Submitted:
16 September 2025
Posted:
17 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Early CAR T-Cell Therapy Approvals and REMS Implementation
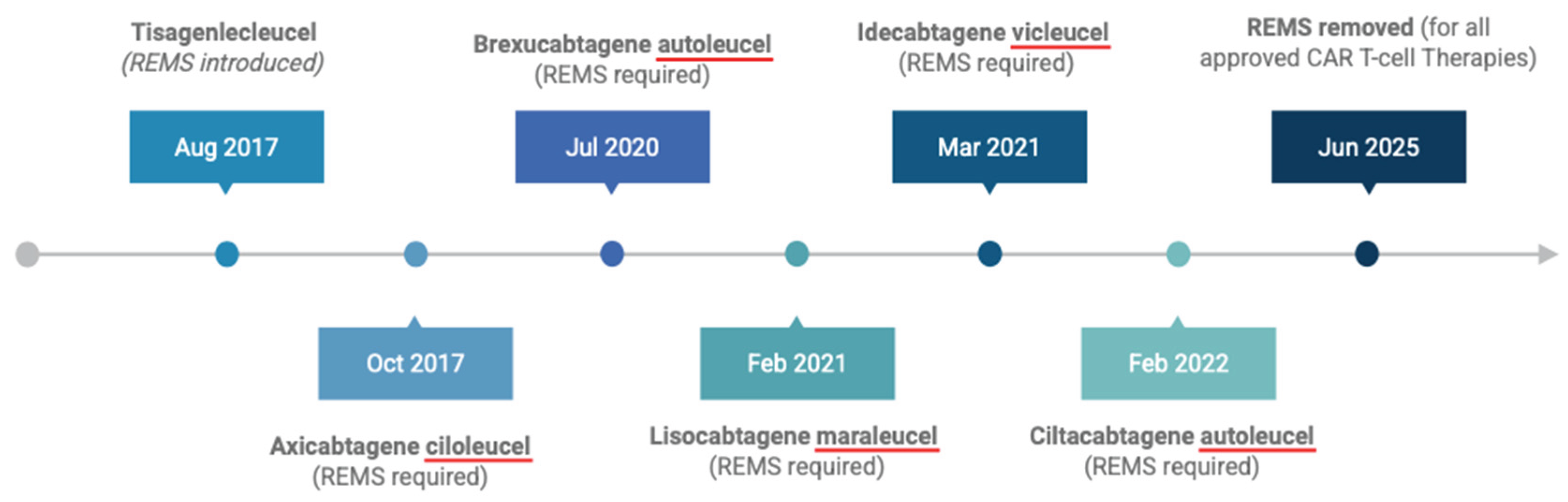
3. CRS and ICANS Onset Across CAR T-Cell Therapies
| CAR T-cell Therapy | Target | Indications |
CRS Onset median (range) |
ICANS Onset median (range) |
| Tisagenlecleucel | CD19 | B-ALL (≤25y), LBCL, FL | 3 days (1–51) | 5 days (1–368) |
| Axicabtagene ciloleucel | CD19 | LBCL, FL | 3 days (1–20) | 5 days (1–133) |
| Brexucabtagene autoleucel | CD19 | MCL, B-ALL | 4 days (1–13) | 6 days (1–51) |
| Lisocabtagene maraleucel | CD19 | LBCL, FL, CLL/SLL, MCL | 5 days (1–63) | 8 days (1–63) |
| Idecabtagene vicleucel | BCMA | MM | 1 day (1–27) | 2 days (1–148) |
| Ciltacabtagene autoleucel | BCMA | MM | 7 days (1–23) | 8 days (1–28) |
4. Additional Safety Considerations Beyond CRS and ICANS
5. Regulatory Reform: The June 2025 FDA Decision
| Policy Requirement | Before June 2025 | After June 2025 |
| Institutional certification | Required at REMS-approved sites | Not required |
| Tocilizumab availability | On-site and immediately accessible | Not required |
| Prescriber/staff training | REMS-specific certification | Integrated into product labeling |
| Patient proximity after infusion | 4 weeks within 2-hour radius (often stricter in practice) | 2 weeks within reasonable proximity to a healthcare facility |
| Driving restriction | 8 weeks | 2 weeks |
6. Expanding Access and Addressing Health Equity
7. Adapting Delivery Models in Response to REMS Elimination
7.1. Share-Care Approach
7.2. Organizational Support
7.3. Accreditation Challenges
7.4. Telehealth Integration
7.5. Future Delivery Models
8. Recommendations and Equity-Driven Implementation Models
8.1. Expand Treatment Sites
8.2. Revise Payer Policies
8.3. Standardize Patient and Caregiver Education
8.4. Integrate Telehealth and Remote Monitoring
8.5. Educate Community Providers
8.6. Strengthen Equity Monitoring Systems
9. Challenges in Maintaining Safety After REMS Removal
10. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACCC | Association for Community Cancer Centers |
| ALL | Acute lymphoblastic leukemia |
| ASTCT | American Society for Transplantation and Cellular Therapy |
| BCMA | B-cell maturation antigen |
| B-ALL | B-cell acute lymphoblastic leukemia |
| CAR T | Chimeric antigen receptor T cell |
| CI | Confidence interval |
| CIBMTR | Center for International Blood and Marrow Transplant Research |
| CIDR | Cellular Immunotherapy Data Resource |
| CLL/SLL | Chronic lymphocytic leukemia / small lymphocytic lymphoma |
| CRS | Cytokine release syndrome |
| DEPICT | Diverse and Equitable Participation in Clinical Trials |
| FACT | Foundation for Accreditation of Cellular Therapies |
| FDA | Food and Drug Administration |
| FL | Follicular lymphoma |
| ICANS | Immune effector cell-associated neurotoxicity syndrome |
| LBCL | Large B-cell lymphoma |
| MCL | Mantle cell lymphoma |
| MM | Multiple Myeloma |
| NIH | National Institutes of Health |
| OR | Odds ratio |
| OS | Overall Survival |
| PFS | Progression-free survival |
| REMS | Risk Evaluation and Mitigation Strategies |
| RPM | Remote patient monitoring |
| SEER | Surveillance, Epidemiology, and End Results Program. |
| VTE | Venous thromboembolism |
References
- Maude SL, Laetsch TW, Buechner J, Rives S, Boyer M, Bittencourt H, Bader P, Verneris MR, Stefanski HE, Myers GD; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. New England Journal of Medicine 2018, 378, 439–448. [CrossRef]
- U.S. Food and Drug Administration. FDA Eliminates Risk Evaluation and Mitigation Strategies (REMS) for Autologous Chimeric Antigen Receptor (CAR) T-Cell Immunotherapies. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-eliminates-risk-evaluation-and-mitigation-strategies-rems-autologous-chimeric-antigen-receptor (accessed on 10 August 2025).
- KYMRIAH (tisagenlecleucel) prescribing information. East Hanover (NJ): Novartis Pharmaceuticals Corporation; revised June 2025. https://www.fda.gov/media/107296/download (accessed on 09 August 2025).
- YESCARTA (axicabtagene ciloleucel) prescribing information. Santa Monica (CA): Kite Pharma, Inc.; revised June 2025. https://www.fda.gov/media/108377/download (accessed on 09 August 2025).
- TECARTUS (brexucabtagene autoleucel) prescribing information. Santa Monica (CA): Kite Pharma, Inc.; revised June 2025. https://www.fda.gov/media/140409/download (accessed on 09 August 2025).
- BREYANZI (lisocabtagene maraleucel) prescribing information. Summit (NJ): Bristol-Myers Squibb; revised June 2025. https://www.fda.gov/media/187950/download (accessed on 09 August 2025).
- ABECMA (idecabtagene vicleucel) prescribing information. Summit (NJ): Bristol-Myers Squibb; revised June 2025. https://www.fda.gov/media/147055/download (accessed on 09 August 2025).
- CARVYKTI (ciltacabtagene autoleucel) prescribing information. Horsham (PA): Janssen Biotech, Inc.; revised June 2025. https://www.fda.gov/media/156560/download (accessed on 09 August 2025).
- Ahmed, N.; Wesson, W.; Lutfi, F.; Porter, D.L.; Bachanova, V.; Nastoupil, L.J.; Perales, M.-A.; Maziarz, R.T.; Brower, J.; Shah, G.L.; et al. Optimizing the post-CAR T monitoring period in recipients of axicabtagene ciloleucel, tisagenlecleucel, and lisocabtagene maraleucel. Blood Adv. 2024, 8, 5346–5353. [CrossRef]
- Ventin, M.; Cattaneo, G.; Maggs, L.; Arya, S.; Wang, X.; Ferrone, C.R. Implications of High Tumor Burden on Chimeric Antigen Receptor T-Cell Immunotherapy: A Review. JAMA Oncol. 2024, 10, 115–121. [CrossRef]
- Hill, J.A.; Seo, S.K. How I prevent infections in patients receiving CD19-targeted chimeric antigen receptor T cells for B-cell malignancies. Blood 2020, 136, 925–935. [CrossRef]
- Little JS; Aleissa MM; Beluch K; Gonzalez-Bocco IH; Marty FM; Manne-Goehler J; Koo S; Hammond SP; Jacobson CA. Low incidence of invasive fungal disease following CD19 chimeric antigen receptor T-cell therapy for non-Hodgkin lymphoma. Blood Advances 2022, 6(16), 4821–4830. [CrossRef]
- Jain, T.; Knezevic, A.; Pennisi, M.; Chen, Y.; Ruiz, J.D.; Purdon, T.J.; Devlin, S.M.; Smith, M.; Shah, G.L.; Halton, E.; et al. Hematopoietic recovery in patients receiving chimeric antigen receptor T-cell therapy for hematologic malignancies. Blood Adv. 2020, 4, 3776–3787. [CrossRef]
- 14. Melody M; Gandhi S; Saunders H; Abdel-Rahman Z; Hastings J; Lengerke Diaz P; Gannon N; Truong T; Hathcock M; Khurana A; Johnston P; Ansell S; Bennani N; Paludo J; Bisneto JV; Wang Y; Rosenthal A; Foran J; Ayala E; Murthy HS; Roy V; Castro JE; Lin Y; Kharfan-Dabaja MA. Incidence of thrombosis in relapsed/refractory B-cell lymphoma treated with axicabtagene ciloleucel: Mayo Clinic experience. Leukemia & Lymphoma 2022, 63(6), 1363–1368. [CrossRef]
- Tix, T.; Alhomoud, M.; Shouval, R.; Scheffer Cliff, E.R.; Perales, M.-A.; Cordas dos Santos, D.M.; Rejeski, K. Second Primary Malignancies after CAR T-Cell Therapy: A Systematic Review and Meta-Analysis of 5,517 Lymphoma and Myeloma Patients. Clin. Cancer Res. 2024, 30, 4690–4700. [CrossRef]
- Ellithi, M.; Elsallab, M.; Lunning, M.A.; Holstein, S.A.; Sharma, S.; Trinh, J.Q.; Ma, J.; Maus, M.V.; Frigault, M.J.; D’Angelo, C.R. Neurotoxicity and Rare Adverse Events in BCMA-Directed CAR T Cell Therapy: A Comprehensive Analysis of Real-World FAERS Data. Transplant. Cell. Ther. 2025, 31, 71.e1–71.e14. [CrossRef]
- American Society of Hematology. FDA-Mandated CAR-T Monitoring Period Could Be Halved. 24 July 2024. https://www.hematology.org/newsroom/press-releases/2024/fda-mandated-car-t-monitoring-period-could-be-halved (accessed on 10 August 2025).
- Berberabe, T. CAR T-Cell Therapy Remains Underutilized, Despite Improvements in Access. Targeted Ther. Oncol. 2024, 13(9), 16. https://www.targetedonc.com/view/car-t-cell-therapy-remains-underutilized-despite-improvements-in-access (accessed on 10 August 2025).
- Ahmed N; Shahzad M; Shippey E; Bansal R; Mushtaq MU; Mahmoudjafari Z; Faisal MS; Hoffmann M; Abdallah AO; Divine C; Hamadani M; McGuirk J; Shune L. Socioeconomic and Racial Disparity in Chimeric Antigen Receptor T Cell Therapy Access. Transplant Cell Ther 2022, 28(7), 358–364. [CrossRef]
- Badr, H.; Rouce, R.; Scheurer, M.E.; Lulla, P.; Mims, M.; Reddy, P. Bringing CAR T Cell Therapy Trials to Underserved Populations. Cancer Cell 2023, 41, 2007–2010. [CrossRef]
- Islam N; Budvytyte L; Khera N; Hilal T. Disparities in clinical trial enrollment- focus on CAR-T and bispecific antibody therapies. Current Hematologic Malignancy Reports 2025, 20, 1–10. [CrossRef]
- U.S. Congress. Diverse and Equitable Participation in Clinical Trials (DEPICT) Act of 2022, H.R.6584, 117th Congress. https://www.congress.gov/bill/117th-congress/house-bill/6584 (accessed on 4 September 2025).
- U.S. Congress. NIH Clinical Trial Diversity Act of 2023, H.R.3503 / S.1701, 118th Congress. https://www.congress.gov/bill/118th-congress/senate-bill/1701 (accessed on 4 September 2025).
- Emole, J.; Lawal, O.; Lupak, O.; Dias, A.; Shune, L.; Yusuf, K. Demographic Differences among Patients Treated with Chimeric Antigen Receptor T-Cell Therapy in the United States. Cancer Med. 2022, 11, 4440–4448. [CrossRef]
- Ahmed N; Sun F; Teigland C; Kilgore KM; Mohammadi I; Chambers J; Dieyi C; Feng C; Osborn J; Fu C; Gergis U. Chimeric Antigen Receptor T-Cell Access in Patients with Relapsed/Refractory Large B-Cell Lymphoma: Association of Access with Social Determinants of Health and Travel Time to Treatment Centers. Transplantation and Cellular Therapy 2024, 30(7), 714–725. [CrossRef]
- Karmali, R.; Machhi, R.; Epperla, N.; Shouse, G.; Romancik, J.; Moyo, T.K.; Kenkre, V.; Ollila, T.A.; Fitzgerald, L.; Hess, B.; et al. Impact of Race and Social Determinants of Health on Outcomes in Patients with Aggressive B-Cell NHL Treated with CAR-T Therapy. Blood Adv. 2024, 8, 2592–2599. [CrossRef]
- Snyder, S.; Chung, K.C.; Jun, M.P.; Gitlin, M. Access to Chimeric Antigen Receptor T Cell Therapy for Diffuse Large B Cell Lymphoma. Adv. Ther. 2021, 38, 4659–4674. [CrossRef]
- Davis ES; Franks JA; Bhatia S; Kenzik KM. Urban-rural differences in cancer mortality: Operationalizing rurality. J Rural Health 2024, 40(2), 268–271. [CrossRef]
- Khare S, Williamson S, O'Barr B, Schachter L, Chen A, Hayes-Lattin B, Leonard J, Desai A, Ferreira-Gandolfo P, Christmas K; et al. Sociodemographic Factors Influencing Access to Chimeric Antigen Receptor T-Cell Therapy for Patients with Non-Hodgkin Lymphoma. Clin. Lymphoma Myeloma Leuk. 2025, 25(2), e120–e125. [CrossRef]
- Borgert R. Improving outcomes and mitigating costs associated with CAR T-cell therapy. Am J Manag Care 2021, 27(13 Suppl), S253–S261. [CrossRef]
- Hoffmann MS; Hunter BD; Cobb PW; Varela JC; Muñoz J. Overcoming barriers to referral for chimeric antigen receptor T-cell therapy in patients with relapsed/refractory diffuse large B-cell lymphoma. Transplant Cell Ther 2023, 29(7), 440–448. [CrossRef]
- Ram, R.; Grisariu, S.; Shargian-Alon, L.; Amit, O.; Bar-On, Y.; Stepensky, P.; Yeshurun, M.; Avni, B.; Hagin, D.; Perry, C.; et al. Toxicity and efficacy of chimeric antigen receptor T-cell therapy in patients with diffuse large B-cell lymphoma above the age of 70 years compared to younger patients- a matched control multicenter cohort study. Haematologica 2022, 107, 1111–1118. [CrossRef]
- Chihara, D.; Liao, L.; Tkacz, J.; Franco, A.; Lewing, B.; Kilgore, K.M.; Nastoupil, L.J.; Chen, L. Real-world experience of CAR T-cell therapy in older patients with relapsed/refractory diffuse large B-cell lymphoma. Blood 2023, 142, 1047–1054. [CrossRef]
- Kharfan-Dabaja MA, Mohty R, Easwar N, Johnston P, Iqbal M, Epperla N, Yared J, Ahmed N, Hamadani M, Beitinjaneh A; et al. Chimeric antigen receptor T cell therapy in octogenarians with B-cell lymphoma: a real-world US multicenter collaborative study. Bone Marrow Transplant 2025, 60(5), 632–639. [CrossRef]
- Shouse, G.; Danilov, A.V.; Artz, A. CAR T-Cell Therapy in the Older Person: Indications and Risks. Curr. Oncol. Rep. 2022, 24, 1189–1199. [CrossRef]
- Stolz, S.M.; Musa, A.; Bachofner, A.; Bankova, A.K.; Gourri, E.; Manz, M.G.; Rieger, M.J.; Schneidawind, D.; Wolfensberger, N.; Zenz, T.; Rösler, W. Prophylactic Tocilizumab Prior to Infusion of CD19 CAR T-Cells Reduces Therapy-Related Complications in Older Lymphoma Patients. Ann. Hematol. 2025, 104, 4149–4155. [CrossRef]
- Association of Community Cancer Centers. Bringing CAR T-Cell Therapies to Community Oncology. Association of Community Cancer Centers 2025. https://www.accc-cancer.org/home/learn/precision-medicine/treatment/immunotherapy/car-t-cell-therapy/bringing-car-t-cell-therapies-to-community-oncology (accessed on 10 August 2025).
- Byrne MT; Lyss AJ; Mullangi S. Key Challenges in CAR T-Cell Therapy Access in Community Oncology. JAMA Oncology 2025, 11(5), 481–482. [CrossRef]
- Sumarsono, A.; Case, M.; Kassa, S.; Moran, B. Telehealth as a Tool to Improve Access and Reduce No-Show Rates in a Large Safety-Net Population in the USA. J. Urban Health 2023, 100, 398–407. [CrossRef]
- Ojinnaka, C.O.; Johnstun, L.; Dunnigan, A.; Nordstrom, L.; Yuh, S. Telemedicine Reduces Missed Appointments but Disparities Persist. Am. J. Prev. Med. 2024, 67, 90–96. [CrossRef]
- Jonas Paludo, Radhika Bansal, Adam T Holland, Kelsey L Haugen, Megan T Spychalla, Alli L McClanahan, Tuan A Truong, Matthew A Hathcock, Arushi Khurana, Hassan B. Alkhateeb; et al. Pilot implementation of remote patient monitoring program for outpatient management of CAR-T cell therapy. Blood 2021, 138 (Suppl. 1), 568–570. [CrossRef]
- Devine, S.M.; Horowitz, M.M. Building a Fit for Purpose Clinical Trials Infrastructure to Accelerate the Assessment of Novel Hematopoietic Cell Transplantation Strategies and Cellular Immunotherapies. J. Clin. Oncol. 2021, 39, 534–546. [CrossRef]
- U.S. Food and Drug Administration. Pharmacovigilance in Cell and Gene Therapy: Evolving Challenges in Risk Management and Long-Term Follow-Up. FDA 2023. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/pharmacovigilance-cell-and-gene-therapy (accessed on 11 August 2025).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




