Submitted:
06 October 2025
Posted:
09 October 2025
You are already at the latest version
Abstract
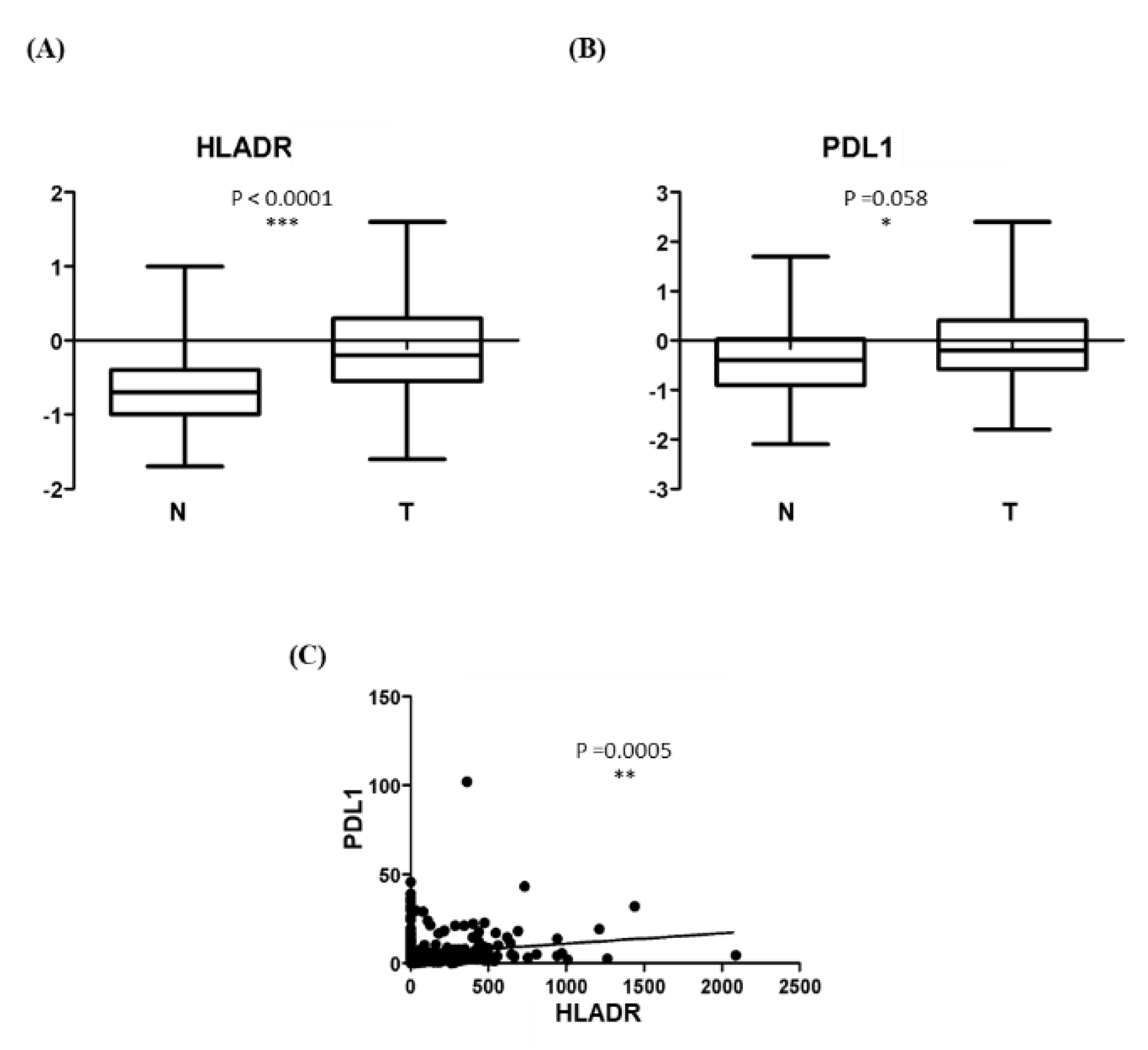
Objective: Oral squamous cell carcinoma (OSCC) is a prevalent form of head and neck squamous cell carcinoma (HNSCC). Traditional treatments include surgery and radiotherapy/chemotherapy, but these often have limited efficacy. Immunotherapy has emerged as a promising treatment option. Recognition of novel biomarkers for immune checkpoints modulation could be helpful for establishing a personalized treatment for OSCC patients. Recently we showed that HNSCC epithelial cells acquire the ability to express class II MHC, HLA-DR. We aimed to investigate HLA-DR role in relation to the presence of the tumor microenvironment represented by CAFs. In addition, since programmed death-ligand 1 protein (PD-L1) is a critical target for cancer immunotherapy in HNSCC, we evaluated HLA-DR and PD-L1 level in healthy and OSCC tissues. Methods: Biopsy specimens from five OSCC patients were used to isolate tumor cells, normal fibroblast and CAFs. HLA-DR expression was evaluated. Cancer Genome Atlas database was used to evaluate HLA-DR and PD-L1 level in normal tissues and HNSCC derived biopsies. Results: Epithelial cancer cells showed de-novo HLA-DR expression. Importantly, HLA-DR expression was observed in both CAFs and normal fibroblasts with a significant higher level in CAFs. Atlas analysis showed an increase of HLA-DR and PD-L1 expression in HNSCC derived biopsies vs normal tissues and a positive correlation between HLA-DR and PD-L1 in cancer samples. Conclusions: Our findings highlighted that tumoral environment could affect OSCC cells inducing HLA-DR expression in non-APC tumoral and CAFs cells. The correlation between PD-L1 and HLA-DR expression has been observed, providing useful insights in targeted immunotherapy strategies for OSCC management.
Keywords:
1. Introduction
2. Materials and Methods
Ethical Consideration
Patients
Primary Cell Cultures
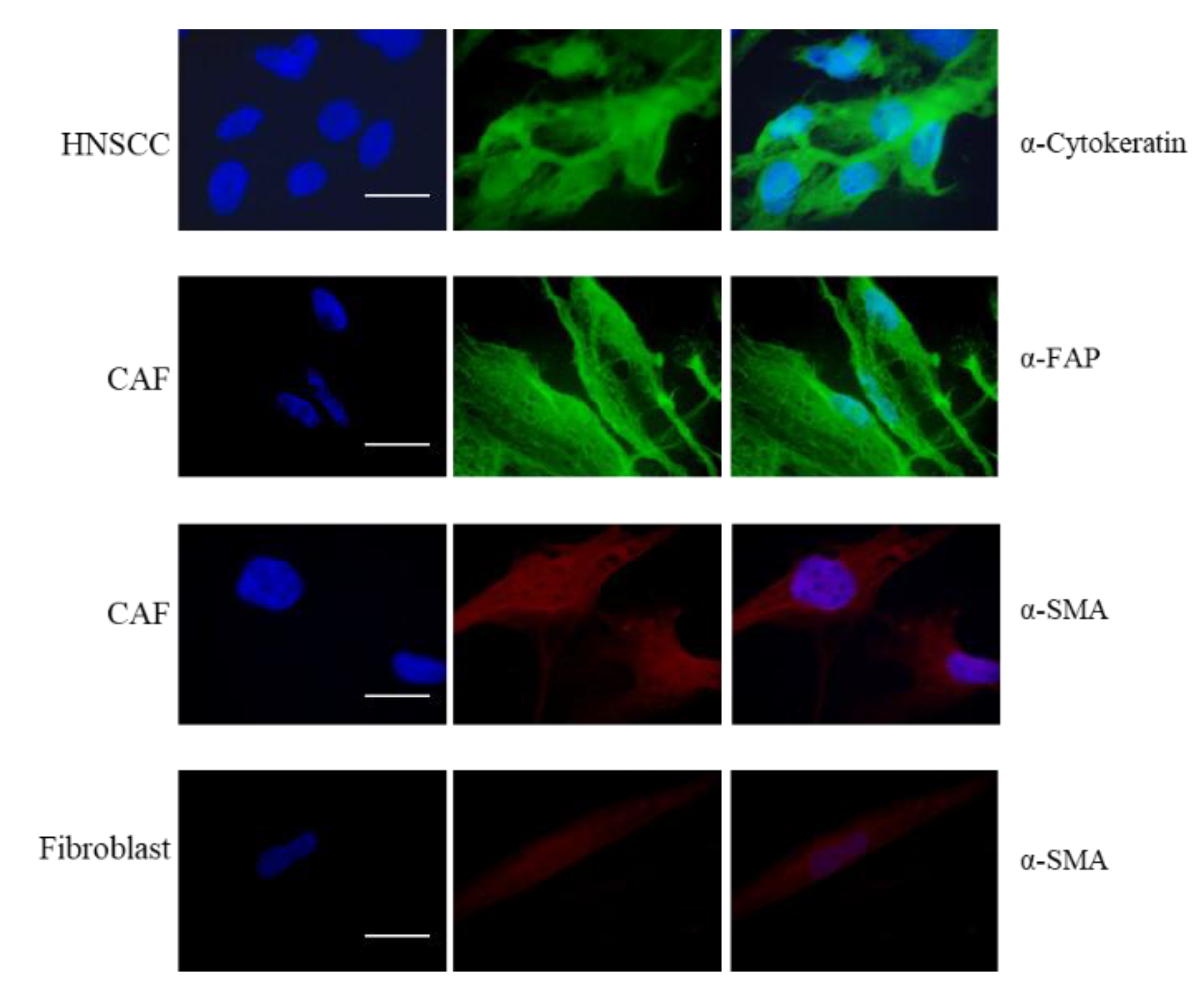
Immunofluorescence Analysis
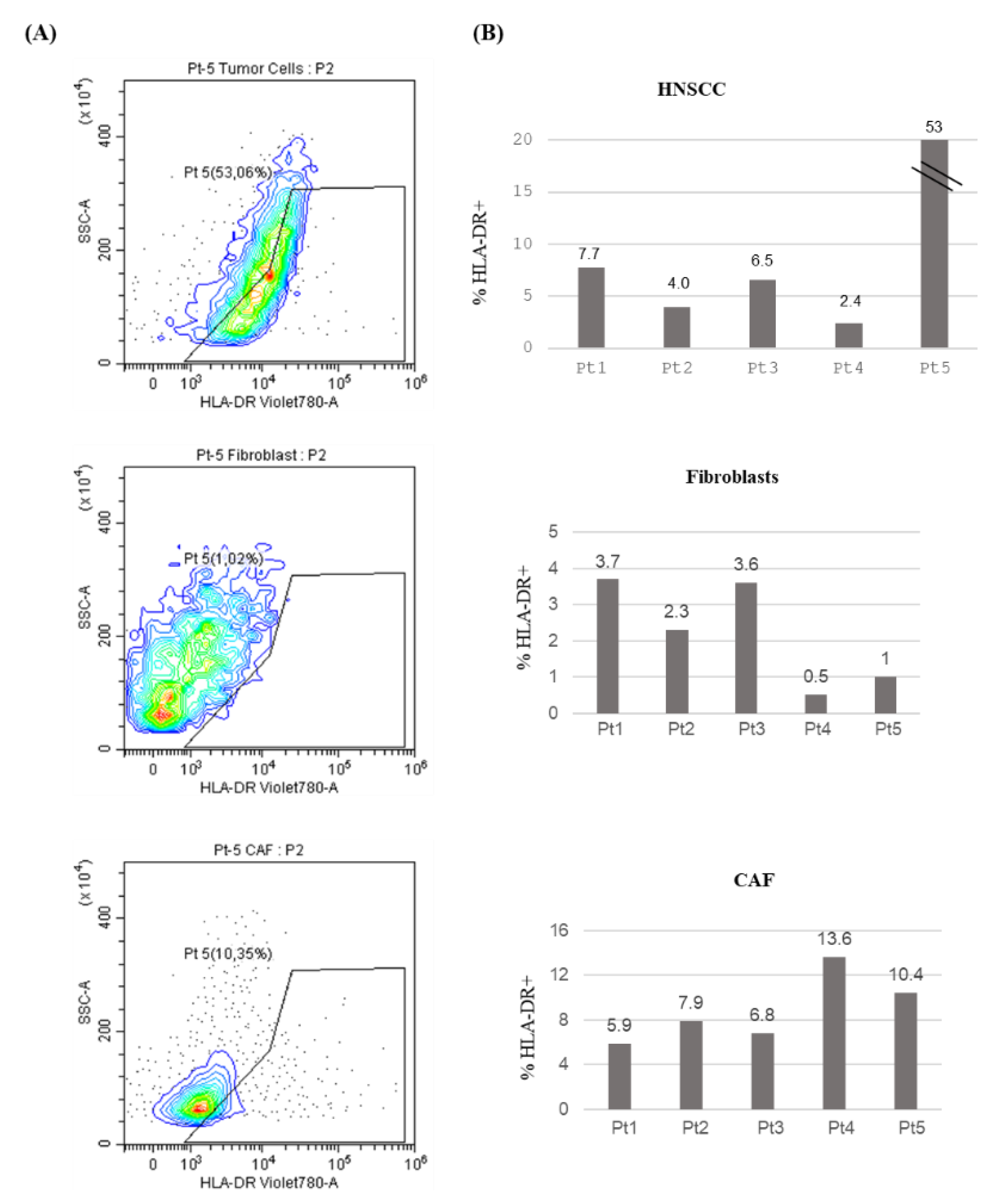
Flow Cytometry – Sorting
Evaluation of HLA-DR Expression
HPV Detection and Genotyping
DNA Detection
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tan, Y.; Wang, Z.; Xu, M.; Li, B.; Huang, Z.; Qin, S.; Nice, E.C.; Tang, J.; Huang, C. Oral Squamous Cell Carcinomas: State of the Field and Emerging Directions. Int J Oral Sci 2023, 15, 44. [Google Scholar] [CrossRef] [PubMed]
- Polz-Gruszka, D.; Morshed, K.; Stec, A.; Polz-Dacewicz, M. Prevalence of Human Papillomavirus (HPV) and Epstein-Barr Virus (EBV) in Oral and Oropharyngeal Squamous Cell Carcinoma in South-Eastern Poland. Infect Agents Cancer 2015, 10, 37. [Google Scholar] [CrossRef]
- Stepan, K.O.; Mazul, A.L.; Larson, J.; Shah, P.; Jackson, R.S.; Pipkorn, P.; Kang, S.Y.; Puram, S.V. Changing Epidemiology of Oral Cavity Cancer in the United States. Otolaryngol Head Neck Surg 2023, 168, 761–768. [Google Scholar] [PubMed]
- El-Bayoumy, K.; Christensen, N.D.; Hu, J.; Viscidi, R.; Stairs, D.B.; Walter, V.; Chen, K.M.; Sun, Y.W.; Muscat, J.E.; JP, R. , Jr An Integrated Approach for Preventing Oral Cavity and Oropharyngeal Cancers: Two Etiologies with Distinct and Shared Mechanisms of Carcinogenesis. Cancer Prev Res 2020, 13, 649–660. [Google Scholar] [CrossRef]
- Coletta, R.D.; Yeudall, W.A.; Salo, T. Grand Challenges in Oral Cancers. Front Oral Health 2020, 1. [Google Scholar] [CrossRef]
- Kaidar-Person, O.; Gil, Z.; Billan, S. Precision medicine in head and neck cancer. Drug Resist Updat 2018, 40, 13–16. [Google Scholar] [CrossRef]
- Tripathi, D.; Gupta, T.; Rai, A.K.; Pandey, P. Pioneering a New Era in Oral Cancer Treatment with Electrospun Nanofibers: A Comprehensive Insight. Anticancer Agents Med Chem. Published online January 2025, 3. [Google Scholar] [CrossRef]
- Lyford-Pike, S.; Peng, S.; Young, G.D. Evidence for a Role of the PD-1:PD-L1 Pathway in Immune Resistance of HPV-Associated Head and Neck Squamous Cell Carcinoma. Cancer Res 2013, 73, 1733–1741. [Google Scholar] [CrossRef]
- Rothschild, U.; Muller, L.; Lechner, A. Immunotherapy in Head and Neck Cancer—Scientific Rationale, Current Treatment Options and Future Directions. Swiss Med Wkly 2018, 148, w14625. [Google Scholar] [CrossRef]
- Turpin, R.; Peltonen, K.; Rannikko, J.H.; Liu, R.; Kumari, A.N.; Nicorici, D.; Lee, M.H.; Mutka, M.; Kovanen, P.E.; Niinikoski, L.; et al. Patient-Derived Tumor Explant Models of Tumor Immune Microenvironment Reveal Distinct and Reproducible Immunotherapy Responses. OncoImmunology 2025, 14, 2466305. [Google Scholar] [CrossRef]
- Partlová, S.; Bouček, J.; Kloudová, K.; Lukešová, E.; Zábrodský, M.; Grega, M.; Fučíková, J.; Truxová, I.; Tachezy, R.; Špíšek, R.; et al. Distinct Patterns of Intratumoral Immune Cell Infiltrates in Patients with HPV-Associated Compared to Non-Virally Induced Head and Neck Squamous Cell Carcinoma. Oncoimmunology 2015, 4, e965570. [Google Scholar] [CrossRef]
- Mandal, R.; Şenbabaoğlu, Y.; Desrichard, A.; Havel, J.J.; Dalin, M.G.; Riaz, N.; Lee, K.W.; Ganly, I.; Hakimi, A.A.; Chan, T.A.; et al. The Head and Neck Cancer Immune Landscape and Its Immunotherapeutic Implications. JCI Insight 2016, 1, e89829. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Li, S.; Jiang, Y.; Chen, T.; An, Z. Unleashing the Power of Immune Checkpoints: A New Strategy for Enhancing Treg Cells Depletion to Boost Antitumor Immunity. Int Immunopharmacol 2025, 147, 113952. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Yao, F.; Wu, L.; Xu, T.; Na, J.; Shen, Z.; Liu, X.; Shi, W.; Zhao, Y.; Liao, Y. Heterogeneity and Interplay: The Multifaceted Role of Cancer-Associated Fibroblasts in the Tumor and Therapeutic Strategies. Clin Transl Oncol 2024, 26, 2395–2417. [Google Scholar] [CrossRef] [PubMed]
- Elyada, E.; Bolisetty, M.; Laise, P.; Flynn, W.F.; Courtois, E.T.; Burkhart, R.A.; Teinor, J.A.; Belleau, P.; Biffi, G.; Lucito, M.S.; et al. Cross-Species Single-Cell Analysis of Pancreatic Ductal Adenocarcinoma Reveals Antigen-Presenting Cancer-Associated Fibroblasts. Cancer Discov 2019, 9, 1102–1123. [Google Scholar] [CrossRef]
- Axelrod, M.L.; Cook, R.S.; Johnson, D.B.; Balko, J.M. Biological Consequences of MHC-II Expression by Tumor Cells in Cancer. Clin Cancer Res. Apr 2019, 25, 2392–2402. [Google Scholar] [CrossRef]
- Johnson, D.B.; Estrada, M.V.; Salgado, R.; Sanchez, V.; Doxie, D.B.; Opalenik, V., SR; AE, F.; E, J.; AS, G.; AR, S.; et al. Melanoma-Specific MHC-II Expression Represents a Tumour-Autonomous Phenotype and Predicts Response to Anti-PD-1/PD-L1 Therapy. Nat Commun 2016, 7, 10582. [Google Scholar] [CrossRef]
- Prampolini, C.; Almadori, G.; Bonvissuto, D.; Barba, M.; Giraldi, L.; Boccia, S.; Paludetti, G.; Galli, J.; Parolini, O.; Settimi, S.; et al. Immunohistochemical Detection of “Ex Novo” HLA-DR in Tumor Cells Determines Clinical Outcome in Laryngeal Cancer Patients. HLA 2021, 98, 517–524. [Google Scholar] [CrossRef]
- Amrane, K.; Le Meur, C.; Besse, B.; Hemon, P.; Le Noac’h, P.; Pradier, O.; Berthou, C.; Abgral, R.; Uguen, A. HLA-DR Expression in Melanoma: From Misleading Therapeutic Target to Potential Immunotherapy Biomarker. Front Immunol 2024, 14, 1285895. [Google Scholar] [CrossRef]
- Hudson, K.; Cross, N.; Jordan-Mahy, N.; Leyland, R. The Extrinsic and Intrinsic Roles of PD-L1 and Its Receptor PD-1: Implications for Immunotherapy Treatment. Frontiers in Immunology 2020, 11, 568931. [Google Scholar] [CrossRef]
- Hirata-Nozaki, Y.; Ohkuri, T.; Ohara, K.; Kumai, T.; Nagata, M.; Harabuchi, S.; Kosaka, A.; Nagato, T.; Ishibashi, K.; Oikawa, K.; et al. PD-L1-Specific Helper T-Cells Exhibit Effective Antitumor Responses: New Strategy of Cancer Immunotherapy Targeting PD-L1 in Head and Neck Squamous Cell Carcinoma. J Transl Med 2019, 17, 207. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, N.; Gangadharan, C.; Pillai, V.; Kuriakose, M.A.; Suresh, A.; Das, M. Establishment and Characterization of Novel Autologous Pair Cell Lines from Two Indian Non Habitual Tongue Carcinoma Patients. Oncol Rep 2022, 48, 150. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjöstedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A Pathology Atlas of the Human Cancer Transcriptome. 2017, 357, eaan2507. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D. MKGE, Christian Wittekind. In TNM Classification of Malignant Tumours; 2016.
- Yan, S.; Zhang, X.; Lin, Q.; Du, M.; Li, Y.; He, S.; Chen, J.; Li, X.; Bei, J.; Chen, S.; et al. Deciphering the Interplay of HPV Infection, MHC-II Expression, and CXCL13+ CD4+ T Cell Activation in Oropharyngeal Cancer: Implications for Immunotherapy. Cancer Immunol Immunother 2024, 73, 206. [Google Scholar] [CrossRef]
- Evans, A.M.; Salnikov, M.; Tessier, T.M.; Mymryk, J.S. Reduced MHC Class I and II Expression in HPV-Negative vs. HPV-Positive Cervical Cancers. Cells 2022, 11, 3911. [Google Scholar] [CrossRef]
- Shamseddine, A.A.; Burman, B.; Lee, N.Y.; Zamarin, D.; Riaz, N. Tumor Immunity and Immunotherapy for HPV-Related Cancers. Cancer Discov 2021, 11, 1896–1912. [Google Scholar]
| Case n. | Age | sex | staging | HPV | outcome |
|---|---|---|---|---|---|
| 1 | 57 | Male | SIII | Negative | DOD |
| 2 | 95 | Male | SIV | Negative | DOD |
| 3 | 56 | Male | SIV | Negative | ALIVE |
| 4 | 69 | Male | SII | Negative | AWED |
| 5 | 69 | Male | SIV | Negative | ALIVE |
| All ( N= 492) |
|
|---|---|
| Age | 61.1 ± 12.0 |
| Sex Male Female |
361 (73.4%) 131 (26.6%) |
| Stage Missing I II III IV |
67 (13.6%) 24 (4.9%) 69 (14.0%) 76 (15.4%) 256 (52.0%) |
| Race american indian or alaska native asian black or african american white missing |
2 (0.4%) 10 (2.0%) 45 (9.1%) 421 (85.6%) 14 (2.8%) |
| HLA-DR expression | 0.70(0.20-89.70) |
| PDL1 expression | 2.90 (1.40-6.25) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




