Submitted:
24 September 2025
Posted:
25 September 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
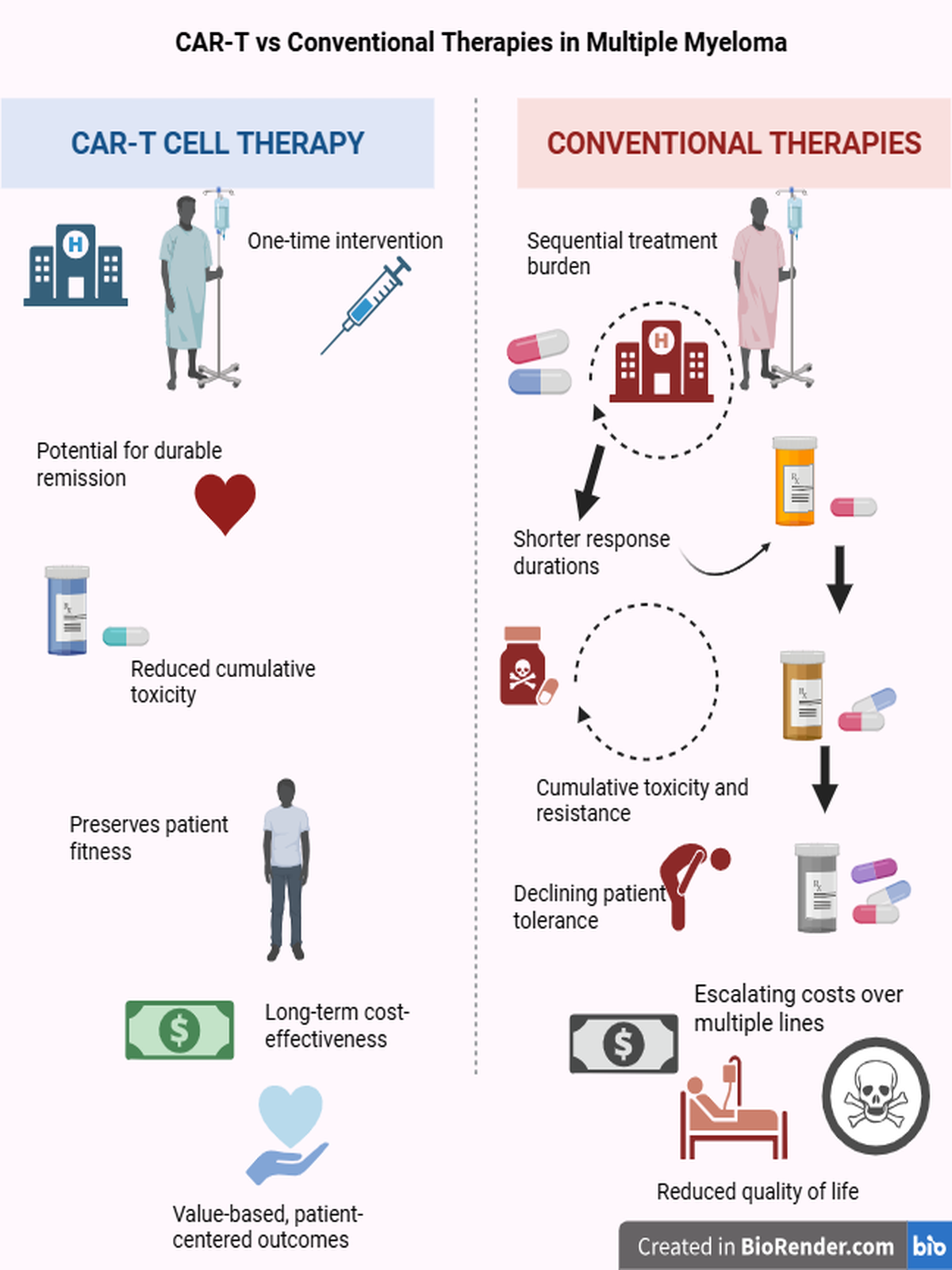
2. Myeloma Therapies
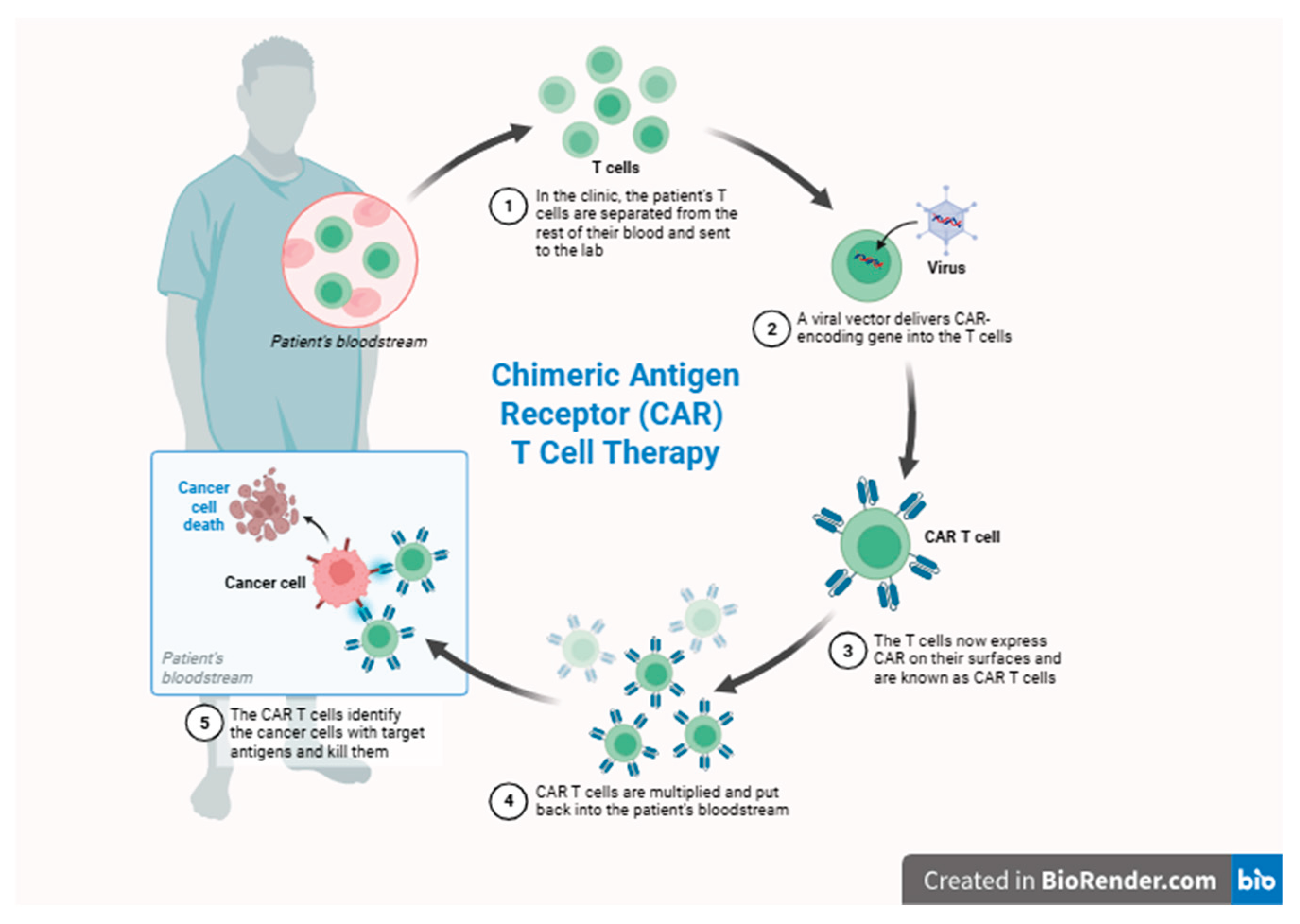
3. From Immunotherapy Origins to Car-T
4. First in Class Approvals: Ide-Cel and Cilta-Cel
5. Why Bcma Is an Ideal Target in Multiple Myeloma
6. Challenges and Future Directions
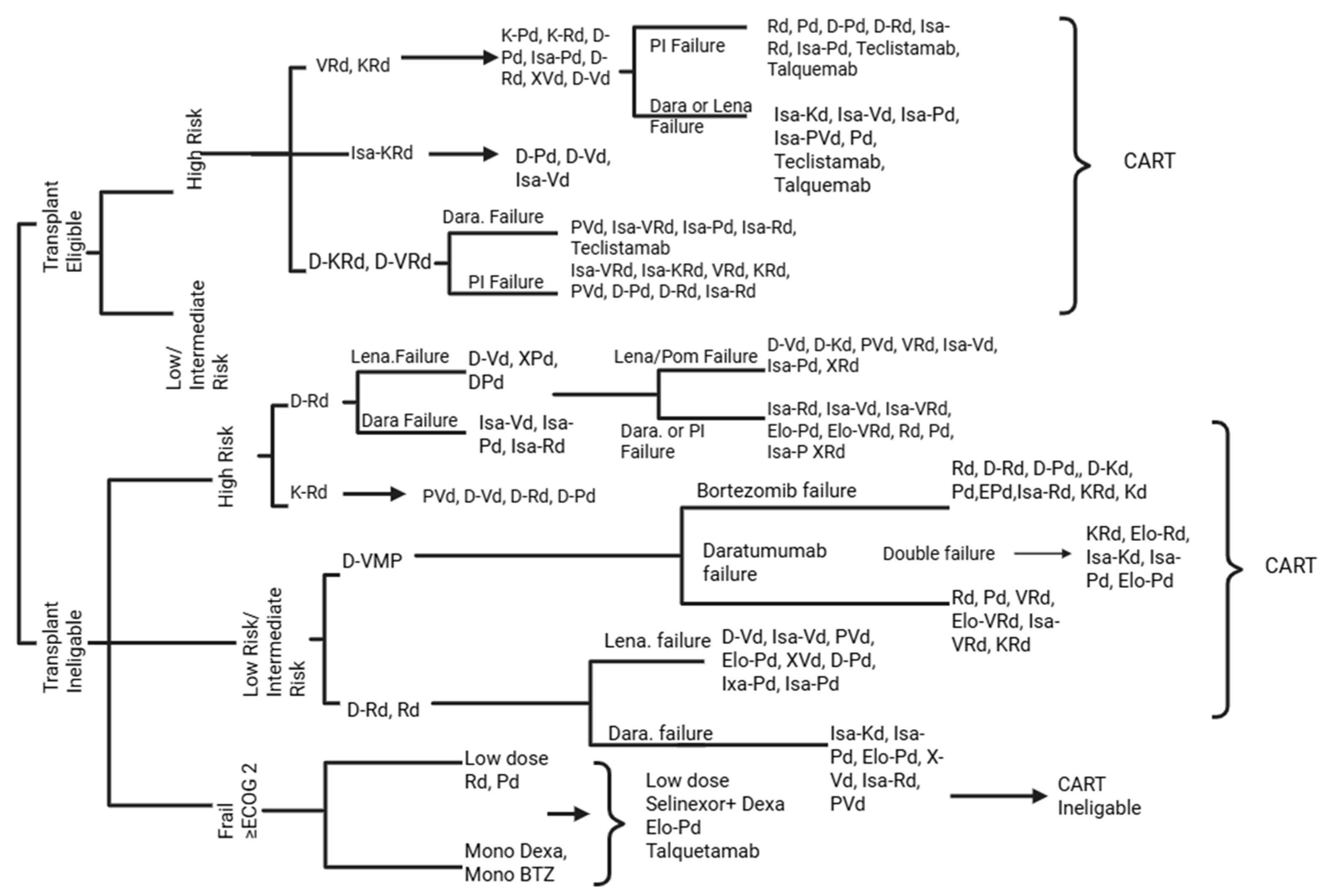
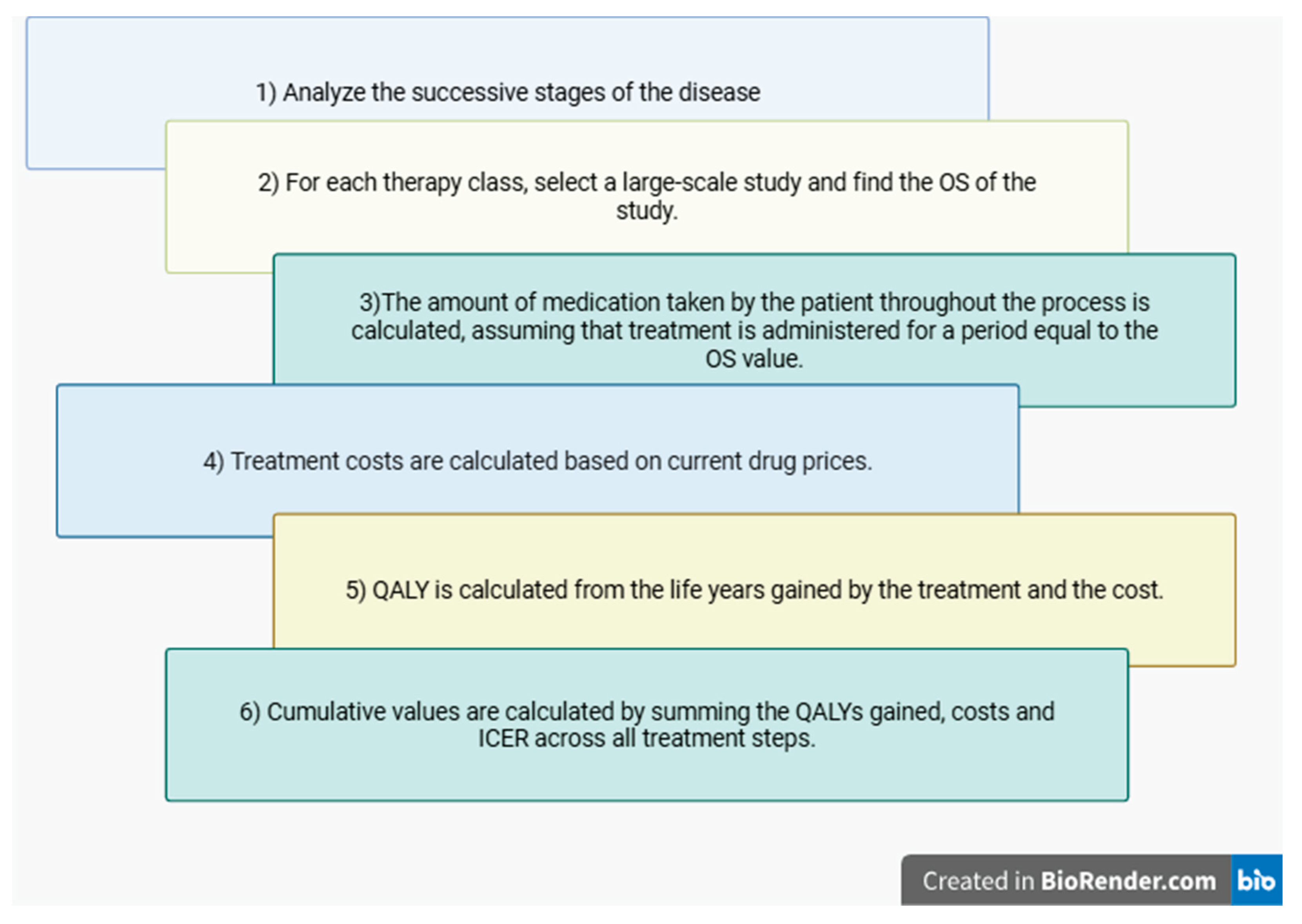
7. Materials and Methods
- ECOG 0 → 0.9
- ECOG 1 → 0.7
- ECOG 2 → 0.5
- ECOG 3 → 0.3
- ECOG 4 → 0.1
- ECOG 5 → 0

- Total ICER values
- QALY outputs
- Across transplant-eligible, ineligible, and frail patient groups
- Policymakers
- Clinicians
- Health economists
8. Results
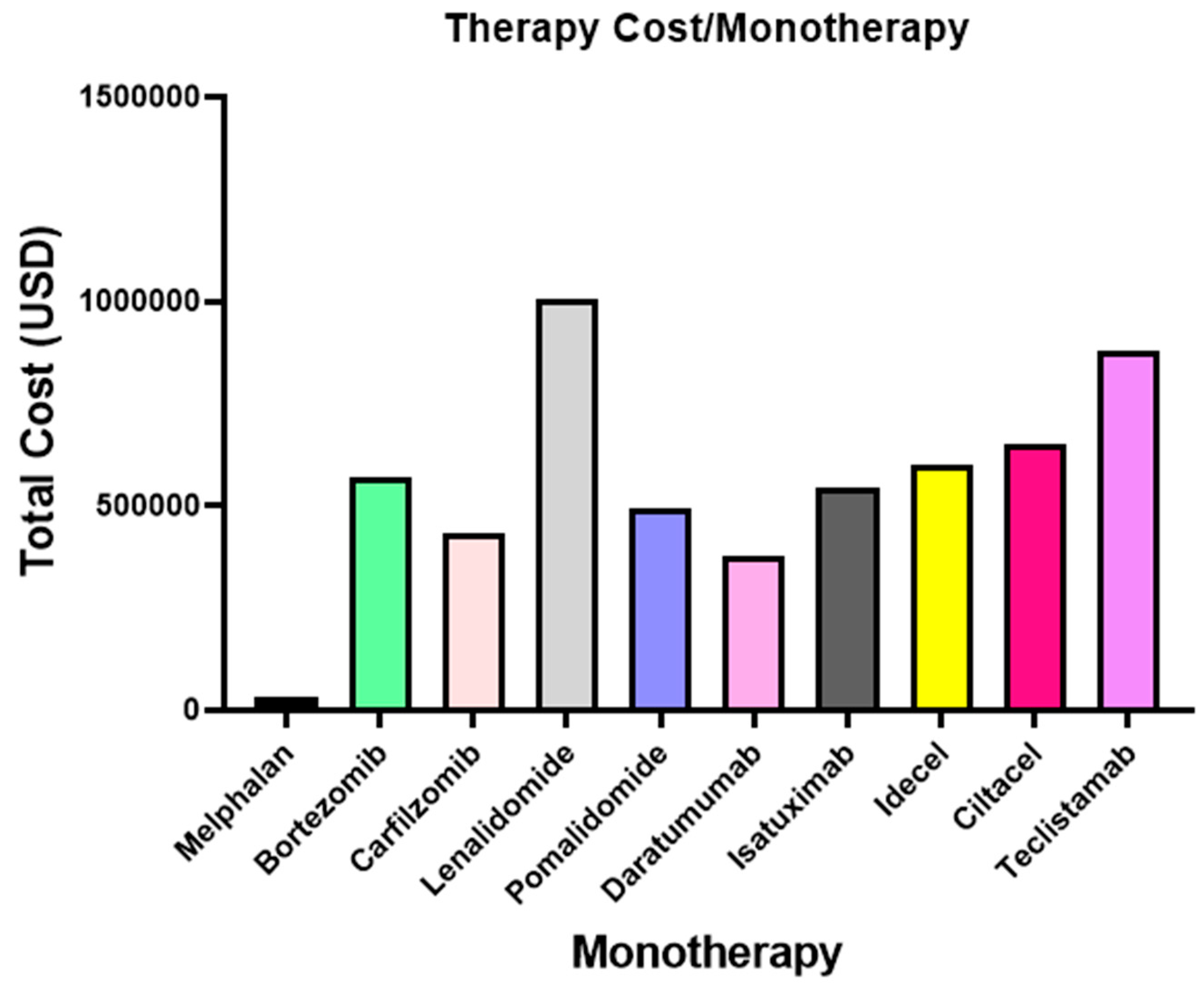
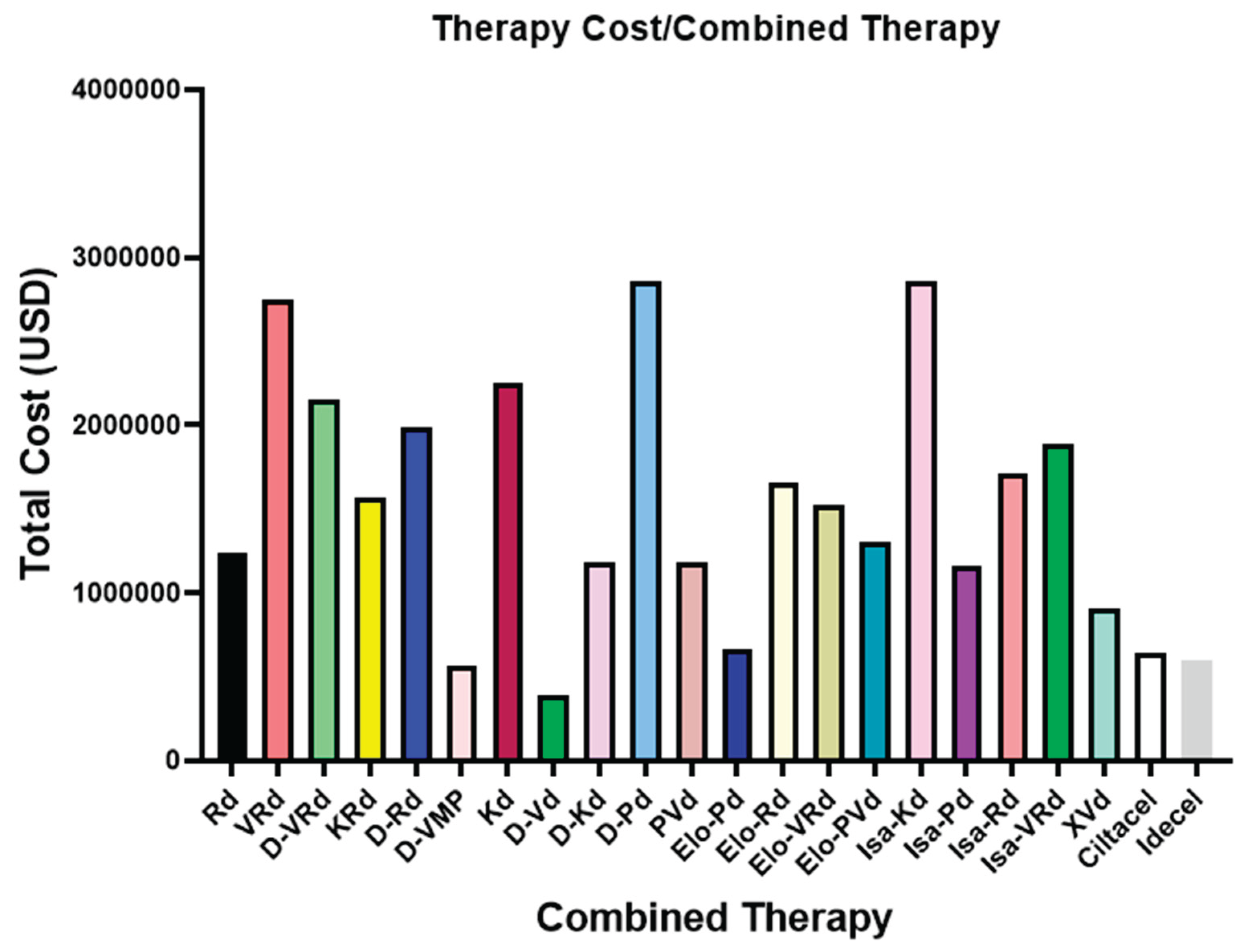
8.1. Therapy Costs
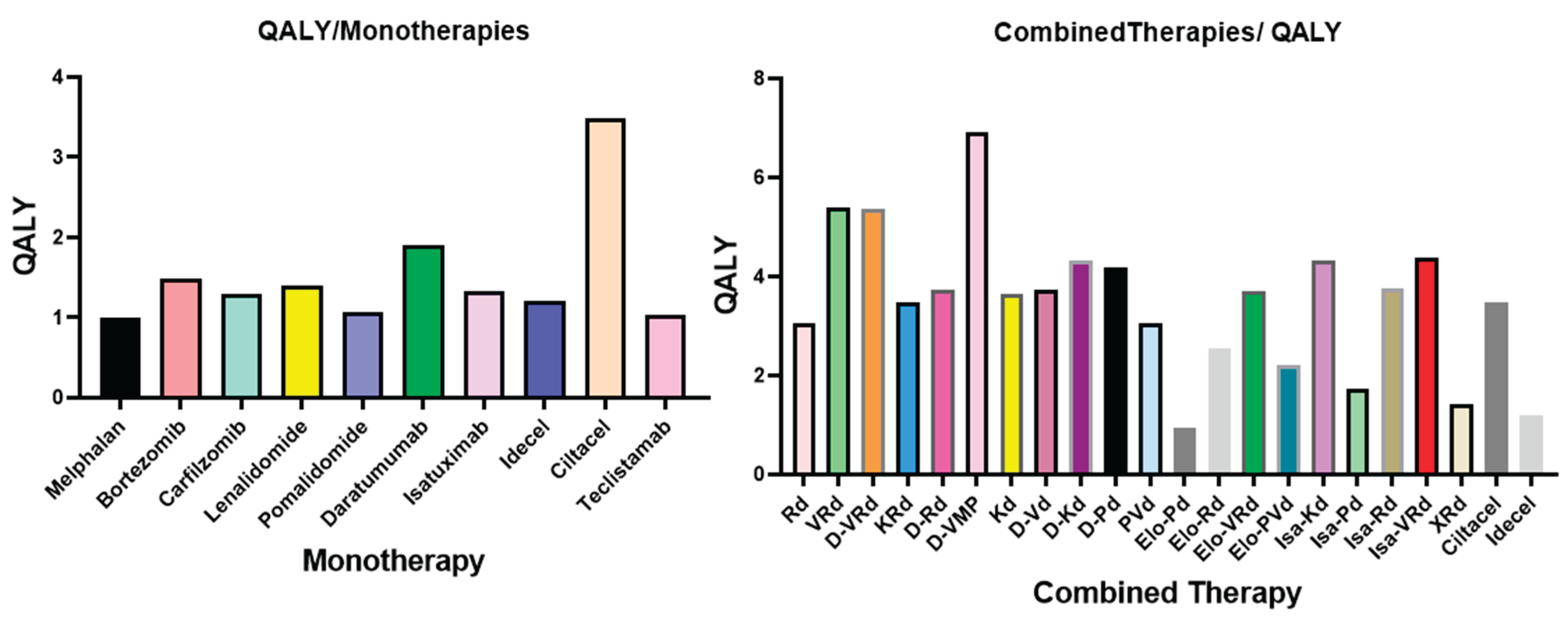
8.2. QALY Outcomes
8.3. ICER Comparisions.
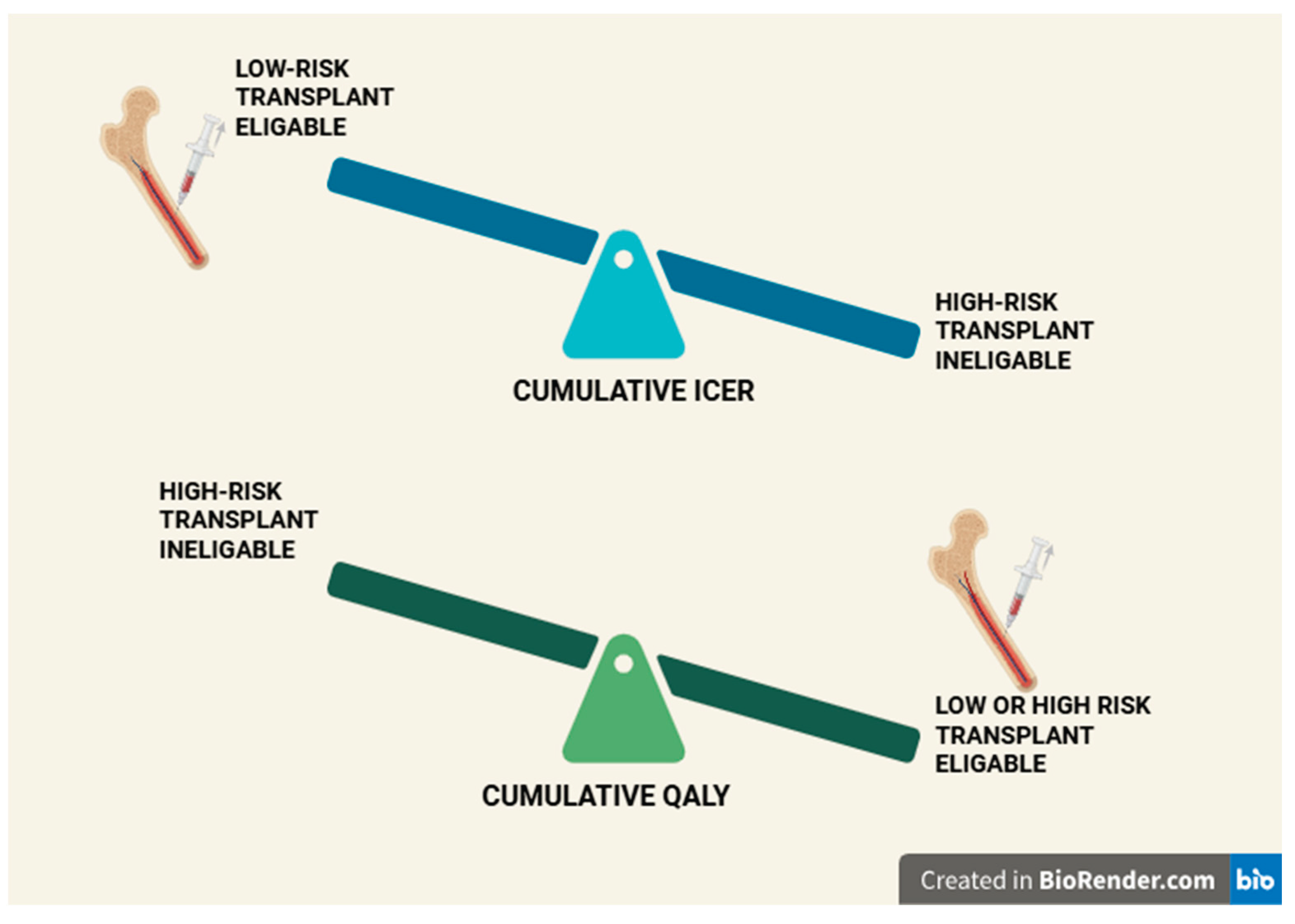
8.4. Aggregated Cost–Effectiveness Outcomes and the See-Saw Model
9. Discussion
10. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | Antibody-Drug Conjugates |
| ALL | Acute Lymphoblastic Leukemia |
| APRIL | A Proliferation-Inducing Ligand |
| ASCT | Autologous Stem Cell Transplantation |
| BAFF | B cell-activating factor belonging to the TNF family |
| BCMA | B-Cell Maturation Antigen |
| BMME | Bone Marrow Microenvironment |
| CAR-T | Chimeric Antigen Receptor T |
| CD | Cluster of Differentiation |
| CLL | Chronic Lymphocytic Leukemia |
| CRAB | (hypercalcaemia, renal impairment, anaemia, bone disease) |
| CRS | Cytokine Release Syndrome |
| CXCR4 | Chemokine Receptor Type 4 |
| ECOG | The Eastern Cooperative Oncology Group |
| FDA | Food and Drug Administration |
| HD | High-Dose |
| HDAC6 | Histone Deacetylase 6 |
| ICAN | Immune Effector Cell-Associated Neurotoxicity |
| ICER | Incremental Cost-Effectiveness Ratios |
| ImiDs: | Immunomodulatory Drugs |
| IL | Interleukin |
| MHC | Major Histocompatibility Complex |
| MM | Multiple Myeloma |
| MRD | Minimal Residual Disease |
| NDMM | Newly Diagnosed Multiple Myeloma |
| ORR | Overall Response Rate |
| PFS | Progression-Free Survival |
| PIs | Proteasome Inhibitors |
| QALYs | Quality-Adjusted Life Years |
| XPO1 | Exportin 1 |
| USD | United States Dollar |
| VGPR | Very Good Partial Response |
| VLA-4 | Integrin α4β1 (very late antigen-4) |
Appendix A
Appendix A.1
| Therapy / Class | Year (Approval / Milestone) | Mechanism / Target | Current Use in MM | Key Toxicities |
|---|---|---|---|---|
| Autologous Stem Cell Transplant (ASCT) [19] | 1983 (MM application) | Hematopoietic rescue | Standard in eligible patients | Myelosuppression, infection risk |
| Bortezomib (first PI) [20,21] | 2003 | Reversible 20S proteasome inhibitor | Backbone of induction (VRd etc.) | Peripheral neuropathy |
| Carfilzomib (2nd-gen PI) [21] | 2012 | Irreversible PI | Relapsed/refractory, combo regimens | Cardiac toxicity |
| Ixazomib (oral PI) [21] | 2015 | Oral PI | Maintenance, frail patients | GI, mild cytopenias |
| Thalidomide (IMiD) reintroduced [11] | 1997 (MM) | Immunomodulation, anti-angiogenesis | Limited due to toxicity | Neuropathy, sedation |
| Lenalidomide (IMiD) [22] | 2005 | IMiD, cytokine modulation, T-cell activation | Frontline & maintenance backbone | Cytopenias, thrombosis |
| Pomalidomide (IMiD) [23] | 2013 | Next-gen IMiD | Relapsed/refractory settings | Cytopenias, infections |
| Daratumumab (anti-CD38 mAb) [1,6] | 2015 | ADCC, CDC, ADCP, direct apoptosis | Widely used frontline & RRMM | Neutropenia, infusion reactions |
| Isatuximab (anti-CD38 mAb) [1,6] | 2020 | Distinct CD38 epitope, direct apoptosis | Combo with Pd in RRMM | Neutropenia, infections |
| Selinexor (XPO1 inhibitor) [1,6] | 2019 | Nuclear export inhibition, p53 reactivation | Triple-class refractory | GI toxicity, cytopenias |
| Venetoclax (BCL-2 inhibitor) [1,6] | Investigation | BCL-2 inhibition | Targeted subgroup (t(11;14)) | Tumor lysis, cytopenias |
| Melflufen (peptide-drug conjugate) [1,6] | 2021 (revoked FDA approval) | Alkylating payload via peptide conjugate | EMA-approved, not FDA | Cytopenias, survival concern |
| HDAC inhibitors (e.g. panobinostat) [1,6] | 2015 (panobinostat FDA) | Histone deacetylase inhibition | Adjunct in refractory disease | GI, fatigue, cytopenias |
| Checkpoint inhibitors (PD-1/PD-L1) [1,6] | Trials (halted in MM) | Immune checkpoint blockade | Investigational only | Immune toxicities |
| Bispecific Abs / CAR-T / NK [1,6] | 2020s | Redirected T/NK cytotoxicity | Rapidly emerging | CRS, neurotoxicity, cytopenias |
Appendix B
References
- Rajkumar SV. Multiple myeloma: 2024 update on diagnosis, risk-stratification, and management. Am J Hematol. 2024 Sep;99(9):1802-1824. doi: 10.1002/ajh.27422. Epub 2024 Jun 28. PMID: 38943315; PMCID: PMC11404783. [CrossRef]
- Binder AF, Walker CJ, Mark TM, Baljevic M. Impacting T-cell fitness in multiple myeloma: potential roles for selinexor and XPO1 inhibitors. Front Immunol. 2023 Oct 26;14:1275329. doi: 10.3389/fimmu.2023.1275329. PMID: 37954586; PMCID: PMC10637355. [CrossRef]
- Kim HY, Moon JY, Ryu H, Choi YS, Song IC, Lee HJ, Yun HJ, Kim S, Jo DY. Bortezomib inhibits the survival and proliferation of bone marrow stromal cells. Blood Res. 2015 Jun;50(2):87-96. doi: 10.5045/br.2015.50.2.87. Epub 2015 Jun 25. PMID: 26157778; PMCID: PMC4486164.
- Krejcik J, Casneuf T, Nijhof IS, Verbist B, Bald J, Plesner T, Syed K, Liu K, van de Donk NW, Weiss BM, Ahmadi T, Lokhorst HM, Mutis T, Sasser AK. Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion, and skews T-cell repertoire in multiple myeloma. Blood. 2016 Jul 21;128(3):384-94. doi: 10.1182/blood-2015-12-687749. Epub 2016 May 24. PMID: 27222480; PMCID: PMC4957162.
- Solly S. Remarks on the pathology of mollities ossium; with cases. Med Chir Trans. 1844;27:435-498.8. doi: 10.1177/095952874402700129. PMID: 20895811; PMCID: PMC2116942.
- El Khatib HH, Abdulla K, Nassar LK, Ellabban MG, Kakarougkas A. Advancements in multiple myeloma therapies: a comprehensive review by disease stage. Lymphatics. 2025;3(1):2. [CrossRef]
- Van Way C 3rd. War and Trauma: A History of Military Medicine - Part II. Mo Med. 2016 Sep-Oct;113(5):336-340. PMID: 30228491; PMCID: PMC6139825.
- Smith SL. War! What is it good for? Mustard gas medicine. CMAJ. 2017 Feb 27;189(8):E321-E322. doi: 10.1503/cmaj.161032. PMID: 28246228; PMCID: PMC5325736.
- Gahrton G, Tura S, Ljungman P, Belanger C, Brandt L, Cavo M, et al. Allogeneic bone marrow transplantation in multiple myeloma. European Group for Bone Marrow Transplantation. N Engl J Med. 1991 Oct 31;325(18):1267-73. doi: 10.1056/NEJM199110313251802. PMID: 1922221.
- ALWALL N. Urethane and stilbamidine in multiple myeloma report on two cases. Lancet. 1947 Sep 13;2(6472):388. doi: 10.1016/s0140-6736(47)90375-9. PMID: 20263550.
- Beninger P. Thalidomide: Following tragedy, a repurposed molecule with continuing opportunities for clinical benefit. Clin Ther. 2025;47(4):249–51. [CrossRef]
- Goodman LS, Wintrobe MM, Dameshek W, Goodman MJ, Gilman A, McLennan MT. Landmark article Sept. 21, 1946: Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl) amine hydrochloride and tris(beta-chloroethyl) amine hydrochloride for Hodgkin's disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. By Louis S. Goodman, Maxwell M. Wintrobe, William Dameshek, Morton J. Goodman, Alfred Gilman and Margaret T. McLennan. JAMA. 1984 May 4;251(17):2255-61. doi: 10.1001/jama.251.17.2255. PMID: 6368885.
- Fu W, Bang SM, Huang H, Kim K, Li W, An G, et.al. Bortezomib, Melphalan, and Prednisone With or Without Daratumumab in Transplant-ineligible Asian Patients With Newly Diagnosed Multiple Myeloma: The Phase 3 OCTANS Study. Clin Lymphoma Myeloma Leuk. 2023 Jun;23(6):446-455.e4. doi: 10.1016/j.clml.2023.02.009. Epub 2023 Mar 4. PMID: 37024420.
- Herzog H, Oliveto EP. A history of significant steroid discoveries and developments originating at the Schering Corporation (USA) since 1948. Steroids. 1992;57(12):617–23. [CrossRef]
- Case DC Jr, Lee DJ 3rd, Clarkson BD. Improved survival times in multiple myeloma treated with melphalan, prednisone, cyclophosphamide, vincristine and BCNU: M-2 protocol. Am J Med. 1977 Dec;63(6):897-903. doi: 10.1016/0002-9343(77)90543-5. PMID: 605911.
- Vazquez S, Gold J, Spirollari E, Akmal S, Hanft SJ. The story of dexamethasone and how it became one of the most widely used drugs in neurosurgery. J Neurosurg. 2023 Nov 24;140(4):1191-1197. doi: 10.3171/2023.9.JNS231099. PMID: 38000066.
- Usmani SZ, Hoering A, Ailawadhi S, Sexton R, Lipe B, Hita SF, et.al; SWOG1211 Trial Investigators. Bortezomib, lenalidomide, and dexamethasone with or without elotuzumab in patients with untreated, high-risk multiple myeloma (SWOG-1211): primary analysis of a randomised, phase 2 trial. Lancet Haematol. 2021 Jan;8(1):e45-e54. doi: 10.1016/S2352-3026(20)30354-9. Epub 2020 Dec 22. PMID: 33357482; PMCID: PMC8601389.
- Thorn CF, Oshiro C, Marsh S, Hernandez-Boussard T, McLeod H, Klein TE, Altman RB. Doxorubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genomics. 2011 Jul;21(7):440-6. doi: 10.1097/FPC.0b013e32833ffb56. PMID: 21048526; PMCID: PMC3116111.
- Morè S, Corvatta L, Manieri VM, Saraceni F, Scortechini I, Mancini G, Fiorentini A, Olivieri A, Offidani M. Autologous Stem Cell Transplantation in Multiple Myeloma: Where Are We and Where Do We Want to Go? Cells. 2022 Feb 10;11(4):606. doi: 10.3390/cells11040606. PMID: 35203257; PMCID: PMC8870632.
- Sogbein O, Paul P, Umar M, Chaari A, Batuman V, Upadhyay R. Bortezomib in cancer therapy: Mechanisms, side effects, and future proteasome inhibitors. Life Sci. 2024 Dec 1;358:123125. doi: 10.1016/j.lfs.2024.123125. Epub 2024 Oct 15. PMID: 39413903.
- Fricker LD. Proteasome Inhibitor Drugs. Annu Rev Pharmacol Toxicol. 2020 Jan 6;60:457-476. doi: 10.1146/annurev-pharmtox-010919-023603. Epub 2019 Sep 3. PMID: 31479618.
- Zhang CW, Wang YN, Ge XL. Lenalidomide use in multiple myeloma (Review). Mol Clin Oncol. 2023 Nov 28;20(1):7. doi: 10.3892/mco.2023.2705. PMID: 38125742; PMCID: PMC10729307.
- Hoy SM. Pomalidomide: A Review in Relapsed and Refractory Multiple Myeloma. Drugs. 2017 Nov;77(17):1897-1908. doi: 10.1007/s40265-017-0833-y. PMID: 29110190.
- Kamrani S, Naseramini R, Khani P, Razavi ZS, Afkhami H, Atashzar MR, Nasri F, Alavimanesh S, Saeidi F, Ronaghi H. Mesenchymal stromal cells in bone marrow niche of patients with multiple myeloma: a double-edged sword. Cancer Cell Int. 2025 Mar 26;25(1):117. doi: 10.1186/s12935-025-03741-x. PMID: 40140850; PMCID: PMC11948648.
- Lopes R, Caetano J, Ferreira B, Barahona F, Carneiro EA, João C. The Immune Microenvironment in Multiple Myeloma: Friend or Foe? Cancers (Basel). 2021 Feb 5;13(4):625. doi: 10.3390/cancers13040625. PMID: 33562441; PMCID: PMC7914424.
- Ghoshal D, Petersen I, Ringquist R, Kramer L, Bhatia E, Hu T, Richard A, Park R, Corbin J, Agarwal S, Thomas A, Ramirez S, Tharayil J, Downey E, Ketchum F, Ochal A, Sonthi N, Lonial S, Kochenderfer JN, Tran R, Zhu M, Lam WA, Coskun AF, Roy K. Multi-Niche Human Bone Marrow On-A-Chip for Studying the Interactions of Adoptive CAR-T Cell Therapies with Multiple Myeloma. bioRxiv [Preprint]. 2024 Apr 12:2024.04.08.588601. doi: 10.1101/2024.04.08.588601. Update in: Biomaterials. 2025 May;316:123016. doi: 10.1016/j.biomaterials.2024.123016. PMID: 38644993; PMCID: PMC11030357.
- Hathi D, Chanswangphuwana C, Cho N, Fontana F, Maji D, Ritchey J, O'Neal J, Ghai A, Duncan K, Akers WJ, Fiala M, Vij R, DiPersio JF, Rettig M, Shokeen M. Ablation of VLA4 in multiple myeloma cells redirects tumor spread and prolongs survival. Sci Rep. 2022 Jan 7;12(1):30. doi: 10.1038/s41598-021-03748-0. PMID: 34996933; PMCID: PMC8741970.
- Fröbel J, Landspersky T, Percin G, Schreck C, Rahmig S, Ori A, et.al. The Hematopoietic Bone Marrow Niche Ecosystem. Front Cell Dev Biol. 2021 Jul 22;9:705410. doi: 10.3389/fcell.2021.705410. PMID: 34368155; PMCID: PMC8339972.
- Menu E, Vanderkerken K. Exosomes in multiple myeloma: from bench to bedside. Blood. 2022 Dec 8;140(23):2429-2442. doi: 10.1182/blood.2021014749. PMID: 35271699; PMCID: PMC10653045.
- Nooka AK, Cohen AD, Lee HC, Badros A, Suvannasankha A, Callander N, Abdallah AO, Trudel S, Chari A, Libby EN, Chaudhry M, Hultcrantz M, Kortüm KM, Popat R, Sborov D, Hakim S, Lewis E, Gorsh B, Bhushan B, McKeown A, Gupta I, Opalinska J, Richardson PG, Lonial S. Single-agent belantamab mafodotin in patients with relapsed/refractory multiple myeloma: Final analysis of the DREAMM-2 trial. Cancer. 2023 Dec 1;129(23):3746-3760. doi: 10.1002/cncr.34987. Epub 2023 Aug 25. PMID: 37622738; PMCID: PMC11055177.
- Beksaç M, Sahli E, Rabea A, Saydam G, Tombak A, Alhuraiji A, Mutahar EY, Hosny M, Alhejazi A, Mattar M, Dal MS, Marashi M, Palamar M. Practical Guidance on the Clinical Management of Belantamab Mafodotin for Patients With Relapsed/Refractory Multiple Myeloma: Recommendations From the Middle East and North Africa Expert Panel. Clin Lymphoma Myeloma Leuk. 2025 May 20:S2152-2650(25)00184-3. doi: 10.1016/j.clml.2025.05.019. Epub ahead of print. PMID: 40579284.
- McCarthy EF. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop J. 2006;26:154-8. PMID: 16789469; PMCID: PMC1888599.
- Eshhar Z, Waks T, Gross G, Schindler DG. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci U S A. 1993 Jan 15;90(2):720-4. doi: 10.1073/pnas.90.2.720. PMID: 8421711; PMCID: PMC45737.
- Maher J, Brentjens RJ, Gunset G, Rivière I, Sadelain M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotechnol. 2002 Jan;20(1):70-5. doi: 10.1038/nbt0102-70. PMID: 11753365.
- Brentjens RJ, Latouche JB, Santos E, Marti F, Gong MC, Lyddane C, et.al. Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat Med. 2003 Mar;9(3):279-86. doi: 10.1038/nm827. Epub 2003 Feb 10. PMID: 12579196.
- Kalos M, Levine BL, Porter DL, Katz S, Grupp SA, Bagg A, June CH. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci Transl Med. 2011 Aug 10;3(95):95ra73. doi: 10.1126/scitranslmed.3002842. PMID: 21832238; PMCID: PMC3393096.
- Kochenderfer JN, Wilson WH, Janik JE, Dudley ME, Stetler-Stevenson M, Feldman SA., et.al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood. 2010 Nov 18;116(20):4099-102. doi: 10.1182/blood-2010-04-281931. Epub 2010 Jul 28. PMID: 20668228; PMCID: PMC2993617.
- Ruella M, Maus MV. Catch me if you can: Leukemia Escape after CD19-Directed T Cell Immunotherapies. Comput Struct Biotechnol J. 2016 Sep 28;14:357-362. doi: 10.1016/j.csbj.2016.09.003. PMID: 27761200; PMCID: PMC5061074.
- Sheykhhasan M, Ahmadieh-Yazdi A, Vicidomini R, Poondla N, Tanzadehpanah H, Dirbaziyan A, et.al. CAR T therapies in multiple myeloma: unleashing the future. Cancer Gene Ther. 2024 May;31(5):667-686. doi: 10.1038/s41417-024-00750-2. Epub 2024 Mar 4. PMID: 38438559; PMCID: PMC11101341.
- Rodriguez-Otero P, Ailawadhi S, Arnulf B, Patel K, Cavo M, Nooka AK, et.al. Ide-cel or Standard Regimens in Relapsed and Refractory Multiple Myeloma. N Engl J Med. 2023 Mar 16;388(11):1002-1014. doi: 10.1056/NEJMoa2213614. Epub 2023 Feb 10. PMID: 36762851.
- Goel U, Zanwar S, Cowan AJ, Banerjee R, Khouri J, Dima D. Ciltacabtagene Autoleucel for the Treatment of Relapsed/Refractory Multiple Myeloma: Efficacy, Safety, and Place in Therapy. Cancer Manag Res. 2025 Feb 19;17:357-372. doi: 10.2147/CMAR.S510408. PMID: 39990276; PMCID: PMC11847446.
- Rasche L, Hudecek M, Einsele H. CAR T-cell therapy in multiple myeloma: mission accomplished? Blood. 2024 Jan 25;143(4):305-310. doi: 10.1182/blood.2023021221. PMID: 38033289.
- Zheng H, Xian H, Zhang W, Lu C, Pan R, Liu H, Xu Z. BCMA-targeted therapies for multiple myeloma: latest updates from 2024 ASH annual meeting. J Hematol Oncol. 2025 Mar 1;18(1):23. doi: 10.1186/s13045-025-01675-5. PMID: 40025529; PMCID: PMC11872297.
- Zugasti I, Espinosa-Aroca L, Fidyt K, Mulens-Arias V, Diaz-Beya M, Juan M, Urbano-Ispizua Á, Esteve J, Velasco-Hernandez T, Menéndez P. CAR-T cell therapy for cancer: current challenges and future directions. Signal Transduct Target Ther. 2025 Jul 4;10(1):210. doi: 10.1038/s41392-025-02269-w. PMID: 40610404; PMCID: PMC12229403.
- Martin T, Dimopoulos MA, Mikhael J, Yong K, Capra M, Facon T, et.al. Isatuximab, carfilzomib, and dexamethasone in patients with relapsed multiple myeloma: updated results from IKEMA, a randomized Phase 3 study. Blood Cancer J. 2023 May 9;13(1):72. doi: 10.1038/s41408-023-00797-8. Erratum in: Blood Cancer J. 2023 Sep 27;13(1):152. doi: 10.1038/s41408-023-00923-6. PMID: 37156782; PMCID: PMC10166682.
- Bahlis NJ, Samaras C, Reece D, Sebag M, Matous J, Berdeja JG., et al. Pomalidomide/Daratumumab/Dexamethasone in Relapsed or Refractory Multiple Myeloma: Final Overall Survival From MM-014. Clin Lymphoma Myeloma Leuk. 2024 Dec;24(12):852-862. doi: 10.1016/j.clml.2024.07.014. Epub 2024 Aug 8. PMID: 39237427.
- Ocio EM, Perrot A, Bories P, San-Miguel JF, Blau IW, Karlin L, et.al. Efficacy and safety of isatuximab plus bortezomib, lenalidomide, and dexamethasone in patients with newly diagnosed multiple myeloma ineligible/with no immediate intent for autologous stem cell transplantation. Leukemia. 2023 Jul;37(7):1521-1529. doi: 10.1038/s41375-023-01936-7. Epub 2023 Jun 14. PMID: 37316728; PMCID: PMC10264885.
- Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et.al. Final analysis of carfilzomib, dexamethasone, and daratumumab vs carfilzomib and dexamethasone in the CANDOR study. Blood Adv. 2023 Jul 25;7(14):3739-3748. doi: 10.1182/bloodadvances.2023010026. PMID: 37163358; PMCID: PMC10368773.
- Briggs AH, Sculpher MJ, Claxton K. Decision modelling for health economic evaluation. Oxford: Oxford University Press; 2006.
- Latimer NR. Survival analysis for economic evaluations alongside clinical trials--extrapolation with patient-level data: inconsistencies, limitations, and a practical guide. Med Decis Making. 2013 Aug;33(6):743-54. doi: 10.1177/0272989X12472398. Epub 2013 Jan 22. PMID: 23341049.
- Alexanian R, Bergsagel DE, Migliore PJ, Vaughn WK, Howe CD. Melphalan therapy for plasma cell myeloma. Blood. 1968 Jan;31(1):1-10. PMID: 5638562.
- Kyle RA, Rajkumar SV. Multiple myeloma. Blood. 2008;111(6):2962–72. [CrossRef]
- Orlowski RZ, Nagler A, Sonneveld P, Bladé J, Hajek R, Spencer A, et.al. Final overall survival results of a randomized trial comparing bortezomib plus pegylated liposomal doxorubicin with bortezomib alone in patients with relapsed or refractory multiple myeloma. Cancer. 2016 Jul 1;122(13):2050-6. doi: 10.1002/cncr.30026. Epub 2016 May 18. PMID: 27191689; PMCID: PMC5701574.
- Iida S, Watanabe T, Matsumoto M, Suzuki K, Sunami K, Ishida T., et.al. Carfilzomib monotherapy in Japanese patients with relapsed or refractory multiple myeloma: A phase 1/2 study. Cancer Sci. 2019 Sep;110(9):2924-2932. doi: 10.1111/cas.14139. Epub 2019 Aug 10. PMID: 31336012; PMCID: PMC6726678.
- Moreau P, Mateos MV, Berenson JR, Weisel K, Lazzaro A, Song K., et.al. Once weekly versus twice weekly carfilzomib dosing in patients with relapsed and refractory multiple myeloma (A.R.R.O.W.): interim analysis results of a randomised, phase 3 study. Lancet Oncol. 2018 Jul;19(7):953-964. doi: 10.1016/S1470-2045(18)30354-1. Epub 2018 Jun 1. Erratum in: Lancet Oncol. 2018 Aug;19(8):e382. doi: 10.1016/S1470-2045(18)30492-3. PMID: 29866475.
- Matsela LM, Cleary S, Wilkinson T. Cost utility and budget impact analysis of dexamethasone compared with bortezomib and lenalidomide for the treatment of second line multiple myeloma from a South African public health perspective. Cost Eff Resour Alloc. 2022 Dec 12;20(1):69. doi: 10.1186/s12962-022-00399-4. PMID: 36510230; PMCID: PMC9746097.
- Kropff M, Baylon HG, Hillengass J, Robak T, Hajek R, Liebisch P., et.al. Thalidomide versus dexamethasone for the treatment of relapsed and/or refractory multiple myeloma: results from OPTIMUM, a randomized trial. Haematologica. 2012 May;97(5):784-91. doi: 10.3324/haematol.2011.044271. Epub 2011 Dec 1. PMID: 22133776; PMCID: PMC3342984.
- Olry de Labry Lima A, Gimeno-Ballester V, Ríos Tamayo R, Epstein D, Matas Hoces A, Ríos Sánchez E, García Mochón L, Alegre-Del Rey EJ. Cost-effectiveness of lenalidomide maintenance in patients with multiple myeloma who have undergone autologous transplant of hematopoietic progenitor cells. Bone Marrow Transplant. 2019 Nov;54(11):1908-1919. doi: 10.1038/s41409-019-0574-5. Epub 2019 May 31. PMID: 31150015.
- Richardson PG, Siegel DS, Vij R, Hofmeister CC, Baz R, Jagannath S., et.al. Pomalidomide alone or in combination with low-dose dexamethasone in relapsed and refractory multiple myeloma: a randomized phase 2 study. Blood. 2014 Mar 20;123(12):1826-32. doi: 10.1182/blood-2013-11-538835. Epub 2014 Jan 13. Erratum in: Blood. 2014 May 15;123(20):3208-9. PMID: 24421329; PMCID: PMC3962162.
- Usmani SZ, Weiss BM, Plesner T, Bahlis NJ, Belch A, Lonial S., et.al. Clinical efficacy of daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma. Blood. 2016 Jul 7;128(1):37-44. doi: 10.1182/blood-2016-03-705210. Epub 2016 May 23. PMID: 27216216; PMCID: PMC4937359.
- Dimopoulos M, Bringhen S, Anttila P, Capra M, Cavo M, Cole C., et.al. Isatuximab as monotherapy and combined with dexamethasone in patients with relapsed/refractory multiple myeloma. Blood. 2021 Mar 4;137(9):1154-1165. doi: 10.1182/blood.2020008209. PMID: 33080623; PMCID: PMC7933767.
- Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ.,et.al. Overall Survival With Daratumumab, Lenalidomide, and Dexamethasone in Previously Treated Multiple Myeloma (POLLUX): A Randomized, Open-Label, Phase III Trial. J Clin Oncol. 2023 Mar 10;41(8):1590-1599. doi: 10.1200/JCO.22.00940. Epub 2023 Jan 4. PMID: 36599114; PMCID: PMC10022849.
- Durie BGM, Hoering A, Sexton R, Abidi MH, Epstein J, Rajkumar SV., et.al. Longer term follow-up of the randomized phase III trial SWOG S0777: bortezomib, lenalidomide and dexamethasone vs. lenalidomide and dexamethasone in patients (Pts) with previously untreated multiple myeloma without an intent for immediate autologous stem cell transplant (ASCT). Blood Cancer J. 2020 May 11;10(5):53. doi: 10.1038/s41408-020-0311-8. PMID: 32393732; PMCID: PMC7214419.
- Yamamoto C, Minakata D, Koyama S, Sekiguchi K, Fukui Y, Murahashi R., et.al. Daratumumab in first-line therapy is cost-effective in transplant-eligible patients with newly diagnosed myeloma. Blood. 2022 Aug 11;140(6):594-607. doi: 10.1182/blood.2021015220. PMID: 35580269; PMCID: PMC9373013.
- Usmani SZ, Facon T, Hungria V, Bahlis NJ, Venner CP, Braunstein M., et al. Daratumumab plus bortezomib, lenalidomide and dexamethasone for transplant-ineligible or transplant-deferred newly diagnosed multiple myeloma: the randomized phase 3 CEPHEUS trial. Nat Med. 2025 Apr;31(4):1195-1202. doi: 10.1038/s41591-024-03485-7. Epub 2025 Feb 5. Erratum in: Nat Med. 2025 Apr;31(4):1366. doi: 10.1038/s41591-025-03581-2. PMID: 39910273; PMCID: PMC12003169.
- Maziarz RT, Devine S, Garrison LP, Agodoa I, Badaracco J, Gitlin M, Perales MA. Estimating the Lifetime Medical Cost Burden of an Allogeneic Hematopoietic Cell Transplantation Patient. Transplant Cell Ther. 2023 Oct;29(10):637.e1-637.e9. doi: 10.1016/j.jtct.2023.06.013. Epub 2023 Jun 24. PMID: 37364775; PMCID: PMC11035010.
- Lee JH, Choi J, Min CK, Park SS, Jo JC, Lee YJ., et.al. Superior outcomes and high-risk features with carfilzomib, lenalidomide, and dexamethasone combination therapy for patients with relapsed and refractory multiple myeloma: results of the multicenter KMMWP2201 study. Haematologica. 2024 Nov 1;109(11):3681-3692. doi: 10.3324/haematol.2024.285534. PMID: 38841794; PMCID: PMC11532698.
- Stewart AK, Rajkumar SV, Dimopoulos MA, Masszi T, Špička I, Oriol A, et.al.; ASPIRE Investigators. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015 Jan 8;372(2):142-52. doi: 10.1056/NEJMoa1411321. Epub 2014 Dec 6. PMID: 25482145.
- Benboubker L, Dimopoulos MA, Dispenzieri A, Catalano J, Belch AR, Cavo M., et.al.; FIRST Trial Team. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. N Engl J Med. 2014 Sep 4;371(10):906-17. doi: 10.1056/NEJMoa1402551. PMID: 25184863.
- Fu W, Bang SM, Huang H, Kim K, Li W, An G, et.al. Bortezomib, Melphalan, and Prednisone With or Without Daratumumab in Transplant-ineligible Asian Patients With Newly Diagnosed Multiple Myeloma: The Phase 3 OCTANS Study. Clin Lymphoma Myeloma Leuk. 2023 Jun;23(6):446-455.e4. doi: 10.1016/j.clml.2023.02.009. Epub 2023 Mar 4. PMID: 37024420.
- Mateos MV, Dimopoulos MA, Cavo M, Suzuki K, Jakubowiak A, Knop S, et.al.; ALCYONE Trial Investigators. Daratumumab plus Bortezomib, Melphalan, and Prednisone for Untreated Myeloma. N Engl J Med. 2018 Feb 8;378(6):518-528. doi: 10.1056/NEJMoa1714678. Epub 2017 Dec 12. PMID: 29231133.
- Mateos MV, Cavo M, Blade J, Dimopoulos MA, Suzuki K, Jakubowiak A., et.al. Overall survival with daratumumab, bortezomib, melphalan, and prednisone in newly diagnosed multiple myeloma (ALCYONE): a randomised, open-label, phase 3 trial. Lancet. 2020 Jan 11;395(10218):132-141. doi: 10.1016/S0140-6736(19)32956-3. Epub 2019 Dec 10. PMID: 31836199.
- Mateos MV, Dimopoulos MA, Gay F, San-Miguel J, Dytfeld D, Spencer A, et al. Iberdomide plus dexamethasone in relapsed or refractory multiple myeloma (CC-220-MM-001): a multicentre, multicohort, open-label, phase 1/2 trial. Lancet Oncol. 2025;26(5):423–35. [CrossRef]
- Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et.al. Final analysis of carfilzomib, dexamethasone, and daratumumab vs carfilzomib and dexamethasone in the CANDOR study. Blood Adv. 2023 Jul 25;7(14):3739-3748. doi: 10.1182/bloodadvances.2023010026. PMID: 37163358; PMCID: PMC10368773.
- Martin T, Dimopoulos MA, Mikhael J, Yong K, Capra M, Facon T, et.al. Isatuximab, carfilzomib, and dexamethasone in patients with relapsed multiple myeloma: updated results from IKEMA, a randomized Phase 3 study. Blood Cancer J. 2023 May 9;13(1):72. doi: 10.1038/s41408-023-00797-8. Erratum in: Blood Cancer J. 2023 Sep 27;13(1):152. doi: 10.1038/s41408-023-00923-6. PMID: 37156782; PMCID: PMC10166682.
- Richardson PG, Perrot A, Miguel JS, Beksac M, Spicka I, Leleu X et.al. Isatuximab-pomalidomide-dexamethasone versus pomalidomide-dexamethasone in patients with relapsed and refractory multiple myeloma: final overall survival analysis. Haematologica. 2024 Jul 1;109(7):2239-2249. doi: 10.3324/haematol.2023.284325. PMID: 38299578; PMCID: PMC11215383.
- Leleu X, Hulin C, Lambert J, Bobin A, Perrot A, Karlin L et.al. Isatuximab, lenalidomide, dexamethasone and bortezomib in transplant-ineligible multiple myeloma: the randomized phase 3 BENEFIT trial. Nat Med. 2024 Aug;30(8):2235-2241. doi: 10.1038/s41591-024-03050-2. Epub 2024 Jun 3. PMID: 38830994; PMCID: PMC11333283.
- Mateos MV, Engelhardt M, Leleu X, Mesa MG, Cavo M, Dimopoulos M, et.al. Impact of prior treatment on selinexor, bortezomib, dexamethasone outcomes in patients with relapsed/refractory multiple myeloma: Extended follow-up subgroup analysis of the BOSTON trial. Eur J Haematol. 2024 Aug;113(2):242-252. doi: 10.1111/ejh.14223. Epub 2024 May 1. PMID: 38693052.
- Moreau P, Garfall AL, van de Donk NWCJ, Nahi H, San-Miguel JF, Oriol A, et.al. Teclistamab in Relapsed or Refractory Multiple Myeloma. N Engl J Med. 2022 Aug 11;387(6):495-505. doi: 10.1056/NEJMoa2203478. Epub 2022 Jun 5. PMID: 35661166; PMCID: PMC10587778.
- Mateos MV, Leleu X, Yimer HA, Song KW, Nooka AK, Goldschmidt H, et al. Talquetamab versus belantamab mafodotin in patients with relapsed/refractory multiple myeloma: an interim analysis of the phase 3 MonumenTAL-3 trial. Lancet Haematol. 2024;11(6):e230–44. [CrossRef]
- Bahlis NJ, Samaras C, Reece D, Sebag M, Matous J, Berdeja JG., et al. Pomalidomide/Daratumumab/Dexamethasone in Relapsed or Refractory Multiple Myeloma: Final Overall Survival From MM-014. Clin Lymphoma Myeloma Leuk. 2024 Dec;24(12):852-862. doi: 10.1016/j.clml.2024.07.014. Epub 2024 Aug 8. PMID: 39237427.
- Dimopoulos M, Weisel K, Moreau P, Anderson LD Jr, White D, San-Miguel J., et.al. Pomalidomide, bortezomib, and dexamethasone for multiple myeloma previously treated with lenalidomide (OPTIMISMM): outcomes by prior treatment at first relapse. Leukemia. 2021 Jun;35(6):1722-1731. doi: 10.1038/s41375-020-01021-3. Epub 2020 Sep 7. PMID: 32895455; PMCID: PMC8179841.
- Richardson PG, Oriol A, Beksac M, Liberati AM, Galli M, Schjesvold F, et.al; OPTIMISMM trial investigators. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed or refractory multiple myeloma previously treated with lenalidomide (OPTIMISMM): a randomised, open-label, phase 3 trial. Lancet Oncol. 2019 Jun;20(6):781-794. doi: 10.1016/S1470-2045(19)30152-4. Epub 2019 May 13. PMID: 31097405.
- Paludo J, Mikhael JR, LaPlant BR, Halvorson AE, Kumar S, Gertz MA, et.al. Pomalidomide, bortezomib, and dexamethasone for patients with relapsed lenalidomide-refractory multiple myeloma. Blood. 2017 Sep 7;130(10):1198-1204. doi: 10.1182/blood-2017-05-782961. Epub 2017 Jul 6. PMID: 28684537; PMCID: PMC5606008.
- Dimopoulos MA, Dytfeld D, Grosicki S, Moreau P, Takezako N, Hori M, et.al. Elotuzumab Plus Pomalidomide and Dexamethasone for Relapsed/Refractory Multiple Myeloma: Final Overall Survival Analysis From the Randomized Phase II ELOQUENT-3 Trial. J Clin Oncol. 2023 Jan 20;41(3):568-578. doi: 10.1200/JCO.21.02815. Epub 2022 Aug 12. PMID: 35960908; PMCID: PMC9870233.
- Gentile M, Specchia G, Derudas D, Galli M, Botta C, Rocco S, et.al. Elotuzumab, lenalidomide, and dexamethasone as salvage therapy for patients with multiple myeloma: Italian, multicenter, retrospective clinical experience with 300 cases outside of controlled clinical trials. Haematologica. 2021 Jan 1;106(1):291-294. doi: 10.3324/haematol.2019.241513. PMID: 32107338; PMCID: PMC7776255.
- Bruzzese A, Derudas D, Galli M, Martino EA, Rocco S, Conticello C, et.al. Elotuzumab plus lenalidomide and dexamethasone in relapsed/refractory multiple myeloma: Extended 3-year follow-up of a multicenter, retrospective clinical experience with 319 cases outside of controlled clinical trials. Hematol Oncol. 2022 Oct;40(4):704-715. doi: 10.1002/hon.3031. Epub 2022 Jun 1. PMID: 35608183.
- Usmani SZ, Hoering A, Ailawadhi S, Sexton R, Lipe B, Hita SF, et.al; SWOG1211 Trial Investigators. Bortezomib, lenalidomide, and dexamethasone with or without elotuzumab in patients with untreated, high-risk multiple myeloma (SWOG-1211): primary analysis of a randomised, phase 2 trial. Lancet Haematol. 2021 Jan;8(1):e45-e54. doi: 10.1016/S2352-3026(20)30354-9. Epub 2020 Dec 22. PMID: 33357482; PMCID: PMC8601389.
- Yee AJ, Laubach JP, Campagnaro EL, Lipe BC, Nadeem O, Friedman RS, et.al. Elotuzumab in combination with pomalidomide, bortezomib, and dexamethasone in relapsed and refractory multiple myeloma. Blood Adv. 2025 Mar 11;9(5):1163-1170. doi: 10.1182/bloodadvances.2024014717. PMID: 39626297; PMCID: PMC11925518.
- Lawton S, Bishton MJ, Clark P, Thackray K, Smith D. Daratumumab, bortezomib and dexamethasone for previously treated myeloma – comparing real-world outcomes in England to the CASTOR phase III clinical trial. Blood. 2024;144(Suppl 1):3778. [CrossRef]
- Sonneveld P, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, et.al. Overall Survival With Daratumumab, Bortezomib, and Dexamethasone in Previously Treated Multiple Myeloma (CASTOR): A Randomized, Open-Label, Phase III Trial. J Clin Oncol. 2023 Mar 10;41(8):1600-1609. doi: 10.1200/JCO.21.02734. Epub 2022 Nov 22. PMID: 36413710; PMCID: PMC10022857.
| Agent/Regimen | Year Introduced | Origin/Context | Current Status in MM Therapy |
|---|---|---|---|
| Melphalan | 1953 | Derived from nitrogen mustard (Great War I) | Still used in transplant conditioning (HD Melphalan) [12,13]. |
| Prednisone | 1950’s | US Army synthesis; corticosteroid | Still used, often in combination regimens [13,14]. |
| M2 Protocol | 1977 | Multi-agent alkylator+ steroid regimen | Historical relevance, rarely used today [15]. |
| Thalidomide | 1953 1997 |
Sedative, Anti-myeloma | Limited due to toxicity; replaced by analogs [11] |
| Dexamethasone | 1960’s | Synthetic glucocorticoid | Still used in most frontline and salvage regimens [16,17]. |
| Doxorubicin | 1969 | Anthracycline (from Streptomyces peucetius) | Still used selectively, limited by cardiotoxicity [18]. |
| Treatment Group | Risk | Path | Total Cost (USD) | Total QALYs | Total ICER (Cost/ QALY) |
Cumulative ICER (USD/QALY) |
|---|---|---|---|---|---|---|
| TE | High | D-VRd→(ASCT)→D-Kd→PVd→CART→ Teclistamab→ | 6,163,033.28 USD | 18.1 | 340,499 | 3,240,643.15 |
| TE | Low / Intermediate |
D-VRd→KRd→ Isa-Kd→CART→Teclistamab | 8,426,332 USD | 18.576 | 453,613.9 | 3,419,842.55 |
| TI | High | D-Rd→Isa-Kd→Elo-Pd→CART→ Teclistamab | 7,043,070 USD | 13.528 | 520,629 | 5,093,778.66 |
| TI | Low/ Intermediate | D-VMP→ Isa-Kd→Elo-Pd→CART→Teclistamab | 5,625,073.5 USD | 16.693 | 336,972 | 3,594,452.68 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




