Submitted:
11 October 2025
Posted:
14 October 2025
You are already at the latest version
Abstract
Keywords:
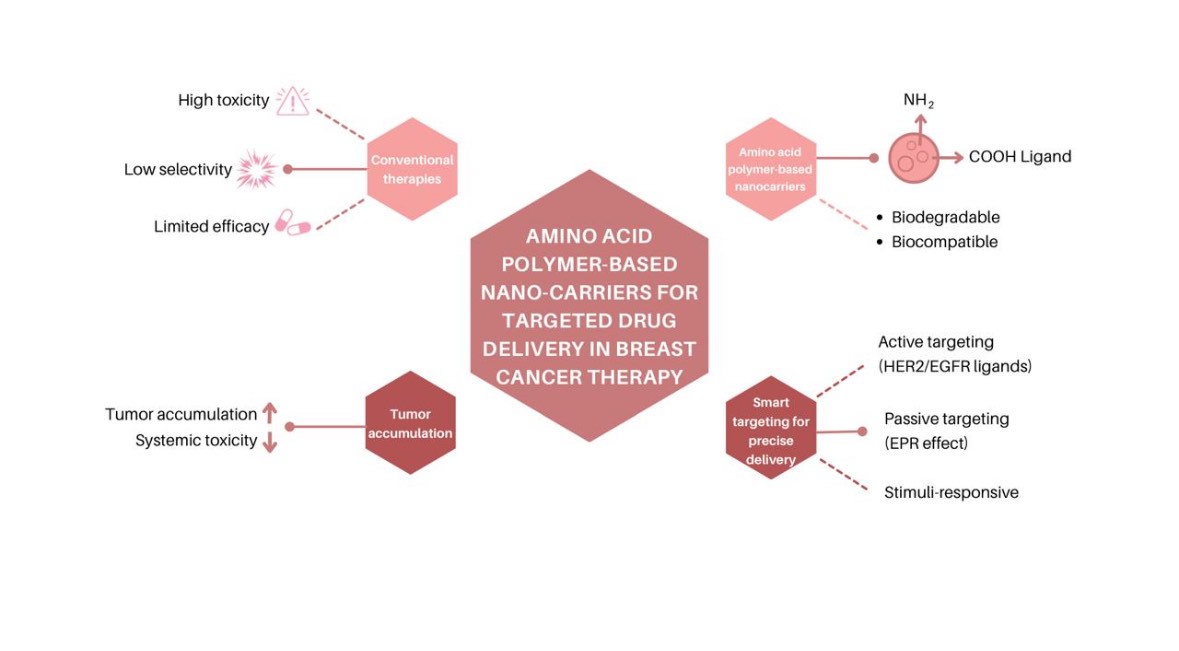
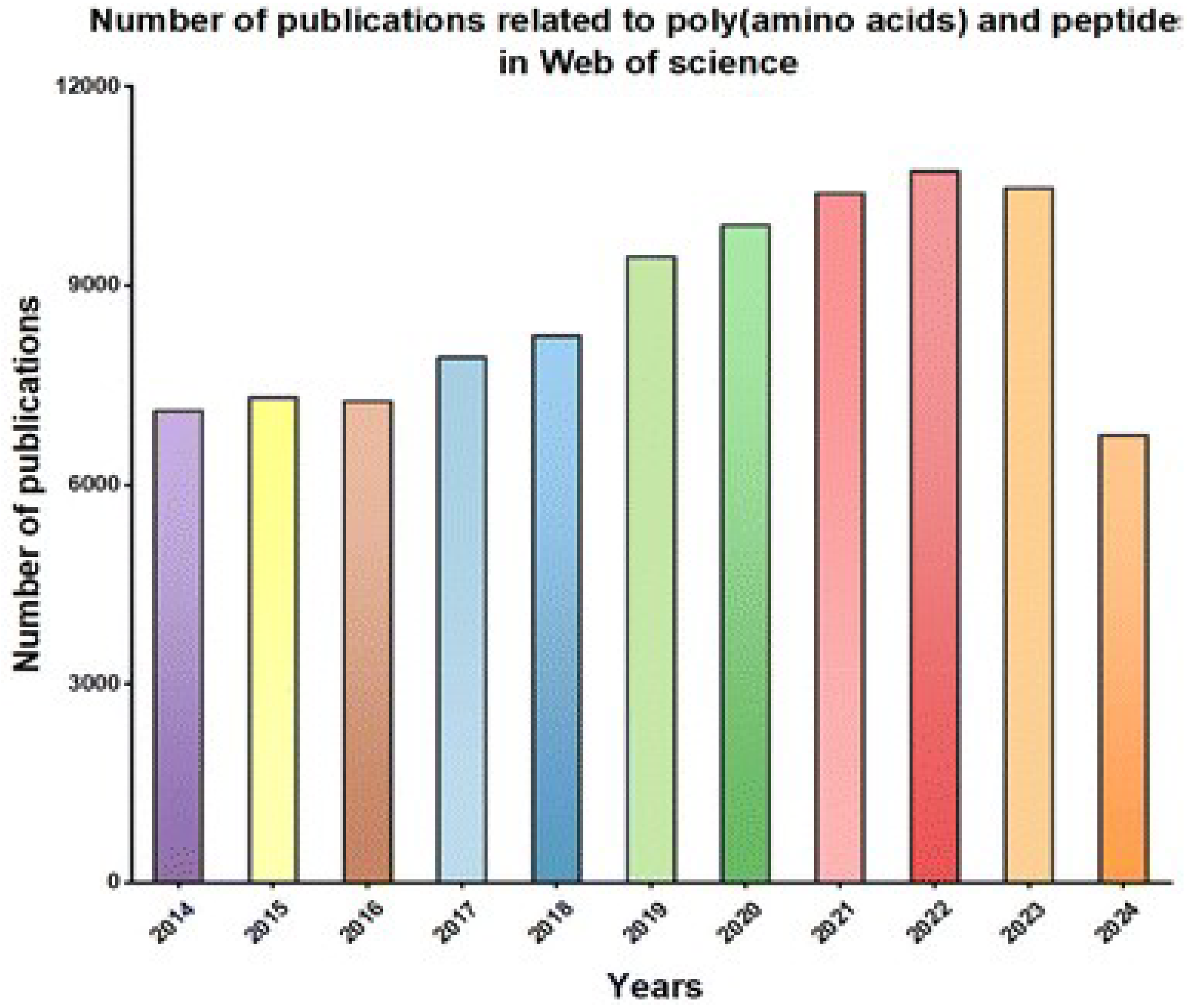
1. Introduction
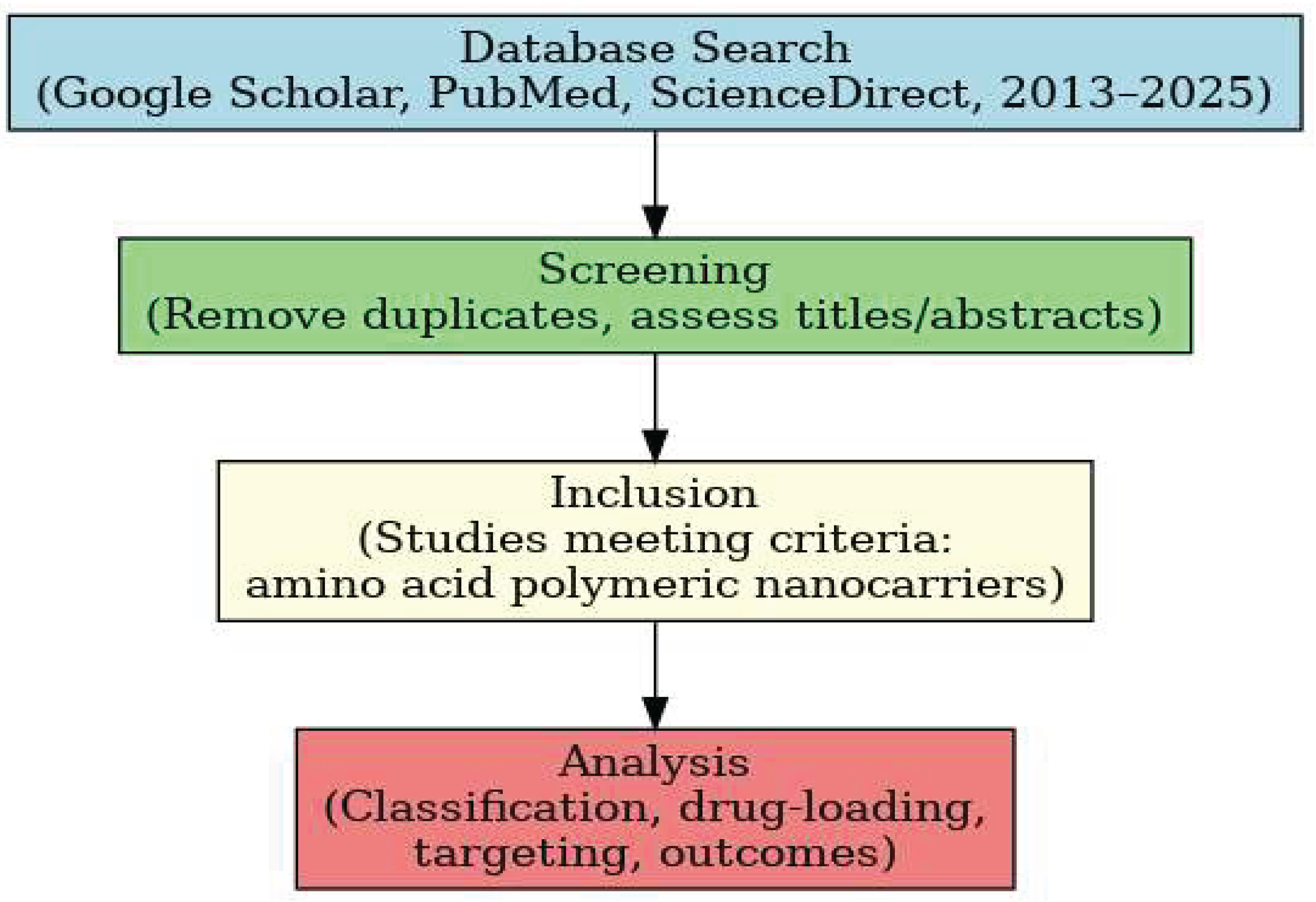
2. Methodology
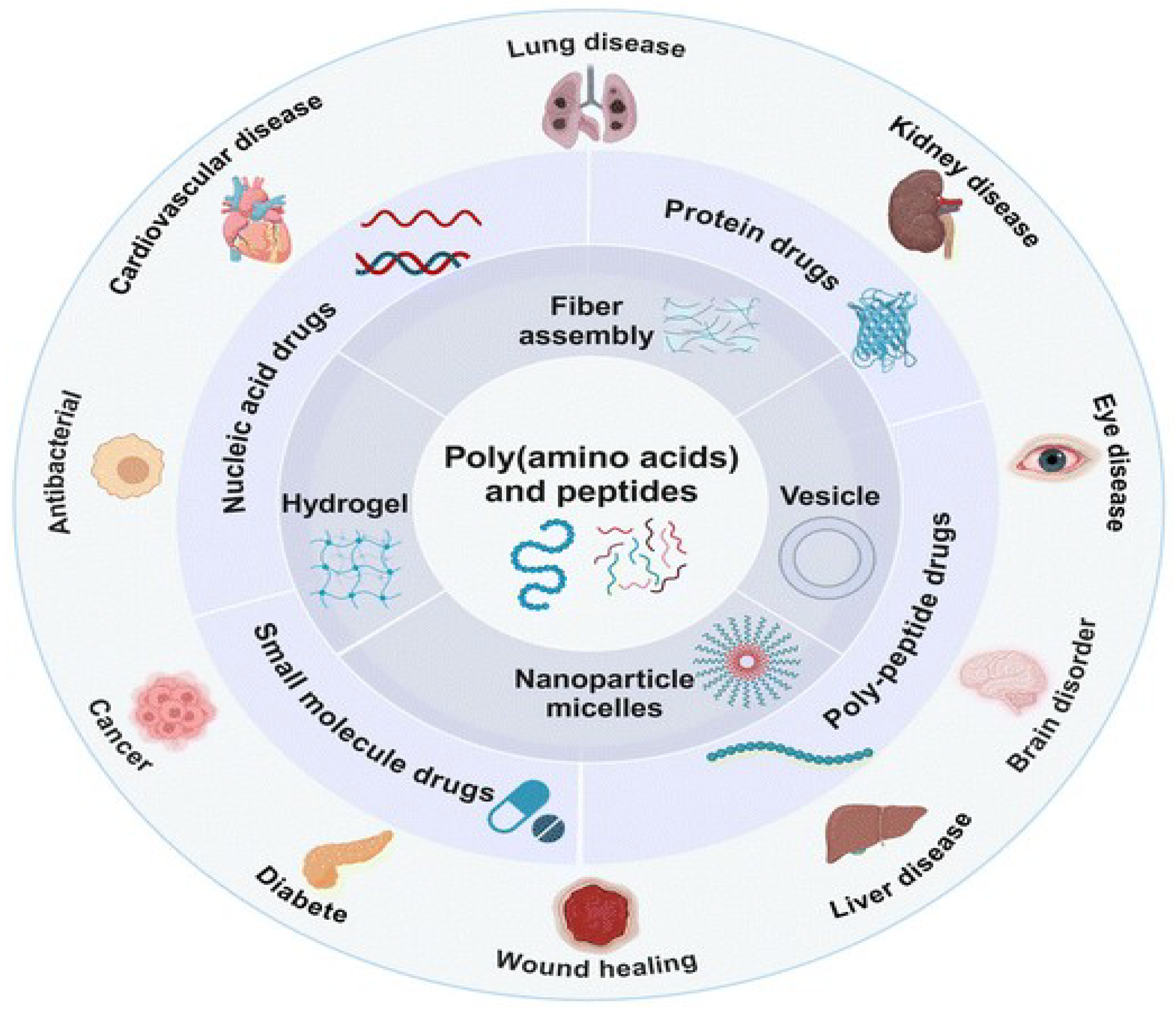
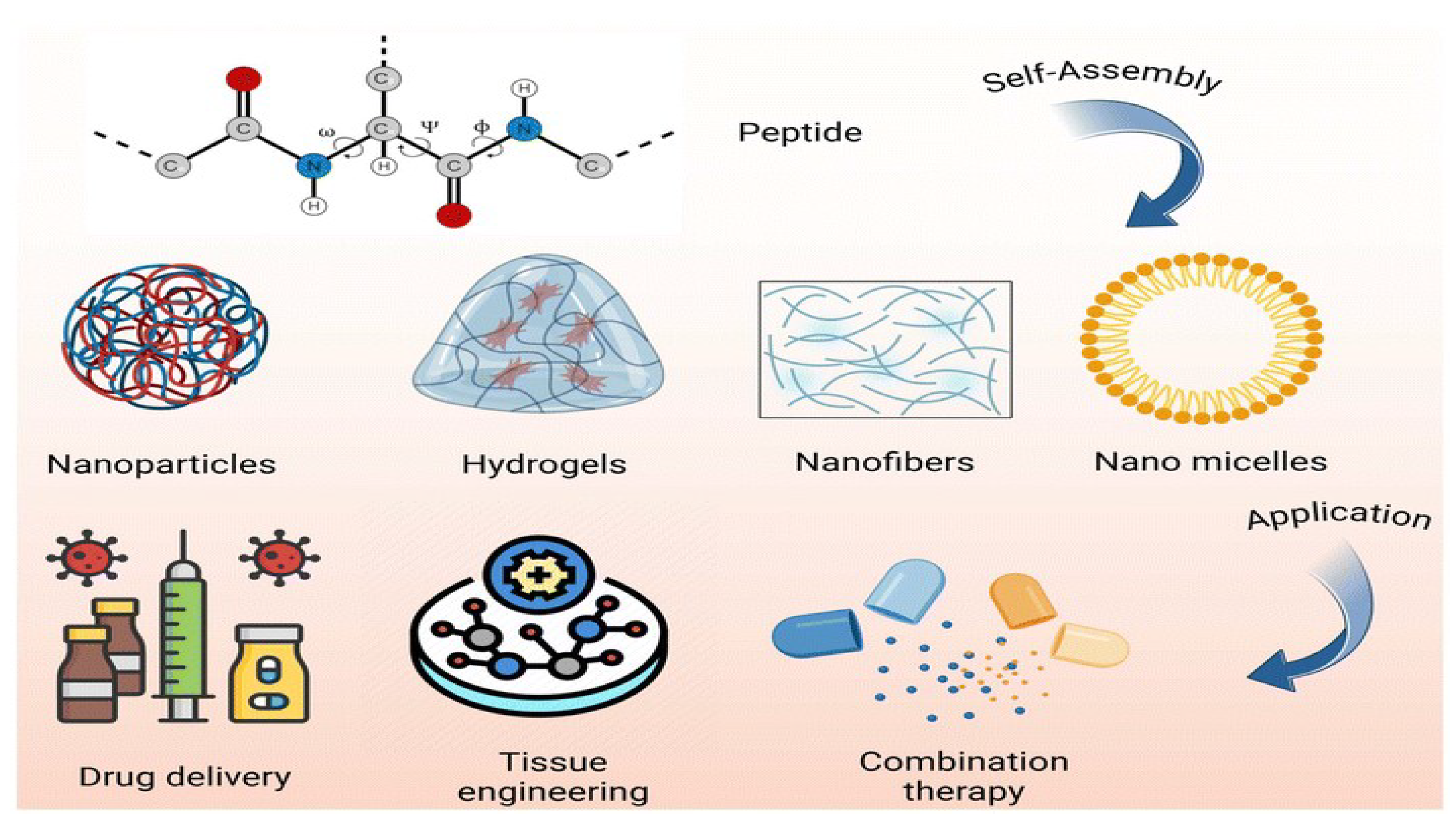
3. Amino Acid Polymers: Building Blocks for Smart Nano-carriers
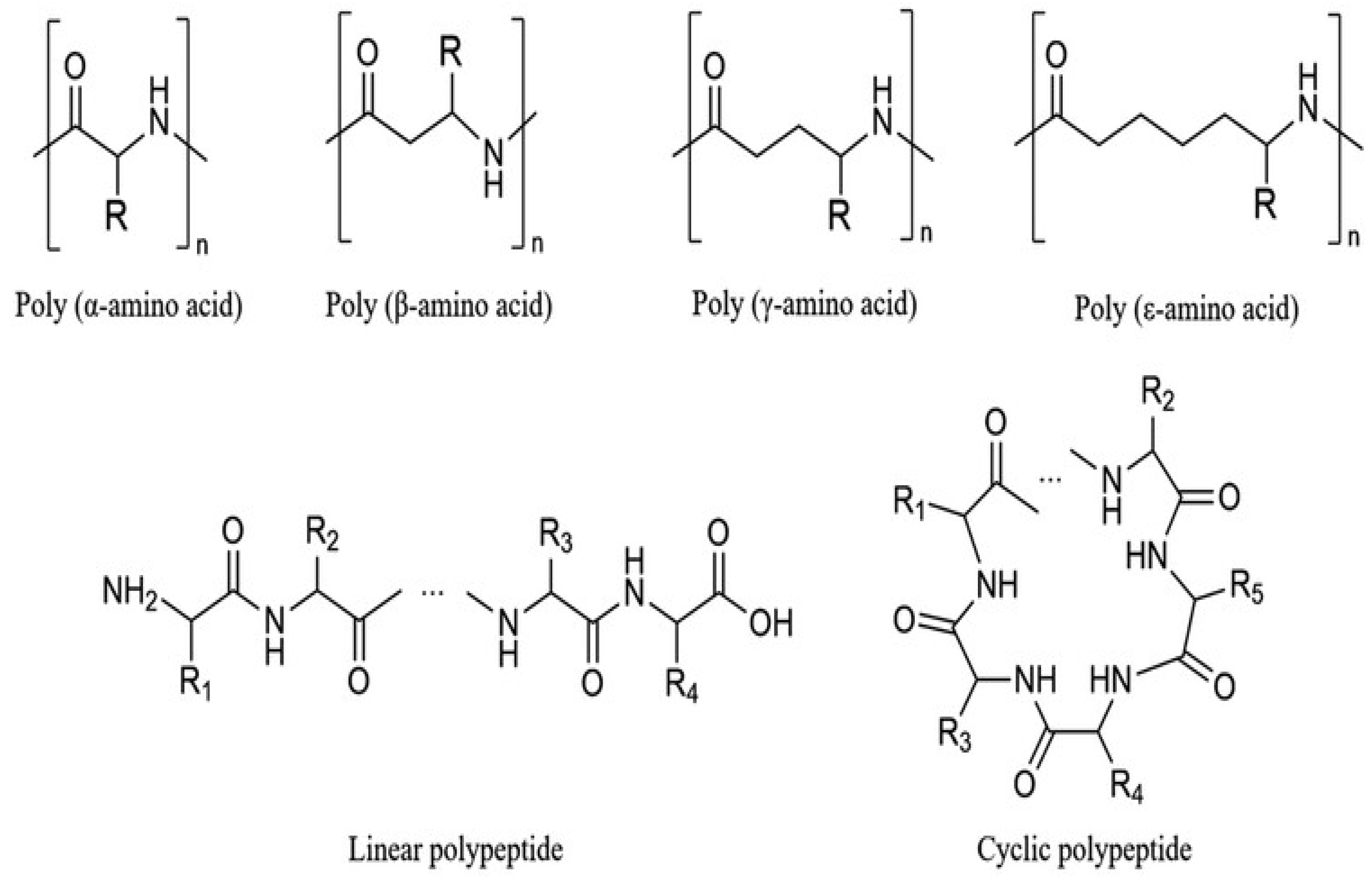
3.1. Classification: Natural vs. Synthetic Amino Acid Polymers
3.2. Drug-Loading Functionalities and Structural Properties
3.3. Biodegradability and Compatibility with Tumor Environment
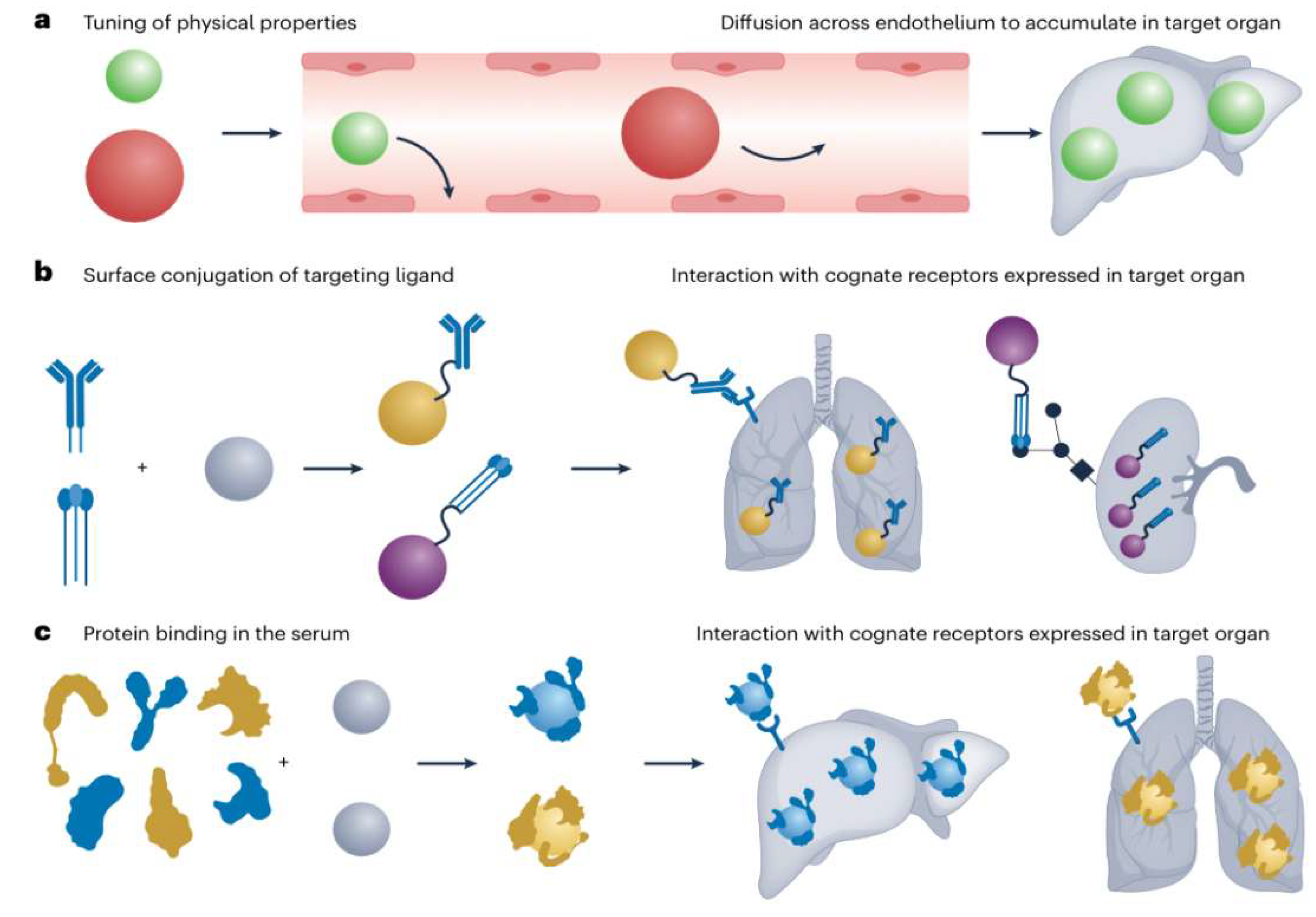
- Ligand Functionalization: This strategy involves conjugating specific ligands onto the surface of poly (amino acid) carriers to enable active recognition and selective binding to target cells or tissues. Such functionalization mitigates nonspecific uptake, thereby minimizing off-target effects and enhancing therapeutic outcomes.
- Passive Targeting: Leveraging the enhanced permeability and retention (EPR) effect, nanoparticles exploit the leaky vasculature and deficient lymphatic drainage characteristic of tumor tissues to accumulate preferentially at pathological sites.
- Stimuli-Responsive Release: Poly (amino acid) carriers can be engineered to respond to local environmental stimuli—such as acidic pH within tumor microenvironments—triggering site-specific drug release. For instance, polyglutamic acid nanoparticles are designed to degrade under acidic conditions prevalent in tumors, ensuring localized therapeutic delivery.
- Cell-Penetrating Peptides (CPPs): CPPs, exemplified by TAT peptides, facilitate the translocation of therapeutic molecules across cellular membranes, significantly enhancing intracellular drug delivery and therapeutic efficacy.
3.4. Strategies for Polymer Modification and Conjugation
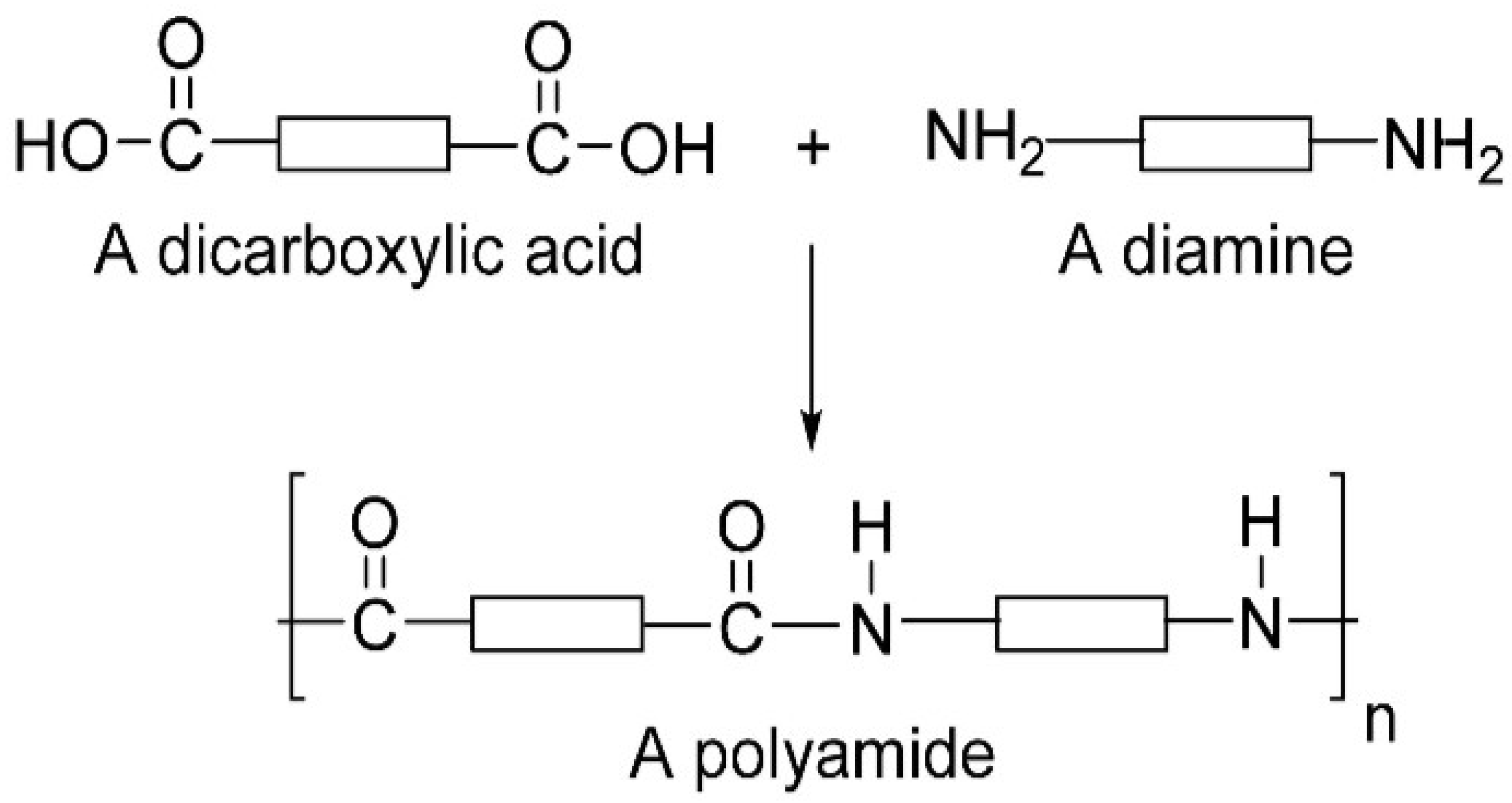
3.4.1. Strategic Chemical Routes for Poly (amino acid) Fabrication
3.4.2. Enzyme-Guided Approaches in Peptide Formation
3.4.3. Biosynthetic Systems for Amino Acid Polymer Production
3.4.4. Directed Self-Assembly of Functional Peptide Structures
4. Targeted Drug Delivery Strategies in Breast Cancer
4.1. Understanding Breast Tumor Microenvironment
4.2. Passive vs. Active Targeting Using Polymer-Based Nano-carriers
4.3. Ligand-Mediated Targeting (e.g., HER2, EGFR)
4.4. Stimuli-Responsive Release Systems (pH, enzymes, redox)
5. Literature-Based Applications in Breast Cancer Therapy
5.1. Summary of Preclinical Findings on Amino Acid Polymer Carriers
5.2. Release Profiles, Tumor Penetration, and Study Limitations
6. Challenges Highlighted in Existing Literature
7. Research Gaps and Future Study Directions
7.1. Unexplored Polymers or Ligand Strategies
7.2. Combinational Drug Delivery Approaches
7.3. Integration with Computational and AI-Guided Design
7.4. Recommendations Across Reviewed Papers
8. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ali, D.K. et al. (2022) ‘Reduction and pH Dualresponsive Biobased Poly(disulfide-amide) Nanoparticles Using Cystine Amino Acid for Targeting Release of Doxorubicin Anticancer Drug’, Journal of Polymers and the Environment, 30(11), pp. 4809–4820. Available at:. [CrossRef]
- Araújo, J.L. et al. (2023) ‘Benefits of using polymeric nanoparticles in breast cancer treatment: a systematic review’, 3 Biotech. Springer Science and Business Media Deutschland GmbH. Available at:. [CrossRef]
- Atoufi Z., Z.P., M.G.H., A.A., B.Z., K.S.K. (2017) ‘A novel bio electro active alginate-aniline tetramer/ agarose scaffold for tissue engineering: synthesis, characterization, drug release and cell culture study’, Journal of Biomaterials Science, Polymer Edition, 28(15), pp. 1617–1638.
- Bel’skaya, L. V., Gundyrev, I.A. and Solomatin, D. V. (2023) ‘The Role of Amino Acids in the Diagnosis, Risk Assessment, and Treatment of Breast Cancer: A Review’, Current Issues in Molecular Biology. Multidisciplinary Digital Publishing Institute (MDPI), pp. 7513–7537. Available at:. [CrossRef]
- Boddu, S.H.S. et al. (2021) ‘Polyamide/poly(Amino acid) polymers for drug delivery’, Journal of Functional Biomaterials. MDPI. Available at:. [CrossRef]
- Bray, F. et al. (2021) ‘The ever-increasing importance of cancer as a leading cause of premature death worldwide’, Cancer, 127(16), pp. 3029–3030. Available at:. [CrossRef]
- Cardoso, V.F. et al. (2018a) ‘Fluorinated polymers as smart materials for advanced biomedical applications’, Polymers. MDPI AG. Available at:. [CrossRef]
- Cardoso, V.F. et al. (2018b) ‘Fluorinated polymers as smart materials for advanced biomedical applications’, Polymers. MDPI AG. Available at:. [CrossRef]
- Chaudhari, R., Patel, V. and Kumar, A. (2024) ‘Cutting-edge approaches for targeted drug delivery in breast cancer: beyond conventional therapies’, Nanoscale Advances. Royal Society of Chemistry, pp. 2270–2286. Available at:. [CrossRef]
- Chen M.C., L.M.H., W.K.W., L.Z.W., L.B.H., C.D.H. (2015) ‘Near-Infrared Light-Responsive Composite Microneedles for On-Demand Transdermal Drug Delivery’, Biomacromolecules, 16(5), pp. 1598–1607.
- Chen, S. et al. (2023) ‘Estimates and Projections of the Global Economic Cost of 29 Cancers in 204 Countries and Territories from 2020 to 2050’, JAMA Oncology, 9(4), pp. 465–472. Available at:. [CrossRef]
- Chung M.F., C.W.T., L.H.Y., H.C.W., H.H.C., Y.C.M., S.H.W. (2014) ‘Inflammation-Induced Drug Release by using a pH-Responsive Gas-Generating Hollow-Microsphere System for the Treatment of Osteomyelitis’, Advanced Healthcare Materials, 3(11), pp. 1854–1861.
- Davaran, S. et al. (2017) ‘Novel dual stimuli-responsive ABC triblock copolymer: RAFT synthesis, “schizophrenic” micellization, and its performance as an anticancer drug delivery nanosystem’, Journal of Colloid and Interface Science, 488, pp. 282–293. Available at:. [CrossRef]
- DeSantis, C.E. et al. (2016) ‘Breast cancer statistics, 2015: Convergence of incidence rates between black and white women’, CA: A Cancer Journal for Clinicians, 66(1), pp. 31–42. Available at:. [CrossRef]
- Dias, J.T. et al. (no date) ‘DNA as Molecular Local Thermal Probe for Magnetic Hyperthermia Analysis.** [*] ((Title(s) Initial(s) Surename(s) of Author(s) including Corresponding Author(s))) ((Department)) ((Institution)) ((Address 1)) Fax: (+) E-mail: Homepage ((optional)): ((Title(s) Initial(s) Surename(s) of Author(s))) ((Department)) ((Institution))’. Available at:. [CrossRef]
- Dilliard, S.A. and Siegwart, D.J. (2023) ‘Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs’, Nature Reviews Materials. Nature Research, pp. 282–300. Available at:. [CrossRef]
- Duan J., D.J., Z.T., S.Z., D.J., Z.Y., M.X.P. iron oxide nanoparticles for systemic siRNA delivery in experimental arthritis. (2014) ‘Polyethyleneimine-Functionalized Iron Oxide Nanoparticles for Systemic siRNA Delivery in Experimental Arthritis’, Nanomedicine, 9(6).
- Duan, Z. et al. (2018) ‘PEGylated Multistimuli-Responsive Dendritic Prodrug-Based Nanoscale System for Enhanced Anticancer Activity’, ACS Applied Materials & Interfaces, 10(42), pp. 35770–35783. Available at:. [CrossRef]
- Fan, J. et al. (2016) ‘Light-Responsive Biodegradable Nanomedicine Overcomes Multidrug Resistance via NO-Enhanced Chemosensitization’, ACS Applied Materials and Interfaces, 8(22), pp. 13804–13811. Available at:. [CrossRef]
- Ferlay, J.; E.M.; L.F.; C.M.; M.L.; P.M.; Z.A.; S.I.; B.F. (2020) Global Cancer Observatory: Cancer Today, IARC.
- Gai, M. et al. (2017) ‘Polylactic acid nano- and microchamber arrays for encapsulation of small hydrophilic molecules featuring drug release via high intensity focused ultrasound’, Nanoscale, 9(21), pp. 7063–7070. Available at:. [CrossRef]
- Grewal, I.K. et al. (2021) ‘Polymeric nanoparticles for breast cancer therapy: A comprehensive review’, Biointerface Research in Applied Chemistry. AMG Transcend Association, pp. 11151–11171. Available at:. [CrossRef]
- Guida, F. et al. (2022) ‘Global and regional estimates of orphans attributed to maternal cancer mortality in 2020’, Nature Medicine, 28(12), pp. 2563–2572. Available at:. [CrossRef]
- Guisasola E., B.A., T.M., A.D., M.M., de la F.J. s. M., V.-R.M. (2015) ‘ Magnetic-Responsive Release Controlled by Hot Spot Effect’, Langmuir, 31(46), pp. 12777–12782.
- Hardy, J.G. et al. (2016) ‘Hydrogel-Forming Microneedle Arrays Made from Light-Responsive Materials for On-Demand Transdermal Drug Delivery’, Molecular Pharmaceutics, 13(3), pp. 907–914. Available at:. [CrossRef]
- Hervault, A. et al. (2016) ‘Doxorubicin loaded dual pH- and thermo-responsive magnetic nanocarrier for combined magnetic hyperthermia and targeted controlled drug delivery applications’, Nanoscale, 8(24), pp. 12152–12161. Available at:. [CrossRef]
- Van Hove, A.H., G. Beltejar, M.J. and Benoit, D.S.W. (2014) ‘Development and invitro assessment of enzymatically-responsive poly(ethylene glycol) hydrogels for the delivery of therapeutic peptides’, Biomaterials, 35(36), pp. 9719–9730. Available at:. [CrossRef]
- Huang, J. et al. (2015) ‘Layer-by-layer assembled milk protein coated magnetic nanoparticle enabled oral drug delivery with high stability in stomach and enzyme-responsive release in small intestine’, Biomaterials, 39, pp. 105–113. Available at:. [CrossRef]
- Huebsch, N. et al. (2014) ‘Ultrasound-triggered disruption and self-healing of reversibly cross-linked hydrogels for drug delivery and enhanced chemotherapy’, Proceedings of the National Academy of Sciences of the United States of America, 111(27), pp. 9762–9767. Available at:. [CrossRef]
- Hussain, M. et al. (2020) ‘Ultra-high-molecular-weight-polyethylene (UHMWPE) as a promising polymer material for biomedical applications: A concise review’, Polymers. MDPI AG. Available at:. [CrossRef]
- J. Zhang, Z.-F.Y.Y.W.W.-H.C.G.-F.L.S.-X.C.R.-X.Z. and X.-Z.Z.J.A. (2013) ‘ Multifunctional Envelope-Type Mesoporous Silica Nanoparticles for Tumor-Triggered Targeting Drug Delivery Jing’, American Chemical society , 135(13), pp. 5068–5037.
- Kashyap, S. et al. (2016) ‘Enzyme and Thermal Dual Responsive Amphiphilic Polymer Core–Shell Nanoparticle for Doxorubicin Delivery to Cancer Cells’, Biomacromolecules, 17(1), pp. 384–398. Available at:. [CrossRef]
- Kearney, C.J. et al. (2015) ‘Switchable Release of Entrapped Nanoparticles from Alginate Hydrogels’, Advanced Healthcare Materials, 4(11), pp. 1634–1639. Available at:. [CrossRef]
- Kocak, G., Tuncer, C. and Bütün, V. (2017) ‘PH-Responsive polymers’, Polymer Chemistry. Royal Society of Chemistry, pp. 144–176. Available at:. [CrossRef]
- Krukiewicz K., Z.P., H.A.P., T.R., B.S., Z.J.K. (2016) ‘An electrically controlled drug delivery system based on conducting poly(3,4-ethylenedioxypyrrole) matrix’, Bioelectrochemistry, 108, pp. 13–20.
- Lee H., H.W., J.S., C.Y., C.Y. (2015) ‘Electroactive Polypyrrole Nanowire Arrays: Synergistic Effect of Cancer Treatment by On-Demand Drug Release and Photothermal Therapy’, Langmuir, 31(14), pp. 4264–4269.
- Lee, J. et al. (2018) ‘Glucose-Responsive Trehalose Hydrogel for Insulin Stabilization and Delivery’, Macromolecular Bioscience, 18(5). Available at:. [CrossRef]
- Li, H. et al. (2018) ‘Dual temperature and pH responsive nanofiber formulations prepared by electrospinning’, Colloids and Surfaces B: Biointerfaces, 171, pp. 142–149. Available at:. [CrossRef]
- Li S., H.K., C.W., S.Y., S.W., L.F., W.Y., L.X.J. (2014) ‘pH-responsive biocompatible fluorescent polymer nanoparticles based on phenylboronic acid for intracellular imaging and drug delivery’, Nanoscale, (22), pp. 13701–13709.
- Liang Y., G.W., P.X., D.X., S.C., W.H., H.B. (2016) ‘Near infrared light responsive hybrid nanoparticles for synergistic therapy’, Biomaterials, 100, pp. 76–90.
- Liu, Y. et al. (2025) ‘Amino acid metabolism in breast cancer: pathogenic drivers and therapeutic opportunities’, Protein and Cell. Oxford University Press, pp. 506–531. Available at:. [CrossRef]
- Maier-Hauff K., R.R., S.R., G.U., W.P., T.B., F.A., von D.A., W.N., F.R. (2007) ‘Intracranial Thermotherapy using Magnetic Nanoparticles Combined with External Beam Radiotherapy: Results of a Feasibility Study on Patients with Glioblastoma Multiforme’, Journal of Neuro-Oncology , 81, pp. 53–60.
- Mohapatra A., H.M.A., L.D., G.M., J.J.A., M.B.I., H.W.O., B.J.D., M.S.R., F.T. (2018) ‘Magnetic stimulus responsive vancomycin drug delivery system based on chitosan microbeads embedded with magnetic nanoparticles’, J Biomed Mater Res B Appl Biomater, 106(6), pp. 2169–2176.
- Mongkolkitikul S., P.N., S.A. (2018) ‘Electrically controlled release of ibuprofen from conductive poly(3-methoxydiphenylamine)/crosslinked pectin hydrogel’, European Journal of Pharmaceutical Sciences, 112, pp. 20–27.
- N. C. Paxton, M.C.A.P.M.L. and M.A.W. (2019) ‘Biomedical applications of polyethylene’, European Polymer Journal, 118, pp. 412–428.
- rtiz de Solorzano, I. et al. (2019) ‘Cleavable and thermo-responsive hybrid nanoparticles for on-demand drug delivery’, Journal of Colloid and Interface Science, 533, pp. 171–181. Available at:. [CrossRef]
- Pafiti, K. et al. (2016) ‘Hydrogel Composites Containing Sacrificial Collapsed Hollow Particles as Dual Action pH-Responsive Biomaterials’, Biomacromolecules, 17(7), pp. 2448–2458. [CrossRef]
- Park, M. et al. (2022) ‘Breast Cancer Metastasis: Mechanisms and Therapeutic Implications’, International Journal of Molecular Sciences. MDPI. Available at:. [CrossRef]
- Pearson S., V.D., K.Y.Y., D.A., L.H., S.M., B.L. (2015) ‘Light-responsive azobenzene-based glycopolymer micelles for targeted drug delivery to melanoma cells’, European Polymer Journal, 69, pp. 616–627.
- Qi, X. et al. (2015) ‘Hyaluronic acid-grafted polyamidoamine dendrimers enable long circulation and active tumor targeting simultaneously’, Carbohydrate Polymers, 126, pp. 231–239. Available at:. [CrossRef]
- Qu J., Z.X., M.P.X., G.B. (2017) ‘pH-responsive self-healing injectable hydrogel based on N-carboxyethyl chitosan for hepatocellular carcinoma therapy’, Acta Biomater., 58, pp. 168–180.
- Raj, S. et al. (2021) ‘Specific targeting cancer cells with nanoparticles and drug delivery in cancer therapy’, Seminars in Cancer Biology. Academic Press, pp. 166–177. Available at:. [CrossRef]
- Riedinger A., G.P., C.A., G.M.A., C.R., M.L., P.T. (2013) ‘Subnanometer Local Temperature Probing and Remotely Controlled Drug Release Based on Azo-Functionalized Iron Oxide Nanoparticles’, Nano Letters, 13(6), pp. 2399–2406.
- Salari, N. et al. (2021) ‘Hyaluronic acid-based drug nanocarriers as a novel drug delivery system for cancer chemotherapy: A systematic review’, DARU, Journal of Pharmaceutical Sciences. Springer Science and Business Media Deutschland GmbH, pp. 439–447. Available at:. [CrossRef]
- Salgarella, A.R. et al. (2018) ‘Investigation of drug release modulation from poly(2-oxazoline) micelles through ultrasound’, Scientific Reports, 8(1). Available at:. [CrossRef]
- Sharma, R. (2021) ‘Global, regional, national burden of breast cancer in 185 countries: Evidence from GLOBOCAN 2018’, Breast Cancer Research and Treatment, 187, pp. 557–567.
- Son S., S.E., K.B.-S. (2014) ‘Light-Responsive Micelles of Spiropyran Initiated Hyperbranched Polyglycerol for Smart Drug Delivery’, Biomacromolecules, 15(2), pp. 628–634.
- Thirunavukkarasu G.K., C.K., L.H., J.Y.Y., P.I.-K., L.J.Y. (2018) ‘Magnetic field-inducible drug-eluting nanoparticles for image-guided thermo-chemotherapy’, Biomaterials, 180, pp. 240–252.
- Tian Y., Z.J., T.X., R.Q., W.Y., Y.W. (2015) ‘Near-Infrared Light-Responsive Nanogels with Diselenide-Cross-Linkers for On-Demand Degradation and Triggered Drug Release’, Particle & Particle Systems Characterization, 32(5), pp. 547–551.
- Trenker, R. et al. (2022) ‘An effective strategy for ligand-mediated pulldown of the HER2/HER3/NRG1β heterocomplex and cryo-EM structure determination at low sample concentrations’, in Methods in Enzymology. Academic Press Inc., pp. 633–662. Available at:. [CrossRef]
- U. Ali, K.J.B.A.K. and N.A.B. (2015) ‘A Review of the Properties and Applications of Poly (Methyl Methacrylate) (PMMA)’, Polym. Rev., 55(4), pp. 678–705.
- Wang B., L.H., S.L., J.Y., D.X., L.L., J.J., C.H. (2017) ‘Construction of High Drug Loading and Enzymatic Degradable Multilayer Films for Self-Defense Drug Release and Long-Term Biofilm Inhibition’, Biomacromolecules, 19(1).
- Wang, D. et al. (2015) ‘Supramolecular hydrogels constructed by red-light-responsive host-guest interactions for photo-controlled protein release in deep tissue’, Soft Matter, 11(38), pp. 7656–7662. Available at:. [CrossRef]
- Wang T., S.G., W.M., Z.B., F.J. (2015) ‘Voltage/pH-Driven Mechanized Silica Nanoparticles for the Multimodal Controlled Release of Drugs’, ACS Applied Materials & Interfaces, 7(38), pp. 21295–21304.
- Wang, X. et al. (2016) ‘Multi-responsive photothermal-chemotherapy with drug-loaded melanin-like nanoparticles for synergetic tumor ablation’, Biomaterials, 81, pp. 114–124. Available at:. [CrossRef]
- Wang Z., C.Z., L.Z., S.P., D.K., J.E., R.J., Q.X.A. (2014) ‘A multi-stimuli responsive gold nanocage–hyaluronic platform for targeted photothermal and chemotherapy’, Biomaterials, 35(36), pp. 9678–9688.
- Wells, C.M. et al. (2019) ‘Stimuli-responsive drug release from smart polymers’, Journal of Functional Biomaterials. MDPI AG. Available at:. [CrossRef]
- Wendels, S. and Avérous, L. (2021) ‘Biobased polyurethanes for biomedical applications’, Bioactive Materials. KeAi Communications Co., pp. 1083–1106. Available at:. [CrossRef]
- World Health Organization (2018) Global Health Estimates 2016: Disease Burden by Cause, Age, Sex, by Country and by Region, 2000–2016. Geneva, Switzerland.
- Wu, G. et al. (2014) ‘Folate-modified doxorubicin-loaded nanoparticles for tumor-targeted therapy’, Pharmaceutical Biology, 52(8), pp. 978–982. Available at:. [CrossRef]
- Wu, P.C. et al. (2018) ‘Magnetic field distribution modulation of intrathecal delivered ketorolac iron-oxide nanoparticle conjugates produce excellent analgesia for chronic inflammatory pain’, Journal of Nanobiotechnology, 16(1). Available at:. [CrossRef]
- Yang, B. et al. (2017) ‘Honokiol-loaded polymeric nanoparticles: An active targeting drug delivery system for the treatment of nasopharyngeal carcinoma’, Drug Delivery, 24(1), pp. 660–669. Available at:. [CrossRef]
- Yang P., L.D., J.S., D.J., G.J., S.W., W.C. (2014) ‘Stimuli-responsive biodegradable poly(methacrylic acid) based nanocapsules for ultrasound traced and triggered drug delivery system’, Biomaterials, 35(6), pp. 2079–2088.
- Yuan, H. et al. (2024) ‘Recent advances in poly(amino acids), polypeptides, and their derivatives in drug delivery’, Nanoscale. Royal Society of Chemistry, pp. 3549–3584. Available at:. [CrossRef]
- Zare, M. et al. (2021) ‘Silicone-based biomaterials for biomedical applications: Antimicrobial strategies and 3D printing technologies’, Journal of Applied Polymer Science. John Wiley and Sons Inc. Available at:. [CrossRef]
- Zhang C., P.D., L.J., H.J., B.A., G.N., Z.H., L.X., L.K., G.Q. (2017) ‘Enzyme-responsive peptide dendrimer-gemcitabine conjugate as a controlled-release drug delivery vehicle with enhanced antitumor efficacy’, Acta Biomaterialia, 55, pp. 153–162.
- Zhou, X. et al. (2019) ‘Biocompatible Chitosan Nanobubbles for Ultrasound-Mediated Targeted Delivery of Doxorubicin’, Nanoscale Research Letters, 14. Available at:. [CrossRef]
- Zhuang Y, D.H.S.Y.H.L.W.R.T.G. et al (2016) ‘Aptamer-Functionalized and Backbone Redox-Responsive Hyperbranched Polymer for Targeted Drug Delivery in Cancer Therapy’, Biomacromol. , 17(7), pp. 2050–2062.

| Classification | Polymer |
| Natural Polymers | |
| Protein-based polymers | Collagen, albumin, gelatin |
| Polysaccharides | Agarose, alginate, carrageenan, hyaluronic acid, dextran, chitosan, cyclodextrins |
| Synthetic polymers | |
| Biodegradable | |
| Polyesters | Poly (lactic acid), poly (glycolic acid), poly (hydroxy butyrate), poly(ε-caprolactone), poly (β-malic acid), poly(dioxanone) |
| Polyanhydrides | Poly (sebacic acid), poly (adipic acid), poly (terphthalic acid) and various copolymers |
| Polyamides | Poly (imino carbonates), polyomino acids |
| Phosphorus-based polymers | Polyphosphates, polyphosphonates, polyphosphazenes |
| Others | Poly (cyano acrylates), polyurethanes, polyortho esters, polyhydroxyproprans, polyacetals |
| Non-biodegradable polymers | |
| Acrylic polymers | Polymethacrylates, poly (methyl methacrylate), poly hydro (ethyl methacrylate) |
| Cellulose derivatives | Carboxymethyl cellulose, ethyl cellulose, cellulose acetate, cellulose acetate propionate, hydroxyl propyl methyl cellulose |
| Silicones | Polydimethyl siloxane, colloidal silica |
| Others | Polyvinyl pyrrolidone, ethyl vinyl acetate, poloxamers, poloxamines |
| Polymer | Characteristics | Advantages | Disadvantages | Ref. |
| Polytetrafluoroethylene (PTFE) | Chemical inertness; low friction coefficient; non-adhesive properties; thermal stability; flexible and durable | Resistant to chemicals; low friction; flexible and durable | Poor tissue integration; susceptible to deformation; limited mechanical strength; biofilm risk; difficult to process; expensive | (Cardoso et al., 2018a) |
| Polyethylene (PE) | Durability and mechanical strength; chemical inertness; low friction; cost-effectiveness | Durable; chemically inert; cost-effective | Non-biodegradable; poor tissue integration; wear debris; oxidative degradation | (N. C. Paxton, 2019; Hussain et al., 2020) |
| Silicone | Durability and stability; flexibility and elasticity; low immune response; non-degradable; wide range of medical applications | Stable; flexible; low immune response; versatile applications | Lack of biodegradability; poor tissue integration; potential foreign body reaction; limited bioactivity; rigid manufacturing | (Zare et al., 2021) |
| Polyurethane (PU) | Mechanical properties; biostability; versatility in formulation; hemocompatibility; cost and availability | Good mechanical properties; biostable; versatile | Non-biodegradable; possible degradation products; limited biocompatibility; requires surface modification | (Wendels and Avérous, 2021) |
| Polymethyl methacrylate (PMMA) | Mechanical strength and durability; stability and long-term use; non-immunogenic; ease of shaping and processing | Strong; durable; stable; easy to process | Not biodegradable; potential for biofilm formation; exothermic polymerization; brittle | (U. Ali, 2015) |
| Stimuli | Polymer | Major Result(s) | Ref(s) |
| pH | N-carboxyethyl chitosan/dibezaldehyde-terminated poly (ethylene glycol) | pH changes promote chemical and physical modifications that swell the system inducing cargo release | ( Qu et al). |
| pH | Poly (lactic acid)-poly(ethyleneimine) | Burst release of doxorubicin (DOX) as pH shifted from 7.4 to 5.4 | ( Li et al). |
| pH | Poly (lactic-co-glycolic acid) (PLGA) | Morphological change induces drug release | Chung et al), |
| Ph | Poly(acrylamide) | Drug release at pH > 4.0 | (Pafiti et al., 2016) |
| Ultrasound | Poly (ethylene glycol) | Led to a six-fold increase in the cumulative release | (Kearney et al., 2015) |
| Ultrasound | Alginate | Pulsed stimulation outperformed constant stimulation | (Huebsch et al., 2014) |
| Ultrasound | Chitosan | Significant release compared to no stimulus | (Zhou et al., 2019) |
| Ultrasound | Poly (methacrylic acid) (PMAA) | Design a three in one theranostic nanoplatform for imaging and release | ( Yang P., 2014) |
| Ultrasound | Poly(2-oxazoline) micelles | Possible carrier with increased release | (Salgarella et al., 2018) |
| Ultrasound | polylactic acid (PLA) | Long-term encapsulation of small hydrophilic molecules and four times the release profile with HIFU | (Gai et al., 2017) |
| UV | Spiropyran-hyperbranched polyglycerol micelle | Assembly and disassembly of micelle induced by UV light exposure controls the drug release. Superior biocompatibility with cells in the absence of UV | (Son S., 2014) |
| UV | Azobenzene-β-galactose micelle | Short UV exposure (2 min) to release drug; low cytotoxicity of unloaded micelles | (Pearson S., 2015) |
| UV | 2-hydroxyethyl methacrylate and ethylene glycol dimethacrylate | Deliver multiple doses of drug upon UV exposure over a prolonged period of time (≤160 h) | (Hardy et al., 2016) |
| UV | mPEG-PLGA nanoparticle | Reverse multidrug resistance of tumor cells; enhance chemosensitization of cells to DOX therapy | (Fan et al., 2016) |
| NIR | Diselenide-cross-linked poly (methacrylic acid) | Controlled illumination with specific number of irradiation times allowed for on-demand controlled drug release and nanogel degradation. Rapid internalization by HeLa cell and cytotoxic under NIR irradiation | (Tian Y., 2015) |
| NIR | Β-cylcodextrin | Anticancer activity in vitro and in vivo against breast cancer, with accelerated drug release upon NIR exposure | (Liang Y., 2016) |
| NIR | Polycaprolactone | On-demand, stepwise drug-release after multiple cycles of NIR exposure with low off-state leakage | (Chen M.C., 2015) |
| Red light | Tetra-ortho-methoxy-substituted azobenzene & β-cyclodextrin | Responsive to red light instead of UV. Deeper tissue penetration depth | (Wang et al., 2015) |
| AMF | Aminosilan-type shell | EMF stimulation of SPIONS can maintain elevated temperatures of approximately 45 °C in glioblastoma multiforme tumors | Maier-Hauff K., 2007) |
| AMF | Polyethylene glycol w/azo drug linker | SPION local temperature can increase up to 50 °C without inducing significant temperature increases in media at sufficiently low concentrations | (Riedinger A., 2013) |
| AMF | (N-isopropylacrylamide)-(N-hydroxymethyl) acrylamide | SPION stimulation can trigger PNIPAM critical temperature transition without increasing temperature of surrounding media | (Guisasola E., 2015) |
| AMF | Poly (maleic anhydride-alt-1-octadecene) | Distance from the nanoparticle surface can be used to control temperature dependent effects during AMF stimulation | (Dias et al., no date) |
| AMF | PLGA | SPION stimulation induced drug release by increasing temperature above the glass transition of PLGA | (Thirunavukkarasu G.K., 2018) |
| Permanent magnet | Tetramethylazanium hydroxide | Intrathecally delivered SPIONS loaded with NSAIDS produced magnetic field dependent reductions in pain and inflammatory markers in a murine model | (Wu et al., 2018) |
| Permanent magnet | Polyethyleneimine | External magnetic guidance improved accumulation of SPIONS in arthritic joints in a rat model |
(Duan J., 2014) |
| AMF | Chitosan-polyethylene glycol | SPION loaded microbeads can respond to multiple stimuli and increase drug release to efficacious levels as the carrier nears exhaustion | (Mohapatra A., 2018) |
| Electric | Agarose/alginate-aniline tetramer | Conductive tetramers improve hydrogel biocompatability with neural cells and enables repeat stimuli responsive drug release | (Atoufi Z., 2017) |
| Electric | Poly(3,4-ethylenedioxypyrrole) | Stimulation induces rapid release of ionically bound ibuprofen but not ibuprofen physically entrapped in the matrix during electrochemical polymerization | (Krukiewicz K., 2016) |
| Electric | Poly(3-methoxydiphenylamine)/Pectin blend | Stimulation increased hydrogel mesh pore size allowing increased drug elution | (Mongkolkitikul S., 2018) |
| Electric | Polypyrrole | Sacrificial templates can be used to create electrically responsive nanowires | (Lee H., 2015) |
| Electric | Monoferrocene functionalized β-cyclodextrin | Stimulus-induced conformational changes can be used to control polymeric ‘gates’ for on/off delivery using mesoporous particles | (Wang T., 2015) |
| Enzyme | PEGylated alkynylated peptide dendrimer | Minimal release in the absence of Cathepsin B | (Zhang C., 2017) |
| Enzyme | Polydimethylsiloxane, polyethylenimine | Release in the presence of HAS, E. coli, or S. aureus | Wang et al. (Wang B., 2017) |
| Enzyme | Poly (maleic acid) | No release until exposure to intestine protease trypsin | Huang et al. (Huang et al., 2015) |
| Enzyme | Poly (ethylene glycol) | Peptide cleaving at desired sites | (Van Hove, G. Beltejar and Benoit, 2014) |
| Enzyme | Poly (styrenyl ether trehalose), poly (ethylene glycol) | Ability to withstand elevated temperatures with cargo intact | (Lee et al., 2018) |
| Enzyme, NIR | Poly (vinyl pyrrolidone) | Minimal release in the absence of hyaluronidase, NIR promoting more release | (Wang Z., 2014) |
| Enzyme, pH | Poly (ethylene glycol) | Release rate increase at pH 5.4 in presence of cathepsin B and glutathione | (Duan et al., 2018) |
| Enzyme, Thermal | 3-pentadecylphenol, oligoethylene glycol acrylate | Proposed release at tissue based on temperature with intracellular release concurrent with enzyme exposure | (Kashyap et al., 2016) |
| pH, Thermal | Poly (ethylene glycol) methyl ether methacrylate | pH and temperature greatly influence the release of DOX | (Hervault et al., 2016) |
| pH, Thermal | Poly(N-vinylcaprolactam), ethyl cellulose, Eudagrit L100 | Most pronounced release occurred at 25 °C and pH 7.4 | Li et al. (Li et al., 2018) |
| pH, Thermal | Poly (2-succinyloxyethyl methacrylate)-b-(N-isopropylacrylamide)-b-[(N-4-vinylbenzyl),N,N-diethylamine]], [P(SEMA-b-NIPAAm-b-VEA)] | Greatest DOX release observed at 37 °C and pH 4, increase in temperature led to decrease in DOX release | (Davaran et al., 2017) |
| NIR, Thermal | Poly (ethylene glycol) methyl ether methacrylate, poly (vinyl pyrrolidone) | Release was higher at 45 °C with a burst increase synonymous with NIR irradiation | (Ortiz de Solorzano et al., 2019) |
| NIR, pH, Redox | Poly (ethylene glycol), poly(dopamine) | NIR irradiation release is function of exposure time, pH and redox release greatest at pH 7.4 | (Wang et al., 2016) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




