Submitted:
15 October 2025
Posted:
17 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines
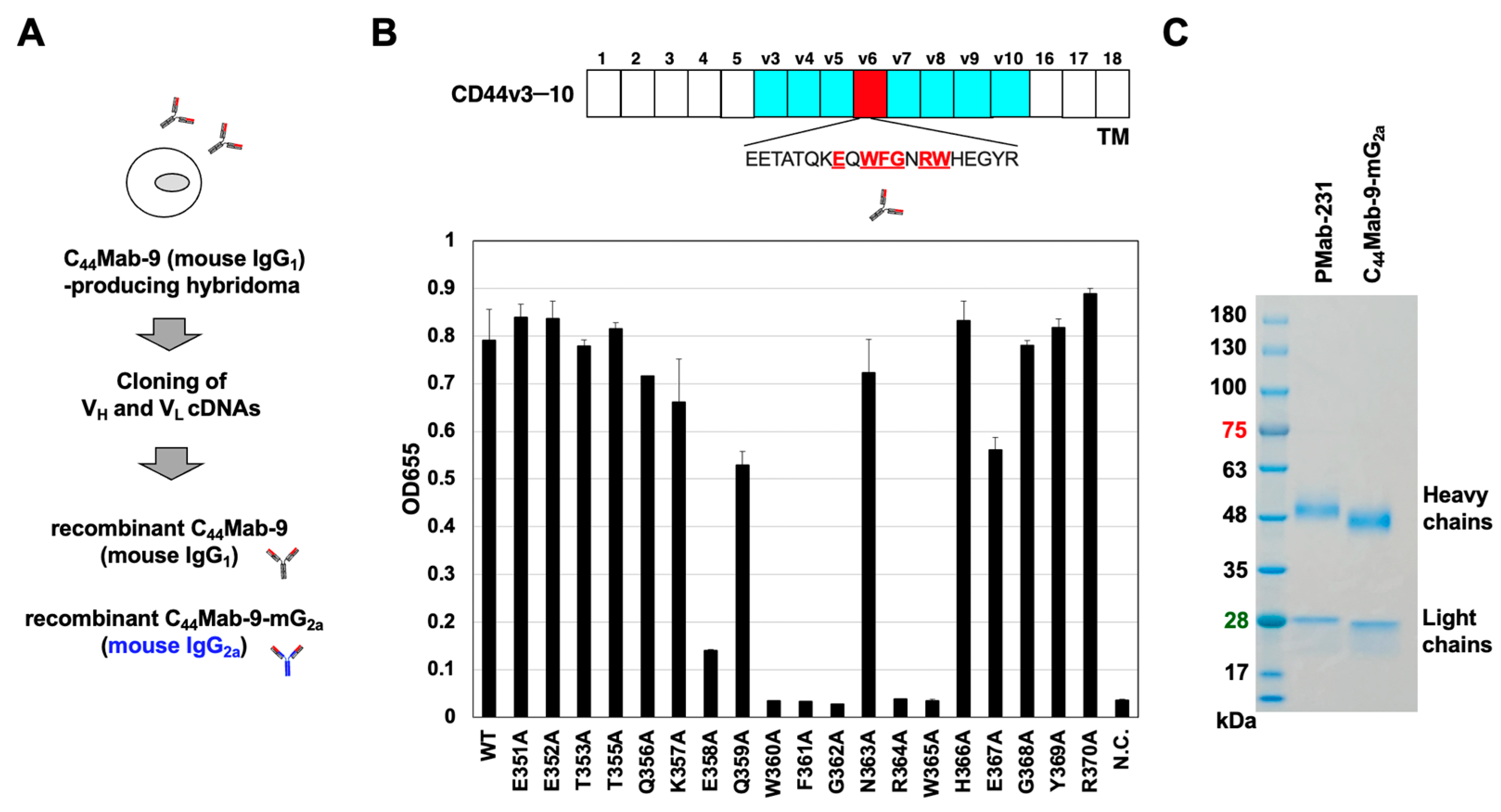
2.2. Recombinant mAb Production
2.3. ELISA
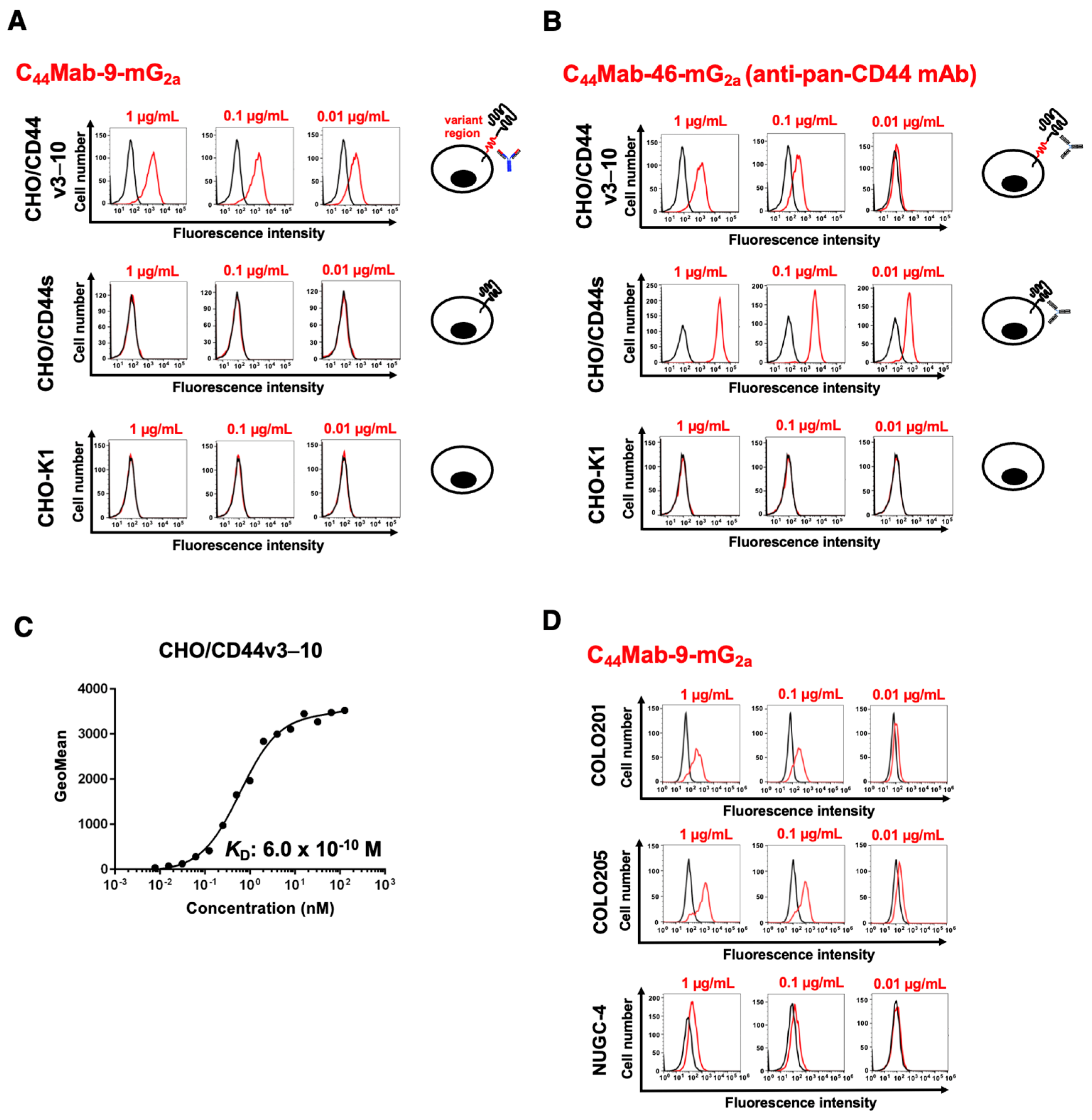
2.4. Flow Cytometry
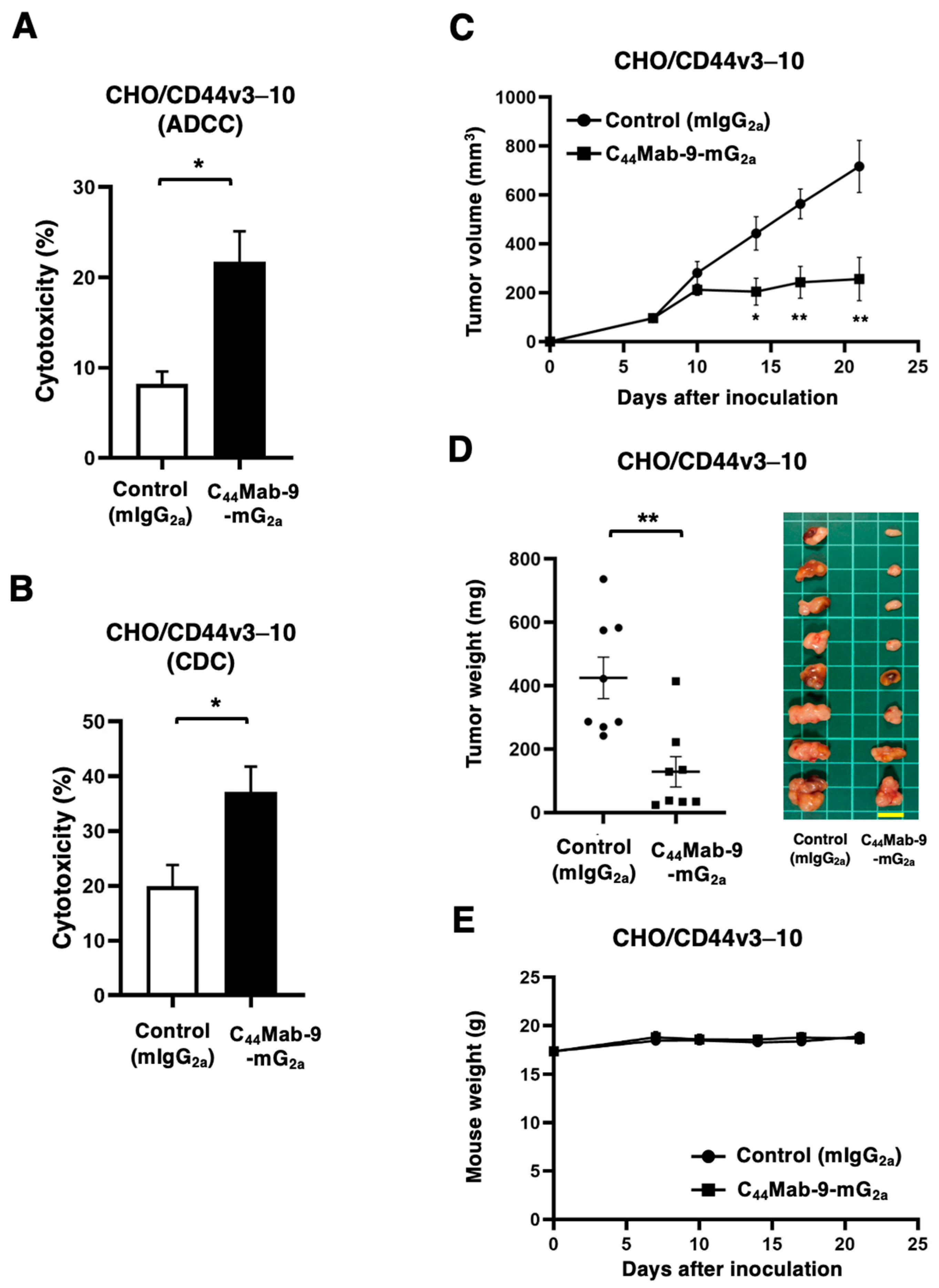
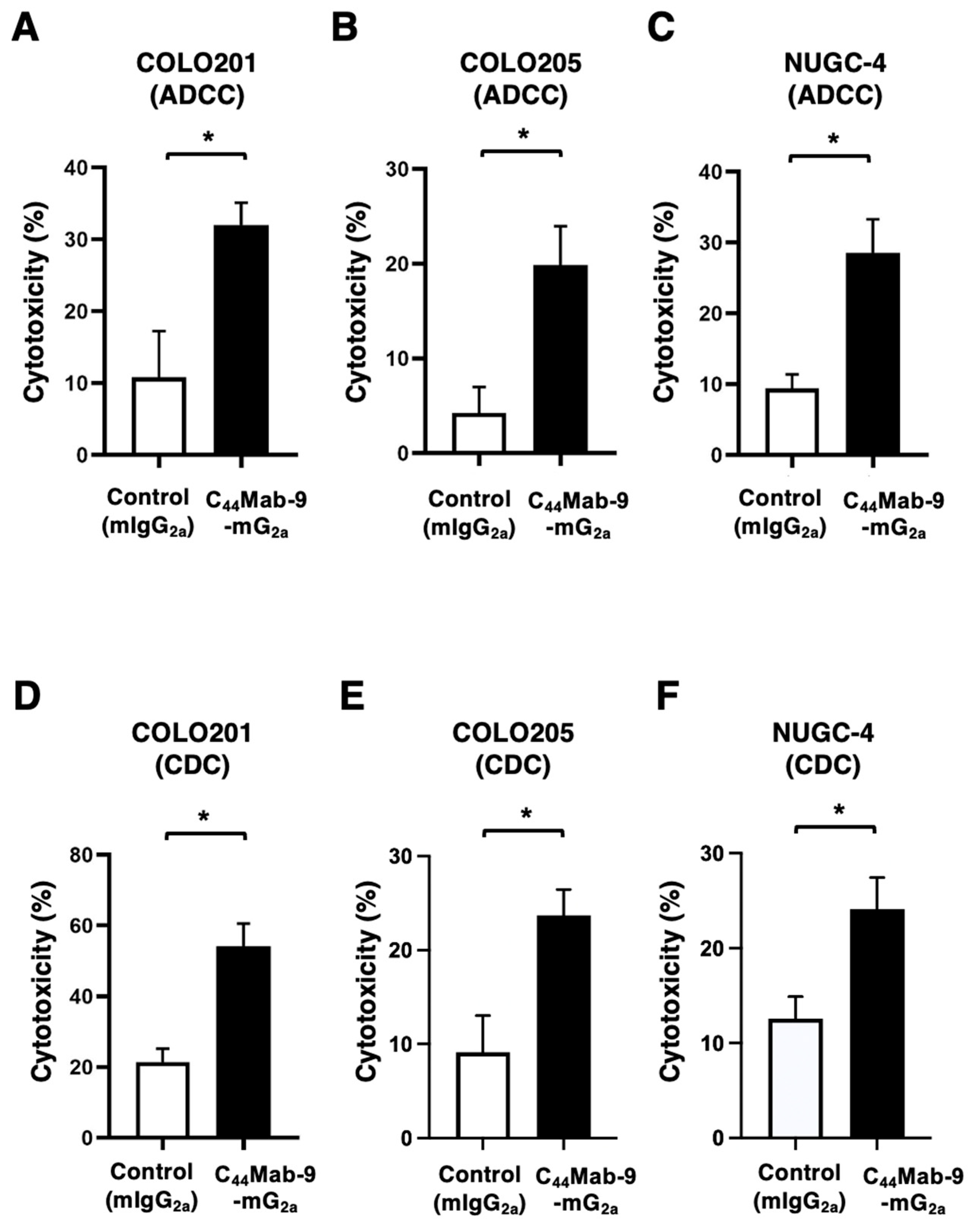
2.5. Measurement of ADCC by C44Mab-9-mG2a
2.6. Measurement of CDC by C44Mab-9-mG2a
2.7. Antitumor Activity of C44Mab-9-mG2a
3. Results
3.1. Production of Recombinant Anti-CD44v6 mAbs
3.2. Flow Cytometry Using C44Mab-9-mG2a
3.3. ADCC, CDC, and Antitumor Effects Against CHO/CD44v3–10 by C44Mab-9-mG2a
3.4. ADCC and CDC Against COLO201, COLO205, and NUGC-4 by C44Mab-9-mG2a
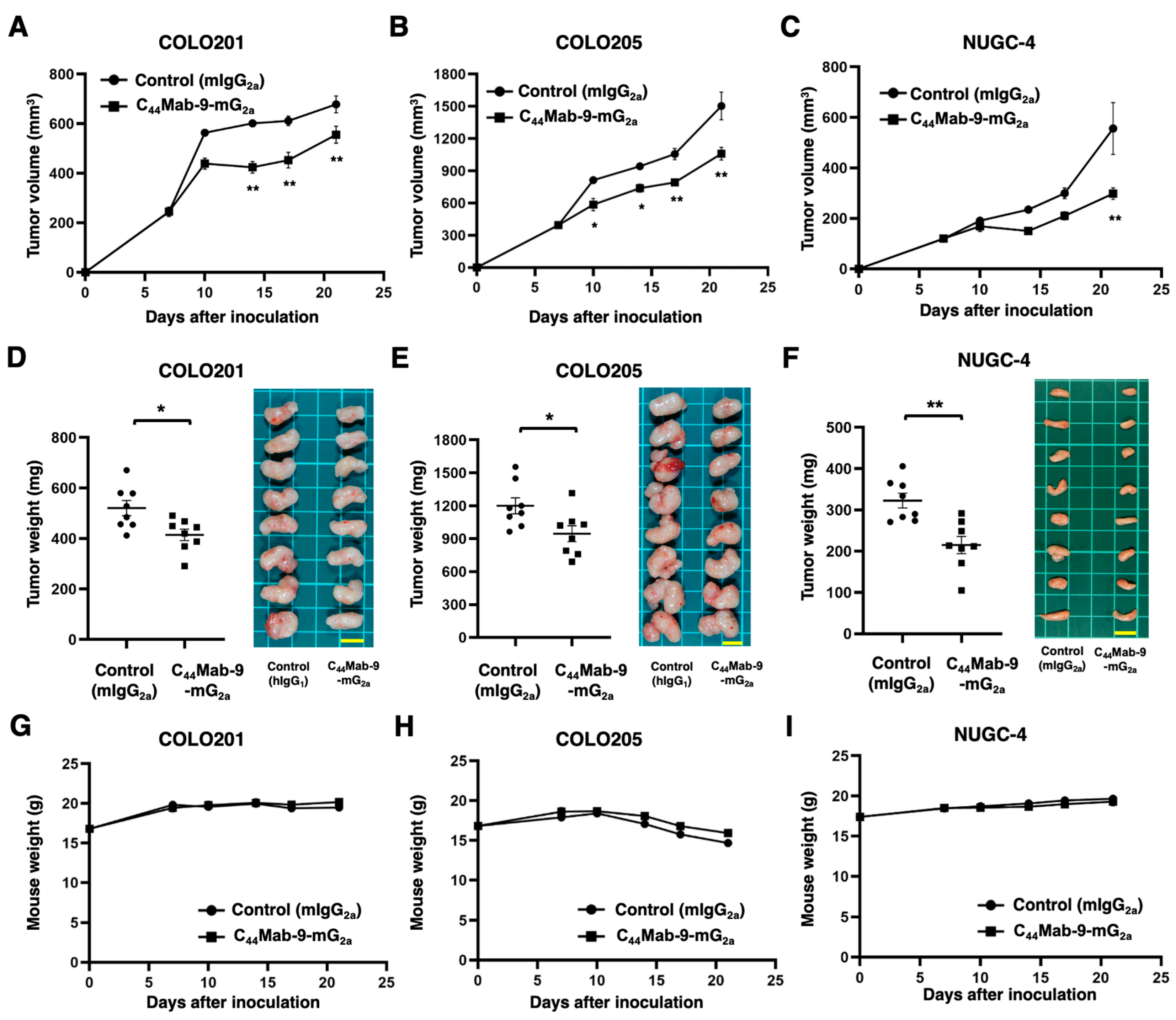
3.4. Antitumor Effects by C44Mab-9-mG2a Against COLO201, COLO205, and NUGC-4 xenografts
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stamenkovic, I.; Amiot, M.; Pesando, J.M.; Seed, B. A lymphocyte molecule implicated in lymph node homing is a member of the cartilage link protein family. Cell 1989;56(6): 1057-1062. [CrossRef]
- Ponta, H.; Sherman, L.; Herrlich, P.A. CD44: from adhesion molecules to signalling regulators. Nat Rev Mol Cell Biol 2003;4(1): 33-45. [CrossRef] [PubMed]
- Zöller, M. CD44: can a cancer-initiating cell profit from an abundantly expressed molecule? Nat Rev Cancer 2011;11(4): 254-267. [CrossRef] [PubMed]
- Guo, Q.; Yang, C.; Gao, F. The state of CD44 activation in cancer progression and therapeutic targeting. Febs j 2021. [CrossRef] [PubMed]
- Slevin, M.; Krupinski, J.; Gaffney, J.; et al. Hyaluronan-mediated angiogenesis in vascular disease: uncovering RHAMM and CD44 receptor signaling pathways. Matrix Biol 2007;26(1): 58-68. [CrossRef]
- Hassn Mesrati, M.; Syafruddin, S.E.; Mohtar, M.A.; Syahir, A. CD44: A Multifunctional Mediator of Cancer Progression. Biomolecules 2021;11(12). [CrossRef]
- Morath, I.; Hartmann, T.N.; Orian-Rousseau, V. CD44: More than a mere stem cell marker. Int J Biochem Cell Biol 2016;81(Pt A): 166-173. [CrossRef]
- Bennett, K.L.; Jackson, D.G.; Simon, J.C.; et al. CD44 isoforms containing exon V3 are responsible for the presentation of heparin-binding growth factor. J Cell Biol 1995;128(4): 687-698. [CrossRef] [PubMed]
- Orian-Rousseau, V.; Chen, L.; Sleeman, J.P.; Herrlich, P.; Ponta, H. CD44 is required for two consecutive steps in HGF/c-Met signaling. Genes Dev 2002;16(23): 3074-3086. [CrossRef]
- Ishimoto, T.; Nagano, O.; Yae, T.; et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc(-) and thereby promotes tumor growth. Cancer Cell 2011;19(3): 387-400. [CrossRef]
- Massagué, J.; Ganesh, K. Metastasis-Initiating Cells and Ecosystems. Cancer Discov 2021;11(4): 971-994. [CrossRef]
- Ganesh, K.; Massagué, J. Targeting metastatic cancer. Nat Med 2021;27(1): 34-44. [CrossRef] [PubMed]
- Ma, L.; Dong, L.; Chang, P. CD44v6 engages in colorectal cancer progression. Cell Death Dis 2019;10(1): 30. [CrossRef]
- Todaro, M.; Gaggianesi, M.; Catalano, V.; et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell 2014;14(3): 342-356. [CrossRef] [PubMed]
- Heider, K.H.; Sproll, M.; Susani, S.; et al. Characterization of a high-affinity monoclonal antibody specific for CD44v6 as candidate for immunotherapy of squamous cell carcinomas. Cancer Immunol Immunother 1996;43(4): 245-253. [CrossRef]
- Heider, K.H.; Mulder, J.W.; Ostermann, E.; et al. Splice variants of the cell surface glycoprotein CD44 associated with metastatic tumour cells are expressed in normal tissues of humans and cynomolgus monkeys. Eur J Cancer 1995;31a(13-14): 2385-2391. [CrossRef] [PubMed]
- Wang, Z.; Tang, Y.; Xie, L.; et al. The Prognostic and Clinical Value of CD44 in Colorectal Cancer: A Meta-Analysis. Front Oncol 2019;9: 309. [CrossRef]
- Verel, I.; Heider, K.H.; Siegmund, M.; et al. Tumor targeting properties of monoclonal antibodies with different affinity for target antigen CD44V6 in nude mice bearing head-and-neck cancer xenografts. Int J Cancer 2002;99(3): 396-402. [CrossRef]
- Riechelmann, H.; Sauter, A.; Golze, W.; et al. Phase I trial with the CD44v6-targeting immunoconjugate bivatuzumab mertansine in head and neck squamous cell carcinoma. Oral Oncol 2008;44(9): 823-829. [CrossRef]
- Tijink, B.M.; Buter, J.; de Bree, R.; et al. A phase I dose escalation study with anti-CD44v6 bivatuzumab mertansine in patients with incurable squamous cell carcinoma of the head and neck or esophagus. Clin Cancer Res 2006;12(20 Pt 1): 6064-6072. [CrossRef]
- Tang, L.; Huang, H.; Tang, Y.; et al. CD44v6 chimeric antigen receptor T cell specificity towards AML with FLT3 or DNMT3A mutations. Clin Transl Med 2022;12(9): e1043. [CrossRef]
- Greco, B.; Malacarne, V.; De Girardi, F.; et al. Disrupting N-glycan expression on tumor cells boosts chimeric antigen receptor T cell efficacy against solid malignancies. Sci Transl Med 2022;14(628): eabg3072. [CrossRef] [PubMed]
- Porcellini, S.; Asperti, C.; Corna, S.; et al. CAR T Cells Redirected to CD44v6 Control Tumor Growth in Lung and Ovary Adenocarcinoma Bearing Mice. Front Immunol 2020;11: 99. [CrossRef]
- Saeidpour Masouleh, S.; Nasiri, K.; Ostovar Ravari, A.; et al. Advances and challenges in CAR-T cell therapy for head and neck squamous cell carcinoma. Biomark Res 2025;13(1): 69. [CrossRef] [PubMed]
- Ciulean, I.S.; Fischer, J.; Quaiser, A.; et al. CD44v6 specific CAR-NK cells for targeted immunotherapy of head and neck squamous cell carcinoma. Front Immunol 2023;14: 1290488. [CrossRef]
- Tawara, M.; Suzuki, H.; Goto, N.; et al. A Novel Anti-CD44 Variant 9 Monoclonal Antibody C44Mab-1 was Developed for Immunohistochemical Analyses Against Colorectal Cancers Curr. Issues Mol. Biol. 2023;45(4): 3658-3673. [CrossRef] [PubMed]
- Suzuki, H.; Kitamura, K.; Goto, N.; et al. A Novel Anti-CD44 Variant 3 Monoclonal Antibody C(44)Mab-6 Was Established for Multiple Applications. Int J Mol Sci 2023;24(9). [CrossRef]
- Kudo, Y.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of a Novel Anti-CD44 Variant 5 Monoclonal Antibody C(44)Mab-3 for Multiple Applications against Pancreatic Carcinomas. Antibodies (Basel) 2023;12(2). [CrossRef]
- Ejima, R.; Suzuki, H.; Tanaka, T.; et al. Development of a Novel Anti-CD44 Variant 6 Monoclonal Antibody C(44)Mab-9 for Multiple Applications against Colorectal Carcinomas. Int J Mol Sci 2023;24(4). [CrossRef]
- Li, G.; Suzuki, H.; Ohishi, T.; et al. Antitumor activities of a defucosylated anti-EpCAM monoclonal antibody in colorectal carcinoma xenograft models. Int J Mol Med 2023;51(2). [CrossRef]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci 2024;25(3). [CrossRef] [PubMed]
- Orian-Rousseau, V. CD44, a therapeutic target for metastasising tumours. Eur J Cancer 2010;46(7): 1271-1277. [CrossRef]
- Maurya, S.K.; Jaramillo-Gómez, J.A.; Rehman, A.U.; et al. Mucin 5AC Promotes Breast Cancer Brain Metastasis through cMET/CD44v6. Clin Cancer Res 2025;31(5): 921-935. [CrossRef]
- Chen, H.; Ling, R.; Lai, J.; et al. CD44v6-mediated regulation of gastric cancer stem cells: a potential therapeutic target. Clin Exp Med 2025;25(1): 80. [CrossRef] [PubMed]
- Stornaiuolo, A.; Valentinis, B.; Sirini, C.; et al. Characterization and Functional Analysis of CD44v6.CAR T Cells Endowed with a New Low-Affinity Nerve Growth Factor Receptor-Based Spacer. Hum Gene Ther 2021;32(13-14): 744-760. [CrossRef]
- Wielenga, V.J.; van der Neut, R.; Offerhaus, G.J.; Pals, S.T. CD44 glycoproteins in colorectal cancer: expression, function, and prognostic value. Adv Cancer Res 2000;77: 169-187.
- Pan, T.; Zhu, J.; Wang, X.; et al. MEN1 Promotes Ferroptosis by Disrupting CD44 Alternative Splicing to Suppress Lung Cancer. Cancer Res 2025. in press. [CrossRef] [PubMed]
- Matzke, A.; Herrlich, P.; Ponta, H.; Orian-Rousseau, V. A five-amino-acid peptide blocks Met- and Ron-dependent cell migration. Cancer Res 2005;65(14): 6105-6110. [CrossRef] [PubMed]
- Tremmel, M.; Matzke, A.; Albrecht, I.; et al. A CD44v6 peptide reveals a role of CD44 in VEGFR-2 signaling and angiogenesis. Blood 2009;114(25): 5236-5244. [CrossRef]
- Orian-Rousseau, V.; Ponta, H. Perspectives of CD44 targeting therapies. Arch Toxicol 2015;89(1): 3-14. [CrossRef]
- Vey, N.; Delaunay, J.; Martinelli, G.; et al. Phase I clinical study of RG7356, an anti-CD44 humanized antibody, in patients with acute myeloid leukemia. Oncotarget 2016;7(22): 32532-32542. [CrossRef]
- Menke-van der Houven van Oordt, C.W.; Gomez-Roca, C.; van Herpen, C.; et al. First-in-human phase I clinical trial of RG7356, an anti-CD44 humanized antibody, in patients with advanced, CD44-expressing solid tumors. Oncotarget 2016;7(48): 80046-80058. [CrossRef]
- Birzele, F.; Voss, E.; Nopora, A.; et al. CD44 Isoform Status Predicts Response to Treatment with Anti-CD44 Antibody in Cancer Patients. Clin Cancer Res 2015;21(12): 2753-2762. [CrossRef]
- Zhang, S.; Wu, C.C.; Fecteau, J.F.; et al. Targeting chronic lymphocytic leukemia cells with a humanized monoclonal antibody specific for CD44. Proc Natl Acad Sci U S A 2013;110(15): 6127-6132. [CrossRef] [PubMed]
- Kaneko, M.K.; Suzuki, H.; Kato, Y. Establishment of a Novel Cancer-Specific Anti-HER2 Monoclonal Antibody H(2)Mab-250/H(2)CasMab-2 for Breast Cancers. Monoclon Antib Immunodiagn Immunother 2024;43(2): 35-43.
- Hosking, M.P.; Shirinbak, S.; Omilusik, K.; et al. Preferential tumor targeting of HER2 by iPSC-derived CAR T cells engineered to overcome multiple barriers to solid tumor efficacy. Cell Stem Cell 2025;32(7): 1087-1101.e1084. [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



