Submitted:
19 September 2025
Posted:
25 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. C. elegans Maintenance and Treatment
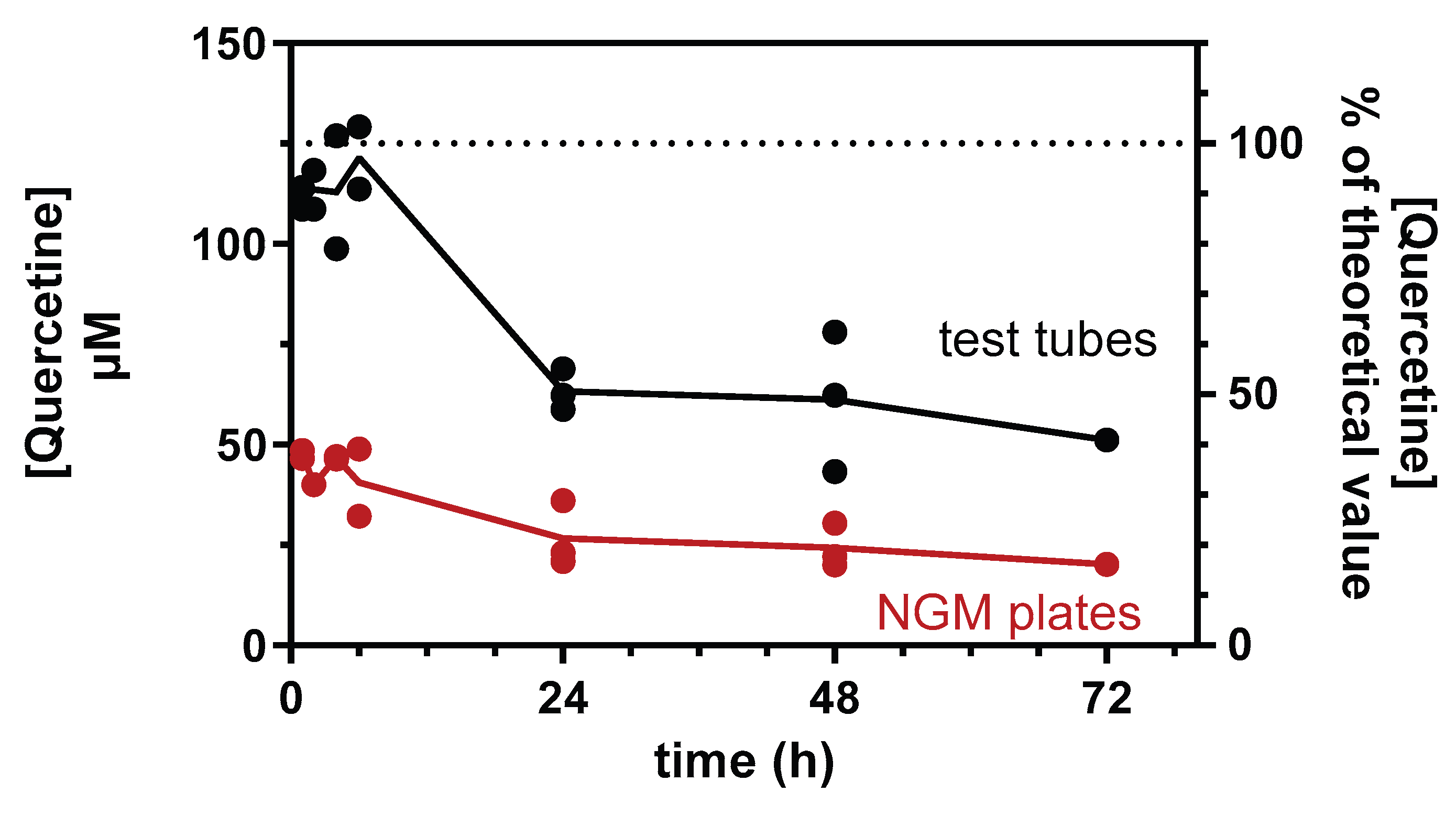
2.3. QF Stability
2.4. Lifespan and Health Span
2.5. Stress Assays
2.6. AGEs Formation
2.7. Gene Expression
2.8. Statistical Analysis
3. Results
3.1. Optimization of Experimental Conditions for QF’s Efficacy Studies
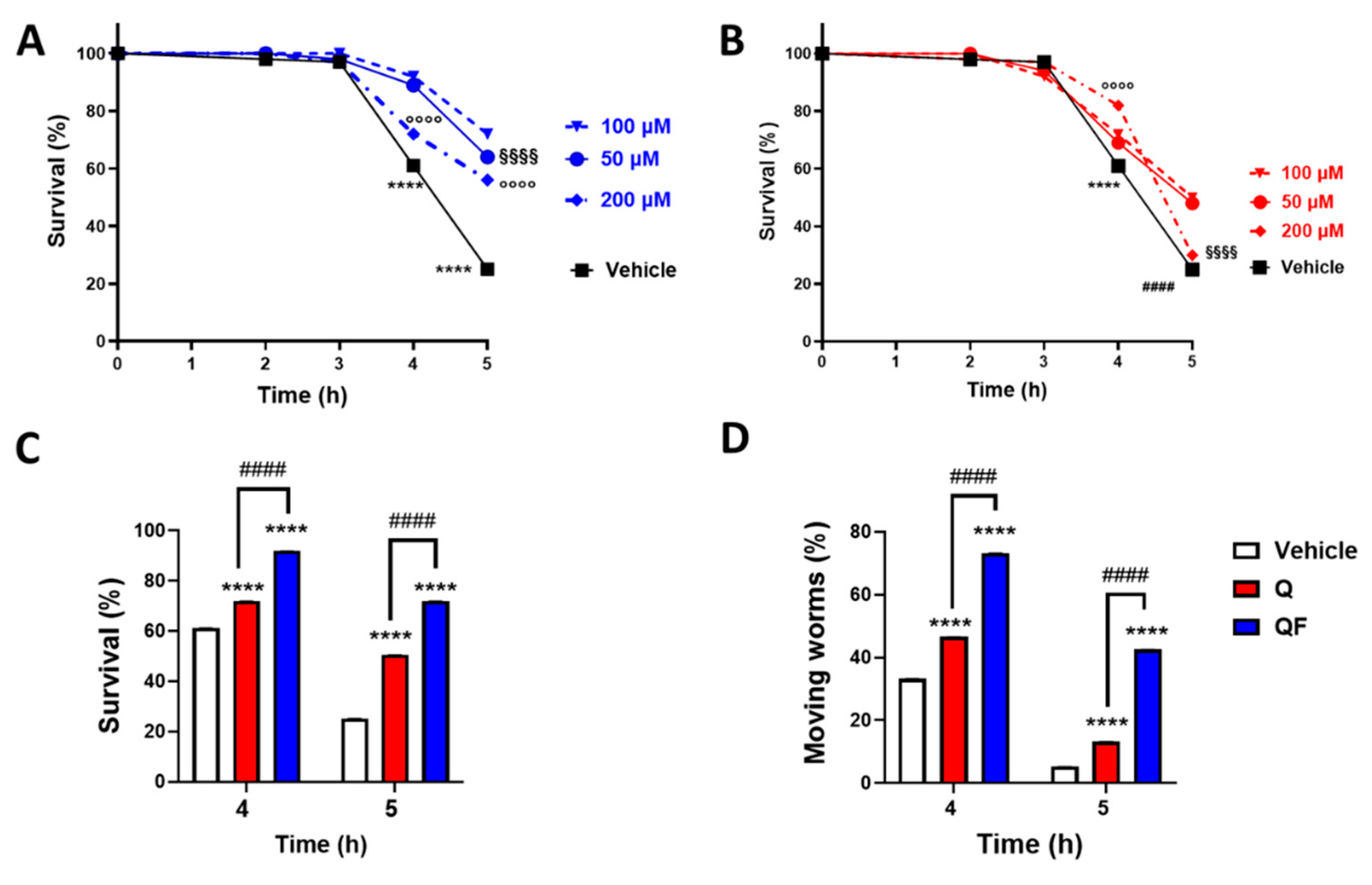
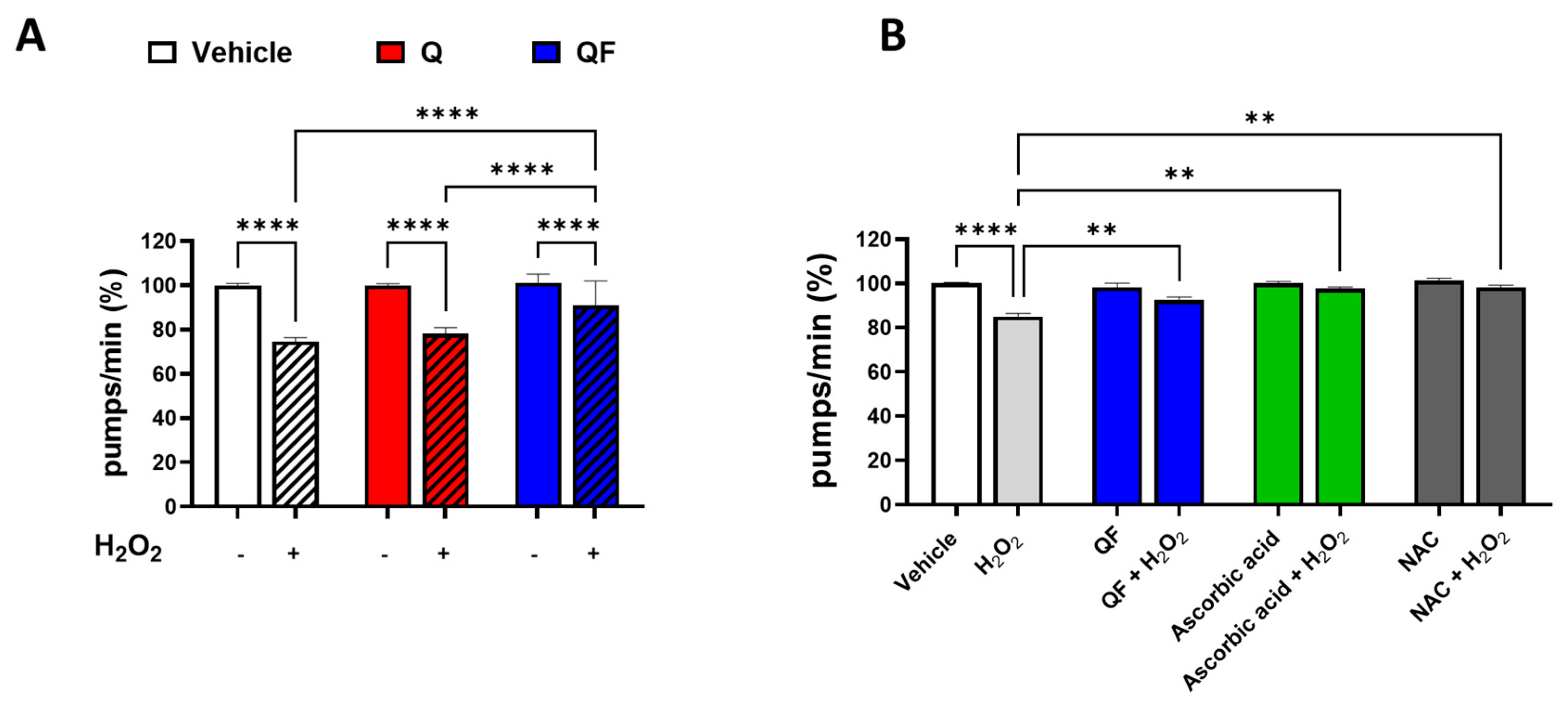
3.2. QF Enhances Worm Resistance to Thermal and Oxidative Stress
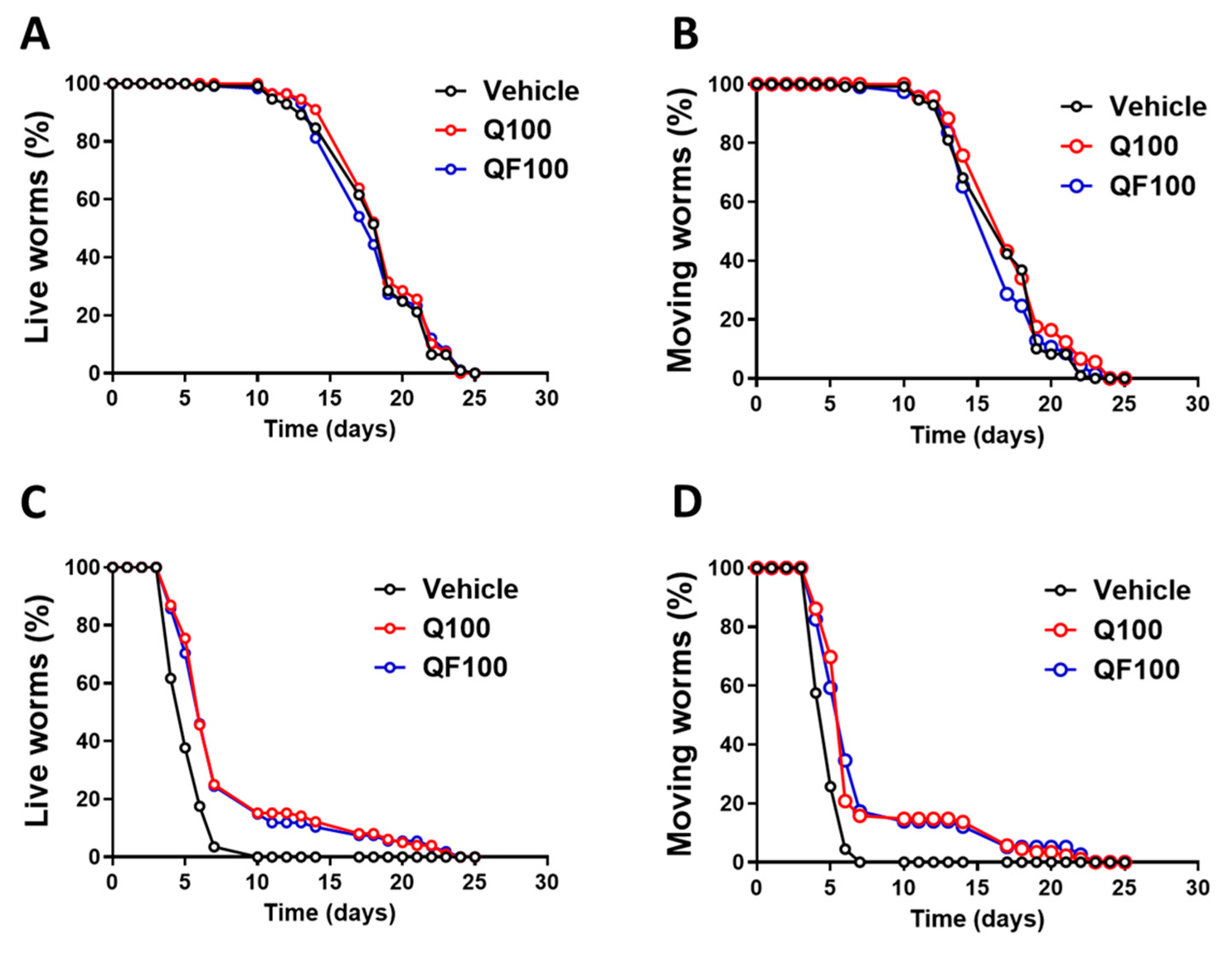
3.3. QF Enhances the Worm’s Lifespan and Health Span
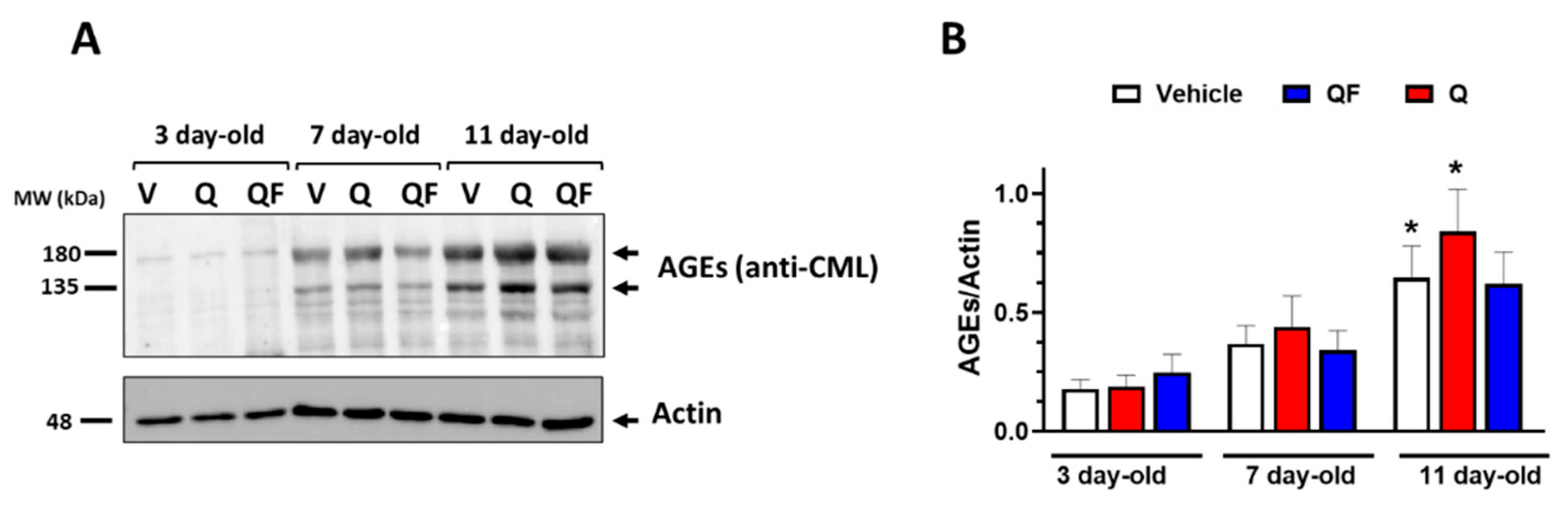
3.4. QF Does not Significantly Reduce AGEs Accumulation
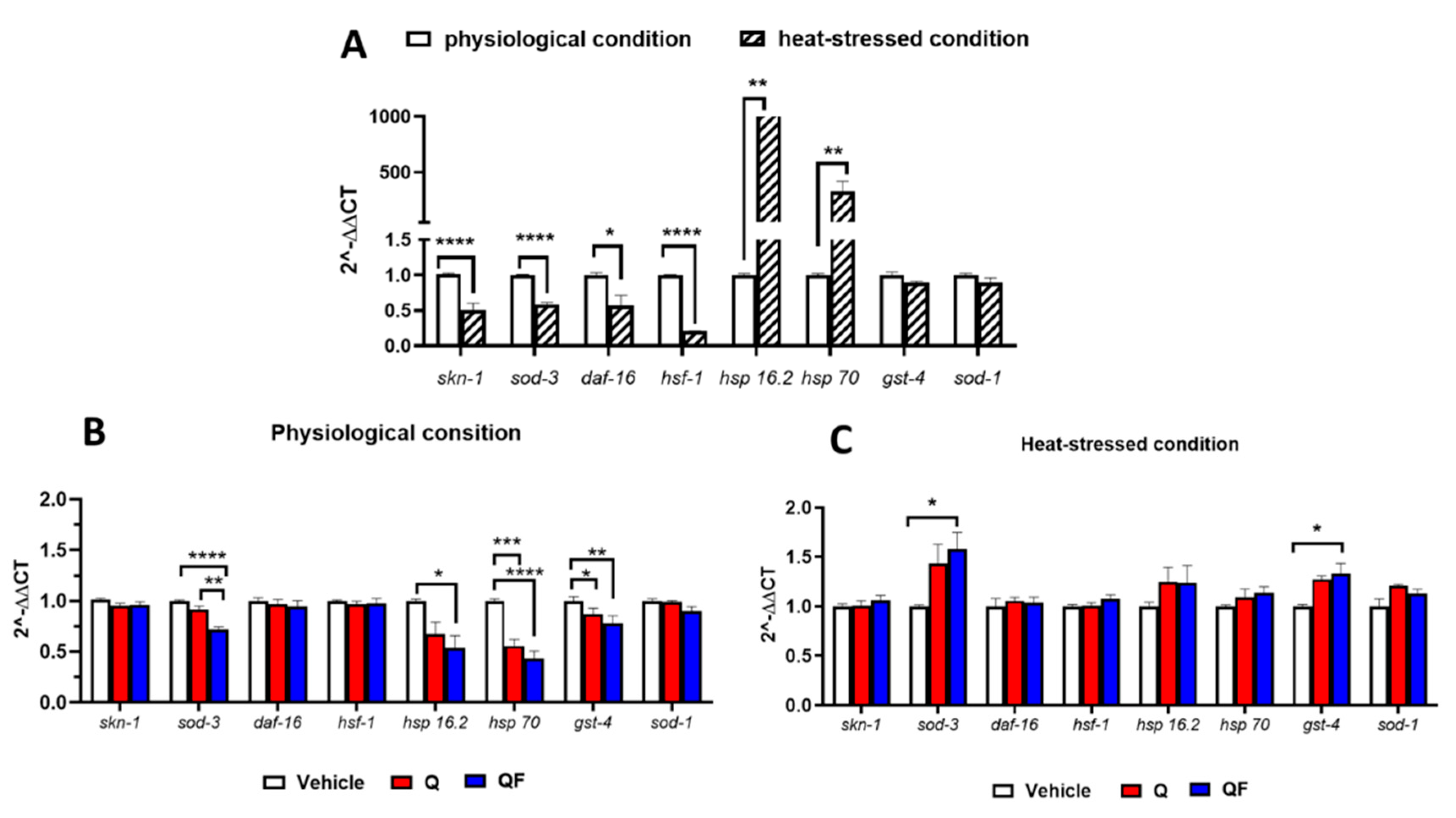
3.5. QF Under Stress Conditions Regulates Gene Expression and Heat-Shock Elements
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Q | Quercetin |
| QF | Quercefit™ Phytosome™ |
| ROS | Reactive oxygen species |
| C. | Caenorhabditis |
| IIS | Insulin/insulin-like growth factor signaling (IIS) pathway |
| AGEs | Advanced glycation end products |
| FOXO4 | Human forkhead box O4 |
| SOD-3 | Superoxide dismutase-3 |
| CTL-1 | Catalase-1 |
| HSP | Small heat shock protein |
| ACN | Acetonitrile |
| CMC | Sodium carboxymethyl cellulose |
| CGC | Caenorhabditis Genetics Centre |
| NGM | Nematode growth medium |
| SDS | Sodium dodecyl sulfate |
| NAC | N-acetylcysteine |
References
- Wissler Gerdes, E.O.; Zhu, Y.; Tchkonia, T.; Kirkland, J.L. Discovery, Development, and Future Application of Senolytics: Theories and Predictions. FEBS J 2020, 287, 2418–2427. [Google Scholar] [CrossRef]
- Ebrahimpour, S.; Zakeri, M.; Esmaeili, A. Crosstalk between Obesity, Diabetes, and Alzheimer’s Disease: Introducing Quercetin as an Effective Triple Herbal Medicine. Ageing Res Rev 2020, 62, 101095. [Google Scholar] [CrossRef]
- McHugh, D.; Gil, J. Senescence and Aging: Causes, Consequences, and Therapeutic Avenues. J Cell Biol 2018, 217, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Viña, J.; Olaso-Gonzalez, G.; Arc-Chagnaud, C.; De la Rosa, A.; Gomez-Cabrera, M.C. Modulating Oxidant Levels to Promote Healthy Aging. Antioxid Redox Signal 2020, 33, 570–579. [Google Scholar] [CrossRef]
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remón, A.; M’hiri, N.; García-Lobato, P.; Manach, C.; Knox, C.; Eisner, R.; Wishart, D.S.; et al. Phenol-Explorer 3.0: A Major Update of the Phenol-Explorer Database to Incorporate Data on the Effects of Food Processing on Polyphenol Content. Database (Oxford) 2013, 2013, bat070. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The Biological Activities, Chemical Stability, Metabolism and Delivery Systems of Quercetin: A Review. Trends in Food Science & Technology 2016, 56, 21–38. [Google Scholar] [CrossRef]
- Ayuda-Durán, B.; González-Manzano, S.; Miranda-Vizuete, A.; Sánchez-Hernández, E.; R Romero, M.; Dueñas, M.; Santos-Buelga, C.; González-Paramás, A.M. Exploring Target Genes Involved in the Effect of Quercetin on the Response to Oxidative Stress in Caenorhabditis Elegans. Antioxidants (Basel) 2019, 8, 585. [Google Scholar] [CrossRef]
- Saul, N.; Pietsch, K.; Menzel, R.; Steinberg, C.E.W. Quercetin-Mediated Longevity in Caenorhabditis Elegans: Is DAF-16 Involved? Mech Ageing Dev 2008, 129, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Sakamoto, K. Quercetin Enhances Motility in Aged and Heat-Stressed Caenorhabditis Elegans Nematodes by Modulating Both HSF-1 Activity, and Insulin-like and P38-MAPK Signalling. PLoS One 2020, 15, e0238528. [Google Scholar] [CrossRef]
- Lan, W.; Xiao, X.; Nian, J.; Wang, Z.; Zhang, X.; Wu, Y.; Zhang, D.; Chen, J.; Bao, W.; Li, C.; et al. Senolytics Enhance the Longevity of Caenorhabditis Elegans by Altering Betaine Metabolism. The Journals of Gerontology, Series A: Biological Sciences and Medical Sciences 2024, 79, glae221. [Google Scholar] [CrossRef]
- Fabian, D.K.; Fuentealba, M.; Dönertaş, H.M.; Partridge, L.; Thornton, J.M. Functional Conservation in Genes and Pathways Linking Ageing and Immunity. Immun Ageing 2021, 18, 23. [Google Scholar] [CrossRef]
- Giunti, S.; Andersen, N.; Rayes, D.; De Rosa, M.J. Drug Discovery: Insights from the Invertebrate Caenorhabditis Elegans. Pharmacology Res & Perspec 2021, 9, e00721. [Google Scholar] [CrossRef]
- Hohmann, M.S.; Habiel, D.M.; Coelho, A.L.; Verri, W.A.; Hogaboam, C.M. Quercetin Enhances Ligand-Induced Apoptosis in Senescent Idiopathic Pulmonary Fibrosis Fibroblasts and Reduces Lung Fibrosis In Vivo. Am J Respir Cell Mol Biol 2019, 60, 28–40. [Google Scholar] [CrossRef]
- Kim, S.R.; Jiang, K.; Ogrodnik, M.; Chen, X.; Zhu, X.-Y.; Lohmeier, H.; Ahmed, L.; Tang, H.; Tchkonia, T.; Hickson, L.J.; et al. Increased Renal Cellular Senescence in Murine High-Fat Diet: Effect of the Senolytic Drug Quercetin. Transl Res 2019, 213, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Özsoy Gökbilen, S.; Becer, E.; Vatansever, H.S. Senescence-Mediated Anticancer Effects of Quercetin. Nutr Res 2022, 104, 82–90. [Google Scholar] [CrossRef]
- Watkins, B.A.; Mitchell, A.E.; Shin, A.C.; Dehghani, F.; Shen, C.-L. Dietary Flavonoid Actions on Senescence, Aging, and Applications for Health. The Journal of Nutritional Biochemistry 2025, 139, 109862. [Google Scholar] [CrossRef] [PubMed]
- Tomou, E.-M.; Papakyriakopoulou, P.; Saitani, E.-M.; Valsami, G.; Pippa, N.; Skaltsa, H. Recent Advances in Nanoformulations for Quercetin Delivery. Pharmaceutics 2023, 15, 1656. [Google Scholar] [CrossRef]
- Ferreira-Silva, M.; Faria-Silva, C.; Carvalheiro, M.C.; Simões, S.; Marinho, H.S.; Marcelino, P.; Campos, M.C.; Metselaar, J.M.; Fernandes, E.; Baptista, P.V.; et al. Quercetin Liposomal Nanoformulation for Ischemia and Reperfusion Injury Treatment. Pharmaceutics 2022, 14, 104. [Google Scholar] [CrossRef] [PubMed]
- Riva, A.; Ronchi, M.; Petrangolini, G.; Bosisio, S.; Allegrini, P. Improved Oral Absorption of Quercetin from Quercetin Phytosome®, a New Delivery System Based on Food Grade Lecithin. Eur J Drug Metab Pharmacokinet 2019, 44, 169–177. [Google Scholar] [CrossRef]
- Šebeková, K.; Brouder Šebeková, K. Glycated Proteins in Nutrition: Friend or Foe? Exp Gerontol 2019, 117, 76–90. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative Stress and Diabetic Complications. Circ Res 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Srikanth, V.; Maczurek, A.; Phan, T.; Steele, M.; Westcott, B.; Juskiw, D.; Münch, G. Advanced Glycation Endproducts and Their Receptor RAGE in Alzheimer’s Disease. Neurobiol Aging 2011, 32, 763–777. [Google Scholar] [CrossRef]
- Kim, J.; Jo, Y.; Cho, D.; Ryu, D. L-Threonine Promotes Healthspan by Expediting Ferritin-Dependent Ferroptosis Inhibition in C. Elegans. Nat Commun 2022, 13, 6554. [Google Scholar] [CrossRef]
- Han, S.K.; Lee, D.; Lee, H.; Kim, D.; Son, H.G.; Yang, J.-S.; Lee, S.-J.V.; Kim, S. OASIS 2: Online Application for Survival Analysis 2 with Features for the Analysis of Maximal Lifespan and Healthspan in Aging Research. Oncotarget 2016, 7, 56147–56152. [Google Scholar] [CrossRef]
- Brinkmann, V.; Romeo, M.; Larigot, L.; Hemmers, A.; Tschage, L.; Kleinjohann, J.; Schiavi, A.; Steinwachs, S.; Esser, C.; Menzel, R.; et al. Aryl Hydrocarbon Receptor-Dependent and -Independent Pathways Mediate Curcumin Anti-Aging Effects. Antioxidants (Basel) 2022, 11. [Google Scholar] [CrossRef]
- Diomede, L.; Romeo, M.; Rognoni, P.; Beeg, M.; Foray, C.; Ghibaudi, E.; Palladini, G.; Cherny, R.A.; Verga, L.; Capello, G.L.; et al. Cardiac Light Chain Amyloidosis: The Role of Metal Ions in Oxidative Stress and Mitochondrial Damage. Antioxid. Redox Signal. 2017, 27, 567–582. [Google Scholar] [CrossRef] [PubMed]
- Komura, T.; Yamanaka, M.; Nishimura, K.; Hara, K.; Nishikawa, Y. Autofluorescence as a Noninvasive Biomarker of Senescence and Advanced Glycation End Products in Caenorhabditis Elegans. NPJ Aging Mech Dis 2021, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Dueñas, M.; Surco-Laos, F.; González-Manzano, S.; González-Paramás, A.M.; Gómez-Orte, E.; Cabello, J.; Santos-Buelga, C. Deglycosylation Is a Key Step in Biotransformation and Lifespan Effects of Quercetin-3-O-Glucoside in Caenorhabditis Elegans. Pharmacol Res 2013, 76, 41–48. [Google Scholar] [CrossRef]
- Back, P.; Braeckman, B.P.; Matthijssens, F. ROS in Aging Caenorhabditis Elegans: Damage or Signaling? Oxid Med Cell Longev 2012, 2012, 608478. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.I.; Pincus, Z.; Slack, F.J. Longevity and Stress in Caenorhabditis Elegans. Aging (Albany NY) 2011, 3, 733–753. [Google Scholar] [CrossRef]
- Rodriguez, M.; Snoek, L.B.; De Bono, M.; Kammenga, J.E. Worms under Stress: C. Elegans Stress Response and Its Relevance to Complex Human Disease and Aging. Trends Genet 2013, 29, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Dues, D.J.; Andrews, E.K.; Schaar, C.E.; Bergsma, A.L.; Senchuk, M.M.; Van Raamsdonk, J.M. Aging Causes Decreased Resistance to Multiple Stresses and a Failure to Activate Specific Stress Response Pathways. Aging (Albany NY) 2016, 8, 777–795. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, K.; Saul, N.; Menzel, R.; Stürzenbaum, S.R.; Steinberg, C.E.W. Quercetin Mediated Lifespan Extension in Caenorhabditis Elegans Is Modulated by Age-1, Daf-2, Sek-1 and Unc-43. Biogerontology 2009, 10, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Kampkötter, A.; Timpel, C.; Zurawski, R.F.; Ruhl, S.; Chovolou, Y.; Proksch, P.; Wätjen, W. Increase of Stress Resistance and Lifespan of Caenorhabditis Elegans by Quercetin. Comp Biochem Physiol B Biochem Mol Biol 2008, 149, 314–323. [Google Scholar] [CrossRef]
- Fitzenberger, E.; Deusing, D.J.; Marx, C.; Boll, M.; Lüersen, K.; Wenzel, U. The Polyphenol Quercetin Protects the Mev-1 Mutant of Caenorhabditis Elegans from Glucose-Induced Reduction of Survival under Heat-Stress Depending on SIR-2.1, DAF-12, and Proteasomal Activity. Mol Nutr Food Res 2014, 58, 984–994. [Google Scholar] [CrossRef]
| Name | Sequence (5’-3’) |
|---|---|
| skn-1- Forward | 5’-GCGACGAGACGAGACGATAA-3’ |
| skn-1-Reverse | 5’-TGAGGTGTTGGACGATGGTG-3’ |
| sod-3- Forward | 5‘-GGCTGTTTCGAAAGGGAATCT-3‘ |
| sod-3-Reverse | 5‘-CCTTTGAAGGTTCTCCACCA-3‘ |
| daf-16 - Forward | 5‘-ATCAGACATCGTTTCCTTCGG-3‘ |
| daf-16 -Reverse | 5‘-TTAACCGTTTCTCTGGACTAGC-3‘ |
| hsf-1 - Forward | 5‘-CGAGGATCCACTCAGACAGC-3‘ |
| hsf-1 -Reverse | 5‘-GTAGTTTGGGTCCGGCACAT-3‘ |
| hsp-16.2- Forward | 5‘-TCCATCTGAGTCTTCTGAGATTGTT-3‘ |
| hsp-16.2 -Reverse | 5‘-GATAGCGTACGACCATCCAAA-3‘ |
| hsp-70- Forward | 5‘-AGCCGGTTGAAAAGGCACT-3‘ |
| hsp-70 -Reverse | 5‘-AGTTGAGGTCCTTCCCATTGAA-3‘ |
| gst-4 - Forward | 5‘-CTTGGCAAGAAAATTTGGACTC-3‘ |
| gst-4 -Reverse | 5‘-GCGTCACTTCCATAGAAAACG-3‘ |
| sod-1- Forward | 5‘-AGGTCTCCAACGCGATTTTT-3‘ |
| sod-1 -Reverse | 5‘-TCGGACTTCTGTGTGATCCAG-3‘ |
| cdc-42 - Forward | 5’-CTGTTGTGGTGGGTCGAGAG-3’ |
| cdc-42 - Reverse | 5’-GTTGACGCAGAAGGGACTGA-3’ |
| y45F10d.4 - Forward | 5’-ATCTTCCCTGGCAACCGAAT-3’ |
| y45F10d.4 - Reverse | 5’-TGGGCGAGCATTGAACAGT-3’ |
| PHYSIOLOGIC CONDITION | HEAT-STRESS CONDITION | |||||
| Vehicle | Q | QF | Vehicle | Q | QF | |
| Sample size | 120 | 120 | 120 | 120 | 123 | 120 |
| Censor | 11 | 16 | 18 | 3 | 8 | 17 |
| Median of survival (days ± SEM) |
18.28 ± 0.32 | 18.80 ± 0.29 | 18.21 ± 0.33 | 5.27 ± 0.13 | 8.16 ± 0.46**** | 7.94 ± 0.48**** |
| Lifespan Improvement rate (%) |
- | 2.8 | 0 | - | 54.8 | 50.7 |
| Median of moving Worms (days ± SEM) |
16.76 ± 0.29 | 17.43 ± 0.30 | 16.57 ± 0.3 | 4.88 ± 0.08 | 7.45 ± 0.43**** | 7.49 ± 0.48**** |
| Health span improvement rate (%) | - | 4.0 | 0 | 0 | 52.7 | 53.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




