Submitted:
09 October 2025
Posted:
13 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
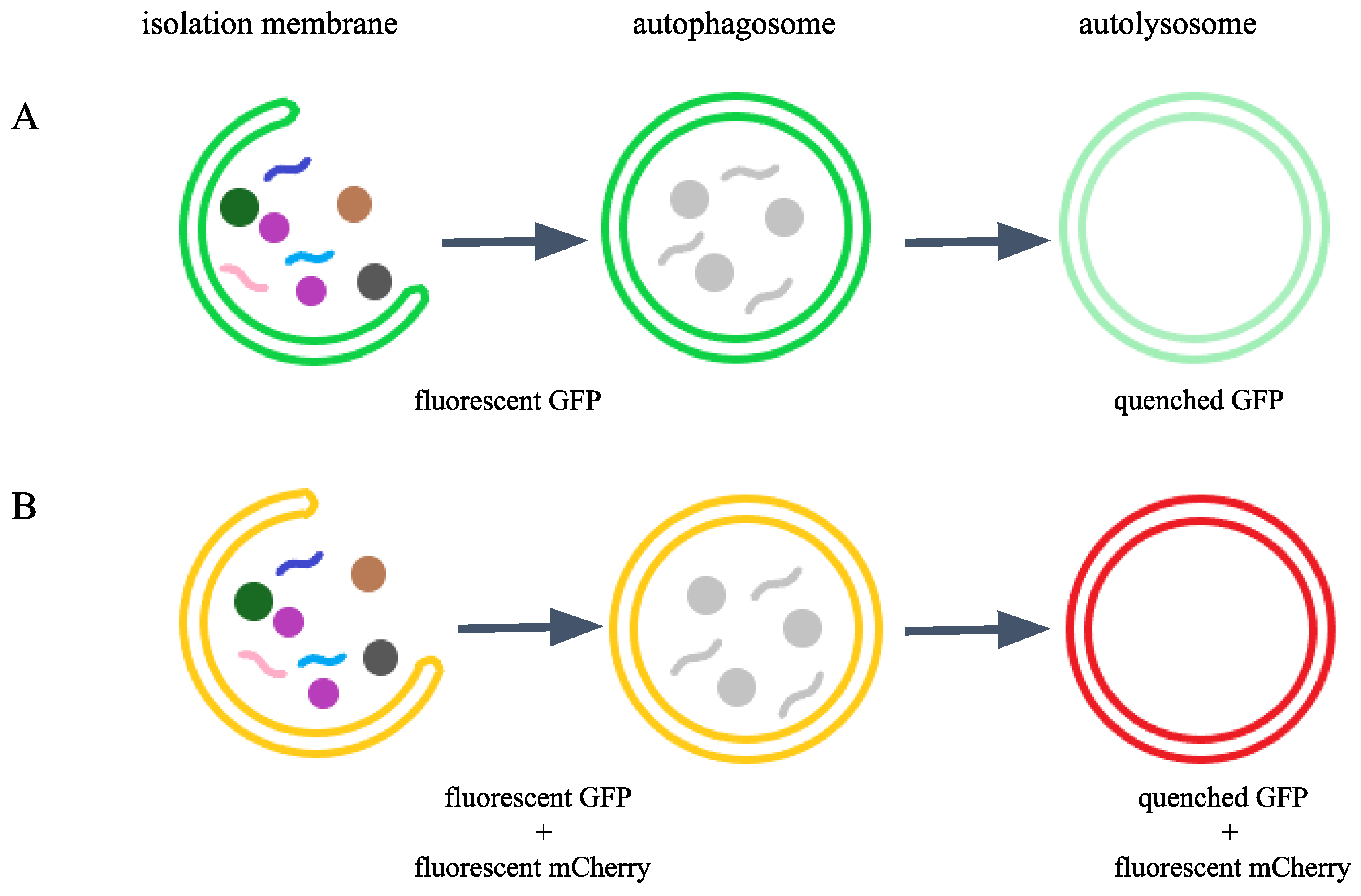
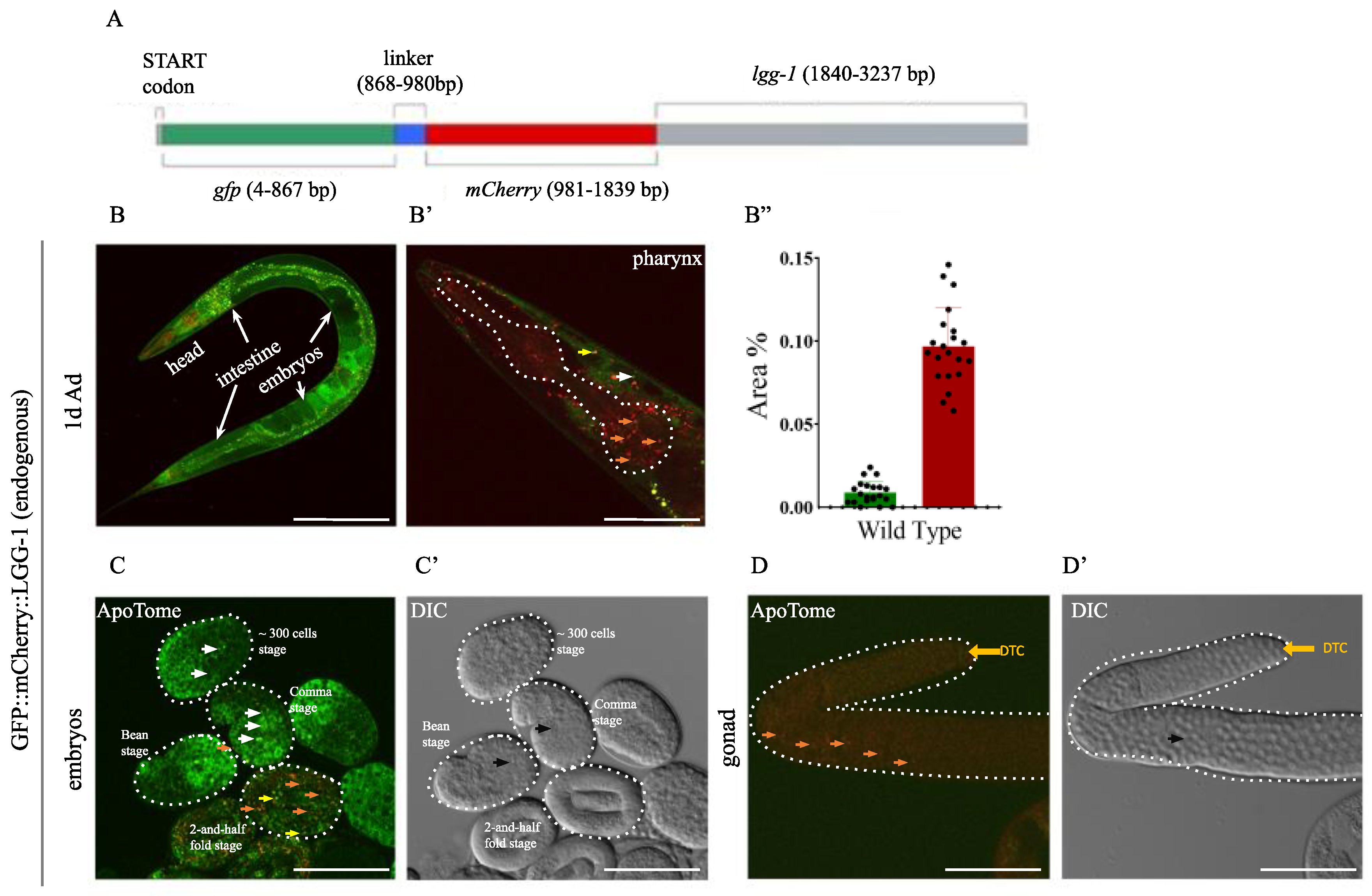
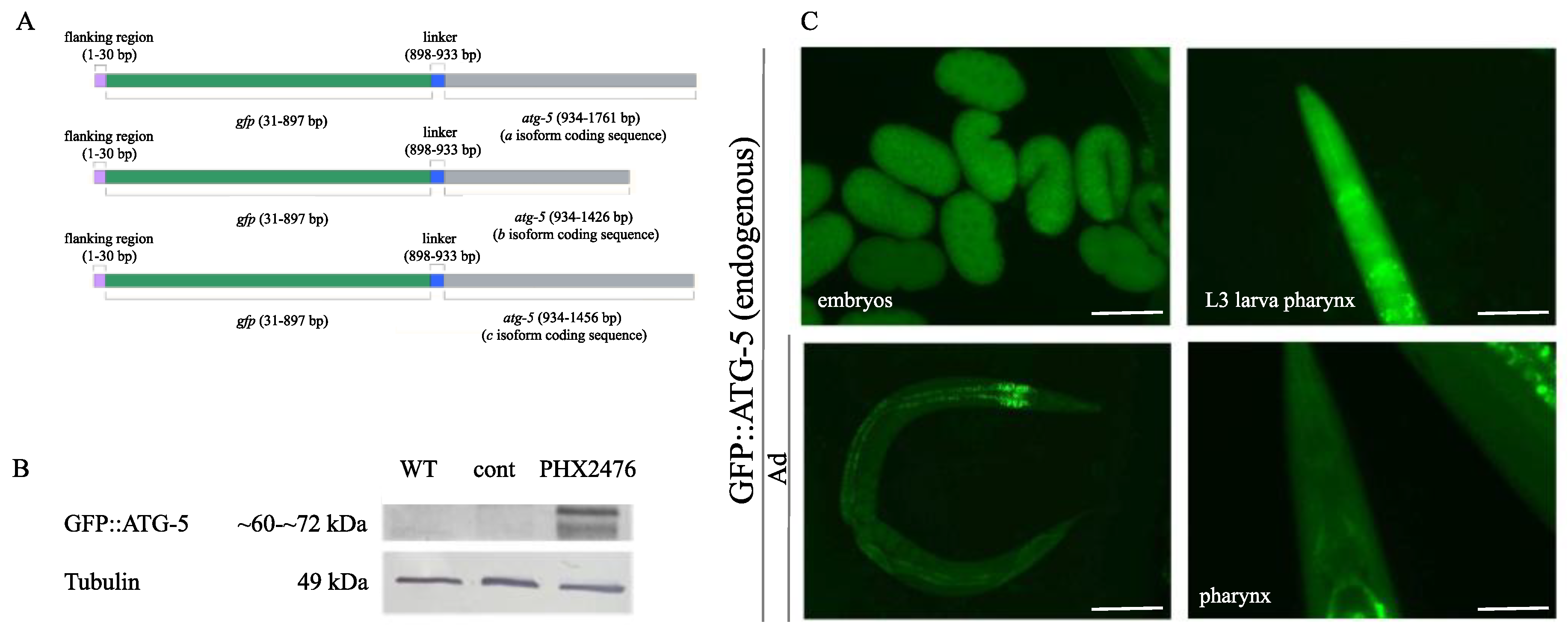
2.1. The Endogenous GFP::mCherry::LGG-1/ATG-8 Reporter is Expressed Ubiquitously in C. elegans Tissues from Early Embryonic Stages Onward
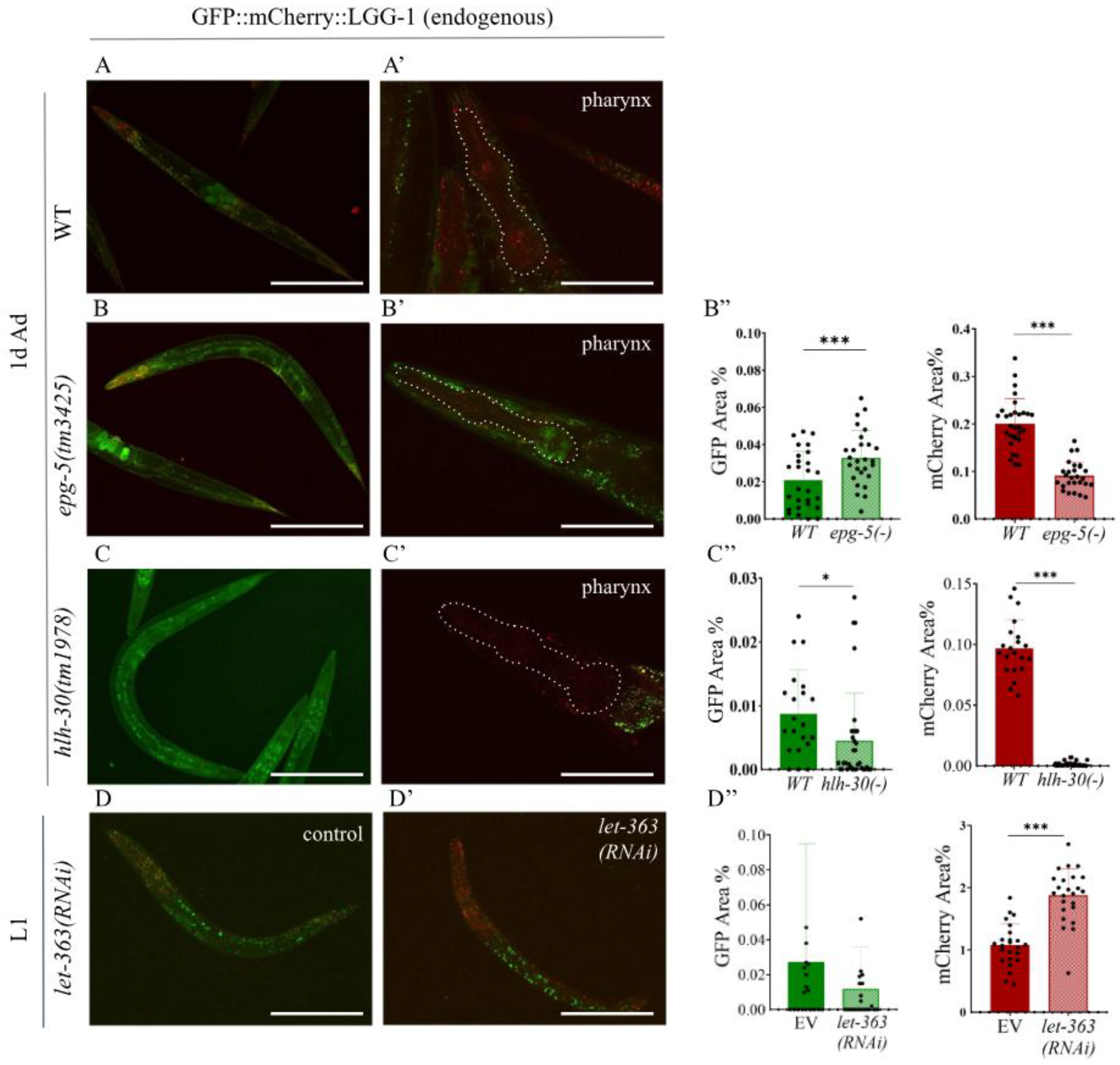
2.2. The Endogenous GFP::mCherry::LGG-1 Reporter Responds to Autophagy-Modulating Stimuli
2.3. Silencing of Let-363/TOR Increases the Number of Autolysosomes in the Endogenous GFP::mCherry::LGG-1 Reporter Strain
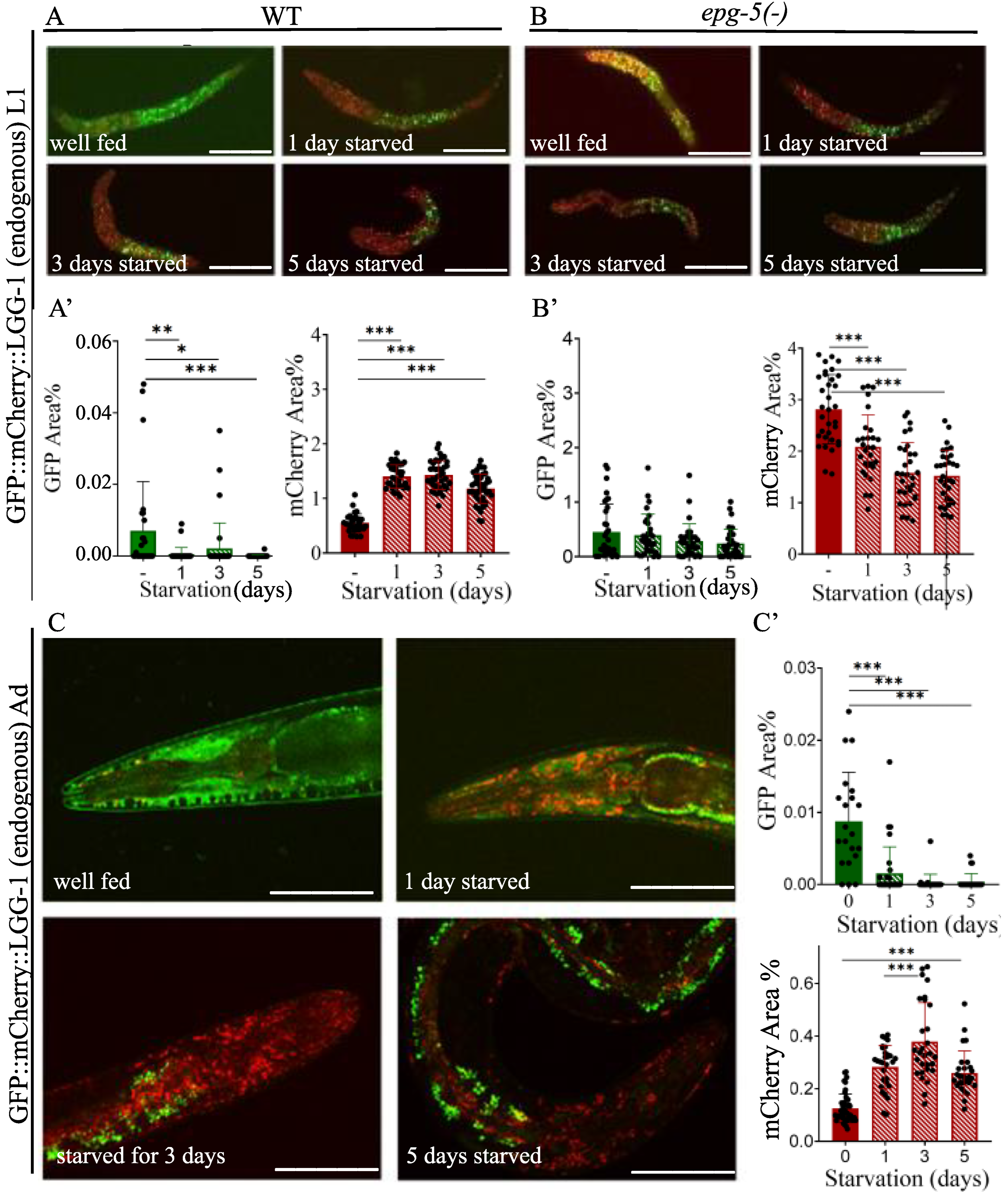
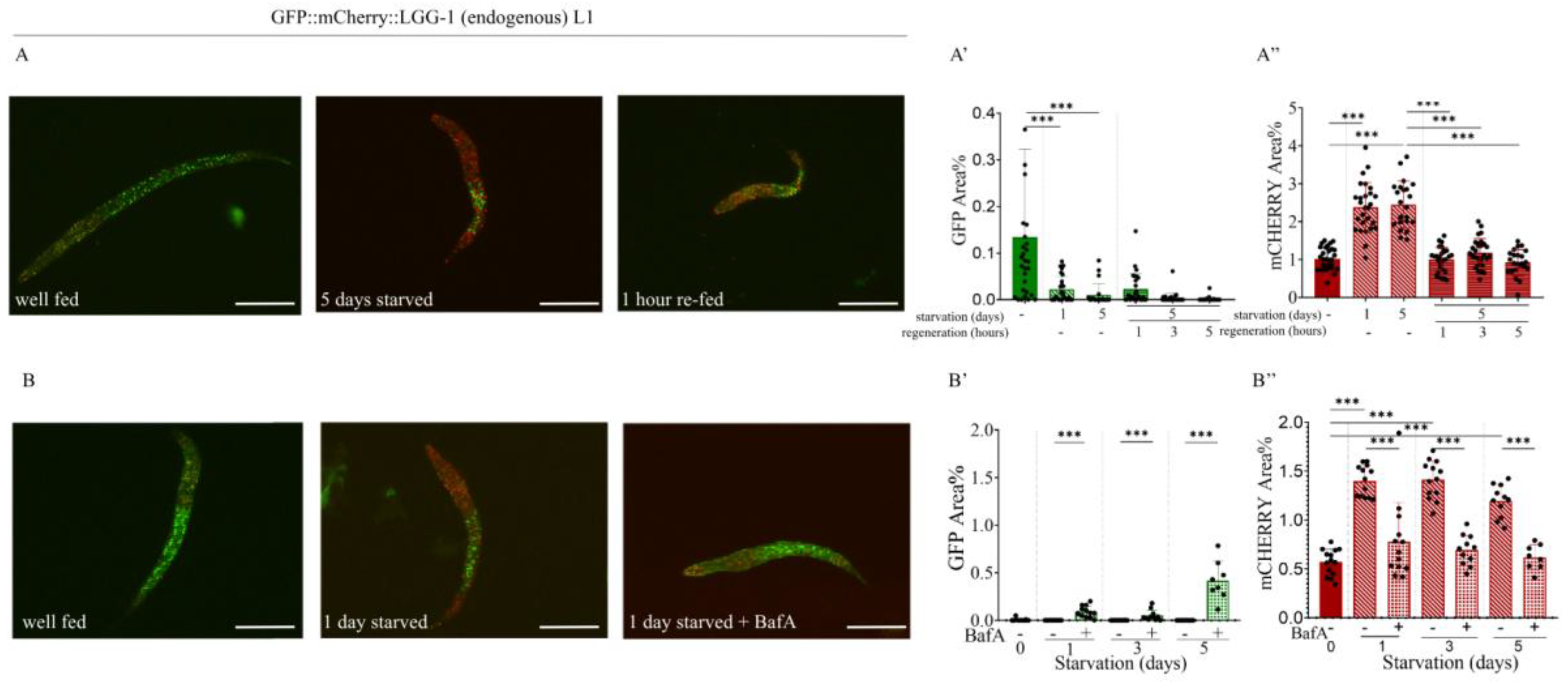
2.4. Starvation Increases the Number of Autolysosomes in L1 larvae and Adult Animals
2.5. Effect of the Autophagy Inhibitor Bafilomycin A1 on the Expression of the Endogenous GFP::mCherry::LGG-1 Reporter
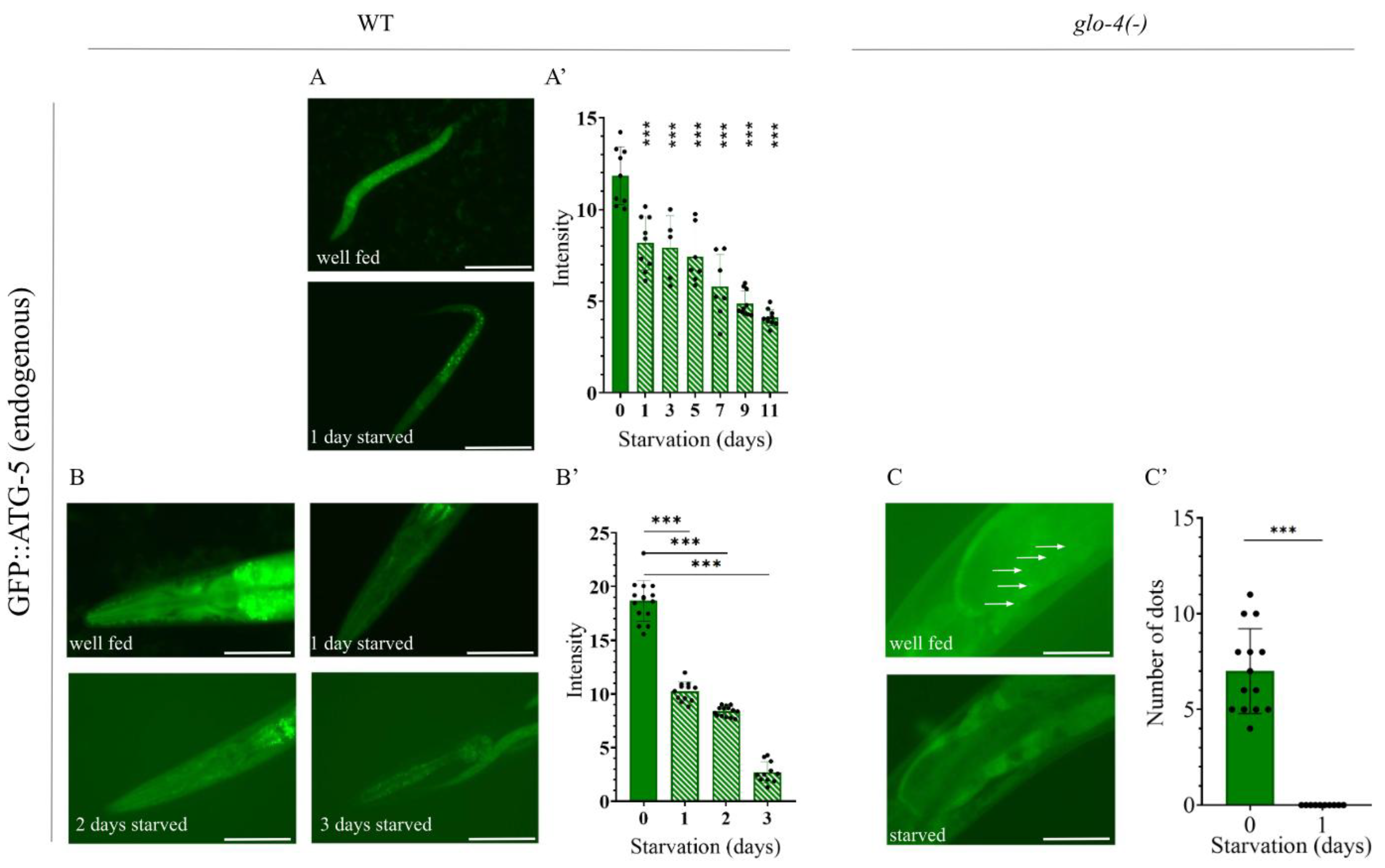
2.6. Analysis of Autophagy Using the Endogenous GFP::ATG-5 Reporter
3. Discussion
4. Materials and Methods
4.1. Strains and Maintenance
4.2. Western Blot Analysis
4.3. Starvation Assay
4.4. RNA Interference
4.5. Bafilomycin A1 Treatment
4.6. Fluorescent Microscopy
4.7. Statistics
5. Conclusion
ACKNOWLEDGEMENTS
AUTHOR CONTRIBUTIONS
ADDITIONAL INFORMATION
References
- M. L. Tóth, T. Sigmond, E. Borsos, J. Barna, P. Erdélyi, K. Takács-Vellai, L. Orosz, A.L. Kovács, G. Csikós, M. Sass, and T. Vellai, „Longevity pathways converge on autophagy genes to regulate life span in Caenorhabditis elegans,” 2008, Autophagy, 4(3), 330–338. [CrossRef]
- N. Mizushima, B. Levine, A. M. Cuervo, and D. J. Klionsky, “Autophagy fights disease through cellular self-digestion.,” Nature, vol. 451, no. February, pp. 1069–1075, 2008. [CrossRef]
- D. J. Klionsky, “The molecular machinery of autophagy: unanswered questions.,” J Cell Sci, vol. 118, pp. 7–18, 2005. [CrossRef]
- M. A. Ortega et al., “Autophagy in Its (Proper) Context: Molecular Basis, Biological Relevance, Pharmacological Modulation, and Lifestyle Medicine,” 2024, Ivyspring International Publisher. [CrossRef]
- A. L. Anding and E. H. Baehrecke, “Cleaning House: Selective Autophagy of Organelles,” Apr. 10, 2017, Cell Press. [CrossRef]
- G. Kroemer, G. Mariño, and B. Levine, “Autophagy and the Integrated Stress Response,” Oct. 2010. [CrossRef]
- E. K. Jo, J. M. Yuk, D. M. Shin, and C. Sasakawa, “Roles of autophagy in elimination of intracellular bacterial pathogens,” Front Immunol, vol. 4, no. MAY, 2013. [CrossRef]
- Y. Pang, L. Wu, C. Tang, H. Wang, and Y. Wei, “Autophagy-Inflammation Interplay During Infection: Balancing Pathogen Clearance and Host Inflammation,” Feb. 22, 2022, Frontiers Media S.A. [CrossRef]
- D. Glick, S. Barth, and K. F. Macleod, “Autophagy: Cellular and molecular mechanisms,” May 2010. [CrossRef]
- Y. Aman et al., “Autophagy in healthy aging and disease,” Aug. 01, 2021, Springer. [CrossRef]
- F. Guo, X. Liu, H. Cai, and W. Le, “Autophagy in neurodegenerative diseases: pathogenesis and therapy,” Jan. 01, 2018, Blackwell Publishing Ltd. [CrossRef]
- J. Nah, J. Yuan, and Y. K. Jung, “Autophagy in neurodegenerative diseases: From mechanism to therapeutic approach,” 2015, Korean Society for Molecular and Cellular Biology. [CrossRef]
- E. Wong and A. M. Cuervo, “Autophagy gone awry in neurodegenerative diseases.,” Nat Neurosci, vol. 13, no. 7, pp. 805–811, 2010. [CrossRef]
- Shen, Y.; Li, M.; Wang, K.; Qi, G.; Liu, H.; Wang, W.; Ji, Y.; Chang, M.; Deng, C.; Xu, F.; Shen, M.; Sun, H. “Diabetic Muscular Atrophy: Molecular Mechanisms and Promising Therapies.”, 2022, Front. Endocrinol.13. [CrossRef]
- Chen, Y., Wang, Z., Ma, Q., & Sun, C. “The role of autophagy in fibrosis: Mechanisms, progression and therapeutic potential”, 2025. International Journal of Molecular Medicine, 55(4). [CrossRef]
- K. Palikaras, E. Lionaki, and N.Tavernarakis, „Coordination of mitophagy and mitochondrial biogenesis during ageing in C. elegans”, 2015, Nature, 521(7553), 525–528. [CrossRef]
- V. I. Korolchuk, A. Mansilla, F. M. Menzies, and D. C. Rubinsztein, “Autophagy Inhibition Compromises Degradation of Ubiquitin-Proteasome Pathway Substrates,” Mol Cell, vol. 33, no. 4, pp. 517–527, Feb. 2009. [CrossRef]
- Y. Chen and D. J. Klionsky, “The regulation of autophagy - unanswered questions.,” J Cell Sci, vol. 124, pp. 161–170, 2011. [CrossRef]
- J. H. Hurley and L. N. Young, “Mechanisms of autophagy initiation,” Annu Rev Biochem, vol. 86, pp. 225–244, Jun. 2017. [CrossRef]
- A. K. Corsi, B. Wightman, and M. Chalfie, “A transparent window into biology: A primer on Caenorhabditis elegans” 2015, Genetics, vol. 200, no. 2, pp. 387–407, 2015. [CrossRef]
- T. Keith Blackwell, A. K. Sewell, Z. Wu, and M. Han, “TOR signaling in caenorhabditis elegans development, metabolism, and aging,” Genetics, vol. 213, no. 2, pp. 329–360, 2019. [CrossRef]
- Hansen, M., Chandra A., Mitani S., et al., “Autophagy and longevity in Caenorhabditis elegans.”, 2018, Trends Cell Biol, 28(3). [CrossRef]
- Prasher D C, “Using GFP to see the light,” Trends Genet., vol. 8, pp. 320–3, 1995. [CrossRef]
- K. D. Piatkevich and V. V. Verkhusha, Guide to red fluorescent proteins and biosensors for flow cytometry, vol. 102. 2011. [CrossRef]
- N. J. Palmisano and A. Meléndez, “Detection of autophagy in Caenorhabditis elegans using GFP::LGG-1 as an autophagy marker,” Cold Spring Harb Protoc, vol. 2016, no. 1, pp. 68–75, Jan. 2016. [CrossRef]
- Y. Chen, V. Scarcelli, and R. Legouis, “Approaches for studying autophagy in caenorhabditis elegans,” Sep. 01, 2017, MDPI. [CrossRef]
- J. T. Chang, C. Kumsta, A. B. Hellman, L. M. Adams, and M. Hansen, “Spatiotemporal regulation of autophagy during Caenorhabditis elegans aging,” Elife, vol. 6, pp. 1–23, 2017. [CrossRef]
- N. Mizushima, and T. Yoshimori, „Autophagic flux measurement: Cargo degradation versus autophagosome formation,” 2025, Trends in Cell Biology, 35(2), 87–98. [CrossRef]
- S. Kimura, T. Noda, and T. Yoshimori, “Dissection of the autophagosome maturation process by a novel reporter protein, tandem fluorescent-tagged LC3,” Autophagy, vol. 3, no. 5, pp. 452–460, 2007. [CrossRef]
- A. Meléndez, Z. Tallóczy, M. Seaman, E.-L. Eskelinen, D. H. Hall, and B. Levine, “Autophagy genes are essential for dauer development and life-span extension in C. elegans,” Science (1979), vol. 301, no. 5638, pp. 1387–91, 2003. [CrossRef]
- K. Noma and Y. Jin, “Rapid integration of multi-copy transgenes using optogenetic mutagenesis in Caenorhabditis elegans,” G3: Genes, Genomes, Genetics, vol. 8, no. 6, pp. 2091–2097, Jun. 2018. [CrossRef]
- V. P. Reddy Chichili, V. Kumar, and J. Sivaraman, “Linkers in the structural biology of protein-protein interactions,” Protein Sci., vol. 22. no. 2, pp. 153-67. [CrossRef]
- Jiang, M., Liu, K., Luo, J., & Dong, Z. (2010). “Autophagy Is a Renoprotective Mechanism During in Vitro Hypoxia and in Vivo Ischemia-Reperfusion Injury.” The American Journal of Pathology, 176(3), 1181–1192. [CrossRef]
- H. Tai et al., “Autophagy impairment with lysosomal and mitochondrial dysfunction is an important characteristic of oxidative stress-induced senescence,” Autophagy, vol. 13, no. 1, pp. 99–113, Jan. 2017. [CrossRef]
- Y. Tian et al., “C. elegans Screen Identifies Autophagy Genes Specific to Multicellular Organisms,” Cell, vol. 141, no. 6, pp. 1042–1055, 2010. [CrossRef]
- H. Zhang and A. Meléndez, “Conserved components of the macroautophagy machinery in Caenorhabditis elegans,” Genetics, Volume 229, Issue 4, Apr. 2025. [CrossRef]
- L. R. Lapierre et al., “The TFEB orthologue HLH-30 regulates autophagy and modulates longevity in Caenorhabditis elegans,” Nat Commun, vol. 4, pp. 1–17, 2013. [CrossRef]
- Y. Mugume, Z. Kazibwe, and D. C. Bassham, “Target of rapamycin in control of autophagy: Puppet master and signal integrator,” Nov. 01, 2020, MDPI AG. [CrossRef]
- X. Long, C. Spycher, Z. S. Han, A. M. Rose, F. Müller, and J. Avruch, “TOR Deficiency in C. elegans Causes Developmental Arrest and Intestinal Atrophy by Inhibition of mRNA Translation,” Current Biology, vol. 12, no. 17, pp. 1448–1461, Sep. 2002. [CrossRef]
- Mizushima, N., Yoshimori, T., & Levine, B. (2010). “Methods in Mammalian Autophagy Research.” Cell, 140(3), 313–326. [CrossRef]
- L. R. Baugh and P. J. Hu, “Starvation responses throughout the caenorhabditis elegans life cycle,” Dec. 01, 2020, Genetics Society of America. [CrossRef]
- A. Mata-Cabana, F. J. Romero-Expósito, and M. Olmedo, “Aging during C. elegans L1 quiescence,” Aging (Albany NY), vol. 12, no. 18, pp. 17756–17758, 2020. [CrossRef]
- A. M. L. Tóth et al., “Longevity pathways converge on autophagy genes to regulate life span in Caenorhabditis elegans,” Autophagy, vol. 4, no. 3, pp. 330–338, Apr. 2008. [CrossRef]
- C. Kang, N. J. You, and L. Avery, “Dual roles of autophagy in the survival of Caenorhabditis elegans during starvation,” Genes Dev, vol. 21, no. 17, pp. 2161–2171, Sep. 2007. [CrossRef]
- Z. Xie, Y. Xie, Y. Xu, H. Zhou, W. Xu, and Q. Dong, “Bafilomycin A1 inhibits autophagy and induces apoptosis in MG63 osteosarcoma cells,” Mol Med Rep, vol. 10, no. 2, pp. 1103–1107, 2014. [CrossRef]
- N. Yuan et al., “Bafilomycin A1 targets both autophagy and apoptosis pathways in pediatric B-cell acute lymphoblastic leukemia,” Haematologica, vol. 100, no. 3, pp. 345–356, 2015. [CrossRef]
- X. Ye, X. J. Zhou, and H. Zhang, “Exploring the role of autophagy-related gene 5 (ATG5) yields important insights into autophagy in autoimmune/autoinflammatory diseases,” Oct. 17, 2018, Frontiers Media S.A. [CrossRef]
- E. Currie, B. King, A. L. Lawrenson, L. K. Schroeder, A. M. Kershner, and G. J. Hermann, “Role of the Caenorhabditis elegans multidrug resistance gene, mrp-4, in gut granule differentiation,” Genetics, vol. 177, no. 3, pp. 1569–1582, Nov. 2007. [CrossRef]
- G. J. Hermann et al., “Genetic analysis of lysosomal trafficking in Caenorhabditis elegans,” Mol Biol Cell, vol. 16, no. 7, pp. 3273–3288, Jul. 2005. [CrossRef]
- J. M. I. Barth, J. Szabad, E. Hafen, and K. Köhler, “Autophagy in Drosophila ovaries is induced by starvation and is required for oogenesis,” Cell Death Differ, vol. 18, no. 6, pp. 915–924, Jun. 2011. [CrossRef]
- K. Shi and C. Tong, “Analyzing Starvation-Induced Autophagy in the Drosophila melanogaster Larval Fat Body,” JoVE, no. 186, p. e64282, 2022. [CrossRef]
- C. K. McPhee and E. H. Baehrecke, “Autophagy in Drosophila melanogaster,” Sep. 2009. [CrossRef]
- N. Mizushima, A. Yamamoto, M. Matsui, T. Yoshimori, and Y. Ohsumi, “In Vivo Analysis of Autophagy in Response to Nutrient Starvation Using Transgenic Mice Expressing a Fluorescent Autophagosome Marker,” Mol Biol Cell, vol. 15, pp. 1101–1111, 2004. [CrossRef]
- H. C. Chapin, M. Okada, A. J. Merz, and D. L. Miller, “Tissue-specific autophagy responses to aging and stress in C. elegans,” Aging (Albany NY)., vol. 6, no. 7, pp. 419–34, 2015. [CrossRef]
- S. H. Y. Lim, M. Hansen, and C. Kumsta, “Molecular Mechanisms of Autophagy Decline during Aging,” Aug. 01, 2024, Multidisciplinary Digital Publishing Institute (MDPI). [CrossRef]
- M. Redman, A. King, C. Watson, and D. King, “What is CRISPR/Cas9?,” Arch Dis Child Educ Pract Ed, vol. 101, no. 4, pp. 213–215, Aug. 2016. [CrossRef]
- R. C. Scott, O. Schuldiner, and T. P. Neufeld, “Role and Regulation of Starvation-Induced Autophagy in the Drosophila Fat Body internal reserve of nutrients. In this catabolic process, cytoplasm is engulfed within double-membrane vesicles known as autophagosomes, which subsequently fuse,” 2004. [Online]. Available: http://www.developmentalcell.
- A. Kuma et al., “The role of autophagy during the early neonatal starvation period,” Nature, vol. 432, no. 7020, pp. 1032–1036, 2004. [CrossRef]
- D. Conte, L. T. MacNei, A. J. M. Walhout, and C. C. Mello, “RNA Interference in Caenorhabditis elegans,” Curr Protoc Mol Biol, vol. 109, pp. 26.3.1-26.3.30, 2015. [CrossRef]
- Á. Sturm, É. Saskoï, K. Tibor, N. Weinhardt, and T. Vellai, “Highly efficient RNAi and Cas9-based auto-cloning systems for C. Elegans research,” Nucleic Acids Res, vol. 46, no. 17, Sep. 2018. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



