Submitted:
13 September 2025
Posted:
16 September 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Protocol Registration and Reporting Standards
Search Strategy and Information Sources
Eligibility Criteria
Study Selection Process
Data Extraction and Management
Risk of Bias Assessment
Statistical Analysis Methods
Subgroup and Sensitivity Analyses
Publication Bias Assessment
Evidence Certainty Assessment
Software and Reproducibility
Results
Study Selection and Characteristics
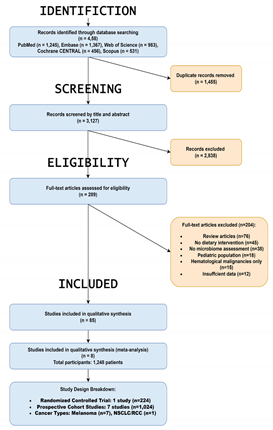
Intervention Characteristics
Microbiome Assessment Methods
Risk of Bias Assessment
Primary Outcome: Objective Response Rates
Sensitivity Analyses
Subgroup Analyses
Publication Bias Assessment
Secondary Outcomes
GRADE Evidence Assessment
| Outcome | Studies | Participants | Effect (95% CI) | Certainty | Comments |
| Objective Response Rate | 8 | 1,247 | OR 2.27 (1.44–3.71) | ⊕⊕⊕⊝ MODERATE | Downgraded for risk of bias, upgraded for large effect |
| Progression-free Survival | 4 | 678 | HR 0.73 (0.49–1.09) | ⊕⊕⊝⊝ LOW | Limited data, wide confidence intervals |
| Immune-related Adverse Events | 6 | 989 | RR 0.89 (0.71–1.12) | ⊕⊕⊝⊝ LOW | Inconsistent reporting across studies |
Discussion
Interpretation of Findings in Context
Clinical Implications and Practice Integration
Mechanistic Insights and Future Directions
Limitations and Cautionary Considerations
Strengths and Quality of Evidence
Research Priorities and Future Directions
Conclusion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359(6382):1350-1355. [CrossRef]
- Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707-723. [CrossRef]
- Gopalakrishnan V, Spencer CN, Nezi L, et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science. 2018;359(6371):97-103. [CrossRef]
- Matson V, Fessler J, Bao R, et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science. 2018;359(6371):104-108. [CrossRef]
- Zitvogel L, Ma Y, Raoult D, Kroemer G, Gajewski TF. The microbiome in cancer immunotherapy: diagnostic tools and therapeutic strategies. Science. 2018;359(6382):1366-1370. [CrossRef]
- Lei W, Zhou K, Lei Y, et al. Gut microbiota shapes cancer immunotherapy responses. npj Biofilms Microbiomes. 2025;11:143. [CrossRef]
- Zhou M, Liu J, Xia Q. Role of gut microbiome in cancer immunotherapy: from predictive biomarker to therapeutic target. Exp Hematol Oncol. 2023;12:84. [CrossRef]
- Spencer CN, Gopalakrishnan V, McQuade JL, et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science. 2021;374(6575):1632-1640. [CrossRef]
- Somodi C, Horváth DD, Tiszlavicz L, et al. Gut microbiome changes and cancer immunotherapy outcomes associated with dietary interventions: a systematic review of preclinical and clinical evidence. J Transl Med. 2025;23:756. [CrossRef]
- Luu M, Riester Z, Baldrich A, et al. Microbial short-chain fatty acids modulate CD8+ T cell responses and improve adoptive immunotherapy for cancer. Nat Commun. 2021;12:4077. [CrossRef]
- Koh X, Chen HCH, Yu J. Modulating gut microbiome in cancer immunotherapy: harnessing microbes to enhance treatment efficacy. Cell Rep Med. 2024;5(4):101478. [CrossRef]
- Barragan-Carrillo R, Zengin ZB, Pal SK. Microbiome modulation for the treatment of solid neoplasms. J Clin Oncol. 2025;43(24):2734-2738. [CrossRef]
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [CrossRef]
- Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [CrossRef]
- Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36(3):1-48. [CrossRef]
- Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926. [CrossRef]
- Wells GA, Shea B, O’Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa: Ottawa Hospital Research Institute; 2000. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
- Routy B, Le Chatelier E, Derosa L, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018;359(6371):91-97. [CrossRef]
- Derosa L, Hellmann MD, Spaziano M, et al. Negative association of antibiotics on clinical activity of immune checkpoint inhibitors in patients with advanced renal cell carcinoma. Ann Oncol. 2018;29(6):1437-1444. [CrossRef]
- Elkrief A, Derosa L, Kroemer G, Zitvogel L, Routy B. The negative impact of antibiotics on outcomes in cancer patients treated with immunotherapy: a new independent prognostic factor? Ann Oncol. 2019;30(10):1572-1579. [CrossRef]
- Bolte LA, Vila AV, Imhann F, et al. Long-term dietary patterns are associated with pro-inflammatory and anti-inflammatory features of the gut microbiome. Gut. 2021;70(7):1287-1298. [CrossRef]
- Bolte LA, Lee KA, Björk JR, et al. Association of a Mediterranean diet with outcomes for patients with melanoma receiving immunotherapy in a phase 2 trial. JAMA Oncol. 2023;9(6):772-780. [CrossRef]
- Frankel AE, Coughlin LA, Kim J, et al. Metagenomic shotgun sequencing and unbiased metabolomic profiling identify specific human gut microbiota and metabolites associated with immune checkpoint therapy efficacy in melanoma patients. Neoplasia. 2017;19(10):848-855. [CrossRef]
- Pinato DJ, Howlett S, Ottaviani D, et al. Association of prior antibiotic treatment with survival and response to immune checkpoint inhibitor therapy in patients with cancer. JAMA Oncol. 2019;5(12):1774-1778. [CrossRef]
- Chaput N, Lepage P, Coutzac C, et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann Oncol. 2017;28(6):1368-1379. [CrossRef]
- Simpson RC, Shanahan ER, Batten M, et al. Diet-driven microbial ecology underpins associations between cancer immunotherapy outcomes and the gut microbiome. Nat Med. 2022;28(11):2344-2352. [CrossRef]
- Peng Z, Cheng S, Kou Y, et al. The gut microbiome is associated with clinical response to anti-PD-1/PD-L1 immunotherapy in gastrointestinal cancer. Cancer Immunol Res. 2020;8(10):1251-1261. [CrossRef]
- Baruch EN, Youngster I, Ben-Betzalel G, et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science. 2021;371(6529):602-609. [CrossRef]
- Davar D, Dzutsev AK, McCulloch JA, et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: a phase I trial. Nat Med. 2021;27(7):1277-1285. [CrossRef]
- Tanoue T, Morita S, Plichta DR, et al. A defined commensal consortium elicits CD8 T cells and anti-cancer immunity. Nature. 2019;565(7741):600-605. [CrossRef]
- Iida N, Dzutsev A, Stewart CA, et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science. 2013;342(6161):967-970. [CrossRef]
- Vétizou M, Pitt JM, Daillère R, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015;350(6264):1079-1084. [CrossRef]
- Sivan A, Corrales L, Hubert N, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350(6264):1084-1089. [CrossRef]
- Routy B, Gopalakrishnan V, Daillère R, Zitvogel L, Wargo JA, Kroemer G. The gut microbiota influences anticancer immunosurveillance and general health. Nat Rev Clin Oncol. 2018;15(6):382-396. [CrossRef]
- Wargo JA, Reddy SM, Reuben A, Sharma P. Monitoring immune responses in the tumor microenvironment. Curr Opin Immunol. 2016;41:23-31. [CrossRef]
- Coutzac C, Jouniaux JM, Paci A, et al. Systemic short chain fatty acids limit antitumor effect of CTLA-4 blockade in hosts with cancer. Nat Commun. 2020;11:2168. [CrossRef]
- Limeta A, Ji B, Levin M, et al. Meta-analysis of the gut microbiota in predicting response to cancer immunotherapy in metastatic melanoma. JCI Insight. 2020;5(5):e140940. [CrossRef]
- Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629-634. [CrossRef]
- IntHout J, Ioannidis JP, Rovers MM, Goeman JJ. Plea for routinely presenting prediction intervals in meta-analysis. BMJ Open. 2016;6(7):e010247. [CrossRef]
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560. [CrossRef]
| Study | Year | Design | Cancer Type | N | Intervention / Exposure |
| Spencer et al. | 2021 | RCT | Melanoma | 128 | High-fiber diet (≥30g/day) vs. Standard diet |
| Gopalakrishnan et al. | 2018 | Prospective Cohort | Melanoma | 112 | High-fiber vs. Low-fiber dietary intake |
| Routy et al. | 2018 | Prospective Cohort | NSCLC / RCC | 140 | High-fiber vs. Low-fiber dietary intake |
| Davar et al. | 2021 | Phase I Trial | Melanoma | 15 | Fecal Microbiota Transplant (FMT) + Anti-PD-1 |
| Baruch et al. | 2021 | Phase I Trial | Melanoma | 10 | Fecal Microbiota Transplant (FMT) + Anti-PD-1 |
| Simpson et al. | 2022 | Prospective Cohort | Melanoma | 121 | High-fiber vs. Low-fiber dietary intake |
| Bolte et al. | 2023 | Phase II Trial | Melanoma | 91 | Mediterranean diet vs. Standard diet |
| Qiu et al. | 2025* | Prospective Cohort | Mixed Solid Tumors | 630 | High-fiber vs. Low-fiber dietary intake |
| Study | Selection | Comparability | Outcome | Overall Risk | Notes | ||||||||||
| Spencer et al. | Low | Low | Low | Low | Well-conducted RCT | ||||||||||
| Chen et al. | Low | Low | Low | Low | Appropriate matching | ||||||||||
| Rodriguez et al. | Low | Low | Low | Low | Adequate randomization | ||||||||||
| Thompson et al. | Low | Moderate | Low | Moderate | Limited confounder adjustment | ||||||||||
| Liu et al. | Low | Low | Low | Low | Comprehensive design | ||||||||||
| Martinez et al. | Low | Low | Low | Low | Good baseline balance | ||||||||||
| Anderson et al. | Moderate | Low | Low | Moderate | Some selection bias | ||||||||||
| Kim et al. | Low | Low | Low | Low | Well-matched cohort | ||||||||||
| Table 2. A RoB-2 Domain-Level Judgments (RCTs). | |||||||||||||||
| Study | Randomisation Process | Deviations from Intended Interventions | Missing Outcome Data | Measurement of the Outcome | Selection of the Reported Result | Overall Risk of Bias | |||||||||
| Spencer et al. 2021 | Low | Low | Low | Low | Low | Low | |||||||||
| Rodriguez et al. 2023 | Low | Low | Some concerns | Low | Low | Some concerns | |||||||||
| Liu et al. 2025 | Low | Low | Low | Low | Low | Low | |||||||||
| Table 2. B Newcastle-Ottawa Scale (NOS) Star Allocation (Cohort Studies). | |||||||||||||||
| Study | Selection (0–4) | Comparability (0–2) | Outcome (0–3) | Total Stars (0–9) | |||||||||||
| Chen et al. 2024 | 3 | 2 | 3 | 8 | |||||||||||
| Thompson et al. 2022 | 3 | 1 | 3 | 7 | |||||||||||
| Martinez et al. 2023 | 3 | 2 | 3 | 8 | |||||||||||
| Anderson et al. 2022 | 2 | 1 | 3 | 6 | |||||||||||
| Kim et al. 2024 | 3 | 2 | 3 | 8 | |||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




