Submitted:
25 September 2025
Posted:
26 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Potential Causes of Myasthenia in ME/CFS
- 1)
- Lack of energy due to mitochondrial dysfunction
- 2)
- Atrophy due to inactivity in the intention to avoid PEM and due to immobilization (deconditioning)
- 3)
- Skeletal muscle damage
- 4)
- Electrophysiological causes leading to insufficient excitation and recruitment of muscle fibers upon neuromuscular activation
3. The Potential Role of the Na+/K+-ATPase in Physiology and Pathophysiology of ME/CFS
4. The Physiological Role of the Na+/K+-ATPase in Muscle Electrophysiology and Metabolism
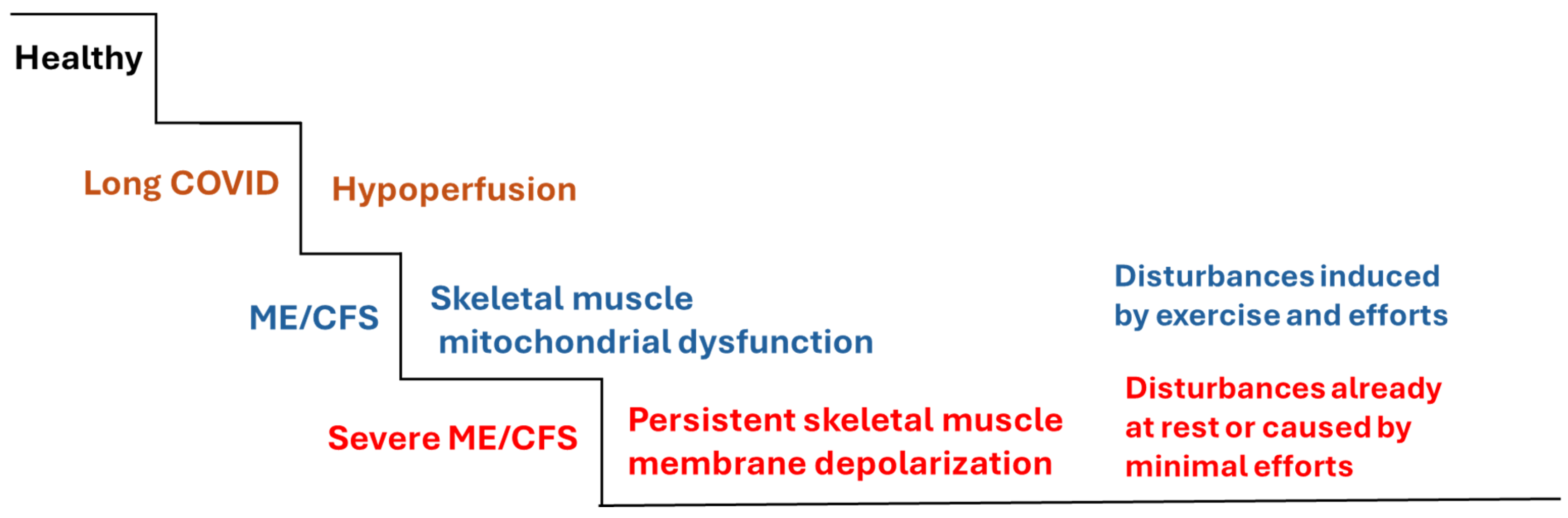
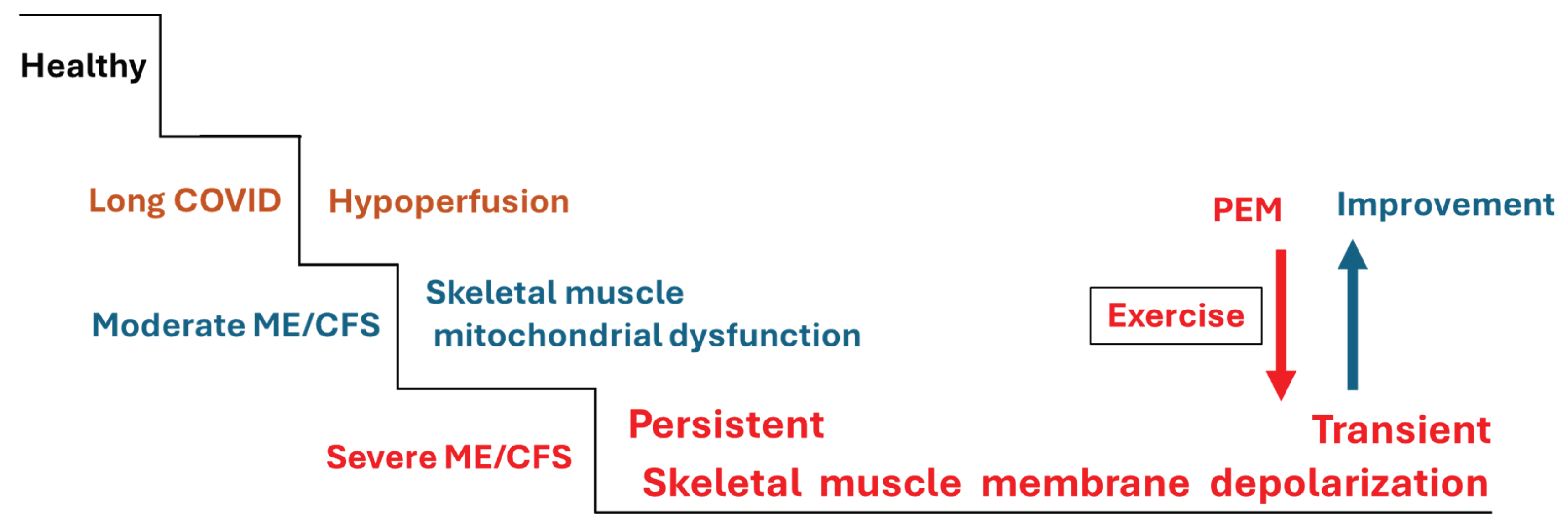
5. Potential Disturbances of Na+/K+-ATPase in ME/CFS
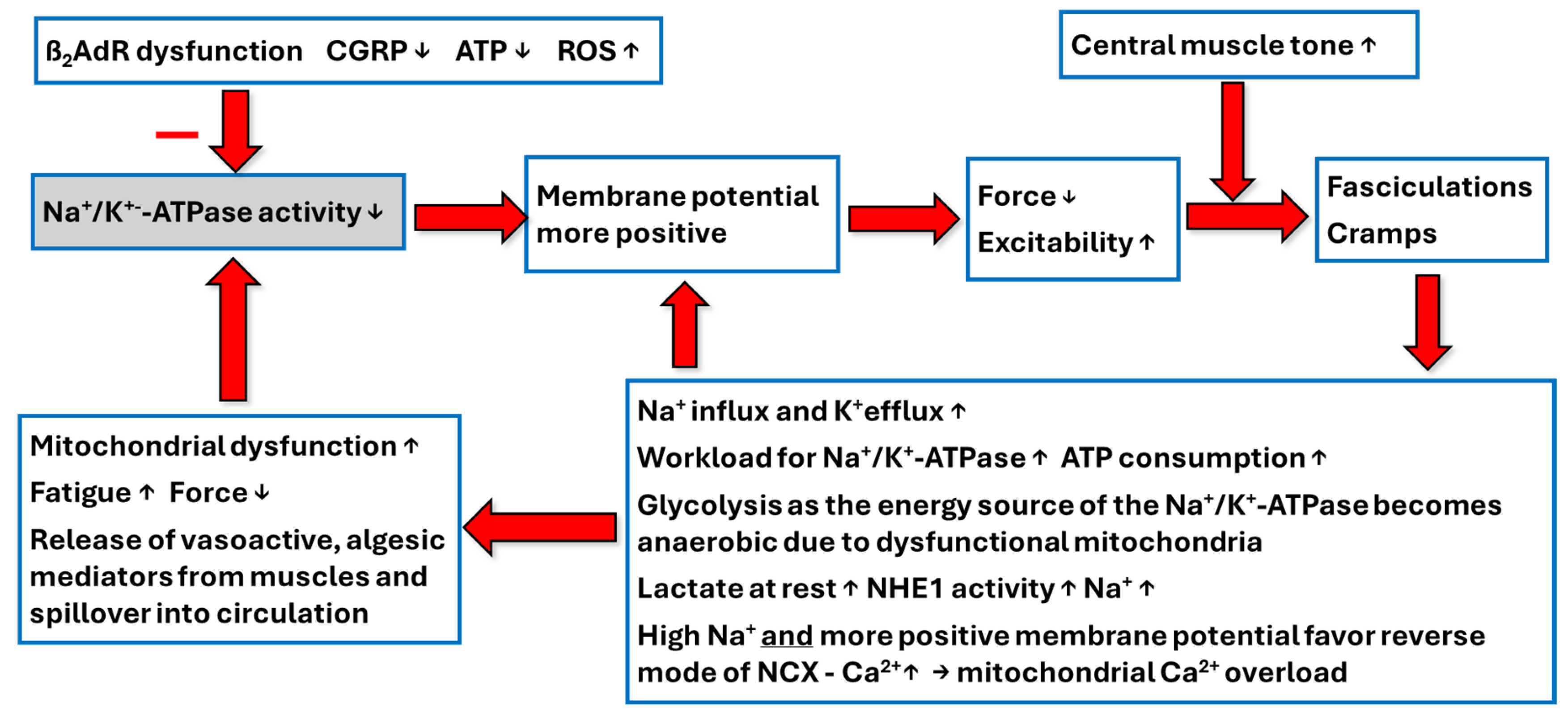
6. High Workload and Energy Consumption of Na+/K+-ATPase by Sarcolemmal Depolarization and Fasciculations
7. Energy Supply of the Na+/K+-ATPase and Its Disturbances
8. Central Muscle Tone and Depolarized Sarcolemma Interact to Cause Inappropriate Excitations
9. Conclusions
References
- Choutka J, Jansari V, Hornig M, Iwasaki A. Unexplained post-acute infection syndromes. Nature Medicine 2022;28:911–23.
- Kedor C, Freitag H, Meyer-Arndt L, Wittke K, Hanitsch LG, Zoller T, et al. A prospective observational study of post-COVID-19 chronic fatigue syndrome following the first pandemic wave in Germany and biomarkers associated with symptom severity. Nature Communications 2022;13:5104. [CrossRef]
- Peter RS, Nieters A, Göpel S, Merle U, Steinacker JM, Deibert P, et al. Persistent symptoms and clinical findings in adults with post-acute sequelae of COVID-19/post-COVID-19 syndrome in the second year after acute infection: A population-based, nested case-control study. PLoS Medicine 2025;22:e1004511.
- Hill E, Mehta H, Sharma S, Mane K, Xie C, Cathey E, et al. Risk factors associated with post-acute sequelae of SARS-CoV-2 in an EHR cohort: a national COVID cohort collaborative (N3C) analysis as part of the NIH RECOVER program. medRxiv 2022.
- Bowe B, Xie Y, Al-Aly Z. Postacute sequelae of COVID-19 at 2 years. Nature Medicine 2023;29:2347–57.
- Steinacker JM, Klinkisch E-M. Halbierte Sichtbarkeit: Aktivität und Rehabilitationsmaßnahmen bei postakuten Infektionssyndromen The (In-) Visible Half: Activity and Rehabilitation Measures for Post-acute Infection Syndromes. Verhaltenstherapie 2025.
- Carruthers BM, van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, et al. Myalgic encephalomyelitis: International Consensus Criteria. J Intern Med 2011;270:327–38. [CrossRef]
- Ballouz T, Menges D, Anagnostopoulos A, Domenghino A, Aschmann HE, Frei A, et al. Recovery and symptom trajectories up to two years after SARS-CoV-2 infection: population based, longitudinal cohort study. Bmj 2023;381.
- van Campen C, Rowe PC, Visser FC. Cerebral Blood Flow Is Reduced in Severe Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients During Mild Orthostatic Stress Testing: An Exploratory Study at 20 Degrees of Head-Up Tilt Testing. Healthcare (Basel) 2020;8. [CrossRef]
- van Campen CMC, Rowe PC, Visser FC. Worsening Symptoms Is Associated with Larger Cerebral Blood Flow Abnormalities during Tilt-Testing in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Medicina 2023;59. [CrossRef]
- Ryabkova VA, Churilov LP, Shoenfeld Y. Neuroimmunology: what role for autoimmunity, neuroinflammation, and small fiber neuropathy in fibromyalgia, chronic fatigue syndrome, and adverse events after human papillomavirus vaccination? International Journal of Molecular Sciences 2019;20:5164.
- Cervia-Hasler C, Brüningk SC, Hoch T, Fan B, Muzio G, Thompson RC, et al. Persistent complement dysregulation with signs of thromboinflammation in active Long Covid. Science 2024;383:eadg7942.
- Haunhorst S, Dudziak D, Scheibenbogen C, Seifert M, Sotzny F, Finke C, et al. Towards an understanding of physical activity-induced post-exertional malaise: Insights into microvascular alterations and immunometabolic interactions in post-COVID condition and myalgic encephalomyelitis/chronic fatigue syndrome. Infection 2024:1–13.
- Conroy K, Bhatia S, Islam M, Jason LA. Homebound versus bedridden status among those with myalgic encephalomyelitis/chronic fatigue syndrome. vol. 9, MDPI; 2021, p. 106.
- Sommerfelt K, Schei T, Angelsen A. Severe and very severe Myalgic encephalopathy/chronic fatigue syndrome me/Cfs in Norway: symptom burden and access to care. J Clin Med 2023;12:1487.
- Paffrath A, Kim L, Kedor C, Stein E, Rust R, Freitag H, et al. Impaired hand grip strength correlates with greater disability and symptom severity in post-COVID Myalgic encephalomyelitis/chronic fatigue syndrome. Journal of Clinical Medicine 2024;13:2153.
- Jäkel B, Kedor C, Grabowski P, Wittke K, Thiel S, Scherbakov N, et al. Hand grip strength and fatigability: correlation with clinical parameters and diagnostic suitability in ME/CFS. Journal of Translational Medicine 2021;19:1–12.
- Nacul LC, Mudie K, Kingdon CC, Clark TG, Lacerda EM. Hand grip strength as a clinical biomarker for ME/CFS and disease severity. Frontiers in Neurology 2018;9:992.
- do Amaral CMSSB, da Luz Goulart C, da Silva BM, Valente J, Rezende AG, Fernandes E, et al. Low handgrip strength is associated with worse functional outcomes in long COVID. Scientific Reports 2024;14:2049. [CrossRef]
- Blitshteyn S, Ruhoy IS, Natbony LR, Saperstein DS. Internal Tremor in Long COVID May Be a Symptom of Dysautonomia and Small Fiber Neuropathy. Neurology International 2024;17:2.
- Zhou T, Sawano M, Arun AS, Caraballo C, Michelsen T, McAlpine LS, et al. Internal tremors and vibrations in long COVID: a cross-sectional study. The American Journal of Medicine 2024.
- Appelman B, Charlton BT, Goulding RP, Kerkhoff TJ, Breedveld EA, Noort W, et al. Muscle abnormalities worsen after post-exertional malaise in long COVID. Nature Communications 2024;15:17. [CrossRef]
- Bizjak DA, Ohmayer B, Buhl JL, Schneider EM, Walther P, Calzia E, et al. Functional and Morphological Differences of Muscle Mitochondria in Chronic Fatigue Syndrome and Post-COVID Syndrome. International Journal of Molecular Sciences 2024;25. [CrossRef]
- Joseph P, Arevalo C, Oliveira RKF, Faria-Urbina M, Felsenstein D, Oaklander AL, et al. Insights From Invasive Cardiopulmonary Exercise Testing of Patients With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. CHEST 2021;160:642–51. [CrossRef]
- de Boer E, Petrache I, Goldstein NM, Olin JT, Keith RC, Modena B, et al. Decreased fatty acid oxidation and altered lactate production during exercise in patients with post-acute COVID-19 syndrome. American Journal of Respiratory and Critical Care Medicine 2022;205:126–9.
- Hoel F, Hoel A, Pettersen IK, Rekeland IG, Risa K, Alme K, et al. A map of metabolic phenotypes in patients with myalgic encephalomyelitis/chronic fatigue syndrome. JCI Insight 2021;6:e149217.
- Vollrath S, Bizjak DA, Zorn J, Matits L, Jerg A, Munk M, et al. Recovery of performance and persistent symptoms in athletes after COVID-19. PLoS One 2022;17:e0277984.
- Guarnieri JW, Haltom JA, Albrecht YES, Lie T, Olali AZ, Widjaja GA, et al. SARS-CoV-2 mitochondrial metabolic and epigenomic reprogramming in COVID-19. Pharmacological Research 2024;204:107170.
- Charlton BT, Slaghekke A, Appelman B, Eggelbusch M, Huijts JY, Noort W, et al. Skeletal muscle properties in long COVID and ME/CFS differ from those induced by bed rest. medRxiv 2025:2025–05.
- Scheibenbogen C, Wirth KJ. Key pathophysiological role of skeletal muscle disturbance in post COVID and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): accumulated evidence. Journal of Cachexia, Sarcopenia and Muscle 2025;16:e13669.
- Wirth KJ, Scheibenbogen C. Pathophysiology of skeletal muscle disturbances in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). J Transl Med 2021;19:162. [CrossRef]
- Semsarian C, Wu M-J, Ju Y-K, Marciniec T, Yeoh T, Allen DG, et al. Skeletal muscle hypertrophy is mediated by a Ca2+-dependent calcineurin signalling pathway. Nature 1999;400:576–81.
- Wirth KJ, Löhn M. Microvascular Capillary and Precapillary Cardiovascular Disturbances Strongly Interact to Severely Affect Tissue Perfusion and Mitochondrial Function in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Evolving from the Post COVID-19 Syndrome. Medicina 2024;60. [CrossRef]
- Clausen T, Andersen SL, Flatman JA. Na(+)-K+ pump stimulation elicits recovery of contractility in K(+)-paralysed rat muscle. J Physiol 1993;472:521–36. [CrossRef]
- Murphy KT, Clausen T. The importance of limitations in aerobic metabolism, glycolysis, and membrane excitability for the development of high-frequency fatigue in isolated rat soleus muscle. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 2007;292:R2001–11.
- Nielsen OB, Clausen T. The significance of active Na+, K+ transport in the maintenance of contractility in rat skeletal muscle. Acta Physiologica Scandinavica 1996;157:199–209.
- Wareham, AC. Effect of denervation and ouabain on the response of the resting membrane potential of rat skeletal muscle to potassium. Pflügers Archiv 1978;373:225–8.
- Petter E, Scheibenbogen C, Linz P, Stehning C, Wirth K, Kuehne T, et al. Muscle sodium content in patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Journal of Translational Medicine 2022;20:580. [CrossRef]
- Clausen, T. Role of Na+, K+-pumps and transmembrane Na+, K+-distribution in muscle function: The FEPS Lecture–Bratislava 2007. Acta Physiologica 2008;192:339–49.
- Pirkmajer S, Chibalin AV. Na,K-ATPase regulation in skeletal muscle. Am J Physiol Endocrinol Metab 2016;311:E1–31. [CrossRef]
- Clausen, T. Na+-K+ Pump Regulation and Skeletal Muscle Contractility. Physiological Reviews 2003;83:1269–324. [CrossRef]
- Nielsen OB, Harrison AP. The regulation of the Na+, K+ pump in contracting skeletal muscle. Acta Physiologica Scandinavica 1998;162:191–200.
- Odoom JE, Kemp GJ, Radda GK. The regulation of total creatine content in a myoblast cell line. Molecular and Cellular Biochemistry 1996;158:179–88.
- Zorzano A, Fandos C, PalacÍN M. Role of plasma membrane transporters in muscle metabolism. Biochemical Journal 2000;349:667–88.
- Page MJ, Di Cera E. Role of Na+ and K+ in enzyme function. Physiological Reviews 2006;86:1049–92.
- Scheibenbogen C, Loebel M, Freitag H, Krueger A, Bauer S, Antelmann M, et al. Immunoadsorption to remove ß2 adrenergic receptor antibodies in Chronic Fatigue Syndrome CFS/ME. PLoS One 2018;13:e0193672. [CrossRef]
- Lohse MJ, Engelhardt S, Eschenhagen T. What is the role of β-adrenergic signaling in heart failure? Circulation Research 2003;93:896–906.
- Abrams RMC, Simpson DM, Navis A, Jette N, Zhou L, Shin SC. Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle & Nerve 2022;65:440–3. [CrossRef]
- Oaklander AL, Nolano M. Scientific Advances in and Clinical Approaches to Small-Fiber Polyneuropathy: A Review. JAMA Neurol 2019. [CrossRef]
- Cabanas H, Muraki K, Balinas C, Eaton-Fitch N, Staines D, Marshall-Gradisnik S. Validation of impaired Transient Receptor Potential Melastatin 3 ion channel activity in natural killer cells from Chronic Fatigue Syndrome/ Myalgic Encephalomyelitis patients. Molecular Medicine 2019;25:14. [CrossRef]
- Cabanas H, Muraki K, Eaton N, Balinas C, Staines D, Marshall-Gradisnik S. Loss of Transient Receptor Potential Melastatin 3 ion channel function in natural killer cells from Chronic Fatigue Syndrome/Myalgic Encephalomyelitis patients. Molecular Medicine 2018;24:44. [CrossRef]
- Eaton-Fitch N, Du Preez S, Cabanas H, Muraki K, Staines D, Marshall-Gradisnik S. Impaired TRPM3-dependent calcium influx and restoration using Naltrexone in natural killer cells of myalgic encephalomyelitis/chronic fatigue syndrome patients. Journal of Translational Medicine 2022;20:94. [CrossRef]
- Alonso-Carbajo L, Alpizar YA, Startek JB, Lopez-Lopez JR, Perez-Garcia MT, Talavera K. Activation of the cation channel TRPM3 in perivascular nerves induces vasodilation of resistance arteries. J Mol Cell Cardiol 2019;129:219–30. [CrossRef]
- Held K, Kichko T, De Clercq K, Klaassen H, Van Bree R, Vanherck J-C, et al. Activation of TRPM3 by a potent synthetic ligand reveals a role in peptide release. Proceedings of the National Academy of Sciences 2015;112:E1363–72. [CrossRef]
- Held K, Tóth BI. TRPM3 in brain (patho) physiology. Frontiers in Cell and Developmental Biology 2021;9:635659.
- Jammes Y, Adjriou N, Kipson N, Criado C, Charpin C, Rebaudet S, et al. Altered muscle membrane potential and redox status differentiates two subgroups of patients with chronic fatigue syndrome. J Transl Med 2020;18:173. [CrossRef]
- Al Masoodi WTM, Radhi SW, Abdalsada HK, Niu M, Al-Hakeim HK, Maes M. Increased galanin-galanin receptor 1 signaling, inflammation, and insulin resistance are associated with affective symptoms and chronic fatigue syndrome due to long COVID. PLoS One 2025;20:e0316373.
- Allain TJ, Bearn JA, Coskeran P, Jones J, Checkley A, Butler J, et al. Changes in growth hormone, insulin, insulinlike growth factors (IGFs), and IGF-binding protein-1 in chronic fatigue syndrome. Biol Psychiatry 1997;41:567–73. [CrossRef]
- Beentjes SV, Miralles Méharon A, Kaczmarczyk J, Cassar A, Samms GL, Hejazi NS, et al. Replicated blood-based biomarkers for myalgic encephalomyelitis not explicable by inactivity. EMBO Molecular Medicine 2025:1–24.
- Al-Hakeim HK, Khairi Abed A, Rouf Moustafa S, Almulla AF, Maes M. Tryptophan catabolites, inflammation, and insulin resistance as determinants of chronic fatigue syndrome and affective symptoms in long COVID. Frontiers in Molecular Neuroscience 2023;16:1194769.
- Agergaard J, Yamin Ali Khan B, Engell-Sørensen T, Schiøttz-Christensen B, Østergaard L, Hejbøl EK, et al. Myopathy as a cause of Long COVID fatigue: Evidence from quantitative and single fiber EMG and muscle histopathology. Clinical Neurophysiology 2023;148:65–75. [CrossRef]
- Jammes Y, Retornaz F. Understanding neuromuscular disorders in chronic fatigue syndrome. F1000Res 2019;8:F1000-Faculty.
- Blaustein MP, Lederer WJ. Sodium/Calcium Exchange: Its Physiological Implications. Physiological Reviews 1999;79:763–854. [CrossRef]
- Dutka TL, Lamb GD. Na+-K+ pumps in the transverse tubular system of skeletal muscle fibers preferentially use ATP from glycolysis. American Journal of Physiology-Cell Physiology 2007;293:C967–77.
- Ghali A, Lacout C, Ghali M, Gury A, Beucher A-B, Lozac’h P, et al. Elevated blood lactate in resting conditions correlate with post-exertional malaise severity in patients with Myalgic encephalomyelitis/Chronic fatigue syndrome. Scientific Reports 2019;9:18817.
- Nelson MJ, Bahl JS, Buckley JD, Thomson RL, Davison K. Evidence of altered cardiac autonomic regulation in myalgic encephalomyelitis/chronic fatigue syndrome: A systematic review and meta-analysis. Medicine 2019;98:e17600.
- Wyller VB, Eriksen HR, Malterud K. Can sustained arousal explain the Chronic Fatigue Syndrome? Behavioral and Brain Functions 2009;5:1–10.
- Löhn M, Wirth KJ. Potential pathophysiological role of the ion channel TRPM3 in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and the therapeutic effect of low-dose naltrexone. Journal of Translational Medicine 2024;22:630. [CrossRef]
- Seljeset S, Liebowitz S, Bright DP, Smart TG. Pre-and postsynaptic modulation of hippocampal inhibitory synaptic transmission by pregnenolone sulphate. Neuropharmacology 2023;233:109530.
- Hoheisel F, Fleischer KM, Rubarth K, Sepulveda N, Bauer S, Konietschke F, et al. Autoantibodies to Arginine-rich Sequences Mimicking Epstein-Barr Virus in Post-COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. medRxiv 2024:2024–12.
- Nakano Y, Wiechert S, Bánfi B. Overlapping activities of two neuronal splicing factors switch the GABA effect from excitatory to inhibitory by regulating REST. Cell Reports 2019;27:860–71.
- Schlauch KA, Khaiboullina SF, De Meirleir KL, Rawat S, Petereit J, Rizvanov AA, et al. Genome-wide association analysis identifies genetic variations in subjects with myalgic encephalomyelitis/chronic fatigue syndrome. Translational Psychiatry 2016;6:e730–e730.
- Artalejo AR, Olivos-Oré LA. Alpha2-adrenoceptors in adrenomedullary chromaffin cells: functional role and pathophysiological implications. Pflügers Archiv-European Journal of Physiology 2018;470:61–6.
- Wirth KJ, Scheibenbogen C, Paul F. An attempt to explain the neurological symptoms of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Journal of Translational Medicine 2021;19:471. [CrossRef]
- Gyires K, Zádori ZS, Török T, Mátyus P. α2-Adrenoceptor subtypes-mediated physiological, pharmacological actions. Neurochemistry International 2009;55:447–53.
- Gyires K, Zádori ZS, Török T, Mátyus P. α2-Adrenoceptor subtypes-mediated physiological, pharmacological actions. Neurochemistry International 2009;55:447–53.
- Scherbakov N, Szklarski M, Hartwig J, Sotzny F, Lorenz S, Meyer A, et al. Peripheral endothelial dysfunction in myalgic encephalomyelitis/chronic fatigue syndrome. ESC Heart Fail 2020;7:1064–71. [CrossRef]
- van Campen CM, Visser FC. The Relation Between Cardiac Output and Cerebral Blood Flow in ME/CFS Patients with a POTS Response During a Tilt Test. Journal of Clinical Medicine 2025;14:3648.
- Rosin DL, Zeng D, Stornetta RL, Norton FR, Riley T, Okusa MD, et al. Immunohistochemical localization of alpha 2A-adrenergic receptors in catecholaminergic and other brainstem neurons in the rat. Neuroscience 1993;56:139–55. [CrossRef]
- Hayashi Y, Maze M. Alpha2 adrenoceptor agonists and anaesthesia. BJA: British Journal of Anaesthesia 1993;71:108–18.
- Wyller VB, Saul JP, Walløe L, Thaulow E. Sympathetic cardiovascular control during orthostatic stress and isometric exercise in adolescent chronic fatigue syndrome. European Journal of Applied Physiology 2008;102:623–32. [CrossRef]
- Van der Stede T, Van de Loock A, Turiel G, Hansen C, Tamariz-Ellemann A, Ullrich M, et al. Cellular deconstruction of the human skeletal muscle microenvironment identifies an exercise-induced histaminergic crosstalk. Cell Metabolism 2025;37:842-856.e7. [CrossRef]
- Wirth K, Scheibenbogen C. A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Recognitions from the finding of autoantibodies against ss2-adrenergic receptors. Autoimmun Rev 2020;19:102527. [CrossRef]
- Retornaz F, Stavris C, Jammes Y. Consequences of sarcolemma fatigue on maximal muscle strength production in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Clinical Biomechanics 2023;108:106055.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



