Submitted:
02 October 2025
Posted:
04 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
| Model | Altered genotype | Narcoleptic-like phenotype† | Advantages | Disadvantages | Ref. | |||||||
| Sleep-wake fragmentation | SOREMs | Wakefulness in the active phase |
Total REM time | Cataplexy | Obesity | |||||||
| Canine | Mutation in OX2 gene | + | + | ↓ | + | Spontaneous genetic mutation | Cost and care; behavioural variability | [17] | ||||
| Murine | Knockout | OX-/- | Prepro-orexin gene knockout | + | + | ↓ | ↑ | + | + | Replicates key narcoleptic symptoms | Lacks the progressive degeneration | [18] |
| OX/AT3 | Selectively ablation of orexin cells through the expression of ataxin-3 transgene causes apoptosis. | + | + | ↓ | ↑ | + | + | Mimics the progressive loss of orexin neurons | Complex to establish; lacks the immune component | [13] | ||
| OX1R-/- | Single or double knockout for orexin receptors | + | Helpful in studying the specific role of OX1R and OX2R | Lacks progressive degeneration; less severe phenotype in single KOs | [19] | |||||||
| OX2R-/- | + | + | [12] | |||||||||
| OX1R-/- OX2R-/- | + | + | ↓ | ↑ | + | [20] | ||||||
| O/E3-/- | O/E3 transcription factor knockout | + | + | ↓ | ↑ | + | Useful in studying the role of O/E3 in regulating sleep-wake controlling neurons. | Broader developmental issues | [21] | |||
| Controlled | OX2R–TD | loxP-flanked transcription- disrupter gene cassette that prevents expression of OX2R |
+ | ↓ | + | Selective and reversible disruption of OX2R in specific regions of the brain | It does not fully replicate the whole narcolepsy phenotype | [22] | ||||
| OX-tTA TetO-DTA | Induction of diphtheria toxin A (DTA) in orexin neurons via tetracycline-transactivator system (tTA) | + | + | ↓ | ↑ | + | + | The extent and timing of neuronal ablation can be controlled; more severe narcolepsy phenotype |
Complex to breed and manage; requires precise regulation of toxin expression | [23] | ||
| Optogenetic | OX/HaloR | Expresses halorhodopsin (HaloR) in orexin neurons | ↓ | Allows real-time control of neuronal activation or inhibition | Involves surgical implantation of optical fibre; does not present full narcolepsy phenotype | [24] | ||||||
| OX/Arch | Expresses archaerhodopsin-3 (Arch) in orexin neurons | + | ↓ | ↑ | + | [25] | ||||||
| OX-tTA TetO-ArchT | Expresses ArchT using the tet-off (tTA) system | + | ↓ | [26] | ||||||||
| Immune-driven | H1N1 infection | Orexin neuron ablation in Rag1-/- mice through H1N1 infection | + | + | ↓ | ↑ | Mimics autoimmune aspects of narcolepsy | Complex to generate; potential variability in immune response; limited immune system representation | [27] | |||
| OX-HA | Expresses hemagglutinin (HA) as a neo-self-antigen in orexin neurons | + | [28] | |||||||||
| †Present (+); increased (↑); decreased (↓). | ||||||||||||
2. Neurochemical Imbalances in Canine Narcolepsy: Insights from Pharmacology
3. Identifying the Genetic Defect in Canine Narcolepsy: Mutation in the Hypocretin-2 Receptor
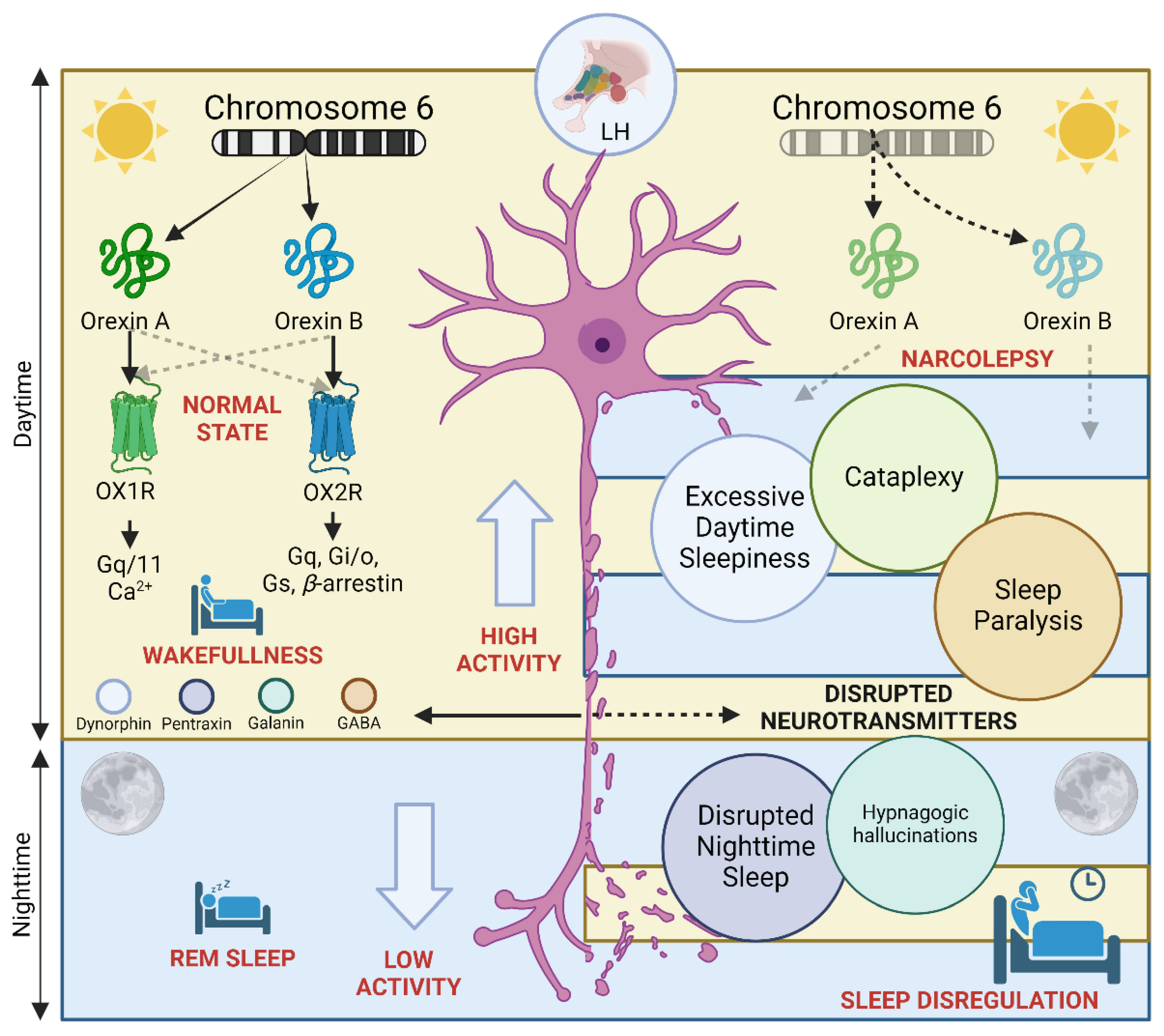
4. Hypocretin and Narcolepsy
4.1. Orexin Receptors and Their Ligands: Molecular Foundations of Arousal Regulation
4.2. Hypocretin Neurons as Integrators of Wakefulness Circuitry
4.3. Towards a Paradigm Shift
5. Murine Models of Narcolepsy
5.1. Sleep Attacks and Narcoleptic Phenotypes in Prepro-Orexin Knockout Mice
5.2. Post-Mortem and Biomarker Evidence in Humans
5.3. The Orexin/Ataxin-3 (ATAX) Model: Neuronal Ablation and Disease Evolution
5.4. Receptor Genetics: Parsing OX1R and OX2R Contributions
6. Other Rodent Models Informing Pathogenesis
6.1. O/E3 and Hypocretin Lineage Differentiation
6.2. Autoimmunity, HLA, and T-Cell Biology
6.3. Distinct Yet Intertwined: MCH and Hypocretin Neurons
6.4. Dual Ablation of Hypocretin and MCH Neurons
6.5. Translational Inflexion: From Models to Disease Modification
7. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
Abbreviations
| REM | Rapid Eye Movement |
| NT1 | Narcolepsy Type 1 |
| NT2 | Narcolepsy Type 2 |
| MCH | Melanin-Concentrating Hormone |
| EDS | Excessive Daytime Sleepiness |
| HLA | Human Leukocyte Antigen |
| CD8 | Cluster of Differentiation 8 (cytotoxic T cell subset) |
References
- Ortega-Robles, E.; Guerra-Crespo, M.; Ezzeldin, S.; Santana-Roman, E.; Palasz, A.; Salama, M.; Arias-Carrion, O. Orexin Restoration in Narcolepsy: Breakthroughs in Cellular Therapy. J Sleep Res 2025. [CrossRef]
- Arias-Carrion, O. Preclinical models of insomnia: advances, limitations, and future directions for drug discovery. Expert Opin Drug Discov 2025, 20, 1061-1074. [CrossRef]
- Bassetti, C.L.A.; Adamantidis, A.; Burdakov, D.; Han, F.; Gay, S.; Kallweit, U.; Khatami, R.; Koning, F.; Kornum, B.R.; Lammers, G.J., et al. Narcolepsy - clinical spectrum, aetiopathophysiology, diagnosis and treatment. Nat. Rev. Neurol. 2019, 15, 519-539. [CrossRef]
- Kornum, B.R.; Knudsen, S.; Ollila, H.M.; Pizza, F.; Jennum, P.J.; Dauvilliers, Y.; Overeem, S. Narcolepsy. Nature Reviews Disease Primers 2017, 3, 16100. [CrossRef]
- Kumar, S.; Sagili, H. Etiopathogenesis and neurobiology of narcolepsy: a review. Journal of Clinical and Diagnostic Research 2014, 8, 190-195. [CrossRef]
- Maycock, T.J.; Rossor, T.; Vanegas, M.; Gringras, P.; Jungbluth, H. Child Neurology: Common Occurrence of Narcolepsy Type 1 and Myasthenia Gravis. Neurology 2024, 103, e209598. [CrossRef]
- Vringer, M.; Zhou, J.; Gool, J.K.; Bijlenga, D.; Lammers, G.J.; Fronczek, R.; Schinkelshoek, M.S. Recent insights into the pathophysiology of narcolepsy type 1. Sleep Med Rev 2024, 78, 101993. [CrossRef]
- Latorre, D.; Sallusto, F.; Bassetti, C.L.A.; Kallweit, U. Narcolepsy: a model interaction between immune system, nervous system, and sleep-wake regulation. Semin Immunopathol 2022, 44, 611-623. [CrossRef]
- Ollila, H.M.; Sharon, E.; Lin, L.; Sinnott-Armstrong, N.; Ambati, A.; Yogeshwar, S.M.; Hillary, R.P.; Jolanki, O.; Faraco, J.; Einen, M., et al. Narcolepsy risk loci outline role of T cell autoimmunity and infectious triggers in narcolepsy. Nat Commun 2023, 14, 2709. [CrossRef]
- Stephansen, J.B.; Olesen, A.N.; Olsen, M.; Ambati, A.; Leary, E.B.; Moore, H.E.; Carrillo, O.; Lin, L.; Han, F.; Yan, H., et al. Neural network analysis of sleep stages enables efficient diagnosis of narcolepsy. Nat Commun 2018, 9, 5229. [CrossRef]
- Lin, L.; Faraco, J.; Li, R.; Kadotani, H.; Rogers, W.; Lin, X.; Qiu, X.; de Jong, P.J.; Nishino, S.; Mignot, E. The sleep disorder canine narcolepsy is caused by a mutation in the hypocretin (orexin) receptor 2 gene. Cell 1999, 98, 365-376. [CrossRef]
- Willie, J.T.; Chemelli, R.M.; Sinton, C.M.; Tokita, S.; Williams, S.C.; Kisanuki, Y.Y.; Marcus, J.N.; Lee, C.; Elmquist, J.K.; Kohlmeier, K.A., et al. Distinct narcolepsy syndromes in Orexin receptor-2 and Orexin null mice: molecular genetic dissection of Non-REM and REM sleep regulatory processes. Neuron 2003, 38, 715-730. [CrossRef]
- Hara, J.; Beuckmann, C.T.; Nambu, T.; Willie, J.T.; Chemelli, R.M.; Sinton, C.M.; Sugiyama, F.; Yagami, K.; Goto, K.; Yanagisawa, M., et al. Genetic ablation of orexin neurons in mice results in narcolepsy, hypophagia, and obesity. Neuron 2001, 30, 345-354. [CrossRef]
- Bonvalet, M.; Ollila, H.M.; Ambati, A.; Mignot, E. Autoimmunity in narcolepsy. Curr Opin Pulm Med 2017, 23, 522-529. [CrossRef]
- Huang, L.; Lai, X.; Liang, X.; Chen, J.; Yang, Y.; Xu, W.; Qin, Q.; Qin, R.; Huang, X.; Xie, M., et al. A promise for neuronal repair: reprogramming astrocytes into neurons in vivo. Biosci Rep 2024, 44. [CrossRef]
- Merkle, F.T.; Maroof, A.; Wataya, T.; Sasai, Y.; Studer, L.; Eggan, K.; Schier, A.F. Generation of neuropeptidergic hypothalamic neurons from human pluripotent stem cells. Development 2015, 142, 633-643. [CrossRef]
- Nishino, S. Canine models of narcolepsy. In The Orexin/Hypocretin System: Physiology and Pathophysiology, Nishino, S., Sakurai, T., Eds. Humana Press: Totowa, NJ, 2006; pp. 233-253. [CrossRef]
- Chemelli, R.M.; Willie, J.T.; Sinton, C.M.; Elmquist, J.K.; Scammell, T.; Lee, C.; Richardson, J.A.; Williams, S.C.; Xiong, Y.; Kisanuki, Y., et al. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell 1999, 98, 437-451. [CrossRef]
- Kisanuki, Y.; Chemelli, R.; Sinton, C.; Williams, S.; Richardson, J.; Hammer, R.; Yanagisawa, M. The role of orexin receptor type-1 (OX1R) in the regulation of sleep. Sleep 2000, 23, A91.
- Kisanuki, Y.Y.; Chemelli, R.M.; Tokita, S.; Willie, J.T.; Sinton, C.M.; Yanagisawa, M. Behavioral and polysomnographic characterization of orexin-1 receptor and orexin-2 receptor double knockout mice. Sleep 2001, 24, A22.
- De La Herrán-Arita, A.K.; Zomosa-Signoret, V.C.; Millán-Aldaco, D.A.; Palomero-Rivero, M.; Guerra-Crespo, M.; Drucker-Colín, R.; Vidaltamayo, R. Aspects of the narcolepsy-cataplexy syndrome in O/E3-null mutant mice. Neuroscience 2011, 183, 134-143. [CrossRef]
- Mochizuki, T.; Arrigoni, E.; Marcus, J.N.; Clark, E.L.; Yamamoto, M.; Honer, M.; Borroni, E.; Lowell, B.B.; Elmquist, J.K.; Scammell, T.E. Orexin receptor 2 expression in the posterior hypothalamus rescues sleepiness in narcoleptic mice. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 4471-4476. [CrossRef]
- Tabuchi, S.; Tsunematsu, T.; Black, S.W.; Tominaga, M.; Maruyama, M.; Takagi, K.; Minokoshi, Y.; Sakurai, T.; Kilduff, T.S.; Yamanaka, A. Conditional ablation of orexin/hypocretin neurons: a new mouse model for the study of narcolepsy and orexin system function. J. Neurosci. 2014, 34, 6495-6509. [CrossRef]
- Tsunematsu, T.; Kilduff, T.S.; Boyden, E.S.; Takahashi, S.; Tominaga, M.; Yamanaka, A. Acute optogenetic silencing of orexin/hypocretin neurons induces slow-wave sleep in mice. J. Neurosci. 2011, 31, 10529-10539. [CrossRef]
- Williams, R.H.; Tsunematsu, T.; Thomas, A.M.; Bogyo, K.; Yamanaka, A.; Kilduff, T.S. Transgenic Archaerhodopsin-3 Expression in Hypocretin/Orexin Neurons Engenders Cellular Dysfunction and Features of Type 2 Narcolepsy. J. Neurosci. 2019, 39, 9435-9452. [CrossRef]
- Tsunematsu, T.; Tabuchi, S.; Tanaka, K.F.; Boyden, E.S.; Tominaga, M.; Yamanaka, A. Long-lasting silencing of orexin/hypocretin neurons using archaerhodopsin induces slow-wave sleep in mice. Behav. Brain Res. 2013, 255, 64-74. [CrossRef]
- Tesoriero, C.; Codita, A.; Zhang, M.D.; Cherninsky, A.; Karlsson, H.; Grassi-Zucconi, G.; Bertini, G.; Harkany, T.; Ljungberg, K.; Liljeström, P., et al. H1N1 influenza virus induces narcolepsy-like sleep disruption and targets sleep-wake regulatory neurons in mice. Proc. Natl. Acad. Sci. U. S. A. 2016, 113, E368-377. [CrossRef]
- Bernard-Valnet, R.; Yshii, L.; Quériault, C.; Nguyen, X.H.; Arthaud, S.; Rodrigues, M.; Canivet, A.; Morel, A.L.; Matthys, A.; Bauer, J., et al. CD8 T cell-mediated killing of orexinergic neurons induces a narcolepsy-like phenotype in mice. Proc. Natl. Acad. Sci. U. S. A. 2016, 113, 10956-10961. [CrossRef]
- Mignot, E.J. History of narcolepsy at Stanford University. Immunol Res 2014, 58, 315-339. [CrossRef]
- Black, J.; Reaven, N.L.; Funk, S.E.; McGaughey, K.; Ohayon, M.M.; Guilleminault, C.; Ruoff, C. Medical comorbidity in narcolepsy: findings from the Burden of Narcolepsy Disease (BOND) study. Sleep Med 2017, 33, 13-18. [CrossRef]
- Nishino, S.; Okura, M.; Mignot, E. Narcolepsy: genetic predisposition and neuropharmacological mechanisms. REVIEW ARTICLE. Sleep Med Rev 2000, 4, 57-99. [CrossRef]
- Mignot, E. Genetic and familial aspects of narcolepsy. Neurology 1998, 50, S16-22. [CrossRef]
- de Lecea, L.; Kilduff, T.S.; Peyron, C.; Gao, X.; Foye, P.E.; Danielson, P.E.; Fukuhara, C.; Battenberg, E.L.; Gautvik, V.T.; Bartlett, F.S., 2nd, et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 322-327. [CrossRef]
- Sakurai, T.; Amemiya, A.; Ishii, M.; Matsuzaki, I.; Chemelli, R.M.; Tanaka, H.; Williams, S.C.; Richardson, J.A.; Kozlowski, G.P.; Wilson, S., et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 1998, 92, 573-585. [CrossRef]
- Wang, C.; Wang, Q.; Ji, B.; Pan, Y.; Xu, C.; Cheng, B.; Bai, B.; Chen, J. The Orexin/Receptor System: Molecular Mechanism and Therapeutic Potential for Neurological Diseases. Front Mol Neurosci 2018, 11, 220. [CrossRef]
- Mahler, S.V.; Smith, R.J.; Moorman, D.E.; Sartor, G.C.; Aston-Jones, G. Multiple roles for orexin/hypocretin in addiction. Prog Brain Res 2012, 198, 79-121. [CrossRef]
- Ishikawa, T.; Kurimoto, E.; Joyal, A.A.; Koike, T.; Kimura, H.; Scammell, T.E. An orexin agonist promotes wakefulness and inhibits cataplexy through distinct brain regions. Curr Biol 2025, 35, 2088-2099 e2084. [CrossRef]
- Scammell, T.E. Narcolepsy. N. Engl. J. Med. 2015, 373, 2654-2662. [CrossRef]
- Brown, R.E.; Basheer, R.; McKenna, J.T.; Strecker, R.E.; McCarley, R.W. Control of sleep and wakefulness. Physiol Rev 2012, 92, 1087-1187. [CrossRef]
- Sutcliffe, J.G.; de Lecea, L. The hypocretins: setting the arousal threshold. Nat Rev Neurosci 2002, 3, 339-349. [CrossRef]
- Ripley, B.; Overeem, S.; Fujiki, N.; Nevsimalova, S.; Uchino, M.; Yesavage, J.; Di Monte, D.; Dohi, K.; Melberg, A.; Lammers, G.J., et al. CSF hypocretin/orexin levels in narcolepsy and other neurological conditions. Neurology 2001, 57, 2253-2258. [CrossRef]
- Postiglione, E.; Barateau, L.; Pizza, F.; Lopez, R.; Antelmi, E.; Rassu, A.L.; Vandi, S.; Chenini, S.; Mignot, E.; Dauvilliers, Y., et al. Narcolepsy with intermediate cerebrospinal level of hypocretin-1. Sleep 2022, 45. [CrossRef]
- Thannickal, T.C.; Moore, R.Y.; Nienhuis, R.; Ramanathan, L.; Gulyani, S.; Aldrich, M.; Cornford, M.; Siegel, J.M. Reduced number of hypocretin neurons in human narcolepsy. Neuron 2000, 27, 469-474. [CrossRef]
- Shan, L.; Linssen, S.; Harteman, Z.; den Dekker, F.; Shuker, L.; Balesar, R.; Breesuwsma, N.; Anink, J.; Zhou, J.; Lammers, G.J., et al. Activated Wake Systems in Narcolepsy Type 1. Ann Neurol 2023, 94, 762-771. [CrossRef]
- Tisdale, R.K.; Yamanaka, A.; Kilduff, T.S. Animal models of narcolepsy and the hypocretin/orexin system: Past, present, and future. Sleep 2021, 44. [CrossRef]
- Miller, E.; Andrews, N.; Stellitano, L.; Stowe, J.; Winstone, A.M.; Shneerson, J.; Verity, C. Risk of narcolepsy in children and young people receiving AS03 adjuvanted pandemic A/H1N1 2009 influenza vaccine: retrospective analysis. BMJ 2013, 346, f794. [CrossRef]
- Sarkanen, T.O.; Alakuijala, A.P.E.; Dauvilliers, Y.A.; Partinen, M.M. Incidence of narcolepsy after H1N1 influenza and vaccinations: Systematic review and meta-analysis. Sleep Med Rev 2018, 38, 177-186. [CrossRef]
- Bernard-Valnet, R.; Yshii, L.; Queriault, C.; Nguyen, X.H.; Arthaud, S.; Rodrigues, M.; Canivet, A.; Morel, A.L.; Matthys, A.; Bauer, J., et al. CD8 T cell-mediated killing of orexinergic neurons induces a narcolepsy-like phenotype in mice. Proc Natl Acad Sci U S A 2016, 113, 10956-10961. [CrossRef]
- Latorre, D.; Kallweit, U.; Armentani, E.; Foglierini, M.; Mele, F.; Cassotta, A.; Jovic, S.; Jarrossay, D.; Mathis, J.; Zellini, F., et al. T cells in patients with narcolepsy target self-antigens of hypocretin neurons. Nature 2018, 562, 63-68. [CrossRef]
- Pedersen, N.W.; Holm, A.; Kristensen, N.P.; Bjerregaard, A.M.; Bentzen, A.K.; Marquard, A.M.; Tamhane, T.; Burgdorf, K.S.; Ullum, H.; Jennum, P., et al. CD8(+) T cells from patients with narcolepsy and healthy controls recognize hypocretin neuron-specific antigens. Nat Commun 2019, 10, 837. [CrossRef]
- Konadhode, R.R.; Pelluru, D.; Blanco-Centurion, C.; Zayachkivsky, A.; Liu, M.; Uhde, T.; Glen, W.B., Jr.; van den Pol, A.N.; Mulholland, P.J.; Shiromani, P.J. Optogenetic stimulation of MCH neurons increases sleep. J Neurosci 2013, 33, 10257-10263. [CrossRef]
- Izawa, S.; Chowdhury, S.; Miyazaki, T.; Mukai, Y.; Ono, D.; Inoue, R.; Ohmura, Y.; Mizoguchi, H.; Kimura, K.; Yoshioka, M., et al. REM sleep-active MCH neurons are involved in forgetting hippocampus-dependent memories. Science 2019, 365, 1308-1313. [CrossRef]
- Hung, C.J.; Ono, D.; Kilduff, T.S.; Yamanaka, A. Dual orexin and MCH neuron-ablated mice display severe sleep attacks and cataplexy. Elife 2020, 9. [CrossRef]
- Evans, R.; Kimura, H.; Alexander, R.; Davies, C.H.; Faessel, H.; Hartman, D.S.; Ishikawa, T.; Ratti, E.; Shimizu, K.; Suzuki, M., et al. Orexin 2 receptor-selective agonist danavorexton improves narcolepsy phenotype in a mouse model and in human patients. Proc Natl Acad Sci U S A 2022, 119, e2207531119. [CrossRef]
- Dauvilliers, Y.; Plazzi, G.; Mignot, E.; Lammers, G.J.; Del Rio Villegas, R.; Khatami, R.; Taniguchi, M.; Abraham, A.; Hang, Y.; Kadali, H., et al. Oveporexton, an Oral Orexin Receptor 2-Selective Agonist, in Narcolepsy Type 1. N Engl J Med 2025, 392, 1905-1916. [CrossRef]
- Scammell, T.E. The neurobiology, diagnosis, and treatment of narcolepsy. Ann. Neurol. 2003, 53, 154-166. [CrossRef]
- van der Hoeven, A.E.; Fronczek, R.; Schinkelshoek, M.S.; Roelandse, F.W.C.; Bakker, J.A.; Overeem, S.; Bijlenga, D.; Lammers, G.J. Intermediate hypocretin-1 cerebrospinal fluid levels and typical cataplexy: their significance in the diagnosis of narcolepsy type 1. Sleep 2022, 45. [CrossRef]
| Models and Mechanisms | Therapies and Future Directions |
|---|---|
| Genetic and neuronal models — including prepro-orexin knockouts, orexin/ataxin-3 ablations, receptor mutants, and canine Hcrtr2 lines — have redefined narcolepsy as a disorder of hypocretin deficiency. These models provided face validity and mechanistic depth but remain locked in descriptive paradigms. | Small-molecule OX2R agonists — Danavorexton and oveporexton deliver the first truly mechanism-targeted therapies in sleep medicine, improving vigilance and reducing cataplexy. They signal a pivot from symptom management toward disease modification. |
| Immune relevance — Human HLA signatures, autoreactive T cells, and Orexin-HA mice prove that adaptive immunity can selectively erase hypocretin neurons. This finding reframes narcolepsy type 1 (NT1) as an autoimmune encephalopathy, placing it within the broader landscape of organ-specific autoimmunity. | Gene therapy — Viral hypocretin delivery or receptor reconstitution offers the possibility of durable circuit repair, with narcolepsy positioned to become the first neuropsychiatric disease amenable to one-time molecular correction. |
| Circuit tools, including optogenetics, chemogenetics, and conditional ablations, have mapped the causal architecture of sleep–wake transitions. Nevertheless, their artificial timing and reversibility risk trivialize a degenerative, chronic disorder into an on–off switch. | Cellular therapies — including stem-cell–derived hypothalamic neurons, astrocyte-to-neuron reprogramming, and patient-specific organoids — foreshadow a regenerative neurology, where lost arousal circuits are rebuilt rather than merely pharmacologically bypassed. |
| Phenotypic blind spots — Current models exaggerate cataplexy and under-represent narcolepsy type 2 (NT2), where partial hypocretin dysfunction, vigilance instability, and cognitive–autonomic burden dominate. Without NT2-specific paradigms, therapeutic discovery risks serving only a fraction of patients. | Integrative approaches — including multi-omics, systems biology, and AI-driven digital phenotyping — promise to bridge the gap between laboratory models and real-world heterogeneity, stratify patients, define novel biomarkers, and accelerate precision trials. |
| Trajectory — The field must break from its descriptive past. From genetic models anchoring hypocretin deficiency → to mechanistic circuit dissection → to immune-mediated causality → to regenerative and precision interventions. | Central Goal — To shift narcolepsy from a “managed” chronic disorder into one of the first preventable and curable neuropsychiatric conditions — a test case for how neuroscience can move from symptom palliation to circuit restoration and disease prevention. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




