Submitted:
06 October 2025
Posted:
07 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
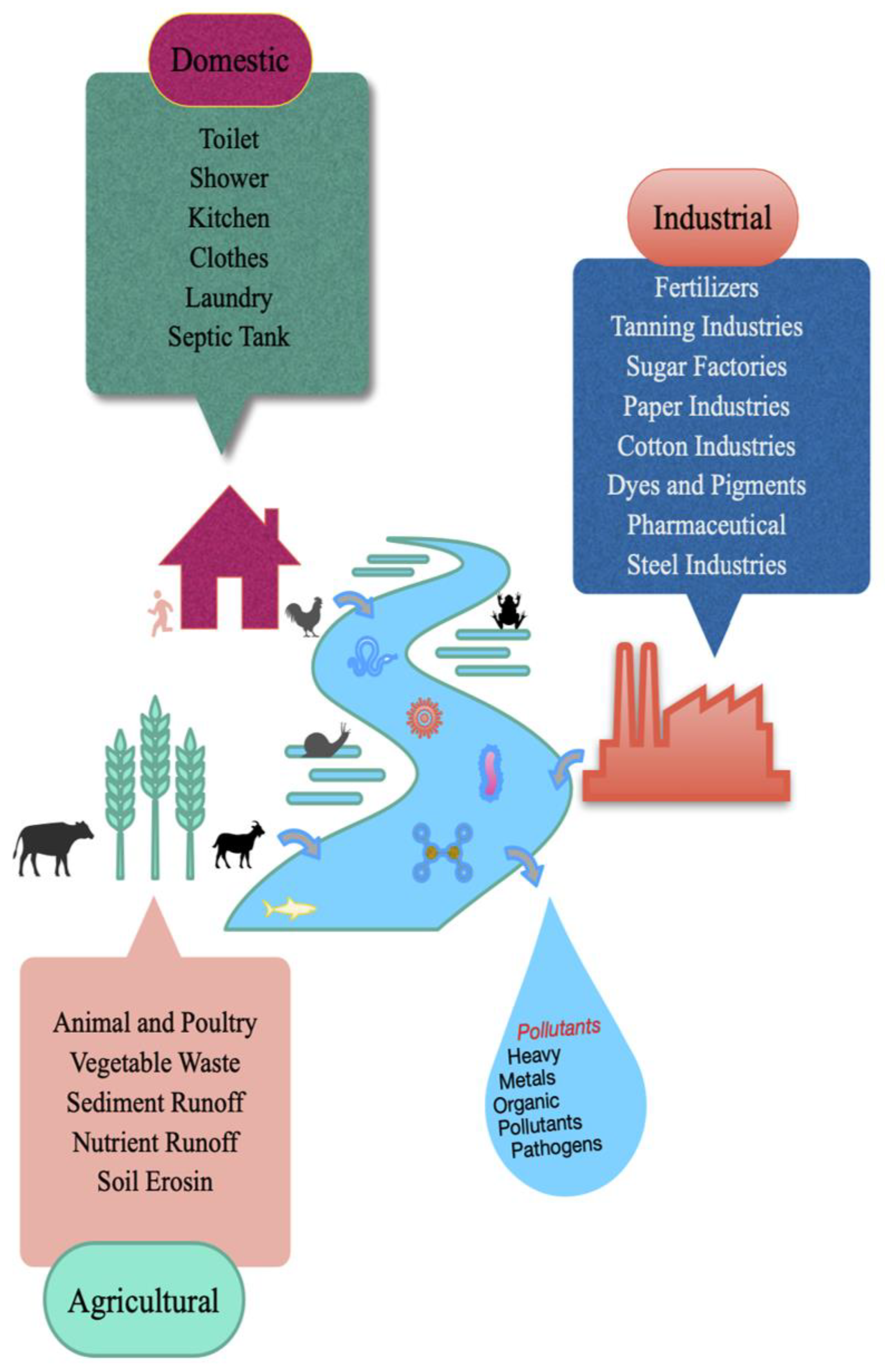
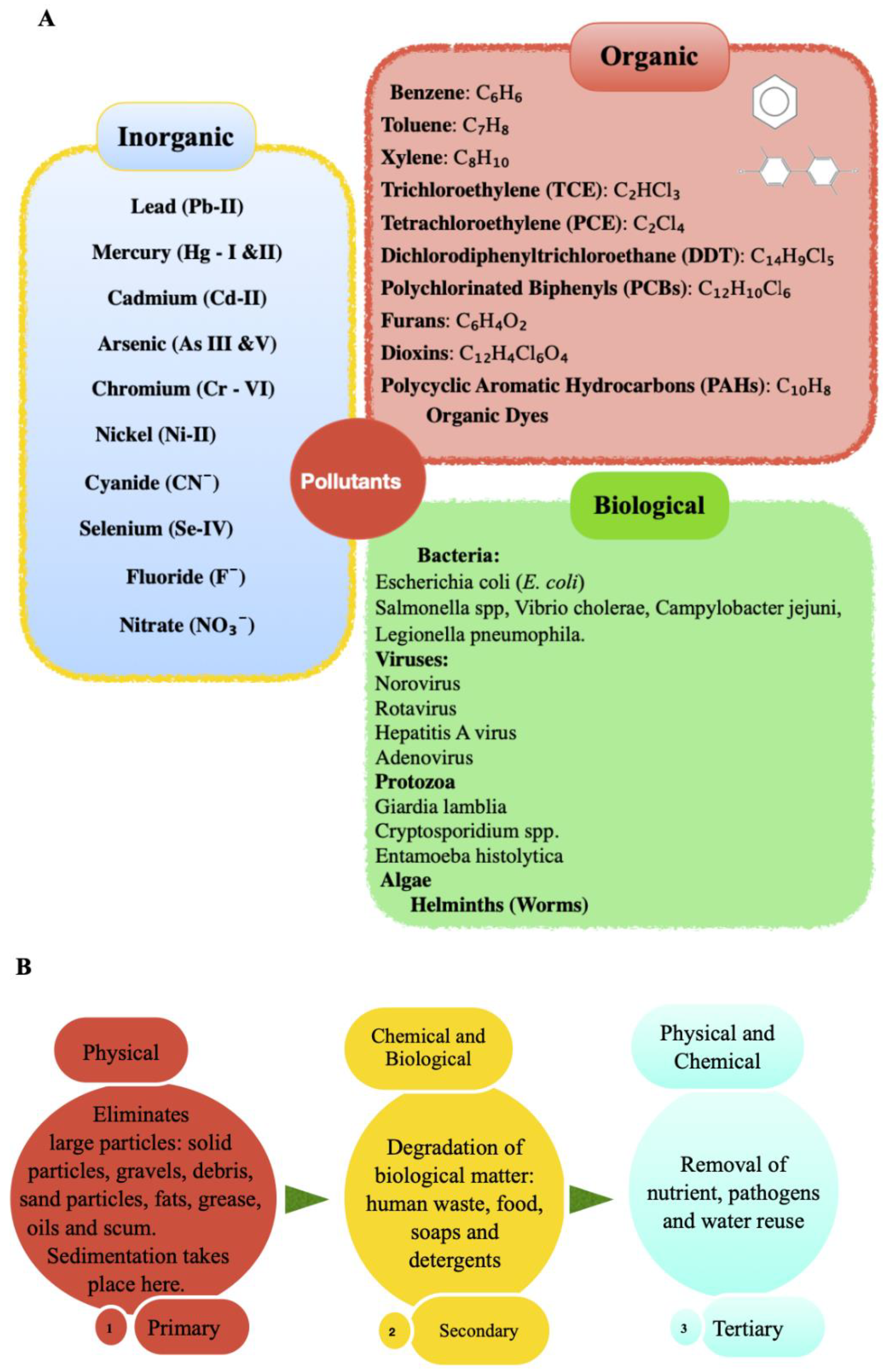
2. Types of Wastewater and Pollutants
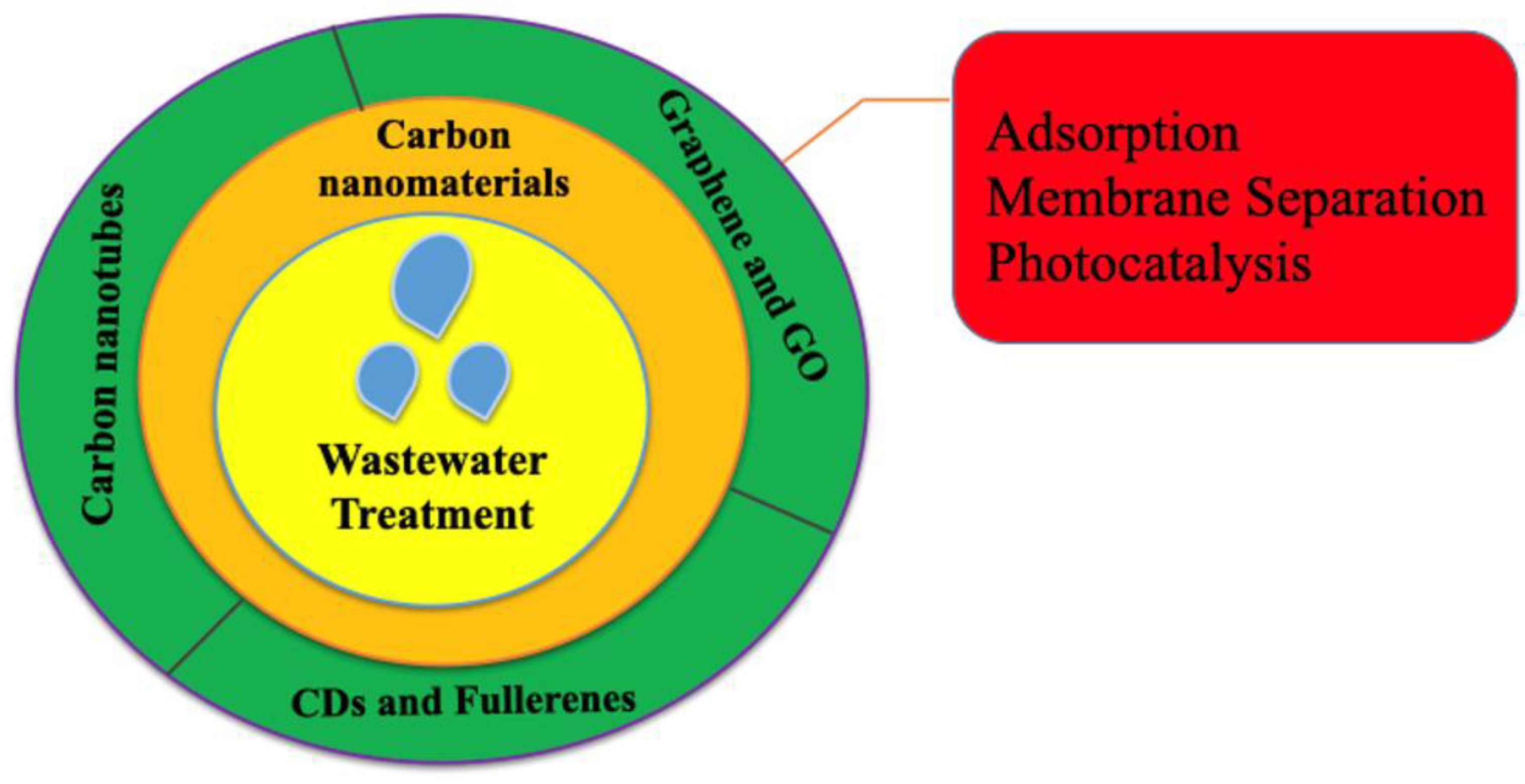
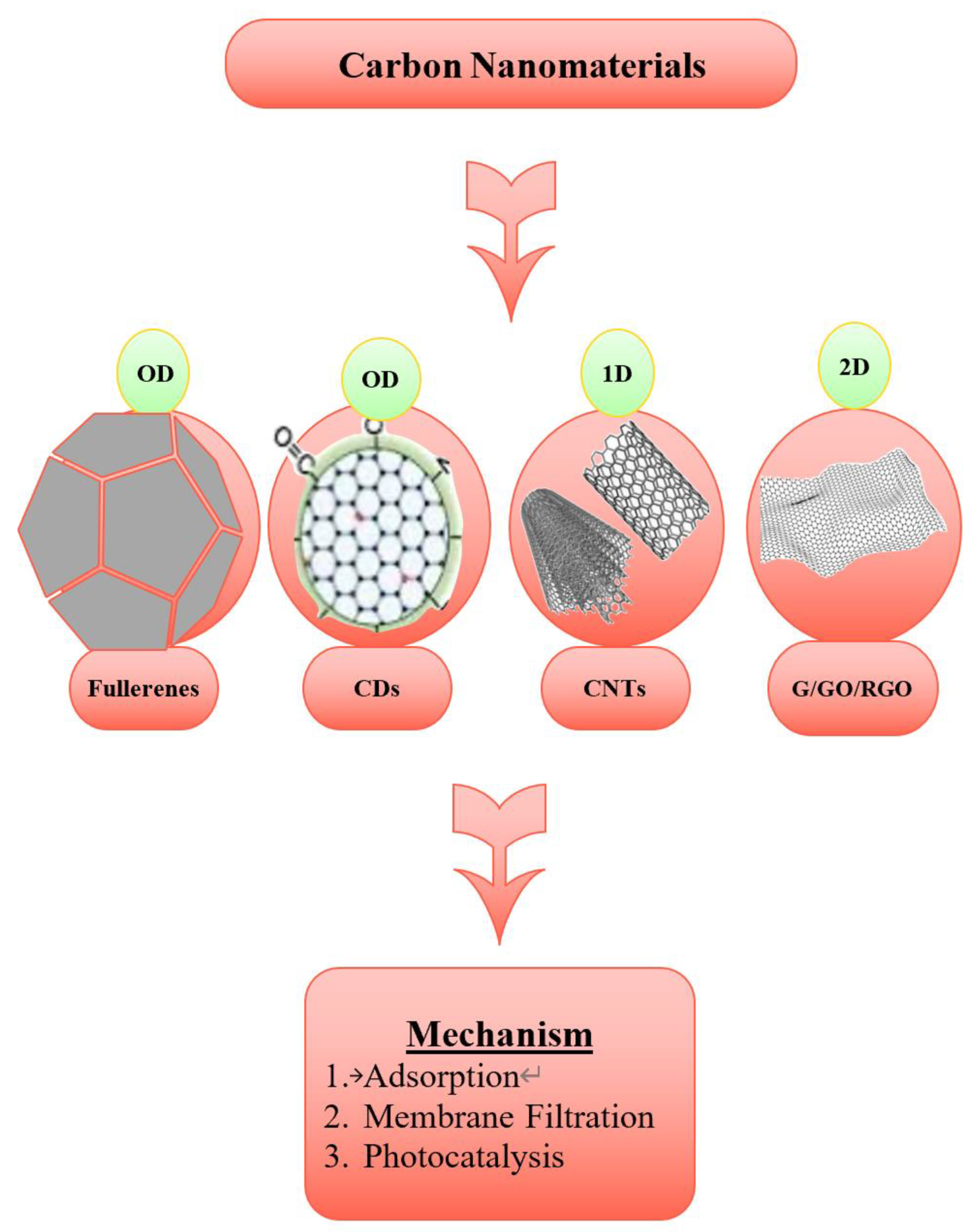
3. Carbon-Based Nanomaterials for Wastewater Treatment: Mechanism Insights
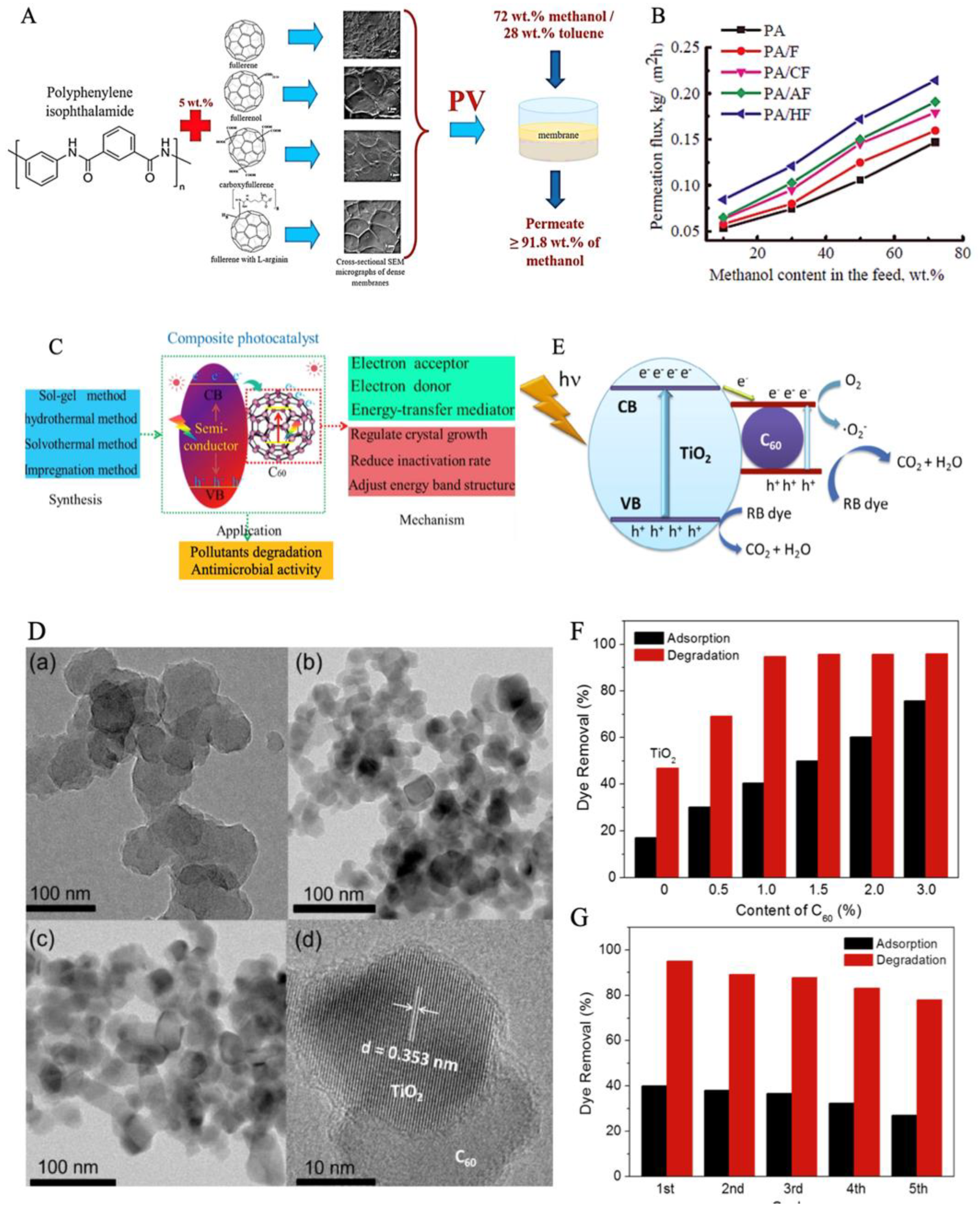
3.1. Fullerenes for Water Treatment
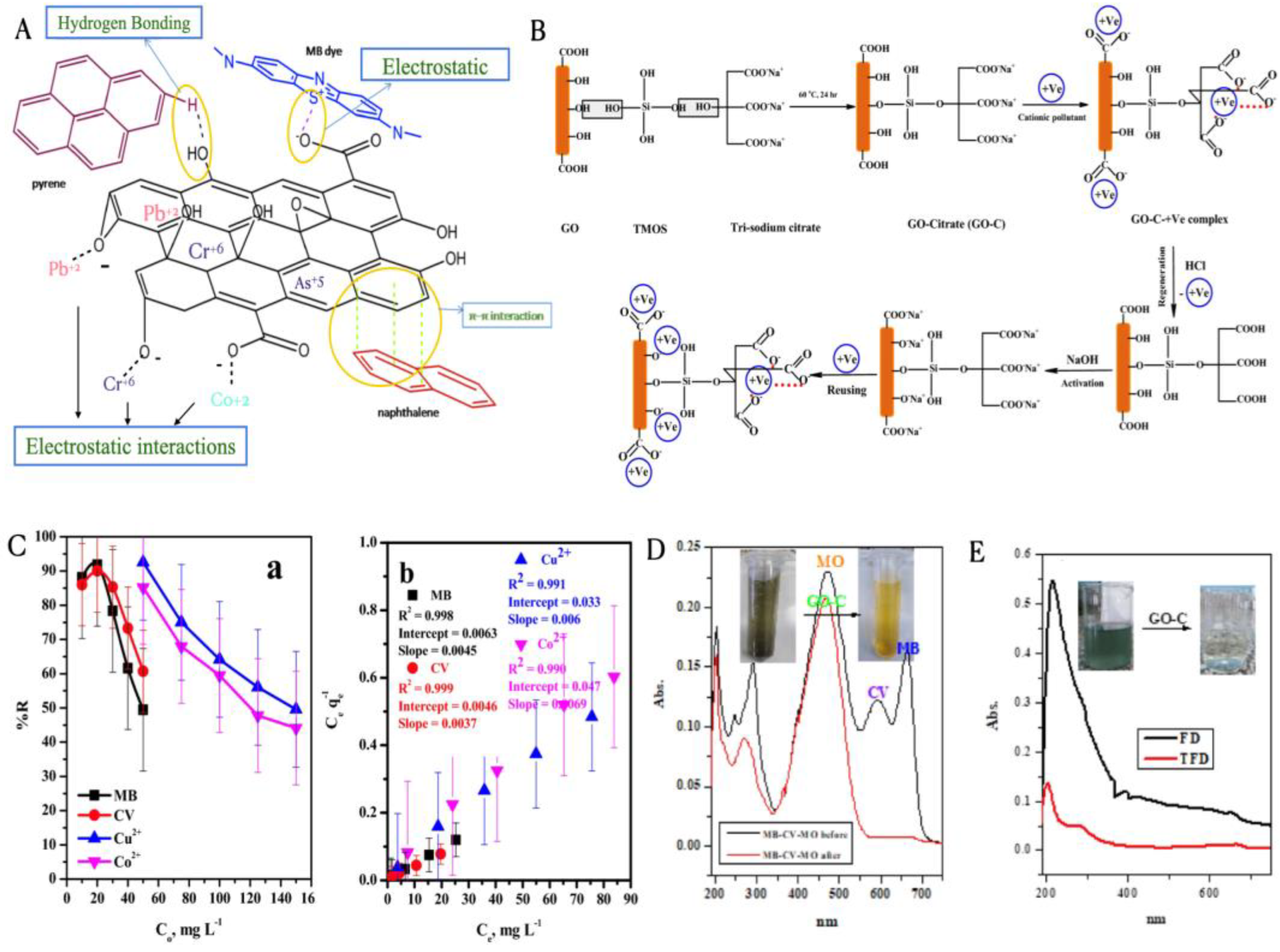
3.1.1. Adsorption
- 1)
- qe / qm =KL Ce / 1+ KL Ce
- 2)
- qe =KF Ce1/n
- 3)
- qe = BT ln (AT Ce)
- 4)
- qe = qm exp (-beta (Ce) 1/n)
- 5)
- Ce / qe (1-Ce) = C / qm
- qe = Amount of adsorbate adsorbed per unit mass of adsorbent at equilibrium
- qm = Maximum adsorption capacity
- KL = Langmuir constant
- Ce = Adsorbate concentration at equilibrium
- KF = Freundlich constant
- n = Adsorption intensity related Freundlich expression
- AT and BT = Temkin constant
- C = BET constant
3.1.2. Membrane Separation
3.1.3. Photocatalytic Degradation of Wastewater
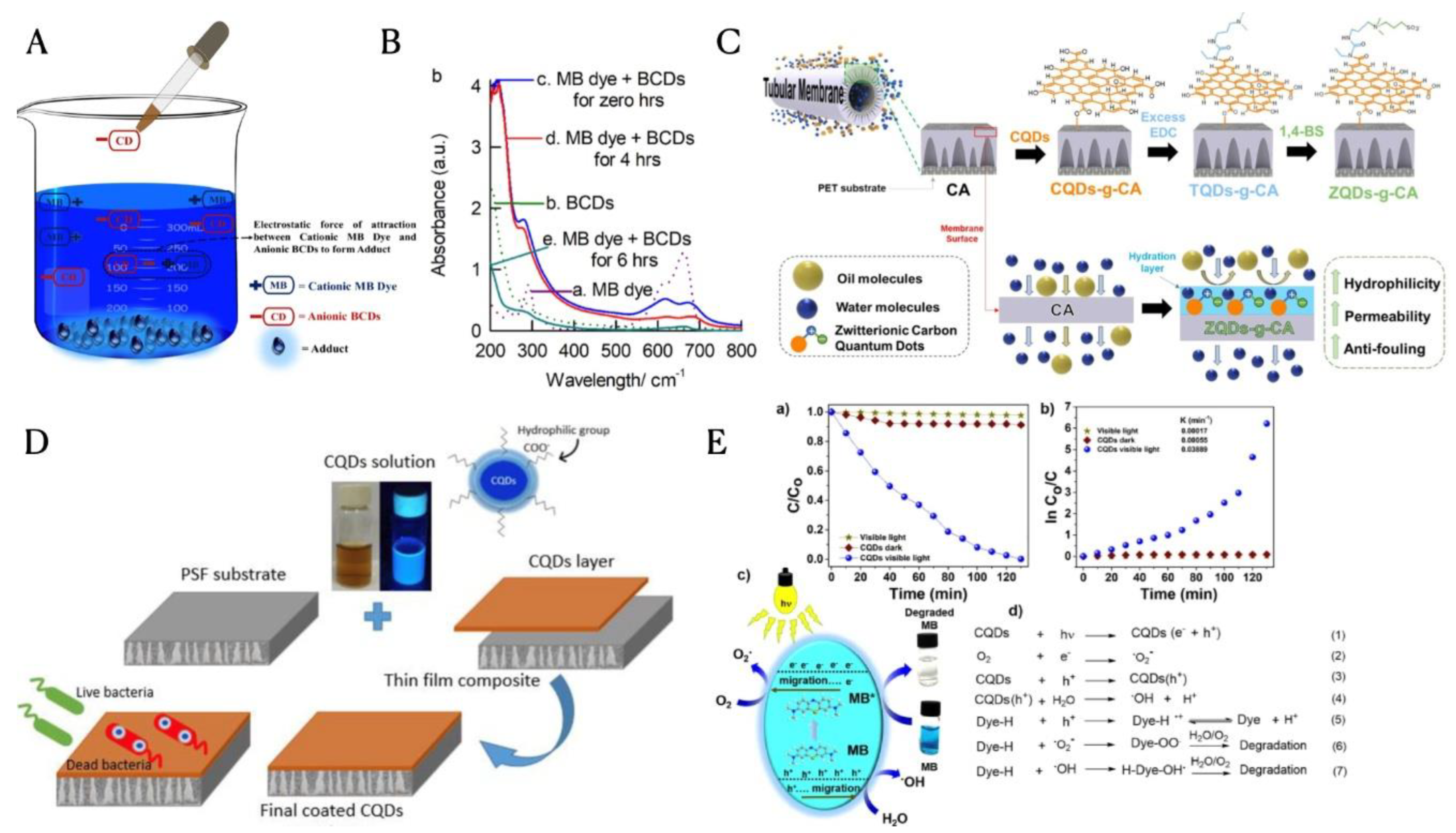
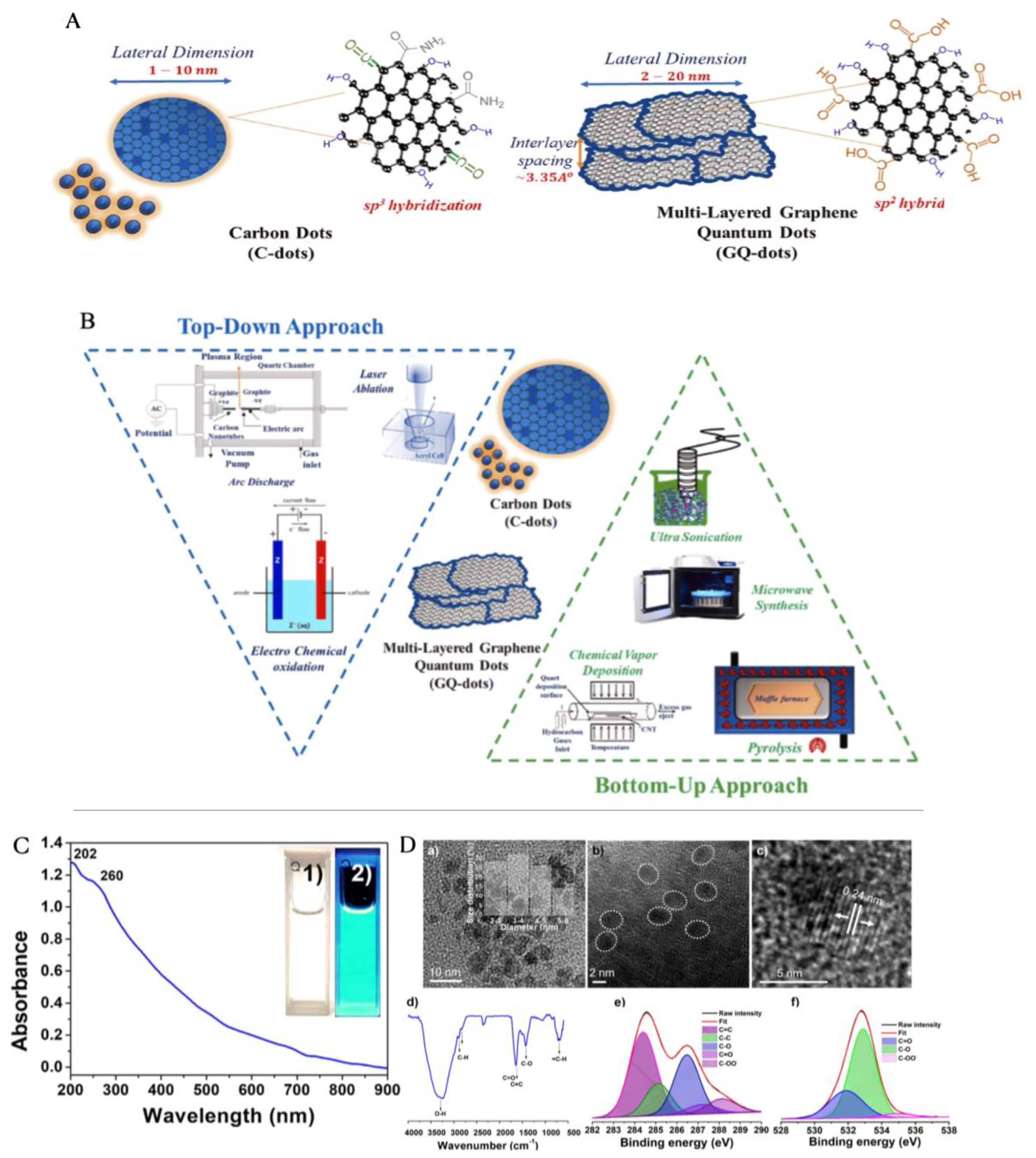
3.2. Carbon Dots for Water Treatment
3.2.1. Adsorption
3.2.2. Membrane Filtration
3.2.3. Photocatalytic Degradation of Wastewater
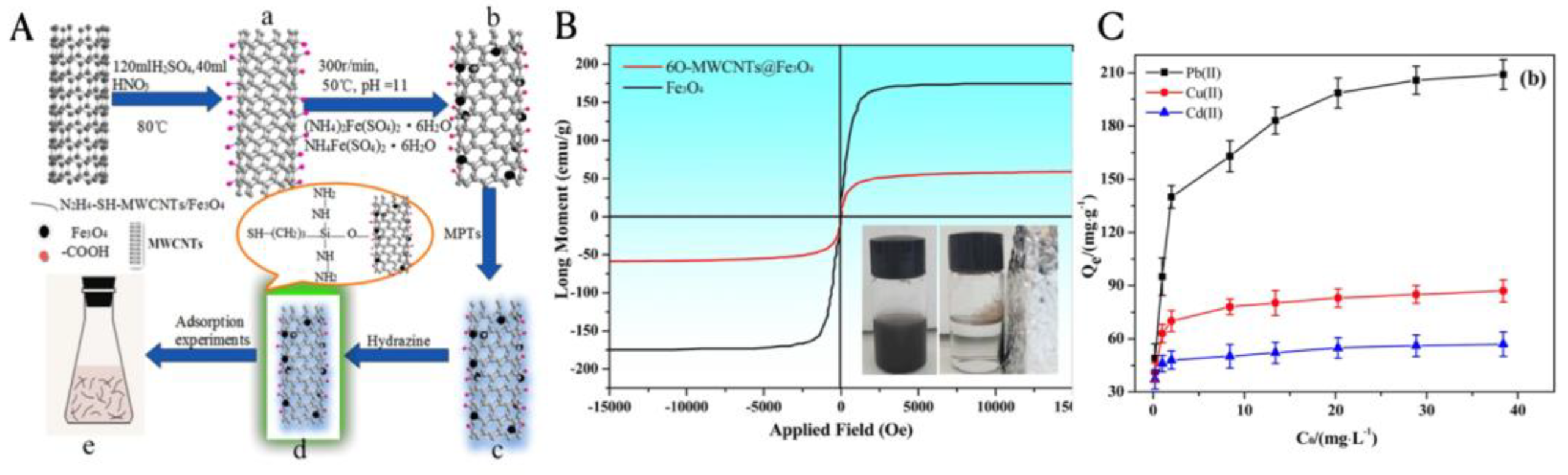
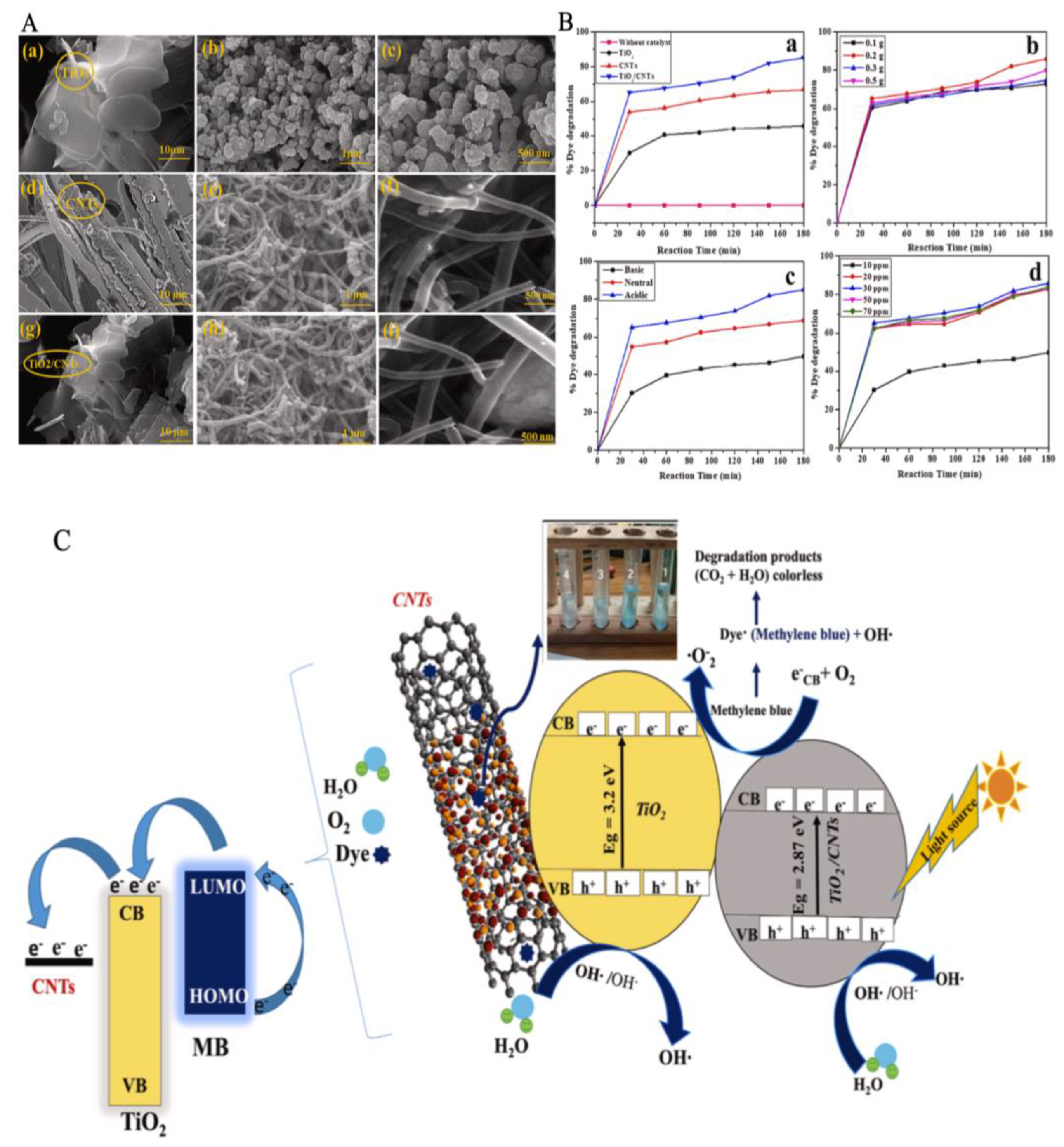
3.3. Carbon Nanotubes for Water Treatment
3.3.1. Adsorption
3.3.2. Membrane Filtration
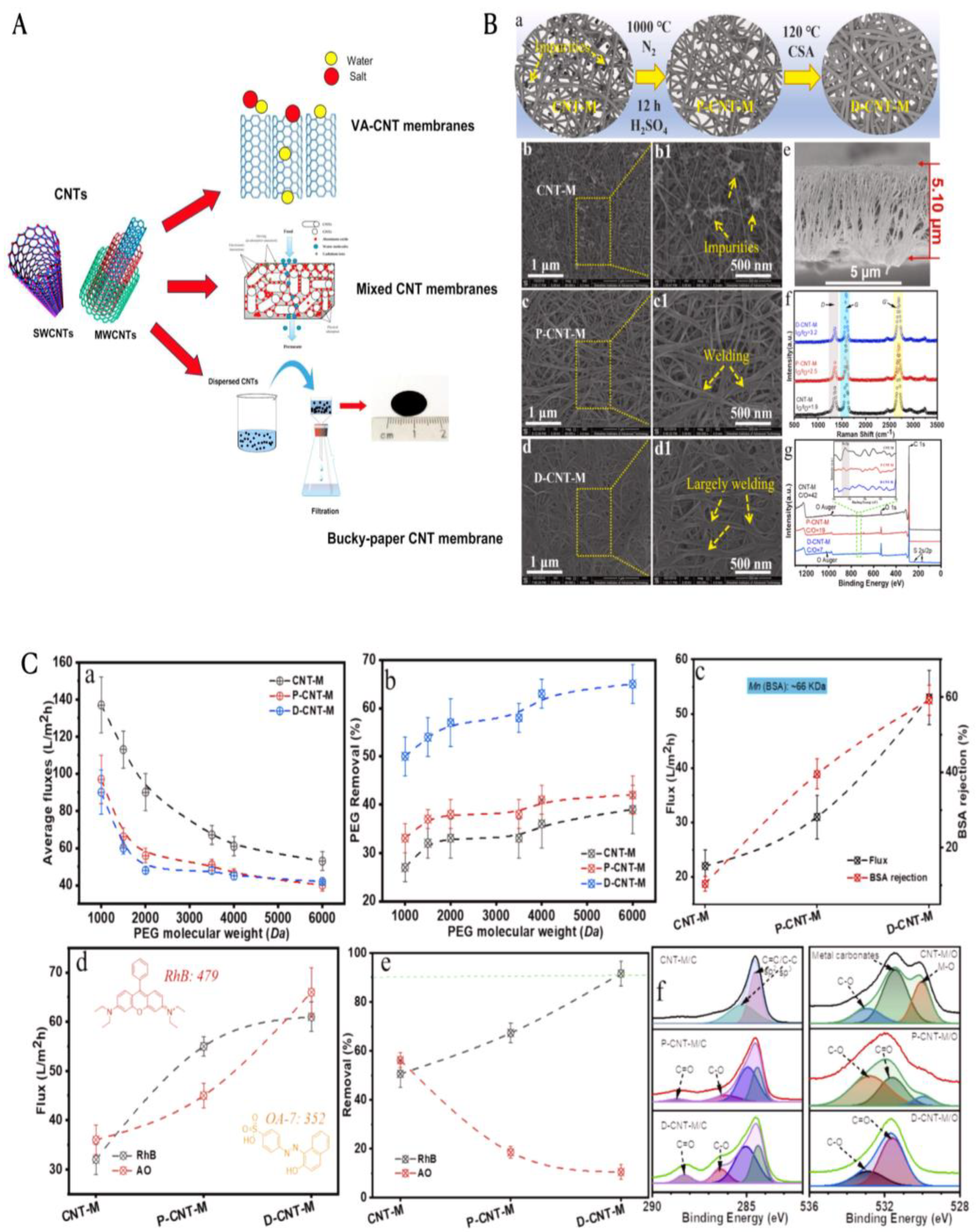
3.3.3. Photocatalytic Degradation of Wastewater
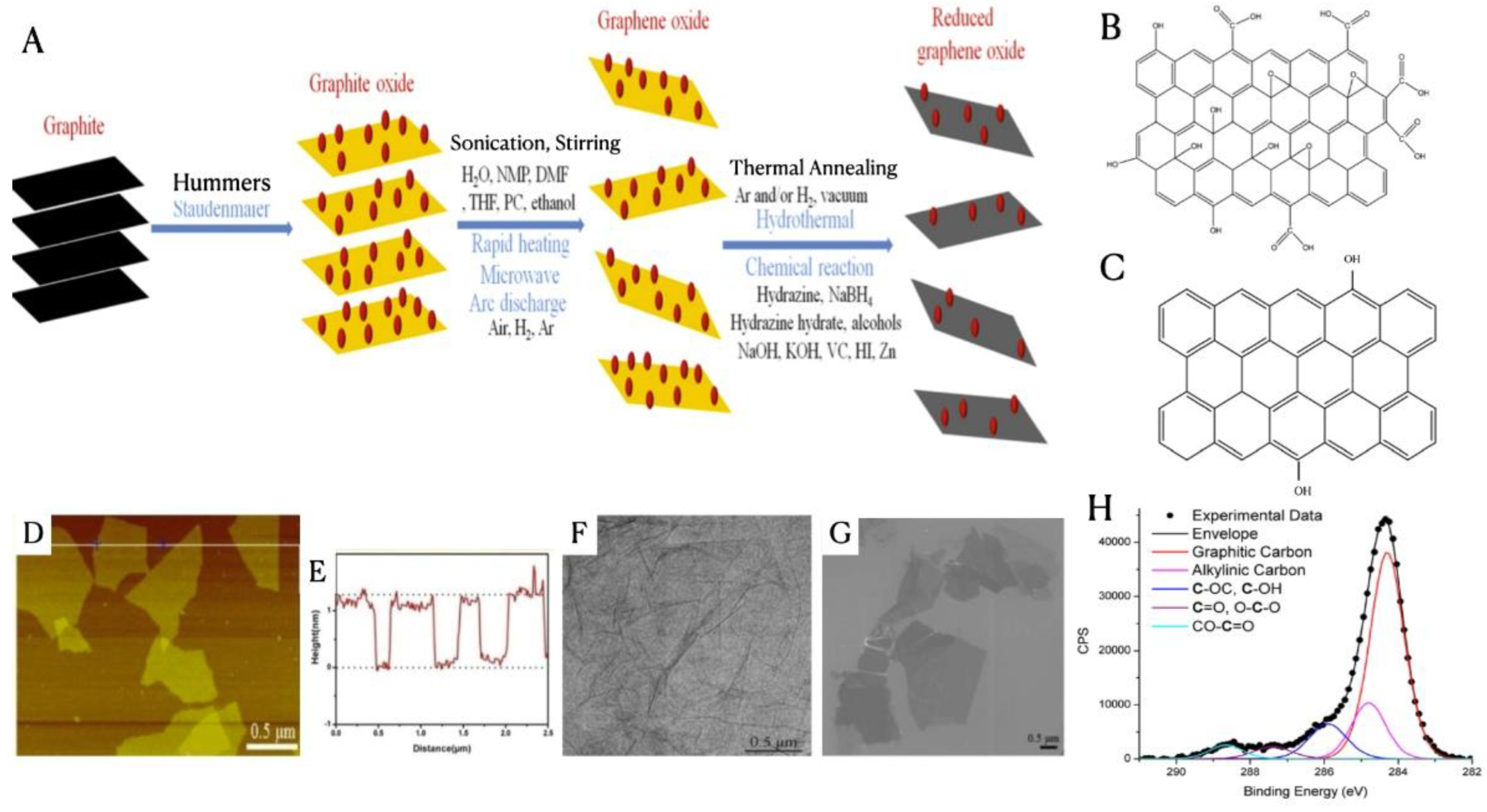
3.4. Graphene-Based Nanomaterials for Water Treatment
3.4.1. Adsorption
3.4.2. Membrane Filtration
3.4.3. Photocatalytic Degradation of Wastewater
4. Antibacterial Activity of Carbon-Based Nanomaterials
5. Comparison of Various CBNs Targeted for Wastewater Treatment
6. Sustainable Practices
6.1. Concerns about CBNs Targeted for Wastewater Treatment
6.2. Systematic Thinking and Precautionary Principle
7. Future Perspective and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barman, S.R.; Gavit, P.; Chowdhury, S.; Chatterjee, K.; and Nain, A. Wastewater Treatment. JACS Au. 2023, 3(11), 2930-2947. [CrossRef]
- UNWWDR. United Nations World Water Development Report 2020. Available online: https://www.unwater.org/publications/un-world-water-development-report-2020 (accessed 2025-04-11).
- World Health Organization, Guidelines for Drinking-Water Quality: Incorporating the First and Second Addenda. World Health Organization, 2022. https://www.who.int/publications/i/item/ 9789241549950 (accessed 2025-04-11).
- Sen, T.K. Agricultural Solid Wastes Based Adsorbent Materials in the Remediation of Heavy Metal Ions from Water and Wastewater by Adsorption: A Review. Molecules 2023, 28(14), 5575. [CrossRef]
- Silva, J.A. Treatment and Reuse for Sustainable Water Resources Management: A Systematic Literature Review. Sustainability 2023, 15(14), 10940. [CrossRef]
- Rashid, R.; Shafiq, I.; Akhter, P.; Iqbal, M.J.; Hussain, M. A state-of-the-art review on wastewater treatment techniques: the effectiveness of adsorption method. Environ. Sci. Pollut. Res. Int. 2021, 28, 9050-9066. [CrossRef]
- Singh, B. J.; Chakraborty, A.; Sehgal, R. A systematic review of industrial wastewater management: Evaluating challenges and enablers. J. Environ. Manag. 2023, 348, 119230. [CrossRef]
- Jassby, D.; Tzahi Y. Cath, T.Y.; Buisson, H. The role of nanotechnology in industrial water treatment. Nat. Nanotechnol. 2018, 13, 670-672. [CrossRef]
- Jain,K.; Patel,A.S.; Vishwas P. Pardhi,V.P.; Flora,S.J.S. Nanotechnology in Wastewater Management: A New Paradigm Towards Wastewater Treatment. Molecules 2021, 26(6), 1797. [CrossRef]
- Devi, M.K.; Yashika, P.R.; Kumar, P.S.; Manikandan, S.; Oviyapriya, M.; V. Varshikaa, V.; Rangasamy, G. Recent advances in carbon-based nanomaterials for the treatment of toxic inorganic pollutants in wastewater. New J. Chem. 2023, 47, 7655-7667. [CrossRef]
- Abbo, H.S.; Gupta, K.C.; Khaligh, N.G.; Titinchi, S.J.J. Carbon Nanomaterials for Wastewater Treatment. ChemBioEng Rev 2021, 8(5), 1-28. [CrossRef]
- Liu, Y.; Phillips, B.; Li, W.; Zhang, Z.; Fang, L.; Jingjing Qiu, J.; Wang, S. Fullerene-Tailored Graphene Oxide Interlayer Spacing for Energy-Efficient Water Desalination. ACS Applied Nano Materials 2018, 1(11), 6168-6175. [CrossRef]
- Mishra, S.; Sundaram, B. Efficacy and challenges of carbon nanotube in wastewater and water treatment, Environ. Nanotechnol. Monit. Manag. 2023, 19, 100764. [CrossRef]
- Tabish, T.A.; Memon, F.A.; Gomez, D.E.; David W. Horsell, D.W.; Zhang, S. A facile synthesis of porous graphene for efficient water and wastewater treatment, Sci. Rep. 2018, 1817. [CrossRef]
- Asghar, F.; Shakoor, B.; Fatima, S.; Munir, S.; Razzaq, H.; Shazia Naheeda, S.; Butler, I.S. Fabrication and prospective applications of graphene oxide-modified nanocomposites for wastewater remediation, RSC Adv., 2022,12, 11750-11768. [CrossRef]
- Kaur, H.; Devi, N.; Siwal, S.S.; Alsanie, W.F.; Thakur, M.K.; Thakur, V.K. Metal−Organic Framework-Based Materials for Wastewater Treatment: Superior Adsorbent Materials for the Removal of Hazardous Pollutants. ACS Omega 2023, 8, 9004-9030. [CrossRef]
- Kesari, K.K.; Soni, R.; Jamal, Q.M.S.; Tripathi, P.; Lal, J.A.; Jha, N.K.et al. Wastewater Treatment and Reuse: a Review of its Applications and Health Implications. Water Air Soil Pollut 2031, 232, 208. [CrossRef]
- Naseem, T.; Durrani, T. The role of some important metal oxide nanoparticles for wastewater and antibacterial applications: A review, J. Environ. Chem. Ecotoxicol. 2021, 3. 59-75. [CrossRef]
- Silva, J.A. Wastewater Treatment and Reuse for Sustainable Water Resources Management: A Systematic Literature Review, Sustainability 2023, 15, 10940. [CrossRef]
- Wastewater Treatment Overview, avengtraining. https://arvengtraining.com/en/waste-water-treatment-overview/ (accessed 2025-04-11).
- Lee, J.D. (1996) Concise Inorganic Chemistry. 5th Edition, Chapman and Hall Ltd., London, 619-621.
- Gollavelli, G.; Ghule, A.V.; Ling, Y.-C. Multimodal Imaging and Phototherapy of Cancer and Bacterial Infection by Graphene and Related Nanocomposites. Molecules 2022, 27, 5588. [CrossRef]
- Gollavelli, G.; Gedda, G.; Mohan, R.; Ling, Y.-C. Status Quo on Graphene Electrode Catalysts for Improved Oxygen Reduction and Evolution Reactions in Li-Air Batteries. Molecules 2022, 27, 7851. [CrossRef]
- Çeçen, F.; Aktaş, Ö. (2011). Water and Wastewater Treatment: Historical Perspective of Activated Carbon Adsorption and its Integration with Biological Processes. In Activated Carbon for Water and Wastewater Treatment (eds F. Çeçen and Ö. Aktaş).
- Kroto, H.; Heath, J.; O’Brien, S.; Curl, R.F.; Smalley, R,E. C60: Buckminsterfullerene. Nature 1985, 318, 162-163 . [CrossRef]
- Discovery of Fullerenes, National Historic Chemical Landmark, Designated October 11, 2010, at the Richard E. Smalley Institute for Nanoscale Science and Technology at Rice University in Houston, Texas.
- Boo, W.O.J. An Introduction to Fullerene Structures Geometry and Symmetry, Journal of Chemical Education, J. Chem. Educ. 1992, 69(8), 605. [CrossRef]
- Poulomi, B.; Bhavna, B. Fullerene and its applications: A review. J INDIAN ACAD ORAL M. 2020, 32(2), 159-163. [CrossRef]
- Kumar, P.A.; Namboodiri, V.V.; Joshi, G.; Mehta, K.P. Fabrication and applications of fullerene-based metal nanocomposites: A review. J. Mater. Res. 2021, 36, 114-128. [CrossRef]
- Dubey, R.; Dutta, D.; Sarkar, A.; Chattopadhyay, P. Functionalized carbon nanotubes: synthesis, properties and applications in water purification, drug delivery, and material and biomedical sciences, Nanoscale Adv., 2021, 3, 5722-5744. [CrossRef]
- Gacem, M.; Modi, S.; Yadav, K.V.; Islam, S.; Patel. A.; Dawane. V.; Jameel. M.; Inwati, K. G.; Piplode, S.; Solanki, S. V.; Basnet, A. Recent Advances in Methods for Synthesis of Carbon Nanotubes and Carbon Nanocomposite and their Emerging Applications: A Descriptive Review. J. Nanomater. 2022(1), 7238602. https://onlinelibrary.wiley.com/doi/10.1155/2022/7238602. [CrossRef]
- Balkanloo, P.G.; Sharifi, K.M.; Marjani, A.P. Graphene quantum dots: synthesis, characterization, and application in wastewater treatment: a review. Mater. Adv., 2023, 4, 4272-4293. [CrossRef]
- Deepa, C.; Rajeshkumar, L.; Ramesh, M. Preparation, synthesis, properties and characterization of graphene-based 2D nano-materials for biosensors and bioelectronics. J Mater Res Technol, 2022, 19, 2657-2694. [CrossRef]
- Cheng, X.; Kan, A.T.; Tomson, M.B. Uptake and Sequestration of Naphthalene and 1,2-Dichlorobenzene by C60. J Nanopart Res 2005, 7, 555-567. [CrossRef]
- Ballesteros, E.; Gallego, M.; Valcárcel, M. Analytical potential of fullerene as adsorbent for organic and organometallic compounds from aqueous solutions, J. Chromatogr. A 2000, 869, 101-110. [CrossRef]
- Alekseeva, O.V.; Bagrovskaya, N.A.; Noskov, A.V., Sorption of heavy metal ions by fullerene and polystyrene/fullerene film composite. Prot Met Phys Chem Surf 2016, 52(3), 443-447. [CrossRef]
- Samonin, V.; Nikonova, V.; Podvyaznikov, M. Sorption properties of activated carbon with respect to metal ions. Protection of Metals 2014, 44, 190-192. [CrossRef]
- Samonin, V.; Nikonova, V.; Podvyaznikov, M, 2014. Carbon adsorbents on the basis of the hydrolytic lignin modified with fullerenes in producing. Russ. J. Appl. Chem. 2014, 87(2), 190-193. [CrossRef]
- Stylianakis, M.M. Distinguished Contributions in the Fields of Biomedical and Environmental Applications Incorporating Nanostructured Materials and Composites. Molecules 2021, 26, 2112. [CrossRef]
- Sudareva, N.; Penkova, A.; Kostereva, T.; Polotskii, A.; Polotskaya, G. Properties of casting solutions and ultrafiltration membranes based on fullerene-polyamide nanocomposites. Express Polym. Lett. 2012, 6, 178-188. [CrossRef]
- Semenov, K.N.; Andrusenko, E.V.; Charykov, N.A.; Litasova, E.V.; Panova, G.G.; Penkova, A.V.; Murin, I.V.; Piotrovskiy, L.B. Carboxylated fullerenes: Physico-chemical properties and potential applications. Prog. Solid State Chem. 2017, 47-48, 19-36. [CrossRef]
- Yan, L.; Zhao, F.; Li, S.; Hu, Z.; Zhao, Y. Low-toxic and safe nanomaterials by surface-chemical design, carbon nanotubes, fullerenes, metallofullerenes, and graphenes. Nanoscale 2011, 3, 362-382. [CrossRef]
- Brunet, L.; Lyon, D.Y.; Hotze, E.M.; Alvarez, P.J.J.; Wiesner, M.R. Comparative Photoactivity and Antibacterial Properties of C60 Fullerenes and Titanium Dioxide Nanoparticles. Environ. Sci. Technol. 2009, 43(12), 4355-4360. [CrossRef]
- Zhang, B.-T.; Zheng, X.; Li, H.-F.; Lin, J.-M. Application of carbon-based nanomaterials in sample preparation: A review. Anal. Chim. Acta 2013, 784, 1-17. [CrossRef]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2018, 148, 702-712. [CrossRef]
- Yashas, S.R.; Shahmoradi, B.; Wantala, K.; Shivaraju, H.P. Potentiality of polymer nanocomposites for sustainable environmental applications: A review of recent advances. Polymer 2021, 233, 124184. [CrossRef]
- Shen, Q.; Xu, S.J.; Xu, Z.L.; Zhang, H.Z.; Dong, Z.Q. Novel thin-film nanocomposite membrane with water-soluble polyhydroxylated fullerene for the separation of Mg2+/Li+ aqueous solution. J. Appl. Polym. Sci. 2019, 136, 48029. [CrossRef]
- Li, Y.; He, G.; Wang, S.; Yu, S.; Pan, F.; Wu, H.; Jiang, Z. Recent advances in the fabrication of advanced composite membranes. J. Mater. Chem. A 2013, 1, 10058-10077. [CrossRef]
- Tan, X.F.; Liu, Y.G.; Gu, Y.L.; Xu, Y.; Zeng, G.M.; Hu, X.J.; Liu, S.B.; Wang, X.; Liu, S.M.; Li, J. Biochar-based nano-composites for the decontamination of wastewater: A review. Bioresour. Technol. 2016, 212, 318-333. [CrossRef]
- Plisko, T.V.; Liubimova, A.S.; Bildyukevich, A.V.; Penkova, A.V.; Dmitrenko, M.E.; Mikhailovskii, V.Y.; Melnikova, G.B.; Semenov, K.N.; Doroshkevich, N.V.; Kuzminova, A.I. Fabrication and characterization of polyamide-fullerenol thin film nanocomposite hollow fiber membranes with enhanced antifouling performance. J. Memb. Sci. 2018, 551, 20-36. [CrossRef]
- Dmitrenko, M.; Penkova, A.; Kuzminova, A.; Atta, R.; Zolotarev, A.; Mazur, A.; Vezo, O.; Lahderanta, E.; Markelov, D.; Ermakov, S. Development and investigation of novel polyphenylene isophthalamide pervaporation membranes modified with various fullerene derivatives. Sep. Purif. Technol. 2019, 226, 241-251. [CrossRef]
- Kausar, A.; Ahmad, I.; Maaza, M.; Eisa, M.H. State-of-the-Art of Polymer/Fullerene C60 Nanocomposite Membranes for Water Treatment: Conceptions, Structural Diversity and Topographies. Membranes 2023, 13, 27. [CrossRef]
- Penkova, A.V.; Dmitrenko, M.E.; Sokolova, M.P.; Chen, B.; Plisko, T.V.; Markelov, D.A.; Ermakov, S.S. Impact of fullerene loading on the structure and transport properties of polysulfone mixed-matrix membranes. J. Mater. Sci. 2016, 51, 7652-7659. [CrossRef]
- Ranjbar, T.; Akbarzadeh, H.; Esmat Mehrjouei, E.; Abbaspour, M.; Salemi, S.; Yaghoubi, H. Molecular insight into C60-grafted graphene oxide as a novel reverse osmosis membrane with low energy consumption for seawater desalination. Desalination 2022, 542, 116062. [CrossRef]
- Liu, Y.; Phillips, B.; Li, Wei.; Zhang, Z.; Fang, L.; Qiu, J.; Wang, S. Fullerene-Tailored Graphene Oxide Interlayer Spacing for Energy-Efficient Water Desalination, ACS Appl. Nano Mater. 2018, 1(11), 6168-6175. [CrossRef]
- Liu, Q.; Guo, C.; Yang, Z.; Yao, H.; Hu, J.; Liu, G.; Jin, W. Enabling efficient water desalination and mitigating membrane fouling by the novel two-dimensional fullerene with unlocking its electrostatic forces. J. Memb. Sci. 22023, 687, 122074. [CrossRef]
- Jani, M.; Arcos-Pareja, J.A.; Ni, M. Engineered Zero-Dimensional Fullerene/Carbon Dots-Polymer Based Nanocomposite Membranes for Wastewater Treatment. Molecules 2020, 25, 4934. [CrossRef]
- Rabenau, T.; Simon, A.; Kremer, R.K.; Sohmen, E. The energy gaps of fullerene C60 and C70 determined from the temperature dependent microwave conductivity. Z. Phys. B:Condens. Matter. 1993, 90, 69-72. [CrossRef]
- Pan, Y.; Liu, X.; Zhang, W.; Liu, Z.; Zeng, G.; Shao, B.; Liang, Q.; He, Q.; Yuan, X.; Huang, D.; Chen, M. Advances in photocatalysis based on fullerene C60 and its derivatives: Properties, mechanism, synthesis, and applications. Appl. Catal. B: Environ. 2020, 265, 118579. [CrossRef]
- Peng, B. Monolayer Fullerene Networks as Photocatalysts for Overall Water Splitting, J. Am. Chem. Soc. 2022, 144(43), 19921-9931. [CrossRef]
- Krishnan, A.; Swarnalal, A.; Das, D.; Krishnan, M.; Saji, V.S.; Shibli, S.M.A. A review on transition metal oxides based photocatalysts for degradation of synthetic organic pollutants. J. Environ. Sci. 2024, 139, 389-417. [CrossRef]
- Yao, S.; Yuan, X.; Jiang, L.; Xiong, T.; Zhang, J. Recent Progress on Fullerene-Based Materials: Synthesis, Properties, Modifications, and Photocatalytic Applications. Materials 2020, 13(13), 2924. [CrossRef]
- Chronopoulos, D.D.; Otyepka, M.; Kokotos, C.G. Photochemical Carbocatalysis: Fullerene-, Carbon Nanotube- or Graphene-Based Metal-Free Photocatalysts for Organic Transformations. ChemCatChem 2024, 16(14), e202400042. [CrossRef]
- Othman, Z.; Sinopoli, A.; Mackey, HR.; Mahmoud, K.A. Efficient Photocatalytic Degradation of Organic Dyes by AgNPs/TiO2/Ti3C2Tx MXene Composites under UV and Solar Light. ACS Omega 2021, 6(49), 33325-33338. [CrossRef]
- Lee, S.Y.; Kang, D.; Jeong, S.; Do, H.T.; Kim, J.H. Photocatalytic Degradation of Rhodamine B Dye by TiO2 and Gold Nanoparticles Supported on a Floating Porous Polydimethylsiloxane Sponge under Ultraviolet and Visible Light Irradiation, ACS Omega 2020, 5(8), 4233-4241. [CrossRef]
- Zhang, X.; Wang, Q.; Zou, L-H.; You, Z-W. Facile fabrication of titanium dioxide/fullerene nanocomposite and its enhanced visible photocatalytic activity, J. Colloid Interface Sci. 2016, 466, 56-61. [CrossRef]
- Lim, J.; Monllor-Satoca, D.; Jang, J.S.; Lee, S.; Choi, W. Visible light photocatalysis of fullerol-complexed TiO2 enhanced by Nb doping. Appl. Catal. B 2014(152-153), 233-240. [CrossRef]
- Krishna, V.; Noguchi, N.; Koopman, B.; Moudgil, B. Enhancement of titanium dioxide photocatalysis by water-soluble fullerenes. J. Colloid Interface Sci. 2006, 304(1), 166-171. [CrossRef]
- Youssef, Z.; Colombeau, L.; Yesmurzayeva, N.; Baros, F.; Vanderesse, R.; Hamieh, T.; Toufaily, J.; Frochot, C.; Roques-Carmes, T.; Acherar, S. Dye-sensitized nanoparticles for heterogeneous photocatalysis: Cases studies with TiO2, ZnO, fullerene and graphene for water purification, Dyes and Pigments, 2018, 159, 49-71. [CrossRef]
- Fu,H.; Xu,T.; Zhu,S.;†Zhu,Y. Photocorrosion Inhibition and Enhancement of Photocatalytic Activity for ZnO via Hybridization with C60, Environ. Sci. Technol. 2008, 42(21), 8064-8069. [CrossRef]
- Behera, A.; Mansingh, S.; Das, K.K.; Parida, K. Synergistic ZnFe2O4-carbon allotropes nanocomposite photocatalyst for norfloxacin degradation and Cr (VI) reduction. J. Colloid Interface Sci. 2019, 544, 96-111. [CrossRef]
- Tahir, M.B.; Nabi, G.; Rafique, M.; Khalid, NR. Role of fullerene to improve the WO3 performance for photocatalytic applications and hydrogen evolution. Int J Energy Res. 2018, 42(15), 4783-4789. [CrossRef]
- Parambil, A.M.; Rajan, S.; Huang, P.-C.; Shashikumar, U.; Tsai, P.-C.; Rajamani, P.; Lin, Y.-C.; Ponnusamy, V.K. Carbon and Graphene Quantum Dots Based Architectonics for Efficient Aqueous Decontamination by Adsorption Chromatography Technique - Current State and Prospects. Environ. Res. 2024, 251(Part 1), 118541. [CrossRef]
- Das, G.S.; Shim, J.P.; Bhatnagar, A.; Tripathi, K.M.; Kim, T.Y. Biomass-Derived Carbon Quantum Dots for Visible-Light-Induced Photocatalysis and Label-Free Detection of Fe(III) and Ascorbic Acid. Sci. Rep. 2019, 9, 15084. [CrossRef]
- Gorle, G.; Gollavelli, G.; Nelli, G.; Ling, Y.-C. Green Synthesis of Blue-Emitting Graphene Oxide Quantum Dots for In Vitro CT26 and In Vivo Zebrafish Nano-Imaging as Diagnostic Probes. Pharmaceutics 2023, 15(2), 632. [CrossRef]
- Zulfajri, M.; Gedda, G.; Ulla, H.; Habibati.; Gollavelli, G.; Huang, G.G. A review on the chemical and biological sensing applications of silver/carbon dots nanocomposites with their interaction mechanisms. Advances in Colloid and Interface Science, 2024, 325, 103115. [CrossRef]
- Bhattacharjee, T.; Konwar, A.; Boruah, J.S.; Chowdhury, D.; Majumdar, G. A sustainable approach for heavy metal remediation from water using carbon dot based composites: A review, J HAZARD MATER ADV, 2023, 10, 100295. [CrossRef]
- Hussen, N.H. Hasan, H.; Khedr, Y,M.F.; Bogoyavlenskiy, A.; Bhat, A.R,; Jamalis, J. Carbon Dot Based Carbon Nanoparticles as Potent Antimicrobial, Antiviral, and Anticancer Agents, ACS Omega 2024, 9(9), 9849-9864. [CrossRef]
- Sabet, M.; Mahdavi, K. Green synthesis of high photoluminescence nitrogen-doped carbon quantum dots from grass via a simple hydrothermal method for removing organic and inorganic water pollutions, Appl. Surf. Sci. 2019, 463, 283-291. [CrossRef]
- Rani, U.A.; Ng, L.Y.; Ng, C.Y.; Mahmoudi, E. A review of carbon quantum dots and their applications in wastewater treatment, Adv. Colloid and Interface Sci. 2020, 278, 102124. [CrossRef]
- Mahmoud, M.E.; Fekry, N.A.; Abdelfattah, A.M. Removal of uranium (VI) from water by the action of microwave-rapid green synthesized carbon quantum dots from starch-water system and supported onto polymeric matrix, J. Hazard. Mater. 2020, 397, 122770. [CrossRef]
- Meng, Y.; Xiao, L.; Muslim, A. Hojiahmat, M. Improving the adsorption of poly(o-phenylenediamine) to heavy metal ions in aqueous solution through its composite with carbon dots. J Polym Res 2021, 28, 404. [CrossRef]
- Perumal, S.; Atchudan, R.; Thirukumaran, P.; Dong Ho Yoon, D.H.; Yong Rok Lee, Y.R. In Woo Cheong, Simultaneous removal of heavy metal ions using carbon dots-doped hydrogel particles, Chemosphere 2022, 286, 131760. [CrossRef]
- Li, J.; Wang, L.; Jiang, G.; Wan, Y.; Wang, J.; Li, Y.; Pi, F. Luminescent carbon dots-rooted polysaccharide crosslinked hydrogel adsorbent for sensitive determination and efficient removal of Cu2+. Food Chemistry, 447, 2024, 138977. [CrossRef]
- Li, M.; Zhang, P.; Mao, J .; Li, J.; Zhang, Y.; Xu, B.; Zhou, J.; Cao, Q.; Xiao, H. Construction of cellulose-based hybrid hydrogel beads containing carbon dots and their high performance in the adsorption and detection of mercury ions in water. J. Environ. Manage. 2024, 359, 121076. [CrossRef]
- Wei, T.; Ni, H.; Ren, X.; Zhou, W.; Gao, H.; Hu, S. Fabrication of nitrogen-doped carbon dots biomass composite hydrogel for adsorption of Cu (II) in wastewater or soil and DFT simulation for adsorption mechanism. Chemosphere 2024, 361, 142432. [CrossRef]
- You, X-Y.; Yin, W-M.; Wang, Y.; Wang, C.; Zheng, W-X.; Guo, Y -R.; Li, S.; Pan, Q-J. Enrichment and immobilization of heavy metal ions from wastewater by nanocellulose/carbon dots-derived composite. Int. J. Bio. Macromol. 2024, 255, 128274. [CrossRef]
- Thakur, A.; Kumar, A.; Ambrish Singh, A. Adsorptive removal of heavy metals, dyes, and pharmaceuticals: Carbon-based nanomaterials in focus. Carbon, 2024, 217, 118621. [CrossRef]
- Mahmoud, M.E.; Fekry, N.A.; Abdelfattah, A.M. Engineering nanocomposite of graphene quantum dots/carbon foam/alginate/zinc oxide beads for efficacious removal of lead and methylene, J. Ind. Eng. Chem. 2022, 115, 365-377. [CrossRef]
- Parambil, A.M.; Rajan, S.; Huang, P-C.; Shashikumar, U.; Tsai, P-C.; Rajamani, P.; Lin, Y-C.; Ponnusamy, V.K.; Carbon and graphene quantum dots based architectonics for efficient aqueous decontamination by adsorption chromatography technique - Current state and prospects, Environ. Res. 2024, 251( Part 1), 118541. [CrossRef]
- Fiorentini, E.F.; Rodríguez,E.M.V.; Bonilla-Petriciolet, A.; Escudero, L.B. Carbon Dots in Organic Pollutant Removal, Carbon Dots: Recent Developments and Future Perspectives, Chapter 12, pp 259-275. ACS Symposium Series Vol. 1465, eISBN: 9780841296992. [CrossRef]
- Manikandan, V.; Lee, N.Y. Green synthesis of carbon quantum dots and their environmental applications, Environ Res. 2022, 212(Pt B), 113283. [CrossRef]
- Waseem, B,Z.; Muniraj, S.; Kumar, A.S. Neem biomass derived carbon quantum dots synthesized via one step ultrasonification method for ecofriendly methylene blue dye removal. Sci Rep 2024, 14, 9706. [CrossRef]
- Lu, S.; Yuan, G.; Zhu, Y.; Yu, S. Carbon dots crosslinked chitosan/cellulose sponge capture of methyl blue by an adsorption process, Luminescence 2021, 36(6), 1459. [CrossRef]
- Hashemzadeh, F.; Khoshmardan, M.E.; Sanaei, D.; Ghalhari, M.R.; Sharifan, H.; Inglezakis, V.J.; Arcibar-Orozco, J.A.; Shaikh, W.A.; Khan, E.; Biswas, J.K. Adsorptive removal of anthracene from water by biochar derived amphiphilic carbon dots decorated with chitosan, Chemosphere 352, 2024, 141248. [CrossRef]
- Yu, S.; S. Lu, S.; Zheng, G. Reusable flexible poly(vinyl alcohol)/chitosan-based polymer carbon dots composite film for acid blue 93 dye adsorption, Luminescence 2023, 38(9), 1552. [CrossRef]
- Teymoorian, T.; Hashemi, N.; Mousazadeh, M.H. Entezarian, Z. N. S doped carbon quantum dots inside mesoporous silica for effective adsorption of methylene blue dye. SN Appl. Sci. 2021, 3, 305. [CrossRef]
- Gallareta-Olivares, G.; Rivas-Sanchez, A.; Cruz-Cruz, A.; Hussain, S.M.; González-González, R.B.; Cárdenas-Alcaide, M.F.; Iqbal, H.M.N.; Parra-Saldívar, R. Metal-doped carbon dots as robust nanomaterials for the monitoring and degradation of water pollutants. Chemosphere 2023, 312(Part 1), 137190. [CrossRef]
- Parambil, A.M.; Priyadarshini, E.; Goutam, R.; Tsai, P-C.; Huang, P-C.; Rajamani, P.; Lin, Y-C.; Ponnusamy, V.K. Self-assembled mesoporous silica decorated with biogenic carbon dot nanospheres hybrid nanomaterial for efficient removal of aqueous Methoxy-DDT via a Short-Bed Adsorption column technique. Environ Res. 2024, 260, 119653. [CrossRef]
- Huang, T-Y.; Lin, Y-F.; Hu, S-R.; Huang, C-C.; Huang, Y-F. Chang H-T. Carbon-dot liposome–based synthesis of gold nanocatalysts for efficient reduction of 4-nitrophenol in wastewater. SUSTAIN MATER TECHNO. 2024, 40, e00896. [CrossRef]
- Nguyen, M.B.; Doan, H.V.; Tan, D.L.H.; Lam, T .D. Advanced g-C3N4 and bimetallic FeNi-BTC integration with carbon quantum dots for removal of microplastics and antibiotics in aqueous environments. J. Environ. Chem. Eng. 2024, 12, 112965. [CrossRef]
- Wang, W-R.; Chen, P-Y.; Deng, J.; Chen, Y.; Liu, H-J. Carbon-dot hydrogels as superior carbonaceous adsorbents for removing perfluorooctane sulfonate from water, J. Chem. Eng. 2022, 435, Part 2, 13502. [CrossRef]
- Hellal, A.; Abdelsalam, H.; Tawfik, W. Ibrahim. M.A. Removal of Atrazine from contaminated water by functionalized graphene quantum dots. Opt Quant Electron 2024, 56, 374. [CrossRef]
- Kausar, A.; Ahmad, I. Graphene quantum dots—Nascent adsorbent nanomaterials for water treatment, Environ Nanotechnol Monit Manag 2024, 21, 100943. [CrossRef]
- Oves, M.; Ansari, M.O.; Iqbal M.I.; Ismail. Graphene quantum dot application in water purification, Editor(s): Mohammad Oves, Khalid Umar, Iqbal M.I. Ismail, Mohamad Nasir Mohamad Ibrahim, In Woodhead Publishing Series in Electronic and Optical Materials, Graphene Quantum Dots, Woodhead Publishing, 2023, 113-132. [CrossRef]
- Wee, S. . -h.; Tye, C. -T., T.; Bhatia, S. Membrane separation process—Pervaporation through zeolite membrane, Sep. Purif. Technol. 2008, 63(3), 500-516. [CrossRef]
- Ezugbe, E.O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10(5), 89. https://doi: 10.3390/membranes10050089.
- Khraisheh, M.; Elhenawy, S.; AlMomani, F.; Al-Ghouti, M.; Hassan, MK.; Hameed, BH. Recent Progress on Nanomaterial-Based Membranes for Water Treatment. Membranes 2021, 11(12), 995; [CrossRef]
- Petukhov, D.I.; Johnson, D.J. Membrane modification with carbon nanomaterials for fouling mitigation: A review. Adv. Colloid Interface Sci. 2024, 327, 103140. [CrossRef]
- Chadha, U.; Selvaraj, S.K.; Thanu, S.V.; Cholapadath, V.; Abraham, A.M.; Zaiyan M,M.; Manoharan, M.; Paramsivam, V. A review of the function of using carbon nanomaterials in membrane filtration for contaminant removal from wastewater. Mater. Res. Express 2022, 9, 012003 . [CrossRef]
- Yuan, Z.; Wu, X.; Jiang, Y.; Li, Y.; Huang, J.; Hao, L.; Zhang,J .; Wang, J. Carbon dots-incorporated composite membrane towards enhanced organic solvent nanofiltration performance. J. Membr. Sci. 2018, 549, 1-11. [CrossRef]
- Koulivand, H.; Shahbazi, A.; Vatanpour, V.; Rahmandoust, M. Development of carbon dot-modified polyethersulfone membranes for enhancement of nanofiltration, permeation and antifouling performance. Sep. Purif. Technol. 2020, 230, 115895. [CrossRef]
- Zhao, H.; Zhang, D.; Sun, H.; Zhao, Y.; Xie, M. Adsorption and detection of heavy metals from aqueous water by PVDF/ATP-CDs composite membrane. Colloids Surf. A: Physicochem. Eng. Asp. 2022, 641, 128573. [CrossRef]
- Carballo, G.V.; Hsu, Y.X.; Yang, H.L.; Lin, H.Y.; Huang, C. C.; Li, C.L.; Leron, R.B.; Tsai, H. A.; Lee, K.R. Novel in-situ zwitterionization of carbon quantum dots on membrane surface for oil/water separation. Sep. Purif. Technol. 2024, 348, 127757. [CrossRef]
- Yang, H.L.; Huang, C.T.; Lin, H.Y.; Chen, Y.H.; Tsai, H.A.; Lee, K.R. Zwitterionic carbon quantum dots incorporated ultrafiltration membrane for efficient removal of copper ion. Sep. Purif. Technol. 2024, 331, 125709. [CrossRef]
- Mahat, N.A.; Shamsudin, S.A.; Jullok, N.; Ma’Radzi, A.H. Carbon quantum dots embedded polysulfone membranes for antibacterial performance in the process of forward osmosis, Desalination 2020, 493, 114618. [CrossRef]
- Gan, J.Y.; Chong, W.C.; Sim, L.C.; Koo, C.H.; Pang, Y.L.; Mahmoudi, E.; Mohammad, A.W. Novel Carbon Quantum Dots/Silver Blended Polysulfone Membrane with Improved Properties and Enhanced Performance in Tartrazine Dye Removal. Membranes 2020, 10, 175. [CrossRef]
- Viscusi, G.; Mottola, S.; Tohamy, HA.S.; Gorrasi, G.; De Marco, I. Design of Cellulose Acetate Electrospun Membranes Loaded with N-doped Carbon Quantum Dots for Water Remediation. In: Mannina, G., Ng, H.Y. (eds) Frontiers in Membrane Technology. IWA-RMTC 2024. Lecture Notes in Civil Engineering, vol 525. Springer, Cham. [CrossRef]
- Tshangana, C.; Muleja, A. Graphene oxide quantum dots membrane: a hybrid filtration-advanced technology system to enhance process of wastewater reclamation. Chem. Pap. 78, 2024, 1317-1333. [CrossRef]
- Zhang, C.; Wei, K.; Zhang, W.; Bai, Y.; Sun, Y.; Gu. J. Graphene Oxide Quantum Dots Incorporated into a Thin Film Nanocomposite Membrane with High Flux and Antifouling Properties for Low-Pressure Nanofiltration. ACS Appl. Mater. Interfaces 2017, 9(12), 11082–11094. https://pubs.acs.org/doi/abs/10.1021/acsami.6b12826.
- Li, J.; Gong, J.L.; Si Qun Tang, S.Q.; Zhou, H.Y.; Liang Xiu Tang, L.X.; Zhao, J. Efficient decolorization performance of graphene quantum dots modulated nanofiltration membranes with tunable pore size for precise molecular sieving, Desalination 2024, 583, 117666. [CrossRef]
- Yi-Fang, M.; Yi-Han, H.; Shu-Heng, H.; Ma, R.; Yi-Ding, M.; Zhi-Hai ,C. Simultaneous regulation of pore size and surface charge of nanofiltration membrane using carbon quantum dots for improved selective separation. Sep. Purif. Technol. 2023, 317, 123870. [CrossRef]
- Zahmatkesh, S.; Bing-Jie, Ni.; Klemeš, J.J.; Bokhari, A.; Hajiaghaei-Keshteli, M. Carbon quantum dots-Ag nanoparticle membrane for preventing emerging contaminants in oil produced water. J. Water Process Eng. 2022, 50, 103309. [CrossRef]
- Jani, M.; Arcos-Pareja, J.A.; Ni, M. Engineered Zero-Dimensional Fullerene/Carbon Dots-Polymer Based Nanocomposite Membranes for Wastewater Treatment. Molecules 2020, 25, 4934. [CrossRef]
- Anand, A.; Unnikrishnan, B.; Ju-Yi Mao, Chin-Jung, Lin.; Jui-Yang, Lai.; Chih-Ching ,H,. Carbon-based low-pressure filtration membrane for the dynamic disruption of bacteria from contaminated water. Water Res. 2022, 212, 118121. [CrossRef]
- Ge, J.; Lian, L.; Wang, X.; Cao, X.; Gao, W.; Lou, D. Coating layered double hydroxides with carbon dots for highly efficient removal of multiple dyes. J. Hazard. Mater. 2022, 424, 127613. [CrossRef]
- Saini, D.; Garg, A.K.; Dalal, C.; Anand, S.R.; Sonkar, S.K.; Sonker, A.K.; Westman, G. Visible-Light-Promoted Photocatalytic Applications of Carbon Dots: A Review. ACS Appl. Nano Mater. 2022, 5, 3087-3109. [CrossRef]
- Ikram, Z.; Azmat, E.; Perviaz, M. Degradation Efficiency of Organic Dyes on CQDs As Photocatalysts: A Review, ACS Omega. ACS Omega. 2024, 9(9), 10017-10029. [CrossRef]
- Saputra, M.A.; Piliang, A.F.R.; Dellyansyah.; Marpongahtun.; Andriayani.; Goei, R.; Ramadhan H.T.S. R.; Gea, S. Synthesis, properties, and utilization of carbon quantum dots as photocatalysts on degradation of organic dyes: A mini review. Catal. Commun. 2024, 187, 106914. [CrossRef]
- Sbacchi, M.; Mamone, M.; Morbiato,L.; Gobbo, P.; Filippini,G.; Prato, M. Shining Light on Carbon Dots: New Opportunities in Photocatalysis, ChemCatChem 2023, 15, e202300667. [CrossRef]
- Tripti, T.; Singh, P.; Rani, N. et al. Carbon dots as potential candidate for photocatalytic treatment of dye wastewater. Environ Sci Pollut Res Int. 2024, 31, 6738-6765. [CrossRef]
- Zhao, F.; Li, X.; Zuo, M.; Liang, Y.; Qin, P.; Wang, H.; Wu ,Z.; Luo, L.; Liu, C.; Leng, L. Preparation of photocatalysts decorated by carbon quantum dots (CQDs) and their applications: A review. J. Environ. Chem. Eng. 2023, 11(2), 109487. [CrossRef]
- Nizam, N.U.M.; Hanafiah, M.M.; Mahmoudi, E. et al. Synthesis of highly fluorescent carbon quantum dots from rubber seed shells for the adsorption and photocatalytic degradation of dyes. Sci Rep 2023, 13, 12777. [CrossRef]
- Meena, S.; Sethi, M.; Saini, S.; Kumar,K.; Saini, P.; Meena, S.; Kashyap, S.; Yadav, M.; Meena, M.L.; Dandia, A.; Nirmal, N.K.; Parewa, V. Molecular surface-dependent light harvesting and photo charge separation in plant-derived carbon quantum dots for visible-light-driven OH radical generation for remediation of aromatic hydrocarbon pollutants and real wastewater. J. Colloid Interface Sci. . 2024, 660, 756-770. [CrossRef]
- Zandipak, R.; Bahramifar, N.; Younesi, H.; Zolfigol, M.A. Electro-photocatalyst effect of N-S-doped carbon dots and covalent organic triazine framework heterostructures for boosting photocatalytic degradation of phenanthrene in water. Chemosphere 2024, 364, 142980. [CrossRef]
- Bhoyar, T.; Saraswat, N.; Jyothirmai, M.V.; Gupta, A.; Malla, S.K.; Park, J.; Vidyasagar, D.; Umare, S.S. Nitrogen-Doped Graphitic Carbon Dots Embedded in Carbon Nitride Scaffolds for Water Decontaminatio. ACS Appl. Nano Mater. 2023, 6(5). 3484-3496. [CrossRef]
- Silva, V.; Louros, V.L.; Silva, C.P.; Tacão, M.; Otero, M.; Calisto, V.; Lima, D.L.D. A solar flow photo-reactor for antibiotic removal from aquaculture effluents using TiO2/carbon quantum dots. Chemosphere 2024, 348, 140723. [CrossRef]
- Choi, N.; Tang, C.; Park, Y.; Du, A.; Ayoko, G.A.; Hwang, Y.; Chae, S. Visible-light-driven photocatalytic degradation of tetracycline using citric acid and lemon juice-derived carbon quantum dots incorporated TiO2 nanocomposites. Sep. Purif. Technol. 2024, 350, 127836. [CrossRef]
- Shafiee, A.; Andrew J.; Carrier, A.J.; Nganou, C.; Ehsan, M.F.; Aibaghi, B.; Oakes, K.D.; Zhang, X. Mechanistic insight into the enhanced photodegradation by black titanium dioxide nanofiber-graphene quantum dot composites. Appl. Surf. Sci. 2023, 636, 157836. [CrossRef]
- Shiwei. Xu.; S. Zhang.; M. Guo.; B. Liu.; Y. Zhang. Electrostatic Attraction Enables Strong Interaction between TiO2 and Colloidal Carbon Quantum Dots for Efficient Visible Light Photocatalytic Degradation of Antibiotics. [CrossRef]
- Xu, Z.; Ma, R.; Zhang, C.; Chen, J .; Fan, J.; Shi, Q. A novel quaternary ammonium structure of carbon dots modified TiO2 for fast reduction of Cr(VI) over a wide pH range under sunlight. J. Chem. Eng. 2024, 489, 151363. [CrossRef]
- Shalini, S.; Sasikala, T.; Tharani, D.; Venkatesh, R.; Muthulingam, S. Novel green CQDs/ZnO binary photocatalyst synthesis for efficient visible light irradiation of organic dye degradation for environmental remediation. J. Mol. Liq. 2024, 410, 125525. [CrossRef]
- Kamel, G.N.; El-Shaheny, R.; Shabana, R.A.; Hassan, A.H.E. Inherent photocatalytic activity of luminescent multi-doped carbon dots manufactured from expired medicine and its application for efficient water remediation and nanosensing. Microchem J. 2024, 201, 110576. [CrossRef]
- Rani, U.A.; Ng, L.Y.; Ng, C.Y.; Mahmoudi, E.; Yee-Sern, Ng.; Mohammad, A.W. Sustainable production of nitrogen-doped carbon quantum dots for photocatalytic degradation of methylene blue and malachite green. J. Water Process Eng. 2021, 40, 101816. [CrossRef]
- Torsello. M.; Ben-Zichri. S.; Pesenti. L.; et al. Carbon Dot/Polylactic Acid Nanofibrous Membranes for Solar-Mediated Oil Absorption/Separation: Performance, Environmental Sustainability, Ecotoxicity and Reusability. Heliyon 2024, 10(4), , e25417. [CrossRef]
- Kausar, A. Carbonaceous nanofillers in polymer matrix, Editor(s): Kausar, A. In Woodhead Publishing Series in Composites Science and Engineering, Polymeric Nanocomposites with Carbonaceous Nanofillers for Aerospace Applications, Woodhead Publishing, 2023, 23-53. [CrossRef]
- Mishra, Y.; Mishra, V.; Chattaraj, A.; Aljabali, A.A.A.; El-Tanani, M.; Farani, M.R.; Huh, Y.S.; Serrano-Aroca, A.; Murtaza , M.; Tambuwala. Carbon nanotube-wastewater treatment nexus: Where are we heading to? Environ. Res. 2023, 238, 117088. [CrossRef]
- Yu, F.; Wu, Y.; Li, X.; Ma ,J. Kinetic and thermodynamic studies of toluene, ethylbenzene, and m-xylene adsorption from aqueous solutions onto KOH-activated multiwalled carbon nanotubes, J. Agric. Food Chem. 2012, 60, 12245-12253. [CrossRef]
- Baby , R.; Saifullah ,B.; Hussein ,MZ. Carbon Nanomaterials for the Treatment of Heavy Metal-Contaminated Water and Environmental Remediation. Nanoscale Res Lett. 2019, 14(1), 341. [CrossRef]
- Ogunsola, S.S.; Oladipo, M.E.; Oladoye, P.O.; Kadhom, M. Carbon nanotubes for sustainable environmental remediation: A critical and comprehensive review. Nano-Structures & Nano-Objects 2024, 37, 101099. [CrossRef]
- Ma, J.; Yu, F.; Zhou, L.; Jin, L.; Yang, M.; Luan, J.; Tang, Y.; Fan, H.; Yuan, Z.; Chen, J. Enhanced adsorptive removal of methyl orange and methylene blue from aqueous solution by alkali-activated multiwalled carbon nanotubes. ACS Appl. Mater. Interfaces 2012, 4, 5749-5760. [CrossRef]
- Rao, R.; Pint C, L.; Islam. A, E.; Weatherup, R.S.; Hofmann, S.; et al. Carbon Nanotubes and Related Nanomaterials: Critical Advances and Challenges for Synthesis toward Mainstream Commercial Applications. ACS Nano 2018, 12(12), 11756-11784. [CrossRef]
- Krishna, R.H.; Chandraprabha, M.N.; Samrat, K.; Murthy, T.P.K.; Manjunatha, C.; Kumar, S.G. Carbon nanotubes and graphene-based materials for adsorptive removal of metal ions – A review on surface functionalization and related adsorption mechanism. Appl. Surf. Sci. Adv. 2023, 16, 100431. [CrossRef]
- Jiang, L.; Li, S.; Yu, H.; Zou, Z.; Hou, X.; Shen, F.; Li, C.; Yao, X. Amino and thiol modified magnetic multi-walled carbon nanotubes for the simultaneous removal of lead, zinc, and phenol from aqueous solutions. Appl. Surf. Sci. 2016, 369, 398-413. [CrossRef]
- Anitha, K.; Namsani, S.; Singh, J.K. Removal of Heavy Metal Ions Using a Functionalized Single-Walled Carbon Nanotube: A Molecular Dynamics Study. J. Phys. Chem. A 2015, 119(30) , 8349-8358. [CrossRef]
- Wang, H. J.; Zhou, A.L.; Peng, F.; Yu, H.; L.F. Chen. Adsorption characteristic of acidified carbon nanotubes for heavy metal Pb(II) in aqueous solution. Mater. Sci. Eng. A. 2007, 466, 201-206. [CrossRef]
- Kończy, k, J.; Żarska, S.; Ciesielski, W. Adsorptive removal of Pb(II) ions from aqueous solutions by multi-walled carbon nanotubes functionalised by selenophosphoryl groups: Kinetic, mechanism, and thermodynamic studies. Colloids Surf. A: Physicochem. Eng. Asp. 2019, 575, 271-282. [CrossRef]
- Gull, M., Hussain, S., Tariq, A. Khalid, Z.; Bibi, F.; Waseem, M. Organic and inorganic acids-functionalized CNTs for the adsorption of Cu2+ and Ni2+ ions from aqueous solution. Chem. Pap. 2023, 77, 6519-6531. [CrossRef]
- Oliveira, A.R.; Correia, A.A.; Rasteiro, M.G. Heavy Metals Removal from Aqueous Solutions by Multiwall Carbon Nanotubes: Effect of MWCNTs Dispersion. Nanomaterials 2021, 11(8), 2082. https://doi: 10.3390/nano11082082.
- Wang, Z.; Xu, W.; Jie, F. Zhao, Z.; Zhou, K.; Liu, H. The selective adsorption performance and mechanism of multiwall magnetic carbon nanotubes for heavy metals in wastewater. Sci Rep 2021, 11, 16878. [CrossRef]
- Alkallas, F.H.; Alghamdi, S.M.; Alsubhe, E.; Albeydani, O.; Elsharkawy, W.B.; Mwafy, E.A. et al. Evaluation of synthesized MWCNTs-COO@Fe3O4 nanocomposite based on laser-assisted method for highly-efficient clean water. Mater. Chem. Phys. 2024, 316, 129093. [CrossRef]
- Tijani, J.O.; Abdulkareem, A.S.; Mustapha, S.; Ndamitso, M.M.; Bada, S.O.; Sagadevan, S. Polyethyleneglycol-polyhydroxylbutyrate functionalized carbon nanotubes for industrial electroplating wastewater treatment. Sep. Purif. Technol. 2024, 332, 125736. [CrossRef]
- Hoang, A.T.; Nižetić, S.; Cheng, C.K.; Luque, R.; Thomas, S.; Banh, T.L.; Pham, V.V.; Nguyen, X.P. Heavy metal removal by biomass-derived carbon nanotubes as a greener environmental remediation: A comprehensive review. Chemosphere, 287, 2022, 131959. [CrossRef]
- Al-Amrani, W.A.; Onaizi, S.A. Adsorptive removal of heavy metals from wastewater using emerging nanostructured materials: A state-of-the-art review. Sep. Purif. Technol. 2024, 343, 127018. [CrossRef]
- Bhasin, C.P.; Pathan, A.; Patel, R.V. An Evaluation of Carbon Nanotube-based and Activated Carbon-based Nanocomposites for Fluoride and Other Pollutant Removal from Water: A Review. Current Nanomaterials 2023, 9, 16-40. [CrossRef]
- Sharma, M.; Dandautiya, R. Advancements in Nanotechnology for Heavy Metal Remediation in Wastewater Treatment: Challenges and Opportunities. e3s web of conferences 2024, 39, 01015. [CrossRef]
- Zare, K.; Sadegh, H.; Shahryari-ghoshekandi, R.; Maazinejad, B.; Ali, V.; Tyagi, I. et al. Enhanced removal of toxic Congo red dye using multi walled carbon nano- tubes: kinetic, equilibrium studies and its comparison with other adsorbents. J Mol Liq 2015, 212, 266-271. [CrossRef]
- Machado, F.M.; Bergmann, C.P.; Lima, E.C.; Royer, B.; de Souza, F. E.; Jauris, I. M. et al. Adsorption of reactive blue 4 dye from water solutions by carbon nanotubes: experiment and theory. Phys. Chem. Chem. Phys., 2012, 14, 11139-11153. [CrossRef]
- Mashkoor, F.; Nasar, A.; Inamuddin. Carbon nanotube-based adsorbents for the removal of dyes from waters: A review. Environ Chem Lett 2020, 18, 605-629. [CrossRef]
- Dastgerdi, Z.H.; Meshkat, S.S.; Esrafili, M.D. Enhanced adsorptive removal of Indigo carmine dye performance by functionalized carbon nanotubes based adsorbents from aqueous solution: equilibrium, kinetic, and DFT study. J Nanostruct Chem 2019, 9, 323-334. [CrossRef]
- Sadegh, H.; Ali, G.A.M.; Agarwal, S. Gupta, V.K. Surface Modification of MWCNTs with Carboxylic-to-Amine and Their Superb Adsorption Performance. Int J Environ Res 2019, 13, 523-531. [CrossRef]
- Hu, D.; Wan, X.; Li, X.; Liu, J.; Zhou, C. Synthesis of water-dispersible poly-l-lysine-functionalized magnetic Fe3O4-(GO-MWCNTs) nanocomposite hybrid with a large surface area for high-efficiency removal of tartrazine and Pb(II). Int. J. Biol. Macromol. 2017, 105, 1611-1621. [CrossRef]
- Chen, L.; Jiang, J.; Sheng, L. Removal of Bisphenol A from Water by Single-Walled Carbon Nanotubes Loaded with Iron Oxide Nanoparticles. Appl. Sci. 2024, 14, 3943. [CrossRef]
- Alkallas, F. H.; Alghamdi, S. M.; Rashed, E. A.; Trabelsi, A.B.G.; Nafee, S.S.; Elsharkawy, W.B.; Alsubhe, E.; Alshreef, S.H.; Mostafa, A.M. Nanocomposite Fe3O4-MWCNTs based on femtosecond pulsed laser ablation for catalytic degradation, Diamond and Related Materials. 2023, 140, 110445. [CrossRef]
- Sayana, K.V.; Prajwal, K.; Deeksha, K.J.; Vishalakshi, B.; Vishwanath, T. Magnetized CNTs incorporated MBA cross-linked guar gum nano-composite for methylene blue dye removal. J. Appl. Polym. Sci. 2024, 141, e54868. [CrossRef]
- Xiang, K.; Lu, W.; Wang, J.; Ma, P.; Xu, L. Aminated multi-walled carbon nanotubes doped magnetic flower-like FeSe2 nanosheets towards efficient adsorption in acidic wastewater. J. Mol. Struct. 2024,1305, 137816. [CrossRef]
- Costa, H.P.S.; Duarte, E.D.V.; da Silva, F.V.; da Silva, M.G.C.; Vieira, M.G.A. Green synthesis of carbon nanotubes functionalized with iron nanoparticles and coffee husk biomass for efficient removal of losartan and diclofenac: Adsorption kinetics and ANN modeling studies. Environ. Res. 2024, 251, 118733. [CrossRef]
- Heloisa Pereira de Sá Costa.; Emanuele Dutra Valente Duarte.; Meuris Gurgel Carlos da Silva.; Melissa Gurgel Adeodato Vieira. Adsorption of diclofenac and losartan using multi-walled carbon nanotubes functionalized with iron nanoparticles via the green route: Equilibrium, thermodynamics, and machine learning studies. J. Water Process Eng. 2024, 58, 104923. [CrossRef]
- Licona-Aguilar, A, I.; Torres-Huerta, A.M.; Domínguez-Crespo, M.A.; Negrete-Rodríguez, M.L.X.; Conde-Barajas, E. et al. Valorization of agroindustrial orange peel waste during the optimization of activated carbon–multiwalled carbon nanotubes–zinc oxide composites used in the removal of methylene blue in wastewater. J. Chem. Eng. 2024, 492, 152102. [CrossRef]
- Aljeboree, A.M.; Hussein, S.A.; Jawad, M.A.; Alkaim, A.F. Hydrothermal synthesis of eco-friendly ZnO/CNT nanocomposite and efficient removal of Brilliant Green cationic dye. Results Chem. 2024, 7, 101364. [CrossRef]
- Eskandarian, L.; Arami, M.; Pajootan, E. Evaluation of Adsorption Characteristics of Multiwalled Carbon Nanotubes Modified by a Poly(propylene imine) Dendrimer in Single and Multiple Dye Solutions: Isotherms, Kinetics, and Thermodynamics, J. Chem. Eng. Data 2014, 59, 444-454. [CrossRef]
- Radia, N.D.; Aljeboree, A.M.; Mhammed, A.A. Enhanced removal of crystal violet from aqueous solution using carrageenan hydrogel nanocomposite/MWCNTs. Inorg. Chem. Commun. 2024, 112803. [CrossRef]
- Grmasha, R.A.; Al-sareji, O.J.; Meiczinger, M.; Stenger-Kovács, C.; Al-Juboori, R.A.; Jakab, M. et al.K.S. A sustainable nano-hybrid system of laccase@M-MWCNTs for multifunctional PAHs and PhACs removal from water, wastewater, and lake water. Environ. Res. 2024, 246, 118097. [CrossRef]
- Ezugbe, E,O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10(5), 89. [CrossRef]
- Othman, N.H.; Alias, N.H.; Fuzil, N.S.; Marpani, F.; Shahruddin, M.Z.; Chew, C.M. et al. A Review on the Use of Membrane Technology Systems in Developing Countries. Membranes 2022, 12(1), 30. [CrossRef]
- Barrejón, M.; Prato, M. Carbon Nanotube Membranes in Water Treatment Applications. Adv. Mater. Interfaces 2022, 9, 2101260. [CrossRef]
- Ihsanullah, Carbon nanotube membranes for water purification: Developments, challenges, and prospects for the future. Sep. Purif. Technol. 2019, 209, 307-337. [CrossRef]
- Membrane Preparation, Editor(s): Ian D. Wilson, Encyclopedia of Separation Science, Academic Press, 2000, Pages 1755-1764. [CrossRef]
- Lee, B.; Baek, Y.; Lee, M.; Jeong, D.H., Yoon, J. et al. A carbon nanotube wall membrane for water treatment. Nat. Commun. 2015, 6, 7109. [CrossRef]
- Arahman, N.; Rosnelly, C.M.; Aulia, M.P.; Haikal, R.D.; Yusni.; Ambarita, A.C. et al. Exploring the effect of CNTs and pluronic on characteristics and stability of polyethersulfone (PES) and polyvinylidene fluoride (PVDF) membranes. Case Stud. Chem. Environ. Eng. 2024, 10, 100777. [CrossRef]
- Ngoma, M.M.; Mathaba, M.; Moothi, K. Effect of carbon nanotubes loading and pressure on the performance of a polyethersulfone (PES)/carbon nanotubes (CNT) membrane. Sci Rep 2021, 11, 23805. [CrossRef]
- Zhao, J.; Zeng, D.; Wang, Q.; Lin, Z.; Vogel, F.; Li, W.; Zhang, P. Effects of a dual functional filler, polyethersulfone-g-carboxymethyl chitosan@MWCNT, for enhanced antifouling and penetration performance of PES composite membranes. J. Environ. Manag. 2024, 365, 121611. [CrossRef]
- Wang, Ruijia.; Liu, Hongxu.; Wang, Zicheng.; Zhao, Jingxuan.; Lv, Ziwei.; Qi, Yuchao. et al. Synergistic Interaction of Ionic Liquid Grafted Poly(vinylidene Fluoride) and Carbon Nanotubes to Construct Water Treatment Membranes with Multiple Separation Properties. Langmuir 2024, 40(23), 11903-11913. [CrossRef]
- Macevele, L.E.; Moganedi, K.L.M.; Magadzu, T. Effects of Poly(Vinylidene Fluoride-co-Hexafluoropropylene) Nanocomposite Membrane on Reduction in Microbial Load and Heavy Metals in Surface Water Samples. J. Compos. Sci. 2024, 8, 119. [CrossRef]
- Zhao, J.; Liu, H.; Zhao, Y.; Qi, Y.; Wang, R. et al. Construction of CS-SDAEM long-chain polysaccharide derivative on TA@CNTs coated PVDF membrane with effective oil-water emulsion purification and low contamination rate. Int. J. Biol. Macromol. 2024, 275, 134230. [CrossRef]
- Zhang, Z.; Li, W.; Zhao, B.; Yang, X.; Zhao, C.; Wang, W. et al. Novel CNT/MXene composite membranes with superior electrocatalytic efficiency and durability for sustainable wastewater treatment. J. Chem. Eng. 2024, 495, 153605. [CrossRef]
- Chen, J.; Liu, X.; Hong, S.; Mo, J.; Peng, J.; Su, T. et al. Polylactic acid/ zinc oxide decorated multi-wall carbon nanotubes fibrous membranes and their applications in oil-water separation. Colloids Surf. A: Physicochem. Eng. Asp. 2024, 692, 133837. [CrossRef]
- Golgoli, M.; Farahbakhsh, J.; Najafi, M.; Khiadani, M.; Johns, M.L.; Zargar, M. Resilient forward osmosis membranes against microplastics fouling enhanced by MWCNTs/UiO-66-NH2 hybrid nanoparticles. Chemosphere 2024, 359, 142180. [CrossRef]
- Yang, Y.; Cheng, Y.; Ling, S.; Wan, Y.; Xiong, Z.; Li, C. et al. Scalable, flexible, ultra-strong, free-standing papery all carbon nanotube membrane with excellent separation and antifouling properties. J. Chem. Eng. 2024, 490, 151750. [CrossRef]
- Jasim, H.K.; Al-Ridah, Z.A.; Naje, A.S. Graphene oxide–carbon nanotube composite membrane for enhanced removal of organic pollutants by forward osmosis. Desalin. Water Treat. 2024, 318, 100363. [CrossRef]
- Abumelha, H.M.; Pashameah, R.; Sari, A.A.A.; Bin-Ibrahim, S.F.; Alanazi, M.A.A.; Shah, R. et al. Remarkable photocatalytic activity of MWCs supported on PVF in recycling, solar and photodegradation processes for commercial dyes and real industrial wastewater. Opt. Mater. 2024, 150, 115319. [CrossRef]
- Hye-Jin, L.; Zhang, N.; Ganzoury. M.A.; Wu, Y.; de Lannoy, C-F. Simultaneous Dechlorination and Advanced Oxidation Using Electrically Conductive Carbon Nanotube Membranes. ACS Appl. Mater. Interfaces 2021, 13(29), 34084-34092. [CrossRef]
- Zhang, J.; Dai, M.; Zhang, S.; Dai, M.; Zhang, P.; Wang, S.; He, Z. (2022), Recent Progress on Carbon-Nanotube-Based Materials for Photocatalytic Applications: A Review. Sol. RRL, 6: 2200243. [CrossRef]
- Osorio-Aguilar, D.-M.; Saldarriaga-Noreña, H.-A.; Murillo-Tovar, M.-A.; Vergara-Sánchez, J.; Ramírez-Aparicio, J.; Magallón-Cacho, L.; García-Betancourt, M.-L. Adsorption and Photocatalytic Degradation of Methylene Blue in Carbon Nanotubes: A Review with Bibliometric Analysis. Catalysts 2023, 13, 1480. [CrossRef]
- Mohapatra ,L.; Cheon, D.; Yoo ,SH. Carbon-Based Nanomaterials for Catalytic Wastewater Treatment: A Review. Molecules 2023, 28(4), 1805. [CrossRef]
- Verma, P.; Ubaid, J.; Varadarajan, K.M.; Wardle, B.L.; Kumar, S. Synthesis and Characterization of Carbon Nanotube-Doped Thermoplastic Nanocomposites for the Additive Manufacturing of Self-Sensing Piezoresistive Materials. ACS Appl. Mater. Interfaces 2022, 14(6), 8361-8372. [CrossRef]
- Hughes, K.J.; Iyer, K.A.; Bird, R.E.; Ivanov, J.; Banerjee, S.; Georges, Gilles.; Zhou, Q. A. Review of Carbon Nanotube Research and Development: Materials and Emerging Applications, ACS Appl. Nano Mater. 2024, 7(16), 18695-18173. [CrossRef]
- Karthikeyan, C.; Arunachalam ,P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloys Compd. 2020, 828, 154281. [CrossRef]
- Chen, Y.; Qian, J.; Wang, N.; Xing, J.; Liu, L. In-situ synthesis of CNT/TiO2 heterojunction nanocomposite and its efficient photocatalytic degradation of Rhodamine B dye. Inorg. Chem. Commun. 2020, 119, 108071. [CrossRef]
- Shaban, M.; Ashraf, A.M.; Abukhadra, M.R. TiO2 Nanoribbons/Carbon Nanotubes Composite with Enhanced Photocatalytic Activity; Fabrication, Characterization, and Application. Sci Rep 2018, 8, 781. [CrossRef]
- Pete, KY.; Kabuba, J.; Otieno, B.; Ochieng, A. Modeling adsorption and photocatalytic treatment of recalcitrant contaminant on multi-walled carbon/TiO2 nanocomposite. Environ Sci Pollut Res Int. 2023 , 30(41), 94154-94165. [CrossRef]
- Czech ,B.; Buda, W. Photocatalytic treatment of pharmaceutical wastewater using new multiwall-carbon nanotubes/TiO2/SiO2 nanocomposites. Environ Res. 2015, 137, 176-84. [CrossRef]
- Akter, J.; Hanif, M.A.; Islam, M.A. et al. Selective growth of Ti3+/TiO2/CNT and Ti3+/TiO2/C nanocomposite for enhanced visible-light utilization to degrade organic pollutants by lowering TiO2-bandgap. Sci Rep 2021, 11, 9490. [CrossRef]
- Koo, Y.; Littlejohn, G.; Collins, B.; Yun, Y.; Shanov, V.N.; Schulz, M.; Pai, D.; Sankar, J. Synthesis and characterization of Ag–TiO2–CNT nanoparticle composites with high photocatalytic activity under artificial light, Compos. B Eng. 2014, 57, 105-111. [CrossRef]
- Azzam, E.M.S.; Fathy, N.A.; El-Khouly, S.M.; Sami, R.M. Enhancement the photocatalytic degradation of methylene blue dye using fabricated CNTs/TiO2/AgNPs/Surfactant nanocomposites, J WATER PROCESS ENG. 2019, 28, 311-321. [CrossRef]
- Yang ,Y.; Liu, K.; Sun, F.; Liu ,Y.; Chen ,J. Enhanced performance of photocatalytic treatment of Congo red wastewater by CNTs-Ag-modified TiO2 under visible light. Environ Sci Pollut Res Int. 2022, 29(11), 15516-15525. [CrossRef]
- Chen, J.; Liu, K.; Liu, Y. Synergistic Molecular Mechanism of Degradation in Dye Wastewater by Rhodopseudomonas palustris Intimately Coupled Carbon Nanotube–Silver Modified Titanium Dioxide Photocatalytic Composite with Sodium Alginate. J. Environ. Manag. 2024, 351, 119913. [CrossRef]
- Liu ,K.; Yang ,Y.; Sun, F.; Liu, Y.; Tang, M.; Chen, J. Rapid degradation of Congo red wastewater by Rhodopseudomonas palustris intimately coupled carbon nanotube - Silver modified titanium dioxide photocatalytic composite with sodium alginate. Chemosphere. 2022 Jul;299:134417. [CrossRef]
- Akhter,P.; Ali,F.; Ali,A.; Hussain,M. TiO2 decorated CNTs nanocomposite for efficient photocatalytic degradation of methylene blue, Diamond and Related Materials, 141, 2024, 110702. [CrossRef]
- Gallegos-Cerda, S.D.; Hernández-Varela, J.D.; Chanona Pérez, J.J.; Huerta-Aguilar, C.A.; Victoriano, L .G.; Arredondo-Tamayo, B.; Hernández, O.R. Development of a low-cost photocatalytic aerogel based on cellulose, carbon nanotubes, and TiO2 nanoparticles for the degradation of organic dyes, Carbohydrate Polymers, 324, 2024, 121476. [CrossRef]
- Rostami, M.S.; Khodaei, M.M.; Benassi, E. Surface modified of chitosan by TiO2@MWCNT nanohybrid for the efficient removal of organic dyes and antibiotics. Int. J. Biol. Macromol. 2024, 274(Part 1), 133382. [CrossRef]
- Su-Bin Kim.; Seul-Yi Lee.; Soo-Jin Park. TiO2 / multi-walled carbon nanotube electro spun nanofibers mats for enhanced Cr (VI) photoreduction. Journal of Cleaner Production 2024, 448,141611. [CrossRef]
- Hor-Yan ,Phin.; Yit-Thai, Ong.; Jin-Chung, Sin. Effect of carbon nanotubes loading on the photocatalytic activity of zinc oxide/carbon nanotubes photocatalyst synthesized via a modified sol-gel method, J. Environ. Chem. Eng., 2020, 8, 103222. [CrossRef]
- Hanif,Md.A.; Young-Soon KimJeasmin AkterHong Gun KimLee Ku Kwac, Fabrication of Robust and Stable N-Doped ZnO/Single-Walled Carbon Nanotubes: Characterization, Photocatalytic Application, Kinetics, Degradation Products, and Toxicity Analysis, ACS Omega, ACS Omega 2023, 8(18), 16174–16185. [CrossRef]
- Humayoun ,UB.; Mehmood ,F.; Hassan ,Y.; Rasheed ,A.; Dastgeer ,G.; Anwar ,A.; Sarwar ,N.; Yoon, D. Harnessing Bio-Immobilized ZnO/CNT/Chitosan Ternary Composite Fabric for Enhanced Photodegradation of a Commercial Reactive Dye. Molecules 2023, 28(18), 6461. [CrossRef]
- Foroutan,R.; Peighambardoust,S.J.; Esvandi,Z.; Khatooni,H.; Ramavandi,B. Evaluation of two cationic dyes removal from aqueous environments using CNT/MgO/CuFe2O4 magnetic composite powder: A comparative study, J. Environ. Chem. Eng. 2021, 9, 104752. [CrossRef]
- Rui-Jie Pan.; Jing Wu.; Jin Qu.; Tingting Zhang.; Fan-Zhen Jiao.; Mang Zhao.; Meng-Yan Han.; Xiaofeng L.; Zhong-Zhen Yu. Peak-like three-dimensional CoFe2O4/carbon nanotube decorated bamboo fabrics for simultaneous solar-thermal evaporation of water and photocatalytic degradation of bisphenol A. J. Mater. Sci. Technol. 2024, 179, 40-49. [CrossRef]
- Fidelis, M.Z.; dos Santos, A.S.G.G.; de Paula ,E.T.; Lenzi, G.G.; Soares, O.S.G.P.; Andreo, O.A.B. Nb2O5/MWCNT nanocomposites for the degradation of ibuprofen via photocatalysis and catalytic ozonation. Catal. Commun. 2024, 187, 106853. [CrossRef]
- Isari, AA.; Mehregan, M.; Mehregan, S.; Hayati, F.; Kalantary, R.R.; Kakavandi, B. Sono-photocatalytic degradation of tetracycline and pharmaceutical wastewater using WO3/CNT heterojunction nanocomposite under US and visible light irradiations: A novel hybrid system. J Hazard Mater. 2020, 15(39), 122050. [CrossRef]
- Huo ,H.; Hu, X.; Wang ,H.; Li ,J.; Xie, G.; Tan ,X.; Jin, Q.; Zhou, D.; Li, C.; Qiu ,G.; Liu ,Y. Synergy of Photocatalysis and Adsorption for Simultaneous Removal of Hexavalent Chromium and Methylene Blue by g-C3N4/BiFeO3/Carbon Nanotubes Ternary Composites. Int J Environ Res Public Health. 2019, 16(17), 3219. [CrossRef]
- Mestre ,AS.; Carvalho ,AP. Photocatalytic Degradation of Pharmaceuticals Carbamazepine, Diclofenac, and Sulfamethoxazole by Semiconductor and Carbon Materials: A Review. Molecules 2019, 24(20), 3702. [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V. Electric field effect in atomically thin carbon films. Science 2004, 306, 666-669. [CrossRef]
- Gollavelli, G.; Ling, Y.C. Chapter 21 Ultrathin graphene structure, fabrication and characterization for clinical diagnosis applications. In Smart Nanodevices for Point-of-Care Applications; Kanchi, S., Chokkareddy, R., Rezakazemi, M., Eds.; CRC Press: Boca Raton, FL, USA, 2022; pp. 263-280.
- Gollavelli, G.; Chang, C.-C.; Ling, Y.-C. Facile Synthesis of Smart Magnetic Graphene for Safe Drinking Water: Heavy Metal Removal and Disinfection Control. ACS Sustainable Chem. Eng. 2013, 1, 462-472. [CrossRef]
- Liu, Y. Application of graphene oxide in water treatment 2017 IOP Conf. Ser.: Earth Environ. Sci. 2017, 94, 012060. [CrossRef]
- Peng, W.; Li, H.; Liu, Y.; Song, S. A review on heavy metal ions adsorption from water by graphene oxide and its composites. J. Mol. Liq. 2017, 230, 496-504. [CrossRef]
- Thakur, K.; and Kandasubramanian, B. Graphene and Graphene Oxide-Based Composites for Removal of Organic Pollutants: A ReviewJ. Chem. Eng. Data 2019,64, 833-867. [CrossRef]
- Huang, P.; Xu, C.; Lin, J.; Wang, C.; Wang, X.; Zhang, C.; Zhou, X.; Guo, S.; Cui, D. Folic Acid-conjugated Graphene Oxide loaded with Photosensitizers for Targeting Photodynamic Therapy. Theranostics 2011, 1,240-250. https://www.thno.org/v01p0240.htm. [CrossRef]
- Patel, M. A.; Yang, H.; Chiu, P. L.; Mastrogiovanni, D. D. T.; Flach, C. R.; Savaram, K.; Gomez, L.; Hemnarine, A.; Mendelsohn, R.; Garfunkel, E.; Jiang, H.; He, H. Direct Production of Graphene Nanosheets for Near Infrared Photoacoustic Imaging. ACS Nano 2013, 7, 8147-8157. [CrossRef]
- Mahmood, F.; Ashraf, S.; Shahzad, M.; Li, B.; Asghar, F.; Amjad, W.; Omar, M. M. Graphene Synthesis from Organic Substrates: A Review. Ind. Eng. Chem. Res. 2023, 62, 17314-17327. [CrossRef]
- Verma, G.; Mondal, K.; Islam, M.; Gupta, A. Recent Advances in Advanced Micro and Nanomanufacturing for Wastewater Purification. ACS Appl. Eng. Mater. 2024, 262-285. [CrossRef]
- Yuan, S.-J.; Wang, J.-J.; Dong, B.; Dai, X.-H. Biomass-Derived Carbonaceous Materials with Graphene/Graphene-Like Structures: Definition, Classification, and Environmental Applications. Environ. Sci. Technol. 2023, 57, 17169-17177. [CrossRef]
- Devi, M. K.; Yaashikaa, P. R.; Kumar, P. S.; Manikandan, S.; Oviyapriya, M.; Varshikaa, V.; Rangasamy, G. Recent Advances in Carbon-Based Nanomaterials for the Treatment of Toxic Inorganic Pollutants in Wastewater. New J. Chem. 2023, 47, 7655-7667. [CrossRef]
- Sitko, R.; Turek, E.; Zawisza, B.; Malicka, E.; Talik, E.; Heimann, J.; Gagor, A.; Feist, B.; Wrzalik, R. Adsorption of Divalent Metal Ions from Aqueous Solutions Using Graphene Oxide. Dalton Trans. 2013, 42, 5682-5689. [CrossRef]
- Liu, L.; Li, C.; Bao, C.; Jia, Q.; Xiao, P.; Liu, X.; Zhang, Q. Preparation and Characterization of Chitosan/Graphene Oxide Composites for the Adsorption of Au(III) and Pd(II). Talanta 2012, 93, 350-357. [CrossRef]
- Musico, Y. L. F.; Santos, C. M.; Dalida, M. L. P.; Rodrigues, D. F. Improved Removal of Lead(II) from Water Using a Polymer-Based Graphene Oxide Nanocomposite. J. Mater. Chem. A 2013, 1, 3789-3796. [CrossRef]
- Yang, Y.; Xie, Y.; Pang, L.; Li, M.; Song, X.; Wen, J.; Zhao, H. Preparation of Reduced Graphene Oxide/Poly(acrylamide) Nanocomposite and Its Adsorption of Pb(II) and Methylene Blue. Langmuir 2013, 29, 10727-10736. [CrossRef]
- Patel, M.; Lanakapati, H.; Maheria, K. Efficient Removal of Pb²⁺, Cd²⁺, and Hg²⁺ from Wastewater Using Advanced Graphene Oxide-Zirconium Hybrid Composite. ACS Appl. Eng. Mater. 2024, 2, 1542-1560. [CrossRef]
- Gou, Q.; Cai, X.; Yan, Z.; Gao, Y.; Tang, J.; Xiao, W.; Cai, J. Highly Selective Pb(II) Adsorption by DTPA-Functionalized Graphene Oxide/Carboxymethyl Cellulose Aerogel. Langmuir 2024, 40, 8002-8014. [CrossRef]
- Jabbar, A. A.; Hussain, D. H.; Latif, K. H.; Jasim, A. K.; Al-aqbi, Z. T.; Alghannami, H. S.; Albishri, A. High-Efficiency Adsorption of Uranium from Wastewater Using Graphene Oxide/Graphene Oxide Nanoribbons/Chitosan Nanocomposite Aerogels. ACS Omega 2024, 9, 27260-27268. [CrossRef]
- Ortiz-Quiñonez, J. L.; Cancino-Gordillo, F. E.; Pal, U. Removal of Cr(III) Ions from Water Using Magnetically Separable Graphene-Oxide-Decorated Nickel Ferrite Nanoparticles. ACS Appl. Nano Mater. 2023, 6, 18491-18507. [CrossRef]
- Bagbi, Y.; Solanki, P. R. Fabrication of Mesoporous Silica Nanoparticle-Decorated Graphene Oxide Sheets for the Effective Removal of Lead (Pb²⁺) from Water. ACS Omega 2024, 9, 304-316. [CrossRef]
- Pan, N.; Zhou, F.; Nie, X.; Ma, C.; Lei, Y.; Lei, H.; Li, L. One-Step Synthesis of Silver-Decorated Reduced Graphene Oxide Nanocomposites for Effective Elimination of Iodide Anions from Water. ACS ES&T Water 2024, 4, 719-734. [CrossRef]
- Thakur, K.; Kandasubramanian, B. Graphene and Graphene Oxide-Based Composites for Removal of Organic Pollutants: A Review. J. Chem. Eng. Data 2019, 64, 833-867. [CrossRef]
- Abd-Elhamid, A.I., Elgoud, E.M.A., Emam, S.S. et al. Superior adsorption performance of citrate modified graphene oxide as nano material for removal organic and inorganic pollutants from aqueous solution. Sci Rep 202j2, 12, 9204. [CrossRef]
- Ghadim, E. E.; Manouchehri, F.; Soleimani, G.; Hosseini, H.; Kimiagar, S.; Nafisi, S. Adsorption Properties of Tetracycline onto Graphene Oxide: Equilibrium, Kinetic and Thermodynamic Studies. PLoS One 2013, 8, 79254. [CrossRef]
- Kyzas, G. Z.; Bikiaris, D. N.; Seredych, M.; Bandosz, T. J.; Deliyanni, E. A. Removal of Dorzolamide from Biomedical Waste- waters with Adsorption onto Graphite Oxide/Poly(Acrylic Acid) Grafted Chitosan Nanocomposite. Bioresour. Technol. 2014, 152, 399-406. [CrossRef]
- Sun, L.; Yu, H.; Fugetsu, B. Graphene Oxide Adsorption Enhanced by in Situ Reduction with Sodium Hydrosulfite to Remove Acridine Orange from Aqueous Solution. J. Hazard. Mater. 2012, 203-204, 101-110. [CrossRef]
- Hosseinabadi-Farahani, Z.; Hosseini-Monfared, H.; Mahmoodi, N. M. Graphene Oxide Nanosheet: Preparation and Dye Removal from Binary System Colored Wastewater. Desalin. Water Treat. 2015, 56, 2382−2394. [CrossRef]
- Leão, M. B.; Fernandes, A. N.; Jauris, C. F. de M. Nanocarbon Dimensionality and Oxidation Degree Influence the Sorption of Atenolol: Implications for Water Treatment. ACS Appl. Nano Mater. 2024, 7 (7), 8307–8317. [CrossRef]
- Pervez, M. N.; Jiang, T.; Mahato, J. K.; Ilango, A. K.; Kumaran, Y.; Zuo, Y.; Zhang, W.; Efstathiadis, H.; Feldblyum, J. I.; Yigit, M. V.; Liang, Y. Surface Modification of Graphene Oxide for Fast Removal of Per- and Polyfluoroalkyl Substances (PFAS) Mixtures from River Water. ACS ES&T Water 2024, 4 (7), 2968–2980. [CrossRef]
- Pramanik, A.; Kolawole, O. P.; Gates, K.; Kundu, S.; Shukla, M. K.; Moser, R. D.; Astarlioglu, M. U.; Al-Ostaz, A.; Ray, P. C. 2D Fluorinated Graphene Oxide (FGO)-Polyethyleneimine (PEI) Based 3D Porous Nanoplatform for Effective Removal of Forever Toxic Chemicals, Pharmaceutical Toxins, and Waterborne Pathogens from Environmental Water Samples. ACS Omega 2023, 8 (47), 44942–44954. [CrossRef]
- Agrawal, F.; Gupta, K.; Kaushik, J.; Tripathi, K. M.; Choudhary, S. K.; Sonkar, S. K. Graphene Incorporated Sugar Derived Carbon Aerogel for Pyridine Adsorption and Oil–Water Separation. Langmuir, Article ASAP. [CrossRef]
- Lv, T.; Wu, F.; Zhang, Z.; Liu, Z.; Zhao, Y.; Yu, L.; Zhang, J.; Yu, C.; Zhao, C.; Xing, G. TiVCTX MXene/Graphene Nanosheet-Based Aerogels for Removal of Organic Contaminants from Wastewater. ACS Appl. Nano Mater. 2024, 7 (7), 7312–7326. [CrossRef]
- He, N.; Zhao, X.; Li, Z.; Shi, T.; Li, Z.; Guo, F.; Li, W. Polydopamine Enhanced Interactions of Graphene Nanosheets to Fabricate Graphene/Polydopamine Aerogels with Effectively Clear Organic Pollutants. Langmuir 2024, 40 (18), 9592–9601. [CrossRef]
- Natasha; Khan, A.; Rahman, U. ; U. Sadaf; Yaseen, M.; Abumousa, R. A.; Khattak, R.; Rehman, N.; Bououdina, M.; Humayun, M. Effective Removal of Nile Blue Dye from Wastewater using Silver-Decorated Reduced Graphene Oxide. ACS Omega 2024, 9 (17), 19461–19480. [CrossRef]
- Kabir, M. H.; Hossain, M. S.; Rahman, M. M.; Ashrafuzzaman, M.; Hasan, M.; Pabel, M. Y.; Islam, D.; Bashar, M. S.; Faruque, T.; Yasmin, S. Green Reduction of Waste-Battery-Derived Graphene Oxide by Jute Leaves and Its Application for the Removal of Tetracyclines from Aqueous Media. ACS Sustainable Resource Management 2024, 1 (8), 1812–1823. [CrossRef]
- Herath, T. M.; Zhang, B.; Dwinandha, D.; Fujii, M. Elucidating Adsorption Mechanisms and Characteristics of Emerging Aromatic Organic Contaminants to Graphene Material by Quantum Chemical Calculation Integrated with Interpretable Machine Learning. ACS ES&T Water, Article ASAP. Publication Date (Web): August 2, 2024. [CrossRef]
- Homaeigohar, S.; Elbahri, M. Graphene Membranes for Water Desalination. NPG Asia Mater. 2017, 9, e427. [CrossRef]
- Nam, Y.-T.; Kang, J.-H.; Jang, J.-D.; Bae, J.-H.; Jung, H.-T.; Kim, D.-W. Recent Developments in Nanoporous Graphene Membranes for Organic Solvent Nanofiltration: A Short Review. Membranes 2021, 11, 793. [CrossRef]
- Han, Q.; Zhang, Z.; Li, L.; Yang, W.; Tao, Y.; Bi, K. Dual-Side Fabrication of Nanopores on Bilayer Graphene Membranes for Selective Ion Transport. J. Phys. Chem. C 2023, 127(46), 22646-22653. [CrossRef]
- Reddy, P. R.; Reddy, K. A.; Kumar, A. Comparative Retention Analysis of Intercalated Cations Inside the Interlayer Gallery of Lamellar and Nonlamellar Graphene Oxide Membranes in Reverse Osmosis Process: A Molecular Dynamics Study. J. Phys. Chem. B 2024, 128(21), 5218-5227. [CrossRef]
- Lv, Y.; Dong, L.; Cheng, L.; Gao, T.; Wu, C.; Chen, X.; He, T.; Cui, Y.; Liu, W. Tailoring Monovalent Ion Sieving in Graphene-Oxide Membranes with High Flux by Rationally Intercalating Crown Ethers. ACS Appl. Mater. Interfaces 2023, 15(39), 46261-46268. [CrossRef]
- Wang, Z.; Ma, C.; Xu, C.; Sinquefield, S. A.; Shofner, M. L.; Nair, S. Graphene Oxide Nanofiltration Membranes for Desalination under Realistic Conditions. Nat. Sustain. 2021, 4, 402–408. [CrossRef]
- Gong, D.; Liu, X.; Wu, P.; et al. Water Pumping Effect over the Organic Ions Defined Graphene Oxide Membrane Impulses High Flux Desalination. npj Clean Water 2022, 5, 68. [CrossRef]
- Zhang, H.; Xing, J.; Wei, G.; et al. Electrostatic-Induced Ion-Confined Partitioning in Graphene Nanolaminate Membrane for Breaking Anion–Cation Co-Transport to Enhance Desalination. Nat. Commun. 2024, 15, 4324. [CrossRef]
- Liu, Q.; Toyosi E, O.; Yang, C.; Zhang, R.; Long, M.; Jiang, Z.; Wu, H. Hydrophilic Graphene Oxide Membrane Intercalated by Halloysite Nanotubes for Oil-in-Water Emulsion Separation. ACS Appl. Eng. Mater. 2023, 1(12), 3338-3347. [CrossRef]
- Wu, D.; Sun, M.; Zhang, W.; Zhang, W. Simultaneous Regulation of Surface Properties and Microstructure of Graphene Oxide Membranes for Enhanced Nanofiltration Performance. ACS Appl. Mater. Interfaces 2023, 15(44), 52029-52037. [CrossRef]
- Chen, J.; Liu, X.; Ding, Z.; He, Z.; Jiang, H.; Zhu, K.; Li, Y.; Shi, G. Multistage Filtration Desalination via Ion Self-Rejection Effect in Cation-Controlled Graphene Oxide Membrane under 1 Bar Operating Pressure. Nano Lett. 2023, 23(23), 10884-10891. [CrossRef]
- Ahmad Farid, M. A.; Sabidi, S.; Ruozhu, W.; Lease, J.; Maeda, T.; Tsubota, T.; Andou, Y. Fabrication of Graphene-Based Nanofiltration Membrane from Fractionated Bamboo Lignin for Dye Removal and Desalination. ACS Appl. Eng. Mater. 2023, 1(10), 2791-2808. [CrossRef]
- Soomro, F.; Ali, A.; Ullah, S.; Iqbal, M.; Alshahrani, T.; Khan, F.; Yang, J.; Thebo, K. H. Highly Efficient Arginine Intercalated Graphene Oxide Composite Membranes for Water Desalination. Langmuir 2023, 39(50), 18447-18457. [CrossRef]
- Ali, A.; Vohra, M. I.; Nadeem, A.; Al-Anzi, B. S.; Iqbal, M.; Memon, A. A.; Jatoi, A. H.; Akhtar, J.; Yang, J.; Thebo, K. H. Ultrafast Graphene Oxide-Lignin Biopolymer Nanocomposite Membranes for Separation of Biomolecules, Dyes, and Salts. ACS Appl. Polym. Mater. 2024, 6(8), 4747-4755. [CrossRef]
- Tiwary, S. K.; Singh, M.; Hasan Likhi, F.; Dabade, S.; Douglas, J. F.; Karim, A. Self-Cross-Linking of MXene-Intercalated Graphene Oxide Membranes with Antiswelling Properties for Dye and Salt Rejection. ACS Environ. Au 2024, 4(2), 69-79. [CrossRef]
- Alemayehu, H. G.; Hou, J.; Qureshi, A. A.; Yao, Y.; Sun, Z.; Yan, M.; Wang, C.; Liu, L.; Tang, Z.; Li, L. Discrimination of Xylene Isomers by Precisely Tuning the Interlayer Spacing of Reduced Graphene Oxide Membrane. ACS Nano 2024, 18(28), 18673-18682. [CrossRef]
- Schmidt, S. J.; Dou, W.; Sydlik, S. A. Regeneratable Graphene-Based Water Filters for Heavy Metal Removal at Home. ACS ES&T Water 2023, 3(8), 2179-2185. [CrossRef]
- Bhol, P.; Yadav, S.; Altaee, A.; Saxena, M.; Kumari Misra, P.; Samal, A. K. Graphene-Based Membranes for Water and Wastewater Treatment: A Review. ACS Appl. Nano Mater. 2021, 4(4), 3274-3293. [CrossRef]
- Tian, L.; Zhou, P.; Su, Z.; Graham, N.; Yu, W. Surface Microstructure Drives Biofilm Formation and Biofouling of Graphene Oxide Membranes in Practical Water Treatment. Environ. Sci. Technol. 2024, 58(27), 12281-12291. [CrossRef]
- Khaliha, S.; Bianchi, A.; Kovtun, A.; Tunioli, F.; Boschi, A.; Zambianchi, M.; Paci, D.; Bocchi, L.; Valsecchi, S.; Polesello, S.; Liscio, A.; Bergamini, M.; Brunetti, M.; Navacchia, M. L.; Palermo, V.; Melucci, M. Graphene Oxide Nanosheets for Drinking Water Purification by Tandem Adsorption and Microfiltration. Sep. Purif. Technol. 2022, 300, 121826. [CrossRef]
- Li, X., Yu, J., Wageh, S., Al-Ghamdi, A.A. and Xie, J. Graphene in Photocatalysis: A Review. Small 2016, 12: 6640-6696. [CrossRef]
- Zhu, D.; Zhou, Q. Action and mechanism of semiconductor photocatalysis on degradation of organic pollutants in water treatment: A review. Environmental Nanotechnology, Monitoring & Management 2019, 12, 100255. [CrossRef]
- Zhang, J.; Xiong, Z.; Zhao, X.S. Graphene–metal–oxide composites for the degradation of dyes under visible light irradiation. J. Mater. Chem. 2011, 21, 3634-3640. [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-Graphene Composite as a High Performance Photocatalyst. ACS Nano 2010, 4, 380-386. [CrossRef]
- Huynh, T.P.; Do, T.C.M.V.; Le, P.H. TiO₂ Nanotube Arrays Decorated with Graphene/Graphite Oxide Nanocomposite for the Photocatalytic Degradation of Anticancer Drugs in the Aquatic Environment. ACS Appl. Nano Mater. 2024, 7, 20012–20023. [CrossRef]
- Li, B.; Cao, H. ZnO@graphene composite with enhanced performance for the removal of dye from water. J. Mater. Chem. 2011, 21, 3346-3349. [CrossRef]
- Lu, K.-Q.; Li, Y.-H.; Tang, Z.-R.; Xu, Y.-J. Roles of Graphene Oxide in Heterogeneous Photocatalysis. ACS Materials Au 2021, 1, 37-54. [CrossRef]
- Trivedi, P.A.; Naik, J.B.; Patil, P.B. Exploring graphene and its derivatives for various applications: photocatalysis. Chem. Pap. 2024, 78, 5705-5722. [CrossRef]
- Ramos, P.G.; Rivera, H.; Sánchez, L.A. et al. Graphene-based Semiconductors for Photocatalytic Degradation of Organic Dye from Wastewater: A Comprehensive Review. Water Air Soil Pollut. 2024, 235, 292. [CrossRef]
- Chitkara, M.; Goyalbc, N.; Kumar, A.; Marasamy, L.; Haq, S.; SAldossari, S. A.; Haldhar, R.; Hossain, M. K. Tailoring graphene-oxide and reduced-graphene-oxide with NaNO3 and CaCl2 catalysts with enhanced photo-catalytic degradation of methylene blue dye. RSC Adv., 2024, 14, 8769-8778. [CrossRef]
- Thakur, S.; Badoni, A.; Sharma, P.; Ojha, A.; Swart, H. C.; Kuznetsov, A. Y.; Prakash, J. Standalone Highly Efficient Graphene Oxide as an Emerging Visible Light-Driven Photocatalyst and Recyclable Adsorbent for the Sustainable Removal of Organic Pollutants. Langmuir Article ASAP. [CrossRef]
- Durairaj, S., Sridhar, D.; Ströhle, G.; Li, H.; Chen, A. Bactericidal Effect and Cytotoxicity of Graphene Oxide/Silver Nanocomposites. ACS Appl. Mater. Interfaces 2024, 16, 18300-18310. [CrossRef]
- Jaiswal, K.; Kadamannil, N.N.; Jelinek, R. Carbon nanomaterials in microbial sensing and bactericidal applications. Current Opinion in Colloid & Interface Science 66, 2023, 101719. [CrossRef]
- Konwarh, R.; Gollavelli, G.; Palanisamy, S. B. Designing novel nanosensors for environmental aspects. Nanofabrication for Smart Nanosensor Applications. Pal, K., Gomes, F., Eds.; Elsevier: location, country, 2020, volume, pp. 51-87. [CrossRef]
- Deokar, A. R.; Sinha, M.; Gollavelli, G.; Ling, Y.-C. Antimicrobial Perspectives for Graphene-Based Nanomaterials. Graphene Science Handbook: Applications and Industrialization. Aliofkhazraei, M., Ali, N., Milne, W. I., Ozkan, C. S., Mitura, S., Gervasoni, J. L., Eds.; CRC Press Taylor & Francis, 2016, pp. 488. [CrossRef]
- Gorle, G.; Gollavelli, G.; Nelli, G,; Ling, Y.-C. Green Synthesis of Blue-Emitting Graphene Oxide Quantum Dots for In Vitro CT26 and In Vivo Zebrafish Nano-Imaging as Diagnostic Probes. Pharmaceutics 2023, 15, 632. [CrossRef]
- Kumah, E. A.; Fopa, R. D.; Harati, S. et al. Human and environmental impacts of nanoparticles: a scoping review of the current literature. BMC Public Health 2023, 23, 1059. [CrossRef]
- Mohan, D., Pittman Jr, C.U. Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J. Hazard. Mater. 2007 137(2), 762-811. [CrossRef]
- Liu, S., et al. (2012).Graphene oxide-based materials for efficient removal of heavy metal ions from aqueous solution: A review. Environ. Pollut. 2019, 252(Pt A), 62-73. [CrossRef]
- Pérez, S.; Farré, M.; Barceló, D. Analysis, behavior and ecotoxicity of carbon-based nanomaterials in the aquatic environment. Trends Anal. Chem. 2009, 28(6), 820-832. 2648-2653. [CrossRef]
- Seoktae, Kang.; Meagan S, M.; Menachem, Elimelech. Microbial Cytotoxicity of Carbon-Based Nanomaterials: Implications for River Water and Wastewater Effluent. Environ. Sci. Technol. Lett. 2009, 43(7). https://pubs.acs.org/doi/10.1021/es8031506. [CrossRef]
- Zaytseva, O.; Neumann, G. Carbon nanomaterials: production, impact on plant development, agricultural and environmental applications. Chem. Biol. Technol. Agric. 2016, 3:17, 1-26. https://chembioagro.springeropen.com/articles/10.1186/s40538-016-0070-8. [CrossRef]
- Freixa, A.; Acuña, V.; Sanchís, J.; Farré, M.; Barceló, D.; Sabate, S. Ecotoxicological effects of carbon based nanomaterials in aquatic organisms. Sci. Total Environ. 2018, 619-620, 328-337. [CrossRef]
- Pikula, K.; Johari, S.A.; Santos-Oliveira, R.; Golokhvast, K. Joint Toxicity and Interaction of Carbon-Based Nanomaterials with Co-Existing Pollutants in Aquatic Environments: A Review. Int. J. Mol. Sci. 2024, 25, 11798. [CrossRef]
- Cha, C.; Shin, S.R.; Annabi, N,; Dokmeci, M.R.; Khademhosseini, A. Carbon-Based Nanomaterials: Multifunctional Materials for Biomedical Engineering. ACS Nano 2013, 7(4), 2891-2897. https://pubs.acs.org/doi/10.1021/nn401196a. [CrossRef]
- OSPAR Convention. Precautionary Principle. https://www.ospar.org/convention/principles/precautionary-principle.
- Goldstein, B.D. The Precautionary Principle Also Applies to Public Health Actions. Am J Public Health. 2001, 91(9), 1358-1361. https://ajph.aphapublications.org/doi/10.2105/AJPH.91.9.1358 . [CrossRef]
- Pinto-Bazurco, J.F. The Precautionary Principle. 2020. https://www.iisd.org/articles/deep-dive/precautionary-principle.
- Warshaw, J. The Trend Towards Implementing the Precautionary Principle in US Regulation of Nanomaterials. Dose-Response 2012, 10, 384-396. [CrossRef]
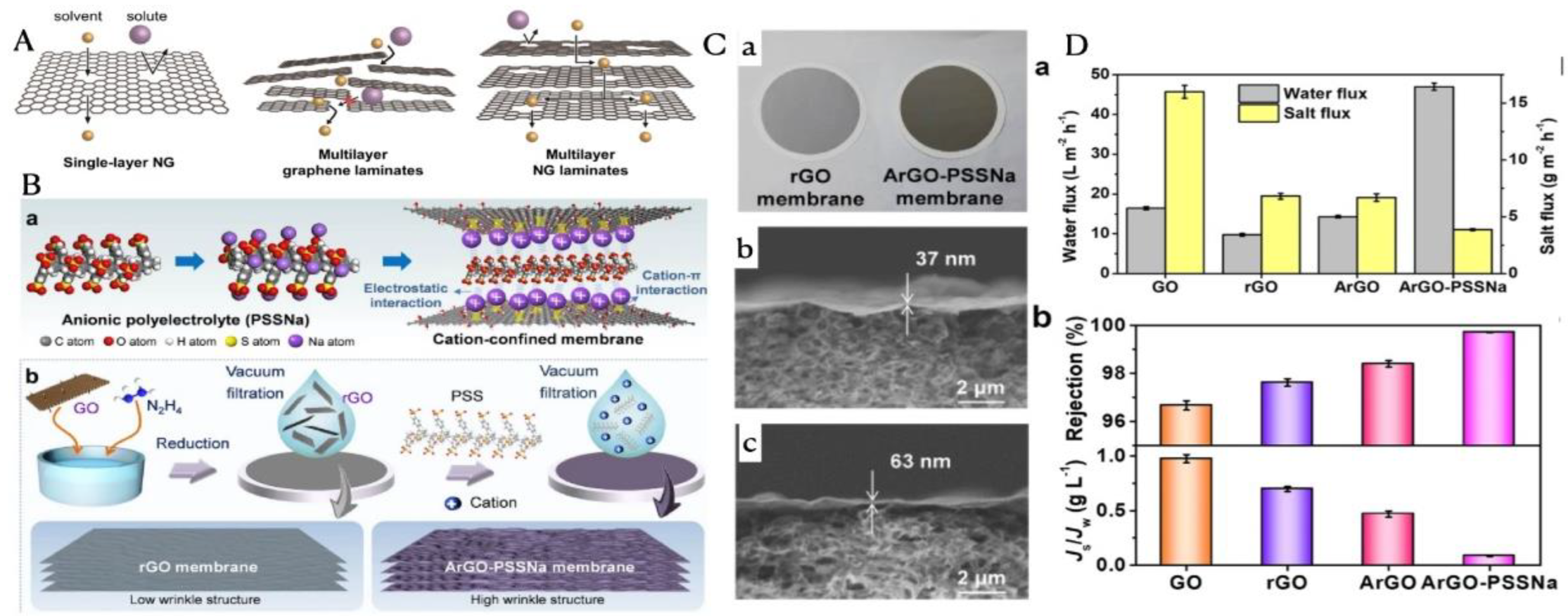
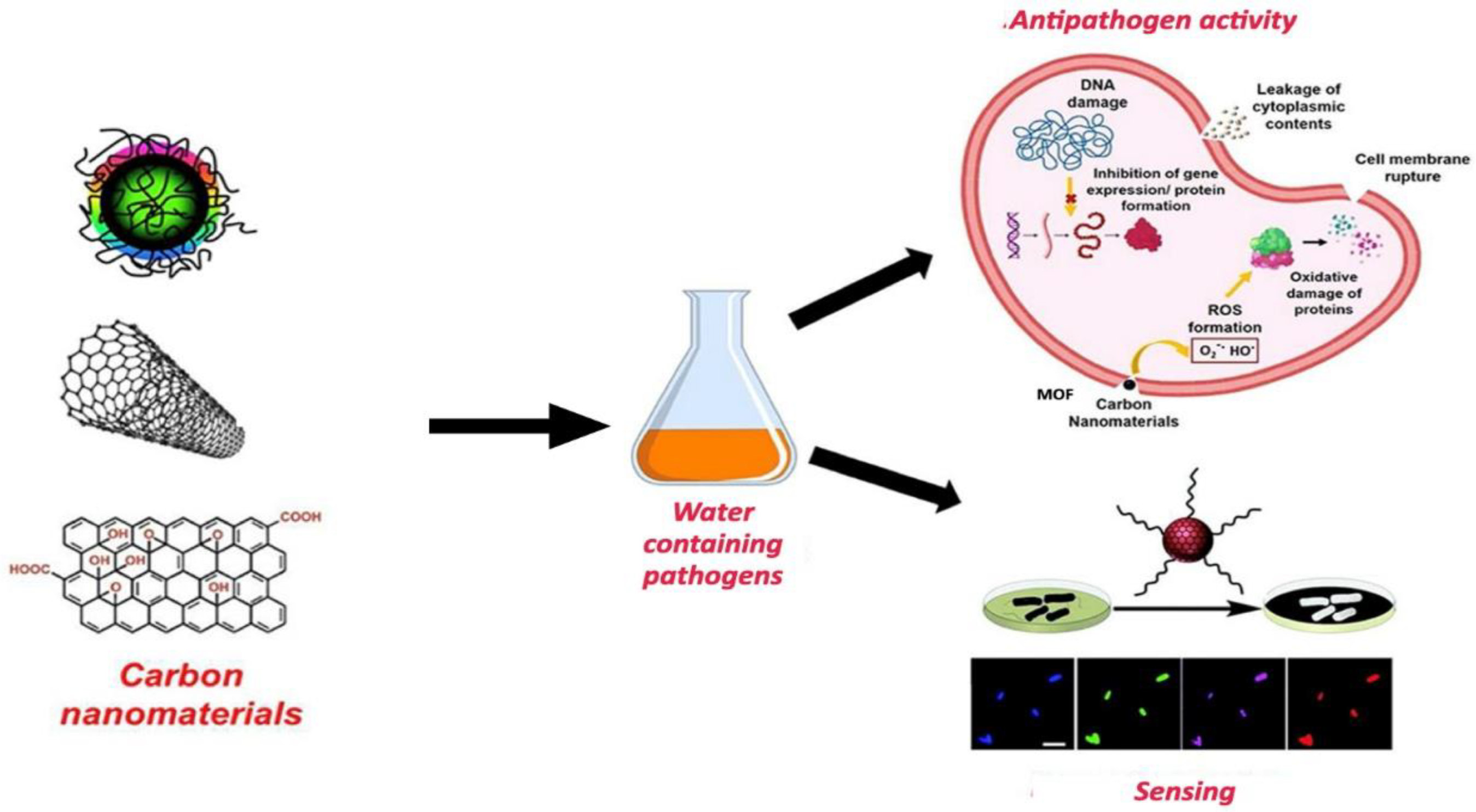
| CBN Type | Target Pollutants | Key Advantages | Main Limitations |
| Activated Carbon | Organic dyes, heavy metals, VOCs | High surface area, low cost, widely available | Poor regeneration, limited selectivity [306]. |
| Fullerenes (C60, etc.) | Pharmaceuticals, pathogens | Good redox properties, antiviral and antibacterial activity | Expensive, limited water dispersion |
| CNTs | Heavy metals, organic pollutants, pathogens | High mechanical strength, effective adsorbent and antimicrobial | High cost, toxicity, separation issues |
| CQDs | Dyes, pharmaceuticals, sensors | Photocatalytic activity, low toxicity, useful in sensing | Lower adsorption capacity than GO/CNTs |
| GO | Heavy metals, dyes, antibiotics | Tunable surface chemistry, strong adsorption, antimicrobial | Potential toxicity, costly synthesis [307]. |
| RGO | Organic micropollutants, heavy metals | Conductive, good electron transfer (useful in Advanced Oxidation Process) | Agglomeration in water, harder to disperse |
| Integration | Application Mode | Key Advantages | Main Challenges |
| Magnetic composites (e.g., GO-Fe₃O₄) | Recovery & reuse | Easy separation with magnets | Reduced surface area, synthesis issues |
| Embedded in composites | Continuous flow reactors | Enhanced selectivity, easier handling | Material compatibility, cost |
| Coated on membranes | Filtration + adsorption | Reduced fouling, reusable | Membrane fabrication complexity |
| Dispersed in batch systems | Adsorption, disinfection | Simple setup, direct contact | Difficult to recover CBNs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




