Submitted:
22 October 2025
Posted:
23 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
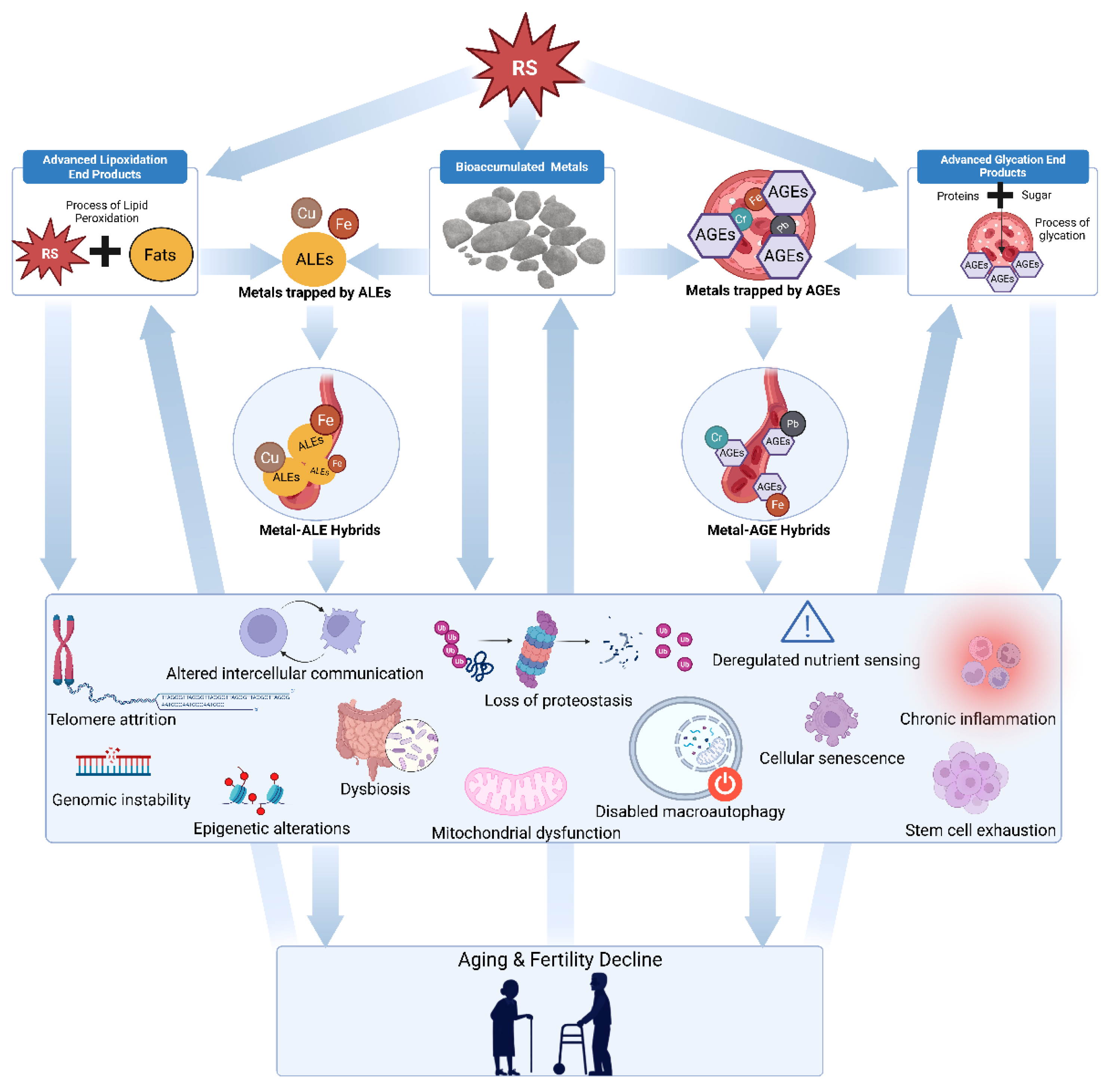
2. ROS, RNS, and RCS in Reproductive Aging
Detoxification System Decline in Aging Ovaries
3. Metal Bioaccumulation in Ovarian Aging
3.1. Iron Accumulation
3.2. Copper and Zinc Dysregulation
3.3. Heavy Metal Exposure
4. Advanced Glycation End Products in Reproductive Aging
4.1. The AGE-RAGE Axis in Ovarian Pathophysiology
4.2. AGE-Mediated Disruption of Ovarian Function
4.3. Dietary AGEs and Reproductive Health
5. Advanced Lipoxidation End Products in Ovarian Aging
5.1. ALE Targeted Proteins
5.2. Mitochondrial Dysfunction and ALE Formation
5.3. Membrane Lipid Deterioration in Aging Oocytes
6. Hormonal Dysfunction and Hypothalamic-Pituitary-Ovarian Axis Changes
6.1. Steroid Hormone Production and Metabolism
6.2. Hypothalamic-Pituitary Network Degeneration and Feedback Failure
7. Clinical Trial Compounds to Delay Female Reproductive Aging
7.1. Reactive Species Therapy
7.2. Metal Chelation Therapy
7.3. Advanced Glycation End Product Prevention Therapy
7.4. Advanced Lipoxidation End Product Prevention Therapy
7.5. Hormone Therapy
7.6. Trace Element Therapy
8. Clinical Translation and Future Directions
Biomarker Development and Clinical Assessment
Future Research Directions
9. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agarwal, A., Aponte-Mellado, A., Premkumar, B. J., Shaman, A., & Gupta, S. (2012). The effects of oxidative stress on female reproduction: a review. Reproductive biology and endocrinology : RB&E, 10, 49. [CrossRef]
- Alkhalaf, A., Kleefstra, N., Groenier, K. H., Bilo, H. J., Gans, R. O., Heeringa, P., Scheijen, J. L., Schalkwijk, C. G., Navis, G. J., & Bakker, S. J. (2012). Effect of benfotiamine on advanced glycation endproducts and markers of endothelial dysfunction and inflammation in diabetic nephropathy. PloS one, 7(7), e40427. [CrossRef]
- Anjos, M. M. D., de Paula, G. R., Yokomizo, D. N., Costa, C. B., Bertozzi, M. M., Verri, W. A., Jr, Alfieri, A. A., Morotti, F., & Seneda, M. M. (2025). Effect of Alpha-Lipoic Acid on the Development, Oxidative Stress, and Cryotolerance of Bovine Embryos Produced In Vitro. Veterinary sciences, 12(2), 120. [CrossRef]
- Arslan, A., Balcioğlu, E., Nisari, M., Yalçin, B., Ülger, M., Güler, E., Uzun, G. B., & Acer, N. (2022). Effect of carnosine on ovarian follicle in rats exposed to electromagnetic field. European Journal of Anatomy, 26(6), 659–668. [CrossRef]
- Athanasiou, D., Voros, C., Soyhan, N., Panagou, G., Sakellariou, M., Mavrogianni, D., Bikouvaraki, E. S., Daskalakis, G., & Pappa, K. (2025). The Molecular Landscape of Nitric Oxide in Ovarian Function and IVF Success: Bridging Redox Biology and Reproductive Outcomes. Biomedicines, 13(7), 1748. [CrossRef]
- Avelino, C. M. S. F., & de Araújo, R. F. F. (2024). Effects of vitamin D supplementation on oxidative stress biomarkers of Iranian women with polycystic ovary syndrome: a meta-analysis study. Revista brasileira de ginecologia e obstetricia : revista da Federacao Brasileira das Sociedades de Ginecologia e Obstetricia, 46, e-rbgo37. [CrossRef]
- Ben-Meir, A., Burstein, E., Borrego-Alvarez, A., Chong, J., Wong, E., Yavorska, T., Naranian, T., Chi, M., Wang, Y., Bentov, Y., Alexis, J., Meriano, J., Sung, H. K., Gasser, D. L., Moley, K. H., Hekimi, S., Casper, R. F., & Jurisicova, A. (2015). Coenzyme Q10 restores oocyte mitochondrial function and fertility during reproductive aging. Aging cell, 14(5), 887–895. [CrossRef]
- Bezdíček, J., Sekaninová, J., Janků, M., Makarevič, A., Luhová, L., Dujíčková, L., & Petřivalský, M. (2025). Reactive oxygen and nitrogen species: multifaceted regulators of ovarian activity†. Biology of reproduction, 112(5), 789–806. [CrossRef]
- Bochynska, S., García-Pérez, M. Á., Tarín, J. J., Szeliga, A., Meczekalski, B., & Cano, A. (2025). The Final Phases of Ovarian Aging: A Tale of Diverging Functional Trajectories. Journal of clinical medicine, 14(16), 5834. [CrossRef]
- Boldyrev, A. A., Aldini, G., & Derave, W. (2013). Physiology and pathophysiology of carnosine. Physiological reviews, 93(4), 1803–1845. [CrossRef]
- Bönhof, G. J., Sipola, G., Strom, A., Herder, C., Strassburger, K., Knebel, B., Reule, C., Wollmann, J. C., Icks, A., Al-Hasani, H., Roden, M., Kuss, O., & Ziegler, D. (2022). BOND study: a randomised double-blind, placebo-controlled trial over 12 months to assess the effects of benfotiamine on morphometric, neurophysiological and clinical measures in patients with type 2 diabetes with symptomatic polyneuropathy. BMJ open, 12(2), e057142. [CrossRef]
- Bourebaba, N., Sikora, M., Qasem, B., Bourebaba, L., & Marycz, K. (2023). Sex hormone-binding globulin (SHBG) mitigates ER stress and improves viability and insulin sensitivity in adipose-derived mesenchymal stem cells (ASC) of equine metabolic syndrome (EMS)-affected horses. Cell communication and signaling : CCS, 21(1), 230. [CrossRef]
- Bozic, I., & Lavrnja, I. (2023). Thiamine and benfotiamine: Focus on their therapeutic potential. Heliyon, 9(11), e21839. [CrossRef]
- Brigelius-Flohé, R., & Flohé, L. (2020). Regulatory phenomena in the glutathione peroxidase superfamily. Antioxidants & Redox Signaling, *33*(7), 498–516. [CrossRef]
- Brinton, R. D., Yao, J., Yin, F., Mack, W. J., & Cadenas, E. (2015). Perimenopause as a neurological transition state. Nature reviews. Endocrinology, 11(7), 393–405. [CrossRef]
- Broekmans, F. J., Soules, M. R., & Fauser, B. C. (2009). Ovarian aging: mechanisms and clinical consequences. Endocrine reviews, 30(5), 465–493. [CrossRef]
- Calabrese, G., Morgan, B., & Riemer, J. (2017). Mitochondrial Glutathione: Regulation and Functions. Antioxidants & redox signaling, 27(15), 1162–1177. [CrossRef]
- Chen, Y., Yang, J., & Zhang, L. (2023). The Impact of Follicular Fluid Oxidative Stress Levels on the Outcomes of Assisted Reproductive Therapy. Antioxidants (Basel, Switzerland), 12(12), 2117. [CrossRef]
- Chen, Y., Zhang, J., Tian, Y., Xu, X., Wang, B., Huang, Z., Lou, S., Kang, J., Zhang, N., Weng, J., Liang, Y., & Ma, W. (2024). Iron accumulation in ovarian microenvironment damages the local redox balance and oocyte quality in aging mice. Redox biology, 73, 103195. [CrossRef]
- Cheng, B., Shi, Y., Wu, Q., Wang, Y., & Ma, Y. (2023). Selenium Protects Follicular Granulosa Cells from Apoptosis Induced by Mercury Through Inhibition of ATF6/CHOP Pathway in Laying Hens. Biological trace element research, 201(11), 5368–5378. [CrossRef]
- Cheraghi, E., Mehranjani, M. S., Shariatzadeh, M. A., Esfahani, M. H., & Ebrahimi, Z. (2016). N-Acetylcysteine improves oocyte and embryo quality in polycystic ovary syndrome patients undergoing intracytoplasmic sperm injection: an alternative to metformin. Reproduction, fertility, and development, 28(6), 723–731. [CrossRef]
- Cicek, N., Eryilmaz, O. G., Sarikaya, E., Gulerman, C., & Genc, Y. (2012). Vitamin E effect on controlled ovarian stimulation of unexplained infertile women. Journal of assisted reproduction and genetics, 29(4), 325–328. [CrossRef]
- Colella, M., Cuomo, D., Peluso, T., Falanga, I., Mallardo, M., De Felice, M., & Ambrosino, C. (2021). Ovarian Aging: Role of Pituitary-Ovarian Axis Hormones and ncRNAs in Regulating Ovarian Mitochondrial Activity. Frontiers in endocrinology, 12, 791071. [CrossRef]
- Cosme, P., Rodríguez, A. B., Garrido, M., & Espino, J. (2022). Coping with Oxidative Stress in Reproductive Pathophysiology and Assisted Reproduction: Melatonin as an Emerging Therapeutical Tool. Antioxidants (Basel, Switzerland), 12(1), 86. [CrossRef]
- Demay, M. B., Pittas, A. G., Bikle, D. D., Diab, D. L., Kiely, M. E., Lazaretti-Castro, M., Lips, P., Mitchell, D. M., Murad, M. H., Powers, S., Rao, S. D., Scragg, R., Tayek, J. A., Valent, A. M., Walsh, J. M. E., & McCartney, C. R. (2024). Vitamin D for the prevention of disease: An Endocrine Society clinical practice guideline. Journal of Clinical Endocrinology and Metabolism.
- Derakhshan, Z., Bahmanpour, S., Nasr-Esfahani, M. H., Masjedi, F., Mirani, M., Dara, M., & Tabei, S. M. B. (2024). Alpha-Lipoic Acid Ameliorates Impaired Steroidogenesis in Human Granulosa Cells Induced by Advanced Glycation End-Products. Iranian journal of medical sciences, 49(8), 515–527. [CrossRef]
- Diamanti-Kandarakis, E., Piperi, C., Patsouris, E., Korkolopoulou, P., Panidis, D., Pawelczyk, L., Papavassiliou, A. G., & Duleba, A. J. (2007). Immunohistochemical localization of advanced glycation end-products (AGEs) and their receptor (RAGE) in polycystic and normal ovaries. Histochemistry and cell biology, 127(6), 581–589. [CrossRef]
- Diamanti-Kandarakis, E., Chatzigeorgiou, A., Papageorgiou, E., Koundouras, D., & Koutsilieris, M. (2016). Advanced glycation end-products and insulin signaling in granulosa cells. Experimental biology and medicine (Maywood, N.J.), 241(13), 1438–1445. [CrossRef]
- Ding, N., Wang, X., Harlow, S. D., Randolph, J. F., Jr, Gold, E. B., & Park, S. K. (2024). Heavy Metals and Trajectories of Anti-Müllerian Hormone During the Menopausal Transition. The Journal of clinical endocrinology and metabolism, 109(11), e2057–e2064. [CrossRef]
- Di Nicuolo, F., D'Ippolito, S., Castellani, R., Rossi, E. D., Masciullo, V., Specchia, M., Mariani, M., Pontecorvi, A., Scambia, G., & Di Simone, N. (2019). Effect of alpha-lipoic acid and myoinositol on endometrial inflammasome from recurrent pregnancy loss women. American Journal of Reproductive Immunology, 82(4), Article e13153. [CrossRef]
- Dong, H., Zhang, Y., Huang, Y., & Deng, H. (2022). Pathophysiology of RAGE in inflammatory diseases. Frontiers in immunology, 13, 931473. [CrossRef]
- Duncan, F. E., Que, E. L., Zhang, N., Feinberg, E. C., O'Halloran, T. V., & Woodruff, T. K. (2016). The zinc spark is an inorganic signature of human egg activation. Scientific reports, 6, 24737. [CrossRef]
- Eddie-Amadi, B. F., Ezejiofor, A. N., Orish, C. N., & Orisakwe, O. E. (2022). Zn and Se abrogate heavy metal mixture induced ovarian and thyroid oxido-inflammatory effects mediated by activation of NRF2-HMOX-1 in female albino rats. Current research in toxicology, 4, 100098. [CrossRef]
- Es-Sai, B., Wahnou, H., Benayad, S., Rabbaa, S., Laaziouez, Y., El Kebbaj, R., Limami, Y., & Duval, R. E. (2025). Gamma-Tocopherol: A Comprehensive Review of Its Antioxidant, Anti-Inflammatory, and Anticancer Properties. Molecules (Basel, Switzerland), 30(3), 653. [CrossRef]
- Fujita, K., Shindo, Y., Katsuta, Y., Goto, M., Hotta, K., & Oka, K. (2023). Intracellular Mg2+ protects mitochondria from oxidative stress in human keratinocytes. Communications biology, 6(1), 868. [CrossRef]
- Gantt, S. L., Denisov, I. G., Grinkova, Y. V., & Sligar, S. G. (2009). The critical iron-oxygen intermediate in human aromatase. Biochemical and biophysical research communications, 387(1), 169–173. [CrossRef]
- Garg, D., & Merhi, Z. (2016). Relationship between Advanced Glycation End Products and Steroidogenesis in PCOS. Reproductive biology and endocrinology : RB&E, 14(1), 71. [CrossRef]
- Garner, T. B., Hester, J. M., Carothers, A., & Diaz, F. J. (2021). Role of zinc in female reproduction. Biology of Reproduction, 104(5), 976–994. [CrossRef]
- Gholami, M., Samaei, S. E., Shaki, F., Etemadinezhad, S., & Sayyad, M. S. (2025). Evaluation of the protective effect of Benfotiamine against neurotoxic effects, depression and oxidative stress induced by noise exposure. Scientific reports, 15(1), 31336. [CrossRef]
- Guarano, A., Capozzi, A., Cristodoro, M., Di Simone, N., & Lello, S. (2023). Alpha Lipoic Acid Efficacy in PCOS Treatment: What Is the Truth?. Nutrients, 15(14), 3209. [CrossRef]
- Gyengesi, E., Paxinos, G., & Andrews, Z. B. (2012). Oxidative stress in the hypothalamus: the importance of calcium signaling and mitochondrial ROS in body weight regulation. Current Neuropharmacology, 10(4), 344 - 353. [CrossRef]
- Hall J. E. (2007). Neuroendocrine changes with reproductive aging in women. Seminars in reproductive medicine, 25(5), 344–351. [CrossRef]
- Hirano, M., Onodera, T., Takasaki, K., Takahashi, Y., Ichinose, T., Nishida, H., Hiraike, H., & Nagasaka, K. (2025). Ovarian aging: pathophysiology and recent developments in maintaining ovarian reserve. Frontiers in Endocrinology, 16, 1619516. [CrossRef]
- Hu, J., Wang, H., Fang, J., Jiang, R., Kong, Y., Zhang, T., Yang, G., Jin, H., Shi, S., Song, N., Qi, L., Huang, X., Wu, Z., & Yao, G. (2025). Ovarian aging-associated downregulation of GPX4 expression regulates ovarian follicular development by affecting granulosa cell functions and oocyte quality. FASEB journal : official publication of the Federation of American Societies for Experimental Biology, 39(6), e70469. [CrossRef]
- Huang, Y., Hu, C., Ye, H., Luo, R., Fu, X., Li, X., Huang, J., Chen, W., & Zheng, Y. (2019). Inflamm-Aging: A New Mechanism Affecting Premature Ovarian Insufficiency. Journal of immunology research, 2019, 8069898. [CrossRef]
- Hummitzsch, K., Kelly, J. E., Hatzirodos, N., Bonner, W. M., Tang, F., Harris, H. H., & Rodgers, R. J. (2024). Expression levels of the selenium-uptake receptor LRP8, the antioxidant selenoprotein GPX1 and steroidogenic enzymes correlate in granulosa cells. Reproduction & fertility, 5(3), e230074. [CrossRef]
- Ji, T., Chen, X., Zhang, Y., Fu, K., Zou, Y., Wang, W., & Zhao, J. (2022). Effects of N-Acetylcysteine on the Proliferation, Hormone Secretion Level, and Gene Expression Profiles of Goat Ovarian Granulosa Cells. Genes, 13(12), 2306. [CrossRef]
- Jiang, Y., Han, Y., Qiao, P., & Ren, F. (2025). Exploring the protective effects of coenzyme Q10 on female fertility. Frontiers in cell and developmental biology, 13, 1633166. [CrossRef]
- Jomova, K., Baros, S., & Valko, M. (2012). Redox active metal-induced oxidative stress in biological systems. Transition Metal Chemistry, 37, 127–134. [CrossRef]
- Ju, W., Zhao, Y., Yu, Y., Zhao, S., Xiang, S., & Lian, F. (2024). Mechanisms of mitochondrial dysfunction in ovarian aging and potential interventions. Frontiers in endocrinology, 15, 1361289. [CrossRef]
- Ju, W., Yan, B., Li, D., Lian, F., & Xiang, S. (2025). Mitochondria-driven inflammation: a new frontier in ovarian ageing. Journal of translational medicine, 23(1), 1005. [CrossRef]
- Kandaraki, E. A., Chatzigeorgiou, A., Papageorgiou, E., Piperi, C., Adamopoulos, C., Papavassiliou, A. G., Koutsilieris, M., & Diamanti-Kandarakis, E. (2018). Advanced glycation end products interfere in luteinizing hormone and follicle stimulating hormone signaling in human granulosa KGN cells. Experimental biology and medicine (Maywood, N.J.), 243(1), 29–33. [CrossRef]
- Kapper, C., Oppelt, P., Ganhör, C., Gyunesh, A. A., Arbeithuber, B., Stelzl, P., & Rezk-Füreder, M. (2024). Minerals and the Menstrual Cycle: Impacts on Ovulation and Endometrial Health. Nutrients, 16(7), 1008. [CrossRef]
- Klein, N. A., & Soules, M. R. (1998). Endocrine changes of the perimenopause. Clinical obstetrics and gynecology, 41(4), 912–920. [CrossRef]
- Kobayashi, N., Machida, T., Takahashi, T., Takatsu, H., Shinkai, T., Abe, K., & Urano, S. (2009). Elevation by oxidative stress and aging of hypothalamic-pituitary-adrenal activity in rats and its prevention by vitamin e. Journal of clinical biochemistry and nutrition, 45(2), 207–213. [CrossRef]
- Kong, B. Y., Bernhardt, M. L., Kim, A. M., O'Halloran, T. V., & Woodruff, T. K. (2012). Zinc maintains prophase I arrest in mouse oocytes through regulation of the MOS-MAPK pathway. Biology of reproduction, 87(1), 11–12. [CrossRef]
- Korytowski, W., Pilat, A., Schmitt, J. C., & Girotti, A. W. (2013). Deleterious cholesterol hydroperoxide trafficking in steroidogenic acute regulatory (StAR) protein-expressing MA-10 Leydig cells: implications for oxidative stress-impaired steroidogenesis. The Journal of biological chemistry, 288(16), 11509–11519. [CrossRef]
- Kornicka-Garbowska, K., Bourebaba, L., Röcken, M., & Marycz, K. (2021). Sex Hormone Binding Globulin (SHBG) Mitigates ER Stress in Hepatocytes In Vitro and Ex Vivo. Cells, 10(4), 755. [CrossRef]
- Krsmanović, L. Z., Stojilković, S. S., Merelli, F., Dufour, S. M., Virmani, M. A., & Catt, K. J. (1992). Calcium signaling and episodic secretion of gonadotropin-releasing hormone in hypothalamic neurons. Proceedings of the National Academy of Sciences of the United States of America, 89(18), 8462–8466. [CrossRef]
- Kumar, P., Liu, C., Hsu, J. W., Chacko, S., Minard, C., Jahoor, F., & Sekhar, R. V. (2021). Glycine and N-acetylcysteine (GlyNAC) supplementation in older adults improves glutathione deficiency, oxidative stress, mitochondrial dysfunction, inflammation, insulin resistance, endothelial dysfunction, genotoxicity, muscle strength, and cognition: Results of a pilot clinical trial. Clinical and translational medicine, 11(3), e372. [CrossRef]
- Kumar, P., Liu, C., Suliburk, J., Hsu, J. W., Muthupillai, R., Jahoor, F., Minard, C. G., Taffet, G. E., & Sekhar, R. V. (2023). Supplementing Glycine and N-Acetylcysteine (GlyNAC) in Older Adults Improves Glutathione Deficiency, Oxidative Stress, Mitochondrial Dysfunction, Inflammation, Physical Function, and Aging Hallmarks: A Randomized Clinical Trial. The journals of gerontology. Series A, Biological sciences and medical sciences, 78(1), 75–89. [CrossRef]
- Kushnir, V. A., Ludaway, T., Russ, R. B., Fields, E. J., Koczor, C., & Lewis, W. (2012). Reproductive aging is associated with decreased mitochondrial abundance and altered structure in murine oocytes. Journal of assisted reproduction and genetics, 29(7), 637–642. [CrossRef]
- Lafuente A. (2013). The hypothalamic-pituitary-gonadal axis is target of cadmium toxicity. An update of recent studies and potential therapeutic approaches. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 59, 395–404. [CrossRef]
- Lai, X.-L., Xiong, W.-J., Li, L.-S., Lan, M.-F., Zhang, J.-X., Zhou, Y.-T., Niu, D., & Duan, X. (2023). Zinc deficiency compromises the maturational competence of porcine oocyte by inducing mitophagy and apoptosis. Ecotoxicology and Environmental Safety, 252, Article 114593. [CrossRef]
- Lava Kumar, S., Kushawaha, B., Mohanty, A., Kumari, A., Kumar, A., Beniwal, R., Kiran Kumar, P., Athar, M., Krishna Rao, D., & Rao, H. B. D. P. (2024). Glutathione peroxidase (GPX1) - Selenocysteine metabolism preserves the follicular fluid's (FF) redox homeostasis via IGF-1- NMD cascade in follicular ovarian cysts (FOCs). Biochimica et biophysica acta. Molecular basis of disease, 1870(6), 167235. [CrossRef]
- Li, C. J., Lin, L. T., Tsai, H. W., Chern, C. U., Wen, Z. H., Wang, P. H., & Tsui, K. H. (2021). The Molecular Regulation in the Pathophysiology in Ovarian Aging. Aging and disease, 12(3), 934–949. [CrossRef]
- Li, M., Hu, S., Sun, J., & Zhang, Y. (2024). The role of vitamin D3 in follicle development. Journal of ovarian research, 17(1), 148. [CrossRef]
- Li, Y., Zhao, T., Li, J., Xia, M., Li, Y., Wang, X., Liu, C., Zheng, T., Chen, R., Kan, D., Xie, Y., Song, J., Feng, Y., Yu, T., & Sun, P. (2022). Oxidative Stress and 4-hydroxy-2-nonenal (4-HNE): Implications in the Pathogenesis and Treatment of Aging-related Diseases. Journal of immunology research, 2022, 2233906. [CrossRef]
- Liew, S. H., Nguyen, Q. N., Strasser, A., Findlay, J. K., & Hutt, K. J. (2017). The ovarian reserve is depleted during puberty in a hormonally driven process dependent on the pro-apoptotic protein BMF. Cell death & disease, 8(8), e2971. [CrossRef]
- Lim, J., & Luderer, U. (2011). Oxidative damage increases and antioxidant gene expression decreases with aging in the mouse ovary. Biology of reproduction, 84(4), 775–782. [CrossRef]
- Lin, G., Li, X., Jin Yie, S. L., & Xu, L. (2024). Clinical evidence of coenzyme Q10 pretreatment for women with diminished ovarian reserve undergoing IVF/ICSI: a systematic review and meta-analysis. Annals of medicine, 56(1), 2389469. [CrossRef]
- Liu, S., Jia, Y., Meng, S., Luo, Y., Yang, Q., & Pan, Z. (2023). Mechanisms of and Potential Medications for Oxidative Stress in Ovarian Granulosa Cells: A Review. International journal of molecular sciences, 24(11), 9205. [CrossRef]
- Liu, W. J., Li, L. S., Lan, M. F., Shang, J. Z., Zhang, J. X., Xiong, W. J., Lai, X. L., & Duan, X. (2024). Zinc deficiency deteriorates ovarian follicle development and function by inhibiting mitochondrial function. Journal of ovarian research, 17(1), 115. [CrossRef]
- Liu, Y., Cheng, G., Li, H., & Meng, Q. (2025). Serum copper to zinc ratio and risk of endometriosis: Insights from a case-control study in infertile patients. Reproductive medicine and biology, 24(1), e12644. [CrossRef]
- Liu, X., Lin, X., Zhang, S., Guo, C., Li, J., Mi, Y., & Zhang, C. (2018). Lycopene ameliorates oxidative stress in the aging chicken ovary via activation of Nrf2/HO-1 pathway. Aging, 10(8), 2016–2036. [CrossRef]
- Lizcano F. (2022). Roles of estrogens, estrogen-like compounds, and endocrine disruptors in adipocytes. Frontiers in endocrinology, 13, 921504. [CrossRef]
- Lord, T., Martin, J. H., & Aitken, R. J. (2015). Accumulation of electrophilic aldehydes during postovulatory aging of mouse oocytes causes reduced fertility, oxidative stress, and apoptosis. Biology of reproduction, 92(2), 33. [CrossRef]
- Lord, T. (2025). The role of reactive oxygen species and oxidative stress in post-ovulatory ageing and apoptosis of the mammalian oocyte [Doctoral dissertation, University of Newcastle]. Open Research Newcastle. https://hdl.handle.net/1959.13/1310421.
- Lou, Y., Yang, T., Xia, C., Yang, S., Deng, H., Zhu, Y., Fang, J., Zuo, Z., & Guo, H. (2025). Effects of Copper on Steroid Hormone Secretion, Steroidogenic Enzyme Expression, and Transcriptomic Profiles in Yak Ovarian Granulosa Cells. Veterinary sciences, 12(5), 428. [CrossRef]
- Lu, C., He, J. C., Cai, W., Liu, H., Zhu, L., & Vlassara, H. (2004). Advanced glycation endproduct (AGE) receptor 1 is a negative regulator of the inflammatory response to AGE in mesangial cells. Proceedings of the National Academy of Sciences of the United States of America, 101(32), 11767–11772. [CrossRef]
- Luddi, A., Capaldo, A., Focarelli, R., Gori, M., Morgante, G., Piomboni, P., & De Leo, V. (2016). Antioxidants reduce oxidative stress in follicular fluid of aged women undergoing IVF. Reproductive biology and endocrinology : RB&E, 14(1), 57. [CrossRef]
- Mahsa Poormoosavi, S., Behmanesh, M. A., Varzi, H. N., Mansouri, S., & Janati, S. (2021). The effect of follicular fluid selenium concentration on oocyte maturation in women with polycystic ovary syndrome undergoing in vitro fertilization/Intracytoplasmic sperm injection: A cross-sectional study. International journal of reproductive biomedicine, 19(8), 689–698. [CrossRef]
- Maldonado-Cárceles, A. B., Souter, I., Li, M. C., Mitsunami, M., Dimitriadis, I., Ford, J. B., Mínguez-Alarcón, L., Chavarro, J. E., & EARTH Study Team (2025). Antioxidant Intake and Ovarian Reserve in Women Attending a Fertility Center. Nutrients, 17(3), 554. [CrossRef]
- Mashhadi, F., Sedghi, Z., Hemmat, A., Rivaz, R., & Roudi, F. (2025). Nutritional interventions for enhancing female fertility: A comprehensive review of micronutrients and their impact. Nursing Research and Practice, 2137328. [CrossRef]
- Md Amin, N. A., Sheikh Abdul Kadir, S. H., Arshad, A. H., Abdul Aziz, N., Abdul Nasir, N. A., & Ab Latip, N. (2022). Are Vitamin E Supplementation Beneficial for Female Gynaecology Health and Diseases?. Molecules (Basel, Switzerland), 27(6), 1896. [CrossRef]
- Mei, Y., Liu, J., Dong, Z., Ke, S., Su, W., Luo, Z., Zhao, J., Li, Y., Yang, M., Wu, J., Su, W., & Li, A. (2025). Heavy metals exposure and HPG-axis related hormones in women across the lifespan: An integrative epidemiological and bioinformatic perspective. Ecotoxicology and environmental safety, 303, 118962. [CrossRef]
- Meng, X., Zhang, J., Wan, Q., Huang, J., Han, T., Qu, T., & Yu, L. L. (2023). Influence of Vitamin D supplementation on reproductive outcomes of infertile patients: a systematic review and meta-analysis. Reproductive biology and endocrinology : RB&E, 21(1), 17. [CrossRef]
- Merhi Z. (2014). Advanced glycation end products and their relevance in female reproduction. Human reproduction (Oxford, England), 29(1), 135–145. [CrossRef]
- Merhi, Z., Goldsammler, M., Charron, M. J., & Buyuk, E. (2025). Dietary advanced glycation end products (AGEs) and superovulation with gonadotropins alter RAGE expression in the ovaries differently at each follicular stage of development. Molecular and cellular endocrinology, 599, 112482. [CrossRef]
- Mezzetti, A., Pierdomenico, S. D., Costantini, F., Romano, F., De Cesare, D., Cuccurullo, F., Imbastaro, T., Riario-Sforza, G., Di Giacomo, F., Zuliani, G., & Fellin, R. (1998). Copper/zinc ratio and systemic oxidant load: effect of aging and aging-related degenerative diseases. Free radical biology & medicine, 25(6), 676–681. [CrossRef]
- Mihalas, B. P., De Iuliis, G. N., Redgrove, K. A., McLaughlin, E. A., & Nixon, B. (2017). The lipid peroxidation product 4-hydroxynonenal contributes to oxidative stress-mediated deterioration of the ageing oocyte. Scientific reports, 7(1), 6247. [CrossRef]
- Mikwar, M., MacFarlane, A. J., & Marchetti, F. (2020). Mechanisms of oocyte aneuploidy associated with advanced maternal age. Mutation research. Reviews in mutation research, 785, 108320. [CrossRef]
- Miler, E. A., Nudler, S. I., Quinteros, F. A., Cabilla, J. P., Ronchetti, S. A., & Duvilanski, B. H. (2010). Cadmium induced-oxidative stress in pituitary gland is reversed by removing the contamination source. Human & experimental toxicology, 29(10), 873–880. [CrossRef]
- Mocchegiani E. (2007). Zinc and ageing: third Zincage conference. Immunity & ageing : I & A, 4, 5. [CrossRef]
- Mokhtari, S., Mahdavi, A. H., Jafarpour, F., Andani, M. R., Dattilo, M., & Nasr-Esfahani, M. H. (2024). Taurine, alpha lipoic acid and vitamin B6 ameliorate the reduced developmental competence of immature mouse oocytes exposed to methylglyoxal. Scientific reports, 14(1), 17937. [CrossRef]
- Mouanness, M., & Merhi, Z. (2022). Impact of Dietary Advanced Glycation End Products on Female Reproduction: Review of Potential Mechanistic Pathways. Nutrients, 14(5), 966. [CrossRef]
- Moldogazieva, N. T., Zavadskiy, S. P., Astakhov, D. V., & Terentiev, A. A. (2023). Lipid peroxidation: Reactive carbonyl species, protein/DNA adducts, and signaling switches in oxidative stress and cancer. Biochemical and biophysical research communications, 687, 149167. [CrossRef]
- National Institute of Diabetes and Digestive and Kidney Diseases. (2024). Trace elements and metals. In LiverTox: Clinical and research information on drug-induced liver injury. U.S. Department of Health and Human Services. https://www.ncbi.nlm.nih.gov/books/NBK548854/.
- National Institutes of Health, Office of Dietary Supplements. (2025). Magnesium – Health professional fact sheet. U.S. Department of Health and Human Services. https://ods.od.nih.gov/factsheets/Magnesium-HealthProfessional/.
- Nelson-Goedert, N. (2025). The conglomerate theory of aging: A unified model of metal, AGE, and ALE accumulation. Preprints. [CrossRef]
- Nelson-Goedert, N. (2025). The conglomerate theory of oncogenesis: A unified biophysical framework for understanding cancer through bioaccumulation. Preprints. [CrossRef]
- Nelson-Goedert, N. (2025). Geropsychosis: A biophysical framework for understanding psychiatric symptoms as accelerated aging pathologies. Preprints. [CrossRef]
- Ni, Z., Li, Y., Song, D., Ding, J., Mei, S., Sun, S., Cheng, W., Yu, J., Zhou, L., Kuang, Y., Li, M., Cai, Z., & Yu, C. (2022). Iron-overloaded follicular fluid increases the risk of endometriosis-related infertility by triggering granulosa cell ferroptosis and oocyte dysmaturity. Cell death & disease, 13(7), 579. [CrossRef]
- Noviyana, A., Soetrisno, S., Prayitno, A., Cilmiaty, R., Laqif, A., Widyaningsih, V., & Dirgahayu, P. (2025). Magnesium supplementation improves ovarian folliculogenesis in type 1 diabetes mellitus. Pharmacia, 72, 1–9. [CrossRef]
- Niu, Y. J., Zhou, W., Nie, Z. W., Zhou, D., Xu, Y. N., Ock, S. A., Yan, C. G., & Cui, X. S. (2020). Ubiquinol-10 delays postovulatory oocyte aging by improving mitochondrial renewal in pigs. Aging, 12(2), 1256–1271. [CrossRef]
- Office of Dietary Supplements. (2025, September 4). Selenium fact sheet for consumers [Fact sheet]. https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/.
- Pamphlett, R., Kum Jew, S., Doble, P. A., & Bishop, D. P. (2019). Elemental Analysis of Aging Human Pituitary Glands Implicates Mercury as a Contributor to the Somatopause. Frontiers in endocrinology, 10, 419. [CrossRef]
- Pamplona R. (2008). Membrane phospholipids, lipoxidative damage and molecular integrity: a causal role in aging and longevity. Biochimica et biophysica acta, 1777(10), 1249–1262. [CrossRef]
- Pennarossa, G., De Iorio, T., Gandolfi, F., & Brevini, T. A. L. (2022). Impact of Aging on the Ovarian Extracellular Matrix and Derived 3D Scaffolds. Nanomaterials (Basel, Switzerland), 12(3), 345. [CrossRef]
- Pertynska-Marczewska, M., & Diamanti-Kandarakis, E. (2017). Aging ovary and the role for advanced glycation end products. Menopause (New York, N.Y.), 24(3), 345–351. [CrossRef]
- Pollack, A. Z., Schisterman, E. F., Goldman, L. R., Mumford, S. L., Albert, P. S., Jones, R. L., & Wactawski-Wende, J. (2011). Cadmium, lead, and mercury in relation to reproductive hormones and anovulation in premenopausal women. Environmental health perspectives, 119(8), 1156–1161. [CrossRef]
- Prokopieva, V. D., Yarygina, E. G., Bokhan, N. A., & Ivanova, S. A. (2016). Use of Carnosine for Oxidative Stress Reduction in Different Pathologies. Oxidative medicine and cellular longevity, 2016, 2939087. [CrossRef]
- Que, E. L., Duncan, F. E., Bayer, A. R., Philips, S. J., Roth, E. W., Bleher, R., Gleber, S. C., Vogt, S., Woodruff, T. K., & O'Halloran, T. V. (2017). Zinc sparks induce physiochemical changes in the egg zona pellucida that prevent polyspermy. Integrative biology : quantitative biosciences from nano to macro, 9(2), 135–144. [CrossRef]
- Rakha, S. I., Elmetwally, M. A., El-Sheikh Ali, H., Balboula, A. Z., Mahmoud, A. M., & Zaabel, S. M. (2022). Lycopene Reduces the In Vitro Aging Phenotypes of Mouse Oocytes by Improving Their Oxidative Status. Veterinary sciences, 9(7), 336. [CrossRef]
- Rance N. E. (2009). Menopause and the human hypothalamus: evidence for the role of kisspeptin/neurokinin B neurons in the regulation of estrogen negative feedback. Peptides, 30(1), 111–122. [CrossRef]
- Rijal, S., Jang, S. H., Cho, D. H., & Han, S. K. (2022). Hydrogen peroxide suppresses excitability of gonadotropin-releasing hormone neurons in adult mouse. Frontiers in endocrinology, 13, 939699. [CrossRef]
- Rungratanawanich, W., Qu, Y., Wang, X., Essa, M. M., & Song, B. J. (2021). Advanced glycation end products (AGEs) and other adducts in aging-related diseases and alcohol-mediated tissue injury. Experimental & molecular medicine, 53(2), 168–188. [CrossRef]
- Santiago-Andres, Y., Hernández Álvarez, E., Ochoa Gutierrez, D., Morton Bermea, O., & Fiordelisio, T. (2025). In vivo effects of cadmium on signaling and secretion of pituitary gonadotrophs in male mice are time-dependent. The Journal of endocrinology, 267(1), e250161. [CrossRef]
- Sasaki, H., Hamatani, T., Kamijo, S., Iwai, M., Kobanawa, M., Ogawa, S., Miyado, K., & Tanaka, M. (2019). Impact of Oxidative Stress on Age-Associated Decline in Oocyte Developmental Competence. Frontiers in endocrinology, 10, 811. [CrossRef]
- Schmid, U., Stopper, H., Heidland, A., & Schupp, N. (2008). Benfotiamine exhibits direct antioxidative capacity and prevents induction of DNA damage in vitro. Diabetes/Metabolism Research and Reviews, 24(5), 371–377. [CrossRef]
- Sissi, C., & Palumbo, M. (2009). Effects of magnesium and related divalent metal ions in topoisomerase structure and function. Nucleic acids research, 37(3), 702–711. [CrossRef]
- Szczuko, M., Szydłowska, I., & Nawrocka-Rutkowska, J. (2021). A Properly Balanced Reduction Diet and/or Supplementation Solve the Problem with the Deficiency of These Vitamins Soluble in Water in Patients with PCOS. Nutrients, 13(3), 746. [CrossRef]
- Shao, Y., Ma, L., Zhou, J., Wu, K., & Tang, X. (2024). Impact of dietary antioxidants on female infertility risk: evidence from NHANES. Scientific reports, 14(1), 22623. [CrossRef]
- Shen, L., Liu, J., Luo, A., & Wang, S. (2023). The stromal microenvironment and ovarian aging: mechanisms and therapeutic opportunities. Journal of ovarian research, 16(1), 237. [CrossRef]
- Shi, Y. Q., Zhu, X. T., Zhang, S. N., Ma, Y. F., Han, Y. H., Jiang, Y., & Zhang, Y. H. (2023). Premature ovarian insufficiency: a review on the role of oxidative stress and the application of antioxidants. Frontiers in endocrinology, 14, 1172481. [CrossRef]
- Smits, M. A. J., Schomakers, B. V., van Weeghel, M., Wever, E. J. M., Wüst, R. C. I., Dijk, F., Janssens, G. E., Goddijn, M., Mastenbroek, S., Houtkooper, R. H., & Hamer, G. (2023). Human ovarian aging is characterized by oxidative damage and mitochondrial dysfunction. Human reproduction (Oxford, England), 38(11), 2208–2220. [CrossRef]
- Sundas, R., Ejaz, R., Ashfaq, S., Qadeer, S., Azam, A., Saeed, W., Khan, N., Saleem, R., & Husna, A. U. (2024). Effects of magnesium supplementation on anti-Mullerian hormone level in premenopausal women. Journal of Population Therapeutics and Clinical Pharmacology, 31(2), 298–306. [CrossRef]
- Tang, W., Wang, K., Feng, Y., Tsui, K. H., Singh, K. K., Stout, M. B., Wang, S., & Wu, M. (2025). Exploration of the mechanism and therapy of ovarian aging by targeting cellular senescence. Life medicine, 4(1), lnaf004. [CrossRef]
- Tatone, C., Carbone, M. C., Falone, S., Aimola, P., Giardinelli, A., Caserta, D., Marci, R., Pandolfi, A., Ragnelli, A. M., & Amicarelli, F. (2006). Age-dependent changes in the expression of superoxide dismutases and catalase are associated with ultrastructural modifications in human granulosa cells. Molecular human reproduction, 12(11), 655–660. [CrossRef]
- Terasaka, T., Adakama, M. E., Li, S., Kim, T., Terasaka, E., Li, D., & Lawson, M. A. (2017). Reactive Oxygen Species Link Gonadotropin-Releasing Hormone Receptor Signaling Cascades in the Gonadotrope. Frontiers in endocrinology, 8, 286. [CrossRef]
- Thornton, K., Merhi, Z., Jindal, S., Goldsammler, M., Charron, M. J., & Buyuk, E. (2020). Dietary Advanced Glycation End Products (AGEs) could alter ovarian function in mice. Molecular and cellular endocrinology, 510, 110826. [CrossRef]
- Tóbon-Velasco, J. C., Cuevas, E., & Torres-Ramos, M. A. (2014). Receptor for AGEs (RAGE) as mediator of NF-kB pathway activation in neuroinflammation and oxidative stress. CNS & neurological disorders drug targets, 13(9), 1615–1626. [CrossRef]
- Tong, L., Zhang, X., Chen, J., He, H., Zhang, W., Wan, Z., & Zhang, H. (2025). The associations between dietary advanced glycation-end products intake and self-reported infertility in U.S. women: data from the NHANES 2013-2018. Scientific reports, 15(1), 1158. [CrossRef]
- Tran, B. N., Ha, T. B. N., Bui, P. M., Dam, H. T., To, K. A., Pham, T. A., & Le, T. (2025). Magnesium chelation of low molecular weight peptides from protein-rich industrial wastes: Production and properties. Journal, genetic engineering & biotechnology, 23(3), 100528. [CrossRef]
- Urzúa, U., Marín, A., & Castellón, E. A. (2025). Oxidative Stress, Parity History, and Remnant Follicles in the Aged Ovary: Insights on Ovarian Cancer Risk and Protection. Antioxidants (Basel, Switzerland), 14(7), 759. [CrossRef]
- van der Reest, J., Nardini Cecchino, G., Haigis, M. C., & Kordowitzki, P. (2021). Mitochondria: Their relevance during oocyte ageing. Ageing research reviews, 70, 101378. [CrossRef]
- Walker, D. M., & Gore, A. C. (2017). Epigenetic impacts of endocrine disruptors in the brain. Frontiers in neuroendocrinology, 44, 1–26. [CrossRef]
- Wang, F., Zhang, Z., Han, J., Zheng, J., Wang, X., & Wang, Z. (2023). Discovery of microglia gonadotropin-releasing hormone receptor and its potential role in polycystic ovarian syndrome. Molecular medicine reports, 27(4), 77. [CrossRef]
- Wang, G., Yang, R., & Zhang, H. (2025). Ovarian vascular aging: a hidden driver of mid-age female fertility decline. npj aging, 11(1), 24. [CrossRef]
- Wang, X., Ding, N., Harlow, S. D., Randolph, J. F., Jr, Mukherjee, B., Gold, E. B., & Park, S. K. (2021). Urinary metals and metal mixtures and timing of natural menopause in midlife women: The Study of Women's Health Across the Nation. Environment international, 157, 106781. [CrossRef]
- Wang, X., Ding, N., Harlow, S. D., Randolph, J. F., Jr, Mukherjee, B., Gold, E. B., & Park, S. K. (2023). Exposure to heavy metals and hormone levels in midlife women: The Study of Women's Health Across the Nation (SWAN). Environmental pollution (Barking, Essex : 1987), 317, 120740. [CrossRef]
- Weiss, G., Skurnick, J. H., Goldsmith, L. T., Santoro, N. F., & Park, S. J. (2004). Menopause and hypothalamic-pituitary sensitivity to estrogen. JAMA, 292(24), 2991–2996. [CrossRef]
- Xiao, C., & Lai, D. (2025). Impact of oxidative stress induced by heavy metals on ovarian function. Journal of applied toxicology : JAT, 45(1), 107–116. [CrossRef]
- Xu, C., Liu, W. B., Zhang, D. D., Shi, H. J., Zhang, L., & Li, X. F. (2018). Benfotiamine, a Lipid-Soluble Analog of Vitamin B1, Improves the Mitochondrial Biogenesis and Function in Blunt Snout Bream (Megalobrama amblycephala) Fed High-Carbohydrate Diets by Promoting the AMPK/PGC-1β/NRF-1 Axis. Frontiers in physiology, 9, 1079. [CrossRef]
- Xu, Y., Nisenblat, V., Lu, C., Li, R., Qiao, J., Zhen, X., & Wang, S. (2018). Pretreatment with coenzyme Q10 improves ovarian response and embryo quality in low-prognosis young women with decreased ovarian reserve: a randomized controlled trial. Reproductive biology and endocrinology : RB&E, 16(1), 29. [CrossRef]
- Yan, F., Zhao, Q., Li, Y., Zheng, Z., Kong, X., Shu, C., Liu, Y., & Shi, Y. (2022). The role of oxidative stress in ovarian aging: a review. Journal of ovarian research, 15(1), 100. [CrossRef]
- Yang, H., Qazi, I. H., Pan, B., Angel, C., Guo, S., Yang, J., Zhang, Y., Ming, Z., Zeng, C., Meng, Q., Han, H., & Zhou, G. (2019). Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice. Antioxidants (Basel, Switzerland), 8(12), 634. [CrossRef]
- Yang, L., Chen, Y., Liu, Y., Xing, Y., Miao, C., Zhao, Y., Chang, X., & Zhang, Q. (2021). The Role of Oxidative Stress and Natural Antioxidants in Ovarian Aging. Frontiers in pharmacology, 11, 617843. [CrossRef]
- Yang, M., Shen, X., Lu, D., Peng, J., Zhou, S., Xu, L., & Zhang, J. (2023). Effects of vitamin D supplementation on ovulation and pregnancy in women with polycystic ovary syndrome: A systematic review and meta-analysis. Frontiers in Endocrinology, 14, 1148556. [CrossRef]
- Yang, P. M., Chen, H. Z., Huang, Y. T., Hsieh, C. W., & Wung, B. S. (2017). Lycopene inhibits NF-κB activation and adhesion molecule expression through Nrf2-mediated heme oxygenase-1 in endothelial cells. International journal of molecular medicine, 39(6), 1533–1450. [CrossRef]
- Yao, X., Zhang, G., Guo, Y., El-Samahy, M., Wang, S., Wan, Y., Han, L., Liu, Z., Wang, F., & Zhang, Y. (2017). Vitamin D receptor expression and potential role of vitamin D on cell proliferation and steroidogenesis in goat ovarian granulosa cells. Theriogenology, 102, 162–173. [CrossRef]
- Yao, Y., Tang, Y., Qin, H., Meng, R., Zhang, C., Zhang, Y., Yang, Y., Qiao, P., Liu, J., & Su, J. (2023). Zinc supplementation promotes oocyte maturation and subsequent embryonic development in sheep. Theriogenology, 206, 161-169. [CrossRef]
- Yiqin, C., Yan, S., Peiwen, W., Yiwei, G., Qi, W., Qian, X., Panglin, W., Sunjie, Y., & Wenxiang, W. (2022). Copper exposure disrupts ovarian steroidogenesis in human ovarian granulosa cells via the FSHR/CYP19A1 pathway and alters methylation patterns on the SF-1 gene promoter. Toxicology letters, 356, 11–20. [CrossRef]
- Zhai, Q., Narbad, A., & Chen, W. (2015). Dietary strategies for the treatment of cadmium and lead toxicity. Nutrients, 7(1), 552–571. [CrossRef]
- Zhang, M., Sun, L., Zhang, Z., Shentu, L., Zhang, Y., Li, Z., Zhang, Y., & Zhang, Y. (2022). Alpha-lipoic acid supplementation restores the meiotic competency and fertilization capacity of porcine oocytes induced by arsenite. Frontiers in cell and developmental biology, 10, 943757. [CrossRef]
- Zhang, W., Cui, Y., & Liu, J. (2023). The association between blood heavy metals level and sex hormones among postmenopausal women in the US. Frontiers in endocrinology, 14, 1175011. [CrossRef]
- Zhu, Y., Sun, H., Gao, T., Hou, S., Li, Y., Xu, Y., Zhang, Q., & Feng, D. (2025). Ovarian remodeling and aging-related chronic inflammation and fibrosis in the mammalian ovary. Journal of ovarian research, 18(1), 133. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



