Submitted:
02 October 2025
Posted:
03 October 2025
You are already at the latest version
Abstract
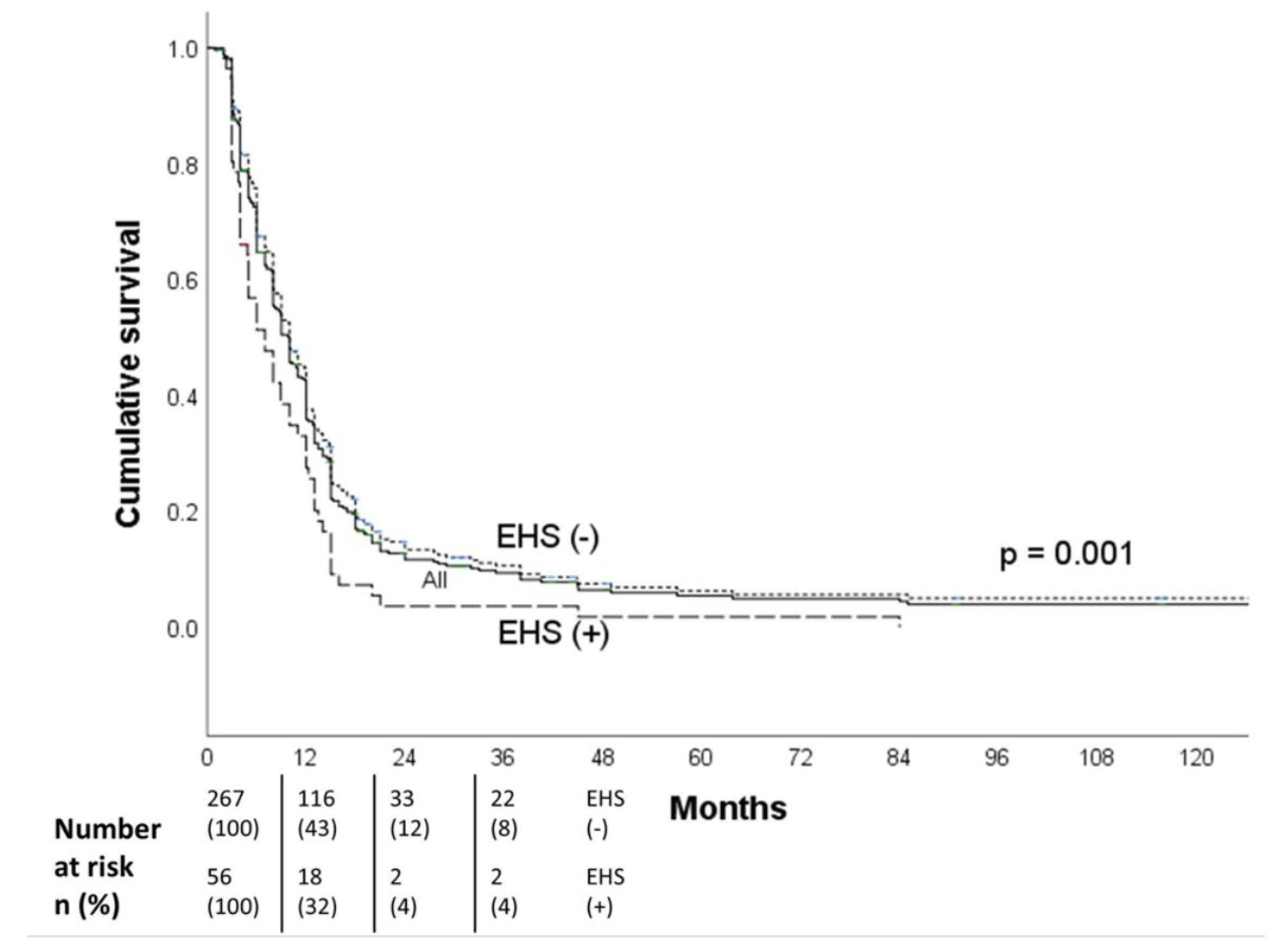
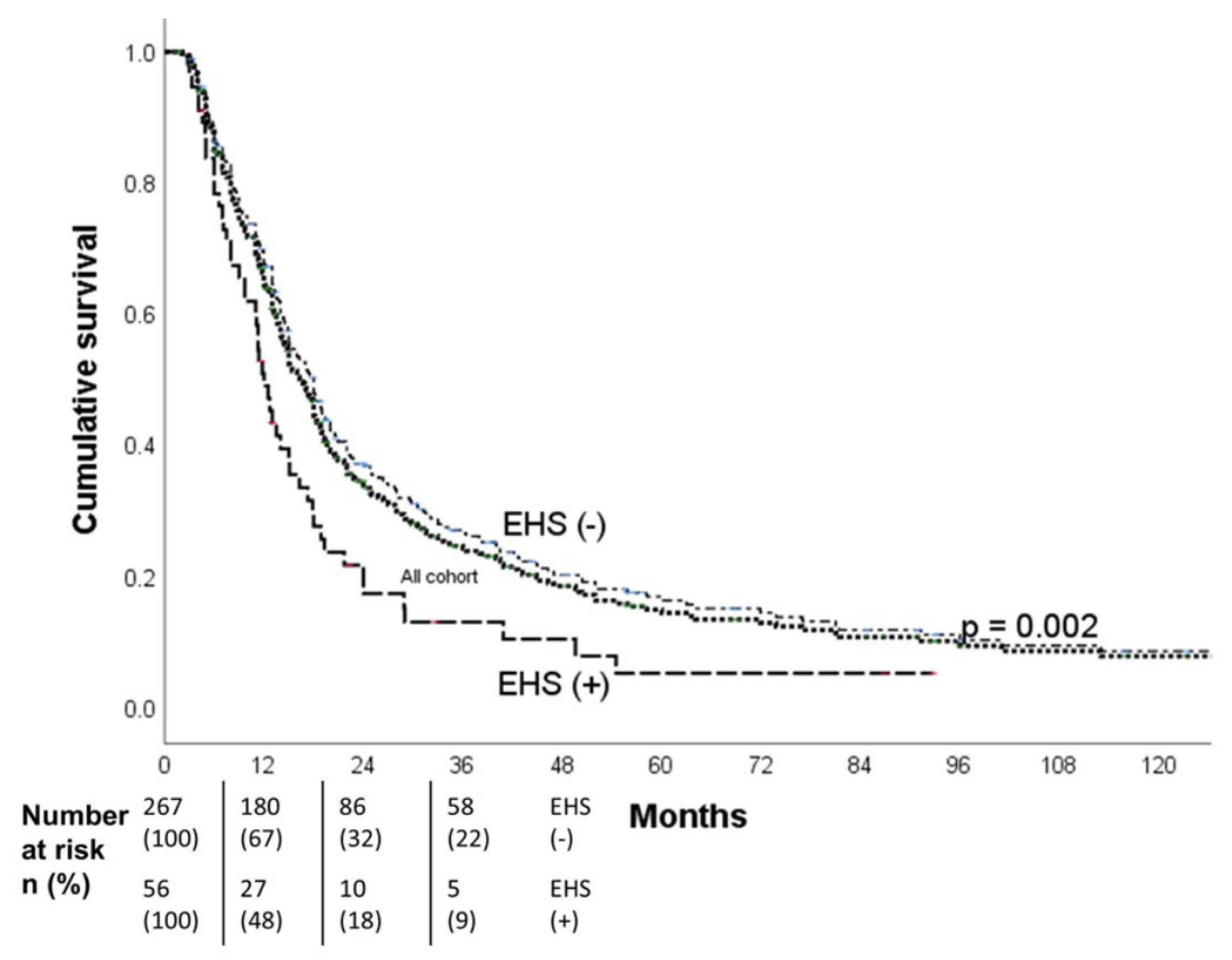
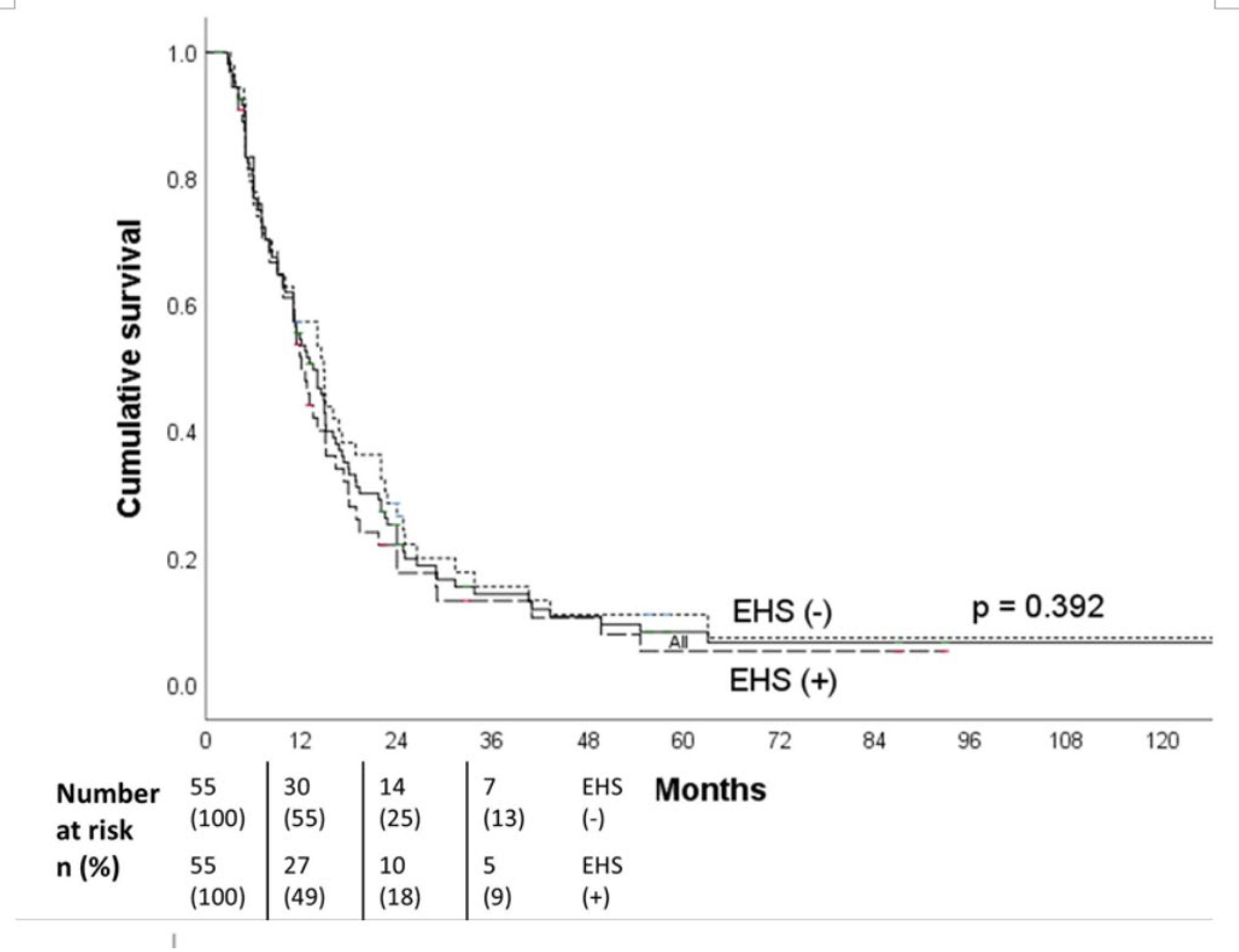
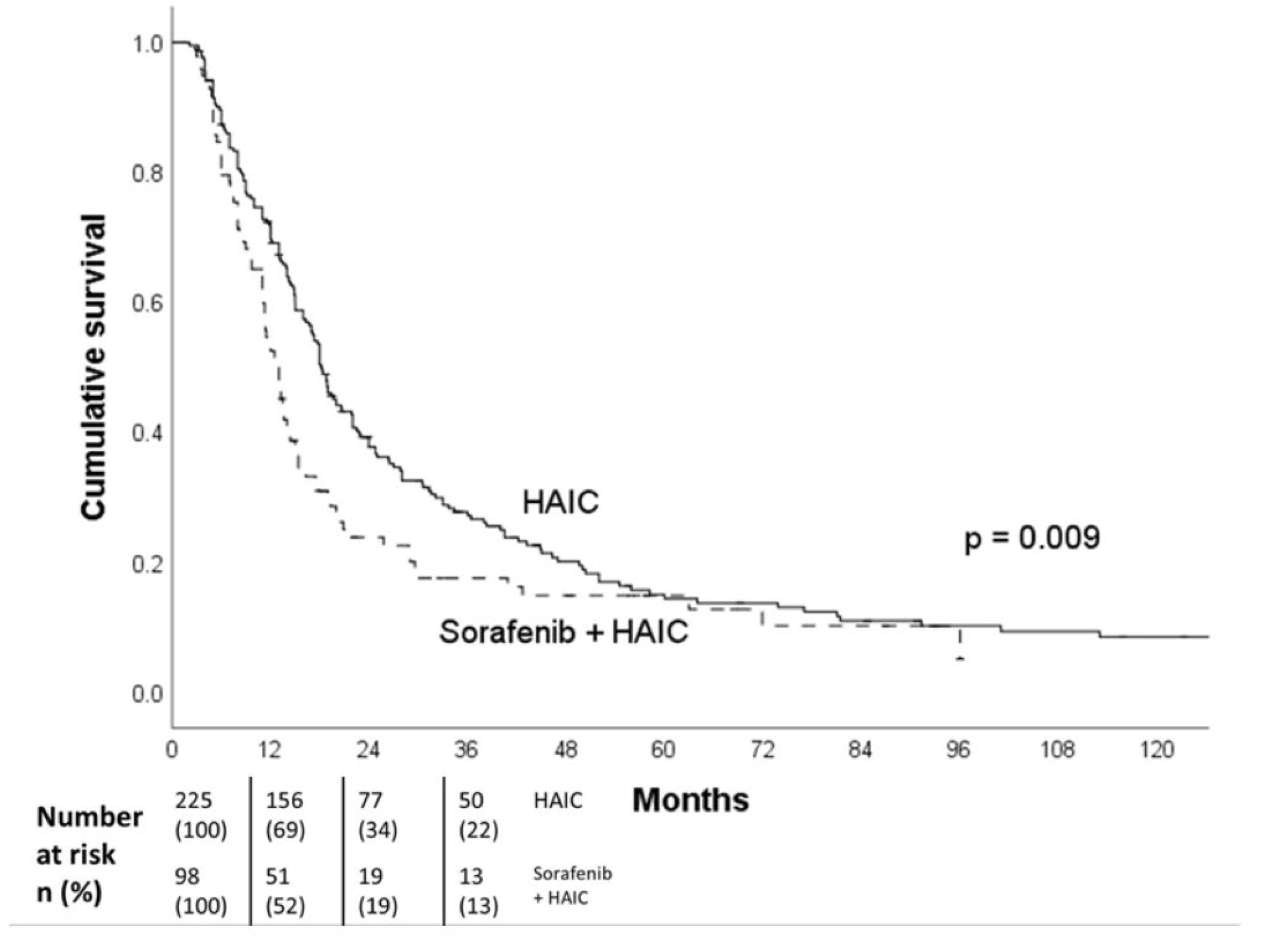
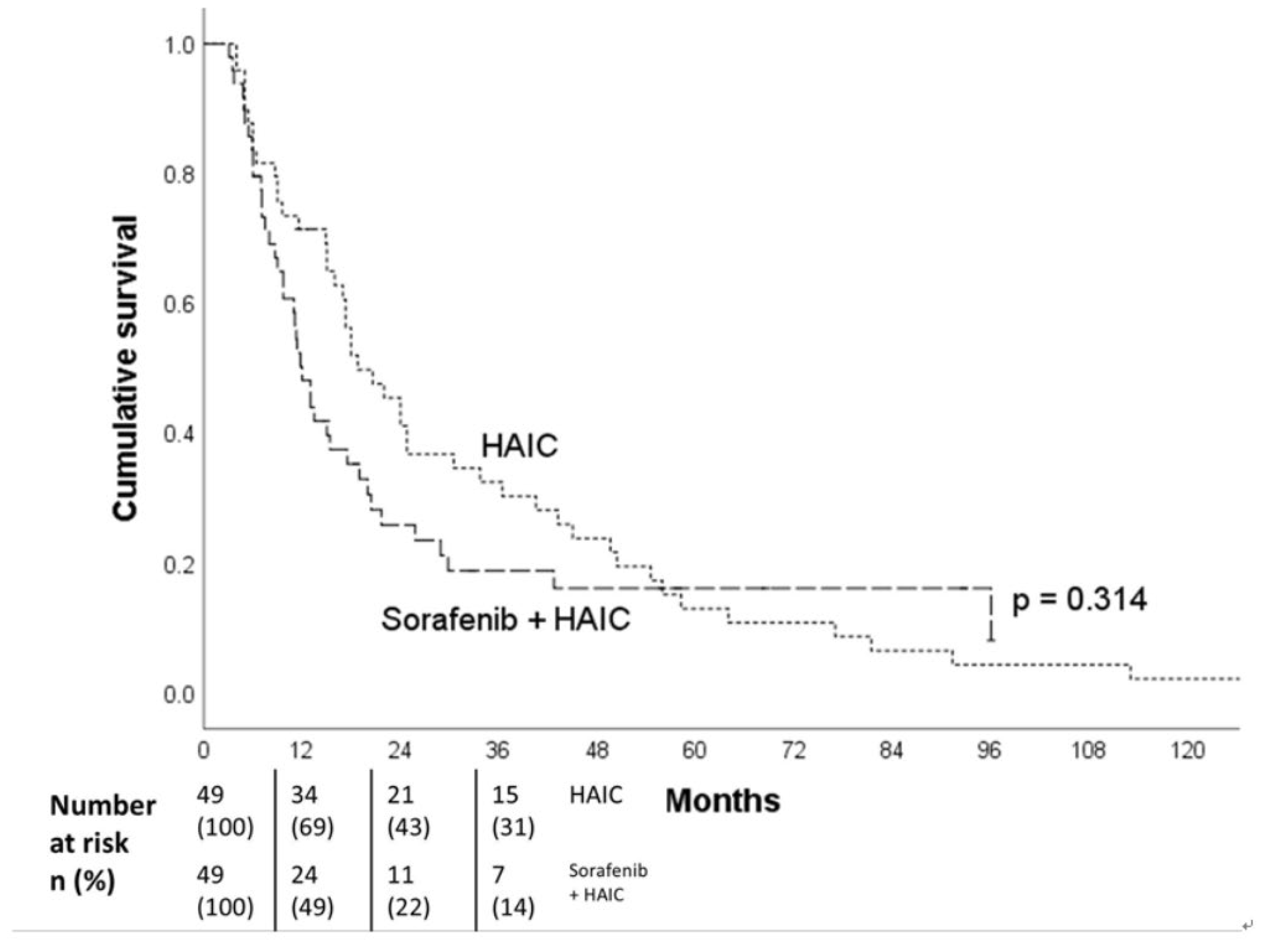
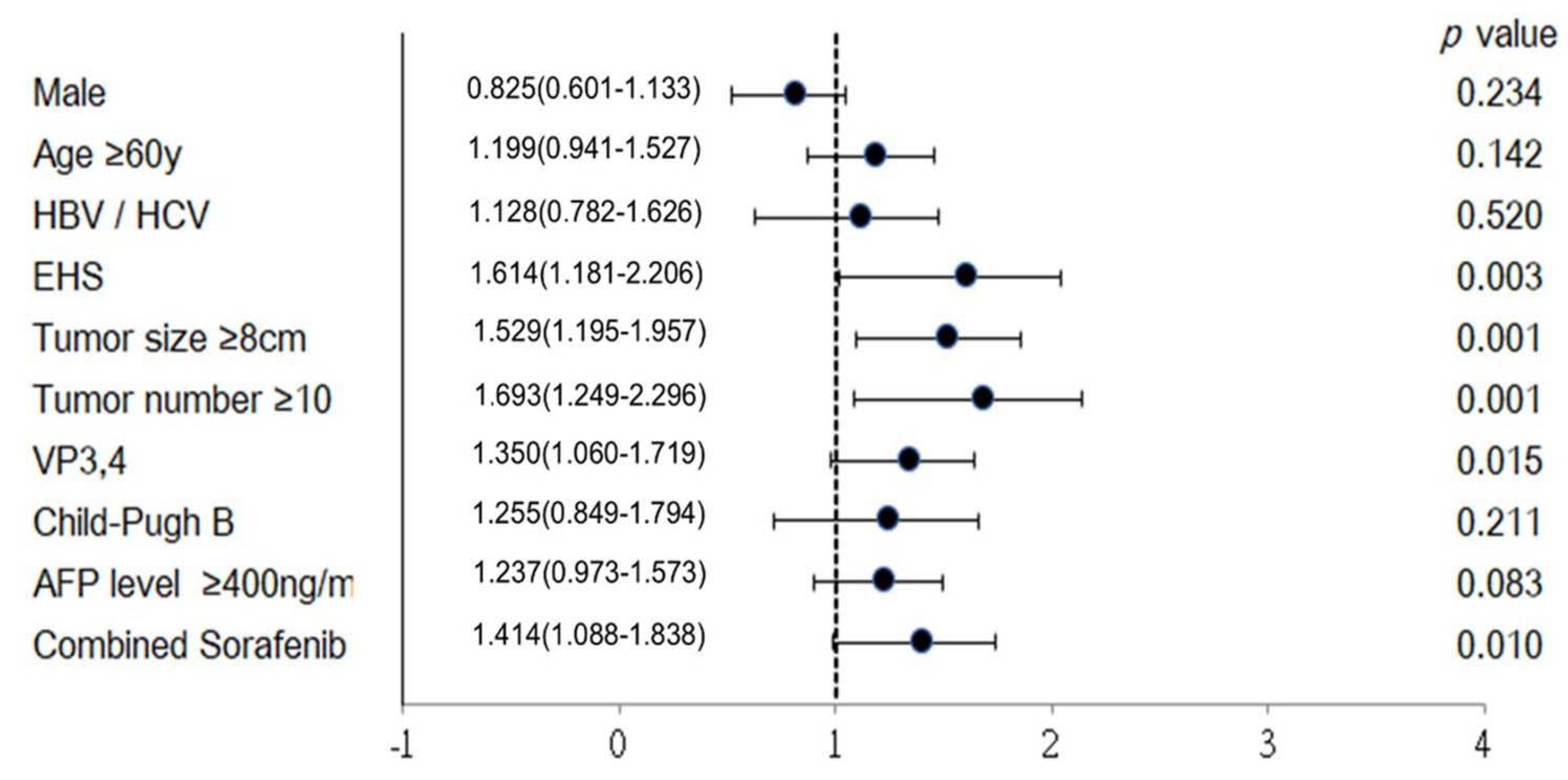
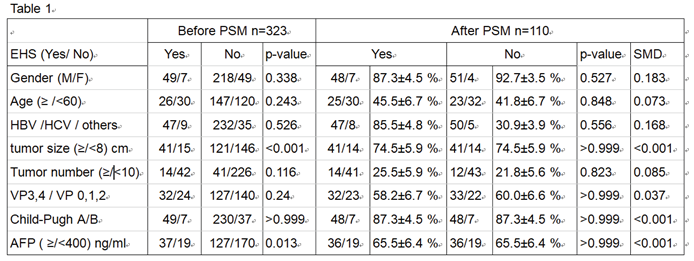
Purpose: To study the efficacy and safety of hepatic artery infusion chemotherapy (HAIC) in the treatment of unresectable hepatocellular carcinoma (HCC) with extrahepatic spread (EHS). Materials and Methods: 323 patients with unresectable HCC received HAIC plus lipiodol microvascular embolization. Treatment responses were evaluated based on mRECIST criteria. The objective response rate (ORR), progression free survival (PFS) and overall survival (OS) of patients with EHS were compared to those without. Subgroup analyses of patients with or without major portal vein tumor thrombosis (PVTT) were performed both before and after propensity score matching (PSM). The survival analysis was calculated with Kaplan-Meier method and compared using the log-rank test. All the statistical analyses were performed by SPSS (version 26.0). Result: The overall ORR was 59.1%.The median OS of the initial all cohort and patients with positive and negative EHS were 16.3, 12.0 and 18.0 months, respectively (p=0.002). In the subgroup analysis, there was no statistical difference of survival in patients with major PVTT between the with-EHS and without-EHS groups (13.0 vs 15.0 months, p=0.407). However, the median OS in patients with EHS was significantly shorter than those without EHS (11.4 vs 19.4 months, p<0.001) in the subgroup of non-major PVTT patients. After PSM, there were no significant survival differences between the EHS and non-EHS groups in any patient cohort or sub-cohort analysis. Conclusion: For unresectable HCC, controlling intrahepatic tumors through HAIC is more important than controlling extrahepatic tumors, especially in patients with major PVTT. Locoregional HAIC can be performed in patients with EHS.
Keywords:
1. Introduction
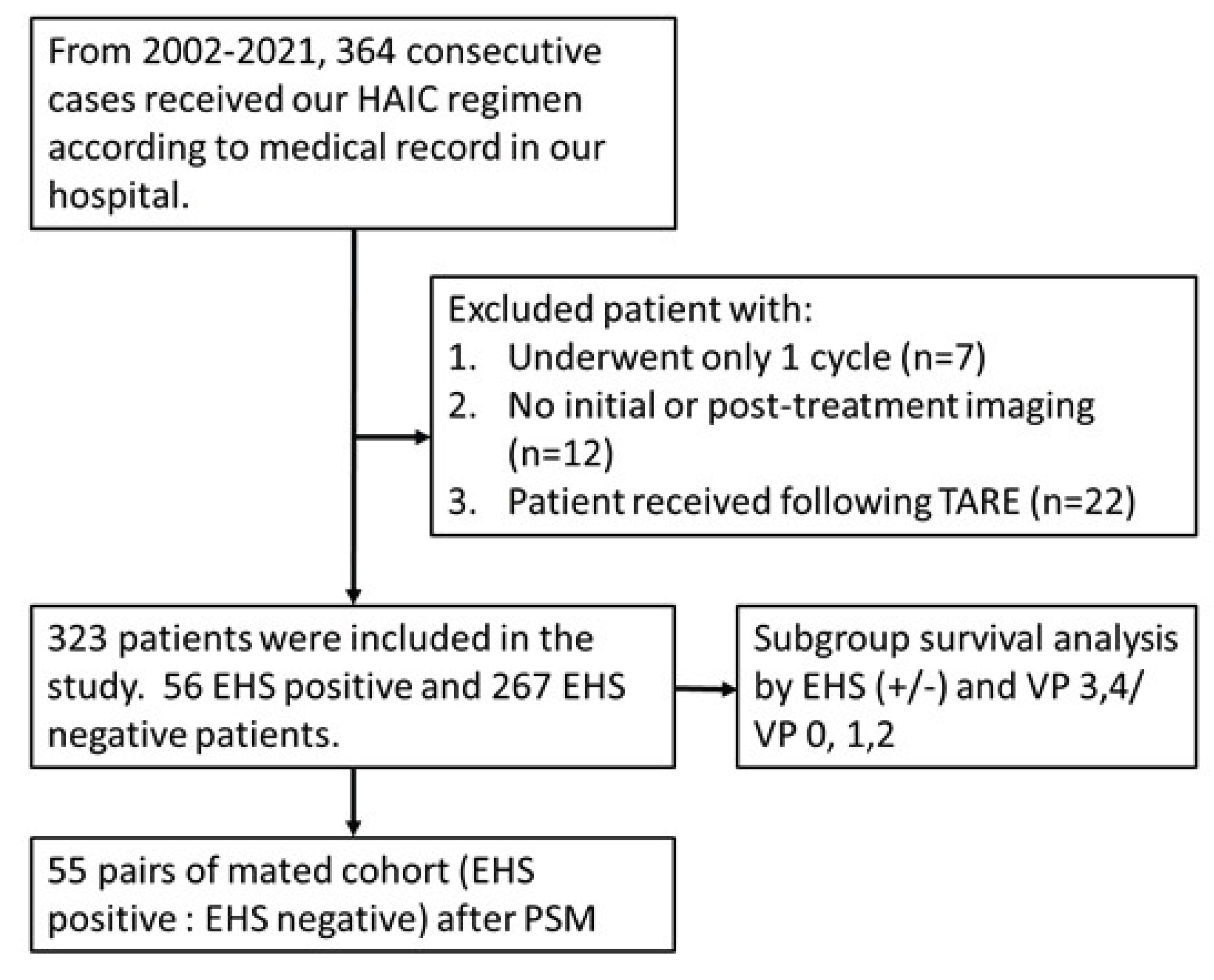
2. Method
3. Result
4. Discussion
5. Limitations
6. Conclusion
Author Contributions
Data Availability Statement
Acknowledgments
References
- Reig, M., Forner, A., Rimola, J., Ferrer-Fàbrega, J., Burrel, M., Garcia-Criado, Á., Kelley, R. K., Galle, P. R., Mazzaferro, V., Salem, R., Sangro, B., Singal, A. G., Vogel, A., Fuster, J., Ayuso, C., & Bruix, J. (2022). BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. Journal of Hepatology, 76(3), 681-693. [CrossRef]
- Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol.2009;10(1),25-34. [CrossRef]
- Rimassa L, and Santoro A.Sorafenib therapy in advanced hepatocellular carcinoma: the SHARP trial. Expert Rev Anticancer Ther. 2009;9(6), 739-745.
- Jackson R, , Psarelli EE, Berhane S, Khan H and Johnson P. Impact of Viral Status on Survival in Patients Receiving Sorafenib for Advanced Hepatocellular Cancer: A Meta-Analysis of Randomized Phase III Trials. J Clin Oncol. 2017; 35(6), 622-628.
- Nakano M, Tanaka M, Kuromatsu R, Nagamatsu H, Tajiri N, Satani M, et al. Sorafenib for the treatment of advanced hepatocellular carcinoma with extrahepatic metastasis: a prospective multicenter cohort study. Cancer Med. 2015;4(12), 1836-1843. [CrossRef]
- Chuma M, Uojima H, Hiraoka A, Kobayashi S, Toyoda H, Tada T, et al. Analysis of efficacy of lenvatinib treatment in highly advanced hepatocellular carcinoma with tumor thrombus in the main trunk of the portal vein or tumor with more than 50% liver occupation: A multicenter analysis. Hepatol Res. 2021;51(2), 201-215. [CrossRef]
- Zhuang BW, Li W, Xie XH, Hu HT, Lu MD, Xie XY. Sorafenib versus hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma: a systematic review and meta-analysis. Jpn J Clin Oncol. 2019;49(9), 845-855.
- Liu M,Shi J, Mou T, Wang Y, Wu Z, and ShenA. Systematic review of hepatic arterial infusion chemotherapy versus sorafenib in patients with hepatocellular carcinoma with portal vein tumor thrombosis. J Gastroenterol Hepatol. 2020; 35(8), 1277-1287. [CrossRef]
- Zheng K, Zhu X, Fu S, Cao G, Li WQ, Xu L, et al. Sorafenib Plus Hepatic Arterial Infusion Chemotherapy versus Sorafenib for Hepatocellular Carcinoma with Major Portal Vein Tumor Thrombosis: A Randomized Trial.Radiology. 2022;303(2), 455-464. [CrossRef]
- He M, Li Q, Zou R, Shen JX, Fang WQ, Tan GS, et al. Sorafenib Plus Hepatic Arterial Infusion of Oxaliplatin, Fluorouracil, and Leucovorin vs Sorafenib Alone for Hepatocellular Carcinoma With Portal Vein Invasion: A Randomized Clinical Trial. JAMA Oncol. 2019;5(7), 953-960.
- Iwamoto H, Niizeki T, Nagamatsu H, Ueshima K, Tani J, Kuzuya T, et al. The Clinical Impact of Hepatic Arterial Infusion Chemotherapy New-FP for Hepatocellular Carcinoma with Preserved Liver Function. Cancers (Basel). 2022;14(19), 4873. [CrossRef]
- Li MF, Liang HL, Chiang CL, Tsai WL, Chen WC, Tsai CC,et al. New Regimen of Combining Hepatic Arterial Infusion Chemotherapy and Lipiodol Embolization in Treating Hepatocellular Carcinoma with Main Portal Vein Invasion. J Pers Med 2022;13(1), 88.
- Nakashima T,Okuda K, Kojiro M, Jimi A, Yamaguchi R, Sakamoto K, et al. Pathology of hepatocellular carcinoma in Japan. 232 Consecutive cases autopsied in ten years. Cancer 1983; 51(5), 863-877.
- Kudo M,Matsui O, Izumi N, Iijima H, Kadoya M, ImaiY, et al. JSH Consensus-Based Clinical Practice Guidelines for the Management of Hepatocellular Carcinoma: 2014 Update by the Liver Cancer Study Group of Japan.Liver Cancer. 2014; 3(3-4), 458-468. [CrossRef]
- Kim B,Won JH, Kim J, Kwon Y, Cho HJ, Huh J, et al. Hepatic Arterial Infusion Chemotherapy for Advanced Hepatocellular Carcinoma: Radiologic and Clinical Factors Predictive of Survival. AJR Am J Roentgenol. 2021; 216(6), 1566-1573. [CrossRef]
- Sung PS, Yang K, Bae SH, Oh JS, Chun HJ, Nam HC, et al. Reduction of Intrahepatic Tumour by Hepatic Arterial Infusion Chemotherapy Prolongs Survival in Hepatocellular Carcinoma. Anticancer Res. 2019;39(7), 3909-3916. [CrossRef]
- Lyu N, Kong Y, Pan T, Mu L, Li S, Liu Y, et al. Hepatic Arterial Infusion of Oxaliplatin, Fluorouracil, and Leucovorin in Hepatocellular Cancer with Extrahepatic Spread. J Vasc Interv Radiol. 2019;30(3), 349-357. [CrossRef]
- Liang RB, Zhao Y, He MK, Wen DS, Bu XY, Huang YX, et al. Hepatic Arterial Infusion Chemotherapy of Oxaliplatin, Fluorouracil, and Leucovorin With or Without Sorafenib as Initial Treatment for Advanced Hepatocellular Carcinoma.Front Oncol.2021; 11, 619461. [CrossRef]
- Shi J, Lai ECH, Li N, Guo WX, Xue J, Lau WY, et al. A new classification for hepatocellular carcinoma with portal vein tumor thrombus.J Hepatobiliary Pancreat Sci. 201;18(1), 74-80. [CrossRef]
- Liang HL, Huang JS, Lin YH, Lai KH, Yang CF, Pan HB. Hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma by placing a temporary catheter via the subclavian route. Acta Radiol. 2007;48(7), 734-740. [CrossRef]
- Lencioni R, Eric C H Lai, Nan Li, Guo WX, Xue J, Lau WY,et al.Objective response by mRECIST as a predictor and potential surrogate end-point of overall survival in advanced HCC.J Hepatol. 2017;66(6), 1166-1172.
- Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al.
- Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008; 359, 378–390.
- Quirk M, Kim YH, Saab S, Lee EW. Management of hepatocellular carcinoma with portal vein thrombosis. World JGastroenterol.2015; 21, 3462–3471. [CrossRef]
- Kudo M. Changing the Treatment Paradigm for Hepatocellular Carcinoma Using Atezolizumab plus Bevacizumab Combination Therapy. Cancers (Basel). 2021;13(21), 5475. [CrossRef]
- Liu BJ, Gao S, Zhu X, Guo JH, Kou FX, Liu SX, et al.Combination Therapy of Chemoembolization and Hepatic Arterial Infusion Chemotherapy in Hepatocellular Carcinoma with Portal Vein Tumor Thrombosis Compared with Chemoembolization Alone: A Propensity Score-Matched Analysis. Biomed Res Int. 2021; 6670367. [CrossRef]
- Dong H, Jian Y, Wang M, Liu F, Zhang Q, Peng Z,et al. Hepatic artery intervention combined with immune-targeted therapy is superior to sequential therapy in BCLC-C hepatocellular carcinoma. J Cancer Res Clin Oncol. 2023; 149(8), 5405-5416.
 |
| Median OS (m) | Extrahepatic spread | p-value | |
|---|---|---|---|
| Yes | No | ||
| VP3/4 subgroup | 13.0 (9.2-16.8) | 15.0 (12.2-17.8) | 0.407 |
| VP ≤2 subgroup | 11.4 (10.6-12.2) | 19.4 (16.8-22.0) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




