Submitted:
29 September 2025
Posted:
05 October 2025
You are already at the latest version
Abstract
Keywords:
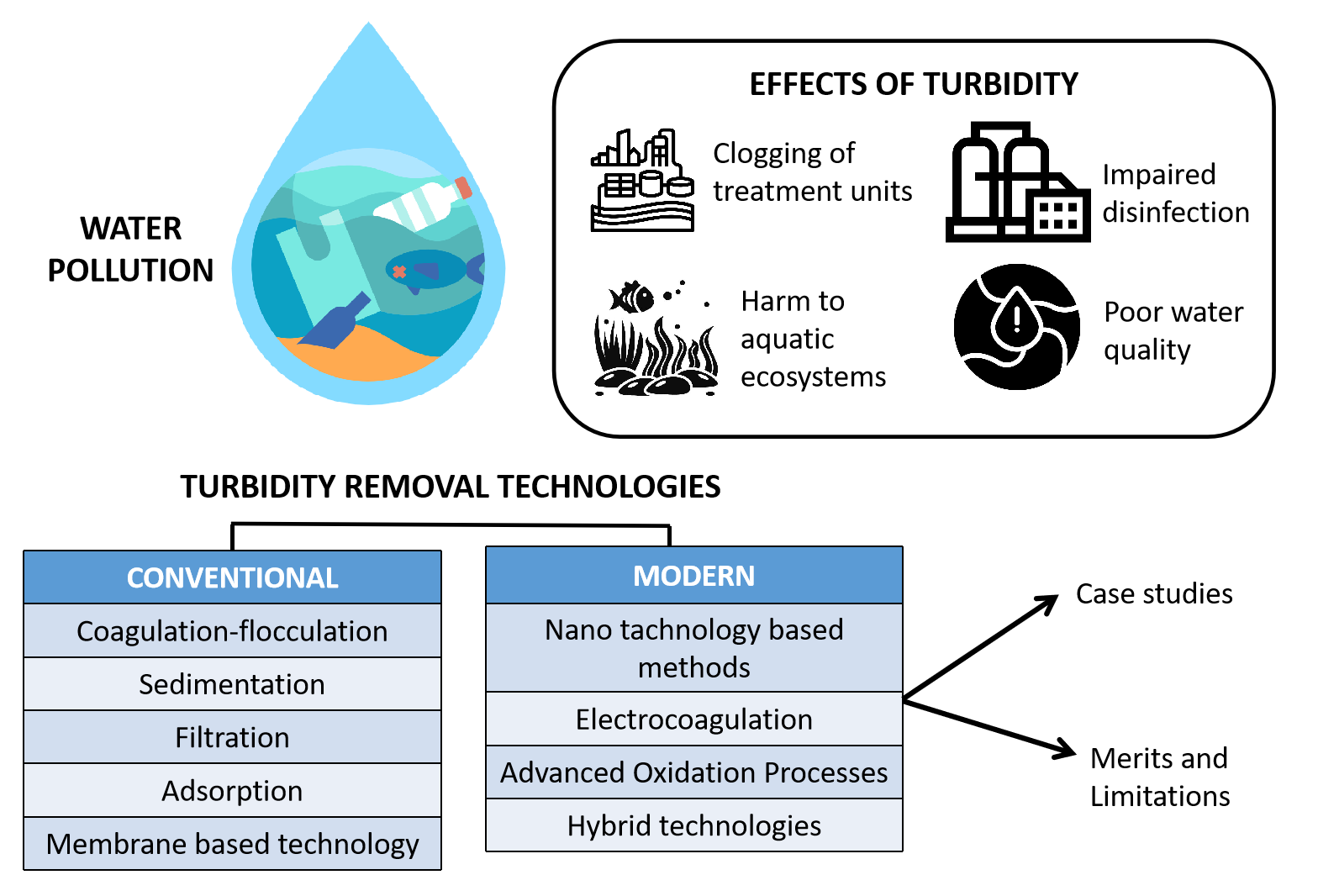
1. Introduction
2. Conventional Treatment Techniques in Removing Turbidity
2.1. Coagulation (with Flocculation)
2.2. Sedimentation
2.3. Filtration
Roughing Filtration
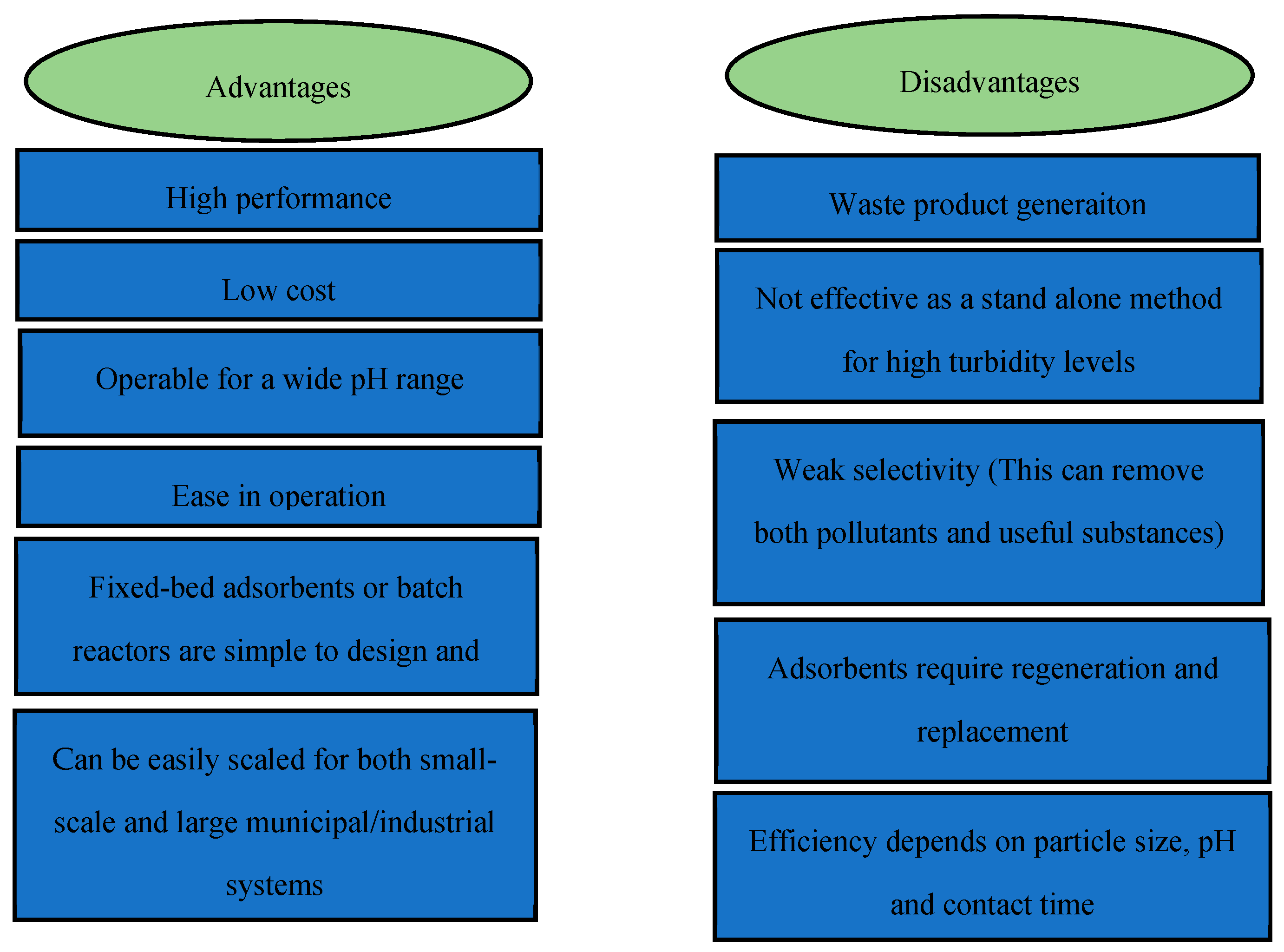
2.4. Adsorption
2.5. Membrane Based Technologies
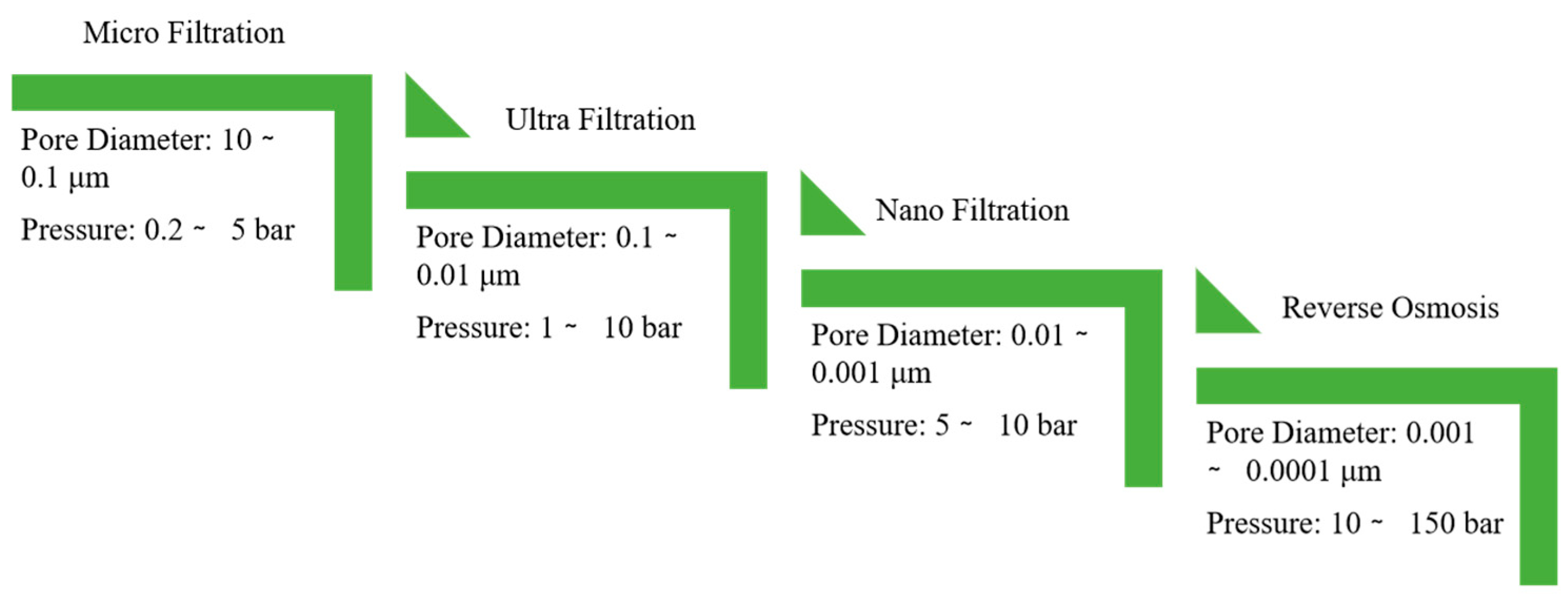
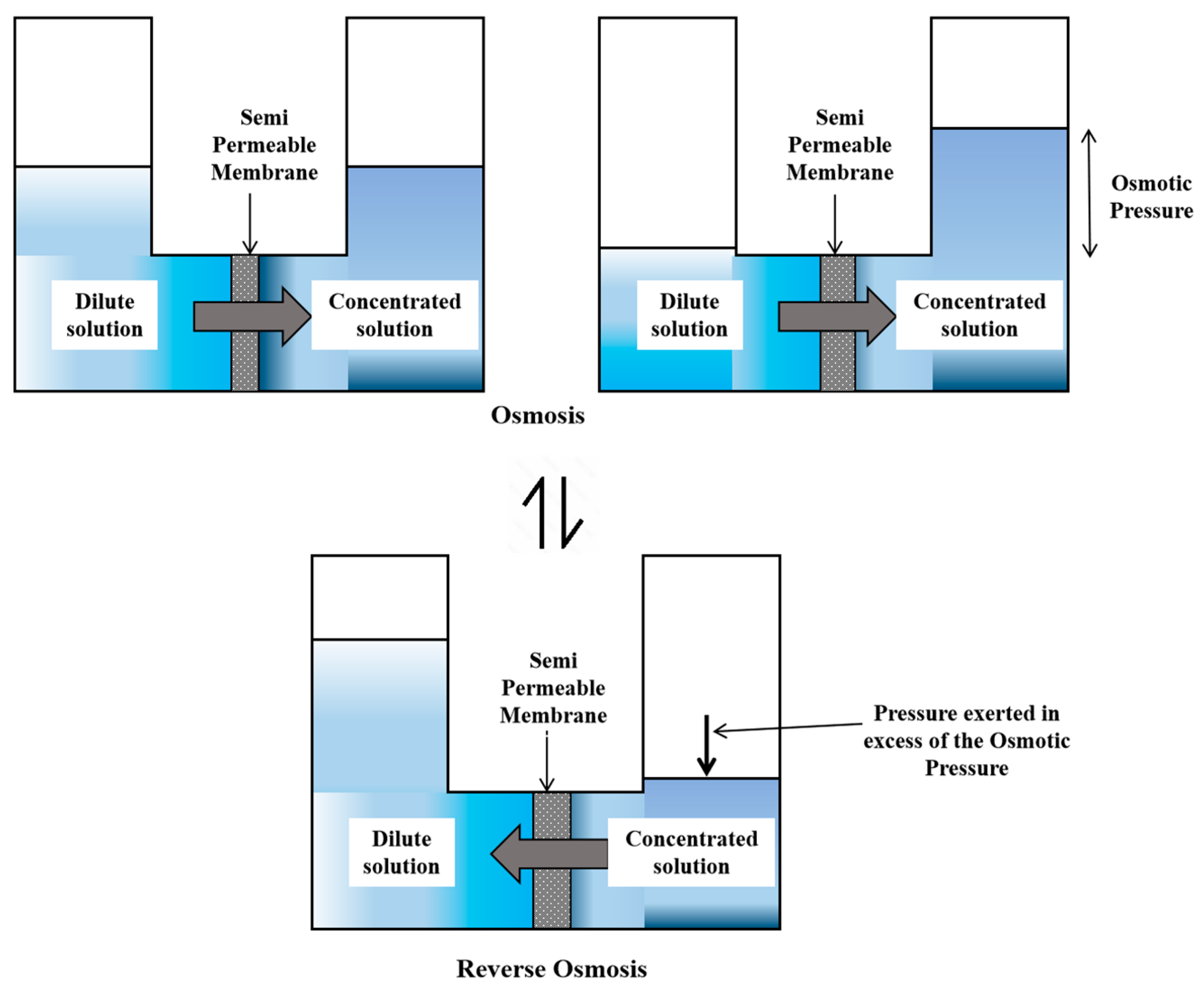
2.5.1. Reverse Osmosis (RO)
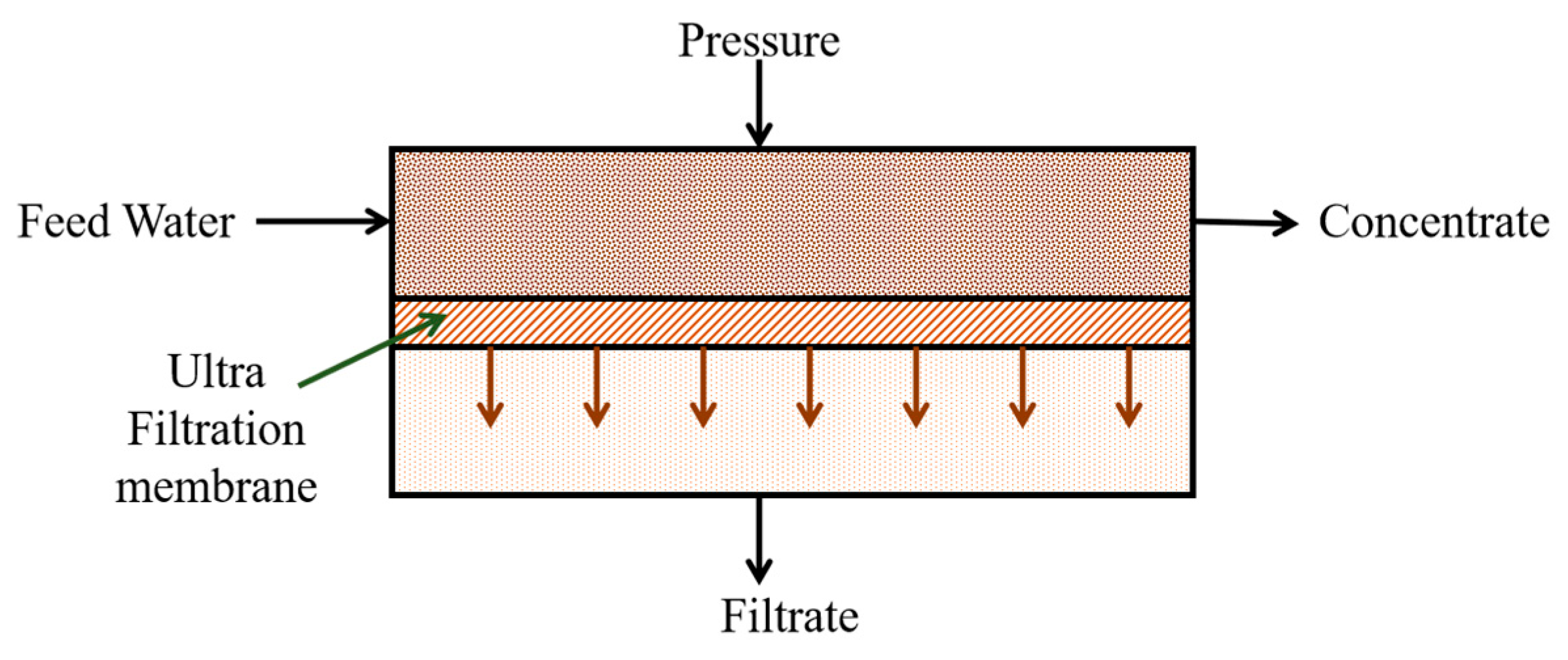
2.5.2. Ultrafiltration (UF)
2.5.3. Microfiltration (MF)
3. Emerging Techniques to Remove Turbidity
3.1. Nano Technology Based Methods
3.1.1. Nano Adsorbents
3.1.2. Nano Engineered Membranes
Nanofiltration Membranes
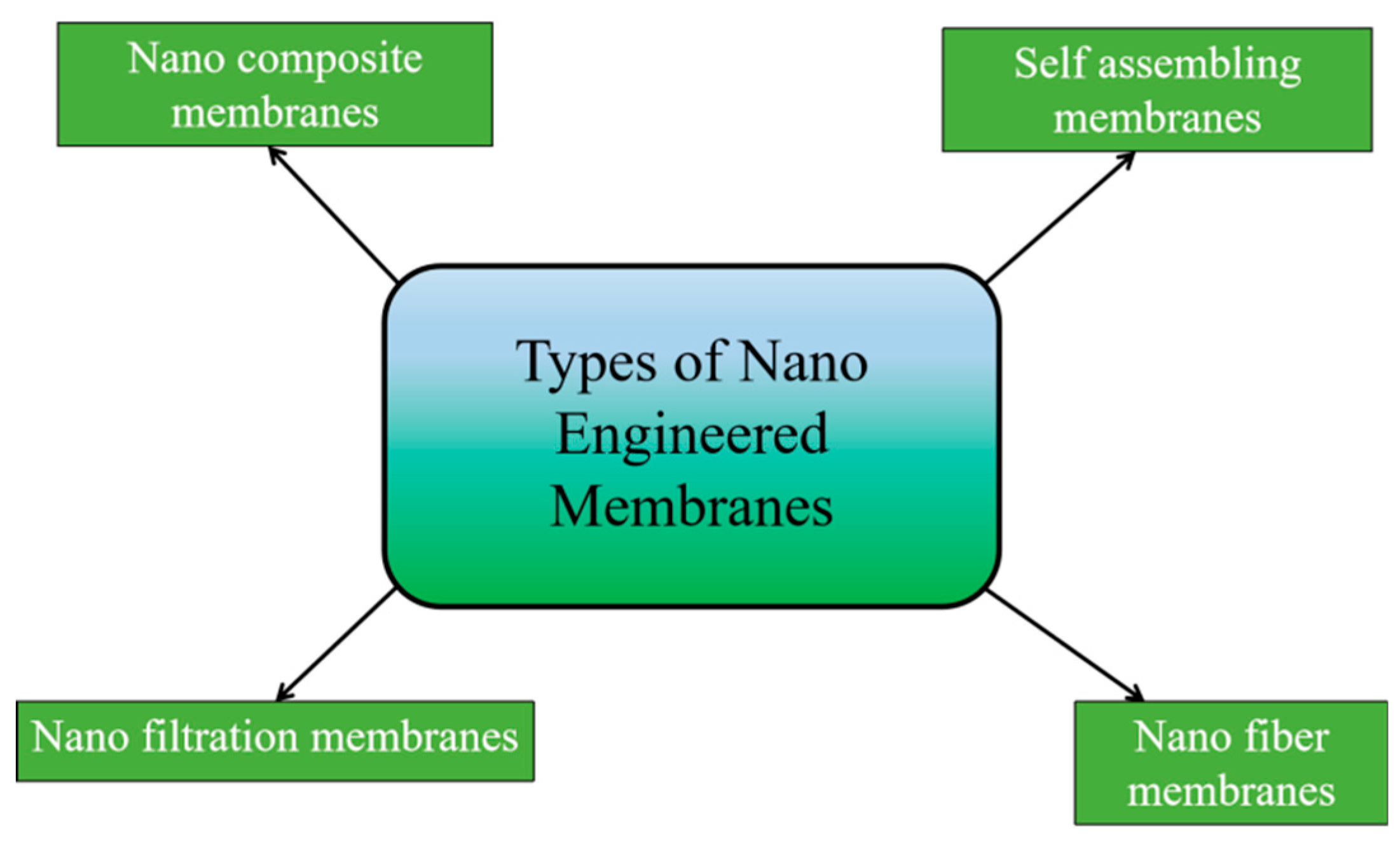
Nanocomposite Membranes
Self-Assembling membranes
Nano Fiber Membranes
3.1.3. Nano Photocatalysis
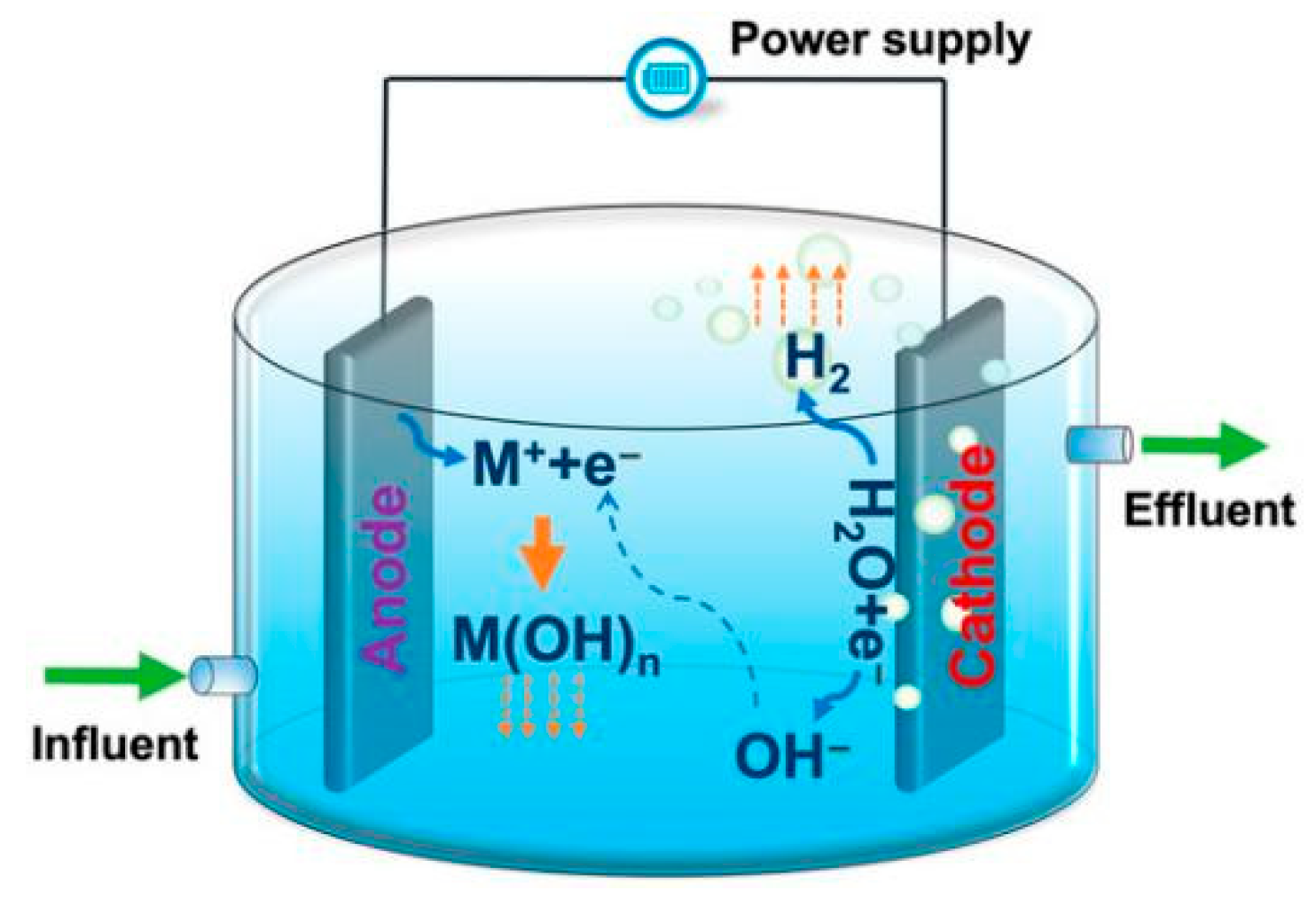
3.2. Electro-Coagulation (EC)
3.3. Advanced Oxidation Processes (AOPs)
3.3.1. Photocatalysis
3.3.2. UV/Hydrogen Peroxide (H2O2)
3.3.3. Ozone/Hydrogen Peroxide (H2O2)
3.3.4. Fenton Processes
3.4. Hybrid Technologies of Removing Turbidity
- Coagulation (with Flocculation) + Membrane Filtration
- Coagulation + Ballasted Flocculation
- Membrane Bioreactor (MBR) + Coagulation
- Ultrasound-Assisted Coagulation
3.4.1. Coagulation (with Flocculation) with Membrane Filtration
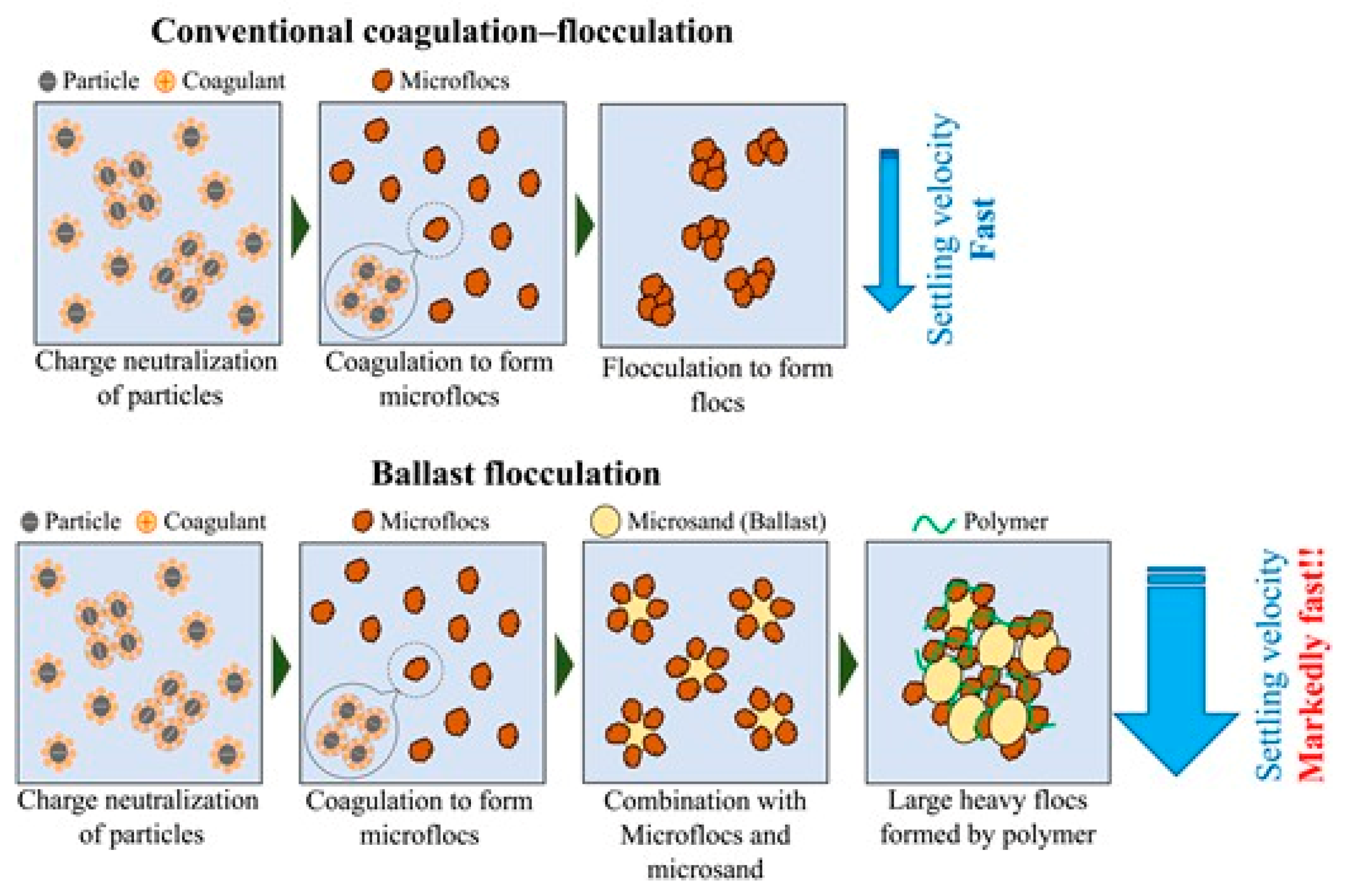
3.4.2. Coagulation with Ballasted Flocculation
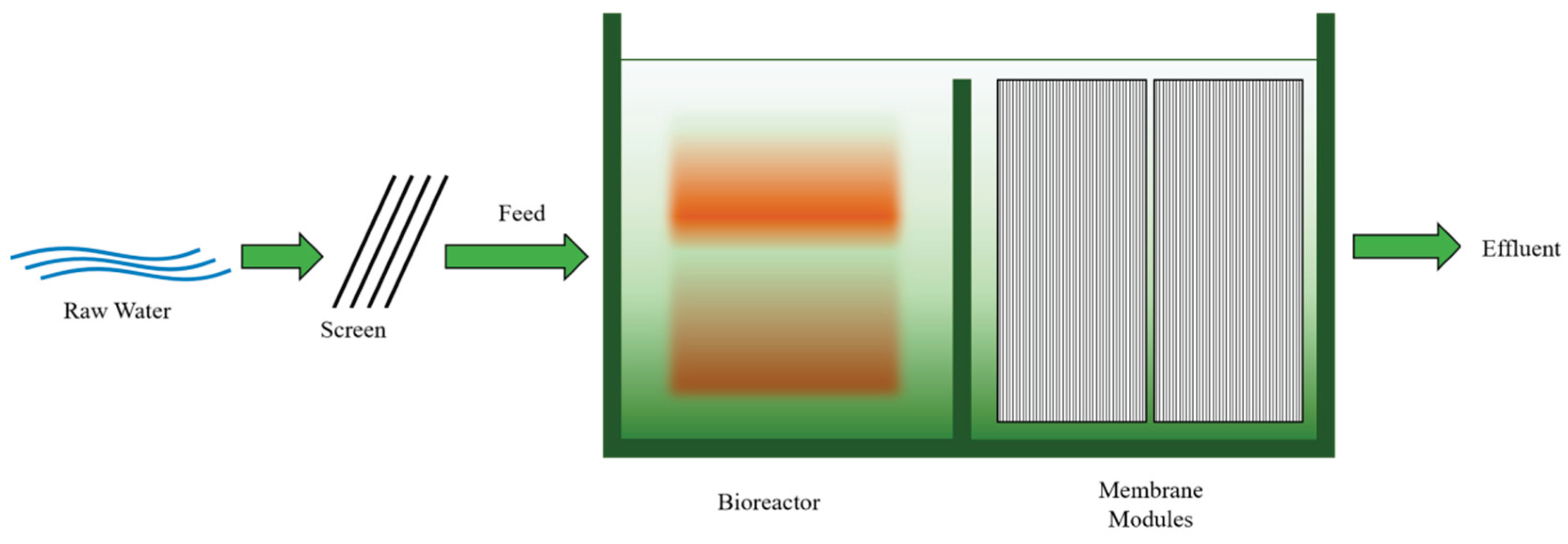
3.4.3. Coagulation with Membrane Bio Reactor technique
3.4.4. Ultrasound Assisted Coagulation
4. Advantages and Disadvantages of the Emerging Technologies
5. Conclusions and Future Directions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| NTU | Nephelometric Turbidity Units |
| UV | Ultra Violet |
| CSG | Coal Seam Gas |
| PMF | Pebble Matrix Filtration |
| RO | Reverse Osmosis |
| NF | Nano Filtration |
| UF | Ultra Filtration |
| MF | Micro Filtration |
| CNT | Carbon Nano Tubes |
| AOP | Advanced Oxidation Process |
| EC | Electro Coagulation |
References
- Mayur Bhadarka, Vaghela, D.T., Bamaniya Pinak Kamleshbhai, Hardik Sikotariya and Verma, P. (2024). Water Pollution: Impacts on Environment. [online] ResearchGate. Available at: https://www.researchgate.net/publication/383021412_Water_Pollution_Impacts_on_Environment.
- USGS (2018). Turbidity and Water | U.S. Geological Survey. [online] USGS. Available at: https://www.usgs.gov/special-topics/water-science-school/science/turbidity-and-water [Accessed : 10/06/2025].
- Fondriest Environmental, Inc. (2014). Turbidity, Total Suspended Solids & Water Clarity - Environmental Measurement Systems. [online] Environmental Measurement Systems. Available at: https://www.fondriest.com/environmental-measurements/parameters/water-quality/turbidity-total-suspended-solids-water-clarity/ [Accessed : 10/06/2025].
- AQUALABO (2024). Understanding the turbidity of water: causes, measurement and solutions - Aqualabo. [online] Aqualabo. Available at: https://www.aqualabo.fr/en/understanding-the-turbidity-of-water-causes-measurement-and-solutions/ [Accessed : 14/06/2025].
- Grobbelaar, J.U. (2009). Turbidity. [online] ScienceDirect. Available at: https://www.sciencedirect.com/science/article/abs/pii/B9780123706263000752.
- YSI (2024). Turbidity measurement and monitoring in water quality analysis | YSI. [online] www.ysi.com. Available at: https://www.ysi.com/parameters/turbidity [Accessed : 14/06/2025].
- Soros, A., Amburgey, J.E., Stauber, C.E., Sobsey, M.D. and Casanova, L.M. (2019). Turbidity reduction in drinking water by coagulation-flocculation with hitosan polymers. Journal of Water and Health, [online] 17(2), pp.204–218. [CrossRef]
- Olszak, N. (2022). How to Treat Turbidity in Water | Complete Water Solutions. [online] COMPLETE WATER SOLUTIONS. Available at: https://complete-water.com/resources/how-to-treat-turbidity-in-water [Accessed : 14/06/2025].
- Lee, C.S., Robinson, J. and Chong, M.F. (2014). A review on application of flocculants in wastewater treatment. Process Safety and Environmental Protection, 92(6), pp.489–508. [CrossRef]
- Ho, Y.-C., Chua, S.-C. and Chong, F.-K. (2020). Coagulation-Flocculation Technology in Water and Wastewater Treatment. Handbook of Research on Resource Management for Pollution and Waste Treatment, pp.432–457. [CrossRef]
- Iwuozor, K.O. (2019). Prospects and Challenges of Using Coagulation-Flocculation Method in the Treatment of Effluents. Advanced Journal of Chemistry-Section a, 2(2), pp.105–127. [CrossRef]
- Lmipumps.com. (2018). Coagulation and Flocculation in Water Treatment. [online] Available at: https://www.lmipumps.com/en/technologies/coagulation-and-flocculation-in-water-treatment/ [Accessed : 14/06/2025].
- Ashraf, S.N., Rajapakse, J., Millar, G. and Dawes, L., 2016. Performance analysis of chemical and natural coagulants for turbidity removal of river water in coastal areas of Bangladesh. In: Kabir, S.M.Z. and Hasan, M.Z., eds. Proceedings of the 1st AISD International Multidisciplinary Conference: International Multidisciplinary Conference on Sustainable Development (IMCSD) 2016. Australia: Australian Institute for Sustainable Development (AISD), pp.172–182. Available at: Fhttps://scholar.google.com.au/citations?view_op=view_citation&hl=en&user=UCssR4oAAAAJ&citation_for_view=UCssR4oAAAAJ:9yKSN-GCB0IC.
- Nishat Ashraf, S., Rajapakse, J., Dawes, L.A. and Millar, G.J. (2018). Coagulants for removal of turbidity and dissolved species from coal seam gas associated water. Journal of Water Process Engineering, 26, pp.187–199. [CrossRef]
- forestier, jean-pierre (2024). How to Eliminate Turbidity in Well Water for Clear and Healthy Water? [online] Diproclean.com. Available at: https://www.diproclean.com/en/solutions-impurities-water-house-xsl-537_538_545.html [Accessed : 15/06/2025].
- Www.htt.io. (2024). Htt.io. [online] Available at: https://www.htt.io/learning-center/understanding-sedimentation-water-treatment [Accessed : 15/06/2025].
- BOQU (2024). How to Reduce Turbidity in Water: Solutions and Technologies. [online] Boquinstrument.com. Available at: https://www.boquinstrument.com/a-how-to-reduce-turbidity-in-water-solutions-and-technologies.html [Accessed : 15/06/2025].
- www.etch2o.com. (2023). What is Sedimentation in Wastewater Treatment? - Etch2o. [online] Available at: https://www.etch2o.com/what-is-sedimentation-in-wastewater-treatment/ [Accessed : 15/06/2025].
- Ephrem Guchi (2015). Review on Slow Sand Filtration in Removing Microbial Contamination and Particles from Drinking Water. American Journal of Food and Nutrition, [online] 3(2), pp.47–55. Available at: https://www.researchgate.net/publication/275097978_Review_on_Slow_Sand_Filtration_in_Removing_Microbial_Contamination_and_Particles_from_Drinking_Water_Ephrem_Guchi_1.
- T, M. (2017). Rapid vs slow filtration. [online] Biosand filter. Available at: https://biosandfilter.org/biosand-filter/rapid-vs-slow-filtration/ [Accessed : 16/06/2025].
- U.S. EPA Area-Wide Optimization Program (AWOP) Water Quality Goals and Operational Criteria for Optimization of Slow Sand Filtration. (n.d.). Available at: https://www.epa.gov/system/files/documents/2024-04/water-quality-goals-and-slow-sand-filtration.pdf [Accessed : 16/06/2025].
- Wegelin, M. (1996). Surface Water Treatment by Roughing Filters: A Design, Construction and Operation Manual, pp.v-3.
- Slow Sand Filtration | SSWM - Find tools for sustainable sanitation and water management! Sswm.info. Available at: https://sswm.info/sswm-university-course/module-6-disaster-situations-planning-and-preparedness/further-resources-0/slow-sand-filtration [Accessed : 16/06/2025].
- Tamakhu, G. and Amatya, I.M. (2021). Turbidity removal by rapid sand filter using anthracite coal as capping media. Journal of Innovations in Engineering Education, 4(1), pp.69–73. [CrossRef]
- Adel, K., M. Negm, M. Abdelrazik and E. Wahb (2014). The Use of Roughing Filters in Water Purification. Scientific Journal of October 6 University, 2(1), pp.50–58. Available at: https://www.researchgate.net/publication/333683287_The_Use_of_Roughing_Filters_in_Water_Purification.
- Nkwonta, O.I., Olufayo, O.A., Ochieng, G.M., Adeyemo, J.A. and Otieno, F.O.A. (2010). Turbidity Removal: Gravel and Charcoal as Roughing Filtration Media. SSRN Electronic Journal. Availabel at: . [CrossRef]
- Rajapakse, J.P. and Fenner, R.A. (2011). Evaluation of Alternative Media for Pebble Matrix Filtration Using Clay Balls and Recycled Crushed Glass. Journal of Environmental Engineering, 137(6), pp.517–524. [CrossRef]
- Pure Aqua. Inc. (2022). Turbidity In Water Treatment. [online] Available at: https://pureaqua.com/turbidity-in-water-treatment/ [Accessed : 16/06/2025].
- Akhtar, M.S., Ali, S. and Zaman, W. (2024). Innovative Adsorbents for Pollutant Removal: Exploring the Latest Research and Applications. Molecules, 29(18), p.4317. [CrossRef]
- Danamark (2018). Adsorption Is The Key | Resources | Danamark Watercare. [online] Danamark. Available at: https://danamark.com/resources/adsorption-water-filtration/ [Accessed : 16/06/2025].
- Lee, S. (2025). Adsorbents in Water Treatment: A Comprehensive Guide. [online] Numberanalytics.com. Available at: https://www.numberanalytics.com/blog/ultimate-guide-adsorbents-water-treatment [Accessed : 16/06/2025].
- Metcalf and Eddy (2013). Wastewater Engineering : Treatment and Resource Recovery. 5th ed. Burr Ridge, Il: Mcgraw-Hill Medical Pub.
- Hai Nguyen Tran (2023). Adsorption Technology for Water and Wastewater Treatments. Water, 15(15), pp.2857–2857. [CrossRef]
- Crittenden, J.C., Trussell, R.R., Hand, D.W., Howe, K.J. and Tchobanoglous, G., 2012. MWH’s Water Treatment: Principles and Design. 3rd ed. Hoboken: John Wiley & Sons.
- Sadegh, H. and A. M. Ali, G. (n.d.). Potential Applications of Nanomaterials in Wastewater Treatment:Nanoadsorbents Performance. In: Advanced Treatment Techniques for Industrial Wastewater. [online] IGI Global, pp.51–52. Available at: https://www.researchgate.net/publication/325904204_Potential_Applications_of_Nanomaterials_in_Wastewater_Treatment_Nanoadsorbents_Performance.
- Interfilter.com. (2025). What is a membrane filter and how does membrane filtration work? [online] Available at: https://www.interfilter.com/news/what-is-a-membrane-filter-and-how-does-membrane-filtration-work [Accessed : 17/06/2025].
- Hancock, N. (2016). Ultrafiltration, Nanofiltration and Reverse Osmosis. [online] Safe Drinking Water Foundation. Available at: https://www.safewater.org/fact-sheets-1/2017/1/23/ultrafiltrationnanoandro [Accessed : 17/06/2025].
- Aziz, S., Abdur Rehman Mazhar, Ubaid, A., Syed, Riaz, Y., Tariq Talha and Jung, D.-W. (2024). A comprehensive review of membrane-based water filtration techniques. Applied Water Science, 14(8). [CrossRef]
- Reverse osmosis membrane fabrication and modification technologies and future trends: A review. (2020). Advances in Colloid and Interface Science, [online] 276, p.102100. [CrossRef]
- Li, X., Jiang, L. and Li, H. (2018). Application of Ultrafiltration Technology in Water Treatment. IOP Conference Series: Earth and Environmental Science, 186(3), p.012009. [CrossRef]
- Michał Bodzek, Konieczny, K. and Rajca, M. (2023). Membranes in water and wastewater disinfection– review. [CrossRef]
- Cuartucci, M. (2020). Ultrafiltration, a cost-effective solution for treating surface water to potable standard. Water Practice and Technology, 15(2), pp.426–436. [CrossRef]
- Synderfiltration.com. (2014). Microfiltration Membranes | Synder Filtration. [online] Available at: https://synderfiltration.com/microfiltration/membranes/ [Accessed : 17/06/2025].
- Hakami, M.W., Alkhudhiri, A., Al-Batty, S., Zacharof, M.-P., Maddy, J. and Hilal, N. (2020). Ceramic Microfiltration Membranes in Wastewater Treatment: Filtration Behavior, Fouling and Prevention. Membranes, 10(9), p.248. [CrossRef]
- Park, W., Jeong, S., Im, S.-J. and Jang, A. (2020). High turbidity water treatment by ceramic microfiltration membrane: Fouling identification and process optimization. Environmental Technology & Innovation, 17, p.100578. [CrossRef]
- Tsakiris, G. and Alexakis, D. (2013). Karstic spring water quality: the effect of groundwater abstraction from the recharge area. Desalination and Water Treatment, 52(13-15), pp.2494–2501. [CrossRef]
- Meng, S., Zhang, M., Yao, M., Qiu, Z., Hong, Y., Lan, W., Xia, H. and Jin, X. (2019). Membrane Fouling and Performance of Flat Ceramic Membranes in the Application of Drinking Water Purification. Water, 11(12), p.2606. [CrossRef]
- Ezugbe, E.O. and Rathilal, S. (2020). Membrane Technologies in Wastewater Treatment: A Review. Membranes, [online] 10(5), p.89. [CrossRef]
- Stoquart, C., Servais, P., Bérubé, P.R. and Barbeau, B. (2012). Hybrid Membrane Processes using activated carbon treatment for drinking water: A review. Journal of Membrane Science, 411-412, pp.1–12. [CrossRef]
- Nataraj, S.K., Hosamani, K.M. and Aminabhavi, T.M. (2006). Distillery wastewater treatment by the membrane-based nanofiltration and reverse osmosis processes. Water Research, 40(12), pp.2349–2356. [CrossRef]
- Gehrke, I., Geiser, A. and Somborn-Schulz, A. (2015). Innovations in nanotechnology for water treatment. Nanotechnology, Science and Applications, 8, p.1. [CrossRef]
- Tripathy, J., Mishra, A., Pandey, M., Thakur, R.R., Chand, S., Rout, P.R. and Shahid, M.K. (2024). Advances in Nanoparticles and Nanocomposites for Water and Wastewater Treatment: A Review. Water, [online] 16(11), p.1481. [CrossRef]
- Khan, N.A., Khan, S.U., Ahmed, S., Farooqi, I.H., Dhingra, A., Hussain, A. and Changani, F. (2019). Applications of Nanotechnology in Water and Wastewater Treatment: A Review. Asian Journal of Water, Environment and Pollution, 16(4), pp.81–86. [CrossRef]
- Anjum, M., Miandad, R., Waqas, M., Gehany, F. and Barakat, M.A. (2019). Remediation of wastewater using various nano-materials. Arabian Journal of Chemistry, 12(8), pp.4897–4919. [CrossRef]
- Bala Anegbe, Ifijen, I.H., Muniratu Maliki, Uwidia, I.E. and Aigbodion, A.I. (2024). Graphene oxide synthesis and applications in emerging contaminant removal: a comprehensive review. Environmental Sciences Europe, 36(1). [CrossRef]
- Moro, K., Ello, A.S., Koffi, K.R. and N’goran Séverin Eroi (2023). Mixed Maghemite/Hematite Iron Oxide Nanoparticles Synthesis for Lead and Arsenic Removal from Aqueous Solution. Journal of Nanomaterials, [online] 2023, pp.1–8. [CrossRef]
- Pandey, N., Shukla, S.K. and Singh, N.B. (2017). Water purification by polymer nanocomposites: an overview. Nanocomposites, 3(2), pp.47–66. [CrossRef]
- Pérez-Botella, E., Valencia, S. and Rey, F. (2022). Zeolites in Adsorption Processes: State of the Art and Future Prospects. Chemical Reviews, 122(24), pp.17647–17695. [CrossRef]
- Naseer, A. (2024). Role of nanocomposites and nano adsorbents for heavy metals removal and dyes. An overview. Desalination and Water Treatment, 320, p.100662. [CrossRef]
- Lokeshappa B (2021). The Role of Nanocompositers and Adsorbents in the Turbidity Removal Process. International Journal of Advances in Engineering and Management (IJAEM), [online] 3, p.246. Available at: https://ijaem.net/issue_dcp/The%20Role%20of%20Nanocompositers%20and%20Adsorbents%20in%20the%20Turbidity%20Removal%20Process.pdf.
- Aboubaraka, A.E., Aboelfetoh, E.F. and Ebeid, E.-Z.M. (2017). Coagulation effectiveness of graphene oxide for the removal of turbidity from raw surface water. Chemosphere, 181, pp.738–746. [CrossRef]
- Khan, S.A., Rafiq, M.T., Feng, Y., Ali, H.M., Waleed A. A. Alsakkaf, Shah, F.A., Ali, A., Ahmed, T. and Aziz, R. (2025). Nanocellulose based titanium dioxide nanocomposites for the wastewater treatment of textile industry. Frontiers in Environmental Science, 13. [CrossRef]
- Khiadani, M., Zarrabi, M. and Foroughi, M. (2013). Urban runoff treatment using nano-sized iron oxide coated sand with and without magnetic field applying. Journal of Environmental Health Science and Engineering, [online] 11(1). [CrossRef]
- Shon, H.K., Phuntsho, S., Chaudhary, D.S., Vigneswaran, S. and Cho, J. (2013). Nanofiltration for water and wastewater treatment – a mini review. Drinking Water Engineering and Science, 6(1), pp.47–53. [CrossRef]
- Hilal, N., Al-Zoubi, H., Darwish, N.A., Mohamma, A.W. and Abu Arabi, M. (2004). A comprehensive review of nanofiltration membranes:Treatment, pretreatment, modelling, and atomic force microscopy. Desalination, 170(3), pp.281–308. Available at: https://www.sciencedirect.com/science/article/pii/S0011916404800328.
- Jhaveri, J.H. and Murthy, Z.V.P. (2015). Nanocomposite membranes. Desalination and Water Treatment, 57(55), pp.26803–26819. [CrossRef]
- Whitesides, G.M. (2002). Self-Assembly at All Scales. Science, 295(5564), pp.2418–2421. [CrossRef]
- Burhannudin Sutisna, Musteata, V., Pulido, B., Puspasari, T., Detlef-M. Smilgies, Nikos Hadjichristidis and Nunes, S.P. (2019). High flux membranes, based on self-assembled and H-bond linked triblock copolymer nanospheres. Journal of Membrane Science, 585, pp.10–18. [CrossRef]
- Sahu, A., Dosi, R., Kwiatkowski, C., Schmal, S. and Poler, J.C. (2023). Advanced Polymeric Nanocomposite Membranes for Water and Wastewater Treatment: A Comprehensive Review. Polymers, 15(3), p.540. [CrossRef]
- Asmatulu, R. and Khan, W.S. (2019). Chapter 1 - Introduction to electrospun nanofibers. [online] ScienceDirect. Available at: https://www.sciencedirect.com/science/article/abs/pii/B9780128139141000018.
- Abdulhamid, M.A. and Muzamil, K. (2023). Recent progress on electrospun nanofibrous polymer membranes for water and air purification: A review. Chemosphere, [online] 310, p.136886. [CrossRef]
- Asmatulu, R., Muppalla, H., Veisi, Z., Khan, W., Asaduzzaman, A. and Nuraje, N. (2013). Study of Hydrophilic Electrospun Nanofiber Membranes for Filtration of Micro and Nanosize Suspended Particles. Membranes, 3(4), pp.375–388. [CrossRef]
- Ong, C.B., Ng, L.Y. and Mohammad, A.W. (2018). A review of ZnO nanoparticles as solar photocatalysts: Synthesis, mechanisms and applications. Renewable and Sustainable Energy Reviews, 81, pp.536–551. [CrossRef]
- Gómez-Pastora, J., Dominguez, S., Bringas, E., Rivero, M.J., Ortiz, I. and Dionysiou, D.D. (2017). Review and perspectives on the use of magnetic nanophotocatalysts (MNPCs) in water treatment. Chemical Engineering Journal, 310, pp.407–427. [CrossRef]
- Nilesh A. Fagnekar (2015). REMOVAL OF TURBIDITY USING ELECTROCOAGULATION. International Journal of Research in Engineering and Technology, 04(06), pp.537–543. [CrossRef]
- Tech, G.W. (2024). Electrocoagulation: The Future of Water Treatment Technology - Genesis Water Technologies. [online] Genesis Water Technologies. Available at: https://genesiswatertech.com/blog-post/electrocoagulation-the-future-of-water-treatment-technology/.
- Electrocoagulation Water Treatment - VentilAQUA. [online] Available at: https://ventilaqua.com/chemical-treatment/electrocoagulation-water-treatment/ [Accessed : 19/06/2025].
- Mao, Y., Zhao, Y. and Cotterill, S. (2023). Examining Current and Future Applications of Electrocoagulation in Wastewater Treatment. Water, [online] 15(8), p.1455. [CrossRef]
- Makwana, A.R. (2018). Factors Influencing Electrocoagulation Treatment of UASB Reactor Effluent. Water science and technology library, pp.55–64. [CrossRef]
- Ebba, M., Asaithambi, P. and Alemayehu, E. (2021). Investigation on operating parameters and cost using an electrocoagulation process for wastewater treatment. Applied Water Science, 11(11). [CrossRef]
- Zailani, L.W.M. and Zin, N.S.M. (2018). Application of Electrocoagulation In Various Wastewater And Leachate Treatment-A Review. IOP Conference Series: Earth and Environmental Science, 140, p.012052. [CrossRef]
- Rahmani, A. (2008). Removal of water turbidity by the electrocoagulation method. PubMed, 8(1), pp.18–24. Available at: https://www.researchgate.net/publication/235366690_Removal_of_water_turbidity_by_the_electrocoagulation_method.
- Kobya, M., Hiz, H., Senturk, E., Aydiner, C. and Demirbas, E. (2006). Treatment of potato chips manufacturing wastewater by electrocoagulation. Desalination, [online] 190(1-3), pp.201–211. [CrossRef]
- C.B. Shivayogimath (2013). TREATMENT OF SOLID WASTE LEACHATE BY ELECTROCOAGULATION TECHNOLOGY. International Journal of Research in Engineering and Technology, 02(13), pp.266–269. [CrossRef]
- Sadeddin, K., Naser, A. and Firas, A. (2011). Removal of turbidity and suspended solids by electro-coagulation to improve feed water quality of reverse osmosis plant. Desalination, 268(1-3), pp.204–207. [CrossRef]
- Fahad Al-Ajmi, Al-Marri, M. and Fares Almomani (2024). Electrocoagulation Process as an Efficient Method for the Treatment of Produced Water Treatment for Possible Recycling and Reuse. Water, 17(1), pp.23–23. Avaiblable at: . [CrossRef]
- Behera, U.S., Poddar, S. and Byun, H.-S. (2025). Electrocoagulation treatment of wastewater collected from Haldia industrial region: Performance evaluation and comparison of process optimization. Water Research, 268, p.122716. [CrossRef]
- Aslan, N. and Cebeci, Y. (2007). Application of Box–Behnken design and response surface methodology for modeling of some Turkish coals. Fuel, 86(1-2), pp.90–97. [CrossRef]
- Ali, M., Kumar, A., Yvaz, A. and Salah, B. (2023). Central composite design application in the optimization of the effect of pumice stone on lightweight concrete properties using RSM. Case Studies in Construction Materials, p.e01958. [CrossRef]
- Rajapakse, R., Jay, R., Waterman, P., Millar, G. and Sumanaweera, S., 2014. Emerging water treatment technologies for decentralised systems: An overview of selected systems suited for application in towns and settlements in remote and very remote regions of Australia and vulnerable and lagging rural regions in Sri Lanka. In: K. Charters, ed. Sustainable Economic Growth for Regional Australia (SEGRA) Conference 2014 Proceedings. Australia: SEGRA, pp.1–28.
- Wang, C., Li, W., Zhang, Z., Lei, D., Che, G., Gou, C., Zhang, J. and Hao, Z. (2024). A novel iron sulfide phase with remarkable hydroxyl radical generation capability for contaminants degradation. Water Research, [online] 251, p.121166. [CrossRef]
- Ghime, D. and Ghosh, P. (2020). Advanced Oxidation Processes: A Powerful Treatment Option for the Removal of Recalcitrant Organic Compounds. [online] www.intechopen.com. IntechOpen. Available at: https://www.intechopen.com/chapters/70086.
- Oturan, M.A. and Aaron, J.-J. (2014). Advanced Oxidation Processes in Water/Wastewater Treatment: Principles and Applications. A Review. Critical Reviews in Environmental Science and Technology, 44(23), pp.2577–2641. [CrossRef]
- Dedeh Rosmaniar Sofia, Eko Sulistio Hanam, Sunardi Sunardi, Dadan Sumiarsa and I Made Joni (2024). Hydroxyl Radical-Based Advanced Oxidation Processes of Red Reactive Dyes by Ultrafine Bubbles Method. Water, 16(12), pp.1678–1678. Available at: https://www.mdpi.com/2073-4441/16/12/1678.
- Tufail, A., Price, W.E. and Hai, F.I. (2020). A critical review on advanced oxidation processes for the removal of trace organic contaminants: A voyage from individual to integrated processes. Chemosphere, 260, p.127460. [CrossRef]
- American Water Chemicals (2025). Advanced Oxidation Processes - Definition | AWC. [online] American Water Chemicals, Inc. Available at: https://www.membranechemicals.com/water-treatment/advanced-oxidation-plants/ [Accessed : 19/06/2025].
- ERDC’s Photocatalytic Anti-Microbial Water Treatment. [online] ERDC Innovation. Available at: https://www.erdcinnovation.org/technology/photocatalytic-water-treatment/ [Accessed : 19/06/2025].
- Malpani, S. (2024). Photocatalysis in Wastewater Treatment: Harnessing the Power of Light - Save The Water. [online] Save The Water. Available at: https://savethewater.org/photocatalysis-in-wastewater-treatment-harnessing-the-power-of-light/.
- Al-Dawery, S., Reddy, Al-Mashrafiya, K., Al-Fraji, B., Muataz and Al-Daweri, S. (n.d.). Disinfection of biologically treated wastewater using photocatalysis process with artificial UV light and natural Solar radiation. [online]. [CrossRef]
- Caressa Munien, Emmanuel Kweinor Tetteh, Govender, T., Shivek Jairajh, Mguni, L.L. and Sudesh Rathilal (2023). Turbidity and COD Removal from Municipal Wastewater Using a TiO2 Photocatalyst—A Comparative Study of UV and Visible Light. 13(8), pp.4766–4766. [CrossRef]
- Ali, N.S., Kalash, K.R., Ahmed, A.N. and Albayati, T.M. (2022). Performance of a solar photocatalysis reactor as pretreatment for wastewater via UV, UV/TiO2, and UV/H2O2 to control membrane fouling. Scientific Reports, 12(1). [CrossRef]
- Spartanwatertreatment.com. (2024). Available at: https://spartanwatertreatment.com/advanced-oxidation-uv-peroxide/ [Accessed : 19/06/2025].
- Muruganandham, M., Suri, R.P.S., Jafari, Sh., Sillanpää, M., Lee, G.-J., Wu, J.J. and Swaminathan, M. (2014). Recent Developments in Homogeneous Advanced Oxidation Processes for Water and Wastewater Treatment. International Journal of Photoenergy, 2014, pp.1–21. [CrossRef]
- Watereurope.eu. (2018). Water Europe Marketplace. [online] Available at: https://mp.watereurope.eu/d/technology/1024 [Accessed : 19/06/2025].
- Collivignarelli, M.C., Pedrazzani, R., Sorlini, S., Abbà, A. and Bertanza, G. (2017). H2O2 Based Oxidation Processes for the Treatment of Real High Strength Aqueous Wastes. Sustainability, [online] 9(2), p.244. Available at:. [CrossRef]
- Glaze, W.H., Kang, J.-W. and Chapin, D.H. (1987). The Chemistry of Water Treatment Processes Involving Ozone, Hydrogen Peroxide and Ultraviolet Radiation. Ozone: Science & Engineering, 9(4), pp.335–352. [CrossRef]
- Sindelar, H.R., Brown, M.T. and Boyer, T.H. (2014). Evaluating UV/H2O2, UV/percarbonate, and UV/perborate for natural organic matter reduction from alternative water sources. Chemosphere, 105, pp.112–118. [CrossRef]
- Spartanwatertreatment.com. (2023). Available at: https://spartanwatertreatment.com/advanced-oxidation-peroxide-ozone/ [Accessed : 19/06/2025].
- Jensby, A. (2025). Benefits of Advanced Oxidation Processes (AOP) - ULTRAAQUA. [online] ULTRAAQUA. Available at: https://ultraaqua.com/blog/aop-advanced-oxidation-processes/ [Accessed : 19/06/2025].
- Paode, R.D., Chandrakanth, M.S., Amy, G.L., Gramith, J.T. and Ferguson, D.W. (2008). Ozone Versus Ozone/Peroxide Induced Particle Destabilization And Aggregation: A Pilot Study. Ozone: Science and Engineering, [online] 17(1), pp.25–51. [CrossRef]
- Walling, S.A., Um, W., Corkhill, C.L. and Hyatt, N.C. (2021). Fenton and Fenton-like wet oxidation for degradation and destruction of organic radioactive wastes. npj Materials Degradation, 5(1). [CrossRef]
- Pignatello, J.J., Oliveros, E. and MacKay, A. (2006). Advanced Oxidation Processes for Organic Contaminant Destruction Based on the Fenton Reaction and Related Chemistry. Critical Reviews in Environmental Science and Technology, 36(1), pp.1–84. [CrossRef]
- Du, Y., Zhou, M. and Lei, L. (2005). Reduction of Fe3+ by intermediates during Fenton oxidation of p-chlorophenol. Huagong Xuebao/Journal of Chemical Industry and Engineering (China), [online] 56(10), pp.1942–1947. Available at: https://www.researchgate.net/publication/286055298_Reduction_of_Fe3_by_intermediates_during_Fenton_oxidation_of_p-chlorophenol.
- Aleixo, N.A., Schmeing, A., Ailton Barbosa Perdão, Silva, J.D., Subtil, E.L., Bueno, R. and Gomes, H. (2023). Advanced oxidation processes in the mitigation of antibiotic-resistant genes in effluents. Elsevier eBooks, pp.341–367. [CrossRef]
- Vasquez-Medrano, R., Prato-Garcia, D. and Vedrenne, M. (2018). Ferrioxalate-Mediated Processes. Advanced Oxidation Processes for Waste Water Treatment, pp.89–113. [CrossRef]
- Jair Juarez, Silva, José Luiz Vieira and Felipe (2020). Treatment of swine wastewater using the Fenton process with ultrasound and recycled iron. Revista Ambiente & Água, 15(3), pp.1–1. [CrossRef]
- Ishak, A.R., Hamid, F.S., Mohamad, S. and Tay, K.S. (2017). Removal of organic matter from stabilized landfill leachate using Coagulation-Flocculation-Fenton coupled with activated charcoal adsorption. Waste Management & Research, 35(7), pp.739–746. [CrossRef]
- Favero, B.M., Favero, A.C., Silvio Roberto Taffarel and Souza, F.S. (2018). Evaluation of the efficiency of coagulation/flocculation and Fenton process in reduction of colour, turbidity and COD of a textile effluent. Environmental Technology, 41(12), pp.1580–1589. [CrossRef]
- Zaman, B., N Hardyanti, Pramesti, I.A. and William, G.S. (2024). The effect of fenton oxidation on the quality of pharmaceutical industry wastewater: a case study of BOD5, COD, TOC, and turbidity parameters. IOP Conference Series Earth and Environmental Science, 1414(1), pp.012036–012036. [CrossRef]
- Xu, M., Wu, C. and Zhou, Y. (2020). Advancements in the Fenton Process for Wastewater Treatment. [online] www.intechopen.com. IntechOpen. Available at: https://www.intechopen.com/chapters/70242.
- de Moura Gomes, L., da Silva Duarte, J.L., Pereira, N.M., Martínez-Huitle, C.A., Tonholo, J. and de Paiva e Silva Zanta, C.L. (2014). Development of a system for treatment of coconut industry wastewater using electrochemical processes followed by Fenton reaction. Water Science and Technology, 69(11), pp.2258–2264. [CrossRef]
- Gowtham, K.L. and Ganesan, R. (2025). Comparative study on removal of turbidity in aquaculture wastewater by electro fenton process with minimal national standards. AIP Conference Proceedings, [online] 3267, p.020002. [CrossRef]
- Nishi, L., Marquetotti, A., Lucia, A., Carvalho, M., Francisco, G. and Bergamasco, R. (2013). Application of Hybrid Process of Coagulation/Flocculation and Membrane Filtration for the Removal of Protozoan Parasites from Water. InTech eBooks. [CrossRef]
- Bouchareb, R., Derbal, K., Özay, Y., Bilici, Z. and Dizge, N. (2020). Combined natural/chemical coagulation and membrane filtration for wood processing wastewater treatment. Journal of Water Process Engineering, 37, p.101521. [CrossRef]
- Zularisam, A.W., Ismail, A.F., Salim, M.R., Sakinah, M. and Matsuura, T. (2009). Application of coagulation–ultrafiltration hybrid process for drinking water treatment: Optimization of operating conditions using experimental design. Separation and Purification Technology, 65(2), pp.193–210. [CrossRef]
- Merabti, L., Ouassila Benhabiles, Zahia Tigrine, Mounir Mellal, Chekir, N., Mahidine, S., Djilali Tassalit, Merouane Bentchikou, Douadi, A., Jammeli, L., Imen Khouni and Seif (2025). Hybrid Coagulation–Membrane Filtration Techniques for Sustainable Soap Wastewater Treatment. Water, 17(10), pp.1411–1411. [CrossRef]
- Zafisah, N.S., Ang, W.L., Mohammad, A.W., Hilal, N. and Johnson, D.J. (2020). Interaction between ballasting agent and flocs in ballasted flocculation for the removal of suspended solids in water. Journal of Water Process Engineering, [online] 33, p.101028. [CrossRef]
- Suzuki, Y., Kaku, R., Takahashi, K., Kanai, M., Tamai, S., Annaka, Y. and Chuganji, N. (2022). Optimum Conditions for High-Speed Solid–Liquid Separation by Ballast Flocculation. SSRN Electronic Journal. Available at: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=4138347.
- Gaikwad, V.T. and Munavalli, G.R. (2019). Turbidity removal by conventional and ballasted coagulation with natural coagulants. Applied Water Science, 9(5). [CrossRef]
- Trinh, T., van den Akker, B., Stuetz, R.M., Coleman, H.M., Le-Clech, P. and Khan, S.J. (2012). Removal of trace organic chemical contaminants by a membrane bioreactor. Water Science and Technology, 66(9), pp.1856–1863. [CrossRef]
- What is Membrane Bioreactor (MBR) Technology? - Coftec. [online] Coftec. Available at: https://www.coftec.ie/what-is-membrane-bioreactor-mbr-technology/ [Accessed : 19/06/2025].
- Chen, W. and Liu, J. (2012). The possibility and applicability of coagulation-MBR hybrid system in reclamation of dairy wastewater. Desalination, 285, pp.226–231. [CrossRef]
- Hadis Saeedikia, Minoo Ghanbarzadeh, Milad Mahmoudzadeh, Manijeh Khorsi, Masoud Barani, Saeed Bazgir, Tavakoli, O., Amir Heidarinasab, Lesage, G., Heran, M. and Farshid Pajoum Shariati (2025). A Hybrid Treatment System of the Coagulation/Flocculation/Settling Stage Followed by a Membrane Bioreactor (CFS-MBR) for Superior Removal of Cr and Nutrient Pollutants from Tannery Wastewater. Water, 17(10), pp.1432–1432. [CrossRef]
- Longina Stępniak and Stańczyk-Mazanek, E. (2022). Ultrasonic Energy as an Agent to Aid Water Treatment in the Coagulation Process. Energies, [online] 15(14), pp.5186–5186. [CrossRef]
- Jiang, H., Lu, H., Zhou, Y., Liu, Y. and Hao, C. (2023). High-efficiency degradation catalytic performance of a novel Angelica sinensis polysaccharide-silver nanomaterial for dyes by ultrasonic cavitation. Ultrasonics Sonochemistry, 93, p.106289. [CrossRef]
- ZHANG, G., WANG, B., ZHANG, P., WANG, L. and WANG, H. (2006). Removal of Algae by Sonication-Coagulation. Journal of Environmental Science and Health Part A, 41(7), pp.1379–1390. [CrossRef]
- Huang, Y., Ding, S., Li, L., Liao, Q., Chu, W. and Li, H. (2021). Ultrasound-enhanced coagulation for Microcystis aeruginosa removal and disinfection by-product control during subsequent chlorination. Water Research, 201, p.117334. [CrossRef]
- Hairom, N.H.H., Soon, C.F., Mohamed, R.M.S.R., Morsin, M., Zainal, N., Nayan, N., Zulkifli, C.Z. and Harun, N.H. (2021). A review of nanotechnological applications to detect and control surface water pollution. Environmental Technology & Innovation, [online] 24, p.102032. [CrossRef]
- Water and Wastewater. (2024). Use Of Nanoparticles In Water Treatment - Water & Wastewater. [online] Available at: https://www.waterandwastewater.com/use-of-nanoparticles-in-water-treatment/.
- Olawade, D.B., Wada, O.Z., Egbewole, B.I., Oluwaseun Fapohunda, Ige, A.O., Usman, S.O. and Olawale Ajisafe (2024). Metal and metal oxide nanomaterials for heavy metal remediation: novel approaches for selective, regenerative, and scalable water treatment. Frontiers in Nanotechnology, 6. [CrossRef]
- Jackson, J.S., K. Kantamaneni, P. Ganeshu, R. Sunkur and U. Rathnayake (2025). Assessment of the role of nanotechnology in water sector: an expert opinion. International Journal of Energy and Water Resources. [CrossRef]
- Thanigaivel, S., Priya, A.K., Gnanasekaran, L., Hoang, T.K.A., Rajendran, S. and Soto-Moscoso, M. (2022). Sustainable applicability and environmental impact of wastewater treatment by emerging nanobiotechnological approach: Future strategy for efficient removal of contaminants and water purification. Sustainable Energy Technologies and Assessments, 53, p.102484. [CrossRef]
- Nano-Magic for Clean Water: A dive into its implications. [online] Hydromo. Available at: https://hydromo.in/nano-magic-for-clean-water-a-dive-into-its-implications/.
- Anjum, M., Miandad, R., Waqas, M., Gehany, F. and Barakat, M.A. (2019). Remediation of wastewater using various nano-materials. Arabian Journal of Chemistry, 12(8), pp.4897–4919. [CrossRef]
- Mpongwana, N. and Rathilal, S. (2022). A Review of the Techno-Economic Feasibility of Nanoparticle Application for Wastewater Treatment. Water, 14(10), p.1550. [CrossRef]
- Islam, S.M.N., Rahman, S.H., Rahman, M.M., Adyel, T.M., Yesman, R.A., Ahmed, M.S. and Kaiser, N. (2011). Excessive Turbidity Removal from Textile Effluents Using Electrocoagulation Technique. Journal of Scientific Research, 3(3), pp.557–568. Available at: https://www.researchgate.net/publication/215716693_Excessive_Turbidity_Removal_from_Textile_Effluents_Using_Electrocoagulation_Technique.
- Mellifiq. (2025). Electrocoagulation Technology for Superior Water Treatment. [online] Available at: https://mellifiq.com/en/electrocoagulation-technology/.
- Tech, G.W. (2024). Electrocoagulation: The Future of Water Treatment Technology - Genesis Water Technologies. [online] Genesis Water Technologies. Available at: https://genesiswatertech.com/blog-post/electrocoagulation-the-future-of-water-treatment-technology/.
- Mollah, M.Y., Schennach, R., Parga, J.R. and Cocke, D.L. (2001). Electrocoagulation (EC)--science and applications. Journal of Hazardous Materials, [online] 84(1), pp.29–41. [CrossRef]
- Electrocoagulation. [online] Racoman.com. Available at: https://www.racoman.com/blog/electrocoagulation-wastewater-treatment-explained [Accessed : 19/06/2025].
- Ingelsson, M., Yasri, N. and Roberts, E.P.L. (2020). Electrode passivation, faradaic efficiency, and performance enhancement strategies in electrocoagulation—a review. Water Research, 187, p.116433. [CrossRef]
- Phu, T.K.C., Nguyen, P.L. and Phung, T.V.B. (2025). Recent progress in highly effective electrocoagulation-coupled systems for advanced wastewater treatment. iScience, [online] 28(3), p.111965. [CrossRef]
- Kuokkanen, V., Kuokkanen, T., Rämö, J. and Lassi, U. (2013). Recent Applications of Electrocoagulation in Treatment of Water and Wastewater—A Review. Green and Sustainable Chemistry, 03(02), pp.89–121. [CrossRef]
- Chow, H. and Pham, A.L.-T. (2021). Mitigating Electrode Fouling in Electrocoagulation by Means of Polarity Reversal: The Effects of Electrode Type, Current Density, and Polarity Reversal Frequency. Water Research, 197, p.117074. [CrossRef]
- Kumari, P. and Kumar, A. (2023). ADVANCED OXIDATION PROCESS: A remediation technique for organic and non-biodegradable pollutant. Results in Surfaces and Interfaces, [online] 11, p.100122. [CrossRef]
- Deng, Y. and Zhao, R. (2015). Advanced Oxidation Processes (AOPs) in Wastewater Treatment. Current Pollution Reports, [online] 1(3), pp.167–176. [CrossRef]
- Srivastav, M., Gupta, M., Agrahari, S.K. and Detwal, P. (2019). Removal of Refractory Organic Compounds from Wastewater by Various Advanced Oxidation Process - A Review. Current Environmental Engineering, 6(1), pp.8–16. [CrossRef]
- Gopalakrishnan, G., Jeyakumar, R.B. and Somanathan, A. (2023). Challenges and Emerging Trends in Advanced Oxidation Technologies and Integration of Advanced Oxidation Processes with Biological Processes for Wastewater Treatment. Sustainability, 15(5), p.4235. [CrossRef]
- Tadele Assefa Aragaw and Fekadu Mazengiaw Bogale (2023). Role of coagulation/flocculation as a pretreatment option to reduce colloidal/bio-colloidal fouling in tertiary filtration of textile wastewater: A review and future outlooks. 11. Avialable at: . [CrossRef]
- Hashmi, Z., Fahrurrozi, Zaini, J., Lim, J.W., Abu Bakar, M.S., Dani Nandiyanto, A.B. and Bilad, M.R. (2025). Current progress on filtration techniques for recovery of microalgae-based products. Algal Bioreactors, pp.71–98. [CrossRef]
| Traditional Treatment Techniques | Modern Treatment Techniques |
|---|---|
| Coagulation (with flocculation) | Nano Technology based methods |
| Filtration and Roughing Filtration | Electro Coagulation |
| Sedimentation | Advanced Oxidation Processes |
| Adsorption | Hybrid Technologies |
| Membrane based technologies |
| Absorbent | Characteristics | |
| Carbon based nanomaterials | Carbon Nano Tubes (CNT) |
|
| Graphene and graphene oxide |
|
|
| Metal-based nano particles | Iron oxide nano particles (e.g. Hematite which has magnetic properties)) |
|
| Other metal oxides (Manganese, Zinc, Magnesium oxides, Titaniumoxide) |
|
|
| Polymer based nano particles |
|
|
| Zeolites |
|
|
| Nanocomposites |
|
|
| Bio Adsorbents | ||
| Study | Nano adsorbent | Water type | Initial turbidity level | Turbidity removal | Reference |
| Graphene Oxide Coagulant | Graphene oxide | Surface water | 20-200 NTU | ≥95% removal | [61] |
| Nano Cellulose Titaium Oxide Composite | Nano Cellulose/ Titaium Oxide |
Textile wastewater | 11.6–83.4 NTU | 97.5-100% | [62] |
| Iron Oxide coated with sand | Nano iron oxide | Strom water runoff | N/A | 90.8-95.7% | [63] |
| Treatment Technique | Pros | Cons |
| Nano Technology based methods | High removal efficiency and surface area: Nano materials, due to their extremely high surface area-to-volume ratio, can effectively interact with suspended particles [138,139] | Environmental and Health Risks: The environmental and health implications of releasing nano particles into ecosystems has not yet being fully understood. Thus, there is a risk that Nano particles can pose risks to aquatic life and human health [144]. |
| Scalability and Integration: Nano particle-based water treatment methods can be integrated into existing water treatment processes which can enhance their capabilities. They are also scalable, from lab-scale to industrial-scale applications [139,140] | Stability, fouling and regeneration issues: In natural water systems, the performance may be compromised and fouling may become an major issue [139]. | |
| Multi functionality and specificity : Different nano materials can be engineered for various pollutant types, combining turbidity reduction with disinfection or chemical removal [141,142] . | High Production Costs: Producing nano particles with the necessary requirement can be costly. As such, adopting such a technology will be rejected by certain countries, particularly, developing countries [139]. | |
| Energy and Cost Efficiency: Nanotechnology-based water purification processes often require less energy and fewer chemicals compared to traditional methods, making them more environmentally friendly and cost-effective in the long run [141,142]. | Regulatory Hurdles: The absence of clear regulations for using nano particles in water treatment makes their application more difficult. Governments and regulatory agencies need to develop guidelines to ensure they are used safely and effectively [139]. | |
| Small-Scale Applications: Nanotechnology can be applied in small-scale systems, making it suitable for decentralized water treatment in remote or resource-limited areas [143]. | High operational cost: The production of nano materials, the necessity of specialized equipment for fabrication gives rise to higher operating costs [145]. | |
| Electro Coagulation (EC) | High turbidity removal efficiency: EC can remove 90–99% of turbidity efficiently, even in highly polluted water [146]. | Electrode surface fouling and replacement: Electrodes corrode and may require frequent cleaning or replacement due to surface fouling (this may occur due to deposition of unwanted materials on the electrode surface [151]. |
| No need for chemical additives: Coagulants are generated electro chemically insitu, reducing the need for external chemicals [147]. | High energy consumption: Especially for water with high conductivity or extended treatment times [152]. | |
| Compact and easily operational system: EC units are modular, compact, and relatively simple to automate [148]. | pH sensitivity: EC performance depends on the initial pH level of the water to be treated and may require adjustment [153]. | |
| Lower sludge volume: The sludge produced by EC is generally denser and easier to dewater compared to chemical coagulation [149]. | Scaling and maintenance: Scale formation (precipitation from mineral salts on the electrode surfaces) on electrodes can hinder current flow and reduce efficiency [154]. | |
| Wide range of applicability: Effective for various types of water: such as surface water, industrial wastewater, oil-in-water emulsions, etc [150]. | Sludge disposal : While the sludge produced is less than chemical coagulation, this sludge still requires proper treatment/disposal. |
|
| Advanced Oxidation Processes | Effective in oxidizing organic turbidity sources: AOPs can degrade humic substances and colloidal organics contributing to turbidity [155]. | Cannot be used for treating large amount of waste, so it is generally used as a secondary treatment for the removal of trace amount of organic, non-biodegradable contaminants [155]. |
| Disinfection alongside turbidity reduction: AOPs (especially UV/H₂O₂, O₃/H₂O₂) inactivate microorganisms, improving clarity [156]. | High energy and chemical costs: UV, ozone, and hydrogen peroxide systems are expensive to install and operate [158] | |
| Reduces refractory compounds: Removes low molecular weight dissolved organics that contribute to “non-filterable” turbidity [157]. | High turbidity can reduce AOP efficiency: Suspended solids scatter light and consume oxidants, reducing the effectiveness of AOPs like UV/H₂O₂ or ozonation [8]. | |
| Hybrid Technologies | Combining methods: such as coagulation with membrane filtration, or non-chemical approaches like pebble matrix filtration (PMF) integrated with other selected technologies, can achieve more complete removal of suspended solids and colloids, thereby reducing the load on downstream treatment processes. [159]. | Higher capital and operational costs: More units and controls mean more cost for installation, operation, and maintenance. |
| Improved membrane lifespan: Pre-treatments like coagulation or EC reduce membrane fouling and extend membrane life [160]. | Increased operational complexity:Requires skilled operators and careful system integration. | |
| Reduced chemical or energy use: The integration of the method combined may reduce dosage or operational costs for each individual technology. | Space requirements: Combining systems increases the system footprint compared to a single unit. | |
| More stable water quality: Hybrid systems can buffer raw water variability better than single-unit systems. | Sludge management: If coagulation or electro coagulation is involved, additional sludge is generated that needs handling. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




