Submitted:
15 September 2025
Posted:
15 September 2025
You are already at the latest version
Abstract
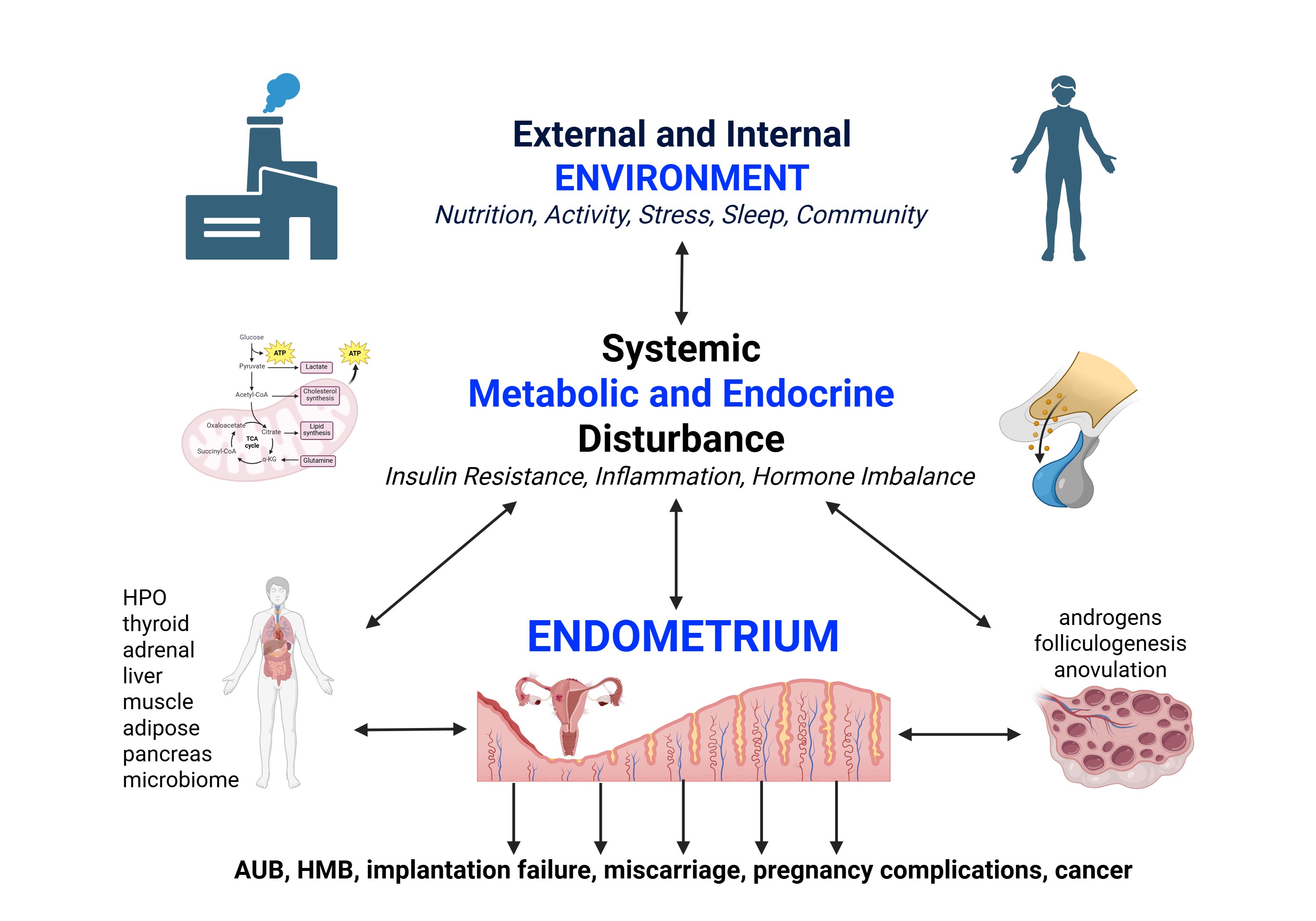
Keywords:
1. Introduction
2. Scope and Methodology
3. Endometrial Function in Reproductive-Age Women
3.1. Normal endometrial physiology
3.2. Regulation of hypothalamic-pituitary gonadotrophin hormones
3.3. Hormonal control of ovarian hormones—indirect control of the endometrium
3.3.1. Sex steroid regulation of the endometrium—systemic endocrine control
3.3.2. Intracrine metabolism in the endometrium—local hormonal control
3.4. The endometrium as a component of the mucosal immune system
3.5. Endometrial inflammation and leukocyte function
3.6. Role of vasoconstriction, hypoxia, and hemostasis in menstruation
3.7. Endometrial stem cells
4. Pathophysiology of Dysfunctional Endometrium
4.1. Classification of abnormal uterine bleeding (AUB) and heavy menstrual bleeding (HMB)
4.2. Association between IR, hyperinsulinemia, and menstrual dysfunction
4.3. Impact of IR and hyperinsulinemia on endometrial dysfunction
4.3.1. Indirect effect of IR and hyperinsulinemia on endometrial dysfunction via altered ovarian hormone production
4.3.2. Direct effect of IR and hyperinsulinemia on endometrial dysfunction
4.4. Chronic systemic inflammation (CSI) and endometrial dysfunction
4.5. Molecular changes in the dysfunctional endometrium in ovulatory and anovulatory PCOS
4.5.1. Endometrial changes in ovulatory PCOS
4.5.2. Endometrial changes in anovulatory PCOS
4.6. Reduced vasoconstriction, angiogenesis, and matrix remodeling in HMB
4.7. Genetic insights into endometrial changes in PCOS
4.8. Genetic insights into endometrial changes in AUB and HMB
4.9. Role of the microbiome (MB) in PCOS, AUB and HMB
4.9.1. Role of the microbiome in PCOS
4.9.2. Mechanistic links between the gut microbiota and endometrial dysfunction
4.9.3. Role of the endometrial microbiome in endometrial dysfunction
4.9.4. Role of the microbiome in AUB and HMB
4.10. PCOS endometrium-derived organoids and endometrial dysfunction
4.11. Impact of lifestyle changes on endometrial dysfunction where no cause is identified
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKT | Serene/threonine specific kinase (also known as protein kinase B) |
| AR | Androgen receptor |
| ASC | Apoptosis-associated speck-like protein containing a caspase recruitment |
| domain | |
| AUB | Abnormal uterine bleeding |
| BMI | Body mass index |
| cAMP | Cyclic adenosine 3’5’-monophosphate |
| CDKN1B | Cyclin-dependent kinase inhibitor 1B |
| CHO | Carbohydrate |
| COEIN | Coagulopathy, Ovulatory Dysfunction, Endometrial, Iatrogenic, |
| Not Otherwise Classified | |
| COX-2 | Cyclooxygenase-2 |
| CRP | C-reactive protein |
| CSI | Chronic systemic inflammation |
| CXCR4 | Chemokine receptor type 4 |
| CYP19A1 | Aromatase |
| CYR61 | Cysteine-rich angiogenesis inducer 61 |
| DEG | Differentially expressed genes |
| DHEA | Dehydroepiandrosterone |
| DHEAS | Dehydroepiandrosterone sulphate |
| EEO | Endometrial epithelial organoid |
| ER | Estrogen receptor |
| E2 | 17β-estradiol |
| FIGO | International Federation of Gynecology and Obstetrics |
| FOXO | Forkhead box protein O |
| FSH | Follicle stimulating hormone |
| GABA | Gamma-aminobutyric acid |
| GWAS | Genome-wide association studies |
| GLUT4 | glucose transporter type 4 |
| GnRH | Gonadotropin releasing hormone |
| HA | Hyperandrogenism |
| HIF | Hypoxia inducible factor |
| HMB | Heavy menstrual bleeding |
| HOXA10 | Homeobox 10 |
| HPO | Hypothalamic-pituitary-ovarian |
| HSD | Hydroxysteroid dehydrogenase |
| ICAM 1 | Intercellular adhesion molecule 1 |
| IFN | Interferon |
| IGF-1 | Insulin-like growth factor-1 |
| IGFBP | Insulin-like growth factor binding protein |
| IL | Interleukin |
| IR | Insulin resistance |
| IRS1 | Insulin receptor substrate 1 |
| KNDy | Kisspeptin, neurokinin B, dynorphin-y |
| LIF | Leukemia inhibitory factor |
| LH | Luteinizing hormone |
| LPS | Lipopolysaccharide |
| MAPK | Mitogen-activated protein kinase |
| MB | Microbiome |
| MMP | Matrix metalloproteinase |
| mRNA | Messenger ribose nucleic acid |
| miRNA | microRNA |
| NFκB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NICE | National Institute for Health and Care Excellence |
| NLRP3 | Nod-Like receptor family pyrin domain containing 3 |
| OSMR | Oncostatin M receptor |
| PALM | Polyp, Adenomyosis, Leiomyoma, Malignancy or Hyperplasia |
| PAX6 | Heterogenous paired box 6 |
| PCOS | Polycystic ovary syndrome |
| PG | Prostaglandin |
| PI3K | Phosphoinositide 3-kinase |
| PKA | Protein Kinase A |
| PAEP | Progesterone-associated endometrial protein |
| PR | Progesterone receptor |
| P4 | Progesterone |
| RNA | Ribose nucleic acid |
| STAT3 | Signal transducer and activator of transcription 3 |
| T cells | T lymphocytes |
| TGF | Transforming growth factor |
| TNF-α | Tumour necrosis factor-α |
| T2DM | Type 2 diabetes mellitis |
| VEGF | Vascular endothelial growth factor |
| VWF | von Willebrand factor |
| WT1 | Wilms tumour-1 |
| Wnt | Wingless-related integration site |
References
- Parker J, O’Brien C, Hawrelak J, Gersh FL. Polycystic Ovary Syndrome: An Evolutionary Adaptation to Lifestyle and the Environment. Int J Environ Res Public Health 2022, 19, 1336. [CrossRef] [PubMed]
- Dumesic DA, Abbott DH, Chazenbalk GD, Scholar G. An Evolutionary Model for the Ancient Origins of Polycystic Ovary Syndrome. J Clin Med. 2023;12(6120):1–16.
- Dumesic DA, Padmanabhan V, Abbott DH. Polycystic ovary syndrome : an evolutionary metabolic adaptation. Reproduction. 2025;169(e250021).
- Shaw LMA, Elton S. Polycystic ovary syndrome: A transgenerational evolutionary adaptation. BJOG An Int J Obstet Gynaecol. 2008;115(2):144–8.
- Azziz R, Dumesic DA, Goodarzi MO. Polycystic ovary syndrome: An ancient disorder? Fertil Steril [Internet]. 2011;95(5):1544–8. [CrossRef]
- Charifson MA, Trumble BC. Evolutionary origins of polycystic ovary syndrome: An environmental mismatch disorder. Evol Med Public Heal. 2019;(1):50–63.
- Teede HJ, Misso ML, Costello MF, Dokras A, Laven J, Moran L, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Eur J Endocrinol [Internet]. 2023;189:G43–64. [CrossRef]
- Wechsung K, Neumann U, Balint N, Wiegand S, Klinikum CV, Entwicklung P Der, et al. Polycystic Ovary Syndrome – Support and Prevention in Adolescence. Geburtsh Frauenheilk [Internet]. 2025;August:1–7. Available from. Available online: https://www.thieme-connect.de/products/ejournals/abstract/10.1055/a-2622-6321.
- Parker, J. Pathophysiological Effects of Contemporary Lifestyle on Evolutionary-Conserved Survival Mechanisms in Polycystic Ovary Syndrome. Life. 2023;13(4):1056.
- Balen A, Rajkowha M. Polycystic ovary syndrome - A systemic disorder? Best Pract Res Clin Obstet Gynaecol. 2003;17(2):263–74.
- Teede HJ, Moran LJ, Morman R, Gibson M, Dokras A, Berry L, et al. Polycystic ovary syndrome perspectives from patients and health professionals on clinical features, current name, and renaming: a longitudinal international online survey. eClinicalMedicine [Internet]. 2025;84:103287. [CrossRef]
- Zhang J, Zhang J, Zheng Z, Zhang R. Evolution of polycystic ovary syndrome and related infertility in women of child-bearing age: A global burden of disease study 2021 analysis. Eur J Obstet Gynecol Reprod Biol [Internet]. 2025;314(August):114659. [CrossRef]
- Parker J, O’Brien C, Yeoh C, Gersh FL, Brennecke S. Reducing the Risk of Pre-Eclampsia in Women with Polycystic Ovary Syndrome Using a Combination of Pregnancy Screening, Lifestyle, and Medical Management Strategies. J Clin Med. 2024;13(1774):1–33.
- Palomba S, Piltonen TT, Giudice LC. Endometrial function in women with polycystic ovary syndrome: A comprehensive review. Hum Reprod Update. 2021;27(3):584–618.
- Mousa, A; Tay, CT; Teede H. Technical Report for the 2023 International Evidence-based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Monash University; 2023.
- Giudice, LC. Endometrium in PCOS: Implantation and predisposition to endocrine CA. Best Pract Res Clin Endocrinol Metab. 2006;20(2):235–44.
- Eriksson G, Li C, Sparovec TG, Dekanski A, Torstensson S, Risal S, et al. Single-cell profiling of the human endometrium in polycystic ovary syndrome. Nat Med. 2025;(Epub ahead of print).
- Wong FC, Kim CE, Garcia-Alonso L, Vento-Tormo R. The human endometrium: atlases, models, and prospects. Curr Opin Genet Dev. 2025;92(102341):1–14.
- Yang J, Yang L, Zhou Y, Cao F, Fang H, Ma H, et al. Molecular subtype of recurrent implantation failure reveals distinct endometrial etiology of female infertility. J Transl Med. 2025;23(1):1–18.
- Lou L, Kong S, Sun Y, Zhang Z, Wang H. Human Endometrial Organoids: Recent Research Progress and Potential Applications. Front Cell Dev Biol. 2022;10(February):1–7.
- Fornes R, Ormazabal P, Rosas C, Gabler F, Vantman D, Romero C, et al. Changes in the expression of insulin signaling pathway molecules in endometria from polycystic ovary syndrome women with or without hyperinsulinemia. Mol Med. 2010;16(3–4):129–36.
- Gonzalez D, Thackeray H, Lewis PD, Mantani A, Brook N, Ahuja K, et al. Loss of WT1 expression in the endometrium of infertile PCOS patients: A hyperandrogenic effect? J Clin Endocrinol Metab. 2012;97(3):957–66.
- Mehdinejadiani S, Amidi F, Mehdizadeh M, Barati M. Effects of letrozole and clomiphene citrate on Wnt signaling pathway in endometrium of polycystic ovarian syndrome and healthy women. Biol Reprod. 2019;100(3):641–8.
- Xue, Z, Li, J, Feng, J et al. Research Progress on the Mechanism Between Polycystic Ovary Syndrome and Abnormal Endometrium. Front physio. 2021;12(788772).
- Sanchez-Garrido MA, Tena-Sempere M. Metabolic dysfunction in polycystic ovary syndrome: Pathogenic role of androgen excess and potential therapeutic strategies. Mol Metab [Internet]. 2020;35(February):100937. [CrossRef]
- Oróstica L, Poblete C, Romero C, Vega M. Pro-Inflammatory Markers Negatively Regulate IRS1 in Endometrial Cells and Endometrium from Women with Obesity and PCOS. Reprod Sci. 2020;27(1):290–300.
- Retis-Resendiz AM, González-García IN, León-Juárez M, Camacho-Arroyo I, Cerbón M, Vázquez-Martínez ER. The role of epigenetic mechanisms in the regulation of gene expression in the cyclical endometrium. Clin Epigenetics [Internet]. 2021;13(1):1–23. [CrossRef]
- Khatun M, Meltsov A, Lavogina D, Loid M, Kask K, Arffman RK, et al. Decidualized endometrial stromal cells present with altered androgen response in PCOS. Sci Rep [Internet]. 2021;11(1):1–11. [CrossRef]
- Ding N, Wang R, Wang P, Wang F. Metabolism-related proteins as biomarkers for predicting prognosis in polycystic ovary syndrome. Proteome Sci. 2024;22(1):1–13.
- Reavey JJ, Walker C, Murray AA, Brito-Mutunayagam S, Sweeney S, Nicol M, et al. Obesity is associated with heavy menstruation that may be due to delayed endometrial repair. J Endocrinol. 2021;249(2):71–82.
- Parker J, Hofstee P, Brennecke S. Prevention of Pregnancy Complications Using a Multimodal Lifestyle, Screening, and Medical Model. J Clin Med. 2024;13(15):4344.
- Hoffman, MK. The great obstetrical syndromes and the placenta. BJOG An Int J Obstet Gynaecol. 2023;130(S3):8–15.
- Hopkins BD, Goncalves MD, Cantley LC. Insulin–PI3K signalling: an evolutionarily insulated metabolic driver of cancer. Nat Rev Endocrinol. 2020;16(5):276–83.
- Burton GJ, Jauniaux E. The human placenta: new perspectives on its formation and function during early pregnancy. Proc R Soc B Biol Sci. 2023;290:20230191.
- Dias Da Silva I, Wuidar V, Zielonka M, Pequeux C. Unraveling the Dynamics of Estrogen and Progesterone Signaling in the Endometrium: An Overview. Cells. 2024;13(15):1–30.
- Vannuccini S, Ph D, Jain V, Sc M, Critchley H, Sc D. From menarche to menopause, heavy menstrual bleeding is the underrated compass in reproductive health. Fertil Steril [Internet]. 2022;118(4):625–36. [CrossRef]
- American Academy of Pediatrics Committee on Adolescence AC of O and GC on AH. Menstruation in Girls and Adolescents : Using the Menstrual Cycle as a Vital Sign. Pediatrics [Internet]. 2006;118(5):2245–50. Available from. Available online: www.pediatrics.org/cgi/doi/10.1542/%0Apeds.2006-2481.
- American College of Obstetricians and Gynecologists. Menstruation in Girls and Adolescents: Using the Menstrual Cycle as a Vital Sign. Committee Opinion No. 651. Obstet Gynecol. 2015;126(6):e143–6.
- Houghton, LC. Menstruation as the Next Vital Sign. JAMA Netw open. 2024;7(5):e2412778.
- Vollmar AKR, Ph D, Mahalingaiah S, Jukic AM, Ph D. The menstrual cycle as a vital sign: a comprehensive review. Fertil Steril Rev [Internet]. 2025;6(1):100081. [CrossRef]
- Wang YX, Arvizu M, Rich-Edwards JW, Stuart JJ, Manson JAE, Missmer SA, et al. Menstrual cycle regularity and length across the reproductive lifespan and risk of premature mortality: prospective cohort study. BMJ. 2020;371:m3464.
- Moore AM, Coolen LM, Porter DT, Goodman RL, Lehman MN. KNDy cells revisited. Endocrinology. 2018;159(9):3219–34.
- Stevenson H, Bartram S, Charalambides MM, Murthy S, Petitt T, Pradeep A, et al. Kisspeptin-neuron control of LH pulsatility and ovulation. Front Endocrinol (Lausanne). 2022;13(November):1–9.
- Nagae M, Uenoyama Y, Okamoto S, Tsuchida H, Ikegami K, Goto T, et al. Direct evidence that KNDy neurons maintain gonadotropin pulses and folliculogenesis as the GnRH pulse generator. Proc Natl Acad Sci U S A. 2021;118(5):1–11.
- Marques P, Skorupskaite K, George JT, Anderson RA. Physiology of GNRH and Gonadotropin Secretion. Endotext [Internet]. 2024;13 October:1–35. Available from: http://www.ncbi.nlm.nih. 2590.
- Hillier SG, Whitelaw PF, Smyth CD. Follicular oestrogen synthesis: the “two-cell, two-gonadotrophin” model revisited. Mol Cell Endocrinol. 1994;100(1–2):51–4.
- Konings G, Brentjens L, Delvoux B, Linnanen T, Cornel K, Koskimies P, et al. Intracrine regulation of estrogen and other sex steroid levels in endometrium and non-gynecological tissues; Pathology, physiology, and drug discovery. Front Pharmacol. 2018;Sep 19(9):940.
- Liu T, Huang Y, Lin H. Estrogen disorders: Interpreting the abnormal regulation of aromatase in granulosa cells (Review). Int J Mol Med. 2021;47(5):1–12.
- Das, SK. Cell cycle regulatory control for uterine stromal cell decidualization in implantation. Reproduction. 2009;137(6):889–99.
- Hewitt SC, Winuthayanon W, Lierz SL, Hamilton KJ, Donoghue LJ, Ramsey JT, et al. Role of ERα in mediating female uterine transcriptional responses to IGF1. Endocrinology. 2017;158(8):2427–35.
- Zhu H, Hou CC, Luo LF, Hu YJ, Yang WX. Endometrial stromal cells and decidualized stromal cells: Origins, transformation and functions. Gene [Internet]. 2014;551(1):1–14. [CrossRef]
- Vasquez YM, Wang X, Wetendorf M, Franco HL, Mo Q, Wang T, et al. FOXO1 regulates uterine epithelial integrity and progesterone receptor expression critical for embryo implantation. PLoS Genet. 2018;14(11):1–27.
- Mueller MD, Vigne JL, Minchenko A, Lebovic DI, Leitman DC, Taylor RN. Regulation of vascular endothelial growth factor (VEGF) gene transcription by estrogen receptors α and β. Proc Natl Acad Sci U S A. 2000;97(20):10972–7.
- King AE, Critchley HOD. Oestrogen and progesterone regulation of inflammatory processes in the human endometrium. J Steroid Biochem Mol Biol [Internet]. 2010;120(2–3):116–26. [CrossRef]
- Yilmaz BD, Sison CAM, Yildiz S, Miyazaki K, Coon V J, Yin P, et al. Genome-wide estrogen receptor-α binding and action in human endometrial stromal cells. F S Sci [Internet]. 2020;1(1):59–66. [CrossRef]
- Nakamura M, Takakura M, Fujii R, Maida Y, Bono Y, Mizumoto Y, et al. The PRB-dependent FOXO1/IGFBP-1 axis is essential for progestin to inhibit endometrial epithelial growth. Cancer Lett [Internet]. 2013;336(1):68–75. [CrossRef]
- Itoh H, Kishore AH, Lindqvist A, Rogers DE, Word RA. Transforming growth factor β1 (TGFβ1) and progesterone regulate matrix metalloproteinases (MMP) in human endometrial stromal cells. J Clin Endocrinol Metab. 2012;97(6):888–97.
- Labrie, F. Intracrinology. Mol Cell Endocrinol. 1991;78(3):13–8.
- Gibson DA, Simitsidellis I, Collins F, Saunders PTK. Endometrial intracrinology: Oestrogens, androgens and endometrial disorders. Int J Mol Sci. 2018;19(10):3276.
- Alanazi, S. Recent Advances in Liquid Chromatography–Mass Spectrometry (LC–MS) Applications in Biological and Applied Sciences. Anal Sci Adv. 2025;6(1):1–12.
- Huhtinen K, Desai R, Stah̊le M, Salminen A, Handelsman DJ, Perheentupa A, et al. Endometrial and endometriotic concentrations of estrone and estradiol are determined by local metabolism rather than circulating levels. J Clin Endocrinol Metab. 2012;97(11):4228–35.
- Huhtinen K, Saloniemi-Heinonen T, Keski-Rahkonen P, Desai R, Laajala D, Stahle M, et al. Intra-tissue steroid profiling indicates differential progesterone and testosterone metabolism in the endometrium and endometriosis lesions. J Clin Endocrinol Metab. 2014;99(11):E2188–97.
- Cloke B, Huhtinen K, Fusi L, Kajihara T, Yliheikkilä M, Ho KK, et al. The androgen and progesterone receptors regulate distinct gene networks and cellular functions in decidualizing endometrium. Endocrinology. 2008;149(9):4462–74.
- Ishikawa T, Harada T, Kubota T, Aso T. Testosterone inhibits matrix metalloproteinase-1 production in human endometrial stromal cells in vitro. Reproduction. 2007;133(6):1233–9.
- Grzechocińska B, Dabrowski F, Cyganek A, Panek G, Wielgoś M. The role of metalloproteinases in endometrial remodelling during menstrual cycle. Ginekol Pol. 2017;88(6):337–42.
- Staun-Ram E, Goldman S, Gabarin D, Shalev E. Expression and importance of matrix metalloproteinase 2 and 9 (MMP-2 and -9) in human trophoblast invasion. Reprod Biol Endocrinol. 2004;2:1–13.
- Zhou X, Wu Y, Zhu Z, Lu C, Zhang C, Zeng L, et al. Mucosal immune response in biology, disease prevention and treatment. Signal Transduct Target Ther [Internet]. 2025;10(1):1–32. [CrossRef]
- Silva-sanchez A, Randall TD. Anatomical Uniqueness of the Mucosal for the Induction and Regulation of Mucosal Immunity and Tolerance [Internet]. Mucosal Vaccines. Elsevier Inc.; 2020. 19–54 p. [CrossRef]
- Kim S, Ndwandwe C, Devotta H, Kareem L, Yao L, O’Mahony L. Role of the microbiome in regulation of the immune system. Allergol Int [Internet]. 2025;74(2):187–96. [CrossRef]
- Lunjani N, Ahearn-Ford S, Dube FS, Hlela C, O’Mahony L. Mechanisms of microbe-immune system dialogue within the skin. Genes Immun [Internet]. 2021;22(5–6):276–88. [CrossRef]
- Park, JC. Chang, L. Kwon, HK. Im S. Beyond the gut: decoding the gut–immune–brain axis in health and disease. Cell Mol Immunol [Internet]. 2025;14 August(On-line ahead of print). [CrossRef]
- Choden T, Cohen NA. The gut microbiome and the immune system. Explor Med. 2022;3(3):219–33.
- Parker J, O’Brien C, Hawrelak J. A narrative review of the role of gastrointestinal dysbiosis in the pathogenesis of polycystic ovary syndrome. Obstet Gynecol Sci. 2021;Epub ahead():1–15.
- Tremellen K, Pearce K. Dysbiosis of Gut Microbiota (DOGMA) - A novel theory for the development of Polycystic Ovarian Syndrome. Med Hypotheses [Internet]. 2012;79(1):104–12. [CrossRef]
- Colonetti T, Limas Carmo Teixeira D, Grande AJ, Rodrigues Uggioni ML, Generoso J, Harding S, et al. The role of intestinal microbiota on pre-eclampsia: Systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2023;291(April):49–58.
- Ishimwe, JA. Maternal microbiome in preeclampsia pathophysiology and implications on offspring health. Physiol Rep. 2021;9(10):1–19.
- Medina-Bastidas D, Camacho-Arroyo I G-GE. Current findings in endometrial microbiome: impact on uterine diseases. Reproduction. 2022;163(5):R81–96.
- Monin L, Whettlock EM, Male V. Immune responses in the human female reproductive tract. Immunology. 2020;160(2):106–15.
- Bister J, Crona Guterstam Y, Strunz B, Dumitrescu B, Haij Bhattarai K, Özenci V, et al. Human endometrial MAIT cells are transiently tissue resident and respond to Neisseria gonorrhoeae. Mucosal Immunol. 2021;14(2):357–65.
- Wira CR, Rodriguez-Garcia M, Patel M V. The role of sex hormones in immune protection of the female reproductive tract. Nat Rev Immunol. 2015;15(4):217–30.
- Chatterjee SK, Saha S, Munoz MNM. Activation of mucosal immunity and novel prophylactic and therapeutic strategy in combating COVID-19. Explor Immunol. 2021;1(5):374–97.
- Evans J, Salamonsen LA. Inflammation, leukocytes and menstruation. Rev Endocr Metab Disord. 2012;13(4):277–88.
- Azlan A, Salamonsen LA, Hutchison J, Evans J. Endometrial inflammasome activation accompanies menstruation and may have implications for systemic inflammatory events of the menstrual cycle. Hum Reprod. 2020;35(6):1363–76.
- Maybin JA, Critchley HOD. Menstrual physiology: Implications for endometrial pathology and beyond. Hum Reprod Update. 2015;21(6):748–61.
- Grzybowska ME, Barcikowska Z. Inflammatory Markers in Dysmenorrhea and Therapeutic Options. Int J Environ Res Public Health. 2020;17(1991):1–14.
- Critchley HOD, Maybin JA, Armstrong GM, Williams ARW. Physiology of the endometrium and regulation of menstruation. Physiol Rev. 2020;100(3):1149–79.
- Maybin JA, Murray AA, Saunders PTK, Hirani N, Carmeliet P, Critchley HOD. Hypoxia and hypoxia inducible factor-1α are required for normal endometrial repair during menstruation. Nat Commun [Internet]. 2018;9(1). [CrossRef]
- Davies J, Kadir RA. Endometrial haemostasis and menstruation. Rev Endocr Metab Disord. 2012;13(4):289–99.
- Gelety TJ, Chaudhuri G. Haemostatic mechanism in the endometrium: role of cyclo-oxygenase products and coagulation factors. Br J Pharmacol. 1995;114(5):975–80.
- Cousins FL, Filby CE, Gargett CE. Endometrial Stem/Progenitor Cells–Their Role in Endometrial Repair and Regeneration. Front Reprod Heal. 2021;3(January):1–14.
- Gargett CE, Nguyen HPT, Ye L. Endometrial regeneration and endometrial stem/progenitor cells. Rev Endocr Metab Disord. 2012;13(4):235–51.
- Kong Y, Shao Y, Ren C, Yang G. Endometrial stem/progenitor cells and their roles in immunity, clinical application, and endometriosis. Stem Cell Res Ther [Internet]. 2021;12(1):1–16. [CrossRef]
- Nair R, Agarwal P, Gadre MA, Vasanthan KS, Seetharam RN. Stem cell treatments for female reproductive disorders : a comprehensive review. J Ovarian Res. 2025;18(161):1–27.
- Sarvestani M, Rajabzadeh A, Mazoochi T, Samimi M, Navari M, Moradi F. Use of placental-derived mesenchymal stem cells to restore ovarian function and metabolic profile in a rat model of the polycystic ovarian syndrome. BMC Endocr Disord. 2024;24(1):1–11.
- Chugh RM, Park HS, Esfandyari S, Elsharoud A, Ulin M, Al-Hendy A. Mesenchymal stem cell-conditioned media regulate steroidogenesis and inhibit androgen secretion in a PCOS cell model via BMP-2. Int J Mol Sci. 2021;22(17):1–15.
- Choi JH, Seok J, Lim SM, Kim TH, Kim GJ. Microenvironmental changes induced by placenta-derived mesenchymal stem cells restore ovarian function in ovariectomized rats via activation of the PI3K-FOXO3 pathway. Stem Cell Res Ther. 2020;11(1):1–13.
- Jafarzadeh H, Nazarian H, Ghaffari Novin M, Shams Mofarahe Z, Eini F, Piryaei A. Improvement of oocyte in vitro maturation from mice with polycystic ovary syndrome by human mesenchymal stromal cell–conditioned media. J Cell Biochem. 2018;119(12):10365–75.
- Xie Q, Xiong X, Xiao N, He K, Chen M, Peng J, et al. Mesenchymal Stem Cells Alleviate DHEA-Induced Polycystic Ovary Syndrome (PCOS) by Inhibiting Inflammation in Mice. Stem Cells Int. 2019;Sep 12:201(9782373).
- Ciprietti M, Bueds C, Vankelecom H, Vriens J. Organoids as powerful models of endometrium epithelium in transcriptomic, cellular and functional mimicry. Cell Mol Life Sci. 2025;82(1):1–19.
- Munro MG, Critchley HOD, Fraser IS, Haththotuwa R, Kriplani A, Bahamondes L, et al. The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions. Int J Gynecol Obstet. 2018;143(3):393–408.
- Rodriguez MB, Dias S, Jordan V, Lethaby A, Lensen SF, Wise MR, et al. Interventions for heavy menstrual bleeding; overview of Cochrane reviews and network meta-analysis. Cochrane Database Syst Rev. 2022;5(5):CD013180.
- Pouraliroudbaneh S, Marino J, Riggs E, Saber A, Jayasinghe Y, Peate M. Heavy menstrual bleeding and dysmenorrhea in adolescents: A systematic review of self-management strategies, quality of life, and unmet needs. Int J Gynecol Obstet. 2024;167(1):16–41.
- Munro MG, Critchley HOD, Fraser IS. The FIGO systems for nomenclature and classification of causes of abnormal uterine bleeding in the reproductive years: Who needs them? Am J Obstet Gynecol [Internet]. 2012;207(4):259–65. [CrossRef]
- Comishen KJ, Bhatt M, Yeung K, Irfan J, Zia A, Sidonio RF, et al. Etiology and diagnosis of heavy menstrual bleeding among adolescent and adult patients: a systematic review and meta-analysis of the literature. J Thromb Haemost [Internet]. 2025;23(3):863–76. [CrossRef]
- Cole SK, Billewicz WZ, Thomson AM. Sources of Variation in Menstrual Blood Loss. BJOG An Int J Obstet Gynaecol. 1971;78(10):933–9.
- Sinharoy SS, Chery L, Patrick M, Conrad A, Ramaswamy A, Stephen A, et al. Prevalence of heavy menstrual bleeding and associations with physical health and wellbeing in low-income and middle-income countries: a multinational cross-sectional study. Lancet Glob Heal [Internet]. 2023;11(11):e1775–84. [CrossRef]
- National Institute for Health and Care Excellence (NICE). Heavy Menstrual Bleeding: Assessment and Management [Internet]. [NICE Guideline NG88]: Updated . Available from: http://www.nice.org.uk/guidance/ng88. Accessed 10 August 2025.
- Australian Commission on Safety and Quality in health Care. Heavy Menstrual Bleeding Clinical Care Standard: June 2024. [Internet]. Available from: https://www.safetyandquality.gov.au/standards/clinical-care-standards/heavy-menstrual-bleeding-clinical-care-standard. Accessed.
- Warner P, Harriet L, Whitaker R, Anthony R, John C, Douglas A, et al. Low dose dexamethasone as treatment for women with heavy menstrual bleeding: A response-adaptive randomised placebo-controlled dose-finding parallel group trial ( DexFEM ). EBioMedicine [Internet]. 2021;69:103434. [CrossRef]
- Mardon, AK. White, S. Howe, D. O’Shea, M. Eathorne, A. Gannott, M. Schott, A. Armour M. Problematic Periods Costing Young Women—The Impact of Menstrual Symptoms on Work and Study. ANZJOG. 2024;Dec 19(on line ahead of print.).
- Ponzo S, Wickham A, Bamford R, Radovic T, Zhaunova L, Peven K, et al. Menstrual cycle-associated symptoms and workplace productivity in US employees: A cross-sectional survey of users of the Flo mobile phone app. Digit Heal. 2022;8:1–12.
- Bigambo FM, Wang D, Zhang Y, Mzava SM, Dai R, Wang X. Current situation of menstruation and gynecological diseases prevalence among Chinese women: a cross-sectional study. BMC Womens Health [Internet]. 2022;22(1):1–12. [CrossRef]
- Ashworth, G. Management of heavy menstrual bleeding in Australian general practice An analysis of BEACH data. Aust J Gen Pract. 2021;50(8):573–9.
- Wilson L, Copp T, Hickey M, Jenkinson B, Jordan SJ, Thompson R, et al. Women who experience heavy menstrual bleeding: prevalence and characteristics from young adulthood to midlife, Australia, 2000–2021: a longitudinal cohort survey study. Med J Aust. 2025;222(4):191–7.
- Serhat E, Cogendez E, Selcuk S, Asoglu MR, Arioglu PF, Eren S. Is there a relationship between endometrial polyps and obesity, diabetes mellitus, hypertension? Arch Gynecol Obstet. 2014;290(5):937–41.
- Harmon QE, Patchel S, Denslow S, Wegienka G, Baird DD. Body Mass Index and Uterine Fibroid Development: A Prospective Study. J Clin Endocrinol Metab [Internet]. 2024;109(11):e2016–23. [CrossRef]
- Keizer AL, Semmler A, Kok HS, van Kesteren PJM, Huirne JAF, Hehenkamp WJK. Modifiable prognostic factors in uterine fibroid development: a systematic review of literature. J Obstet Gynaecol (Lahore) [Internet]. 2024;44(1):2288225. [CrossRef]
- Jasper EA, Mautz BS, Hellwege JN, Piekos JA, Jones SH, Zhang Y, et al. A phenome-wide association study of uterine fibroids reveals a marked burden of comorbidities. Commun Med. 2025;5(1):1–15.
- Palomba, S. Is fertility reduced in ovulatory women with polycystic ovary syndrome? An opinion paper. Hum Reprod. 2021;36(9):2421–8.
- Patel, S. Disruption of aromatase homeostasis as the cause of a multiplicity of ailments: A comprehensive review. J Steroid Biochem Mol Biol [Internet]. 2017;168:19–25. [CrossRef]
- Szukiewicz D, Trojanowski S, Kociszewska A, Szewczyk G. Modulation of the Inflammatory Response in Polycystic Ovary Syndrome (PCOS)—Searching for Epigenetic Factors. Int J Mol Sci. 2022;23(14663):1–27.
- Hapangama DK, Bulmer JN. Pathophysiology of heavy menstrual bleeding. Women’s Heal. 2016;12(1):3–13.
- Girling JE, Lockhart MG, Olshansky M, Paiva P, Woodrow N, Marino JL, et al. Differential Gene Expression in Menstrual Endometrium from Women with Self-Reported Heavy Menstrual Bleeding. Reprod Sci. 2017;24(1):28–46.
- Li X, Wang F, Chen M, Ling L, Zhao F, Peng D. The association between endometrial polyps and insulin resistance from the expression of PI3K and AKT proteins perspective. BMC Womens Health [Internet]. 2024;24(366):1–7. [CrossRef]
- Oliva MM, Gambioli R, Forte G, Porcaro G, Aragona C, Unfer V. Unopposed estrogens: current and future perspectives. Eur Rev Med Pharmacol Sci. 2022;26(8):2975–89.
- Nijkang NP, Anderson L, Markham R, Manconi F. Endometrial polyps: Pathogenesis, sequelae and treatment. SAGE Open Med. 2019;7:1–12.
- Zhong⋅ X, Li⋅ Y, Liang W, Hu Q, Zeng A, Ding M, et al. Clinical and metabolic characteristics of endometrial lesions in polycystic ovary syndrome at reproductive age. BMC Womens Health. 2023;23(1):1–9.
- Cakmak H, Taylor HS. Implantation failure: Molecular mechanisms and clinical treatment. Hum Reprod Update. 2011;17(2):242–53.
- Yusuf M, Amri MF, Ugusman A, Hamid AA, Wahab NA, Mokhtar MH. Hyperandrogenism and Its Possible Effects on Endometrial Receptivity : A Review. int J Mol Sci. 2023;24:12026.
- Salamonsen LA, Dimitriadis E. Infertility and the Endometrium. Clin Exp Obstet Gynecol. 2022;49(9):1–10.
- Skliutė G, Baušytė R, Ramašauskaitė D, Navakauskienė R. Characterization of Epigenetic and Molecular Factors in Endometrium of Females with Infertility. Biomedicines. 2022;10(6):1–12.
- Khodaei MM, Noori Z, Zare F, Meshkin A. Ferroptosis and recurrent miscarriage: a critical review of pathophysiology and emerging therapeutic targets. Front Cell Dev Biol. 2025;13:1559300.
- Gusarova TA, Nizyaeva N V., Mikhalev SA, Tikhonova NB, Orgadeeva DA, Mikhaleva LM, et al. Morphological and molecular features of decidual endometrial cells in miscarriage. Morphology. 2023;161(1):37–49.
- Muter J, Kong CS, Nebot MT, Tryfonos M, Vrljicak P, Brighton PJ, et al. Stalling of the endometrial decidual reaction determines the recurrence risk of miscarriage. Sci Adv. 2025;11(26):1–14.
- Bacon SJ, Zhu Y, Ghosh P. Early spiral arteriole remodeling in the uterine–placental interface: A rat model. J Anat. 2024;244(6):1054–66.
- Elawad T, Scott G, Bone JN, Elwell H, Lopez CE, Filippi V, et al. Risk factors for pre-eclampsia in clinical practice guidelines: Comparison with the evidence. BJOG An Int J Obstet Gynaecol. 2022;131(1):46–62.
- Kibret KT, Chojenta C, Gresham E, Tegegne TK, Loxton D. Maternal dietary patterns and risk of adverse pregnancy (hypertensive disorders of pregnancy and gestational diabetes mellitus) and birth (preterm birth and low birth weight) outcomes: A systematic review and meta-analysis. Public Health Nutr. 2019;22(3):506–20.
- Paula WO, Patriota ESO, Gonçalves VSS, Pizato N. Maternal Consumption of Ultra-Processed Foods-Rich Diet and Perinatal Outcomes: A Systematic Review and Meta-Analysis. Nutrients. 2022;14:3242.
- Sanderson PA, Critchley HOD, Williams ARW, Arends MJ, Saunders PTK. New concepts for an old problem: The diagnosis of endometrial hyperplasia. Hum Reprod Update. 2017;23(2):232–54.
- Damor, CB. Damor P. Molecular Profiling of Endometrial Hyperplasia and its Progression to Endometrial Carcinoma: A Histopathological and Genomic Correlation Study. Int J Life Sci Biotechnol Pharma Res [Internet]. 2025;14(6):150–3. Available from: https://www.ijlbpr.com/uploadfiles/28vol14issue6pp150-153.20250607043048.
- Bostan IS, Mihaila M, Roman V, Radu N, Neagu MT, Bostan M, et al. Landscape of Endometrial Cancer: Molecular Mechanisms, Biomarkers, and Target Therapy. Cancers (Basel). 2024;16(11):1–24.
- Balhara N, Yadav R, Chauhan MB. Role of signaling pathways in endometrial cancer. Mol Biol Rep. 2025;52(1):1–18.
- Brower M, Brennan K, Pall M, Azziz R. The severity of menstrual dysfunction as a predictor of insulin resistance in PCOS. J Clin Endocrinol Metab. 2013;98(12):E1967–71.
- Li X, Yang D, Pan P, Azziz R, Yang D. The Degree of Menstrual Disturbance Is Associated With the Severity of Insulin Resistance in PCOS. Front Endo. 2022;13:873726.
- Ezeh, U. Ezeh, C. Pisarska, MD. Azziz R. Menstrual dysfunction in polycystic ovary syndrome: association with dynamic state insulin resistance rather than hyperandrogenism. Fertil Steril [Internet]. 2021;115(6):1557–68. [CrossRef]
- Salcido, AC, Shehata, H. Berry, A. Riba C. Insulin resistance and other risk factors of cardiovascular disease amongst women with abnormal uterine bleeding. J Insul Resist [Internet]. 2022;5(1):1–7. [CrossRef]
- Solomon CG, Hu FB, Dunaif A, Rich-edwards J, Hunter DJ, Colditz GA, et al. Long or Highly Irregular Menstrual Cycles as a Marker for Risk of Type 2 Diabetes Mellitus. Jama. 2001;286(19):2421–6.
- Roumain, J. Charles, M. A. Courten, M. P. D. Hanson, R. L. Brodie, T. D. Pettitt, D. J. Knowler WC. The Relationship of Menstrual Irregularity to Type 2 Diabetes in Pima Indian Women. Diabetes Care. 1998;21(3):1–4.
- Strowitzki T, Capp E, Eye H Von. The degree of cycle irregularity correlates with the grade of endocrine and metabolic disorders in PCOS patients. Eur J Obstet Gynecol [Internet]. 2010;149(2):178–81. [CrossRef]
- Cooper GS, Ephross SA, Sandler DP. Menstrual patterns and risk of adult-onset diabetes mellitus ଝ. J Clin Epidemiol. 2000;53:1170–3.
- Wang Y, Shan Z, Arvizu M, Pan A, Manson JE, Missmer SA. Associations of Menstrual Cycle Characteristics Across the Reproductive Life Span and Lifestyle Factors With Risk of Type 2 Diabetes. JAMA Netw Open. 2020;3(12):e2027928.
- Wang YX, Shan Z, Arvizu M, Pan A, Manson JAE, Missmer SA, et al. Associations of Menstrual Cycle Characteristics Across the Reproductive Life Span and Lifestyle Factors With Risk of Type 2 Diabetes. JAMA Netw open. 2020;3(12):e2027928.
- Foryś E, Baran A, Dziurdzia A, Jarosz-wójcik E, Matusik P, Gawlik A, et al. Are menstrual disorders in adolescent girls related to metabolic disorders? Paediatr Endocrinol Diabetes, Metab [Internet]. 2023;29(2):75–82. [CrossRef]
- Onalan R, Onalan G, Tonguc E, Ozdener T. Body mass index is an independent risk factor for the development of endometrial polyps in patients undergoing in vitro fertilization. Fertil Steril. 2009;91(4):1056–60.
- Kacalska-Janssen O, Rajtar-Ciosek A, Zmaczyński A, Wyroba J, Milewicz T, Krzyczkowska-Sendrakowska M KJ. Markers of insulin resistance in perimenopausal women with endometrial pathology. Ginekol Pol. 2013;84(11):922–9.
- He Y, Zeng Q, Li X, Liu B, Wang P. The Association between Subclinical Atherosclerosis and Uterine Fibroids. PLoS One. 2013;8(2):1–10.
- Salcedo AC, Yun J, Carter C, Hart E. Therapeutic Carbohydrate Restriction as a Metabolic Modality for the Prevention and Treatment of Abnormal Uterine Bleeding. Nutrients [Internet]. 2023;15:3760. [CrossRef]
- Uimari O, Auvinen J, Jokelainen J, Puukka K, Ruokonen A, Järvelin M, et al. Uterine fibroids and cardiovascular risk. Hum Reprod. 2016;31(12):2689–703.
- Laughlin-tommaso SK, Fuchs EL, Wellons MF, Lewis CE, Calderon-margalit R, Stewart EA, et al. Uterine Fibroids and the Risk of Cardiovascular Disease in the Coronary Artery Risk Development in Young Adult Women ’s Study. J Women’s. 2019;28(1):46–52.
- Final Annual Report RCOG. NHMBA. Final Annual Report: National Heavy Menstrual Bleeding Audit [Internet]. 2014. Available from: https://www.hqip.org.uk/wp-content/uploads/2018/02/HwNYNM.
- Soldani R, Cagnacci A, Yen SSC. Insulin insulin-like growth factor I (IGF-I) and IGF-II enhance basal and gonadotrophin-releasing hormone-stimulated luteinizing hormone release from rat anterior pituitary cells in vitro. Eur J Endocrinol. 1994;131(6):641–5.
- Sliwowska JH, Fergani C, Gawałek M, Skowronska B, Fichna P, Lehman MN. Insulin: Its role in the central control of reproduction. Physiol Behav. 2014;133:197–206.
- Nestler JE, Jakubowicz DJ, De Vargas AF, Brik C, Quintero N, Medina F. Insulin stimulates testosterone biosynthesis by human thecal cells from women with polycystic ovary syndrome by activating its own receptor and using inositolglycan mediators as the signal transduction system. J Clin Endocrinol Metab. 1998;83(6):2001–5.
- Baillargeon JP, Nestler JE. Commentary: Polycystic ovary syndrome: A syndrome of ovarian hypersensitivity to insulin? J Clin Endocrinol Metab. 2006;91(1):22–4.
- S Rice, A Khalid, S Khan, M Lacey RH. The effect of hyperinsulinemia on FSH-mediated signalling pathways in granulosa cells - implications for follicle growth in women with PCOS. Hum Reprod [Internet]. 2023;38(Supplement-1):P-643. [CrossRef]
- Nelson-degrave VL, Wickenheisser JK, Hendricks KL, Asano T, Fujishiro M, Legro RS, et al. Alterations in Mitogen-Activated Protein Kinase Kinase and Extracellular Regulated Kinase Signaling in Theca Cells Contribute to Excessive Androgen Production in Polycystic Ovary Syndrome. Mol Endocrinol. 2015;19(2):379–90.
- Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: An update on mechanisms and implications. Endocr Rev. 2012;33(6):981–1030.
- Maclean JA, Hayashi K. Progesterone Actions and Resistance in Gynecological Disorders. Cells. 2022;11:647.
- Garg D, Merhi Z. Relationship between Advanced Glycation End Products and Steroidogenesis in PCOS. Reprod Biol Endocrinol [Internet]. 2016;14(1):1–13. [CrossRef]
- Zuo T, Zhu M, Xu W. Roles of oxidative stress in polycystic ovary syndrome and cancers. Oxid Med Cell Longev. 2016;2016:8589318.
- Zheng X, Pan X, Zhang J, Cao X. Hyperinsulinemia-induced PAX6 expression promotes endometrial epithelial cell proliferation via negatively modulating p27 signaling. Biomed Pharmacother [Internet]. 2018;97(97):802–8. [CrossRef]
- Maybin JA, Critchley HOD, Jabbour HN. Inflammatory pathways in endometrial disorders. Mol Cell Endocrinol [Internet]. 2011;335(1):42–51. [CrossRef]
- Matsuyama S, Whiteside S, Li SY. Implantation and Decidualization in PCOS: Unraveling the Complexities of Pregnancy. Int J Mol Sci. 2024;25:1203.
- Bremer AA, Miller WL. The serine phosphorylation hypothesis of polycystic ovary syndrome: a unifying mechanism for hyperandrogenemia and insulin resistance. Fertil Steril. 2008;89(5):1039–48.
- Lee M, Yoon J, Kim H, Kim YS, Lyu SW, Lee BS. Hyperandrogenic Milieu Dysregulates the Expression of Insulin Signaling Factors and Glucose Transporters in the Endometrium of Patients with Polycystic Ovary Syndrome. Reprod Sci. 2020;27(8):1637–47.
- Nagao H, Cai W, Wewer NJ, Steger M, Batista TM, Pan H. Distinct signaling by insulin and IGF-1 receptors and their extra- and intracellular domains. PNAS. 2021;118(17):e2019474118.
- Wang Y, Hua S, Tian W, Zhang L, Zhao J, Zhang H, et al. Mitogenic and anti-apoptotic effects of insulin in endometrial cancer are phosphatidylinositol 3-kinase/Akt dependent. Gynecol Oncol [Internet]. 2012;125(3):734–41. [CrossRef]
- Jiang N, Zhao W, Shen H, Du D, Li X. Hyperinsulinemia impairs decidualization via AKT-NR4A1 signaling: new insight into polycystic ovary syndrome ( PCOS ) -related infertility. J Ovarian Res [Internet]. 2024;17(1):31. [CrossRef]
- Kooijman, R. Regulation of apoptosis by insulin-like growth factor ( IGF ) -I. Cytokine Growth Factor Rev. 2006;17:305–23.
- Foley, PJ. Effect of low carbohydrate diets on insulin resistance and the metabolic syndrome. Curr Opin Endocrinol Diabetes Obes. 2021;28(5):463–8.
- Wolosowicz M, Prokopiuk S, Kaminski TW. Recent Advances in the Treatment of Insulin Resistance Targeting Molecular and Metabolic Pathways: Fighting a Losing Battle? Med. 2022;58:472.
- Robinson S, Kiddy D, Gelding S V., Willis D, Niththyananthan R, Bush A, et al. The relationship of insulin insensitivity to menstrual pattern in women with hyperandrogenism and polycystic ovaries. Clin Endocrinol (Oxf). 1993;39(3):351–5.
- Niu J, Lu M, Liu B. Association between insulin resistance and abnormal menstrual cycle in Chinese patients with polycystic ovary syndrome. J Ovarian Res [Internet]. 2023;16(1):1–11. [CrossRef]
- Dey R, Bhattacharya K, Basak AK, Paul N, Bandyopadhyay R, Chaudhuri GR, et al. Inflammatory perspectives of polycystic ovary syndrome: role of specific mediators and markers. Middle East Fertil Soc J [Internet]. 2023;28(33):1–12. [CrossRef]
- Vasyukova E, Zaikova E, Kalinina O, Gorelova I, Pyanova I, Bogatyreva E, et al. Inflammatory and Anti-Inflammatory Parameters in PCOS Patients Depending on Body Mass Index: A Case-Control Study. Biomedicines. 2023;11(10):1–13.
- Zhai Y, Pang Y. Systemic and ovarian inflammation in women with polycystic ovary syndrome. J Reprod Immunol [Internet]. 2022;151(April):103628. [CrossRef]
- Ye Z, Zhao J, Li R. Effects of immune cells and cytokines on the endometrial immune microenvironment in polycystic ovary syndrome. Gynecol Obstet Clin Med [Internet]. 2022;2(4):181–5. [CrossRef]
- Wang J, Yin T, Liu S. Dysregulation of immune response in PCOS organ system. Front Immunol. 2023;14(May):1–13.
- Liu S, Hong L, Mo M, Xiao S, Chen C, Li Y, et al. Evaluation of endometrial immune status of polycystic ovary syndrome. J Reprod Immunol [Internet]. 2021;144(20):103282. [CrossRef]
- Smith OPM, Jabbour HN, Critchley HOD. Cyclooxygenase enzyme expression and E series prostaglandin receptor signalling are enhanced in heavy menstruation. Hum Reprod. 2007;22(5):1450–6.
- Milne SA, Perchick GB, Boddy SC, Jabbour HN. Expression, Localization, and Signaling of PGE2 and EP2/EP4 Receptors in Human Nonpregnant Endometrium across the Menstrual Cycle. J Clin Endocrinol Metab. 2015;86(October):4453–9.
- Eijkeren MA Van, Christiaens GCML. Effects of mefenamic acid on menstrual hemostasis in essential menorrhagia. Am J Obstet Gynecol [Internet]. 1992;166(5):1419–28. [CrossRef]
- Khajehei M, Abdali K, Tabatabaee H. The effect of mefenamic acid and naproxen on heavy menstrual bleeding : A placebo-controlled study. South African J Obstet Gynecolgy. 2013;19(2):31–4.
- Rees MCP, Bernal SL, Ca R, Turnbull AC, Hospital JR, Ox O. Effect of Fenamates on Prostaglandin E Receptor Binding. Lancet. 1988;September:541–2.
- Shetty SS, Deepthi D, Harshitha S, Sonkusare S, Naik PB, N SK, et al. Environmental pollutants and their effects on human health. Heliyon [Internet]. 2023;9(9):e19496. [CrossRef]
- Stegehuis N, Kotsirilos V, Parker J. The Impact of Microparticulate Air Pollution in Polycystic Ovary Syndrome: A Narrative Review. Clin Exp Obstet Gynecol. 2024;51(10):233.
- Parker, J. O’Brien C, Hawrelak J. A narrative review of the role of gastrointestinal dysbiosis in the pathogenesis of polycystic ovary syndrome. Obstet Gynecol Sci. 2022;65(1):14–28.
- Ameho S, Klutstein M. The effect of chronic inflammation on female fertility. Reproduction [Internet]. 2025;169(e240197). [CrossRef]
- Drizi A, Djokovic D, Laganà AS, Herendael B Van. Impaired inflammatory state of the endometrium: A multifaceted approach to endometrial inflammation. Current insights and future directions. Menopause Rev. 2020;19(2):90–100.
- Palomba S, Daolio J, La Sala GB. Oocyte Competence in Women with Polycystic Ovary Syndrome. Trends Endocrinol Metab [Internet]. 2017;28(3):186–98. [CrossRef]
- Piltonen, TT. Polycystic ovary syndrome: Endometrial markers. Best Pract Res Clin Obstet Gynaecol [Internet]. 2016;37:66–79. [CrossRef]
- Quezada S, Avellaira C, Johnson MC, Gabler F, Fuentes A, Vega M. Evaluation of steroid receptors, coregulators, and molecules associated with uterine receptivity in secretory endometria from untreated women with polycystic ovary syndrome. Fertil Steril [Internet]. 2006;85(4):1017–26. [CrossRef]
- Li X, Feng Y, Lin JF, Billig H, Shao R. Endometrial progesterone resistance and PCOS. J Biomed Sci. 2014;21(1):1–7.
- Marquardt RM, Kim TH, Shin JH, Jeong JW. Progesterone and estrogen signaling in the endometrium: What goes wrong in endometriosis? Int J Mol Sci. 2019;20(15):1–28.
- van der Horst PH, Wang Y, van der Zee M, Burger CW, Blok LJ. Interaction between sex hormones and WNT/β-catenin signal transduction in endometrial physiology and disease. Mol Cell Endocrinol [Internet]. 2012;358(2):176–84. [CrossRef]
- Gellersen B, Brosens JJ. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr Rev. 2014;35(6):851–905.
- Salamun V, Rizzo M, Lovrecic L, Hocevar K, Papler Burnik T, Janez A, Jensterle M, Vrtacnik Bokal E, Peterlin B MA. The Endometrial Transcriptome of Metabolic and Inflammatory Pathways During the Window of Implantation Is Deranged in Infertile Obese Polycystic Ovarian Syndrome Women. Metab Syndr Relat Disord. 2022;20(7):384–94.
- Savaris RF, Groll JM, Young SL, DeMayo FJ, Jeong JW, Hamilton AE, et al. Progesterone resistance in PCOS endometrium: A microarray analysis in clomiphene citrate-treated and artificial menstrual cycles. J Clin Endocrinol Metab. 2011;96(6):1737–46.
- Das, SK. Regional development of uterine decidualization: Molecular signaling by Hoxa-10. Mol Reprod Dev. 2010;77(5):387–96.
- Cermik D, Selam B, Taylor HS. Regulation of HOXA-10 expression by testosterone in vitro and in the endometrium of patients with polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88(1):238–43.
- Ali R, Ahmed T, Gul H, Rehman R. An interplay of Progesterone, Leukemia Inhibitor Factor and Interleukin-6 in the window of implantation ; Impact on fertility. Cytokine [Internet]. 2023;170(May):156332. [CrossRef]
- Ujvari D, Hulchiy M, Calaby A, Nybacka A, Byström B, Hirschberg AL. Lifestyle intervention up-regulates gene and protein levels of molecules involved in insulin signaling in the endometrium of overweight/obese women with polycystic ovary syndrome. Hum Reprod. 2014;29(7):1526–35.
- Hagenfeldt, K. The role of prostaglandins and allied substances in uterine haemostasis. Contraception. 1987;36(1):23–35.
- Smith SK, Abel MH, Kelly RW, Baird DT. Prostaglandin Synthesis in the Endometrium of Women with Ovulatory Dysfunctional Uterine Bleeding. Brithish J Obstet Gynaecol. 1981;88(April):434–432.
- Marsh MM, Malakooti N, Taylor NH. Endothelin and neutral endopeptidase in the endometrium of women with menorrhagia. Hum Reprod [Internet]. 1997;12(9):2036–40. [CrossRef]
- Abberton KM, Healy DL, Rogers PAW. Smooth muscle alpha actin and myosin heavy chain expression in the vascular smooth muscle cells surrounding human endometrial arterioles. Hum Reprod [Internet]. 1999;14(12):3095–100. [CrossRef]
- Abberton KM, Taylor NH, Healy DL, Rogers PAW. Vascular smooth muscle cell proliferation in arterioles of the human endometrium. Hum Reprod. 1999;14(4):1072–9.
- Gleeson, N. Devitt, M. Sheppard, BL. Bonnar N. Endometrial fibrinolytic enzymes in women with normal menstruation and dysfunctional uterine bleeding. Br J Obstet Gynaecol. 1993;100(8):768–71.
- Sylus AM, Nandeesha H, Chitra T. Matrix metalloproteinase-9 increases and interleukin-10 reduces with increase in body mass index in polycystic ovary syndrome: A cross-sectional study. Int J Reprod Biomed. 2020;18(8):605–10.
- Middelkoop MA, Don EE, Hehenkamp WJK, Polman NJ, Griffioen AW, Huirne JAF. Angiogenesis in abnormal uterine bleeding: a narrative review. Hum Reprod Update. 2023;29(4):457–85.
- Critchley HOD, Maybin JA, Ch MBB. Molecular and Cellular Causes of Abnormal Uterine Bleeding of Endometrial Origin. Semin Reprod Med. 2011;29(5):400–9.
- Rae M, Mohamad A, Price D, Hadoke PWF, Walker BR, Mason JI, et al. Cortisol Inactivation by 11β-Hydroxysteroid dehydrogenase-2 May Enhance Endometrial Angiogenesis via Reduced Thrombospondin-1 in Heavy Menstruation. J Clin Endocrinol Metab. 2009;94(4):1443–50.
- Villavicencio A, Bacallao K, Avellaira C, Gabler F, Fuentes A, Vega M. Androgen and estrogen receptors and co-regulators levels in endometria from patients with polycystic ovarian syndrome with and without endometrial hyperplasia. Gynecol Oncolo. 2006;103:307–14.
- Guo F, Huang Y, Fernando T, Shi Y. Altered Molecular Pathways and Biomarkers of Endometrial Receptivity in Infertile Women with Polycystic Ovary Syndrome. Reprod Sci [Internet]. 2022;29:3335–45. [CrossRef]
- Koc O, Ozdemirici S, Acet M, Soyturk U. Nuclear factor-κB expression in the endometrium of normal and overweight women with polycystic ovary syndrome. J Obstet Gynaecol (Lahore) [Internet]. 2017;37(7):924–30. [CrossRef]
- Younas K, Quintela M, Thomas S, Garcia-Parra J, Blake L, Whiteland H, et al. Delayed endometrial decidualisation in polycystic ovary syndrome; the role of AR-MAGEA11. J Mol Med. 2019;97(9):1315–27.
- James DW, Quintela M, Lucini L, Alkafri NK, Healey GD, Younas K, et al. Homeobox regulator Wilms Tumour 1 is displaced by androgen receptor at cis-regulatory elements in the endometrium of PCOS patients. Front Endocrinol (Lausanne). 2024;15(April):1–13.
- Thibord F, Cunha J, Džigurski J, Tuftin B, Huffman JE, Pujol-Gualdo N, Cho K, Wilson PWF, Johnsen JM, Raffield LM, Chen MH, Laisk T JA. Genome-wide meta-analysis of heavy menstrual bleeding reveals 36 risk loci. Blood. 2025;146(6):745–58.
- Navarro A, Bariani MV, Park HS, Zota AR, Al-Hendy A. Report of Exosomes Isolated from a Human Uterine Leiomyoma Cell Line and Their Impact on Endometrial Vascular Endothelial Cells. Pharmaceuticals. 2022;15(577):1–15.
- Sadler B, Minard CG, Haller G, Gurnett CA, O’Brien SH, Wheeler A, et al. Whole-exome analysis of adolescents with low VWF and heavy menstrual bleeding identifies novel genetic associations. Blood Adv. 2022;6(2):420–8.
- Wang X, Yang D, Peng H. Female Reproductive Tract Organoids: Applications from Physiology to Pathology. Biomolecules. 2025;15:925.
- Zhao X, Jiang Y, Xi H, Chen L, Feng X. Exploration of the Relationship between Gut Microbiota and Polycystic Ovary Syndrome (PCOS): A Review. Geburtshilfe Frauenheilkd. 2020;80(2):161–71.
- Rizk MG, Thackray VG. Intersection of Polycystic Ovary Syndrome and the Gut Microbiome. J Endocr Soc. 2021;5(2):1–16.
- Hou K, Wu ZX, Chen XY, Wang JQ, Zhang D, Xiao C, et al. Microbiota in health and diseases. Signal Transduct Target Ther. 2022;7(1):1–28.
- Yurtdaş G, Akdevelioğlu Y. A New Approach to Polycystic Ovary Syndrome: The Gut Microbiota. J Am Coll Nutr [Internet]. 2020;39(4):371–82. [CrossRef]
- PARKER JIM, HAWRELAK J, GERSH FL. Nutritional Role of Polyphenols As a Component of a Wholefood Diet in the Management of Polycystic Ovary Syndrome. Australas Coll Nutr Environ Med J [Internet]. 2021;40(2):6–12. Available online: https://search.ebscohost.com/login.aspx?direct=true&db=rzh&AN=154032844&lang=ja&site=ehost-live.
- Parker, J. Hawrelak, J. Gersh F. Nutritional Role of Polyphenols As a Component of a Wholefood Diet in the Management of Polycystic Ovary Syndrome. Australas Coll Nutr Environ Med J [Internet]. 2021;40(2):6–12. Available online: https://search.ebscohost.com/login.aspx?direct=true&db=rzh&AN=154032844&lang=ja&site=ehost-live.
- Tremellen KPK. Nutrition, Fertility, and Human Reproductive Function. In: Nutrition, Fertility, and Human Reproductive Function. Adelaide: CRC Press; 2015. p. 27–50.
- Wu Y, Lin Z, Li C, Lin X, Shan S, Guo B. Epigenetic regulation in metabolic diseases: mechanisms and advances in clinical study. Signal Transduct Target Ther. 2023;8:98.
- Faraji J, Metz GAS. Environmental Epigenetics: New Horizons in Redefining Biological and Health Outcomes. PREPRINT. 2025.
- Scarfo G, Daniele S, Fusi J, Gesi M, Martini C, Franzoni F, et al. Metabolic and Molecular Mechanisms of Diet and Physical Exercise in the Management of Polycystic Ovarian Syndrome. Biomedicines. 2022;10:1305.
- Szczuko M, Kikut J, Szczuko U, Szydłowska I, Nawrocka-rutkowska J, Zi M. Nutrition Strategy and Life Style in Polycystic Ovary Syndrome — Narrative Review. Nutrients. 2021;13:2452.
- Gu Y, Zhou G, Zhou F, Li Y, Wu Q, He H, et al. Gut and Vaginal Microbiomes in PCOS: Implications for Women’s Health. Front Endocrinol (Lausanne). 2022;13(February):1–9.
- Sola-Leyva A, Pérez-Prieto I, Molina NM, Vargas E, Ruiz-Durán S, Leonés-Baños I, et al. Microbial composition across body sites in polycystic ovary syndrome: a systematic review and meta-analysis. Reprod Biomed Online. 2023;47(1):129–50.
- Lin SY, Yang YC, Chang CYY, Lin CC, Hsu WH, Ju SW, et al. Risk of polycystic ovary syndrome in women exposed to fine air pollutants and acidic gases: A nationwide cohort analysis. Int J Environ Res Public Health. 2019;16(23):4816.
- Leceta J, Del Campo R, Jordan S, Klose CSN. Editorial: Immunoregulation at mucosal surfaces. Front Immunol. 2022;13(August):1–5.
- Hampl R, Starka L. Endocrine Disruptors and Gut Microbiome Interactions. Physiol Res. 2020;69(suppl. 2):S211–23.
- Bachmann MC, Bellalta S, Basoalto R, Gómez-Valenzuela F, Jalil Y, Lépez M, et al. The Challenge by Multiple Environmental and Biological Factors Induce Inflammation in Aging: Their Role in the Promotion of Chronic Disease. Front Immunol. 2020;11(October):1–19.
- Thin ZS, Chew J, Yu T, Ong Y, Affendi R, Ali R, et al. Impact of microplastics on the human gut microbiome : a systematic review of microbial composition, diversity, and metabolic disruptions. BMC Gastroenterol. 2025;25(583):1–18.
- Balali H, Morabbi A, Karimian M. Concerning influences of micro/nano plastics on female reproductive health: focusing on cellular and molecular pathways from animal models to human studies. Reprod Biol Endocrinol [Internet]. 2024;22(1):1–35. [CrossRef]
- Sun J, Sui M, Wang T, Teng X, Sun J, Chen M. Detection and quantification of various microplastics in human endometrium based on laser direct infrared spectroscopy. Sci Total Environ [Internet]. 2024;906(23):167760. [CrossRef]
- Kim N, Lee JH, Lee I, Park JH, Jung GS, Lee MJ, et al. Investigation of potential toxic effects of nano- and microplastics on human endometrial stromal cells. Reprod Toxicol [Internet]. 2025;132(24):108848. [CrossRef]
- Qin X, Cao M, Peng T, Shan H, Lian W, Yu Y, et al. Features, Potential Invasion Pathways, and Reproductive Health Risks of Microplastics Detected in Human Uterus. Environ Sci Technol. 2024;58(24):10482–93.
- Gupta N, Yadav VK, Gacem A, Al-Dossari M, Yadav KK, Abd El-Gawaad NS, et al. Deleterious Effect of Air Pollution on Human Microbial Community and Bacterial Flora: A Short Review. Int J Environ Res Public Health. 2022;19(23):1–16.
- Jin Y, Wu S, Zeng Z, Fu Z. Effects of environmental pollutants on gut microbiota. Environ Pollut [Internet]. 2017;222:1–9. [CrossRef]
- Chuang KJ, Chan CC, Su TC, Lee C Te, Tang CS. The effect of urban air pollution on inflammation, oxidative stress, coagulation, and autonomic dysfunction in young adults. Am J Respir Crit Care Med. 2007;176(4):370–6.
- Zhang S, Mwiberi S, Pickford R, Breitner S, Huth C, Koenig W, et al. Longitudinal associations between ambient air pollution and insulin sensitivity: results from the KORA cohort study. Lancet Planet Heal [Internet]. 2021;5(1):e39–49. [CrossRef]
- Li J, Wu Q, Wu XK, Zhou ZM, Fu P, Chen XH, et al. Effect of exposure to second-hand smoke from husbands on biochemical hyperandrogenism, metabolic syndrome and conception rates in women with polycystic ovary syndrome undergoing ovulation induction. Hum Reprod. 2018;33(4):617–25.
- Mahalingaiah S, Missmer SE, Cheng JJ, Chavarro J, Laden F, Hart JE. Perimenarchal air pollution exposure and menstrual disorders. Hum Reprod. 2018;33(3):512–9.
- Liang Y, Lu Q, Chen M, Zhao X, Chu C, Zhang C, et al. Impact of endocrine disrupting chemicals ( EDCs ) on epigenetic regulation in the uterus : a narrative review. Reprod Biol Endocrinol. 2025;23(80):1–23.
- Tricotteaux-Zarqaoui S, Lahimer M, Abou Diwan M, Corona A, Candela P, Cabry R, et al. Endocrine disruptor chemicals exposure and female fertility declining: from pathophysiology to epigenetic risks. Front Public Heal. 2024;12(December):1–12.
- Parent, AS. Damdimopoulou, P. Johansson, HKL. Bouftas, N. Draskau, MK. Franssen, D. Fudvoye, J. van Duursen, M. Svingen T. Endocrine-disrupting chemicals and female reproductive health: a growing concern. Nat Rev Endocrinol [Internet]. 2025;22 May(Online ahead of print). [CrossRef]
- Paramasivam A, Murugan R, Jeraud M, Dakkumadugula A, Periyasamy R, Arjunan S. Additives in Processed Foods as a Potential Source of Endocrine-Disrupting Chemicals: A Review. J Xenobiotics. 2024;14(4):1697–710.
- Qi X, Yun C, Pang Y, Qiao J. The impact of the gut microbiota on the reproductive and metabolic endocrine system. Gut Microbes [Internet]. 2021;13(1):1–22. [CrossRef]
- Ashonibare VJ, Akorede BA, Ashonibare PJ, Akhigbe TM, Akhigbe RE. Gut microbiota-gonadal axis: the impact of gut microbiota on reproductive functions. Front Immunol. 2024;15(February):1–14.
- Moustakli E, Stavros S, Katopodis P, Potiris A, Drakakis P, Dafopoulos S, et al. Gut Microbiome Dysbiosis and Its Impact on Reproductive Health: Mechanisms and Clinical Applications. Metabolites. 2025;15(6):1–15.
- Kong FS, Huang P, Chen JH, Ma Y. The Novel Insight of Gut Microbiota from Mouse Model to Clinical Patients and the Role of NF-κB Pathway in Polycystic Ovary Syndrome. Reprod Sci [Internet]. 2024;31(11):3323–33. [CrossRef]
- Sheldon IM, Roberts MH. Toll-like receptor 4 mediates the response of epithelial and stromal cells to lipopolysaccharide in the endometrium. PLoS One. 2010;5(9):1–10.
- Krikun G, Trezza J, Shaw J, Rahman M, Guller S, Abrahams VM LC. LPS appears to activate human endometrial endothelial cells through TLR-4-dependent and TLR-4-independent mechanisms. Am J Reprod Immunol. 2012;68(3):233–7.
- Yu J, Berga SL, Zou W, Taylor RN. Interleukin-1β inhibits estrogen receptor-α, progesterone receptors A and B and biomarkers of human endometrial stromal cell differentiation: Implications for endometriosis. Mol Hum Reprod. 2019;25(10):625–37.
- Gholizadeh Shamasbi S, Dehgan P, Mohammad-Alizadeh Charandabi S, Aliasgarzadeh A, Mirghafourvand M. The effect of resistant dextrin as a prebiotic on metabolic parameters and androgen level in women with polycystic ovarian syndrome: a randomized, triple-blind, controlled, clinical trial. Eur J Nutr [Internet]. 2019;58(2):629–40. [CrossRef]
- Martinez Guevara D, Vidal Cañas S, Palacios I, Gómez A, Estrada M, Gallego J, et al. Effectiveness of Probiotics, Prebiotics, and Synbiotics in Managing Insulin Resistance and Hormonal Imbalance in Women with Polycystic Ovary Syndrome (PCOS): A Systematic Review of Randomized Clinical Trials. Nutr. 2024;16(22):1–28.
- Kaluanga Bwanga P, Tremblay-Lemoine PL, Timmermans M, Ravet S, Munaut C, Nisolle M, et al. The Endometrial Microbiota: Challenges and Prospects. Med. 2023;59(9):1–10.
- Balla B, Illés A, Tobiás B, Pikó H, Beke A, Sipos M, et al. The Role of the Vaginal and Endometrial Microbiomes in Infertility and Their Impact on Pregnancy Outcomes in Light of Recent Literature. Int J Mol Sci. 2024;25(23):1–23.
- Abdalla W, Nabeel W, Atiyeh I. The role of the microbiome in endometrial carcinoma: Pathogenesis, biomarkers, and therapeutic prospects. J Obs Gynaecol Res. 2025;51:e70070.
- Pelzer ES, Willner D, Buttini M, Huygens F. A role for the endometrial microbiome in dysfunctional menstrual bleeding. Antonie van Leeuwenhoek, Int J Gen Mol Microbiol [Internet]. 2018;111(6):933–43. [CrossRef]
- Stener-Victorin E, Padmanabhan V, Walters KA, Campbell RE, Benrick A, Giacobini P, et al. Animal Models to Understand the Etiology and Pathophysiology of Polycystic Ovary Syndrome. Endocr Rev. 2020;41(4):538–76.
- Hibaoui Y, Feki A. Organoid Models of Human Endometrial Development and Disease. Front Cell Dev Biol. 2020;8(February):1–6.
- Miyazaki K, Dyson MT, Coon V JS, Furukawa Y, Yilmaz BD, Maruyama T, et al. Generation of Progesterone-Responsive Endometrial Stromal Fibroblasts from Human Induced Pluripotent Stem Cells: Role of the WNT/CTNNB1 Pathway. Stem Cell Reports [Internet]. 2018;11(5):1136–55. [CrossRef]
- Gnecco JS, Brown A, Buttrey K, Ives C, Goods BA, Baugh L, et al. Organoid co-culture model of the human endometrium in a fully synthetic extracellular matrix enables the study of epithelial-stromal crosstalk. Med [Internet]. 2023;4(8):554-579.e9. [CrossRef]
- Filby CE, Wyatt KA, Mortlock S, Cousins FL, McKinnon B, Tyson KE, et al. Comparison of organoids from menstrual fluid and hormone-treated endometrium: Novel tools for gynecological research. J Pers Med. 2021;11(12):1314.
- Luyckx L, Wei M, Saarela U, Myllykangas M, Kinnunen J, Arffman R, et al. PCOS endometrium-derived epithelial organoids as a novel model to study endometrial dysfunction. Hum Reprod. 2025;40(8):1535–49.
- Piltonen TT, Chen J, Erikson DW, Spitzer TLB, Barragan F, Rabban JT, et al. Mesenchymal stem/progenitors and other endometrial cell types from women with polycystic ovary syndrome (PCOS) display inflammatory and oncogenic potential. J Clin Endocrinol Metab. 2013;98(9):3765–75.
- Shishehgar F, Ramezani Tehrani F, Mirmiran P, Hajian S, Baghestani AR, Moslehi N. Comparison of Dietary Intake between Polycystic Ovary Syndrome Women and Controls. Glob J Health Sci. 2016;8(9):302.
- Lim SS, Hutchison SK, Van Ryswyk E, Norman RJ, Teede HJ, Moran LJ. Lifestyle changes in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2019;2019(3).
- Benton, ML. The influence of evolutionary history on human health and disease. Nat Rev Genet [Internet]. 2021. [Google Scholar] [CrossRef]
- Gakidou E, Afshin A, Abajobir AA, Abate KH, Abbafati C, Abbas KM, et al. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990-2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1345–422.
- Sabag A, Patten RK, Moreno-Asso A, Colombo GE, Dafauce Bouzo X, Moran LJ, et al. Exercise in the management of polycystic ovary syndrome: A position statement from Exercise and Sports Science Australia. J Sci Med Sport [Internet]. 2024;27(10):668–77. [CrossRef]
- Marsh KA, Steinbeck KS, Atkinson FS, Petocz P, Brand-miller JC. Effect of a low glycemic index compared with a conventional healthy diet on polycystic ovary syndrome. Am J Clin Nutr [Internet]. 2010;92(1):83–92. [CrossRef]
- Moran LJ, Ko H, Misso M, Marsh K, Noakes M, Talbot M, et al. Dietary Composition in the Treatment of Polycystic Ovary Syndrome: A Systematic Review to Inform Evidence-Based Guidelines. J Acad Nutr Diet. 2013;113(4):520–45.
- Medeiros FL, Fernandes AC, Padovan M, Kraemer MV, Bernardo GL, Uggioni PL, et al. The evolution of carbohydrate-restricted diets for type 2 diabetes mellitus: a scoping review. Acad Nutr Diet. 2025;2(2):1–13.
- Nicholas AP, Soto-Mota A, Lambert H, Collins AL. Restricting carbohydrates and calories in the treatment of type 2 diabetes: A systematic review of the effectiveness of “low-carbohydrate” interventions with differing energy levels. J Nutr Sci. 2021;10(e76):1–15.
- Feinman RD, Pogozelski WK, Astrup A, Bernstein RK, Fine EJ, Westman EC, et al. Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition [Internet]. 2015;31(1):1–13. [CrossRef]
- Evert AB, Dennison M, Gardner CD, Timothy Garvey W, Karen Lau KH, MacLeod J, et al. Nutrition therapy for adults with diabetes or prediabetes: A consensus report. Diabetes Care. 2019;42(5):731–54.
- Joseph JJ, Deedwania P, Acharya T, Aguilar D, Bhatt DL, Chyun DA, et al. Comprehensive Management of Cardiovascular Risk Factors for Adults with Type 2 Diabetes: A Scientific Statement from the American Heart Association. Circulation. 2022;145(9):722–59.
- Stranks, Stephen N. Lawlor-Smith L. Managing Type 2 Diabetes with Therapeutic Carbohydrate Reduction [Internet]. 2023. p. 1–9. Available from: https://www.diabetessociety.com.au/wp-content/uploads/2023/11/Managing-Type-2-Diabetes-with-Therapeutic-Carbohydrate-reduction-TCR-November-2023_Final.
- Hite A, Cavan D, Cucuzella M, Cywes R, Ede G. Clinical Guidelines for Therapeutic Carbohydrate Restriction [Internet]. Society of Metabolic Health Practitioners. 2025. p. 1–23. Available from: https://thesmhp.
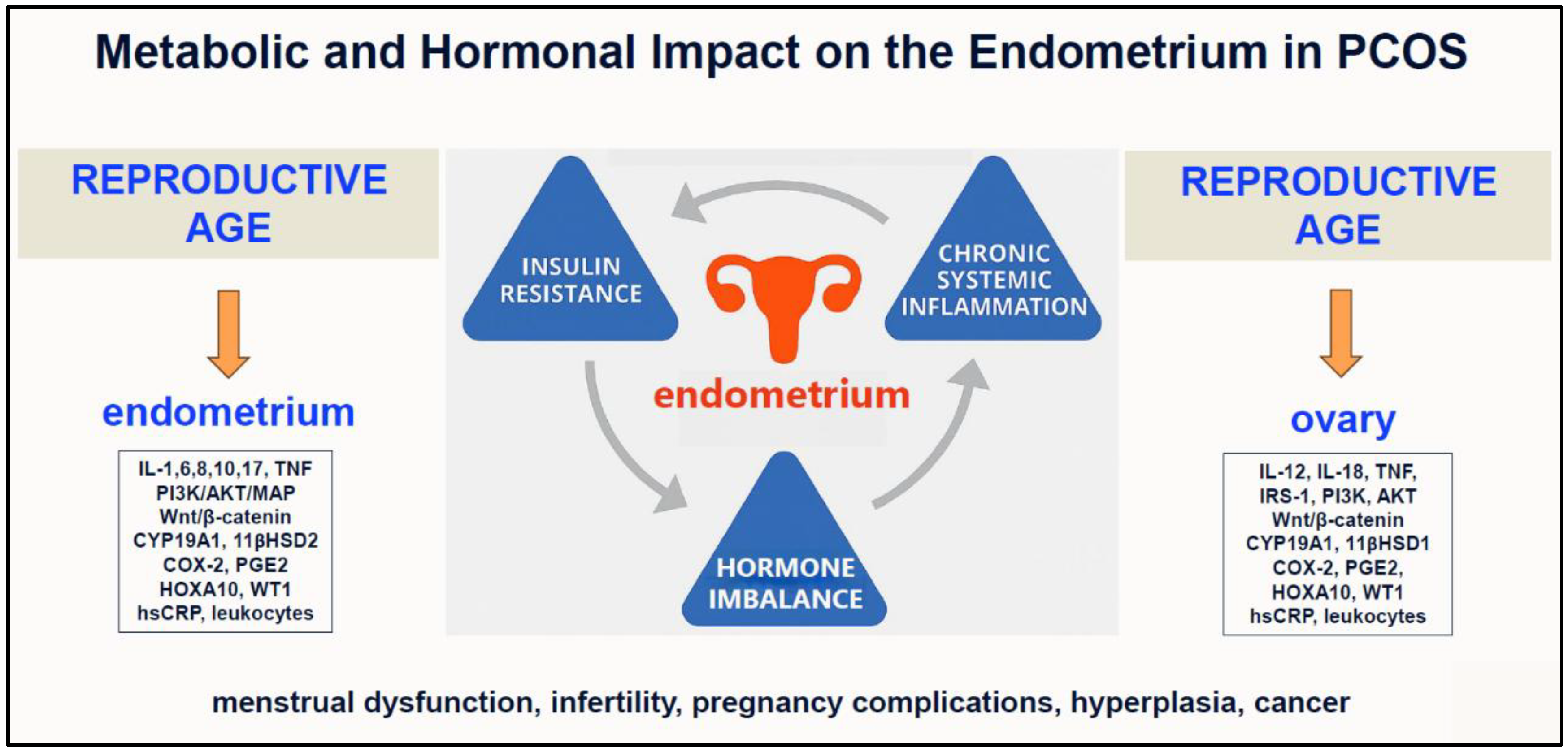
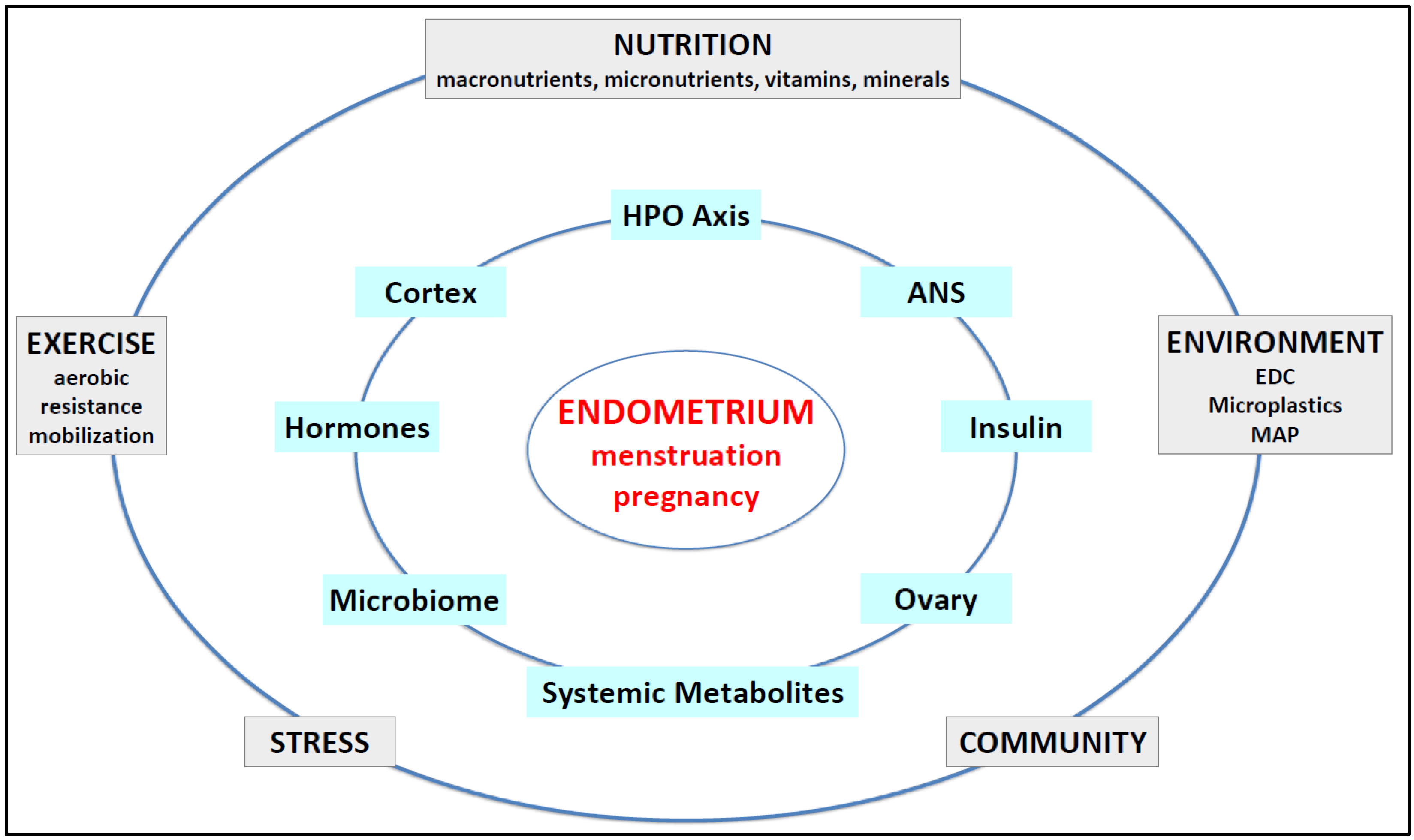
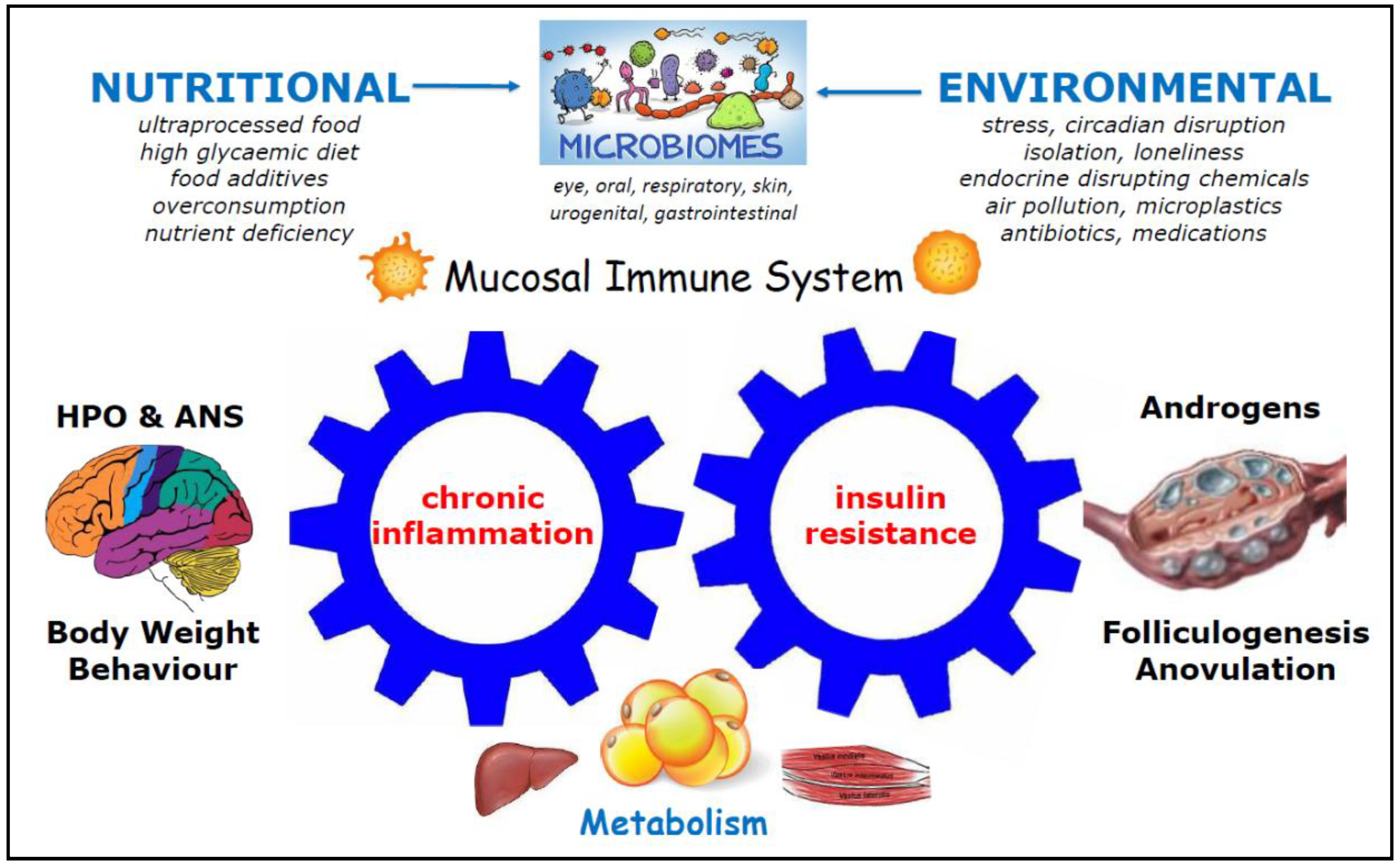
| Endometrial Problem | Pathological Findings and Mechanisms | References |
|---|---|---|
| Heavy menstrual bleeding |
Irregular breakdown of a thickened, hyperplastic endometrium due to unopposed E2 and/or P4 deficiency, vascular fragility, and low-grade inflammation. Alterations in inflammatory mediators, hemostasis, fibrinolysis, tissue and vascular remodeling. |
(122,123) |
| Polyps | Chronic unopposed E2 from anovulatory cycles together with insulin-mediated growth factors (VEGF, TGFβ-1)—and possibly HA—drive excess inflammation and focal endometrial proliferation, cystic glandular changes, stromal fibrosis, and increased vascularity. |
(124–127) |
| Implantation failure |
Defective decidualization, altered epithelium, inadequate spiral artery remodeling, immune cell imbalance, and defective extracellular matrix remodeling. Altered endometrial receptivity markers (reduced LIF, HOXA10, αvβ3 integrin and pinopode formation) driven by HA, IR, inflammation, and obesity. Lifestyle strategies (weight loss, diet, physical activity, circadian alignment) targeting obesity and IR play an important role. |
(19,128,129) |
|
Infertility |
Combined effects of chronic anovulation and impaired endometrial receptivity reduces conception rates. Key drivers include P4 resistance, low LIF/HOXA10/αvβ3-integrin, defective pinopodes, IR, inflammation, and HA. Upregulated genes involving decidualization (HAND2, MUC1, CSF2), angiogenesis (PDGFA), and inflammation (RELA, CXCL10). Altered epigenetic expression of miRNAs. Lifestyle factors, particularly elevated BMI, are significantly associated with infertility. |
(130,131) |
| Miscarriage | Impaired decidualization and trophoblast invasion resulting from P4 resistance, imbalanced cytokines, chronic inflammation, and metabolic dysfunction. Endometrial cells have heightened oxidative stress and dysregulated iron metabolism leading to increased ferroptosis. Depleted antioxidant defenses (impaired glutathione peroxidase 4) compromise endometrial cell viability and placental development. |
(132–134) |
| Pregnancy complications |
Elevated risk of GDM, PE, FGR, PTB, and stillbirth. Placental abnormalities include defective spiral artery remodeling, spiral artery thrombosis, atherosis of basal arterioles, and failure of deep placentation, with co-existing maternal endothelial dysfunction. Underlying maternal CSI, IR, and HA, alter placental physiology and development. Lifestyle factors modify risk of pregnancy complications. |
(13,31,135–138) |
| Hyperplasia | Prolonged estrogen exposure (unopposed by P4), obesity, decreased SHBG, dyslipidemia, elevated FAI, and IR promote abnormal growth of endometrial glands in relation to stroma +/- cytological atypia (EIN). Loss of PTEN expression, PI3K3CA mutations, MMR deficiencies. Modifiable risks include obesity, diet and exercise. |
(122,139,140) |
| Endometrial Cancer |
Progression from untreated atypical hyperplasia under chronic E2 stimulation, compounded by hyperinsulinemia, inflammation, and genetic mutations. Dysregulated signaling pathways (Notch, Wnt/β-catenin, PI3K/AKT/mTOR, MAPK, JAK/STAT, HER2). |
(122,141,142) |
| Study (Year) |
Study Design Population (Country) |
Key Findings | Insulin Resistance Metrics |
Menstrual Cycle Length |
Citation |
|---|---|---|---|---|---|
| Robinson et al. (1993) |
Cross-sectional 72 PCOS 31 Controls (UK) |
↓Insulin sensitivity in PCOS with oligomenorrhea cw controls (p<0.01), but normal in PCOS with eumenorrhea |
IV Insulin Tolerance Test |
>35 days | (182) |
| Strowitzki et al. (2010) |
Cross-sectional 118 HA PCOS (Germany) |
↑HOMA-IR with amenorrhea (4.6) cw eumenorrhea (2.8) (p=0.019) |
HOMA-IR | >35 days | (149) |
| Brower et al. (2013) |
Cross-sectional 494 PCOS 138 Controls (USA) |
Higher mean HOMA-IR (2.2) in PCOS cw controls (1.41), after adjusting for age, BMI, and race |
HOMA-IR fasting insulin |
>35 days | (143) |
| Ezeh et al. (2021) |
Cross-sectional 57 HA PCOS 57 Controls (USA) |
↑Plasma glucose disappearance rate constant (kITT) in amenorrhea (1.98 +/- 0.28) cw eumenorrhea (3.33 +/- 0.51), after adjusting for age, BMI, and ethnicity |
Short Insulin Tolerance Test |
>35 days | (145) |
| Li et al. (2022) |
Cross-sectional 527 PCOS 565 Controls (China) |
↑HOMA-IR, ↑HOMA-β, and ↓QUICKI in women with cycles 45-90 days cw cycle >90 days and controls. No significant difference between cycles <45 and 45-90 days |
HOMA-IR HOMA-β QUICKI |
45-90 days | (144) |
| Niu et al. (2023) |
Retrospective 140 PCOS (China) |
Dose-response relationship between ↑HOMA-IR and cycle length: eumenorrhea (1.61: CI 1.3-1.85), oligomenorrhea (2.02: CI 1.61-2.445) amenorrhea (2.35: CI 1.96-2.75) |
HOMA-IR QUICKI ISI |
>35 days | (183) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




