucl Med. 2013 May;54(5):748–55.
- Rezazadeh F, Kilcline AP, Viola NT. Imaging Agents for PET of Inflammatory Bowel Disease: A Review. J Nucl Med. 2023 Dec 1;64(12):1858–64.
- Pickett JR, Wu Y, Ta HT. VCAM-1 as a common biomarker in inflammatory bowel disease and colorectal cancer: unveiling the dual anti-inflammatory and anti-cancer capacities of anti-VCAM-1 therapies. Cancer Metastasis Rev. 2025 Mar 17;44(2):40.
- Ley K, Burns C. Adhesion molecules in lymphocyte trafficking and colitis. Gastroenterology. 2001 Oct 1;121(4):1008–10.
- Zeng MY, Miralda I, Armstrong CL, Uriarte SM, Bagaitkar J. The roles of NADPH oxidase in modulating neutrophil effector responses. Mol Oral Microbiol. 2019 Apr;34(2):27–38.
- Winterbourn CC, Kettle AJ, Hampton MB. Reactive Oxygen Species and Neutrophil Function. Annu Rev Biochem. 2016 Jun 2;85:765–92.
- Luecken MD, Theis FJ. Current best practices in single-cell RNA-seq analysis: a tutorial. Mol Syst Biol. 2019 Jun 19;15(6):e8746.
- Friedrich M, Pohin M, Powrie F. Cytokine Networks in the Pathophysiology of Inflammatory Bowel Disease. Immunity. 2019 Apr 16;50(4):992–1006.
- Chen ML, Sundrud MS. Cytokine networks and T cell subsets in inflammatory bowel diseases. Inflamm Bowel Dis. 2016 May;22(5):1157–67.
- Mennillo E, Kim YJ, Lee G, Rusu I, Patel RK, Dorman LC, Flynn E, Li S, Bain JL, Andersen C, Rao A, Tamaki S, Tsui J, Shen A, Lotstein ML, Rahim M, Naser M, Bernard-Vazquez F, Eckalbar W, Cho SJ, Beck K, El-Nachef N, Lewin S, Selvig DR, Terdiman JP, Mahadevan U, Oh DY, Fragiadakis GK, Pisco A, Combes AJ, Kattah MG. Single-cell and spatial multi-omics highlight effects of anti-integrin therapy across cellular compartments in ulcerative colitis. Nat Commun. 2024 Feb 19;15(1):1493.
- Lismont C, Revenco I, Li H, Costa CF, Lenaerts L, Hussein MAF, De Bie J, Knoops B, Van Veldhoven PP, Derua R, Fransen M. Peroxisome-Derived Hydrogen Peroxide Modulates the Sulfenylation Profiles of Key Redox Signaling Proteins in Flp-In T-REx 293 Cells. Front Cell Dev Biol. 2022;10:888873.
- Day NJ, Zhang T, Gaffrey MJ, Zhao R, Fillmore TL, Moore RJ, Rodney GG, Qian WJ. A deep redox proteome profiling workflow and its application to skeletal muscle of a Duchenne Muscular Dystrophy model. Free Radical Biology and Medicine. 2022 Nov 20;193:373–84.
- Global redox proteome and phosphoproteome analysis reveals redox switch in Akt | Nature Communications [Internet]. [cited 2025 Sep 4]. Available from: https://www.nature.com/articles/s41467-019-13114-4.
- Li X, Gluth A, Zhang T, Qian WJ. Thiol redox proteomics: Characterization of thiol-based post-translational modifications. Proteomics. 2023 Jul;23(13–14):e2200194.
- Zhang T, Gaffrey MJ, Li X, Qian WJ. Characterization of cellular oxidative stress response by stoichiometric redox proteomics. Am J Physiol Cell Physiol. 2021 Feb 1;320(2):C182–94.
- Global redox proteome and phosphoproteome analysis reveals redox switch in Akt | Nature Communications [Internet]. [cited 2025 Sep 13]. Available from: https://www.nature.com/articles/s41467-019-13114-4.
- Su Z, Burchfield JG, Yang P, Humphrey SJ, Yang G, Francis D, Yasmin S, Shin SY, Norris DM, Kearney AL, Astore MA, Scavuzzo J, Fisher-Wellman KH, Wang QP, Parker BL, Neely GG, Vafaee F, Chiu J, Yeo R, Hogg PJ, Fazakerley DJ, Nguyen LK, Kuyucak S, James DE. Global redox proteome and phosphoproteome analysis reveals redox switch in Akt. Nat Commun. 2019 Dec 2;10(1):5486.
- Ruiz Castro PA, Yepiskoposyan H, Gubian S, Calvino-Martin F, Kogel U, Renggli K, Peitsch MC, Hoeng J, Talikka M. Systems biology approach highlights mechanistic differences between Crohn’s disease and ulcerative colitis. Sci Rep. 2021 Jun 1;11(1):11519.
- Kariyawasam V, Davis T, Ghali M, Tejcek D, R O’Neil T, Verley E, Griffiths M, Campos M, Bonney I, Bulmer AC, Ramaswamy Y, Mitrev N, Corte C, Witting PK, Chami B. Myeloperoxidase Luminol Reaction - A Novel Faecal Assay for Predicting Colonoscopy Findings in Patients with Ulcerative Colitis: A Pilot Cross-Sectional Clinical Study. Adv Healthc Mater. 2025 Aug 21;e01825.
- Turner D, Ricciuto A, Lewis A, D’Amico F, Dhaliwal J, Griffiths AM, Bettenworth D, Sandborn WJ, Sands BE, Reinisch W, Schölmerich J, Bemelman W, Danese S, Mary JY, Rubin D, Colombel JF, Peyrin-Biroulet L, Dotan I, Abreu MT, Dignass A, International Organization for the Study of IBD. STRIDE-II: An Update on the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) Initiative of the International Organization for the Study of IBD (IOIBD): Determining Therapeutic Goals for Treat-to-Target strategies in IBD. Gastroenterology. 2021 Apr;160(5):1570–83.
- Scheppach W, Sommer H, Kirchner T, Paganelli GM, Bartram P, Christl S, Richter F, Dusel G, Kasper H. Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis. Gastroenterology. 1992 Jul;103(1):51–6.
- Breuer RI, Soergel KH, Lashner BA, Christ ML, Hanauer SB, Vanagunas A, Harig JM, Keshavarzian A, Robinson M, Sellin JH, Weinberg D, Vidican DE, Flemal KL, Rademaker AW. Short chain fatty acid rectal irrigation for left-sided ulcerative colitis: a randomised, placebo controlled trial. Gut. 1997 Apr;40(4):485–91.
- Luceri C, Femia AP, Fazi M, Di Martino C, Zolfanelli F, Dolara P, Tonelli F. Effect of butyrate enemas on gene expression profiles and endoscopic/histopathological scores of diverted colorectal mucosa: A randomized trial. Dig Liver Dis. 2016 Jan;48(1):27–33.
- Chulkina M, Rohmer C, McAninch S, Panganiban RP, Villéger R, Portolese A, Ciocirlan J, Yang W, Cohen C, Koltun W, Valentine JF, Cong Y, Yochum G, Beswick EJ, Pinchuk IV. Increased Activity of MAPKAPK2 within Mesenchymal Cells as a Target for Inflammation-Associated Fibrosis in Crohn’s Disease. J Crohns Colitis. 2024 Aug 6;18(7):1147–61.
- Wang Z, Liang XY, Chang X, Nie YY, Guo C, Jiang JH, Chang M. MMI-0100 Ameliorates Dextran Sulfate Sodium-Induced Colitis in Mice through Targeting MK2 Pathway. Molecules. 2019 Aug 3;24(15):2832.
- Deeks ED. Anifrolumab: First Approval. Drugs. 2021 Oct;81(15):1795–802.
| Stimulus / Factor | Target Cell Type | Effect on DUOX2 / H₂O₂ | Pathophysiological Consequence | References |
| IFN-γ (Th1 cytokine) | Intestinal epithelium | Strong upregulation of DUOX2 and H₂O₂ | Drives epithelial oxidative burst; promotes chronic inflammation in IBD. | [47,48] |
| IL-22 (Type 17 cytokine) | Intestinal epithelium | Increases DUOX2 expression | Enhances epithelial host defense; may aid barrier repair and antimicrobial protection. | [49,50] |
| TLR4 agonists (LPS) | Intestinal epithelium | Upregulates DUOX2 and H₂O₂ | Couples bacterial sensing to ROS output; implicated in colitis-associated tumorigenesis. | [37,51] |
| Adherent-invasive E. coli | Intestinal epithelium | Potently induces DUOX2 | Amplifies H₂O₂ release during dysbiosis; promotes mucosal inflammation. | [52,53] |
| Dysbiotic microbiota | Intestinal epithelium | Broad activation of DUOX2 | Marker of disrupted homeostasis; correlates with early preclinical IBD changes. | [54,55] |
| Short-chain fatty acids (butyrate) | Intestinal epithelium | Downregulates DUOX2 | Restores barrier integrity; dampens inflammation by lowering epithelial H₂O₂ output. | [56] |
| HDAC inhibitors | Intestinal epithelium | Mimic butyrate effect, suppress DUOX2 | Potential therapeutic avenue to control DUOX2-mediated oxidative stress. | [57] |
| Inflammatory milieu (IL-8, TNF, IFNα) | Neutrophils (new finding) | Induces de novo DUOX2 expression | Neutrophils gain extra oxidative capacity; amplify cytokine circuits and tissue inflammation in IBD. | [57,58] |
| Strategy / Target | Mechanism of Action | Expected Effect on IBD | References |
| Neutrophil DUOX2 inhibition | Genetic silencing (e.g., conditional knockout) or small-molecule inhibition of DUOX2 | Reduces neutrophil H₂O₂ and cytokine output; suppresses mucosal inflammation; improves colitis in models. ⚠ Risk: impaired pathogen defense. | [31,73] |
| Butyrate / SCFA supplementation | Microbial metabolite; inhibits histone deacetylases (HDACs) | Downregulates epithelial and neutrophil DUOX2; reduces cytokine release and NETosis; enhances barrier integrity. | [74,75,76] |
| JAK inhibitors (e.g., baricitinib, tofacitinib) | Block IFN-α/γ and other cytokine signaling via JAK–STAT pathway | Prevent IFNα-driven neutrophil–fibroblast immunofibrosis; reduce IL-8–mediated recruitment; established anti-inflammatory effect in IBD. | [77,78] |
| CXCR1/2 antagonists (e.g., reparixin) | Block neutrophil chemokine receptors for IL-8/CXCL1/2 | Reduces neutrophil migration into gut mucosa; may lower neutrophil burden and tissue injury. | [79,80] |
| MK2 (p38 MAPK) inhibitors | Inhibit MAPK-activated protein kinase 2 (MK2), required for NADPH oxidase assembly/ROS | Decreases neutrophil-derived ROS and cytokine output; protects against DSS colitis in preclinical studies. | [81,82] |
| TLR4 antagonists (e.g., eritoran) | Block LPS binding to TLR4 on epithelial cells | Prevent LPS-induced DUOX2 overactivation; reduces epithelial oxidative stress and inflammation. | [83,84,85] |
| Probiotics / Fecal microbiota transplantation (FMT) | Restore commensals and SCFA-producers (e.g., Clostridium clusters) | Reduce dysbiosis-driven DUOX2 activation; increase butyrate production; rebalance immune–microbiota crosstalk. | [86] |
| Antioxidants (e.g., N-acetylcysteine, vitamins C/E) | Scavenge reactive oxygen species (ROS) | Neutralize excessive H₂O₂ from DUOX2/NOX; may reduce epithelial oxidative injury, but efficacy is inconsistent. | [15,73,87] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




