Submitted:
31 October 2025
Posted:
04 November 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
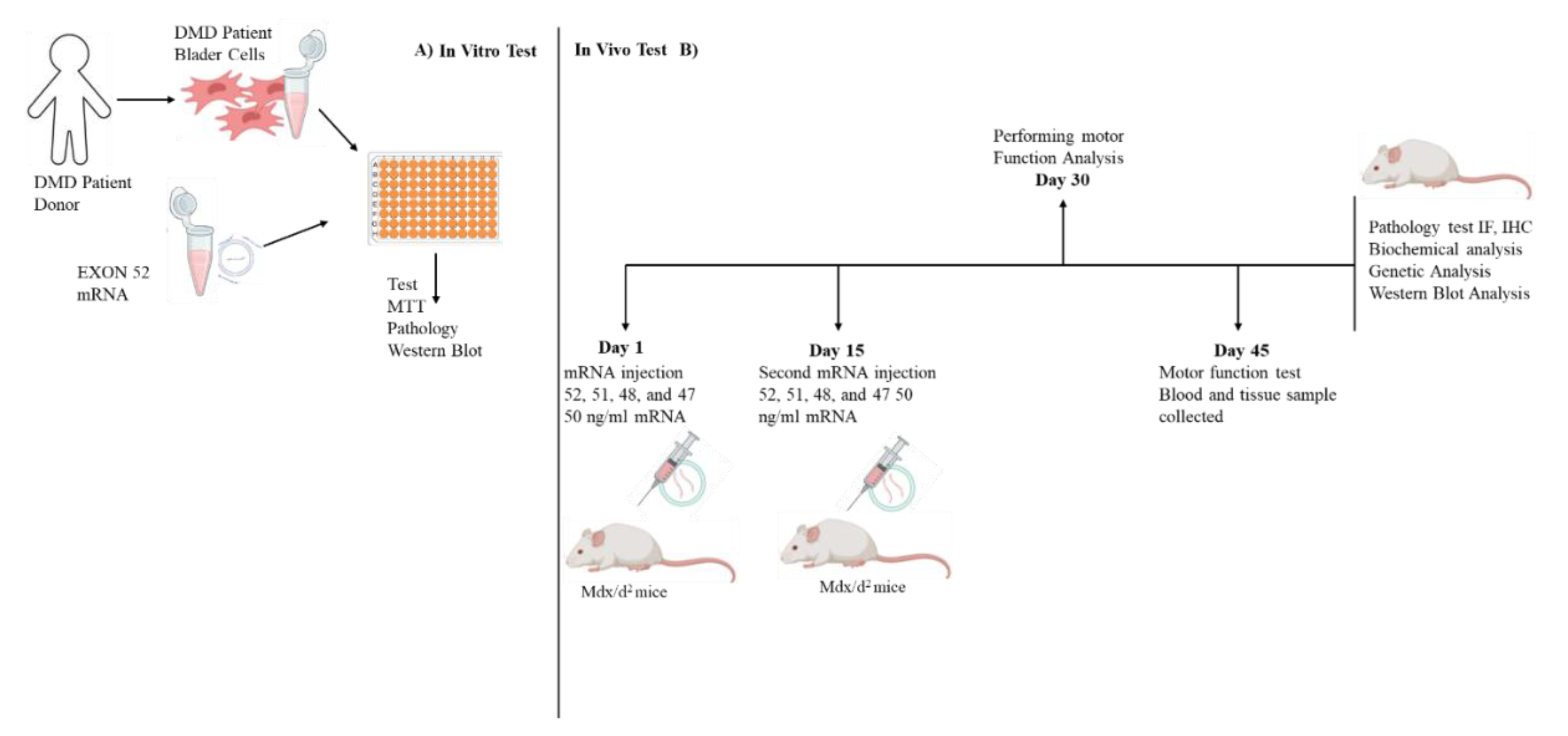
2.1. Pre-Experimental Stages and Patent Process
2.2. Experimental Design
2.3. Primary Cell Culture Preparation
2.3.1. EXON 52 Test
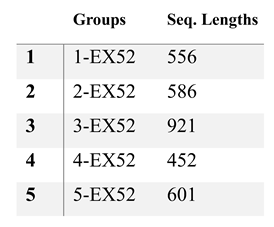 |
2.4. Animal Study
2.5. Treatment Preparation
2.6. Behavioral Tests
2.7. Molecular Analyses
2.7.1. ELISA Tests
2.7.2. Gene Expression
 |
2.7.3. Western Blot
2.7.4. IF (Immunofluorescence) Analysis
2.7.5. IHC (Immunohistochemical) Analysis
2.7.6. Statistical Analysis
3. Results
3.1. EXON 52 In Vitro Test Results
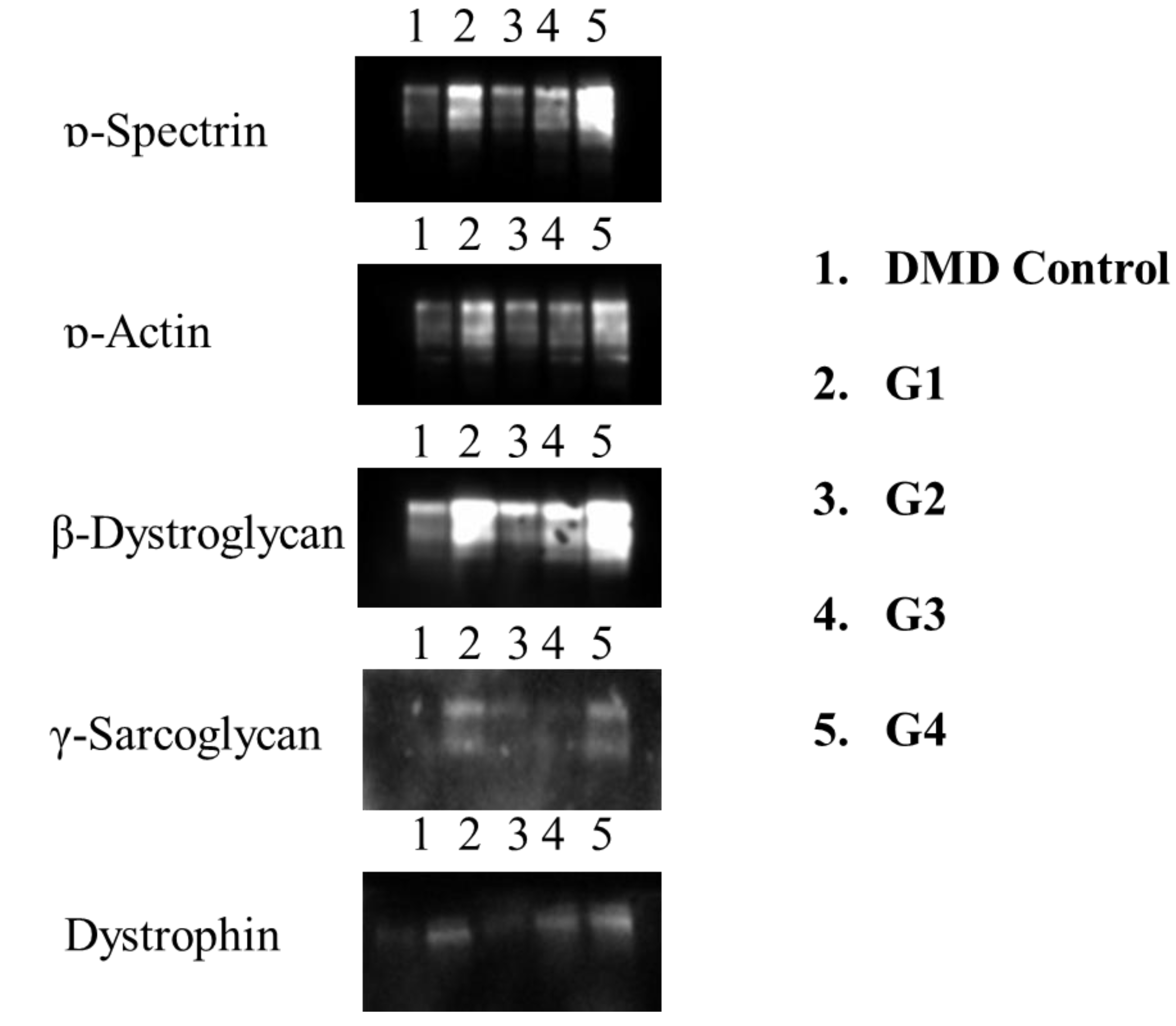
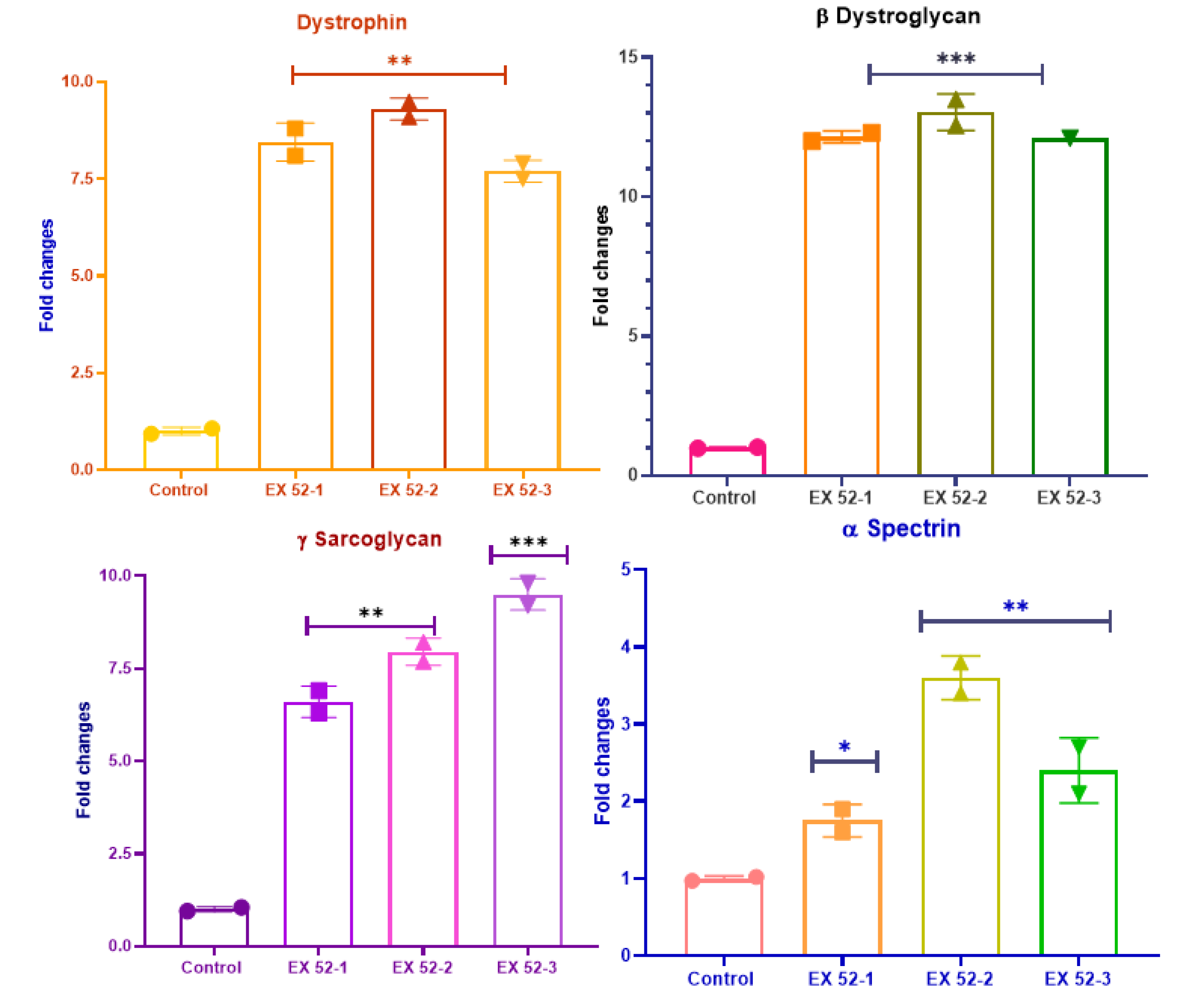
3.1.1. Western Blot Analysis
3.1.2. Genetic Analyses
3.2. In vivo test for EXON 52, 51, 48, and 47 mRNA complex
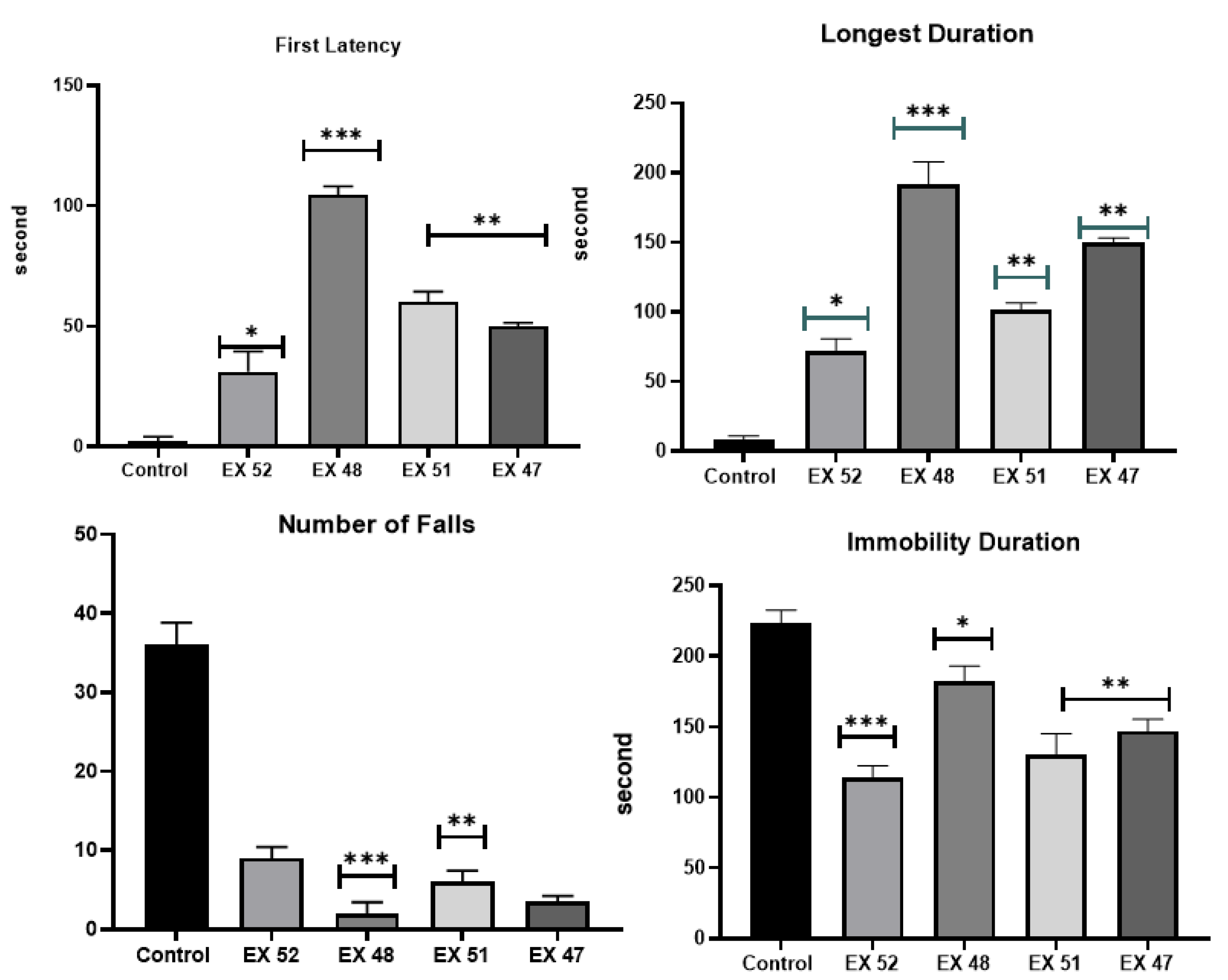
3.2.1. Behavioral Analyses
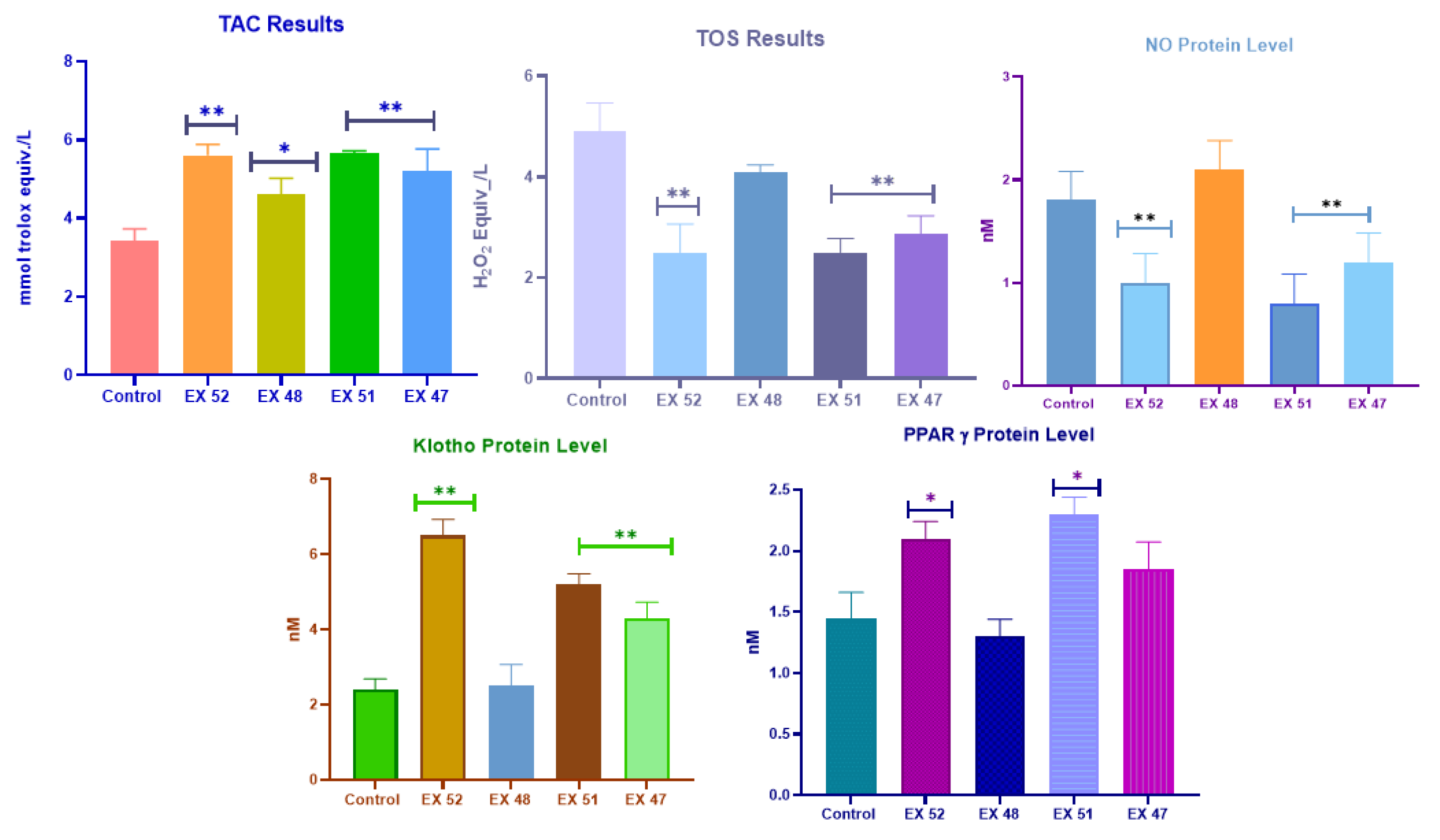
3.2.2. ELISA Analyses
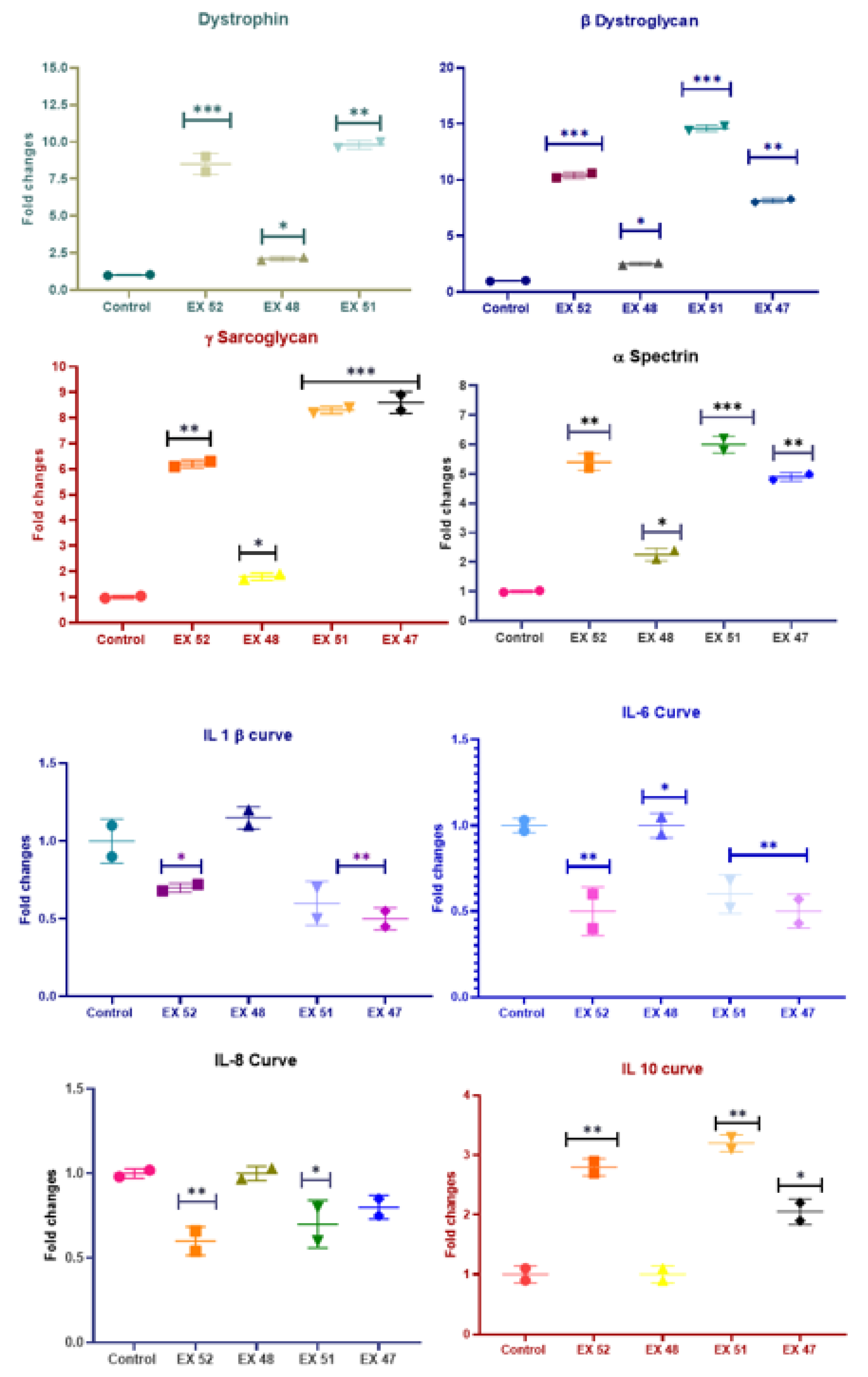
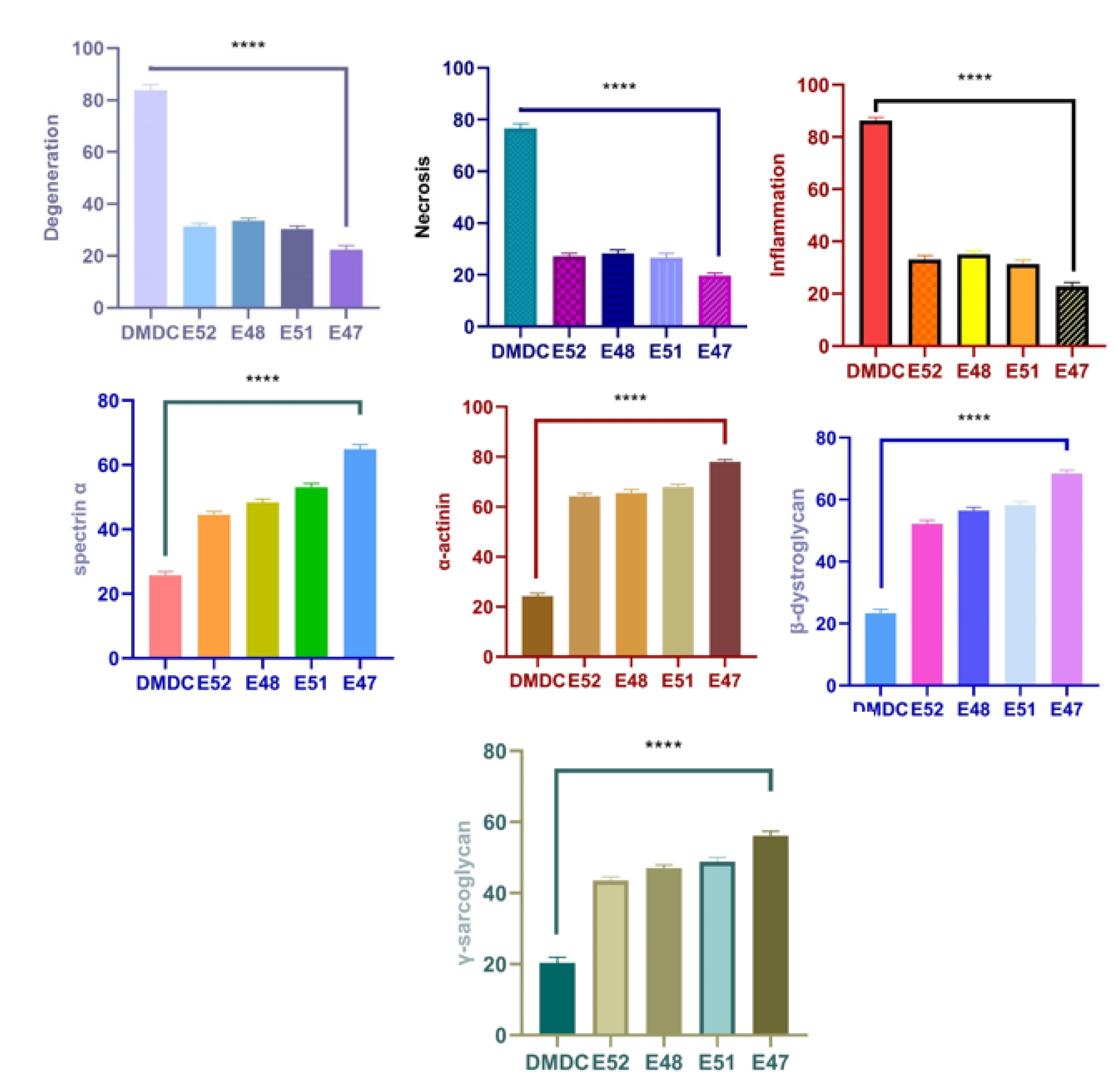
3.3.3. Gene Expression Analyses
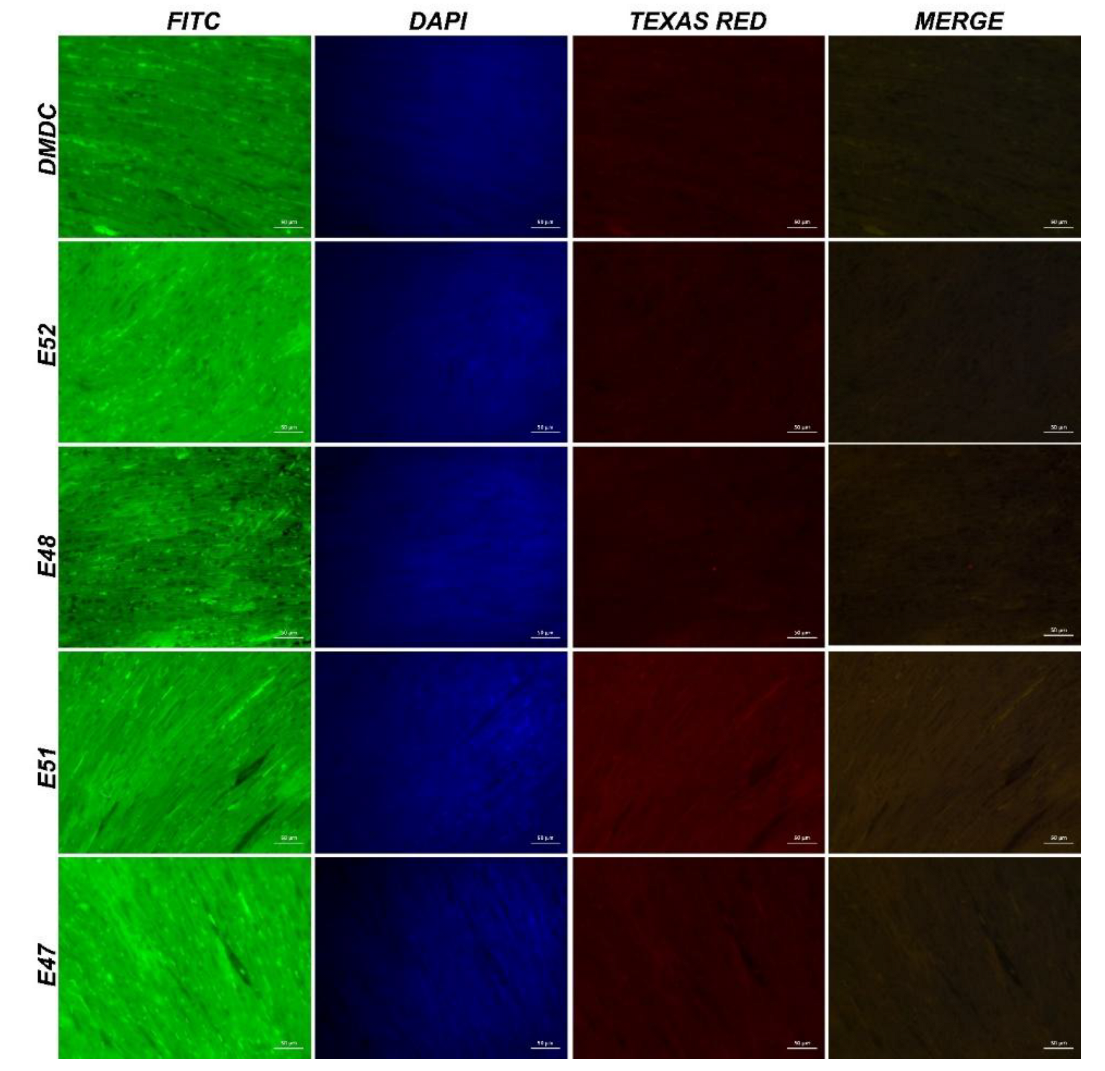
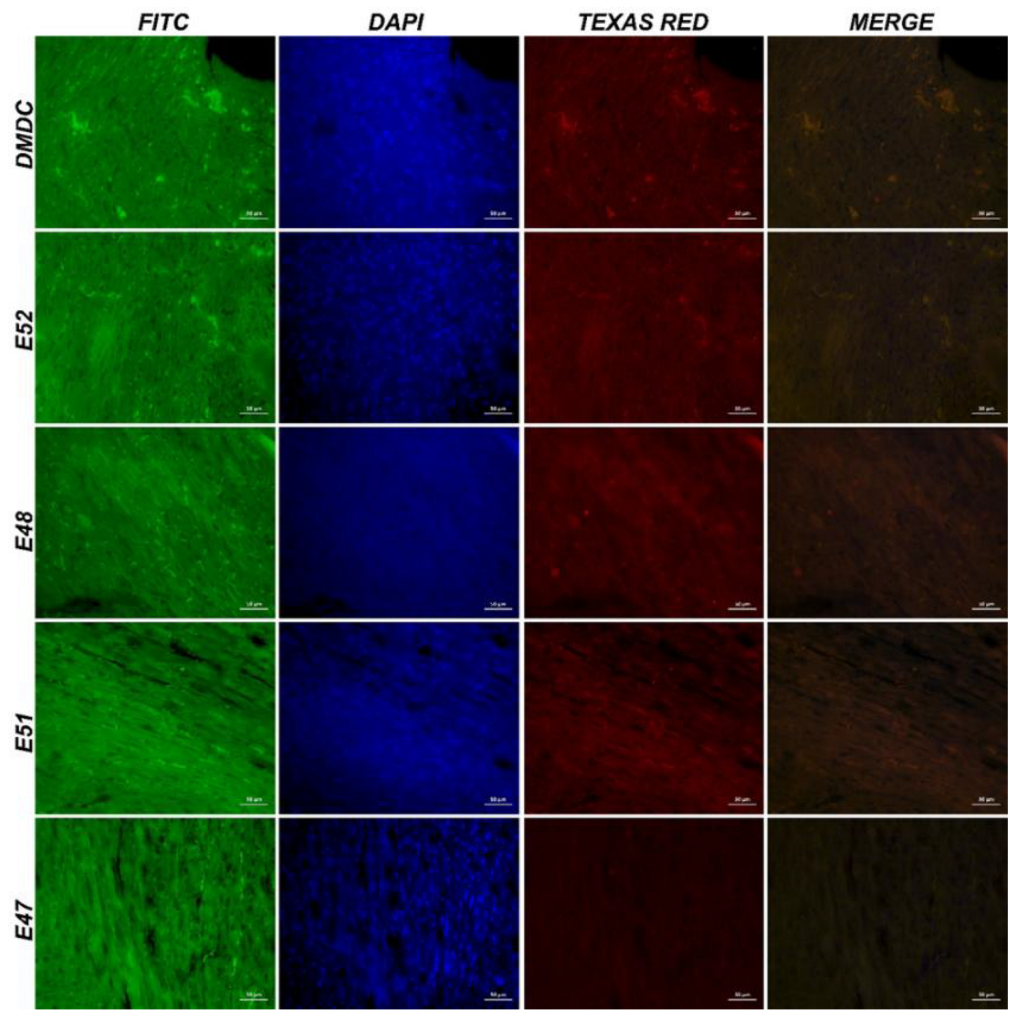
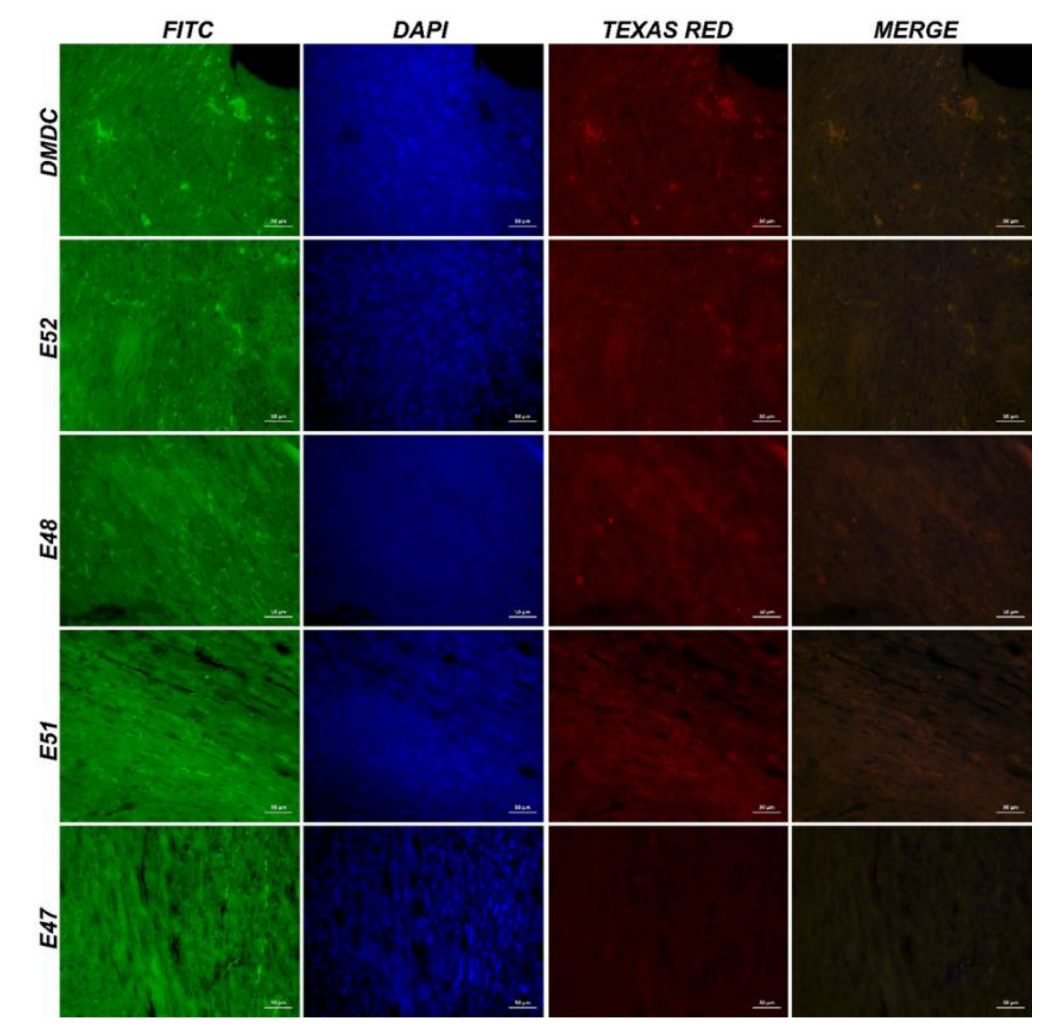
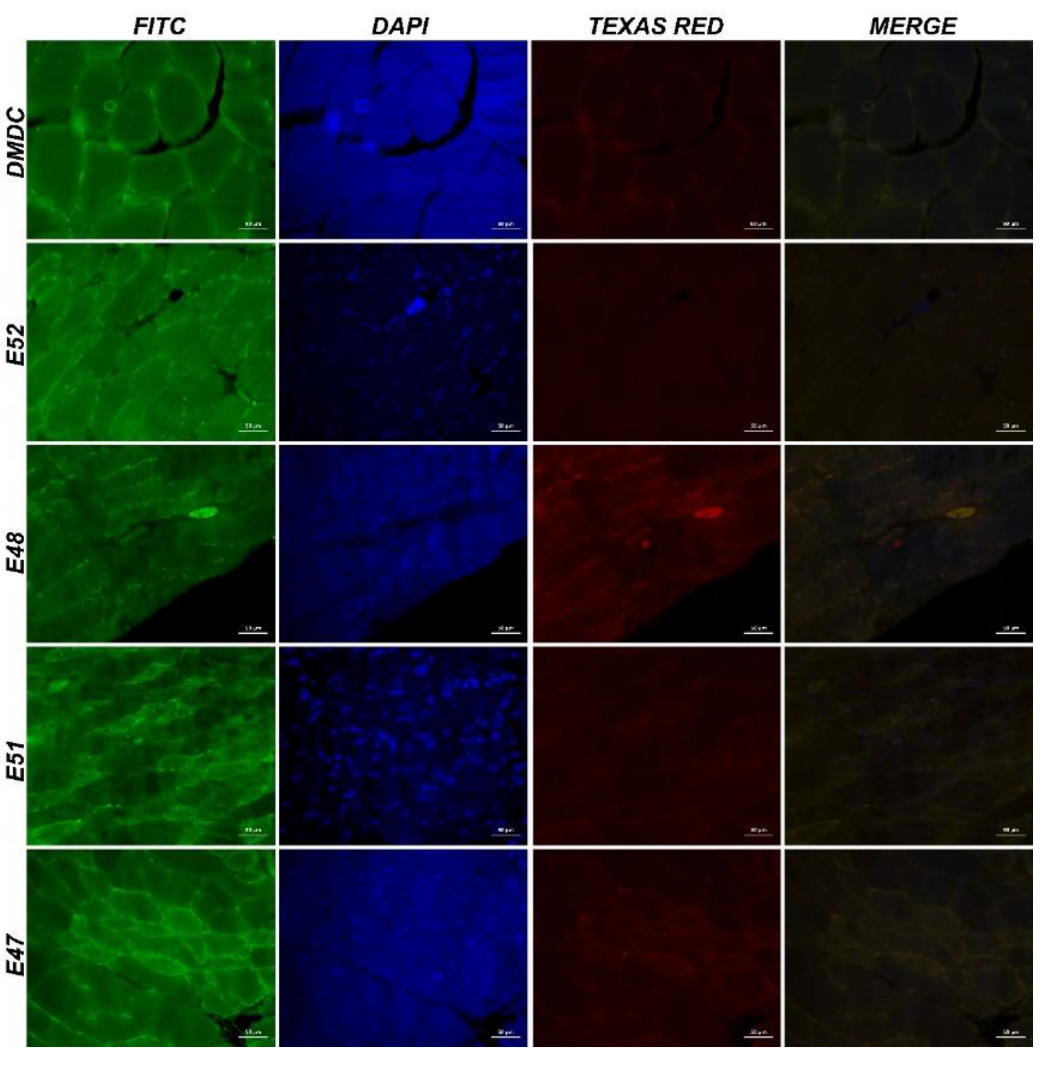
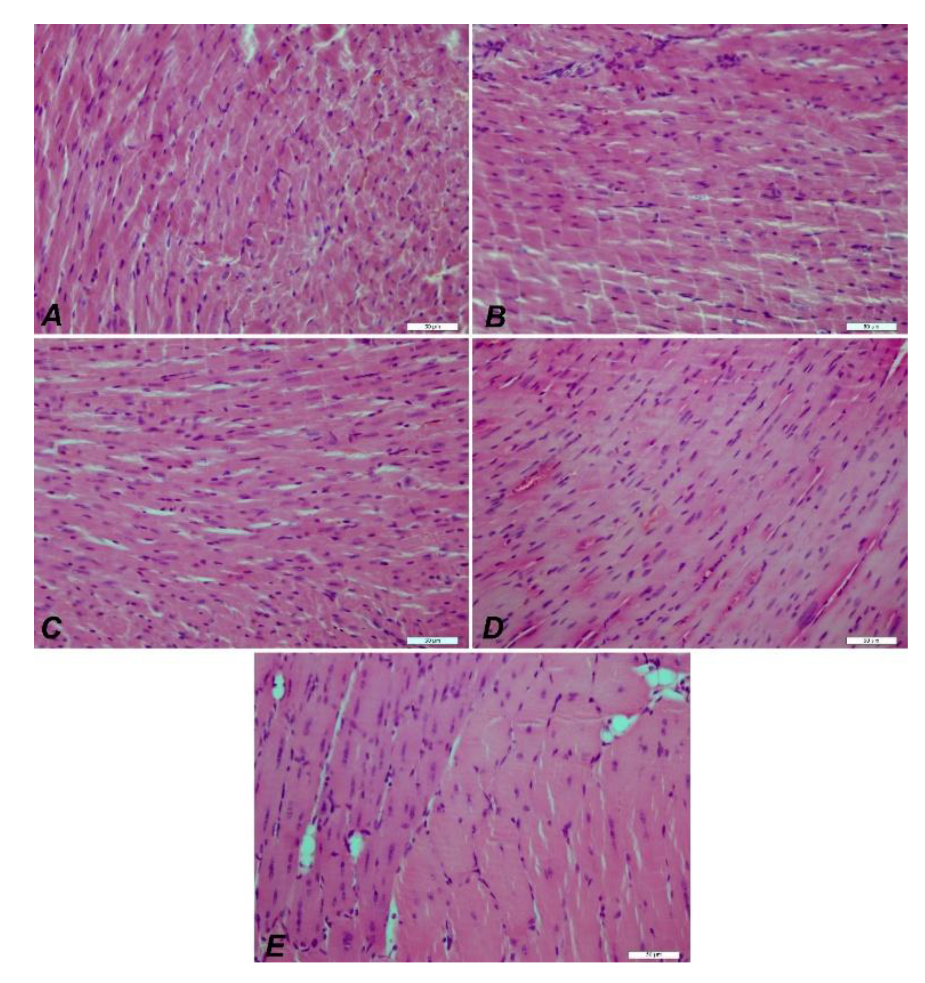
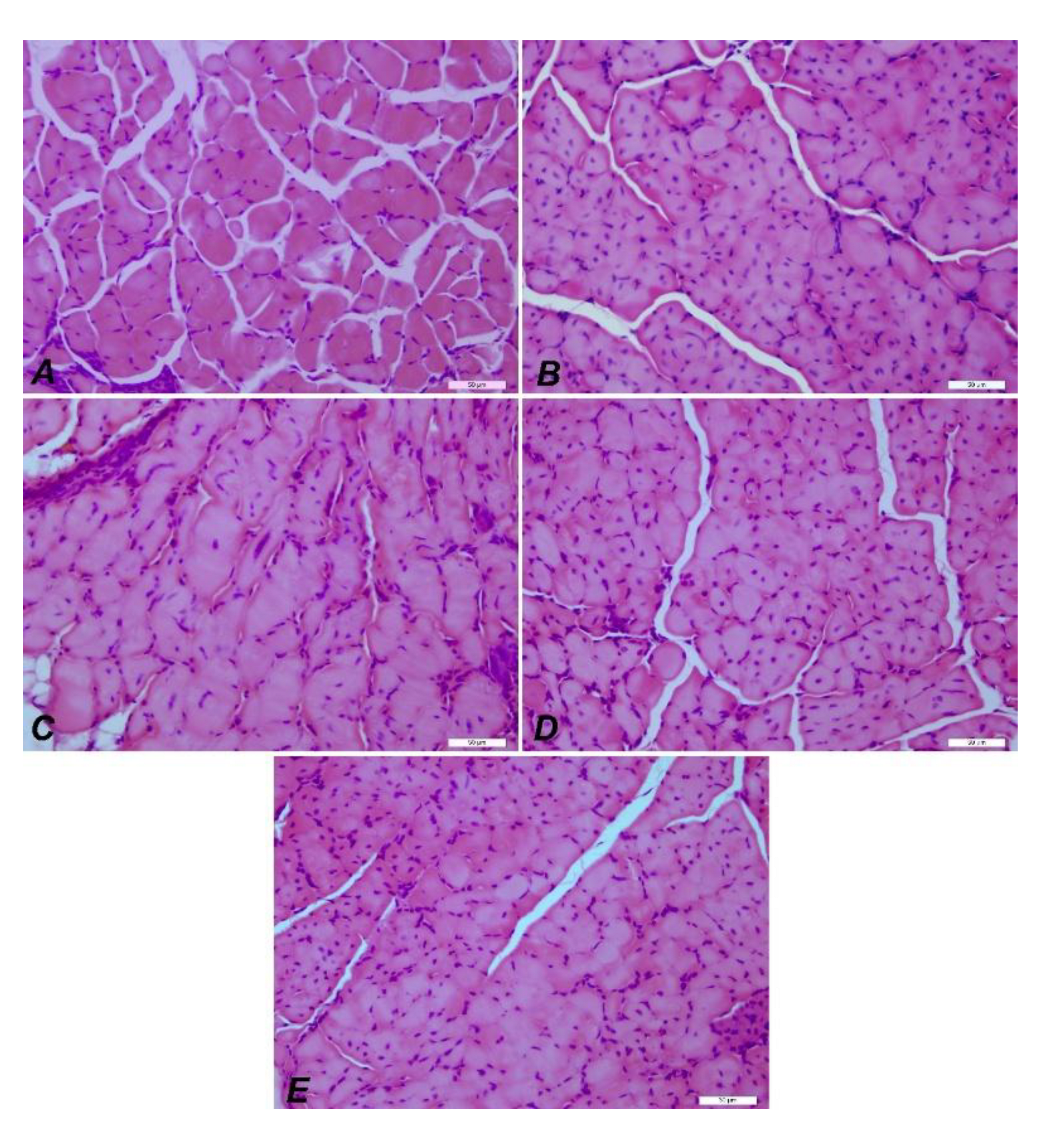
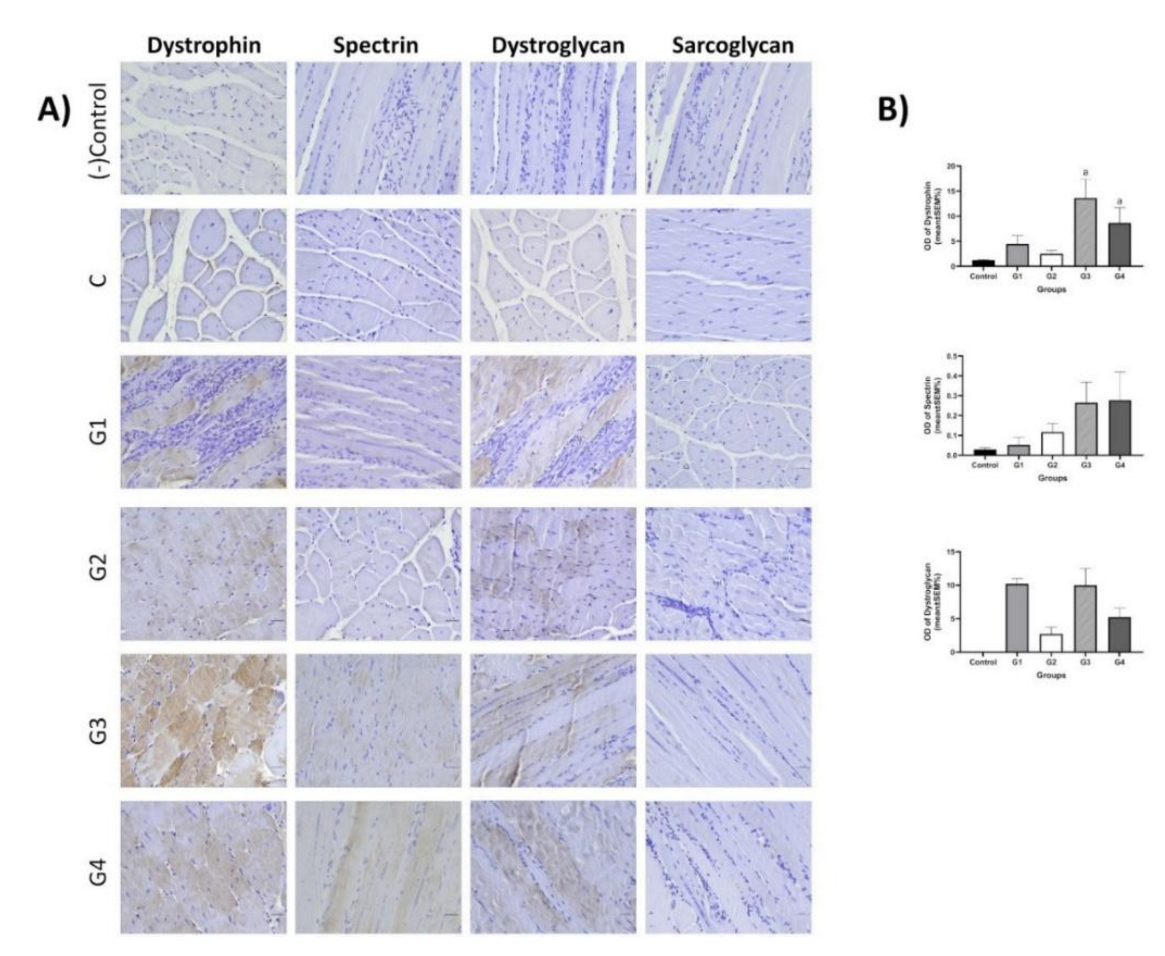
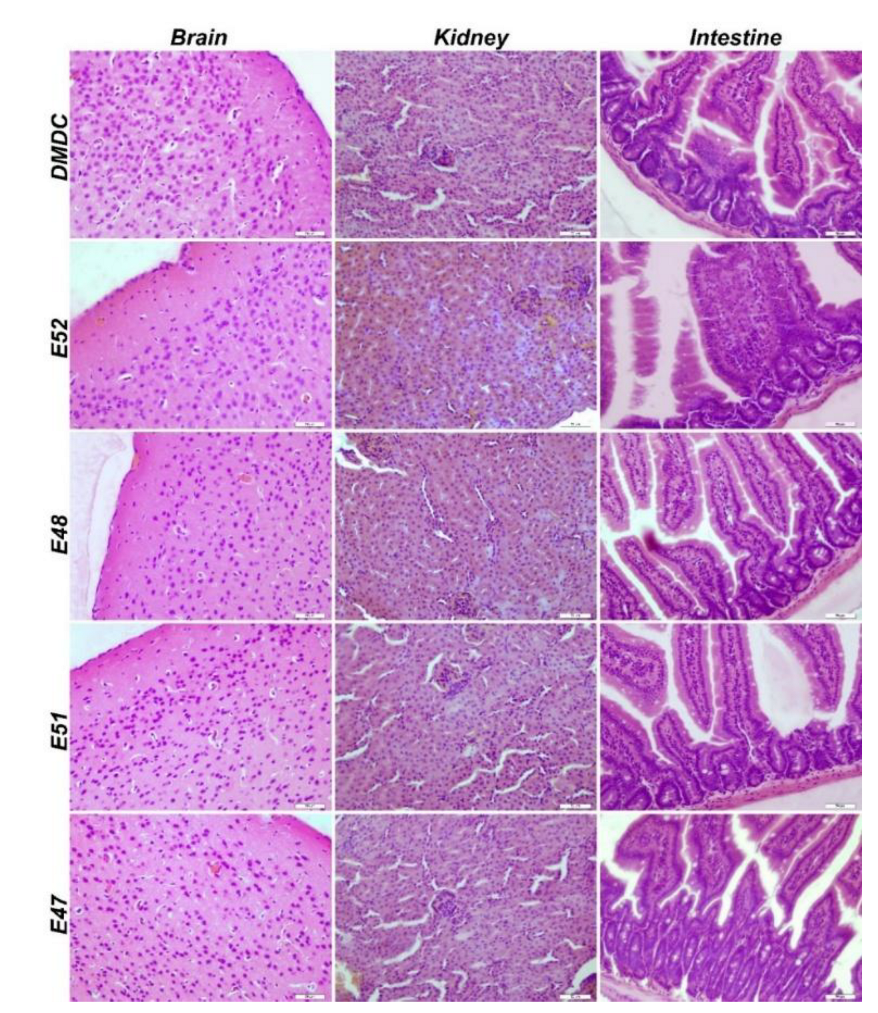
3.3.4. Immunofluorescence and Immunohistochemistry Analysis for Muscles
3.3.5. IHC Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DGC | Dystrophin-glycoprotein complex |
| ECM | Extracellular matrix |
| DMD | Duchenne Muscular Dystrophy |
| IHC | Immunohistochemical |
| IF | Immunofluorescence |
| PPAR- γ | Proliferator-activated receptor γ |
| EX | Exon |
| TAC | Total Antioxidant Capacity |
| TOS | Total Oxidative Stress |
| NO | Nitric Oxide |
References
- Ervasti, J.M. and K.P. Campbell, A role for the dystrophin-glycoprotein complex as a transmembrane linker between laminin and actin. The Journal of cell biology, 1993. 122(4): p. 809-823.
- Constantin, B. , Dystrophin complex functions as a scaffold for signalling proteins. Biochimica et Biophysica Acta (BBA)-Biomembranes, 2014. 1838(2): p. 635-642.
- Gao, Q.Q. and E.M. McNally, The dystrophin complex: structure, function, and implications for therapy. Comprehensive physiology, 2015. 5(3): p. 1223-1239.
- Hughes, D.C. , et al., Age-related differences in dystrophin: impact on force transfer proteins, membrane integrity, and neuromuscular junction stability. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 2017. 72(5): p. 640-648.
- Duan, D. , et al., Duchenne muscular dystrophy. Nature reviews disease primers, 2021. 7(1): p. 13.
- Vo, A.H. and E.M. McNally, Modifier genes and their effect on Duchenne muscular dystrophy. Current opinion in neurology, 2015. 28(5): p. 528-534.
- García-Rodríguez, R. , et al., Premature termination codons in the DMD gene cause reduced local mRNA synthesis. Proceedings of the National Academy of Sciences, 2020. 117(28): p. 16456-16464.
- Mercuri, E., C. G. Bönnemann, and F. Muntoni, Muscular dystrophies. The Lancet, 2019. 394(10213): p. 2025-2038.
- Bies, R.D. , et al., Human and murine dystrophin mRNA transcripts are differentially expressed during skeletal muscle, heart, and brain development. Nucleic acids research, 1992. 20(7): p. 1725-1731.
- Zhang, X.-f., W. Hu, and J. Hu, Neurological impairments in Duchenne muscular dystrophy: A comprehensive review. Acta Neurologica Belgica, 2025: p. 1-12.
- Zhu, Y. , et al., Serum enzyme profiles differentiate five types of muscular dystrophy. Disease Markers, 2015. 2015(1): p. 543282.
- Lo Cascio, C.M. , et al., Gastrointestinal dysfunction in patients with Duchenne muscular dystrophy. PLoS One, 2016. 11(10): p. e0163779.
- Kutluk, M.G. and Ç.S. Doğan, Kidney involvement and associated risk factors in children with Duchenne muscular dystrophy. Pediatric Nephrology, 2020. 35(10): p. 1953-1958.
- Villa, C.R. , et al., Identifying evidence of cardio-renal syndrome in patients with Duchenne muscular dystrophy using cystatin C. Neuromuscular Disorders, 2016. 26(10): p. 637-642.
- Klingler, W. , et al., The role of fibrosis in Duchenne muscular dystrophy. Acta Myologica, 2012. 31(3): p. 184.
- Desguerre, I. , et al., Endomysial fibrosis in Duchenne muscular dystrophy: a marker of poor outcome associated with macrophage alternative activation. Journal of Neuropathology & Experimental Neurology, 2009. 68(7): p. 762-773.
- Sindhurakar, A. , et al., An automated test of rat forelimb supination quantifies motor function loss and recovery after corticospinal injury. Neurorehabilitation and neural repair, 2017. 31(2): p. 122-132.
- Aartsma-Rus, A. and M. van Putten, Assessing functional performance in the mdx mouse model. Journal of visualized experiments: JoVE, 2014(85): p. 51303.
- Taglietti, V. , et al., Duchenne muscular dystrophy trajectory in R-DMDdel52 preclinical rat model identifies COMP as biomarker of fibrosis. Acta neuropathologica communications, 2022. 10(1): p. 60.
- Therapeutics, S. , Sarepta Therapeutics Reports Positive Clinical Results from Phase 2 MOMENTUM Study of SRP-5051 in Patients with Duchenne Muscular Dystrophy Amenable to Skipping Exon 51| Sarepta Therapeutics, Inc.[Internet].(2021).
- Happi Mbakam, C., G. Lamothe, and J.P. Tremblay, Therapeutic strategies for dystrophin replacement in Duchenne muscular dystrophy. Frontiers in Medicine, 2022. 9: p. 859930.
- Aartsma-Rus, A. and A.M. Krieg, FDA approves eteplirsen for Duchenne muscular dystrophy: the next chapter in the eteplirsen saga. Nucleic acid therapeutics, 2017. 27(1): p. 1-3.
- Mantuano, P. , et al., β-Dystroglycan restoration and pathology progression in the dystrophic mdx mouse: outcome and implication of a clinically oriented study with a novel oral dasatinib formulation. Biomolecules, 2021. 11(11): p. 1742.
- Hack, A.A. , et al., γ-Sarcoglycan deficiency leads to muscle membrane defects and apoptosis independent of dystrophin. The Journal of cell biology, 1998. 142(5): p. 1279-1287.
- Guhathakurta, P. , et al., Enhancing interaction of actin and actin-binding domain 1 of dystrophin with modulators: Toward improved gene therapy for Duchenne muscular dystrophy. Journal of Biological Chemistry, 2022. 298(12).
- Wehling-Henricks, M. , et al., Klotho gene silencing promotes pathology in the mdx mouse model of Duchenne muscular dystrophy. Human molecular genetics, 2016. 25(12): p. 2465-2482.
- Suntar, I. , et al., Natural products, PGC-1α, and Duchenne muscular dystrophy. Acta Pharmaceutica Sinica B, 2020. 10(5): p. 734-745.
- Deconinck, N. and B. Dan, Pathophysiology of duchenne muscular dystrophy: current hypotheses. Pediatric neurology, 2007. 36(1): p. 1-7.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




