Submitted:
10 October 2025
Posted:
11 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Western Blotting
2.3. Extraction of Exosomes from Cell Culture Media
3. Results
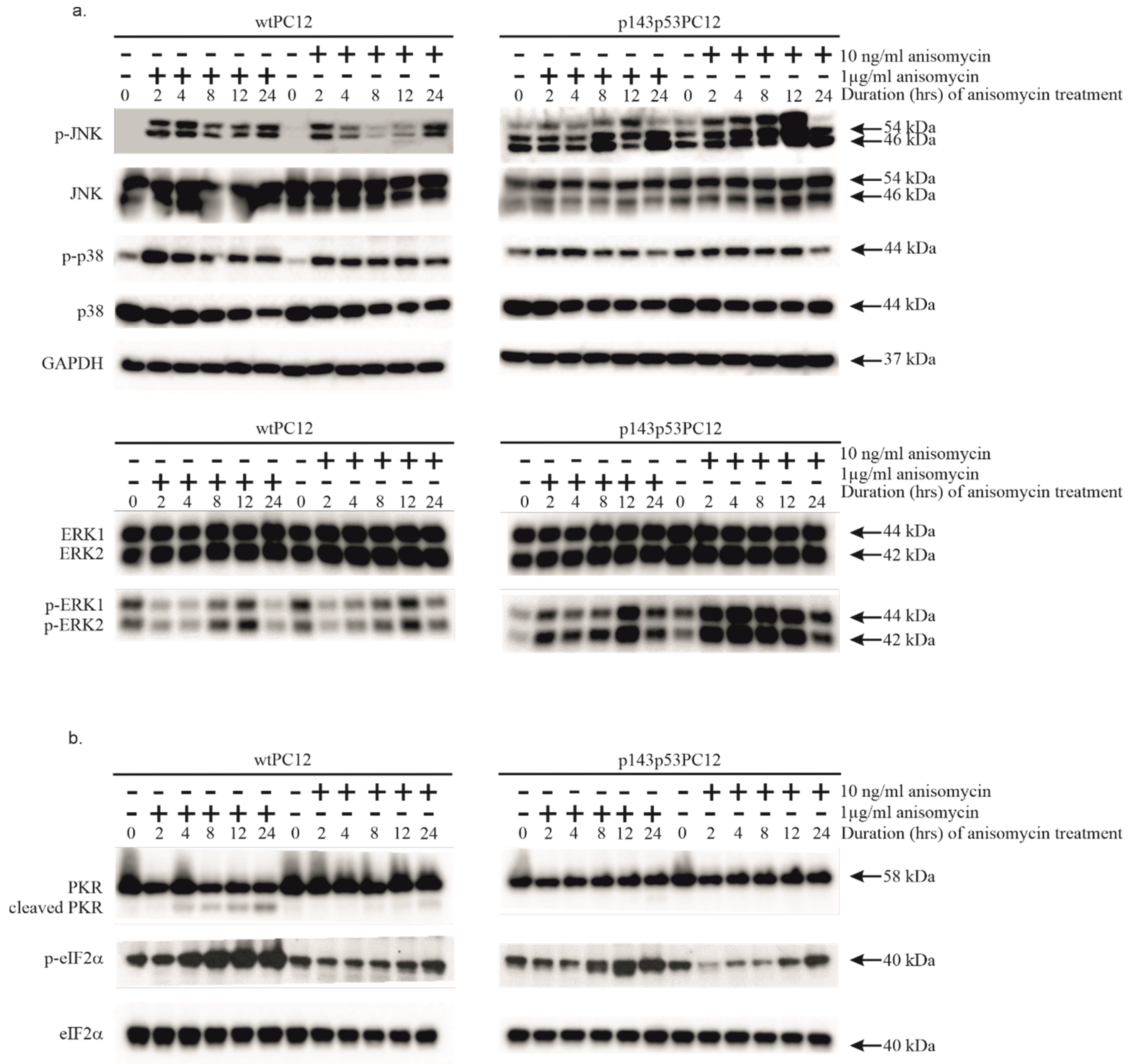
3.1. Both Low and High Concentrations of Anisomycin Causes Activation of Stress Kinases in wtPC12 Cells and PC12 Cells Expressing a Dominant Negative p53 Protein
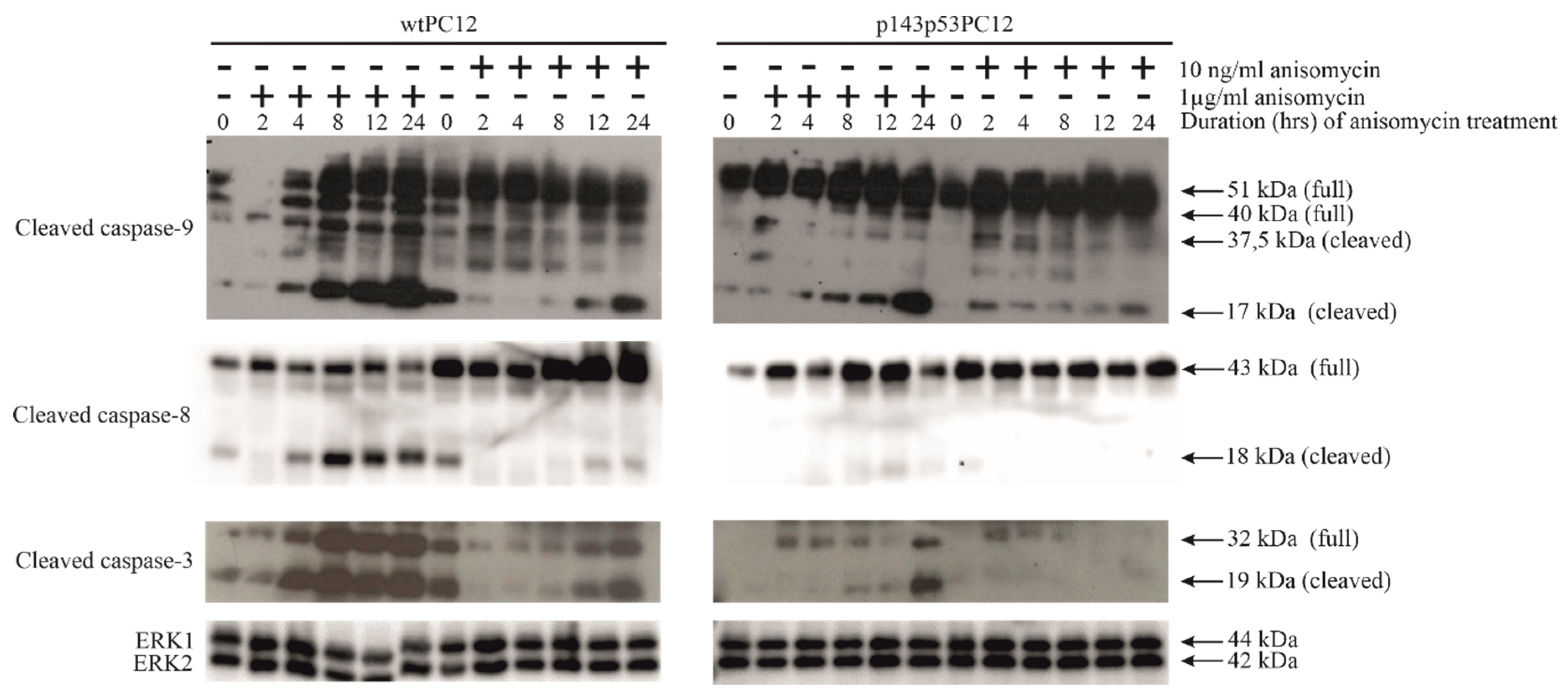
3.2. p53-Dependence of Anisomycin-Induced Caspase Activation
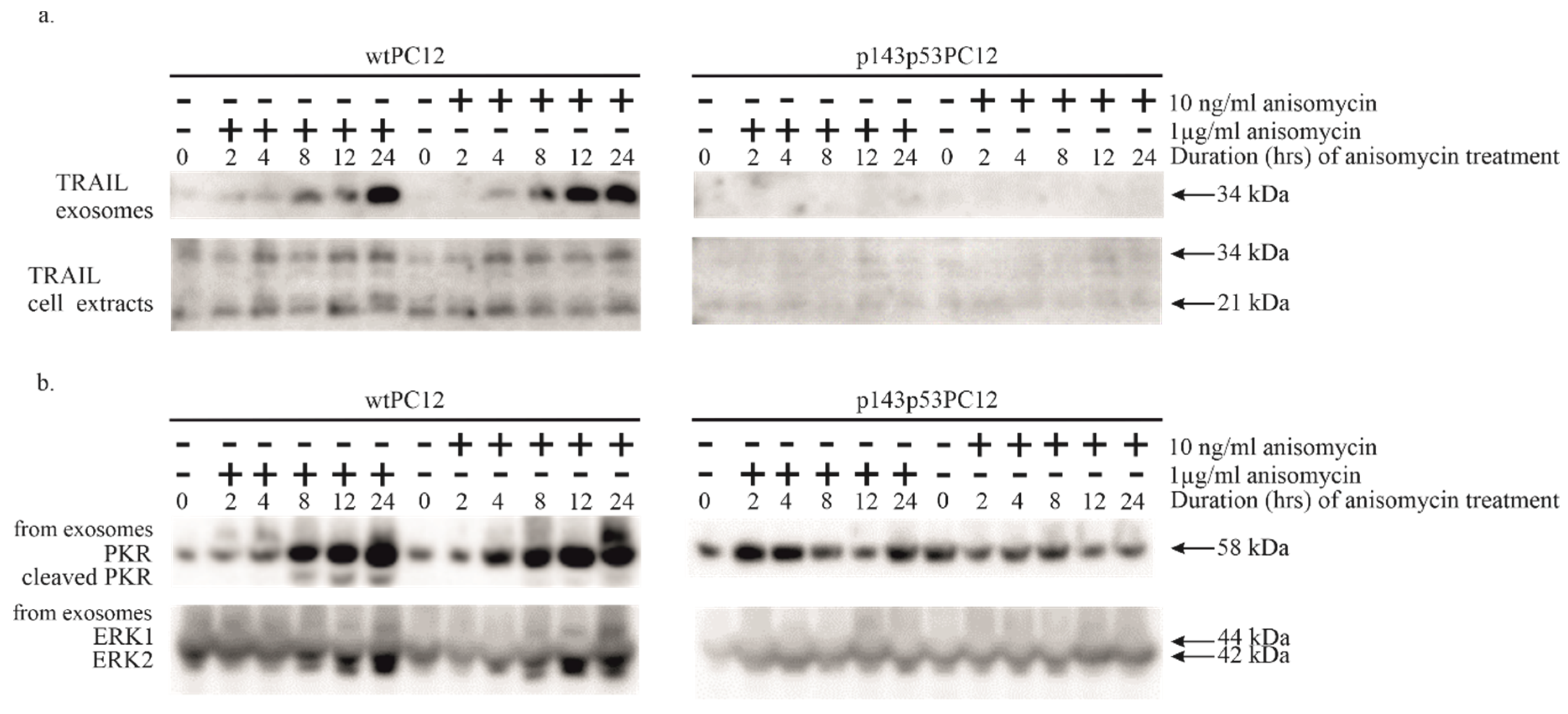
3.3. Potential Involvement of Exosomal Signaling in the Apoptosis of Anisomycin-Treated PC12 Cell Lines
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DR4, DR5 | death receptors 4/5 |
| eIF2α | eukaryotic translation initiation factor 2 α |
| ERK1,2 | extracellular signal regulated kinase 1/2 |
| JNK | c-Jun N-terminal kinase |
| p38 MAPK | p38 mitogen-activated protein kinase |
| p143p53PC12 | PC12 subclone expressing a dominant inhibitory p53 protein |
| PKR | dsRNA-activated protein kinase |
| RSR | ribotoxic stress response |
| TRAIL | tumor necrosis factor-related apoptosis inducing ligand |
| wtPC12 | wild-type PC12, rat pheochromocytoma cell line |
References
- A Iordanov, M.S.; David, P.; Jennifer L., M.; Thanh-Hoai, D.; Jean A., P.; Steven Li-Ye, C.; and Magun, B.E. Ribotoxic Stress Response: Activation of the Stress-Activated Protein Kinase JNK1 by Inhibitors of the Peptidyl Transferase Reaction and by Sequence-Specific RNA Damage to the α-Sarcin/Ricin Loop in the 28S RRNA. Mol Cell Biol 1997, 17, 3373–3381. [Google Scholar] [CrossRef]
- Vázquez, D. Protein Synthesis and Translation Inhibitors. In Inhibitors of Protein Biosynthesis; Springer Berlin Heidelberg: Berlin, Heidelberg, 1979; pp. 1–14. ISBN 978-3-642-81309-2. [Google Scholar]
- Barbacid, M.; Vazquez, D. [3H]Anisomycin Binding to Eukaryotic Ribosomes. J Mol Biol 1974, 84, 603–623. [Google Scholar] [CrossRef]
- Macías-Silva, M.; Vázquez-Victorio, G.; Hernández-Damián, J. Anisomycin Is a Multifunctional Drug: More than Just a Tool to Inhibit Protein Synthesis. Curr Chem Biol 2010, 4, 124–132. [Google Scholar]
- Ouyang, D.-Y.; Wang, Y.; Zheng, Y.T. Activation of C-Jun N-Terminal Kinases by Ribotoxic Stresses. Cell Mol Immunol 2006, 2, 419–425. [Google Scholar]
- Vind, A.C.; Genzor, A.V.; Bekker-Jensen, S. Ribosomal Stress-Surveillance: Three Pathways Is a Magic Number. Nucleic Acids Res 2020, 48, 10648–10661. [Google Scholar] [CrossRef]
- Törocsik B; Szeberényi J Anisomycin Uses Multiple Mechanisms to Stimulate Mitogen-activated Protein Kinases and Gene Expression and to Inhibit Neuronal Differentiation in PC12 Phaeochromocytoma Cells. European Journal of Neuroscience 2000, 12, 527–532. [CrossRef]
- Gal-Ben-Ari, S.; Barrera, I.; Ehrlich, M.; Rosenblum, K. PKR: A Kinase to Remember. Front Mol Neurosci 2019, 11. [Google Scholar] [CrossRef]
- Saelens, X.; Kalai, M.; Vandenabeele, P. Translation Inhibition in Apoptosis: Caspase-Dependent PKR Activation and EIF2-α Phosphorylation. Journal of Biological Chemistry 2001, 276, 41620–41628. [Google Scholar] [CrossRef]
- Kalai, M.; Suin, V.; Festjens, N.; Meeus, A.; Bernis, A.; Wang, X.M.; Saelens, X.; Vandenabeele, P. The Caspase-Generated Fragments of PKR Cooperate to Activate Full-Length PKR and Inhibit Translation. Cell Death Differ 2007, 14, 1050–1059. [Google Scholar] [CrossRef]
- Törőcsik, B.; Szeberényi, J. Anisomycin Affects Both Pro- and Antiapoptotic Mechanisms in PC12 Cells. Biochem Biophys Res Commun 2000, 278, 550–556. [Google Scholar] [CrossRef]
- Hernández Borrero, L.J.; El-Deiry, W.S. Tumor Suppressor P53: Biology, Signaling Pathways, and Therapeutic Targeting. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer 2021, 1876, 188556. [Google Scholar] [CrossRef] [PubMed]
- Lavin, M.F.; Gueven, N. The Complexity of P53 Stabilization and Activation. Cell Death Differ 2006, 13, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.M.; De Queiroz, R.M.; Venkatesh, D.; Prives, C. The Roles and Regulation of MDM2 and MDMX: It Is Not Just about P53. Genes Dev 2021, 35, 575–601. [Google Scholar] [CrossRef] [PubMed]
- Haupt, S.; Berger, M.; Goldberg, Z.; Haupt, Y. Apoptosis - the P53 Network. J Cell Sci 2003, 116, 4077–4085. [Google Scholar] [CrossRef]
- Fridman, J.S.; Lowe, S.W. Control of Apoptosis by P53. Oncogene 2003, 22, 9030–9040. [Google Scholar] [CrossRef]
- Feroz, W.; Sheikh, A.M.A. Exploring the Multiple Roles of Guardian of the Genome: P53. Egyptian Journal of Medical Human Genetics 2020, 21, 49. [Google Scholar] [CrossRef]
- Pavlakis, E.; Neumann, M.; Stiewe, T. Extracellular Vesicles: Messengers of P53 in Tumor–Stroma Communication and Cancer Metastasis. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Yu, X.; Harris, S.L.; Levine, A.J. The Regulation of Exosome Secretion: A Novel Function of the P53 Protein. Cancer Res 2006, 66, 4795–4801. [Google Scholar] [CrossRef]
- Théry, C. Exosomes: Secreted Vesicles and Intercellular Communications. F1000 Biol Rep 3(15). F1000 Biol Rep 2011, 3, 15. [Google Scholar] [CrossRef]
- Dilsiz, N. Hallmarks of Exosomes. Future Sci OA 2022, 8, FSO764. [Google Scholar] [CrossRef]
- Stenqvist, A.-C.; Nagaeva, O.; Baranov, V.; Mincheva-Nilsson, L. Exosomes Secreted by Human Placenta Carry Functional Fas Ligand and TRAIL Molecules and Convey Apoptosis in Activated Immune Cells, Suggesting Exosome-Mediated Immune Privilege of the Fetus. The Journal of Immunology 2013, 191, 5515–5523. [Google Scholar] [CrossRef]
- Pimentel, J.M.; Zhou, J.-Y.; Wu, G.S. The Role of TRAIL in Apoptosis and Immunosurveillance in Cancer. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Gonzalvez, F.; Ashkenazi, A. New Insights into Apoptosis Signaling by Apo2L/TRAIL. Oncogene 2010, 29, 4752–4765. [Google Scholar] [CrossRef] [PubMed]
- Naval, J.; de Miguel, D.; Gallego-Lleyda, A.; Anel, A.; Martinez-Lostao, L. Importance of TRAIL Molecular Anatomy in Receptor Oligomerization and Signaling. Implications for Cancer Therapy. Cancers (Basel) 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Rivoltini, L.; Chiodoni, C.; Squarcina, P.; Tortoreto, M.; Villa, A.; Vergani, B.; Bürdek, M.; Botti, L.; Arioli, I.; Cova, A.; et al. TNF-Related Apoptosis-Inducing Ligand (TRAIL)–Armed Exosomes Deliver Proapoptotic Signals to Tumor Site. Clinical Cancer Research 2016, 22, 3499–3512. [Google Scholar] [CrossRef]
- Ke, C.; Hou, H.; Li, J.; Su, K.; Huang, C.; Lin, Y.; Lu, Z.; Du, Z.; Tan, W.; Yuan, Z. Extracellular Vesicle Delivery of TRAIL Eradicates Resistant Tumor Growth in Combination with CDK Inhibition by Dinaciclib. Cancers (Basel) 2020, 12. [Google Scholar] [CrossRef]
- Jella, K.K.; Nasti, T.H.; Li, Z.; Malla, S.R.; Buchwald, Z.S.; Khan, M.K. Exosomes, Their Biogenesis and Role in Inter-Cellular Communication, Tumor Microenvironment and Cancer Immunotherapy. Vaccines (Basel) 2018, 6. [Google Scholar] [CrossRef]
- Pap, M.; Szeberényi, J. Differential Ras-Dependence of Gene Induction by Nerve Growth Factor and Second Messenger Analogs in PC12 Cells. Neurochem Res 1998, 23, 969–975. [Google Scholar] [CrossRef]
- Balogh, A.; Bátor, J.; Markó, L.; Németh, M.; Pap, M.; Sétáló, G.; Müller, D.N.; Csatary, L.K.; Szeberényi, J. Gene Expression Profiling in PC12 Cells Infected with an Oncolytic Newcastle Disease Virus Strain. Virus Res 2014, 185, 10–22. [Google Scholar] [CrossRef]
- Teng, K.K.; Angelastro, J.M.; Cunningham, M.E.; Greene, L.A. Chapter 21 - Cultured PC12 Cells: A Model for Neuronal Function, Differentiation, and Survival. In Cell Biology (Third Edition); Celis, J.E., Ed.; Academic Press: Burlington, 2006; pp. 171–176. ISBN 978-0-12-164730-8. [Google Scholar]
- Varga, J.; Bátor, J.; Péter, M.; Árvai, Z.; Pap, M.; Sétáló, G.; Szeberényi, J. The Role of the P53 Protein in Nitrosative Stress-Induced Apoptosis of PC12 Rat Pheochromocytoma Cells. Cell Tissue Res 2014, 358, 65–74. [Google Scholar] [CrossRef]
- Fábián, Z.; Vecsernyés, M.; Pap, M.; Szeberényi, J. The Effects of a Mutant P53 Protein on the Proliferation and Differentiation of PC12 Rat Phaeochromocytoma Cells. J Cell Biochem 2006, 99, 1431–1441. [Google Scholar] [CrossRef]
- Abayasiriwardana, K.S.; Barbone, D.; Kim, K.-U.; Vivo, C.; Lee, K.K.; Dansen, T.B.; Hunt, A.E.; Evan, G.I.; Broaddus, V.C. Malignant Mesothelioma Cells Are Rapidly Sensitized to TRAIL-Induced Apoptosis by Low-Dose Anisomycin via Bim. Mol Cancer Ther 2007, 6, 2766–2776. [Google Scholar] [CrossRef]
- Xia, S.; Li, Y.; Rosen, E.M.; Laterra, J. Ribotoxic Stress Sensitizes Glioblastoma Cells to Death Receptor–Induced Apoptosis: Requirements for c-Jun NH2-Terminal Kinase and Bim. Molecular Cancer Research 2007, 5, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Schipp, R.; Varga, J.; Bátor, J.; Vecsernyés, M.; Árvai, Z.; Pap, M.; Szeberényi, J. Partial P53-Dependence of Anisomycin-Induced Apoptosis in PC12 Cells. Mol Cell Biochem 2017, 434, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Varga, J.; Bátor, J.; Nádasdi, G.; Árvai, Z.; Schipp, R.; Szeberényi, J. Partial Protection of PC12 Cells from Cellular Stress by Low-Dose Sodium Nitroprusside Pre-Treatment. Cell Mol Neurobiol 2016, 36, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Pap, M.; Szeberényi, J. Involvement of Proteolytic Activation of Protein Kinase R in the Apoptosis of PC12 Pheochromocytoma Cells. Cell Mol Neurobiol 2008, 28, 443–456. [Google Scholar] [CrossRef]
- Ding, T.; Zhou, Y.; Long, R.; Chen, C.; Zhao, J.; Cui, P.; Guo, M.; Liang, G.; Xu, L. DUSP8 Phosphatase: Structure, Functions, Expression Regulation and the Role in Human Diseases. Cell Biosci 2019, 9, 70. [Google Scholar] [CrossRef]
- Ha, J.; Kang, E.; Seo, J.; Cho, S. Phosphorylation Dynamics of JNK Signaling: Effects of Dual-Specificity Phosphatases (DUSPs) on the JNK Pathway. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Li, M.; Zhou, J.-Y.; Ge, Y.; Matherly, L.H.; Wu, G.S. The Phosphatase MKP1 Is a Transcriptional Target of P53 Involved in Cell Cycle Regulation *. Journal of Biological Chemistry 2003, 278, 41059–41068. [Google Scholar] [CrossRef]
- Yin, Y.; Liu, Y.-X.; Jin, Y.J.; Hall, E.J.; Barrett, J.C. PAC1 Phosphatase Is a Transcription Target of P53 in Signalling Apoptosis and Growth Suppression. Nature 2003, 422, 527–531. [Google Scholar] [CrossRef]
- Shen, W.H.; Wang, J.; Wu, J.; Zhurkin, V.B.; Yin, Y. Mitogen-Activated Protein Kinase Phosphatase 2: A Novel Transcription Target of P53 in Apoptosis. Cancer Res 2006, 66, 6033–6039. [Google Scholar] [CrossRef]
- Xie, S.; Zhang, Q.; Jiang, L. Current Knowledge on Exosome Biogenesis, Cargo-Sorting Mechanism and Therapeutic Implications. Membranes (Basel) 2022, 12. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



