Submitted:
17 October 2025
Posted:
20 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
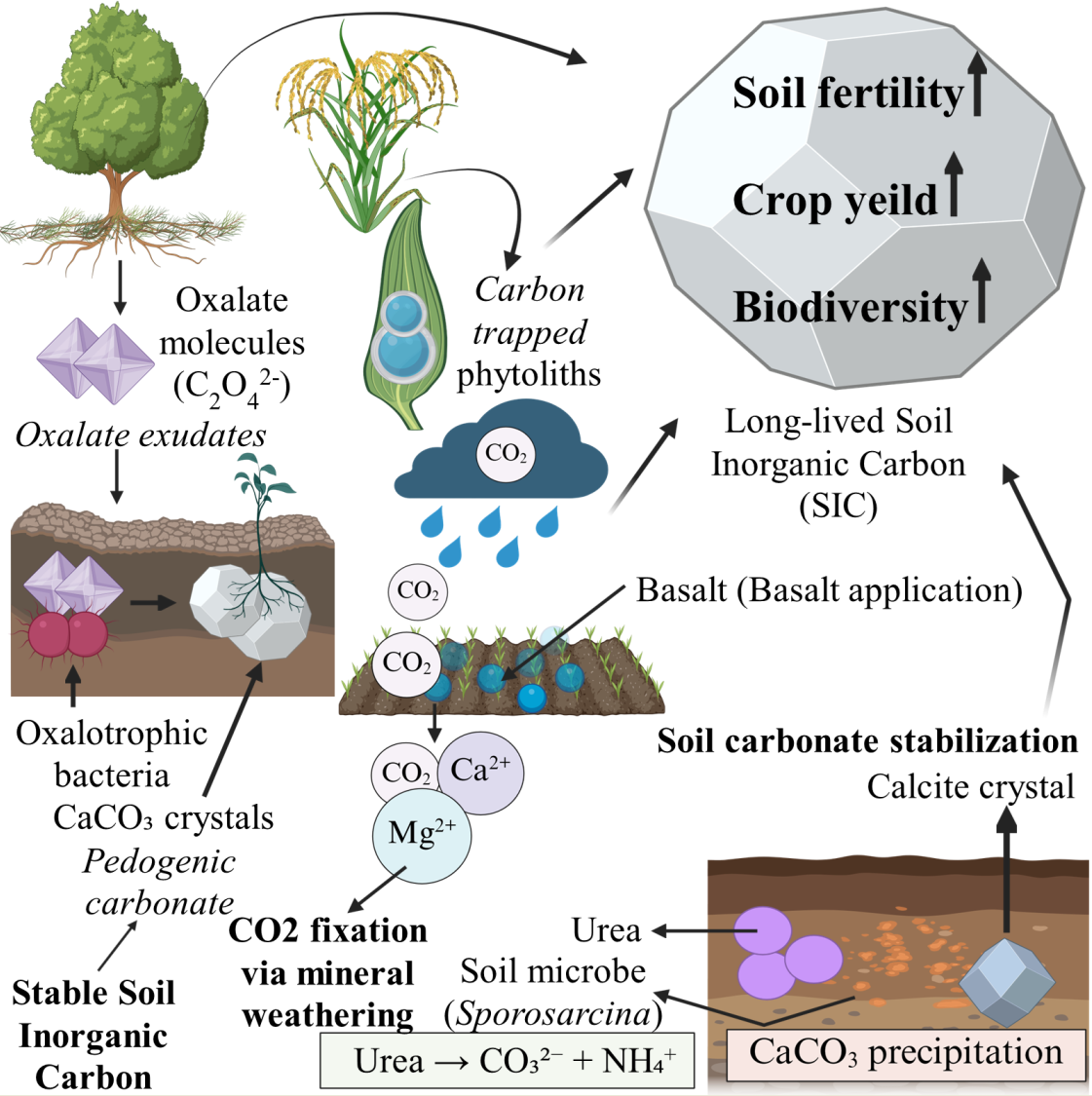
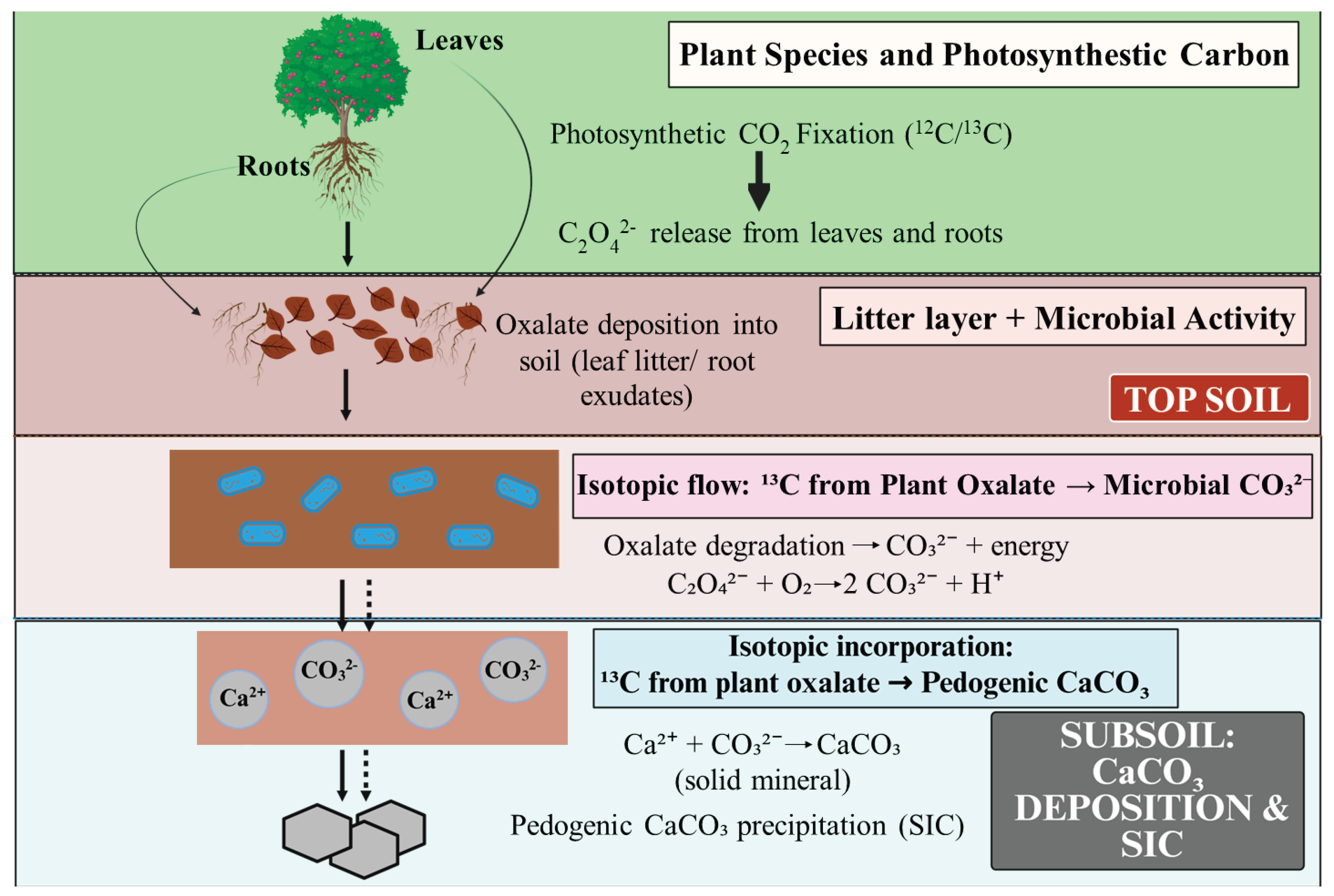
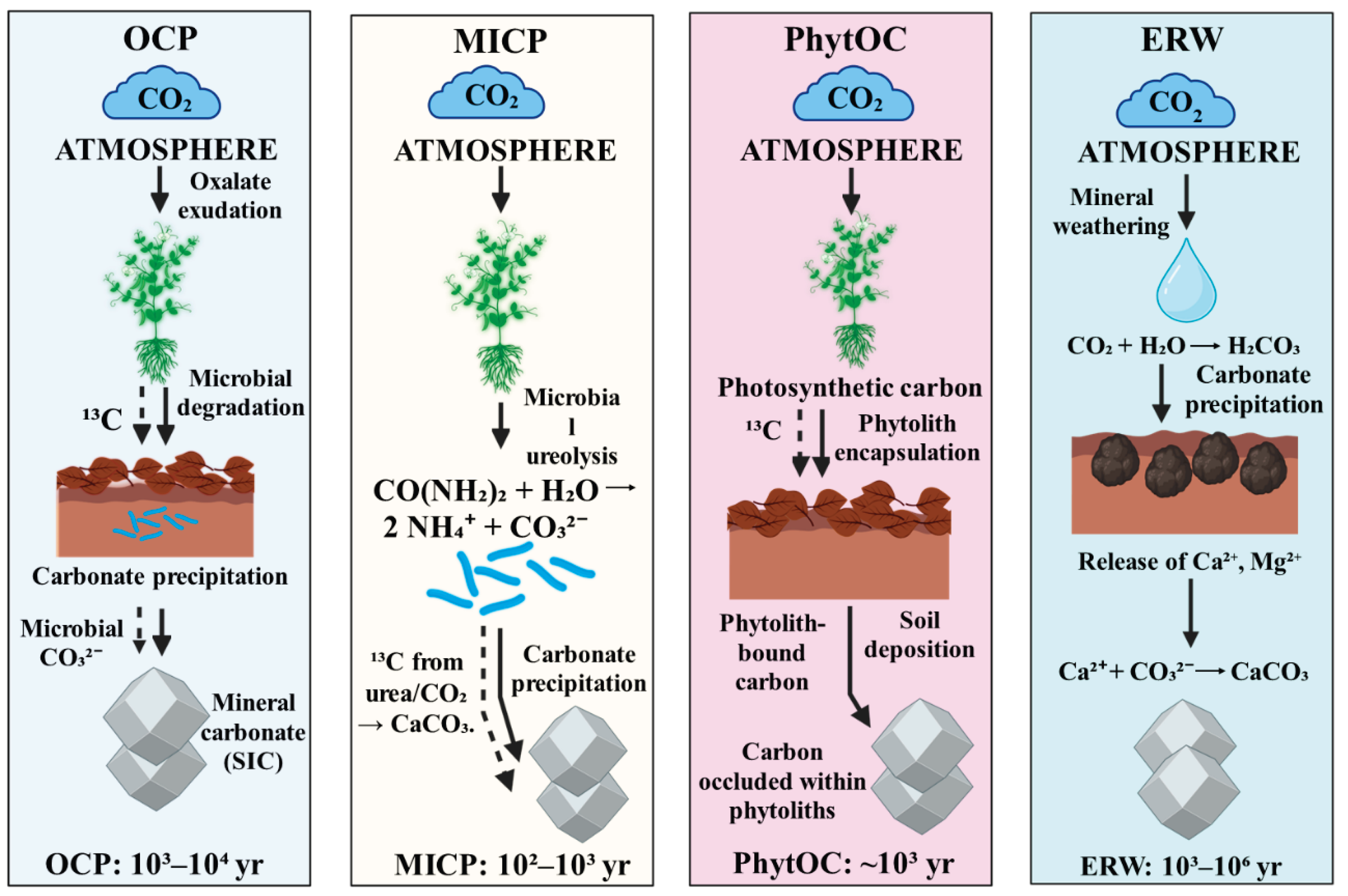
- Oxalate–Carbonate Pathway (OCP): Plants produce calcium oxalate crystals that enter soils via litterfall. Oxalotrophic microorganisms then oxidize oxalate, releasing carbonate and driving calcite precipitation [7]. In OCP systems, biologically produced calcium oxalate is microbially degraded to alkalinity and carbonate, ultimately precipitating pedogenic calcium carbonate under favorable conditions [8]. One key enzymatic route involves oxalate decarboxylation, wherein oxalyl-CoA decarboxylase converts oxalyl-CoA to formyl-CoA with release of CO₂, which can subsequently be hydrated to bicarbonate and drive carbonate mineralization [9]. In wood and litter, brown-rot fungi frequently accumulate calcium oxalate that is subsequently metabolized by oxalotrophic bacteria, sustaining localized alkalinity and carbonate precipitation [10]. Oxalotrophy is widespread among plant-associated Burkholderia/Paraburkholderia lineages, providing a microbial conduit between plant oxalate production and carbonate formation in the rhizosphere [11].
- Microbially Induced Carbonate Precipitation (MICP): Microbial metabolisms such as ureolysis, denitrification, sulfate reduction, or photosynthesis create alkaline conditions or increase bicarbonate availability, leading to carbonate mineral formation [12]. Extracellular carbonic anhydrases catalyze rapid interconversion of CO₂ and bicarbonate, increasing local carbonate alkalinity and enhancing CaCO₃ nucleation in soils and engineered matrices [13]. Ureolytic, endospore-forming bacteria such as Sporosarcina pasteurii can induce calcium carbonate precipitation via urease-driven alkalinization and have been widely tested for biocement and “bioconcrete” applications [14]. Photosynthetic cyanobacteria and microalgae elevate pH and draw down dissolved inorganic carbon, promoting carbonate precipitation while stabilizing biocrusts and improving infiltration [15].
- Enhanced Rock Weathering (ERW): Finely ground silicate minerals (e.g., basalt) are spread onto soils, where plant roots and microbial activity accelerate dissolution, releasing Ca²⁺ and Mg²⁺ that combine with CO₂ to form stable carbonates or bicarbonates [16]. For ERW and carbonate-forming pathways, standardized MRV should pair soil inorganic carbon measurements with mineralogical speciation, isotopic partitioning, and, where relevant, process biomarkers (e.g., ureC for ureolysis; oxc/frc for oxalate catabolism) to constrain mechanisms and durability (Hartmann et al., 2013; Syed et al., 2020). To avoid over-generalization, removal rates should be reported as t CO₂ ha⁻¹ yr⁻¹ with explicit assumptions on Ca/Mg supply, rainfall, and pH buffering, and should be bracketed by evidence level (lab, field, modeled) [17]
- PhytOC: Organic carbon trapped in silica phytoliths produced by plants, often resistant to microbial degradation [5]. In croplands, PhytOC provides a distinct, physically protected carbon pool whose accumulation varies among grasses and cereals across climates [18]. Recent field work from Poland and Estonia indicates that cereal systems can measurably increase PhytOC stocks, highlighting agronomic levers for deployment in temperate regions [19]. Bamboo forests may achieve comparatively high PhytOC yields per hectare, although reported rates are site-specific and sensitive to species and soil silica supply [20].
2. Major Biomineralization Pathways Relevant to Trees and Soils
2.1. Oxalate–Carbonate Pathway (OCP)
2.2. Microbially Induced Carbonate Precipitation (MICP) in Rhizosphere & Trunk Microenvironments
- a)
- Ureolysis – This is the most extensively studied and technologically applied mechanism, mediated by urease-positive bacteria such as Sporosarcina pasteurii (formerly Bacillus pasteurii), as well as Bacillus, Lysinibacillus, Heliobacter, and others. Urease catalyzes the hydrolysis of urea:
- b)
- Denitrification – Facultative anaerobes such as Pseudomonas, Paracoccus, and Thauera reduce nitrate under anaerobic conditions:
- c)
- Sulfate Reduction – Sulfate-reducing bacteria such as Desulfovibrio and Desulfobacter convert sulfate to sulfide using organic matter:
- d)
- Photosynthetic CO₂ Drawdown – Photosynthetic microbes, particularly cyanobacteria (e.g., Synechococcus spp.) and microalgae, deplete dissolved CO₂:
2.3. Phytoliths and Carbon Occlusion (PhytOC)
2.4. Enhanced Rock Weathering (ERW) Synergized with Vegetation
2.5. Intracellular and Structural Mineralization in Plants (Ca-Oxalate, Ca-Carbonate)
3. Microbial Players and Community Ecology
3.1. Oxalotrophs, Ureolytic Bacteria, and Photosynthetic Microbes
3.1.1. Oxalotrophs
3.1.2. Ureolytic Bacteria and Sporosarcina pasteurii
3.1.3. Photosynthetic Microbes (Cyanobacteria and Microalgae)
3.1.4. Functional Redundancy and Specialization
3.2. Fungi and Community Ecology
3.2.1. Microbial Community Assembly, pH Niches, Nutrient Feedbacks, and Resilience
4. Mechanistic Geochemistry: Nucleation, Polymorphs, and Permanence
4.1. Geochemical Pathways from CO2 → Organic C → Oxalate → Carbonate
4.2. Mineral Phases, Transformation Pathways, and Stability
4.3. Inference and Permanence
5. Empirical Evidence — Case Studies and Field Reports
5.1. Classic Ecosystems: Iroko (Ivory Coast) and Documented OCP Sites; Isotopic and Mineralogical Evidence
5.2. Recent Observations: Fig Trees in Basaltic Soils
5.3. Agricultural Systems: PhytOC in Crops and Bamboo
5.4. Experimental MICP Trials in Soils and in Planta (Greenhouse and Field), Dolerite/Basalt Amendments Supplying Divalent Cations
6. Methods and Measurement
6.1. Mineralogical & Microstructural Tools
- X-ray Diffraction (XRD): A fundamental tool for quantitative mineral analysis, used to identify and quantify mineral phases in bulk samples (soil, wood ash, basalt fracture networks). It distinguishes calcite, aragonite, dolomite, quartz, etc., and is key for confirming carbonate precipitation [89].
- Scanning Electron Microscopy (SEM) with Energy Dispersive X-ray Spectroscopy (EDS): SEM provides high-resolution imaging of mineral morphology and micro/nanoscale surface topography (e.g., calcite granules on sand grains, CaOx in plant tissues). Coupled with EDS, it reveals elemental composition (e.g., Ca, C, O), confirming that observed particulates contain calcium and carbon [90].
- Micro-Computed Tomography (µCT): A non-destructive 3D imaging method that visualizes mineral distribution inside samples (e.g., carbonate within basalt fracture networks, void-filling carbonate in root/trunk cross-sections [91].
- Transmission Electron Microscopy (TEM): Enables nanoscale imaging of crystal structures, such as identifying vaterite nanoparticles or organic inclusions. Though less common in field studies, TEM is valuable for fundamental studies of biomineral textures [92].
- Raman Spectroscopy: Provides molecular fingerprints to distinguish CaCO₃ polymorphs (calcite, aragonite, vaterite) through distinct Raman peaks, and can also detect organic carbon signatures. Non-destructive Raman mapping has been applied to assess phytolith carbon content [93].
- Synchrotron-based µXRF and XANES: Micro-X-ray fluorescence (µXRF) enables micron-scale elemental mapping (Ca, Si), while X-ray absorption near-edge spectroscopy (XANES) at the Ca K-edge differentiates between CaCO₃, Ca-oxalate, and other Ca compounds. These have been used, for example, to precisely localize carbonate versus oxalate in fig-tree tissues [30].
- Other Methods: Fourier-transform infrared spectroscopy (FTIR) and thermogravimetric analysis (TGA) can detect Si–O and C–O bonds (phytolith carbon). Acid fumigation, calcimetry, or manometry can measure bulk inorganic carbon by quantifying CO₂ released upon acid reaction [94].
- Isotopic Tracers and Signatures: Isotopic analysis provides definitive fingerprints of the origin, age, and in some cases, conditions of biomineral formation—essential for validating sequestration, additionality, and permanence [95].
- Carbon Isotopes (δ¹³C): C₃ plant organic carbon typically has δ¹³C ~–25‰, while atmospheric CO₂ is ~–8‰ (preindustrial). Newly precipitated carbonates often show intermediate δ¹³C signatures, confirming a biological (photosynthate-derived) origin rather than lithogenic carbonate (~0‰) [96].
- Radiocarbon (¹⁴C): ¹⁴C dating is the primary tool to distinguish modern (recent atmospheric/plant-derived) carbonates from ancient, geologic carbon, which lack radiocarbon [97].
- Clumped Isotopes (∆₄₇): Advanced mass spectrometry of carbonate bonds can sometimes provide formation temperatures. Although still nascent in field studies, it holds potential for differentiating carbonates formed at surface biogenic versus deeper soil conditions [98].
- Oxygen Isotopes (δ¹⁸O): Ratios in carbonates reflect temperature and source water conditions, though interpretation in soils remains complex. Together, δ¹³C and ¹⁴C data provide robust confirmation of carbon origin, while clumped isotopes and δ¹⁸O add insight into formation environments [99].
- Quantifying PhytOC and Soil SIC: Quantifying the carbon sequestered via phytolith-occluded carbon (PhytOC) and soil inorganic carbon (SIC) requires standardized protocols [100].
- Phytolith Extraction: Plant/soil samples are oxidized with strong acids (H₂O₂, HNO₃) and alkali to remove organics, leaving silica residues (phytoliths). Alternatively, HF digestion dissolves silica, releasing occluded carbon. Extracted phytoliths are analyzed for carbon (e.g., combustion in CHN analyzers) [5].
- Soil Inorganic Carbon (SIC): Commonly quantified by treating soils with HCl (fumigation or titration) to release CO₂ from carbonates, which is measured volumetrically or instrumentally (e.g., Scheibler test, pressure transducer) [101].
- Density Fractionation: Heavy-liquid separation (e.g., sodium polytungstate) isolates dense fractions enriched in phytoliths or carbonates. The carbon content of these fractions is analyzed to estimate PhytOC relative to total soil carbon [102].
- In situ Techniques: Advanced imaging such as NanoSIMS or synchrotron Nano-XRF map carbon distribution within individual phytoliths, confirming the presence of occluded carbon at microscale. Though not routine, these approaches validate microscopic localization [103].
- Microbial Community Profiling: Understanding the microbial drivers of biomineralization requires taxonomic, functional, and activity-based analyses.
- Metagenomics: DNA sequencing of soil or litter samples provides a comprehensive inventory of microbial taxa (e.g., Cupriavidus, Oxalobacter, Bacillus, Trichoderma) [31].
- Metatranscriptomics / Metaproteomics: Sequencing or mass spectrometry of RNA/proteins detects active metabolic pathways (e.g., transcripts of oxalate decarboxylase, urease), revealing functional activity [104].
- Targeted Functional Gene Assays (qPCR): Quantifies abundance of biomineralization-associated genes—oxc and frc (oxalate degradation), ureC (urease), cah (carbonic anhydrase) [105].
- Enzyme Activity Assays: Direct measurements of soil urease or oxalate oxidase activity provide functional evidence of biomineralization [106].
- Stable Isotope Probing (SIP): Incubation with labeled substrates (¹³C-oxalate, ¹⁵N-urea) followed by tracking isotope incorporation into microbial biomass links specific taxa to pathways [107].
- MRV Frameworks for Permanence, Leakage, and Additionality: Measurement, Reporting, and Verification (MRV) is critical for carbon accounting, ensuring credibility of biomineralization projects.
- Long-term Monitoring: Repeated soil and plant sampling across years to track inorganic carbon stocks (calcite, phytolith C) [108].
- Flux Measurements: CO₂ fluxes measured via closed chambers or eddy-covariance; distinguishing inorganic vs. organic fluxes may require isotopic tracing or specialized chambers targeting HCO₃⁻ [109].
- Baseline Controls & Field Trials: Paired plots and randomized designs establish baselines and capture variability, proving additionality (that sequestration would not occur without intervention) [35].
- Permanence Trials: Soil cores incubated under altered pH/moisture test stability of newly formed carbonates. Addressing metastable mineral phases is key [110].
- Leakage and Trade-offs: Assessment of unintended effects (e.g., N₂O emissions from urea, nutrient leaching, rock mining/transport for ERW) [16].
- Modeling: Process-based soil carbon models (including inorganic pools) extrapolate findings across scales and time [111].
- Certification Standards: Few carbon-credit protocols explicitly recognize SIC. New standards—similar to those for biochar or SOC—are urgently needed. High MRV costs remain a barrier, but digital MRV (dMRV) solutions may reduce expenses via automation [35].
7. Engineering and Management Pathways
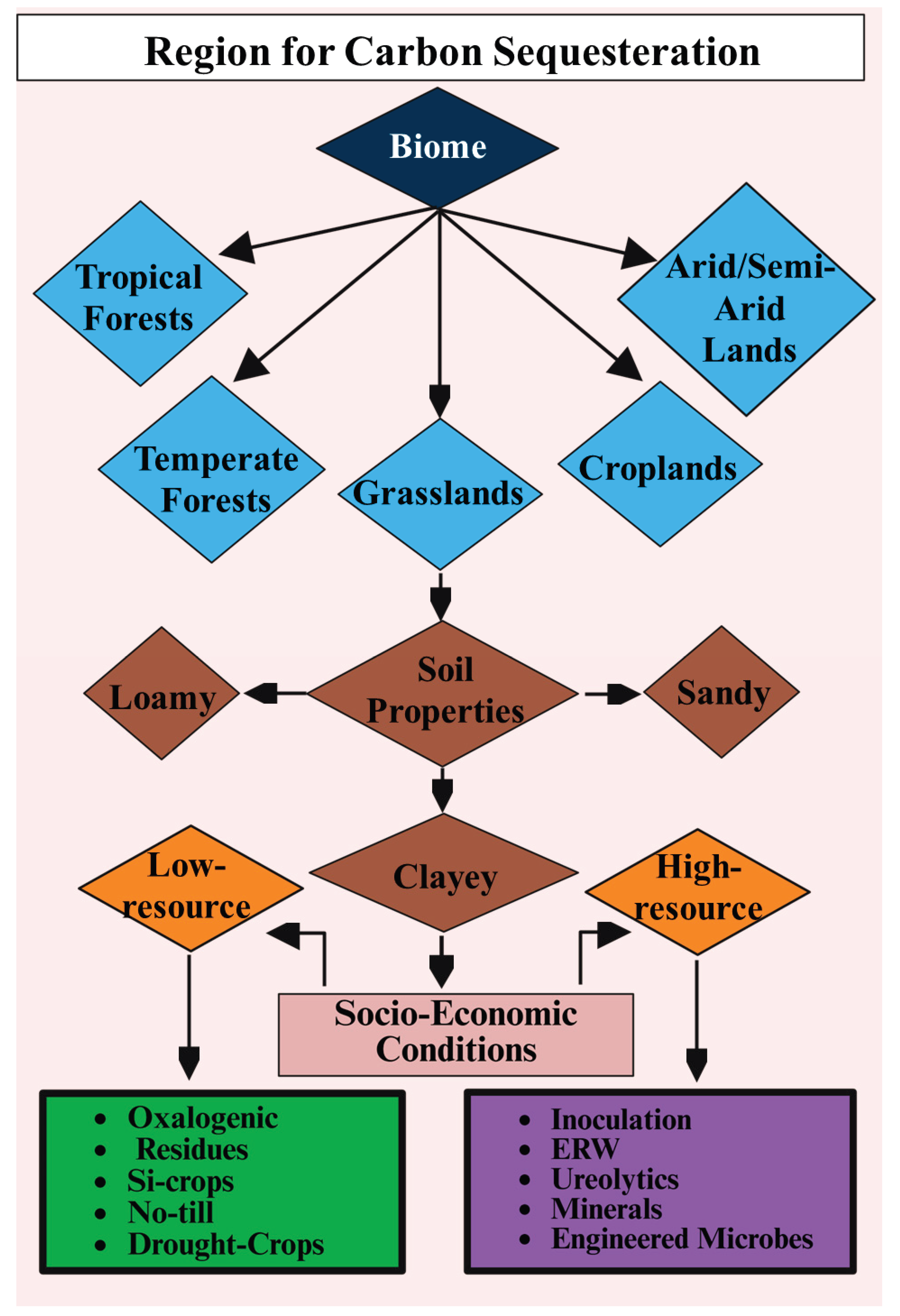
7.1. Low-Intervention Strategies: Selecting and Restoring Oxalogenic Tree Species, Agroforestry Integration, Residue and Fire Management to Maximize OCP and PhytOC
7.2. Active Microbial Augmentation: Inoculation with Oxalotrophs/Ureolytic Strains and Biosafety Considerations
7.3. Mineral Amendments and Enhanced Rock Weathering (ERW) Integration
7.4. Biotechnological Routes: Engineering Cyanobacteria, Plants, and Microbiomes for Enhanced Carbonate Precipitation
8. Scaling, Permanence, and Climate Accounting
8.1. Estimates of Potential C Removal per Hectare Under Optimistic vs. Conservative Scenarios (OCP, PhytOC, MICP + ERW)
8.2. Residence Times and Stability
8.3. Co-Benefits and Trade-Offs
8.4. Inclusion in Carbon Markets and MRV Requirements
9. Risks, Ethical & Governance Considerations
9.1. Environmental Risks
9.2. Biosafety & Regulatory Oversight
9.3. Social and Land-Use Justice
9.4. Global Governance & Climate Policy Implications
10. Knowledge Gaps and Research Priorities (Roadmap)
10.1. Quantitative Field Measurements of Trunk and Soil Carbonate Mass Balances Across Biomes
10.2. Longitudinal Experiments Linking Plant Physiology → Litter Chemistry → Microbial Oxalotrophy → Carbonate Accrual
10.3. Robust MRV Protocols and Standardized Lab-to-Field Pipelines
10.4. Techno-Economic, Biosafety, and Socioeconomic Considerations
11. Roadmap for Translational Action
11.1. Short-Term (1–3 Years)
11.2. Medium-Term (3–7 Years)
11.3. Long-Term (7–20 Years)
12. Conclusions
Author Contributions
Funding
Declaration of generative AI and AI-assisted technologies in the writing process
Conflicts of Interest
References
- Knoll AH. Biomineralization and Evolutionary History. Rev Mineral Geochem. 2003 Jan 1;54(1):329–56.
- Verrecchia EP, Braissant O, Cailleau G. The oxalate–carbonate pathway in soil carbon storage: the role of fungi and oxalotrophic bacteria. In: Gadd GM, editor. Fungi in Biogeochemical Cycles [Internet]. 1st ed. Cambridge University Press; 2006 [cited 2025 Sep 8]. p. 289–310. Available from: https://www.cambridge.org/core/product/identifier/CBO9780511550522A018/type/book_part.
- Raza S, Irshad A, Margenot A, Zamanian K, Li N, Ullah S, Mehmood K, Ajmal Khan M, Siddique N, Zhou J, Mooney SJ, Kurganova I, Zhao X, Kuzyakov Y. Inorganic carbon is overlooked in global soil carbon research: A bibliometric analysis. Geoderma. 2024 Mar;443:116831.
- Trumbore S. Radiocarbon and Soil Carbon Dynamics. Annu Rev Earth Planet Sci. 2009 May 1;37(1):47–66.
- Parr JF, Sullivan LA. Soil carbon sequestration in phytoliths. Soil Biol Biochem. 2005 Jan;37(1):117–24.
- Franceschi VR, Nakata PA. CALCIUM OXALATE IN PLANTS: Formation and Function. Annu Rev Plant Biol. 2005 Jun 1;56(1):41–71.
- Cailleau G, Braissant O, Verrecchia EP. Turning sunlight into stone: the oxalate-carbonate pathway in a tropical tree ecosystem. Biogeosciences. 2011 Jul 5;8(7):1755–67.
- Xia D, Nie W, Li X, Finlay RD, Lian B. Secondary products and molecular mechanism of calcium oxalate degradation by the strain Azospirillum sp. OX-1. Sci Rep. 2024 Oct 9;14(1):23506.
- Sheng X, Liu Y, Zhang R. A theoretical study of the catalytic mechanism of oxalyl-CoA decarboxylase, an enzyme for treating urolithiasis. RSC Adv. 2014 Aug 5;4(67):35777.
- Vlasov DYu, Zelenskaya MS, Izatulina AR, Janson SYu, Frank-Kamenetskaya OV. Oxalate Crystallization under the Action of Brown Rot Fungi. Crystals. 2023 Mar 2;13(3):432.
- Kost T, Stopnisek N, Agnoli K, Eberl L, Weisskopf L. Oxalotrophy, a widespread trait of plant-associated Burkholderia species, is involved in successful root colonization of lupin and maize by Burkholderia phytofirmans. Front Microbiol [Internet]. 2014 [cited 2025 Sep 8];4. Available from: http://journal.frontiersin.org/article/10.3389/fmicb.2013.00421/abstract.
- Hammes F, Verstraete* W. Key roles of pH and calcium metabolism in microbial carbonate precipitation. Rev Environ Sci Biotechnol. 2002 Mar;1(1):3–7.
- Mwandira W, Mavroulidou M, Gunn MJ, Purchase D, Garelick H, Garelick J. Concurrent Carbon Capture and Biocementation through the Carbonic Anhydrase (CA) Activity of Microorganisms -a Review and Outlook. Environ Process. 2023 Dec;10(4):56.
- DeJong JT, Mortensen BM, Martinez BC, Nelson DC. Bio-mediated soil improvement. Ecol Eng. 2010 Feb;36(2):197–210.
- Hazarika A, Yadav M. Biomineralization of carbon dioxide by carbonic anhydrase. Biocatal Agric Biotechnol. 2023 Aug;51:102755.
- Beerling DJ, Kantzas EP, Lomas MR, Wade P, Eufrasio RM, Renforth P, Sarkar B, Andrews MG, James RH, Pearce CR, Mercure JF, Pollitt H, Holden PB, Edwards NR, Khanna M, Koh L, Quegan S, Pidgeon NF, Janssens IA, Hansen J, Banwart SA. Potential for large-scale CO2 removal via enhanced rock weathering with croplands. Nature. 2020 Jul 9;583(7815):242–8.
- Clarkson MO, Larkin CS, Swoboda P, Reershemius T, Suhrhoff TJ, Maesano CN, Campbell JS. A review of measurement for quantification of carbon dioxide removal by enhanced weathering in soil. Front Clim. 2024 Jun 19;6:1345224.
- Song Z, Liu H, Si Y, Yin Y. The production of phytoliths in C hina’s grasslands: implications to the biogeochemical sequestration of atmospheric CO2 . Glob Change Biol. 2012 Dec;18(12):3647–53.
- Rutkowska B, Schröder P, Mench M, Rineau F, Szulc W, Szulc W, Pobereżny J, Tiideberg K, Niedziński T, Loit E. Role of phytolith occluded carbon of cereales plants for climate change mitigation. J Water Land Dev. 2024 Mar 21;50–8.
- Qi L, Sun T, Guo X, Guo Y, Li FY. Phytolith--occluded carbon sequestration potential in three major steppe types along a precipitation gradient in Northern China. Ecol Evol. 2021 Feb;11(3):1446–56.
- Ma W, Tang S, Dengzeng Z, Zhang D, Zhang T, Ma X. Root exudates contribute to belowground ecosystem hotspots: A review. Front Microbiol. 2022 Oct 5;13:937940.
- Gabarrón-Galeote MA, Trigalet S, Van Wesemael B. Effect of land abandonment on soil organic carbon fractions along a Mediterranean precipitation gradient. Geoderma. 2015 Jul;249–250:69–78.
- Beerling DJ, Epihov DZ, Kantola IB, Masters MD, Reershemius T, Planavsky NJ, Reinhard CT, Jordan JS, Thorne SJ, Weber J, Val Martin M, Freckleton RP, Hartley SE, James RH, Pearce CR, DeLucia EH, Banwart SA. Enhanced weathering in the US Corn Belt delivers carbon removal with agronomic benefits. Proc Natl Acad Sci. 2024 Feb 27;121(9):e2319436121.
- Song Z, McGrouther K, Wang H. Occurrence, turnover and carbon sequestration potential of phytoliths in terrestrial ecosystems. Earth-Sci Rev. 2016 Jul;158:19–30.
- Manning DAC. Mineral sources of potassium for plant nutrition. A review. Agron Sustain Dev. 2010 Apr;30(2):281–94.
- Rowley MC, Grand S, Verrecchia ÉP. Calcium-mediated stabilisation of soil organic carbon. Biogeochemistry. 2018 Jan;137(1–2):27–49.
- Pons S, Bindschedler S, Sebag D, Junier P, Verrecchia E, Cailleau G. Biocontrolled soil nutrient distribution under the influence of an oxalogenic-oxalotrophic ecosystem. Plant Soil. 2018 Apr;425(1–2):145–60.
- Dupraz C, Reid RP, Braissant O, Decho AW, Norman RS, Visscher PT. Processes of carbonate precipitation in modern microbial mats. Earth-Sci Rev. 2009 Oct;96(3):141–62.
- Owusu EA, Wu J, Appiah EA, Marfo WA, Yuan N, Ge X, Ling K, Wang S. Carbon Mineralization in Basaltic Rocks: Mechanisms, Applications, and Prospects for Permanent CO2 Sequestration. Energies. 2025 Jul 2;18(13):3489.
- Goldschmidt G. Novel mechanisms of carbon mineralization in woody tissues of Ficus specie. In 2025.
- Zhu T, Dittrich M. Carbonate Precipitation through Microbial Activities in Natural Environment, and Their Potential in Biotechnology: A Review. Front Bioeng Biotechnol [Internet]. 2016 Jan 20 [cited 2025 Sep 8];4. Available from: http://journal.frontiersin.org/Article/10.3389/fbioe.2016.00004/abstract.
- Kimura K, Okuro T. Cyanobacterial Biocrust on Biomineralized Soil Mitigates Freeze–Thaw Effects and Preserves Structure and Ecological Functions. Microb Ecol. 2024 Dec;87(1):69.
- Martin G, Guggiari M, Bravo D, Zopfi J, Cailleau G, Aragno M, Job D, Verrecchia E, Junier P. Fungi, bacteria and soil pH : the oxalate–carbonate pathway as a model for metabolic interaction. Environ Microbiol. 2012 Nov;14(11):2960–70.
- Kelland ME, Wade PW, Lewis AL, Taylor LL, Sarkar B, Andrews MG, Lomas MR, Cotton TEA, Kemp SJ, James RH, Pearce CR, Hartley SE, Hodson ME, Leake JR, Banwart SA, Beerling DJ. Increased yield and CO2 sequestration potential with the C4 cereal Sorghum bicolor cultivated in basaltic rock dust--amended agricultural soil. Glob Change Biol. 2020 Jun;26(6):3658–76.
- Phillips AJ, Gerlach R, Lauchnor E, Mitchell AC, Cunningham AB, Spangler L. Engineered applications of ureolytic biomineralization: a review. Biofouling. 2013 Jul;29(6):715–33.
- Zhang K, Tang CS, Jiang NJ, Pan XH, Liu B, Wang YJ, Shi B. Microbial--induced carbonate precipitation (MICP) technology: a review on the fundamentals and engineering applications. Environ Earth Sci. 2023 May;82(9):229.
- Jain S, Venkataraman A, Wechsler ME, Peppas NA. Messenger RNA-based vaccines: Past, present, and future directions in the context of the COVID-19 pandemic. Adv Drug Deliv Rev. 2021 Dec;179:114000.
- Decho AW. Overview of biopolymer-induced mineralization: What goes on in biofilms? Ecol Eng. 2010 Feb;36(2):137–44.
- Benzerara K, Skouri-Panet F, Li J, Férard C, Gugger M, Laurent T, Couradeau E, Ragon M, Cosmidis J, Menguy N, Margaret-Oliver I, Tavera R, López-García P, Moreira D. Intracellular Ca-carbonate biomineralization is widespread in cyanobacteria. Proc Natl Acad Sci. 2014 Jul 29;111(30):10933–8.
- Yang X, Huang S, Wu Q, Zhang R. Nitrate reduction coupled with microbial oxidation of sulfide in river sediment. J Soils Sediments. 2012 Oct;12(9):1435–44.
- De Tombeur F, Raven JA, Toussaint A, Lambers H, Cooke J, Hartley SE, Johnson SN, Coq S, Katz O, Schaller J, Violle C. Why do plants silicify? Trends Ecol Evol. 2023 Mar;38(3):275–88.
- Koebernick N, Mikutta R, Kaiser K, Klotzbücher A, Nguyen ATQ, Nguyen MN, Klotzbücher T. Controls on phytolith stability upon exposure in paddy soils. Geoderma. 2024 Mar;443:116821.
- Stocks-Fischer S, Galinat JK, Bang SS. Microbiological precipitation of CaCO3. Soil Biol Biochem. 1999 Oct;31(11):1563–71.
- Ren T, Cai A. Global patterns and drivers of soil dissolved organic carbon concentrations. Earth Syst Sci Data. 2025 Jun 25;17(6):2873–85.
- Canarini A, Kaiser C, Merchant A, Richter A, Wanek W. Root Exudation of Primary Metabolites: Mechanisms and Their Roles in Plant Responses to Environmental Stimuli. Front Plant Sci. 2019 Feb 21;10:157.
- Van Paassen LA, Daza CM, Staal M, Sorokin DY, Van Der Zon W, Van Loosdrecht MarkCM. Potential soil reinforcement by biological denitrification. Ecol Eng. 2010 Feb;36(2):168–75.
- Hartmann J, West AJ, Renforth P, Köhler P, De La Rocha CL, Wolf--Gladrow DA, Dürr HH, Scheffran J. Enhanced chemical weathering as a geoengineering strategy to reduce atmospheric carbon dioxide, supply nutrients, and mitigate ocean acidification. Rev Geophys. 2013 Apr;51(2):113–49.
- Cohen PA, Macdonald FA. The Proterozoic Record of Eukaryotes. Paleobiology. 2015 Sep;41(4):610–32.
- Webb MA, Cavaletto JM, Carpita NC, Lopez LE, Arnott HJ. The intravacuolar organic matrix associated with calcium oxalate crystals in leaves of Vitis. Plant J. 1995 Apr;7(4):633–48.
- Koester M, Stock SC, Nájera F, Abdallah K, Gorbushina A, Prietzel J, Matus F, Klysubun W, Boy J, Kuzyakov Y, Dippold MA, Spielvogel S. From rock eating to vegetarian ecosystems — Disentangling processes of phosphorus acquisition across biomes. Geoderma. 2021 Apr;388:114827.
- Bui M, Adjiman CS, Bardow A, Anthony EJ, Boston A, Brown S, Fennell PS, Fuss S, Galindo A, Hackett LA, Hallett JP, Herzog HJ, Jackson G, Kemper J, Krevor S, Maitland GC, Matuszewski M, Metcalfe IS, Petit C, Puxty G, Reimer J, Reiner DM, Rubin ES, Scott SA, Shah N, Smit B, Trusler JPM, Webley P, Wilcox J, Mac Dowell N. Carbon capture and storage (CCS): the way forward. Energy Environ Sci. 2018;11(5):1062–176.
- Liang C, Schimel JP, Jastrow JD. The importance of anabolism in microbial control over soil carbon storage. Nat Microbiol. 2017 Jul 25;2(8):17105.
- Grąz M. Role of oxalic acid in fungal and bacterial metabolism and its biotechnological potential. World J Microbiol Biotechnol. 2024 Jun;40(6):178.
- Syed S, Buddolla V, Lian B. Oxalate Carbonate Pathway—Conversion and Fixation of Soil Carbon—A Potential Scenario for Sustainability. Front Plant Sci. 2020 Dec 21;11:591297.
- Sahin N. Oxalotrophic bacteria. Res Microbiol. 2003 Jul;154(6):399–407.
- Baetz AL, Allison MJ. Localization of Oxalyl-Coenzyme A Decarboxylase, and Formyl-Coenzyme A Transferase in Oxalobacter formigenes Cells. Syst Appl Microbiol. 1992 May;15(2):167–71.
- Gadd GM. Fungal biomineralization. Curr Biol. 2021 Dec;31(24):R1557–63.
- Rahman MM, Hora RN, Ahenkorah I, Beecham S, Karim MR, Iqbal A. State-of-the-Art Review of Microbial-Induced Calcite Precipitation and Its Sustainability in Engineering Applications. Sustainability. 2020 Aug 4;12(15):6281.
- Qin K, Zheng Z, Wang J, Pan H, Tang R. Biomineralization strategy: from material manufacturing to biological regulation. Giant. 2024 Aug;19:100317.
- Yang YY, Jung JY, Song WY, Suh HS, Lee Y. Identification of Rice Varieties with High Tolerance or Sensitivity to Lead and Characterization of the Mechanism of Tolerance. Plant Physiol. 2000 Nov 1;124(3):1019–26.
- Karabourniotis G, Liakopoulos G, Nikolopoulos D, Bresta P. Protective and defensive roles of non-glandular trichomes against multiple stresses: structure–function coordination. J For Res. 2020 Feb;31(1):1–12.
- Quirk J, Leake JR, Banwart SA, Taylor LL, Beerling DJ. Weathering by tree-root-associating fungi diminishes under simulated Cenozoic atmospheric CO2 decline. Biogeosciences. 2014 Jan 23;11(2):321–31.
- Addadi L, Raz S, Weiner S. Taking Advantage of Disorder: Amorphous Calcium Carbonate and Its Roles in Biomineralization. Adv Mater. 2003 Jun 17;15(12):959–70.
- Bindschedler S, Cailleau G, Verrecchia E. Role of Fungi in the Biomineralization of Calcite. Minerals. 2016 May 5;6(2):41.
- Lauchnor EG, Topp DM, Parker AE, Gerlach R. Whole cell kinetics of ureolysis by S porosarcina pasteurii . J Appl Microbiol. 2015 Jun;118(6):1321–32.
- Clemmensen KE, Bahr A, Ovaskainen O, Dahlberg A, Ekblad A, Wallander H, Stenlid J, Finlay RD, Wardle DA, Lindahl BD. Roots and Associated Fungi Drive Long-Term Carbon Sequestration in Boreal Forest. Science. 2013 Mar 29;339(6127):1615–8.
- Bäckhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, Semenkovich CF, Gordon JI. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci. 2004 Nov 2;101(44):15718–23.
- Andrei AŞ, Păuşan MR, Tămaş T, Har N, Barbu-Tudoran L, Leopold N, Banciu HL. Diversity and Biomineralization Potential of the Epilithic Bacterial Communities Inhabiting the Oldest Public Stone Monument of Cluj-Napoca (Transylvania, Romania). Front Microbiol [Internet]. 2017 Mar 7 [cited 2025 Sep 8];08. Available from: http://journal.frontiersin.org/article/10.3389/fmicb.2017.00372/full.
- Wu Y, Pan X, Xie H, Que L, Tang X. Research progress of biomineralization for the diagnosis and treatment of malignant tumors. Front Pharmacol. 2023 Dec 14;14:1335019.
- Elejalde-Cadena NR, Cuéllar-Cruz M, Moreno A. The role of silica and alkaline earth metals with biomolecules in the biomineralization processes: the eggshell’s formation and the crystallization in vivo for x-ray crystallography. Prog Cryst Growth Charact Mater. 2020 Feb;66(1):100473.
- Rahman MdM, Haque TMdA, Sourav NS, Rahman S, Yesmin S, Mia R, Al Noman A, Begum K. Synthesis and investigation of dyeing properties of 8-hydroxyquinoline-based azo dyes. J Iran Chem Soc. 2021 Apr;18(4):817–26.
- Vienne A, Poblador S, Portillo-Estrada M, Hartmann J, Ijiehon S, Wade P, Vicca S. Enhanced Weathering Using Basalt Rock Powder: Carbon Sequestration, Co-benefits and Risks in a Mesocosm Study With Solanum tuberosum. Front Clim. 2022 May 17;4:869456.
- Izatulina AR, Gurzhiy VV, Krzhizhanovskaya MG, Kuz’mina MA, Leoni M, Frank-Kamenetskaya OV. Hydrated Calcium Oxalates: Crystal Structures, Thermal Stability, and Phase Evolution. Cryst Growth Des. 2018 Sep 5;18(9):5465–78.
- Cheng X, Lv H, Liu S, Li C, Li P, Zhou Y, Shi Y, Zhou G. The phytolith carbon sequestration in terrestrial ecosystems: the underestimated potential of bamboo forest. Ecol Process. 2023 Dec 20;12(1):62.
- Renforth P, Manning DAC, Lopez-Capel E. Carbonate precipitation in artificial soils as a sink for atmospheric carbon dioxide. Appl Geochem. 2009 Sep;24(9):1757–64.
- Faehnrich B, Kirchhof G, Hulugalle N, Goebel B. Carbon Sequestration in Australian Vertisols under Conservation Agriculture – Facts and Public Perception. Int Soil Water Conserv Res. 2025 Aug;S2095633925000826.
- Li Z, Delvaux B. Phytolith--rich biochar: A potential Si fertilizer in desilicated soils. GCB Bioenergy. 2019 Nov;11(11):1264–82.
- Sánchez-Román M, Romanek CS, Fernández-Remolar DC, Sánchez-Navas A, McKenzie JA, Pibernat RA, Vasconcelos C. Aerobic biomineralization of Mg-rich carbonates: Implications for natural environments. Chem Geol. 2011 Feb;281(3–4):143–50.
- Gallagher TM, Breecker DO. The Obscuring Effects of Calcite Dissolution and Formation on Quantifying Soil Respiration. Glob Biogeochem Cycles. 2020 Dec;34(12):e2020GB006584.
- Li S, Wu X, Xie J. Biomineralization technology for solidification/stabilization of heavy metals in ecosystem: status and perspective. Front Ecol Evol. 2023 Jun 16;11:1189356.
- Omoregie AI, Palombo EA, Nissom PM. Bioprecipitation of calcium carbonate mediated by ureolysis: A review. Environ Eng Res. 2020 Nov 22;26(6):200379–0.
- Rossi F, Olguín EJ, Diels L, De Philippis R. Microbial fixation of CO2 in water bodies and in drylands to combat climate change, soil loss and desertification. New Biotechnol. 2015 Jan;32(1):109–20.
- Li Z, Bai T, Dai L, Wang F, Tao J, Meng S, Hu Y, Wang S, Hu S. A study of organic acid production in contrasts between two phosphate solubilizing fungi: Penicillium oxalicum and Aspergillus niger. Sci Rep. 2016 Apr 29;6(1):25313.
- Abdolvand Y, Sadeghiamirshahidi M, Keenum I. Denitrification processes, inhibitors, and their implications in ground improvement. Biogeotechnics. 2025 Mar;100176.
- Zheng X, Lu X, Zhou M, Huang W, Zhong Z, Wu X, Zhao B. Experimental Study on Mechanical Properties of Root–Soil Composite Reinforced by MICP. Materials. 2022 May 17;15(10):3586.
- Kantola IB, Blanc--Betes E, Masters MD, Chang E, Marklein A, Moore CE, Von Haden A, Bernacchi CJ, Wolf A, Epihov DZ, Beerling DJ, DeLucia EH. Improved net carbon budgets in the US Midwest through direct measured impacts of enhanced weathering. Glob Change Biol. 2023 Dec;29(24):7012–28.
- Cailleau G, Braissant O, Dupraz C, Aragno M, Verrecchia EP. Biologically induced accumulations of CaCO3 in orthox soils of Biga, Ivory Coast. CATENA. 2005 Jan;59(1):1–17.
- Song Z, Parr JF, Guo F. Potential of Global Cropland Phytolith Carbon Sink from Optimization of Cropping System and Fertilization. Lin S, editor. PLoS ONE. 2013 Sep 16;8(9):e73747.
- Xiao J, Song Y, Li Y. Comparison of Quantitative X-ray Diffraction Mineral Analysis Methods. Minerals. 2023 Apr 18;13(4):566.
- Bernhard Schulz JP. SEM Automated Mineralogy applied for the quantification of mineral and textural sorting in submarine sediment gravity flows. Gospod Surowcami Miner - Miner Resour Manag [Internet]. 2023 Jul 26 [cited 2025 Sep 8]; Available from: https://journals.pan.pl/dlibra/publication/144094/edition/125506/content.
- He C, Sadeghpour H, Shi Y, Mishra B, Roshankhah S. Mapping distribution of fractures and minerals in rock samples using Res-VGG-UNet and threshold segmentation methods. Comput Geotech. 2024 Nov;175:106675.
- Zhou W, Ikuhara YH, Zheng Z, Wang K, Cao B, Chen J. Transmission electron microscopy (TEM) studies of functional nanomaterials. In: Modeling, Characterization, and Production of Nanomaterials [Internet]. Elsevier; 2023 [cited 2025 Sep 8]. p. 467–512. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780128199053000178.
- Behrens G, Kuhn LT, Ubic R, Heuer AH. Raman Spectra of Vateritic Calcium Carbonate. Spectrosc Lett. 1995 Sep;28(6):983–95.
- Roonasi P, Holmgren A. A Fourier transform infrared (FTIR) and thermogravimetric analysis (TGA) study of oleate adsorbed on magnetite nano-particle surface. Appl Surf Sci. 2009 Mar;255(11):5891–5.
- Bartelink EJ, Chesson LA. Recent applications of isotope analysis to forensic anthropology. Forensic Sci Res. 2019 Jan 2;4(1):29–44.
- Catoni, Marcella; DIVAPRA – Chimica Agraria E Pedologia, Università Di Torino, Torino, MONGER, H Curtis MONGER; Department Of Plant And Environmental Science, New Mexico State University, Las Cruces, Bonifacio, Eleonora; DIVAPRA – Chimica Agraria E Pedologia, Università Di Torino, Torino. δ13C DEI CARBONATI PEDOGENETICI E CONDIZIONI AMBIENTALI DI PRECIPITAZIONEδ13C DES CARBONATES PEDOGENETIQUES ET CONDITIONS ENVIRONNEMENTALES DE PRECIPITATIONPEDOGENIC CARBONATE δ13C AND ENVIRONMENTAL PRECIPITATION CONDITIONS. EQA - Int J Environ Qual Vol 7 2011 17-24 [Internet]. 2013 [cited 2025 Sep 8]; Available from: http://eqa.unibo.it/article/view/3830.
- Hajdas I, Ascough P, Garnett MH, Fallon SJ, Pearson CL, Quarta G, Spalding KL, Yamaguchi H, Yoneda M. Radiocarbon dating. Nat Rev Methods Primer. 2021 Sep 9;1(1):62.
- Anderson NT, Kelson JR, Kele S, Daëron M, Bonifacie M, Horita J, Mackey TJ, John CM, Kluge T, Petschnig P, Jost AB, Huntington KW, Bernasconi SM, Bergmann KD. A Unified Clumped Isotope Thermometer Calibration (0.5–1,100 °C) Using Carbonate--Based Standardization. Geophys Res Lett. 2021 Apr 16;48(7):e2020GL092069.
- Cerling TE. The stable isotopic composition of modern soil carbonate and its relationship to climate. Earth Planet Sci Lett. 1984 Dec;71(2):229–40.
- Davamani V, Sangeetha Piriya R, Rakesh SS, Parameswari E, Paul Sebastian S, Kalaiselvi P, Maheswari M, Santhi R. Phytolith-Occluded Carbon Sequestration Potential of Oil Palm Plantation in Tamil Nadu. ACS Omega. 2022 Jan 25;7(3):2809–20.
- Wang X, Wang J, Zhang J. Comparisons of Three Methods for Organic and Inorganic Carbon in Calcareous Soils of Northwestern China. Kirchman DL, editor. PLoS ONE. 2012 Aug 31;7(8):e44334.
- Plaza C, Giannetta B, Benavente I, Vischetti C, Zaccone C. Density-based fractionation of soil organic matter: effects of heavy liquid and heavy fraction washing. Sci Rep. 2019 Jul 12;9(1):10146.
- Alexandre A, Basile-Doelsch I, Delhaye T, Borshneck D, Mazur JC, Reyerson P, Santos GM. New highlights of phytolith structure and occluded carbon location: 3-D X-ray microscopy and NanoSIMS results. Biogeosciences. 2015 Feb 12;12(3):863–73.
- Sessitsch A, Gyamfi S, Stralis-Pavese N, Weilharter A, Pfeifer U. RNA isolation from soil for bacterial community and functional analysis: evaluation of different extraction and soil conservation protocols. J Microbiol Methods. 2002 Oct;51(2):171–9.
- Shahsavari E, Aburto-Medina A, Taha M, Ball AS. A quantitative PCR approach for quantification of functional genes involved in the degradation of polycyclic aromatic hydrocarbons in contaminated soils. MethodsX. 2016;3:205–11.
- Wei S, Cui H, Jiang Z, Liu H, He H, Fang N. Biomineralization processes of calcite induced by bacteria isolated from marine sediments. Braz J Microbiol. 2015 Jun;46(2):455–64.
- Uhlik O, Leewis MC, Strejcek M, Musilova L, Mackova M, Leigh MB, Macek T. Stable isotope probing in the metagenomics era: A bridge towards improved bioremediation. Biotechnol Adv. 2013 Mar;31(2):154–65.
- Lawrence GB, Fernandez IJ, Hazlett PW, Bailey SW, Ross DS, Villars TR, Quintana A, Ouimet R, McHale MR, Johnson CE, Briggs RD, Colter RA, Siemion J, Bartlett OL, Vargas O, Antidormi MR, Koppers MM. Methods of Soil Resampling to Monitor Changes in the Chemical Concentrations of Forest Soils. J Vis Exp. 2016 Nov 25;(117):54815.
- Ma J, Wang ZY, Stevenson BA, Zheng XJ, Li Y. An inorganic CO2 diffusion and dissolution process explains negative CO2 fluxes in saline/alkaline soils. Sci Rep. 2013 Jun 19;3(1):2025.
- Wilhelm RC, Lynch L, Webster TM, Schweizer S, Inagaki TM, Tfaily MM, Kukkadapu R, Hoeschen C, Buckley DH, Lehmann J. Susceptibility of new soil organic carbon to mineralization during dry-wet cycling in soils from contrasting ends of a precipitation gradient. Soil Biol Biochem. 2022 Jun;169:108681.
- Naorem A, Jayaraman S, Dalal RC, Patra A, Rao CS, Lal R. Soil Inorganic Carbon as a Potential Sink in Carbon Storage in Dryland Soils—A Review. Agriculture. 2022 Aug 18;12(8):1256.
- Singh P, Singh R, Singh S, Chauhan RS, Bala S, Pathak N, Singh PK, Tripathi M. Microbial Engineering for a Greener Ecosystem and Agriculture: Recent Advances and Challenges. J Pure Appl Microbiol. 2024 Jun 1;18(2):797–807.
- Dynarski KA, Bossio DA, Scow KM. Dynamic Stability of Soil Carbon: Reassessing the “Permanence” of Soil Carbon Sequestration. Front Environ Sci. 2020 Nov 13;8:514701.
- Rodriguez-Blanco JD, Shaw S, Benning LG. The kinetics and mechanisms of amorphous calcium carbonate (ACC) crystallization to calcite, viavaterite. Nanoscale. 2011;3(1):265–71.
- Abbas G, Yuksel Orhan O. Energy penalties of CO2 storage and transportation. In: Advances and Technology Development in Greenhouse Gases: Emission, Capture and Conversion [Internet]. Elsevier; 2024 [cited 2025 Sep 8]. p. 187–216. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780443190674000115.
- Dooley K, Kartha S. Land-based negative emissions: risks for climate mitigation and impacts on sustainable development. Int Environ Agreem Polit Law Econ. 2018 Feb;18(1):79–98.
- Rosenstock TS, Wilkes A, Jallo C, Namoi N, Bulusu M, Suber M, Mboi D, Mulia R, Simelton E, Richards M, Gurwick N, Wollenberg E. Making trees count: Measurement and reporting of agroforestry in UNFCCC national communications of non-Annex I countries. Agric Ecosyst Environ. 2019 Nov;284:106569.
- Jose S. Agroforestry for ecosystem services and environmental benefits: an overview. Agrofor Syst. 2009 May;76(1):1–10.
| Pathways | Mechanism | Key Organisms/Plants | Main Carbonate/Products | Environmental Controls | References |
| Oxalate–Carbonate Pathway (OCP) | Plants produce Ca²⁺-oxalate crystals; soil bacteria and fungi oxidize oxalate, elevating pH and HCO₃⁻, leading to CaCO₃ precipitation. One key enzymatic route involves oxalate decarboxylation, wherein oxalyl-CoA decarboxylase converts oxalyl-CoA to formyl-CoA with release of CO₂, which can be hydrated to HCO₃⁻, driving carbonate mineralization. In wood and litter, brown-rot fungi frequently accumulate Ca-oxalate that is subsequently metabolized by oxalotrophic bacteria, sustaining localized alkalinity and carbonate precipitation. | Oxalogenic plants (e.g. Milicia, Ficus), oxalotrophic microbes including Burkholderia/Paraburkholderia lineages | Calcium carbonate (calcite/vaterite) in soil and wood | Requires Ca²⁺ availability, pH 7–8, and sufficient moisture | [53] |
| Microbial-Induced Carbonate Precipitation (MICP) | Microbial metabolisms (ureolysis, denitrification, photosynthesis, sulfate reduction) generate OH⁻ and HCO₃⁻, driving CaCO₃ precipitation. Extracellular carbonic anhydrases catalyze rapid interconversion of CO₂ and HCO₃⁻, enhancing carbonate alkalinity and CaCO₃ nucleation. Ureolytic, endospore-forming bacteria (e.g. Sporosarcina pasteurii) induce precipitation via urease-driven alkalinization, with applications in biocement and “bioconcrete.” Photosynthetic cyanobacteria and microalgae elevate pH and draw down dissolved inorganic carbon, stabilizing biocrusts and promoting carbonate precipitation. | Ureolytic bacteria (Sporosarcina spp.), denitrifiers, cyanobacteria, microalgae | Calcium carbonate (calcite) in soil matrices or biofilms | Influenced by rhizosphere exudates, anaerobic microzones | [14] |
| Phytolith Carbon (PhytOC) | Plants take up soluble silica, forming amorphous SiO₂ phytoliths that entrap occluded organic C. This physically protected C pool varies among grasses, cereals, and bamboo, with measurable contributions in croplands and high yields in bamboo forests under sufficient Si supply. | Grasses, crops, bamboo, other high-Si plants | Amorphous silica (opal) with occluded organic C | Depends on Si availability and neutral soil conditions |
[5] |
| Enhanced Rock Weathering (ERW) | Biogenic acids and root/soil CO₂ dissolve silicate minerals, releasing Ca²⁺/Mg²⁺ that form secondary carbonates. Standardized MRV approaches should pair soil inorganic C measurements with mineralogical speciation, isotopic partitioning, and biomarkers (e.g. ureC, oxc/frc) to constrain mechanisms and durability. | Plant roots, rhizosphere microbes; silicate rocks (basalt, dolerite) | Secondary carbonates (mainly calcite) in soils and waters | Enhanced by rock amendments and microbial exudates | [34] |
| Intracellular Biomineralization |
Specialized plant cells (idioblasts) precipitate Ca-oxalate crystals for storage/defense; these may later transform into CaCO₃ after tissue decay. Other widespread intracellular biominerals include Ca-carbonate and silica. Such crystals contribute to structural reinforcement, ion homeostasis, and defense against herbivory and pathogens. |
Oxalate-accumulating plants (e.g. figs, amaranths); many higher plants forming Ca-oxalate, Ca-carbonate, or silica crystals | Calcium oxalate (whewellite), calcium carbonate, amorphous silica in plant tissues | Driven by ion gradients, stress conditions, and species-specific regulation | [6] |
| Mineral / System | Chemical Formula / Pathway | Occurrence / Source / Case Study | Solubility / Mechanism | Longevity / Evidence | Role in Carbon Sequestration | References |
| Whewellite (CaOx·H₂O) | CaC₂O₄·H₂O (monohydrate) | Plant leaves, bark, wood (Ca-oxalate idioblasts) | Very low solubility; degrades by specialized microbes | Stable in tissue; microbially degraded (10–100 yr) | OCP precursor; oxalate substrate for oxalotrophs | [6] |
| Weddellite (CaOx·2H₂O) | CaC₂O₄·2H₂O (dihydrate) | Can form from whewellite recrystallization | Similar low solubility | Similar to whewellite | Transition phase in OCP cycle | [78] |
| Calcite | CaCO₃ | Soil carbonates, wood deposits | Insoluble at pH > 6; soluble in acid; most stable polymorph | Centuries to millennia in soil | Long-term stable carbonate sink | [79] |
| Aragonite | CaCO₃ (polymorph) | Some freshwater shells, precipitates | Transforms to calcite over time; slightly more soluble | Months–years (to transform) | Short-lived, converts to calcite reservoir | [80] |
| Vaterite | CaCO₃ (polymorph) | Rare biominerals (kidney stones, some algae) | Metastable; rapidly converts to calcite in water | Days–months (transforms quickly) | Transitional carbonate form | [14] |
| Phytolith (opal-A) | SiO₂·nH₂O (amorphous) | Grasses, cereals, bamboo (plant silica bodies) | Very low solubility; dissolves only in strong base/biogenic alkali | Thousands of years | Major PhytOC sink; stable C storage | [16] |
| Biogenic Calcite | CaCO₃ with organics | Coral reefs, mussel shells | Similar to calcite; isotopic CO₂ signatures | Geological records (very long) | Permanent geological sequestration | [55] |
| Iroko Trees (Ivory Coast) | OCP | Carbonate precipitation in acidic soils linked to tree metabolism | Isotopic δ13C confirms biogenic origin | δ13C: -14.2‰ in soil carbonates | Demonstrates OCP-driven C storage | [14] |
| Fig Trees (Kenya) | OCP | Novel trunk mineralization of CO₂ to CaCO₃; co-benefits: carbon capture, soil improvement, food | Ficus wakefieldii high-performing | Stable carbonate in trunk | OCP with ecosystem benefits | [28] |
| PhytOC (Cereals/Bamboo) | PhytOC | Long-term C storage in agro-ecosystems; enhanced by management | Very slow turnover | Global PhytOC sink: 26.35 ± 10.22 Tg CO₂/yr | Major agricultural C sequestration route | [2] |
| MICP (Lab Trials) | MICP | Improves soil strength and stability without harming plant growth | Driven by urease/denitrification | Up to +32.6% soil strength, +49.2% cohesion | Engineered carbonate precipitation | [46] |
| ERW (US Corn Belt) | ERW | Enhances crop yields while removing carbon | Mineral weathering in soils | 10.5 ± 3.8 t CO₂/ha over 4 years | Synergistic CDR + productivity | [6] |
| Oxalotrophs | Oxalate decarboxylase (Burkholderiales, Oxalobacteraceae) | Rhizosphere, decaying wood | Oxalate degradation | 50–70% OCP efficiency | OCP driver | [38] |
| Ureolytics | Urease hydrolysis (Sporosarcina, Bacillus) | Anaerobic soils | Rapid carbonate precipitation | Short-term to medium | MICP mechanism | [81] |
| Cyanobacteria | Photosynthetic CO₂ drawdown (Synechococcus, Nostoc) | Epiphytic, soil crusts | CO₂ fixation and local alkalinity | Long-lived in crusts | Local carbonate formation | [82] |
| Fungi | Oxalate production (Aspergillus, Penicillium) | Saprophytic, acidic soils | Oxalate precursor release | Continuous supply | Substrate providers for OCP | [83] |
| Denitrifiers | Nitrate reduction (Pseudomonas, Paracoccus) | Rhizosphere microsites | Produce alkalinity for precipitation | Medium persistence | MICP facilitator | [84] |
| System / Location | Pathway | Key Observations | CDR Rate / Potential | Permanence (approx.) | Evidence Type | References |
| Iroko Tree, Ivory Coast | OCP | Calcite precipitation in soils; ~1 ton CaCO₃ per tree lifetime | 2–4 t C ha⁻¹ yr⁻¹; ~1 ton CaCO₃ / lifetime | 10²–10⁶ years | Isotopic, mineralogical; Ref. 11, 54 | [87] |
| Figs, basaltic soils (Kenya) | OCP + MICP | Trunk calcification | 1–2 t C ha⁻¹ yr⁻¹ | 10²–10⁶ years (inferred from OCP permanence) | Field reports, 2025 conferences | [30] |
| Croplands, Global | PhytOC | Occluded C in phytoliths | 26.35 ± 10.22 Tg CO₂ yr⁻¹; ~0.5–1 t C ha⁻¹ yr⁻¹ | Millennia | Density fractionation; Ref. 18, 55 | [88] |
| Bamboo, Asia | PhytOC | Occluded C in phytoliths | 0.5–1 t C ha⁻¹ yr⁻¹ | Millennia | Density fractionation | [5] |
| US Corn Belt | ERW | Enhanced weathering of soils | 10.5 ± 3.8 t CO₂ ha⁻¹ (cumulative over 4 years) | 10⁴ years | Field trials; Ref. 25, 59 | [16] |
| Global Models | ERW | Modeled large-scale deployment potential | ~2 billion metric tons CO₂ yr⁻¹ | 10⁴ years | Modeling studies; Ref. 59 | [16] |
| MICP trials, Global | MICP + ERW | Soil amendments | 0.5–1 t C ha⁻¹ yr⁻¹ | 10²–10⁴ years (inferred from ERW/MICP permanence) | Experimental data | [35] |
| Impact Area | Co-benefits | Trade-offs | References |
| Soil Fertility | Improved nutrient availability and cycling; reduced soil acidification | Unintended alkalinization in already alkaline soils | [34] |
| Water Availability | Improved soil water retention and infiltration rates | Water-intensive methods may deplete resources in water-scarce regions | [75] |
| Land Use | Integration with food production (agroforestry, fruit trees) | Potential for land-use competition, creating food vs. carbon trade-offs | [87] |
| Biodiversity | Enhanced biodiversity through creation of diverse habitats | Inappropriate scaling can reduce biodiversity | [58] |
| Socio Economic | Increased crop yields and reduced fertilizer costs for farmers; economic resilience and food security | Land dispossession and the creation of “ecological sacrifice zones” | [116] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




