Submitted:
17 September 2025
Posted:
19 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
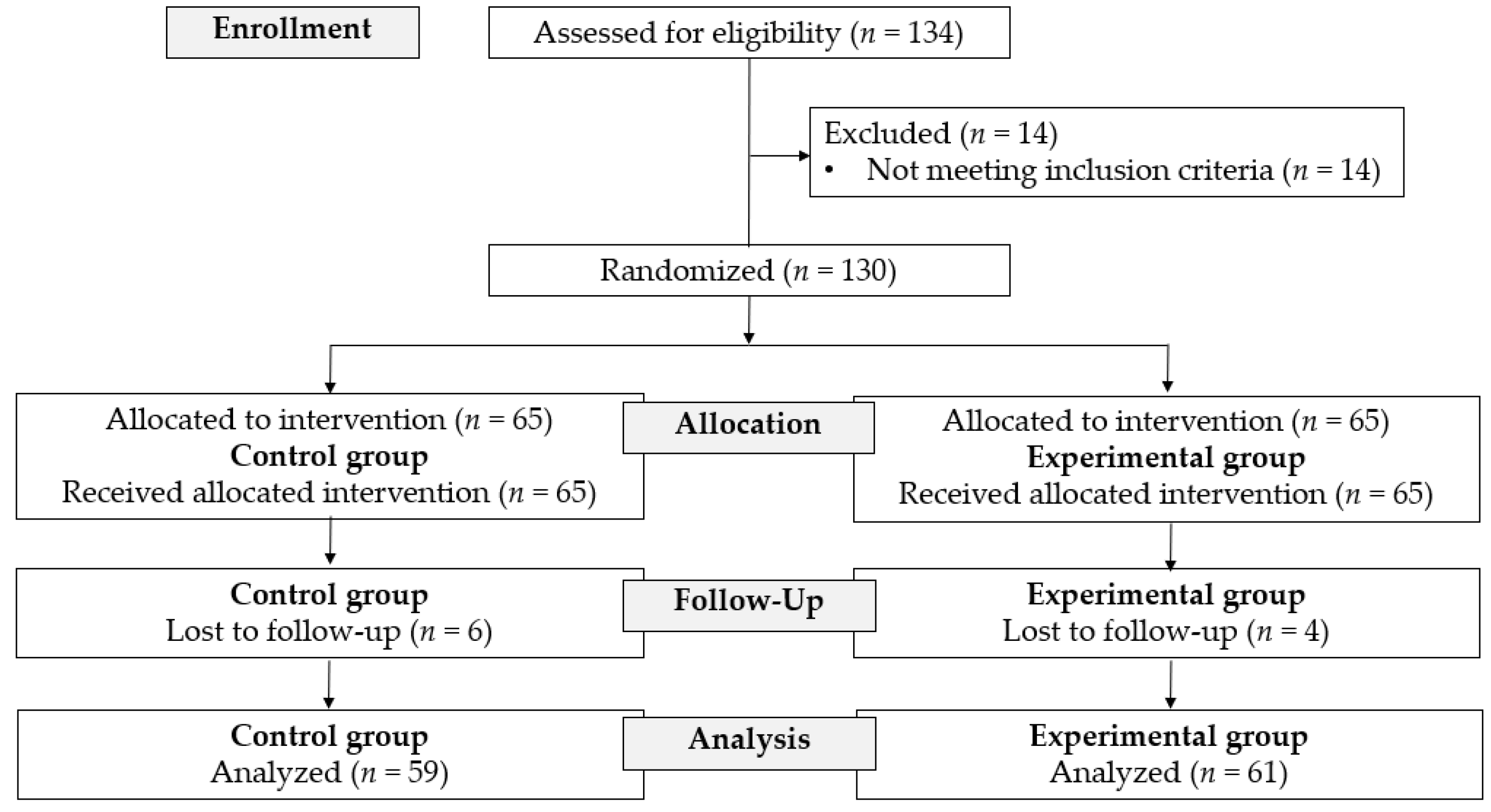
2.1. Study Design
2.2. Participants
2.3. Interventions
2.4. Outcomes
2.5. Sample Size
- Zα/2 = 1.96 (for a two-sided significance level of 0.05)
- Zβ = 0.84 (for 80% power)
- Δ = 0.2 (effect size)
- σ = 0.5 (standard deviation)
2.6. Randomization and Blinding
2.7. Study Procedures
2.8. Data Collection and Compliance
2.9. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Funding
Data availability
Acknowledgments
Declaration of competing interest
References
- World Health Organization, Advice for the public: Coronavirus disease (COVID-19), World Health Organization, 2023.
- S. Perlman, Another decade, another coronavirus, N. Engl. J. Med. 382(8) (2020) 1–2. [CrossRef]
- J.S. Peiris, C.M. Chu, V.C. Cheng, et al., Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study, Lancet 361(9371) (2003) 1767–1772. [CrossRef]
- Q. Li, X. Guan, P. Wu, et al., Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia, N. Engl. J. Med. 382(13) (2020) 1199–1207. [CrossRef]
- C. Huang, Y. Wang, X. Li, et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet 395(10223) (2020) 497–506. [CrossRef]
- N. Chen, M. Zhou, X. Dong, et al., Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study, Lancet 395(10223) (2020) 507–513. [CrossRef]
- H.A. Rothan, S.N. Byrareddy, The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak, J. Autoimmun. 109 (2020) 102433. [CrossRef]
- W. Ni, X. Yang, D. Yang, et al., Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19, Crit. Care 24(1) (2020) 422. [CrossRef]
- R. Wölfel, V.M. Corman, W. Guggemos, et al., Virological assessment of hospitalized patients with COVID-2019, Nature 581(7809) (2020) 465–469. [CrossRef]
- L. Zou, F. Ruan, M. Huang, et al., SARS-CoV-2 viral load in upper respiratory specimens of infected patients, N. Engl. J. Med. 382(12) (2020) 1177–1179. [CrossRef]
- D. Panatto, A. Orsi, B. Bruzzone, et al., Efficacy of the Sentinox spray in reducing viral load in mild COVID-19 and its virucidal activity against other respiratory viruses: results of a randomized controlled trial and an in vitro study, Viruses 14(5) (2022) 1033. [CrossRef]
- S. Alsaleh, A. Alhussien, A. Alyamani, et al., Efficacy of povidone-iodine nasal rinse and mouth wash in COVID-19 management: a prospective, randomized pilot clinical trial (povidone-iodine in COVID-19 management), BMC Infect. Dis. 24 (2024) 271. https://doi.org /10.1186/s12879-024-09137-y.
- M. Idrees, B. McGowan, A. Fawzy, A.A. Abuderman, R. Balasubramaniam, O. Kujan, Efficacy of mouth rinses and nasal spray in the inactivation of SARS-CoV-2: a systematic review and meta-analysis of in vitro and in vivo studies, Int. J. Environ. Res. Public Health 19(19) (2022) 12148. [CrossRef]
- E.R. Anderson, E.I. Patterson, S. Richards, et al., CPC-containing oral rinses inactivate SARS-CoV-2 variants and are active in the presence of human saliva, J. Med. Microbiol. 71 (2022) 001508. [CrossRef]
- N. Okamoto, A. Saito, T. Okabayashi, A. Komine, Virucidal activity and mechanism of action of cetylpyridinium chloride against SARS-CoV-2, J. Oral Maxillofac. Surg. Med. Pathol. 34(6) (2022) 800–804. [CrossRef]
- Z. Low, R. Lani, V. Tiong, C. Poh, S. AbuBakar, P. Hassandarvish, COVID-19 therapeutic potential of natural products, Int. J. Mol. Sci. 24(11) (2023) 9589. [CrossRef]
- A.N.R. Corrêa, P. Weimer, R.C. Rossi, J.F. Hoffmann, L.S. Koester, E.S. Suyenaga, C.D. Ferreira, Lime and orange essential oils and d-limonene as a potential COVID-19 inhibitor: Computational, in chemico, and cytotoxicity analysis, Food Biosci. 51 (2023) 102348. [CrossRef]
- F. Yang, R. Chen, W. Li, et al., D-limonene is a potential monoterpene to inhibit PI3K/Akt/IKK-α/NF-κB p65 signaling pathway in coronavirus disease 2019 pulmonary fibrosis, Front. Med. 8 (2021) 591830. [CrossRef]
- H. Lin, Z. Li, Y. Sun, et al., D-Limonene: promising and sustainable natural bioactive compound, Appl. Sci. 14 (2024) 4605. [CrossRef]
- Falk-Filipsson, A. Löf, M. Hagberg, E.W. Hjelm, Z. Wang, d-limonene exposure to humans by inhalation: uptake, distribution, elimination, and effects on the pulmonary function, J. Toxicol. Environ. Health (1993) 77-88. [CrossRef]
- J.A. Miller, I.A. Hakim, W. Chew, P. Thompson, C.A. Thomson, H.S. Chow, Adipose tissue accumulation of d-limonene with the consumption of a lemonade preparation rich in d-limonene content, Nutr. Cancer 62(6) (2010) 783–788. [CrossRef]
- L.A. Barker, B.W. Bakkum, C. Chapman, The clinical use of monolaurin as a dietary supplement: a review of the literature, J. Chiropr. Med. 18(4) (2019) 305–310. [CrossRef]
- J.J. Kabara, R. Vrable, M.S.F. Lie Ken Jie, Antimicrobial lipids: natural and synthetic acids and monoglycerides, Lipids 12(9) (1977) 753–759. [CrossRef]
- S.J. Projan, S. Brown-Skrobot, P.M. Schlievert, F. Vandenesch, R.P. Novick, Glycerol monolaurate inhibits the production of β-lactamase, toxic shock syndrome toxin-1 and other staphylococcal exoproteins by interfering with signal transduction, J. Bacteriol. 176(14) (1994) 4204–4209. [CrossRef]
- E. Subroto, R. Indiarto, Bioactive monolaurin as an antimicrobial and its potential to improve the immune system and against COVID-19: a review, Food Res. 4(6) (2020) 2355–2365. [CrossRef]
- Y. Weerapol, S. Manmuan, S. Limmatvapirat, C. Limmatvapirat, J. Sirirak, P. Tamdee, S. Tubtimsri, Enhancing the efficacy of monolaurin against SARS-CoV-2 and influenza A (H1N1) with a nanoemulsion formulation, OpenNano 17 (2024) 100207. [CrossRef]
- G.L. Drusano, Infection site concentrations: their therapeutic importance and the macrolide and macrolide-like class of antibiotics, Pharmacotherapy 25(12 Pt 2) (2005) 150S–158S. [CrossRef]
- T. Fazekas, P. Eickhoff, N. Pruckner, et al., Lessons learned from a double-blind randomised placebo-controlled study with a iota-carrageenan nasal spray as medical device in children with acute symptoms of common cold, BMC Complement. Altern. Med. 12 (2012) 147. [CrossRef]
- F. Fais, R. Juskeviciene, V. Francardo, et al., Drug-free nasal spray as a barrier against SARS-CoV-2 and its delta variant: in vitro study of safety and efficacy in human nasal airway epithelia, Int. J. Mol. Sci. 23(7) (2022) 4062. [CrossRef]
- J.S. Pelletier, B. Tessema, S. Frank, J.B. Westover, S.M. Brown, J.A. Capriotti, Efficacy of povidone-iodine nasal and oral antiseptic preparations against severe acute respiratory syndrome-coronavirus 2 (SARS-CoV-2), Ear Nose Throat J. 100(2_suppl) (2021) 192S–196S. [CrossRef]
- K.F. Schulz, D.G. Altman, D. Moher, CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials, BMJ 340 (2010) c332. [CrossRef]
- World Health Organization, Living Guidance for Clinical Management of COVID-19, World Health Organization, 2021.
- J. Ponphaiboon, W. Krongrawa, S. Limmatvapirat, S. Tubtimsri, A. Jittmittraphap, P. Leaungwutiwong, C. Mahidol, S. Ruchirawat, P. Kittakoop, C. Limmatvapirat, In vitro development of local antiviral formulations with potent virucidal activity against SARS-CoV-2 and influenza viruses, Pharmaceutics 17 (2025) 349. [CrossRef]
- C. Mahidol, S. Ruchirawat, W. Sutananta, P. Kittakoop, N. Wanichacheva, S. Limmatvapirat, C. Limmatvapirat, P. Leaungwutiwong, A. Jittmittraphap, S. Tubtimsri, J. Ponphaiboon, and W. Krongrawa, Antiviral product for oral and throat and production method, Thai Petty Patent no. 20893, 2022.
- C. Mahidol, S. Ruchirawat, W. Sutananta, P. Kittakoop, N. Wanichacheva, S. Limmatvapirat, C. Limmatvapirat, P. Leaungwutiwong, A. Jittmittraphap, S. Tubtimsri, J. Ponphaiboon, and W. Krongrawa, Nasal spray product for killing viruses and production method, Thai Petty Patent no. 20894, 2022.
- Gupta, E. Jeyakumar, R. Lawrence, Journey of limonene as an antimicrobial agent, J. Pure Appl. Microbiol. 15 (3) (2021) 1094–1110. [CrossRef]
- E. Subroto, Monoacylglycerols and diacylglycerols for fat-based food products: a review, Food Res. 4 (4) (2020) 932–943. [CrossRef]
- M. Bañó-Polo, L. Martínez-Gil, M.M. Sánchez del Pino, A. Massoli, I. Mingarro, R. Léon, M.J. Garcia-Murria, Cetylpyridinium chloride promotes disaggregation of SARS-CoV-2 virus-like particles, J. Oral Microbiol. 14 (1) (2022) 2030094. [CrossRef]
- Y. Weerapol, S. Manmuan, S. Limmatvapirat, C. Limmatvapirat, J. Sirirak, P. Tamdee, S. Tubtimsri, Enhancing the efficacy of monolaurin against SARS-CoV-2 and influenza A (H1N1) with a nanoemulsion formulation, OpenNano 17 (2024) 100207. [CrossRef]
- H.J. Rodríguez-Casanovas, M.D.I. Rosa, Y. Bello-Lemus, G. Rasperini, A.J. Acosta-Hoyos, Virucidal activity of different mouthwashes using a novel biochemical assay, Healthcare 10 (2022) 63. [CrossRef]
- A.M. Parikh-Das, N.C. Sharma, Q. Du, C.A. Charles, Superiority of essential oils versus 0.075% CPC-containing mouthrinse: a two-week randomized clinical trial, J. Clin. Dent. 24 (3) (2013) 94–99.
- M.Z.I. Chowdhury, K.C. Sikdar, T.C. Turin, Sample size calculation in clinical studies: Some common scenarios, Int. J. Stat. Med. Res. 6 (4) (2017) 152–161. [CrossRef]
- World Health Organization, COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health, World Health Organization, 2023.
- Alhassan, N. Asiamah, F.F. Opuni, A. Alhassan, The Likert scale: exploring the unknowns and their potential to mislead the world, UDS Int. J. Dev. 9 (2) (2022) 867–880. [CrossRef]
- C. Chong, Dividing the emergency department into red, yellow, and green zones to control COVID-19 infection; a letter to editor, Arch. Acad. Emerg. Med. 8 (1) (2020) e60.
- M. Nicola, Z. Alsafi, C. Sohrabi, A. Kerwan, A. Al-Jabir, C. Iosifidis, M. Agha, R. Aghaf, The socio-economic implications of the coronavirus pandemic (COVID-19): a review, Int. J. Surg. 78 (2020) 185–193. [CrossRef]
- A.N.R. Corrêa, P. Weimer, R.C. Rossi, J.F. Hoffmann, L.S. Koester, E.S. Suyenaga, D. Ferreira, Lime and orange essential oils and D-limonene as a potential COVID-19 inhibitor: computational, in chemico, and cytotoxicity analysis, Food Biosci. 51 (2023) 102348. [CrossRef]
- N.Q. Fadilah, A. Jittmittraphap, P. Leaungwutiwong, P. Pripdeevech, D. Dhanushka, C. Mahidol, S. Ruchirawat, P. Kittakoop, Virucidal activity of essential oils from Citrus × aurantium L. against influenza A virus H1N1: limonene as a potential household disinfectant against virus, Nat. Prod. Commun. 17 (1) (2022) 1–7. [CrossRef]
- S.C. Chaudhary, M.S. Siddiqui, M. Athar, M.S. Alam, D-limonene modulates inflammation, oxidative stress and Ras-ERK pathway to inhibit murine skin tumorigenesis, Hum. Exp. Toxicol. 31 (8) (2012) 798–811. [CrossRef]
- Razazi, A. Kakanezhadi, A. Raisi, B. Pedram, O. Dezfoulian, F. Davoodi, D-limonene inhibits peritoneal adhesion formation in rats via anti-inflammatory, anti-angiogenic, and antioxidative effects, Inflammopharmacology 32 (2) (2024) 1077–1089. [CrossRef]
- M.F.N. Meeran, A. Seenipandi, H. Javed, C. Sharma, H.M. Hashiesh, S.N. Goyal, N.K. Jha, S. Ojha, Can limonene be a possible candidate for evaluation as an agent or adjuvant against infection, immunity, and inflammation in COVID-19?, Heliyon 7 (1) (2021) e05703. [CrossRef]
- M. Bacanlı, A.A. Başaran, N. Başaran, The antioxidant and antigenotoxic properties of citrus phenolics limonene and naringin, Food Chem. Toxicol. 81 (2015) 160–170. [CrossRef]
- E.S. Keles, Mild SARS-CoV-2 infections in children might be based on evolutionary biology and linked with host reactive oxidative stress and antioxidant capabilities, New Microbes New Infect. 36 (2020) 100723. [CrossRef]
- Khomich, S.N. Kochetkov, B. Bartosch, A.V. Ivanov, Redox biology of respiratory viral infections, Viruses 10 (8) (2018) 392. [CrossRef]
- M.A. Beck, J. Handy, O.A. Levander, Host nutritional status: the neglected virulence factor, Trends Microbiol. 12 (9) (2004) 417–423. [CrossRef]
- E. Subroto, R. Indiarto, Bioactive monolaurin as an antimicrobial and its potential to improve the immune system and against COVID-19: a review, Food Res. 4 (6) (2020) 2355–2365. [CrossRef]
- D. Herrera, J. Serrano, S. Roldán, M. Sanz, Is the oral cavity relevant in SARS-CoV-2 pandemic?, Clin. Oral Investig. 24 (8) (2020) 2925–2930. [CrossRef]
- G.D. Kumar, A. Mishra, L. Dunn, A. Townsend, I.C. Oguadinma, K.R. Bright, C.P. Gerba, Biocides and novel antimicrobial agents for the mitigation of coronaviruses, Front. Microbiol. 11 (2020) 1351. [CrossRef]
- T. Nagatake, K. Ahmed, K. Oishi, Prevention of respiratory infections by povidone-iodine gargle, Dermatology 204 (Suppl. 1) (2002) 32–36. [CrossRef]
- R. Kawana, T. Kitamura, O. Nakagomi, et al., Inactivation of human viruses by povidone-iodine in comparison with other antiseptics, Dermatology 195 (Suppl. 2) (1997) 29–35. [CrossRef]
- Steyer, M. Marušić, M. Kolenc, T. Triglav, A throat lozenge with fixed combination of cetylpyridinium chloride and benzydamine hydrochloride has direct virucidal effect on SARS-CoV-2, COVID 1 (2) (2021) 435–446. [CrossRef]
- D.L. Popkin, S. Zilka, M. Dimaano, H. Fujioka, C. Rackley, R. Salata, A. Griffith, P.K. Mukherjee, M.A. Ghannoum, F. Esper, Cetylpyridinium chloride (CPC) exhibits potent, rapid activity against influenza viruses in vitro and in vivo, Pathog. Immun. 2 (2) (2017) 253–269. [CrossRef]
- W. Luo, K. Wang, J. Luo, et al., Limonene anti-TMV activity and its mode of action, Pestic. Biochem. Physiol. 194 (2023) 105512. [CrossRef]
- P.M. Schlievert, S.H. Kilgore, K.S. Seo, D.Y.M. Leung, Glycerol monolaurate contributes to the antimicrobial and anti-inflammatory activity of human milk, Sci. Rep. 9 (1) (2019) 14550. [CrossRef]
- S.L.F. Miranda, J.T. Damaceno, M. Faveri, L.C. Figueiredo, G.M.S. Soares, M. Feres, B. Bueno-Silva, In vitro antimicrobial effect of cetylpyridinium chloride on complex multispecies subgingival biofilm, Braz. Dent. J. 31 (2) (2020) 103–108. [CrossRef]
- G. LeBel, K. Vaillancourt, M. Morin, D. Grenier, Antimicrobial activity, biocompatibility and anti-inflammatory properties of cetylpyridinium chloride-based mouthwash containing sodium fluoride and xylitol: An in vitro study, Oral Health Prev. Dent. 18 (1) (2020) 1069–1076. [CrossRef]
- J. Sun, D-limonene: Safety and clinical applications, Altern. Med. Rev. 12 (3) (2007) 259–264. https://pubmed.ncbi.nlm.nih.gov/18072821/.
- R. Tarragó-Gil, M.J. Gil-Mosteo, M. Aza-Pascual-Salcedo, et al., Randomized clinical trial to assess the impact of oral intervention with cetylpyridinium chloride to reduce salivary SARS-CoV-2 viral load, J. Clin. Periodontol. 50 (3) (2023) 288–294. [CrossRef]
- S. Felsenstein, J.A. Herbert, P.S. McNamara, C.M. Hedrich, COVID-19: immunology and treatment options, Clin. Immunol. 215 (2020) 108448. [CrossRef]
- R. Cecchini, A.L. Cecchini, SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression, Med. Hypotheses 143 (2020) 110102. [CrossRef]
| Characteristic | Experimental group (n =61) |
Control group (n =59) |
p value | |
|---|---|---|---|---|
| Age, % (n) | ||||
| 18-30 years | 18.0 (11) | 27.1 (16) | 0.657 | |
| 31-40 years | 27.9 (17) | 22.0 (13) | ||
| 41-50 years | 29.5 (18) | 22.0 (13) | ||
| 51-60 years | 14.8 (9) | 18.6 (11) | ||
| > 60 years | 9.8 (6) | 10.2 (6) | ||
| Sex, % (n) | ||||
| Male | 39.3 (24) | 42.4 (25) | 0.911 | |
| Female | 60.7 (37) | 57.6 (34) | ||
| Underlying disease, % (n) | ||||
| Hypertension | 4.9 (3) | 5.1 (3) | - | |
| Diabetes | 4.9 (3) | 1.7 (1) | ||
| Hyperlipidemia | 3.3 (2) | 5.1 (3) | ||
| Heart disease | 0.0 (0) | 3.4 (2) | ||
| Asthma | 0.0 (0) | 1.7 (1) | ||
| Thrombocytopenia | 1.6 (1) | 0.0 (0) | ||
| Spondylosis | 4.9 (3) | 0.0 (0) | ||
| Renal disease | 3.3 (2) | 0.0 (0) | ||
| Thyroid disease | 1.6 (1) | 0.0 (0) | ||
| No underlying disease | 75.4 (46) | 83.1 (49) | ||
| Symptom and severity level | Day 1 | Day 3 | Day 7 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Experimental group (n =61) |
Control group (n =59) |
p value | Experimental group (n =61) |
Control group (n =59) |
p value | Experimental group (n =61) |
Control group (n =59) |
p value | ||
| Fever and headache, % (n) | 0.038 | < 0.001 | 0.019 | |||||||
| Worse | 13.1 (8) | 10.2 (6) | 4.9 (3) | 6.8 (4) | 0.0 (0) | 0.0 (0) | ||||
| As before | 42.6 (26) | 69.5 (41) | 18.0 (11) | 11.9 (7) | 0.0 (0) | 0.0 (0) | ||||
| Slightly better | 21.3 (13) | 16.9 (10) | 23.0 (14) | 54.2 (32) | 0.0 (0) | 1.7 (1) | ||||
| Moderately improved | 4.9 (3) | 0.0 (0) | 27.9 (17) | 22.0 (13) | 0.0 (0) | 3.4 (2) | ||||
| Much better | 4.9 (3) | 0.0 (0) | 13.1 (8) | 1.7 (1) | 41.0 (25) | 66.1 (39) | ||||
| Completely recovered | 0.0 (0) | 0.0 (0) | 0.0 (0) | 0.0 (0) | 45.9 (28) | 25.4 (15) | ||||
| No previous symptoms | 13.1 (8) | 3.4 (2) | 13.1 (8) | 3.4 (2) | 13.1 (8) | 3.4 (2) | ||||
| Sore throat, % (n) | 0.605 | 0.007 | < 0.001 | |||||||
| Worse | 9.8 (6) | 10.2 (6) | 3.3 (2) | 6.8 (4) | 0.0 (0) | 0.0 (0) | ||||
| As before | 62.3 (38) | 69.5 (41) | 24.6 (15) | 23.7 (14) | 1.6 (1) | 0.0 (0) | ||||
| Slightly better | 19.7 (12) | 16.9 (10) | 34.4 (21) | 50.8 (30) | 13.1 (8) | 1.7 (1) | ||||
| Moderately improved | 1.6 (1) | 0.0 (0) | 23.0 (14) | 13.6 (8) | 18.0 (11) | 33.9 (20) | ||||
| Much better | 3.3 (2) | 0.0 (0) | 11.5 (7) | 1.7 (1) | 37.7 (23) | 57.6 (34) | ||||
| Completely recovered | 0.0 (0) | 0.0 (0) | 0.0 (0) | 0.0 (0) | 26.2 (16) | 3.4 (2) | ||||
| No previous symptoms | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | ||||
| Cough and mucus, % (n) | 0.229 | 0.046 | 0.011 | |||||||
| Worse | 13.1 (8) | 6.8 (4) | 14.8 (9) | 8.5 (5) | 0.0 (0) | 0.0 (0) | ||||
| As before | 63.9 (39) | 78.0 (46) | 29.5 (18) | 32.2 (19) | 0.0 (0) | 0.0 (0) | ||||
| Slightly better | 14.8 (9) | 11.9 (7) | 21.3 (13) | 39.0 (23) | 18.0 (11) | 30.5 (18) | ||||
| Moderately improved | 1.6 (1) | 0.0 (0) | 19.7 (12) | 15.3 (9) | 42.6 (26) | 52.5 (31) | ||||
| Much better | 3.3 (2) | 0.0 (0) | 11.5 (7) | 1.7 (1) | 21.3 (13) | 11.9 (7) | ||||
| Completely recovered | 0.0 (0) | 0.0 (0) | 0.0 (0) | 0.0 (0) | 14.8 (9) | 1.7 (1) | ||||
| No previous symptoms | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | ||||
| Runny nose and nose congestion, % (n) | 0.072 | < 0.001 | < 0.001 | |||||||
| Worse | 14.8 (9) | 8.5 (5) | 8.2 (5) | 8.5 (5) | 0.0 (0) | 0.0 (0) | ||||
| As before | 49.2 (30) | 76.3 (45) | 27.9 (17) | 22.0 (13) | 0.0 (0) | 0.0 (0) | ||||
| Slightly better | 24.6 (15) | 11.9 (7) | 19.7 (12) | 50.8 (30) | 0.0 (0) | 0.0 (0) | ||||
| Moderately improved | 4.9 (3) | 0.0 (0) | 19.7 (12) | 13.6 (8) | 16.4 (10) | 25.4 (15) | ||||
| Much better | 3.3 (2) | 0.0 (0) | 21.3 (13) | 1.7 (1) | 49.2 (30) | 67.8 (40) | ||||
| Completely recovered | 0.0 (0) | 0.0 (0) | 0.0 (0) | 0.0 (0) | 31.1 (19) | 3.4 (2) | ||||
| No previous symptoms | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | 3.3 (2) | 3.4 (2) | ||||
| Satisfaction level | Experimental group (n =61) | Control group (n =59) | |
|---|---|---|---|
| Relieving fever and headache, % (n) | |||
| Very satisfied | 8.2 (5) | 3.4 (2) | |
| Satisfied | 85.2 (52) | 64.4 (38) | |
| Neutral | 6.6 (4) | 32.2 (19) | |
| Unsatisfied | 0.0 (0) | 0.0 (0) | |
| Very unsatisfied | 0.0 (0) | 0.0 (0) | |
| Mean | 4.02 | 3.71 | |
| SD | 0.39 | 0.53 | |
| t score | 3.60 | ||
| p value | < 0.001 | ||
| Relieving sore throat, % (n) | |||
| Very satisfied | 6.6 (4) | 1.7 (1) | |
| Satisfied | 88.5 (54) | 66.1 (39) | |
| Neutral | 4.9 (3) | 32.2 (19) | |
| Unsatisfied | 0.0 (0) | 0.0 (0) | |
| Very unsatisfied | 0.0 (0) | 0.0 (0) | |
| Mean | 4.02 | 3.69 | |
| SD | 0.34 | 0.50 | |
| t score | 4.10 | ||
| p value | < 0.001 | ||
| Relieving cough and mucus, % (n) | |||
| Very satisfied | 4.9 (3) | 3.4 (2) | |
| Satisfied | 86.9 (53) | 55.9 (33) | |
| Neutral | 8.2 (5) | 40.7 (24) | |
| Unsatisfied | 0.0 (0) | 0.0 (0) | |
| Very unsatisfied | 0.0 (0) | 0.0 (0) | |
| Mean | 3.97 | 3.63 | |
| SD | 0.37 | 0.55 | |
| t score | 3.96 | ||
| p value | < 0.001 | ||
| Relieving runny nose and nose congestion, % (n) | |||
| Very satisfied | 13.1 (8) | 3.4 (2) | |
| Satisfied | 85.2 (52) | 59.3 (35) | |
| Neutral | 1.6 (1) | 37.3 (22) | |
| Unsatisfied | 0.0 (0) | 0.0 (0) | |
| Very unsatisfied | 0.0 (0) | 0.0 (0) | |
| Mean | 4.10 | 3.66 | |
| SD | 0.40 | 0.55 | |
| t score | 5.02 | ||
| p value | < 0.001 | ||
| Color, smell, and taste of the products, % (n) | |||
| Very satisfied | 9.8 (6) | 3.4 (2) | |
| Satisfied | 88.5 (54) | 71.2 (42) | |
| Neutral | 1.6 (1) | 25.4 (15) | |
| Unsatisfied | 0.0 (0) | 0.0 (0) | |
| Very unsatisfied | 0.0 (0) | 0.0 (0) | |
| Mean | 4.07 | 3.78 | |
| SD | 0.36 | 0.50 | |
| t score | 3.62 | ||
| p value | < 0.001 | ||
| Overall satisfaction | |||
| Mean | 4.03 | 3.69 | |
| SD | 0.30 | 0.48 | |
| t score | 4.61 | ||
| p value | < 0.001 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




