Submitted:
11 September 2025
Posted:
12 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
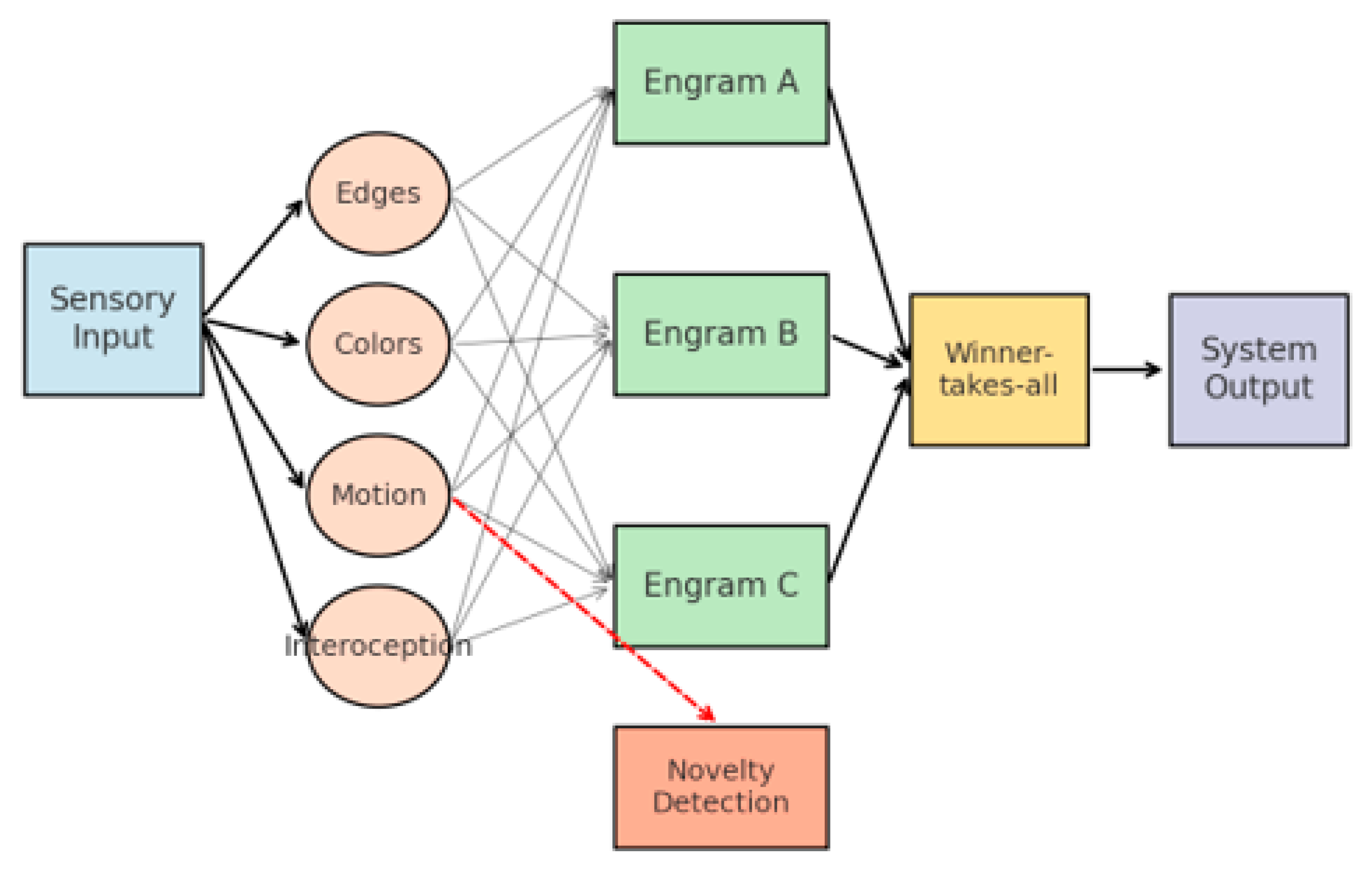
2.1. The Theoretical Framework of the EBPM Model
- Sensory components (e.g., visual edges and colors, auditory timbres, tactile cues),
- Motor components (articular configurations, force vectors),
- Interoceptive and emotional components (visceral state, affective value),
- Cognitive components (associations, abstract rules).
- Stimulus activation – sensory input activates elementary engram units (sub-engrams) such as edges, textures, tones, motor or interoceptive signals.
- Engram co-activation – shared sub-engrams may partially activate multiple parent engrams in parallel.
- Scoring and aggregation – each parent engram accumulates an activation score based on the convergence of its sub-engrams.
- Winner selection – if a single engram surpasses the recognition threshold, it is fully activated and determines the output; if several engrames are partially active, the strongest one prevails through lateral inhibition.
- Novelty detection – if all activation scores remain below threshold, the system enters a novelty state: no complete engram is recognized.
- Attention engagement – attention is mobilized to increase neuronal gain and plasticity, strengthening the binding of co-active sub-engrams into a proto-engram.
- Consolidation vs. fading – if the stimulus/event is repeated or behaviorally relevant, the proto-engram is consolidated into a stable engram; if not, the weak connections gradually fade.
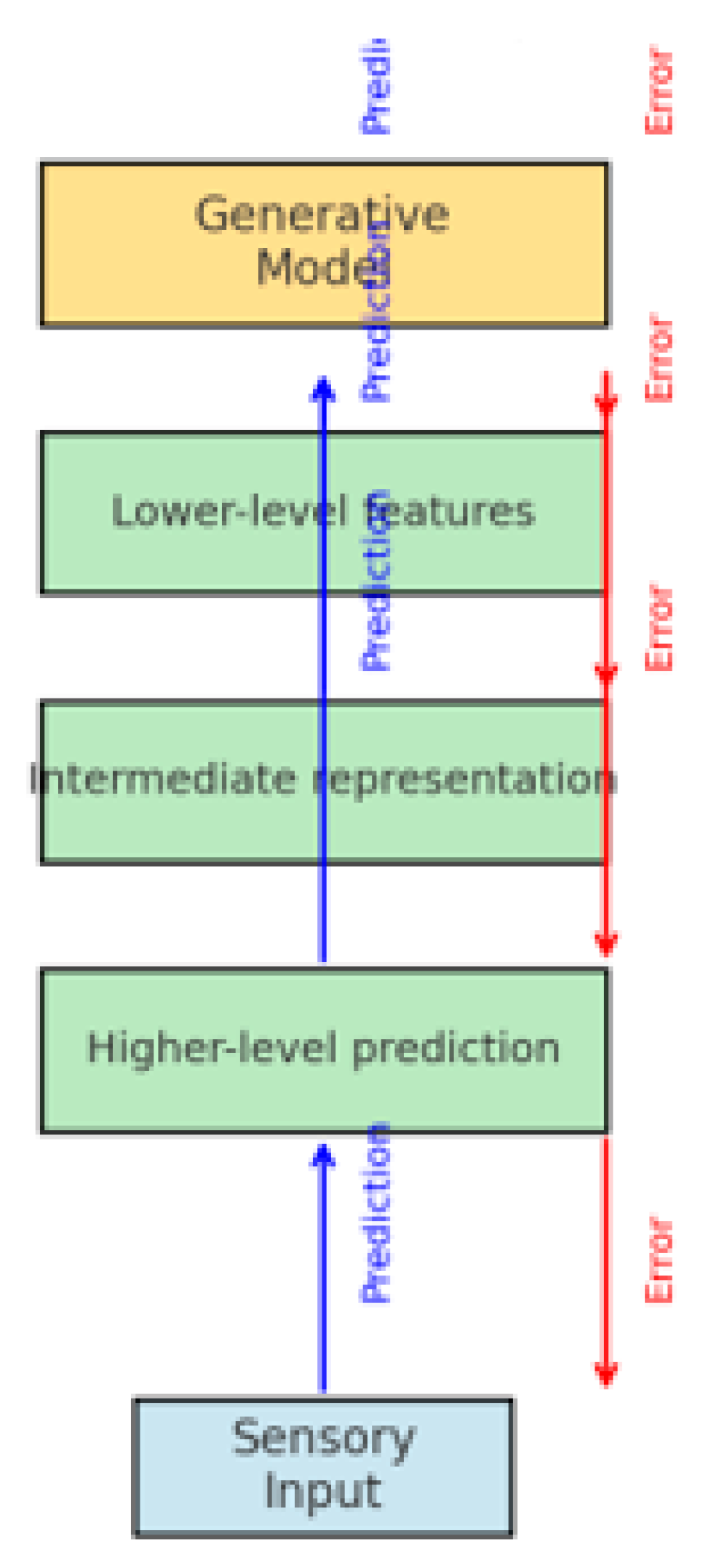
2.2. The Theoretical Framework of Predictive Coding (PC)
- Internal generative models (hierarchical representations of the environment),
- Prediction errors (differences between input and predictions),
- Adaptive updating (learning through error minimization).
2.3. Comparison of EBPM and PC
2.3.1. Theoretical Foundation
2.3.2. Processing Dynamics in the Two Models
- The brain generates hierarchical top–down predictions.
- Sensory input is compared with predictions.
- Prediction errors are transmitted upward and correct the internal model.
- Perception arises as the result of minimizing the difference between prediction and input.
- The input activates elementary engram units.
- The elementary engram units simultaneously activate multiple engrams.
- Through lateral inhibition, the most active engram prevails.
- If no winner exceeds the threshold, attention is triggered and a new engram is formed.
- Perception is the complete reactivation of a pre-existing network, not a reconstruction process.
| Aspect | Predictive Coding (PC) | Engram-Based Program Model (EBPM) |
|---|---|---|
| Basic principle | Predictions compared with input; error is propagated. | Recognition through engrames composed of reusable sub-engrams; full activation only for familiar stimuli. |
| Partial activation | - | Partial/similar stimuli → shared sub-engrams are activated; multiple engrams become partially active. |
| Selection | - | Lateral inhibition: the engram with the most complete activation prevails. |
| Novelty / unfamiliarity | Unclear how new representations are detected and formed | If all engrames remain below threshold: sub-engrams are active, but no parent engram is completed → novelty state. Attention is engaged. |
| Formation of new engrames (role of attention) | Does not specify a concrete biological mechanism. | Automatic (“fire together, wire together”): in the novelty state, attention increases neuronal gain and plasticity; co-active sub-engrams are bound into a proto-engram. Without repetition: the linkage fades/does not finalize. With repetition: it consolidates into a stable engram. |
| Signaling absence | Postulates “omission error” (biologically problematic). | No “negative spike.” Silence at parent-engrams level + fragmentary activation of sub-engrams defines novelty; attention manages exploration and learning. |
| Neuronal logic | Requires an internal comparator/explicit error signals. | Pure spike/silence + lateral inhibition + co-activation-dependent plasticity; attention sets the learning threshold. |
2.3.3. Comparative Advantages and Limitations
| Domain | EBPM | PC Prediction |
|---|---|---|
| Central Mechanism | Direct Activation of Multimodal Engrams | Continuous generation and updating of predictions |
| The Role of Prediction | Non-existent; only goal-driven sensitization | Fundamental for processing |
| Recognition Speed | Instantaneous for familiar stimuli | Slower, requires iterative computation |
| Energy Efficiency | High (without generative simulation) | Lower (increased metabolic cost) |
| Attention Control | Goal-driven (sensitization of relevant networks) | Through the magnitude of prediction errors |
| Biological Plausibility | Supported by studies on engrams and inhibition | Supported by cortical predictive microcircuits |
| Typical Application Domains | Rapid recognition, episodic memories, reflexes | Perception in ambiguous or noisy conditions |
| Domain | EBPM | PC | Similar existing theories |
|---|---|---|---|
| Central Mechanism | Direct multimodal engram activation | Generative predictive coding | Hopfield attractors, episodic replay |
| Recognition Speed | Fast, one-step match | Iterative, slower | Fast (pattern completion) |
| Energy Efficiency | High, no generative loop | Low, high metabolic demand | Variable |
| Flexibility | Limited to stored engrams | High, reconstructive | Active inference |
| Biological Plausibility | Supported by engram studies | Supported by cortical microcircuits | Both partially supported |
2.3.4. Divergent Experimental Predictions
| Domain | EBPM Prediction | PC Prediction | Testable through |
|---|---|---|---|
| fMRI (similarity analysis) | Recognition = reactivation of storage networks | Anticipatory activation in higher cortical areas | fMRI + RSA |
| EEG/MEG (latency) | Rapid recognition of familiar patterns | Longer latencies due to generative processing | EEG/MEG |
| Cue-based recall | A partial cue reactivates the entire engram | The cue triggers generative reconstruction | Behavioral and neurophysiological studies |
| Robotic Implementation | Faster execution of learned procedures | Better adaptive reconstruction with incomplete data | AI/Robotics Benchmarks |
2.3.5. Interpretive Analysis
- EBPM better explains rapid recognition, complex reflexes, and sudden cue-based recall.
- PC is superior in situations with incomplete or ambiguous data, due to its generative reconstruction capability.
- The two models are therefore complementary, each having its own set of advantages and limitations.
2.3.6. EBPM vs. Related Theories — Method-by-Method Comparison
| Method / Framework | What the method does | Identical to EBPM | What it adds vs. EBPM | What EBPM adds |
|---|---|---|---|---|
| Hopfield / Attractor networks [7] | Recurrent dynamics converge to stored patterns (content-addressable recall from partial cues). | Pattern completion from partial input; competition among states. |
Explicit energy formalism; well-characterized attractor dynamics. |
Explicit multimodality (sensory–motor–affective–cognitive) and engram units beyond binary patterns. |
| CLS — Complementary Learning Systems (hippocampus fast; neocortex slow) [8] | Episodic rapid learning + slow semantic consolidation. | Separation of “fast vs. slow” memory routes. | System-level transfer between stores. | EBPM is operational in real time via direct matching; no mandatory inter-system consolidation for recognition. |
| Hippocampal indexing: pattern completion/separation [9,10] | Hippocampus indexes episodes; CA3 supports completion, DG supports separation. | Fast episodic reactivation from indices; completion logic. | Fine neuro-anatomical mapping (CA3/DG) with predictions. | EBPM generalizes to neocortex and to multimodal action-oriented engrams, not only episodic replays. |
| Exemplar / Instance-Based (GCM; Instance Theory) [11,12] | Decisions by similarity to stored instances; latencies fall with instance accrual. | Direct “match to memory” with similarity score. | Quantitative psychophysics of categorization and automatization. | EBPM includes motor/interoceptive bindings and neuronal lateral inhibition/WTA selection. |
| Recognition-by-Components (RBC) [13] | Object recognition via geons and relations. | Sub-engrams akin to primitive features. | Explicit 3D structural geometry. | EBPM is not vision-limited; covers sequences/skills and affective valence. |
| HTM / Sequence Memory (cortical columns) [5] | Sparse codes, sequence learning, local predictions. | Reuse of sparse distributed codes; sequence handling. | Continuous prediction emphasis. | EBPM does not require continuous generative prediction; for familiar inputs it yields lower latency/energy via direct recall. |
| Sparse Distributed Memory (SDM) [14] | Approximate addressing in high-D sparse spaces; noise-tolerant recall. | Proximity-based matching in feature space. | Abstract memory addressing theory. | EBPM specifies engram units and novelty/attention gating with biological microcircuit motifs. |
| Memory-Augmented NNs (NTM/DNC)[15,16] | Neural controllers read/write an external memory differentiably. | Key-based recall from stored traces. | General algorithmic read/write; task-universal controllers. | EBPM is bio-plausible (engram units; STDP-like plasticity) without opaque external memory. |
| Episodic Control in RL [17] | Policies exploit episodic memory for rapid action. | Fast reuse of familiar episodes. | RL-specific credit assignment and returns. | EBPM operates for perception and action with multimodal bindings. |
| Prototypical / Metric-Learning (few-shot) [18] | Classify by distance to learned prototypes in embedding space. | Prototype-like matching ≈ EBPM encoder + similarity. | Episodic training protocols; few-shot guarantees. | EBPM grounds the embedding in neuro-plausible engrams + inhibition, not just vector spaces. |
| Predictive Coding / Active Inference [1], [2,3,19,20] | Top-down generative predictions + iterative error minimization. | Shares attentional modulation and context use. | Robust reconstruction under missing data via generative loops. | EBPM delivers instant responses on familiar inputs (no iterative inference) with lower compute/energy; DIME then falls back to PC for novelty. |
- Extract low-level primitives (edges, textures, timbre, phonemes).
- 2.
- Activate candidate multimodal Engram Units (EUs) from those primitives.
- 3.
- Spread + compete among candidate EUs with lateral inhibition / WTA.
- 4.
- Select the winner (fully activated engram) → recognition / action trigger.
- 5.
- Familiarity/novelty test (no clear winner → novelty branch).
- 6.
- Rapid plasticity (Hebb/STDP-like) to form/adjust new or refined engrams.
- 7.
- Bind multimodal facets (sensory–motor–affective–cognitive) within the engram; optional hippocampal indexing for episode-level links.
- 8.
- Real-time reuse: for familiar inputs, jump straight to Steps 3–4 (no generative loop).
2.3.7. Distinctive Contributions of EBPM (Beyond Attractors/CLS/HTM/PC)
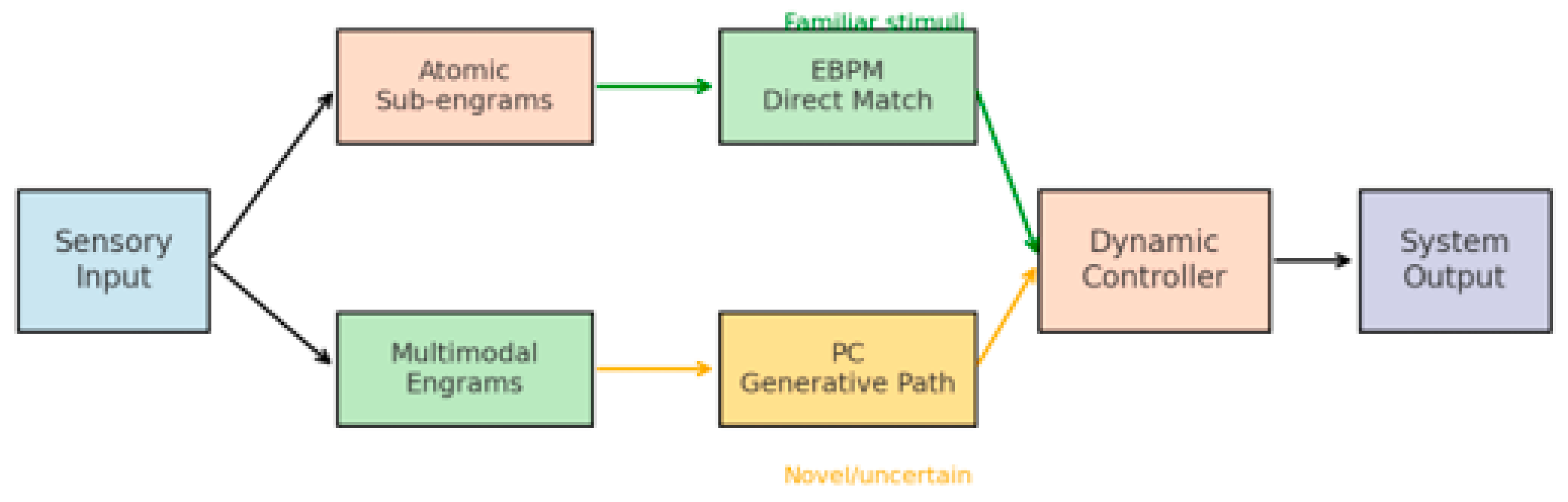
- Multimodal Engram as the Computational Unit. EBPM defines an engram as a compositional binding of sensory, motor, interoceptive/affective, and cognitive facets. This goes beyond classic attractor networks (binary pattern vectors) and metric-learning embeddings by specifying the unit of recall and action as a multimodal assembly.
- Novelty-first Decision with Explicit Abstention. By design, EBPM yields near-zero classification on truly novel inputs. Partial activation of sub-engrams is treated as a reliable novelty flag—not as a misclassification. This sharp separation of recognition versus novelty is absent from PC’s iterative reconstruction loop and from generic prototype schemes.
- Attention-gated Proto-Engram Formation. EBPM assigns a concrete role to attention and neuromodulatory gain (e.g., LC-NE) in binding co-active sub-engrams into a proto-engram during novelty, followed by consolidation. This specifies “when and how new representations form” more explicitly than attractors/CLS/HTM.
- Real-time Reuse without Iterative Inference. For familiar inputs, EBPM supports one-step reactivation of stored engrams (winner selection via lateral inhibition), avoiding costly predictive iterations. This explains ultra-fast recognition and procedural triggering in overlearned contexts.
- Integration-readiness via DIME. EBPM is not an isolated alternative: the DIME controller provides a formal arbitration (α) that privileges EBPM under high familiarity and shifts toward PC under uncertainty, yielding a principled hybrid.
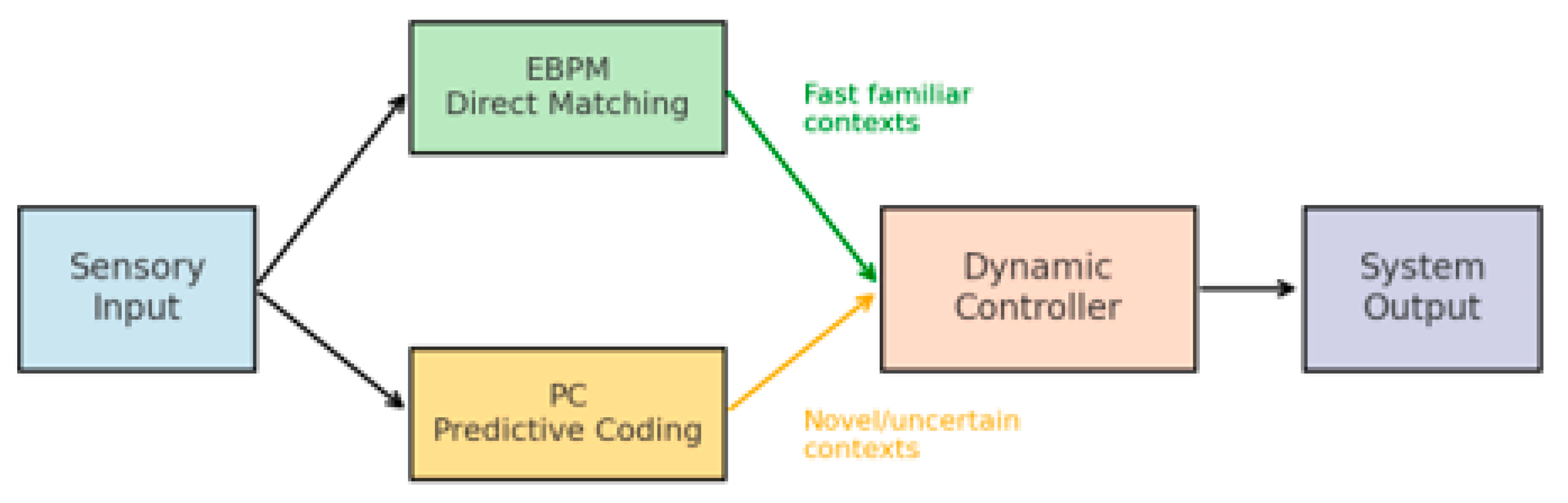
2.4. The DIME Integrative Model
- Dual-path processing: sensory input is processed simultaneously through direct engram matching (EBPM) and top–down prediction (PC).
- Dynamic controller: a central mechanism evaluates the context (level of familiarity, uncertainty, noise) and selects the dominant pathway.
- Adaptive flexibility: in familiar environments → EBPM becomes dominant; in ambiguous environments → PC is engaged.
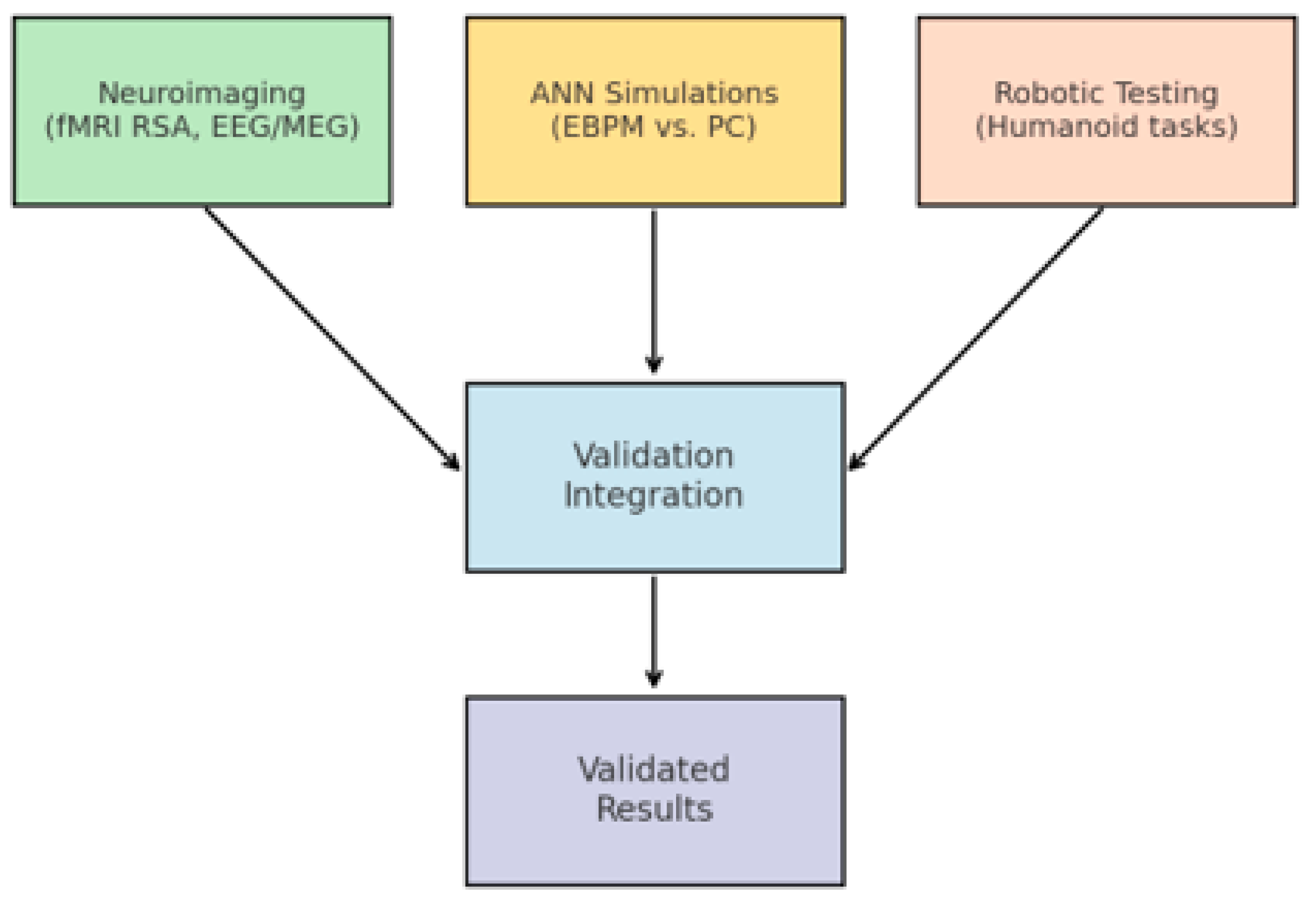
2.5. Methodology of Experimental Validation
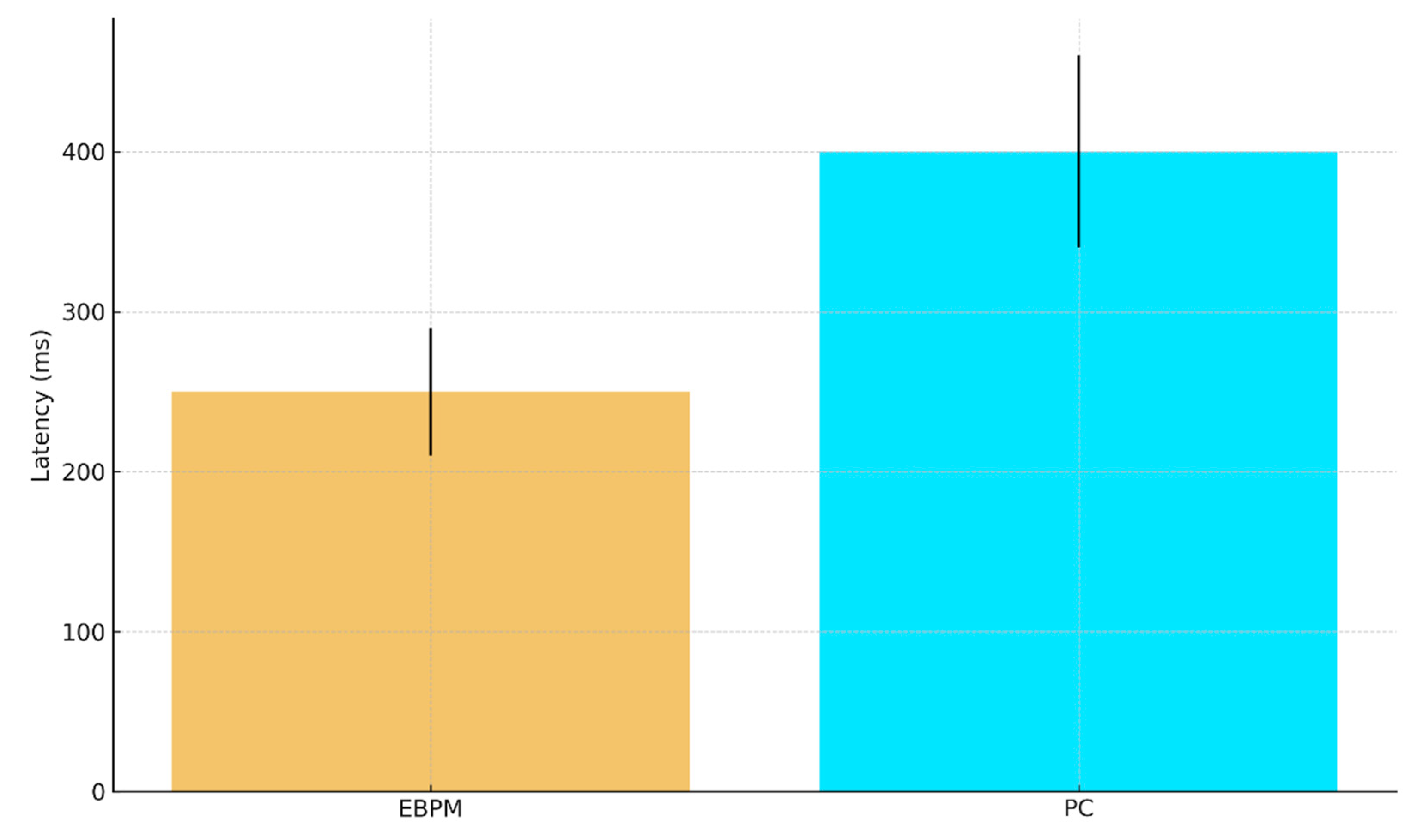
- H1 (EEG/MEG latency). EBPM-dominant trials (familiar stimuli) yield shorter recognition latencies (≈200–300 ms) compared to PC-dominant trials (≈350–450 ms). We base the ≈200–300 ms range on recent EEG evidence indicating that recognition memory signals emerge from around 200 ms post-stimulus across diverse stimulus types [27].
- H2 (fMRI representational similarity). EBPM produces higher RSA between learning and recognition patterns in hippocampal/cortical regions; PC shows anticipatory activation in higher areas under uncertainty.
- H3 (Cue-based recall completeness). Partial cues reinstantiate full engram patterns under EBPM, while PC engages reconstructive dynamics without full reinstatement.
- H4 (Behavioral/robotic efficiency). Under stable familiarity, EBPM leads to lower inference time and energy cost than PC; under novelty/noise, DIME shifts toward PC to preserve robustness.
- H5 (NE-gain modulation). Pupil-indexed or physiological proxies of LC-NE correlate with novelty detection and proto-engram formation during EBPM-dominant novelty trials.
-
Neuroimaging:
- fMRI (representational similarity analysis, RSA) → measures the overlap of patterns between learning and recognition.
- EEG/MEG → measures recognition latencies (EBPM ≈ shorter, PC ≈ longer).
-
Computational simulations:
- Artificial neural networks inspired by EBPM vs. PC.
- Comparison of inference times and computational cost.
-
Robotic experiments:
- Implementation in visuomotor control.
- Measurement of reaction times and energy consumption in navigation tasks.
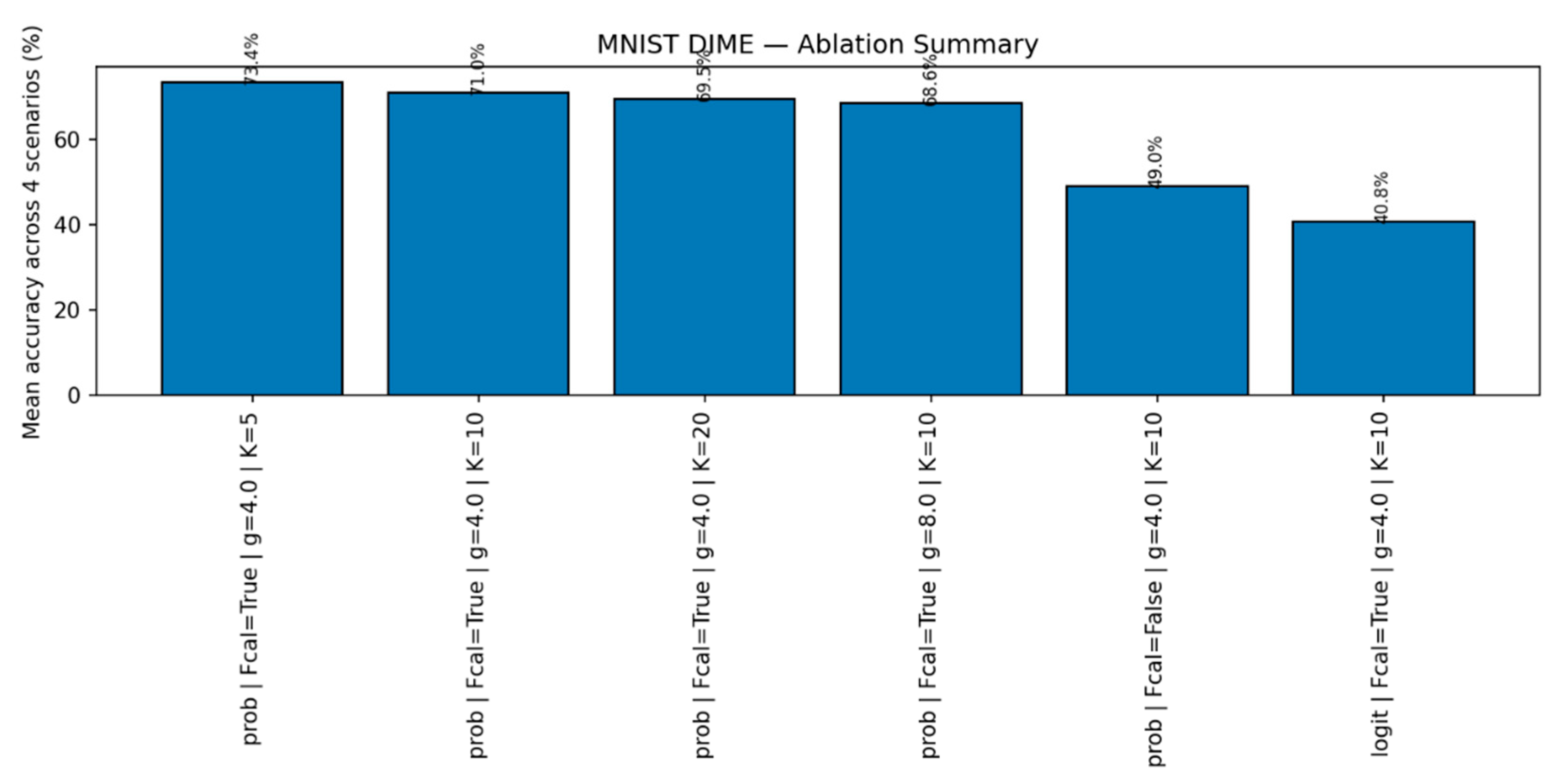
2.6. ANN Simulation Protocol
- (i)
- DIME-Prob (probability-level fusion): p = α·p_EBPM + (1−α)·p_PC, with α driven by a familiarity score calibrated on familiar validation data. On CIFAR-10 we tune {gain = 4.0, thr_shift = 0.75, T_EBPM = 6.0, T_PC = 1.0}.
- (ii)
- (ii) DIME-Lazy (runtime-aware gating, used in robotics): δ = mean(|I_t − I_{t−1}|). If δ ≤ 0.004 → EBPM-only; if δ ≥ 0.015 → PC-only; otherwise fuse with fixed α = 0.65.
3. Results
Preface — Scope of the Simulations (Proof-of-Concept)
3.1. ANN Simulations on Familiar vs. Novel Splits
- Metrics: top-1 accuracy, latency (ms/ex.), and for DIME the mean controller weight ᾱ where applicable.
- These results support the complementarity… (see Section 2.6).
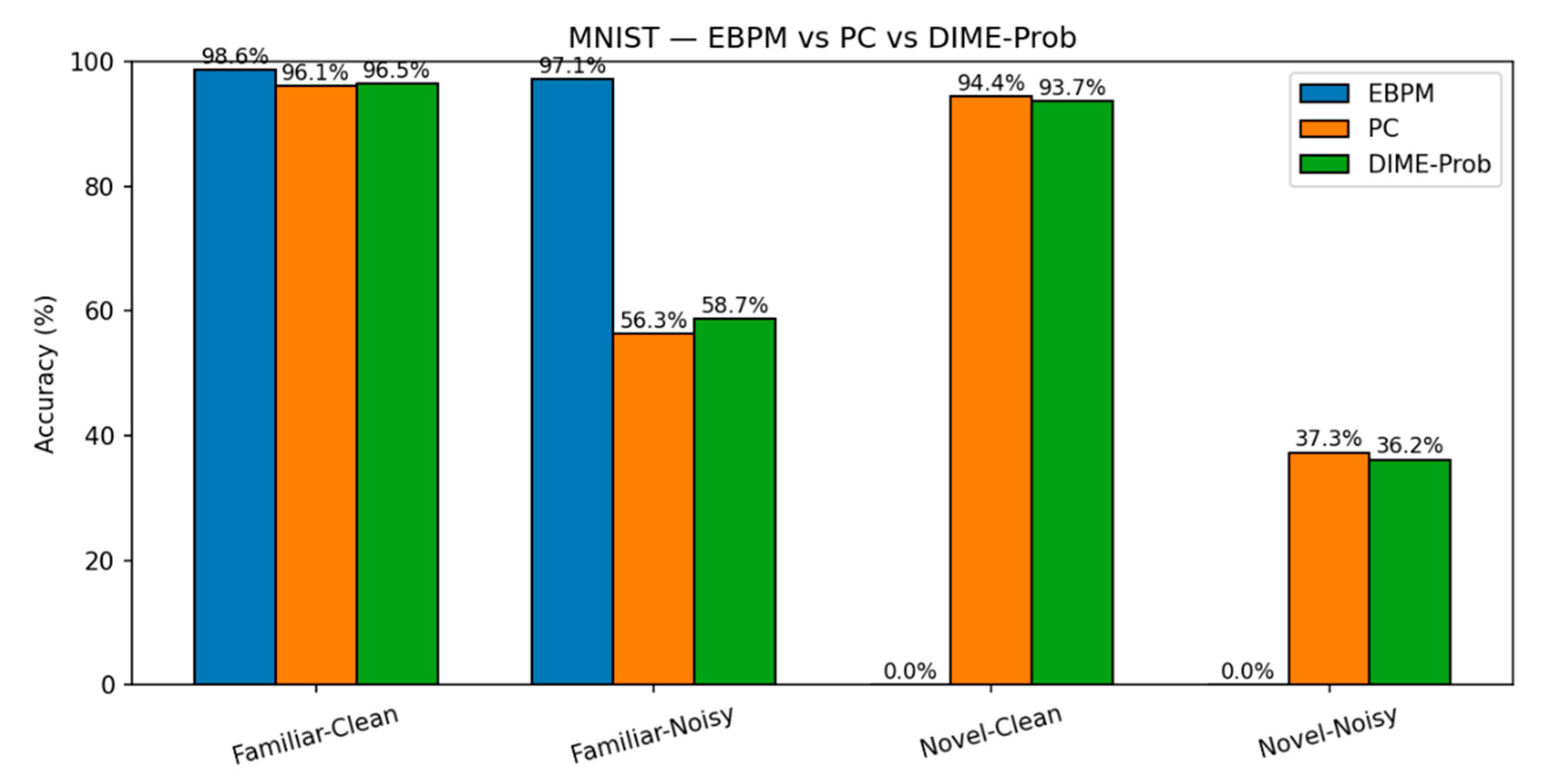
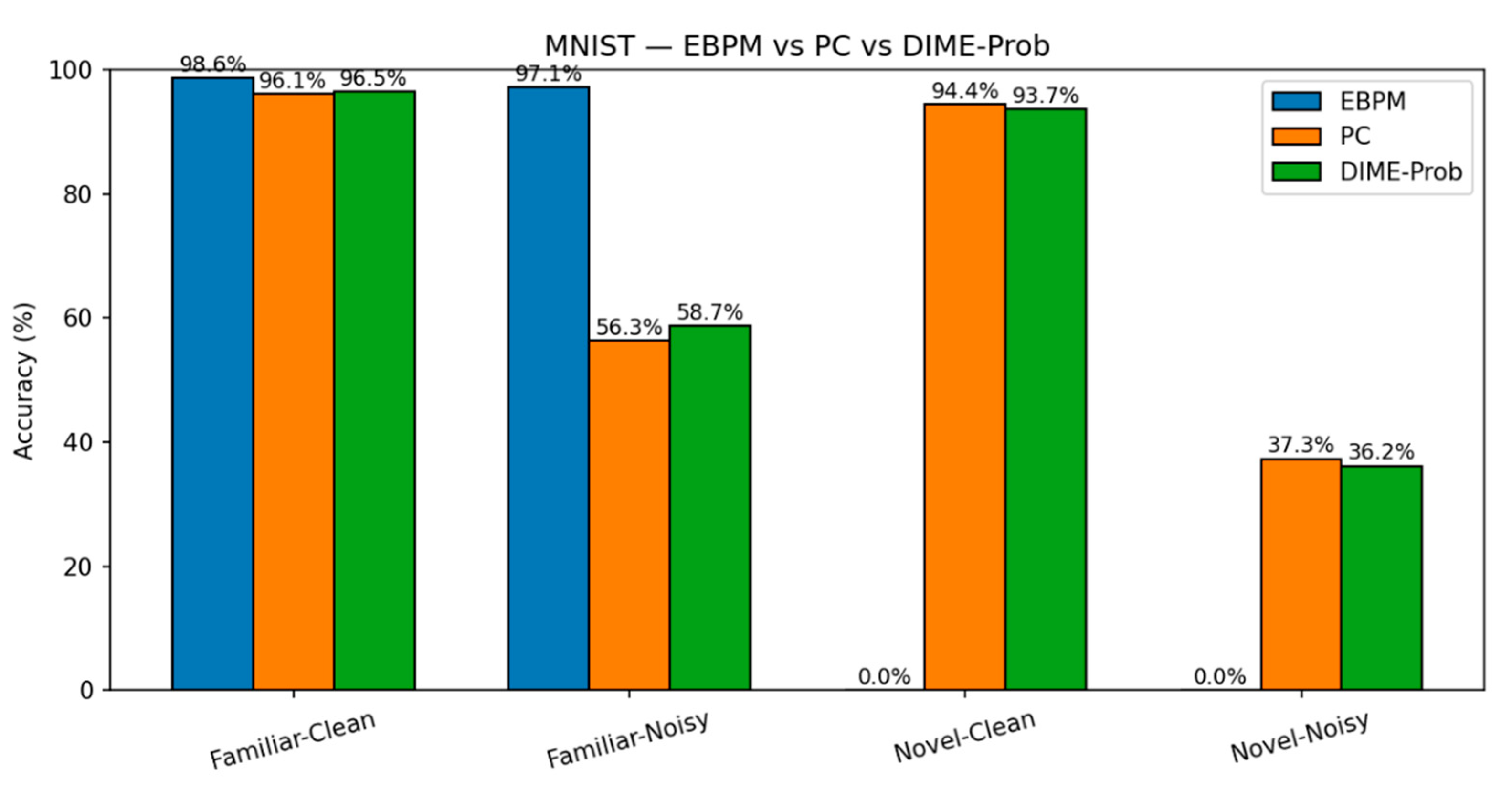
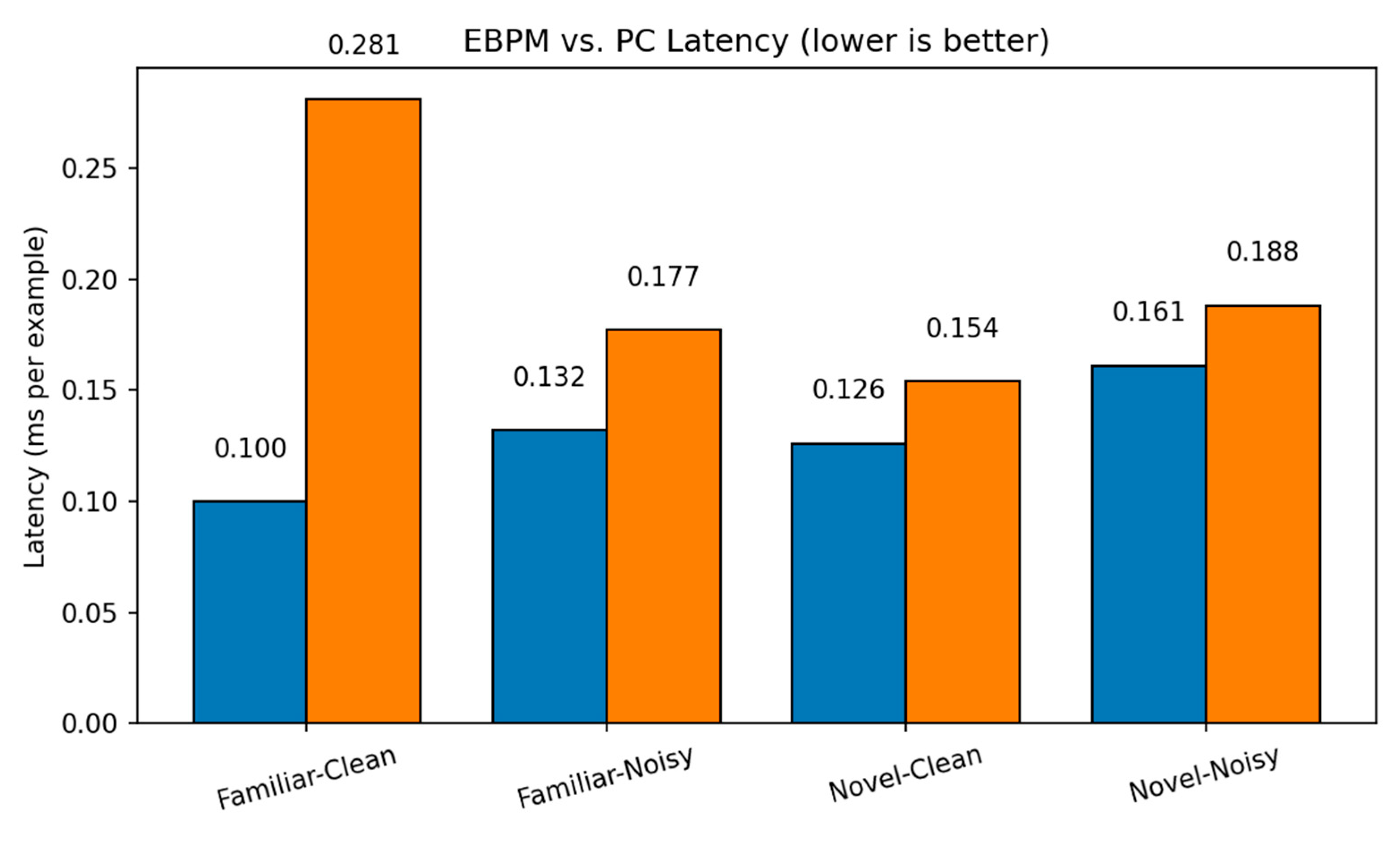
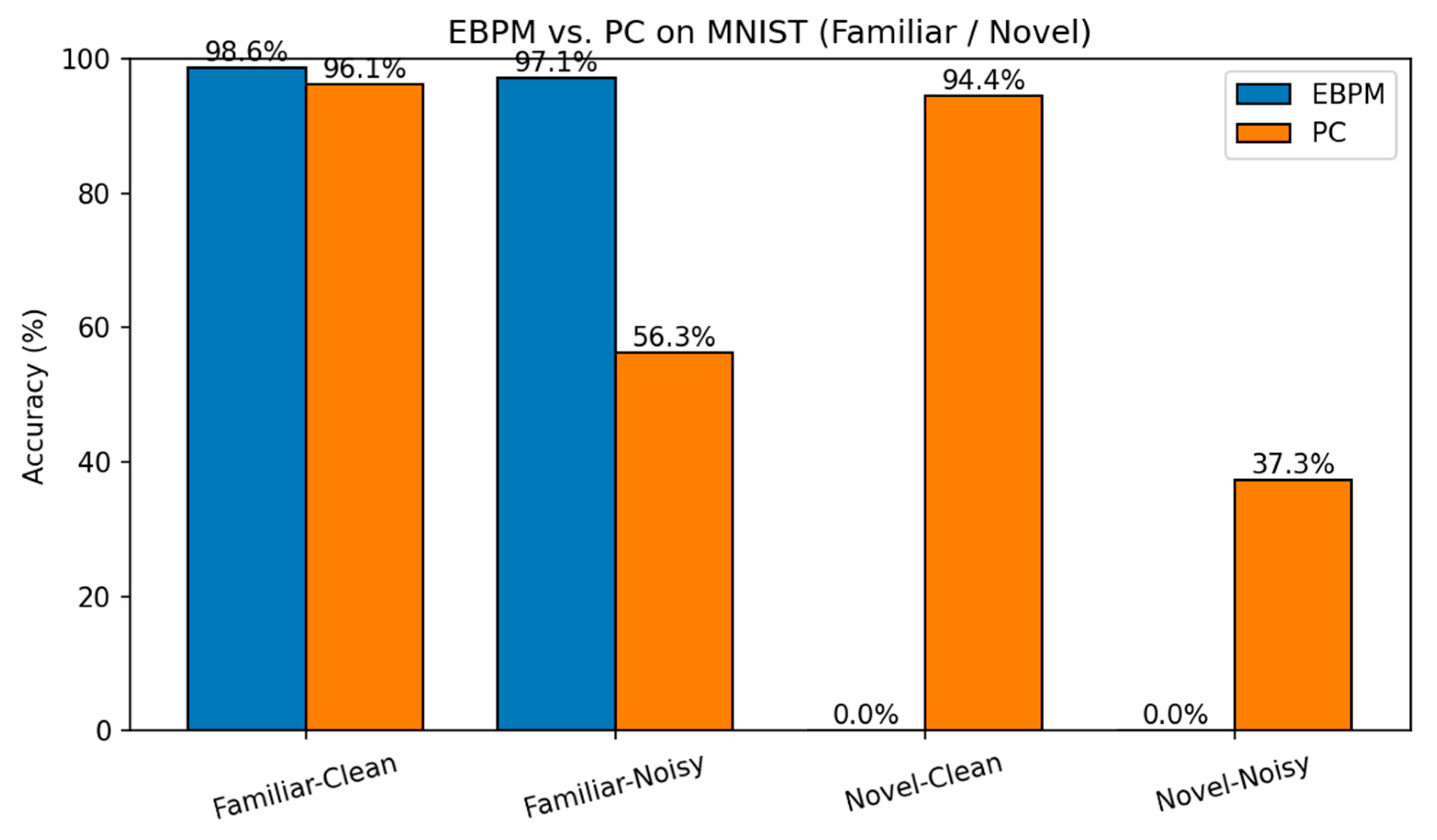
- EBPM — Familiar-Clean: 98.63%, 0.100 ms/ex; Familiar-Noisy: 97.13%, 0.132 ms/ex; Novel: 0.0%.
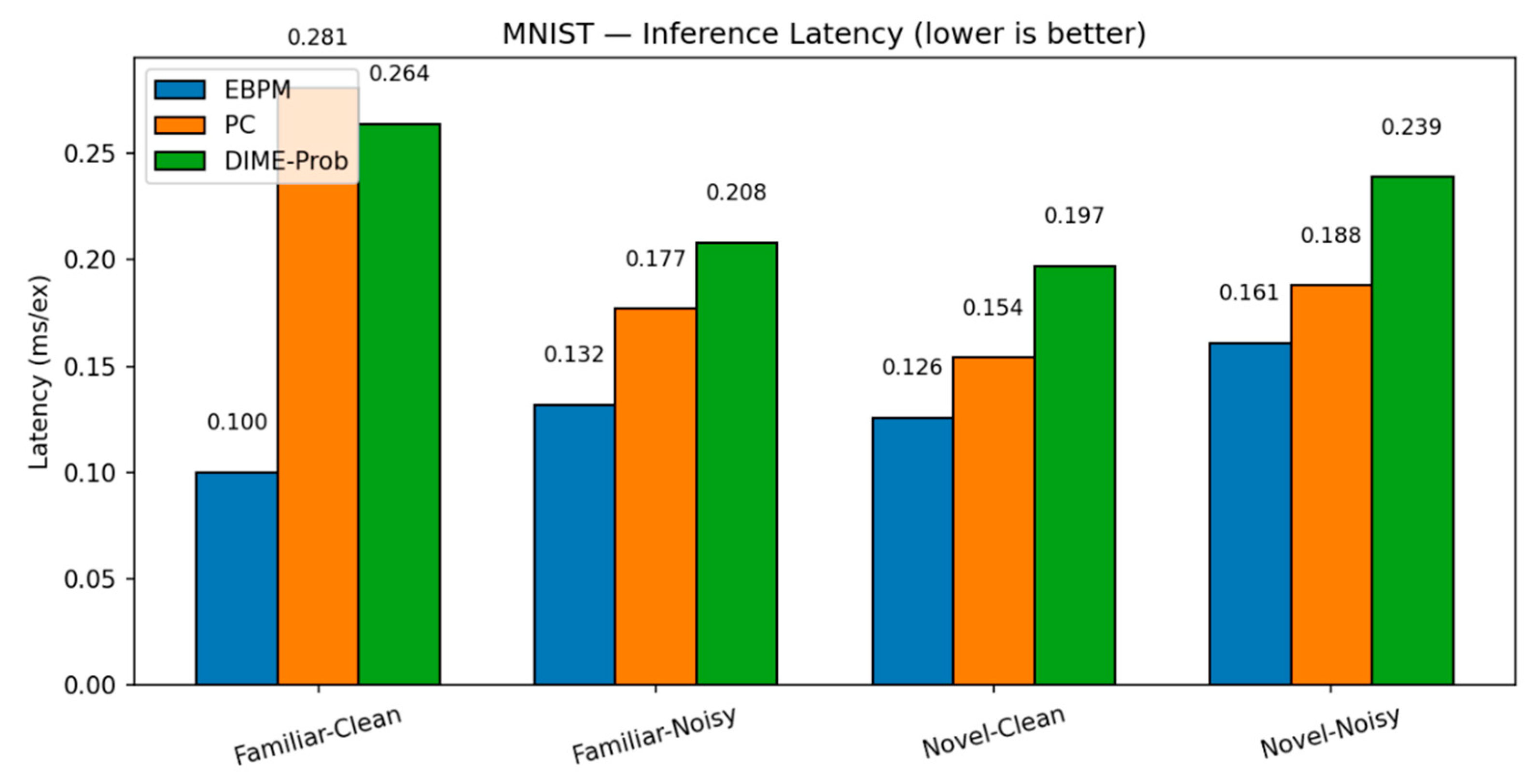
- PC — Familiar-Clean: 96.11%, 0.281 ms/ex; Familiar-Noisy: 56.28%, 0.177 ms/ex; Novel-Clean: 94.40%, 0.154 ms/ex; Novel-Noisy: 37.27%, 0.188 ms/ex.
- DIME (adaptive) — Familiar-Clean: 98.63%, 0.247 ms/ex, ᾱ = 0.993; Familiar-Noisy: 96.93%, 0.188 ms/ex, ᾱ = 0.974; Novel≈0% (as EBPM dominates unless fused).
- DIME-Prob (probability-level fusion) — Familiar-Clean: 96.47%, 0.264 ms; Familiar-Noisy: 58.68%, 0.208 ms; Novel-Clean: 93.65%, 0.197 ms; Novel-Noisy: 36.16%, 0.239 ms.
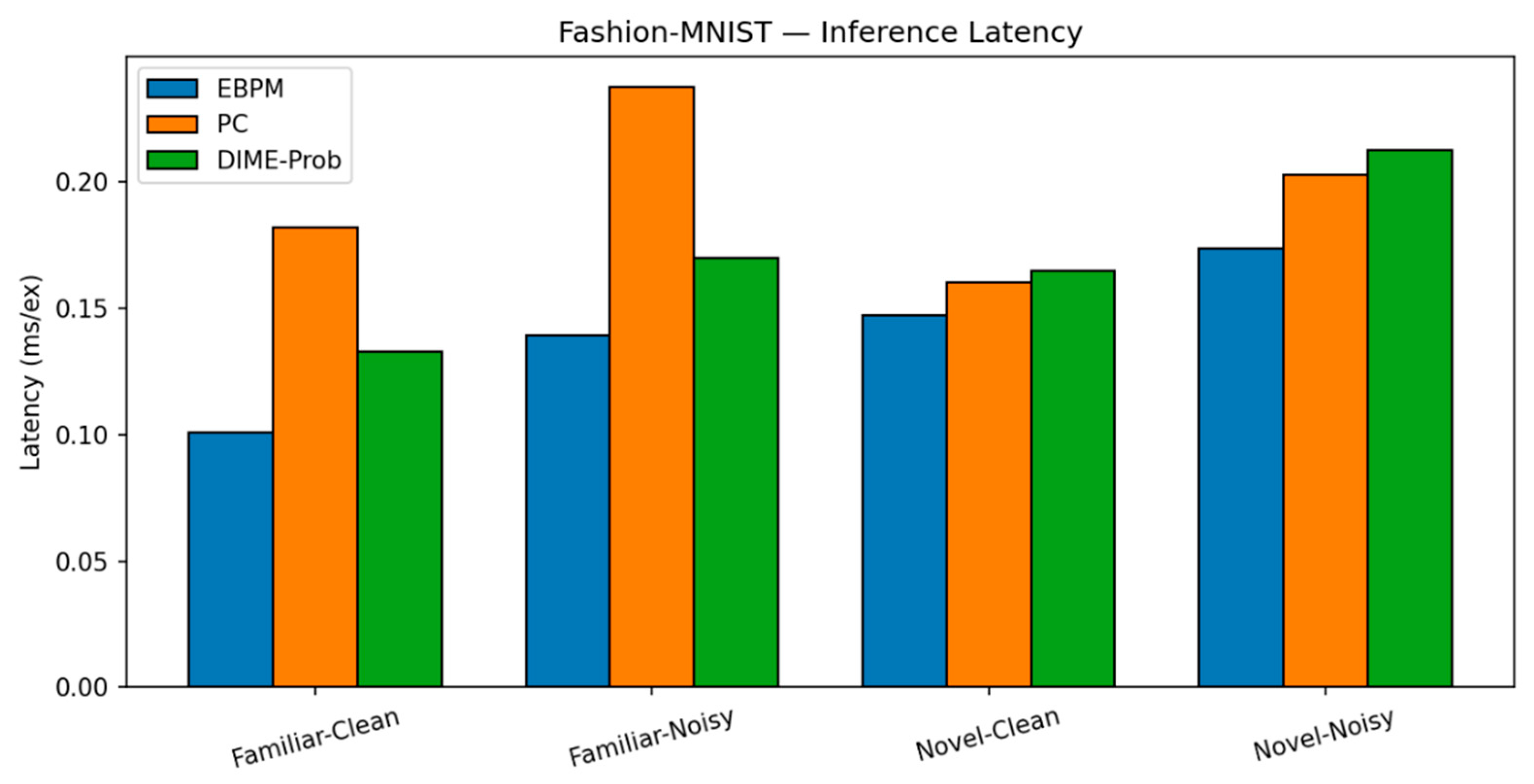
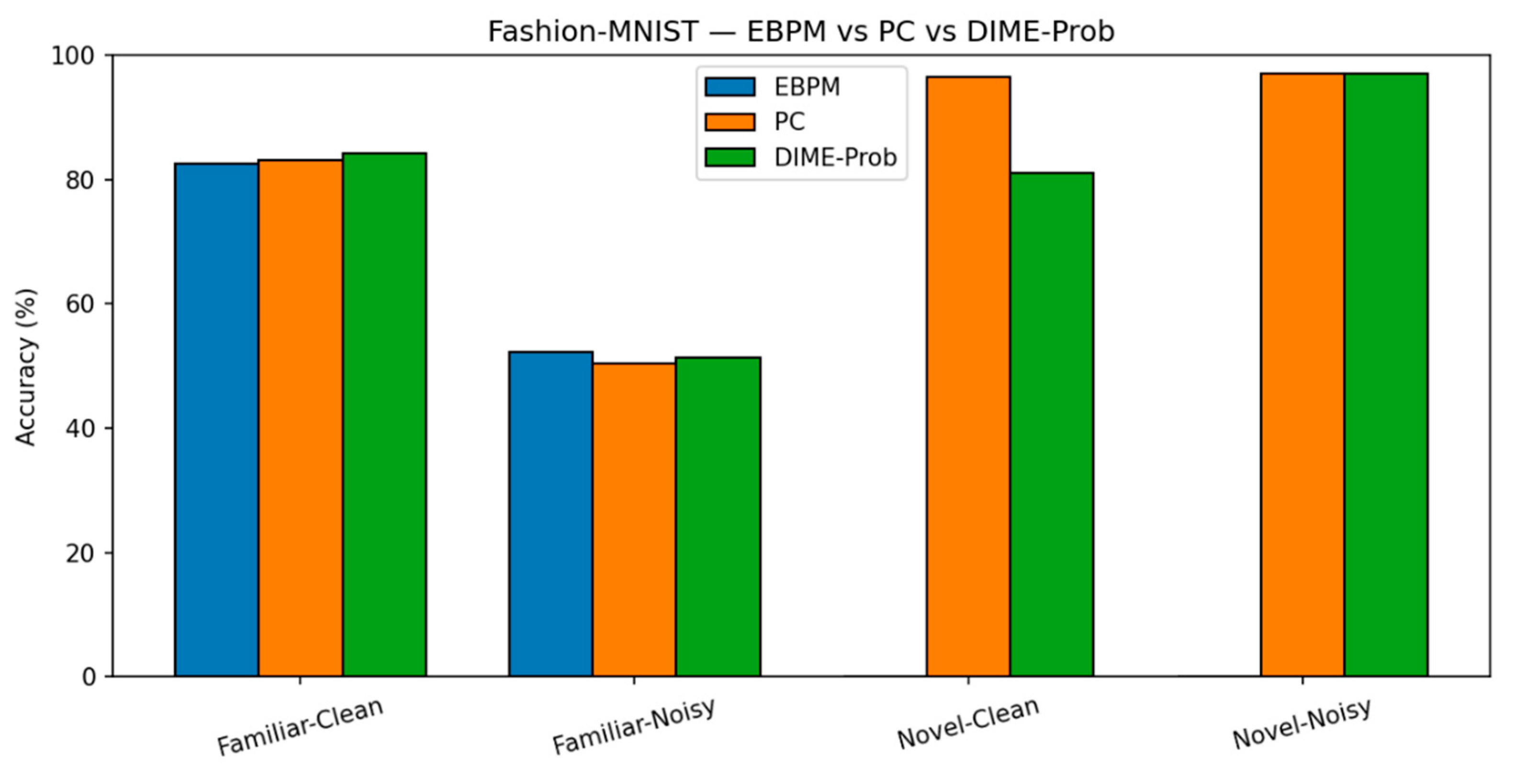
- Fashion-MNIST. Trends are similar to MNIST, with a larger familiar/noise gap.
- EBPM — Familiar-Clean: 82.56%, 0.101 ms; Familiar-Noisy: 52.35%, 0.139 ms; EBPM reports ≈0% accuracy on novel inputs, consistent with its design as a novelty detector: sub-engrams activate but do not yield a full engram. This produces an explicit novelty signal, rather than a forced classification.
- PC — Familiar-Clean: 83.14%, 0.182 ms; Familiar-Noisy: 50.45%, 0.238 ms; Novel-Clean: 96.50%, 0.160 ms; Novel-Noisy: 97.00%, 0.203 ms.
- DIME (adaptive) — Familiar-Clean: 84.14%, 0.133 ms, ᾱ = 0.536; Familiar-Noisy: 51.31%, 0.170 ms, ᾱ = 0.221; Novel-Clean: 81.10%, 0.165 ms, ᾱ = 0.506; Novel-Noisy: 97.10%, 0.213 ms, ᾱ = 0.443.
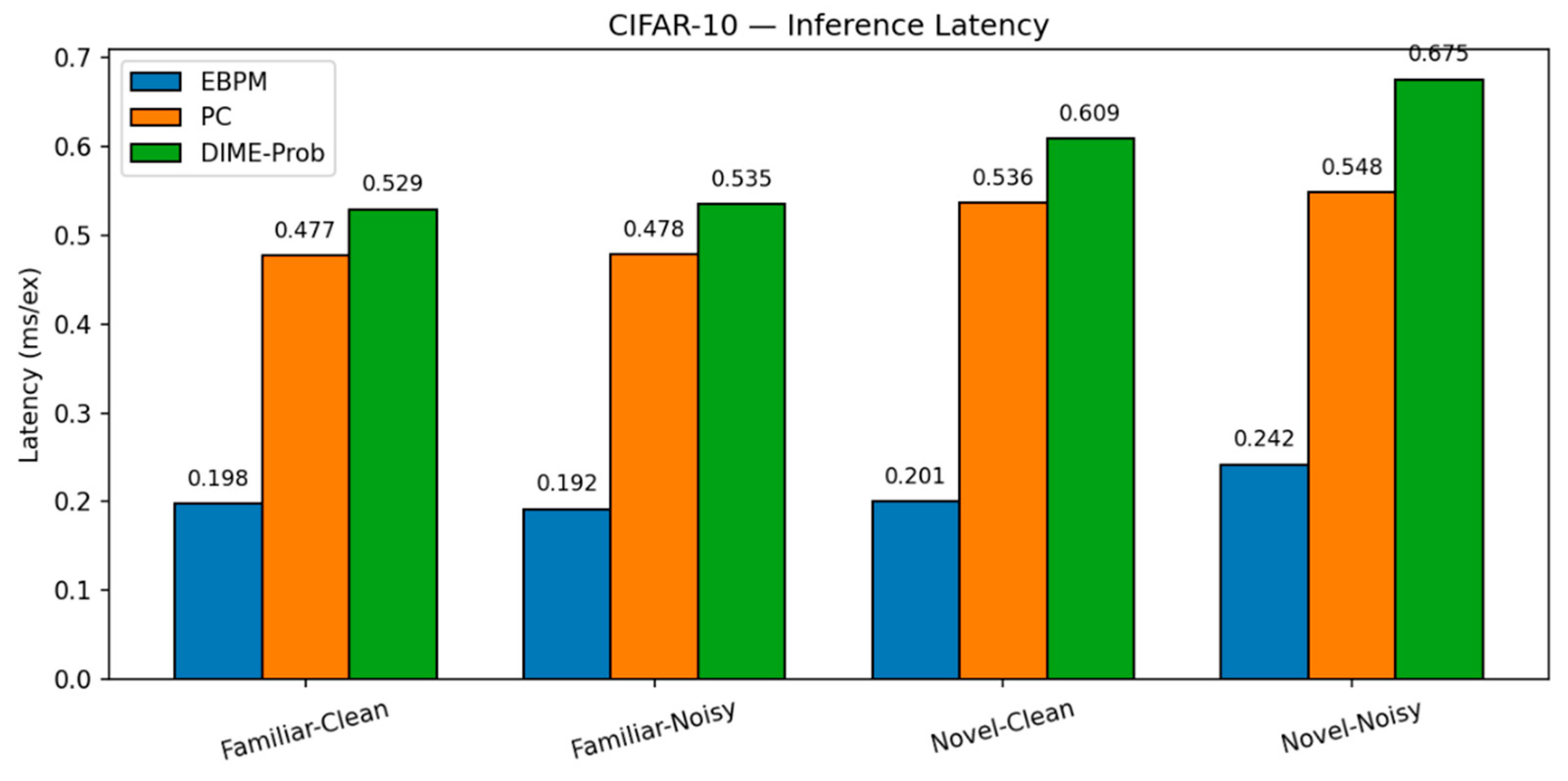
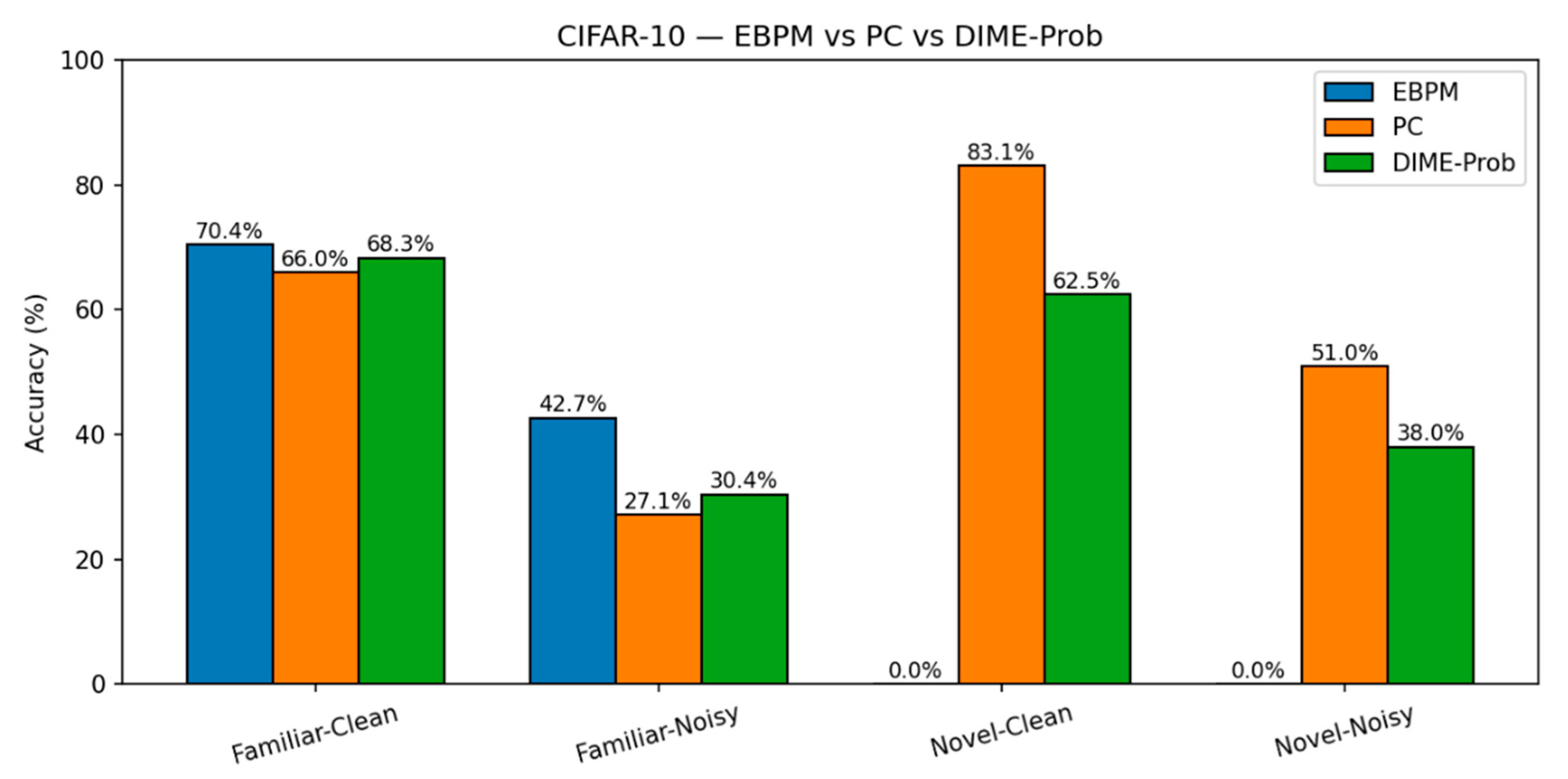
- EBPM — Familiar-Clean: 70.42%, 0.198 ms; Familiar-Noisy: 42.67%, 0.192 ms; Novel: 0%.
-
PC — Familiar-Clean: 65.99%, 0.477 ms; Familiar-Noisy: 27.15%, 0.478 ms; Novel-Clean: 83.10%, 0.536 ms; Novel-Noisy: 50.95%, 0.548 ms.
- DIME-Prob (best tuned) — Familiar-Clean: 68.31%, 0.529 ms; Familiar-Noisy: 30.38%, 0.535 ms; Novel-Clean: 62.50%, 0.609 ms; Novel-Noisy: 38.05%, 0.675 ms.
| Model | Familiar-Clean (acc / ms) |
Familiar-Noisy (acc / ms) | Novel-Clean (acc / ms) |
Novel-Noisy (acc / ms) |
ᾱ |
|---|---|---|---|---|---|
| EBPM | 98.63% / 0.100 | 97.13% / 0.132 | 0.00%* / 0.126 | 0.00%* / 0.161 | - |
| PC | 96.11% / 0.281 | 56.28% / 0.177 | 94.40% / 0.154 | 37.27% / 0.188 | - |
| DIME | 98.63% / 0.247 | 96.93% / 0.188 | 0.00% / 0.135 | 0.00% / 0.289 | 0.99 / 0.97 / – / – |
| DIME-Prob | 96.47% / 0.264 | 58.68% / 0.208 | 93.65% / 0.197 | 36.16% / 0.239 | 0.61/ 0.38/ 0.41/ 0.23 |
| Model | Familiar-Clean (acc / ms) |
Familiar-Noisy (acc / ms) | Novel-Clean (acc / ms) |
Novel-Noisy (acc / ms) |
ᾱ |
|---|---|---|---|---|---|
| EBPM | 82.56% / 0.101 | 52.35% / 0.139 | 0.00% / 0.147 | 0.00% / 0.174 | - |
| PC | 83.14% / 0.182 | 50.45% / 0.238 | 96.50% / 0.160 | 97.00% / 0.203 | - |
| DIME | 84.14% / 0.133 | 51.31% / 0.170 | 81.10% / 0.165 | 97.10% / 0.213 | 0.54 / 0.22 / 0.51 / 0.44 |
| Model | Familiar-Clean (acc / ms) |
Familiar-Noisy (acc / ms) |
Novel-Clean (acc / ms) |
Novel-Noisy (acc / ms) |
|---|---|---|---|---|
| EBPM | 70.42% / 0.198 | 42.67% / 0.192 | 0.00% / 0.201 | 0.00% / 0.242 |
| PC | 65.99% / 0.477 | 27.15% / 0.478 | 83.10% / 0.536 | 50.95% / 0.548 |
| DIME-Prob* | 68.31% / 0.529 | 30.38% / 0.535 | 62.50% / 0.609 | 38.05% / 0.675 |
3.2. Comparative Results and Simulations
- EBPM: performs well in speed and efficiency, but is limited in adaptation and robustness.
- PC: flexible and robust, but entails high costs.
- DIME: integrates the advantages of both, providing an optimal balance between speed, efficiency, and flexibility.
3.3. General Information Flow in DIME
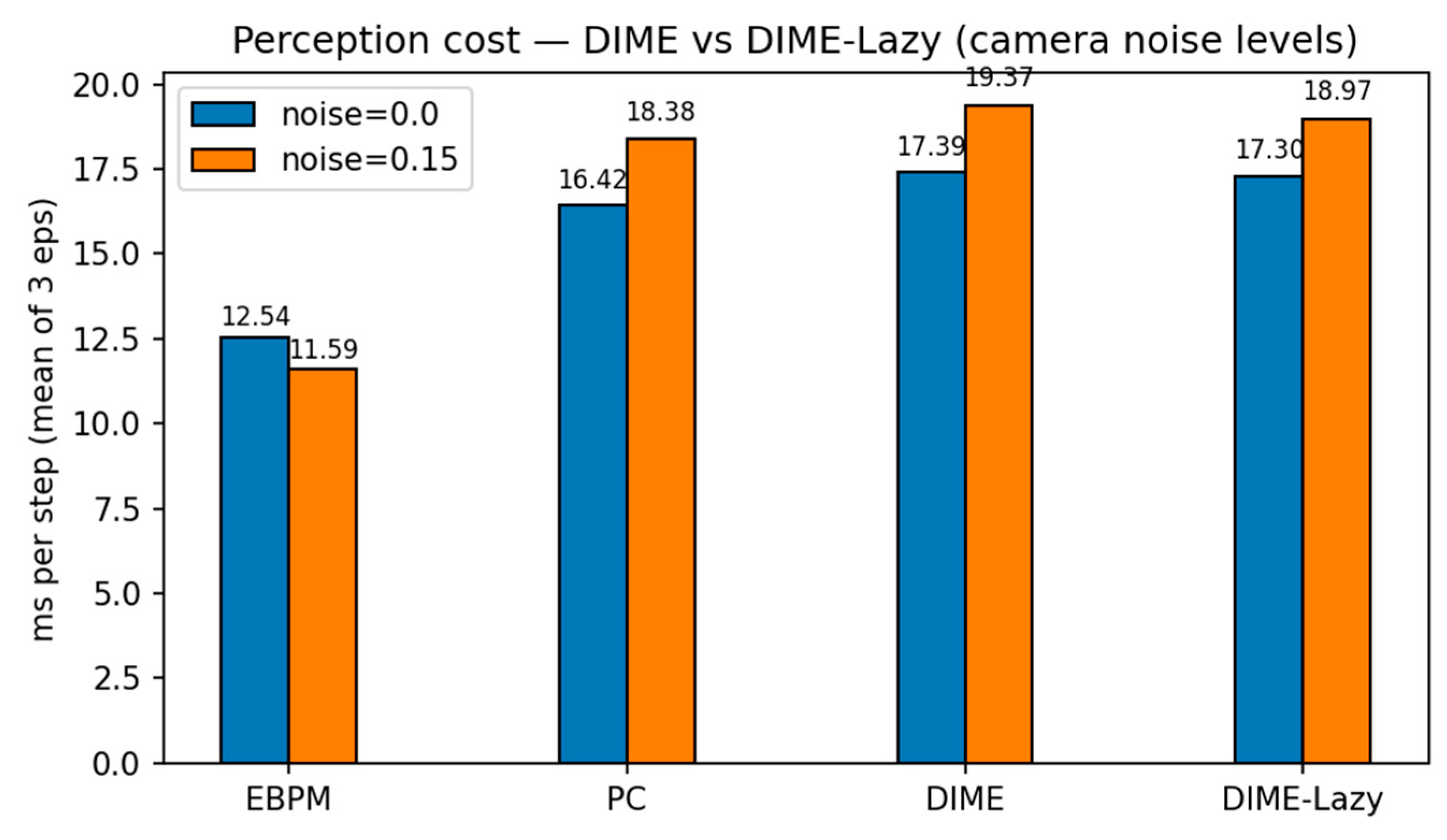
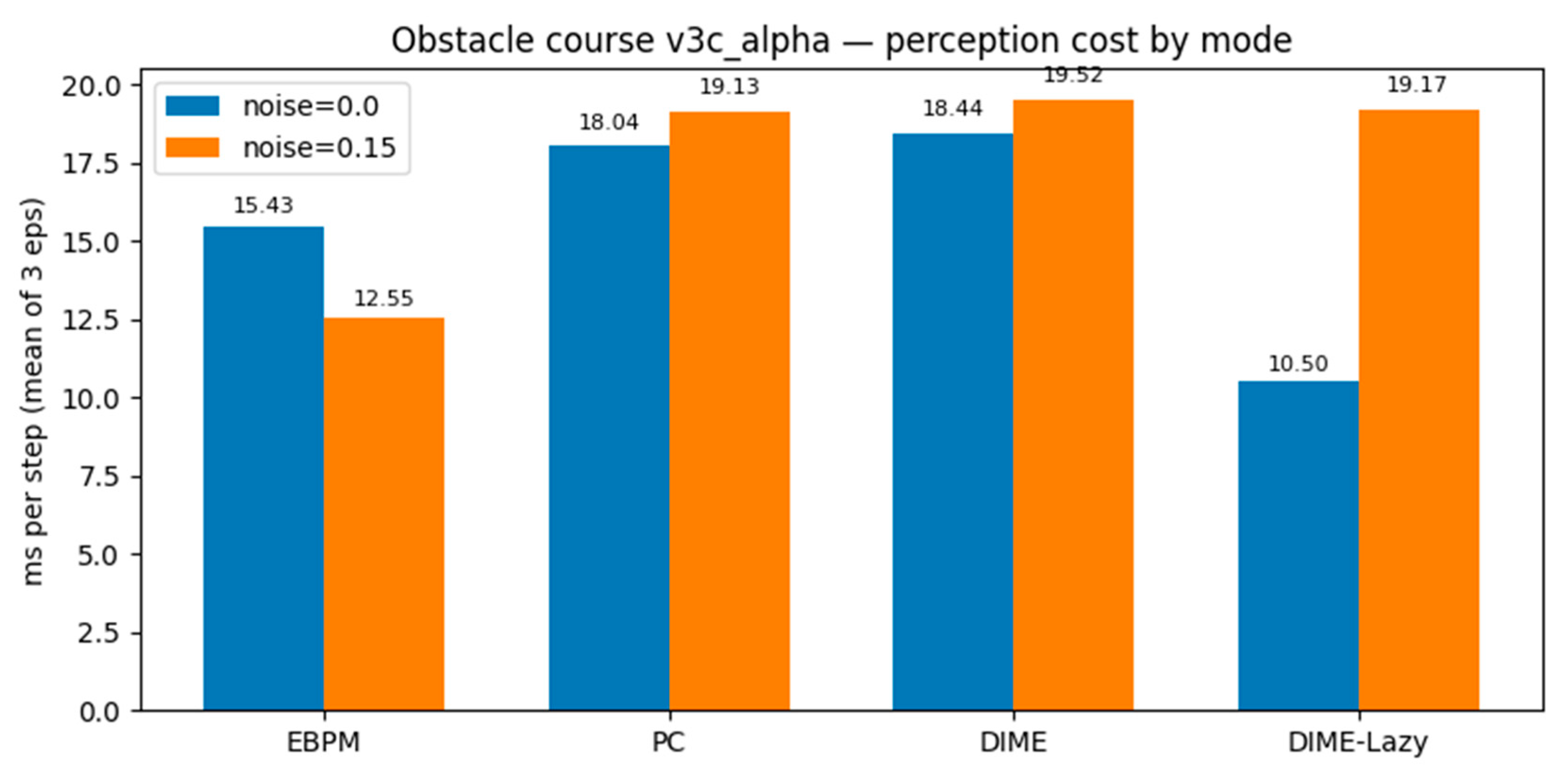
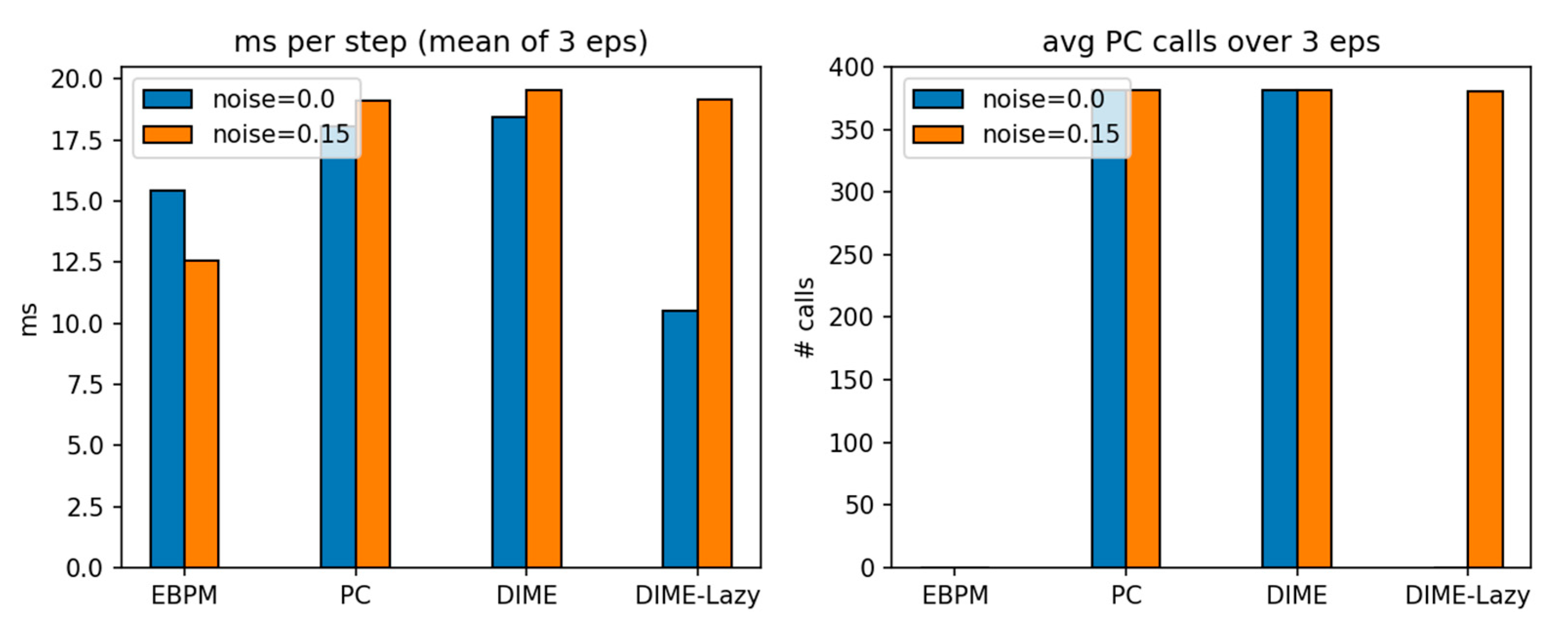
3.4. Virtual Robotics (Obstacle Course): Runtime-Aware DIME-Lazy (α Fixed)

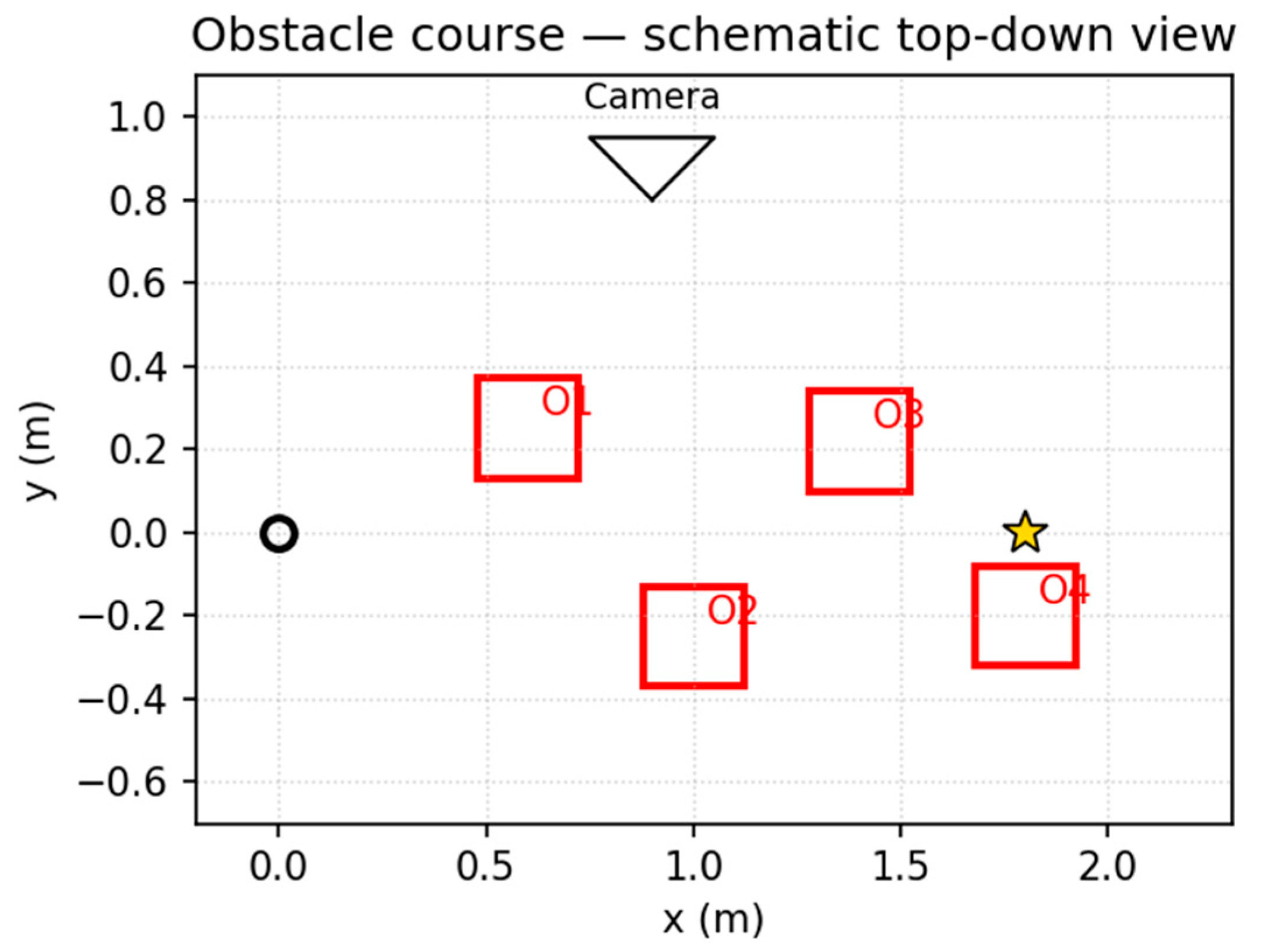
| mode | noise | ms/step (mean) |
steps (mean) |
success | collisions (mean) |
|---|---|---|---|---|---|
| EBPM | 0.00 | 15.43 | 381 | 1.00 | 113 |
| PC | 0.00 | 18.04 | 381 | 1.00 | 113 |
| DIME | 0.00 | 18.44 | 381 | 1.00 | 113 |
| DIME-Lazy | 0.00 | 10.50 | 381 | 1.00 | 113 |
| EBPM | 0.15 | 12.55 | 381 | 1.00 | 113 |
| PC | 0.15 | 19.13 | 381 | 1.00 | 113 |
| DIME | 0.15 | 19.52 | 381 | 1.00 | 113 |
| DIME-Lazy | 0.15 | 19.17 | 381 | 1.00 | 113 |
4. Discussion
4.1. Advantages of EBPM
4.2. Limitations of EBPM
4.3. Advantages of PC
4.4. Limitations of PC
4.5. Threats to Validity and Limitations
4.6. DIME as an Integrative Solution
- EBPM = fast, energy-efficient, but less flexible.
- PC = robust, adaptive, but computationally costly.
- DIME = hybrid integration, balancing both.
4.7. Interdisciplinary Implications
- Neuroscience: EBPM provides an alternative explanation for the phenomenon of memory replay, while DIME may serve as a more faithful framework for understanding the interaction between memory and prediction.
- Artificial Intelligence: EBPM-inspired architectures are more energy-efficient, and their integration with PC-type modules can enhance noise robustness.
5. Conclusions
- in neuroscience, it provides a more comprehensive explanation of the interaction between memory and prediction,
- in artificial intelligence, it suggests hybrid architectures that are more energy-efficient,
- in robotics, it paves the way for autonomous systems that can combine rapid recognition with adaptability to unfamiliar environments.
Data & Code Availability
Appendix A
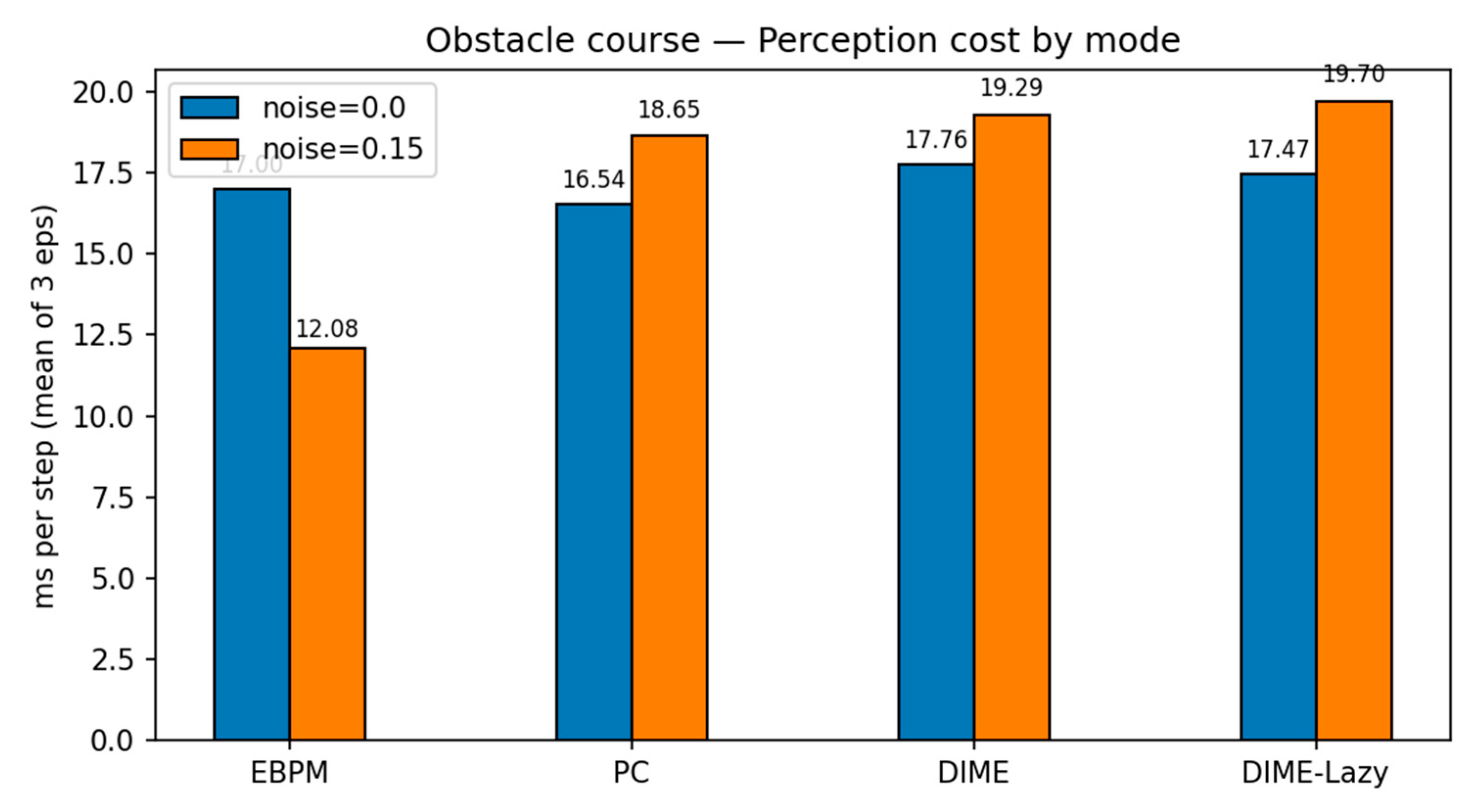
Appendix B
| File (relative path) | What it contains |
|---|---|
| results_csv/pybullet_obstacles_metrics_v3c_alpha.csv | v3c_alpha (α=0.65): ms/step mean, steps, success, collisions, avg PC calls per mode & noise |
| results_csv/pybullet_obstacles_metrics_v3c.csv | v3c (dyn gating, no fixed α): same fields |
| results_csv/pybullet_obstacles_metrics_v3b.csv | v3b: success & collisions breakdown |
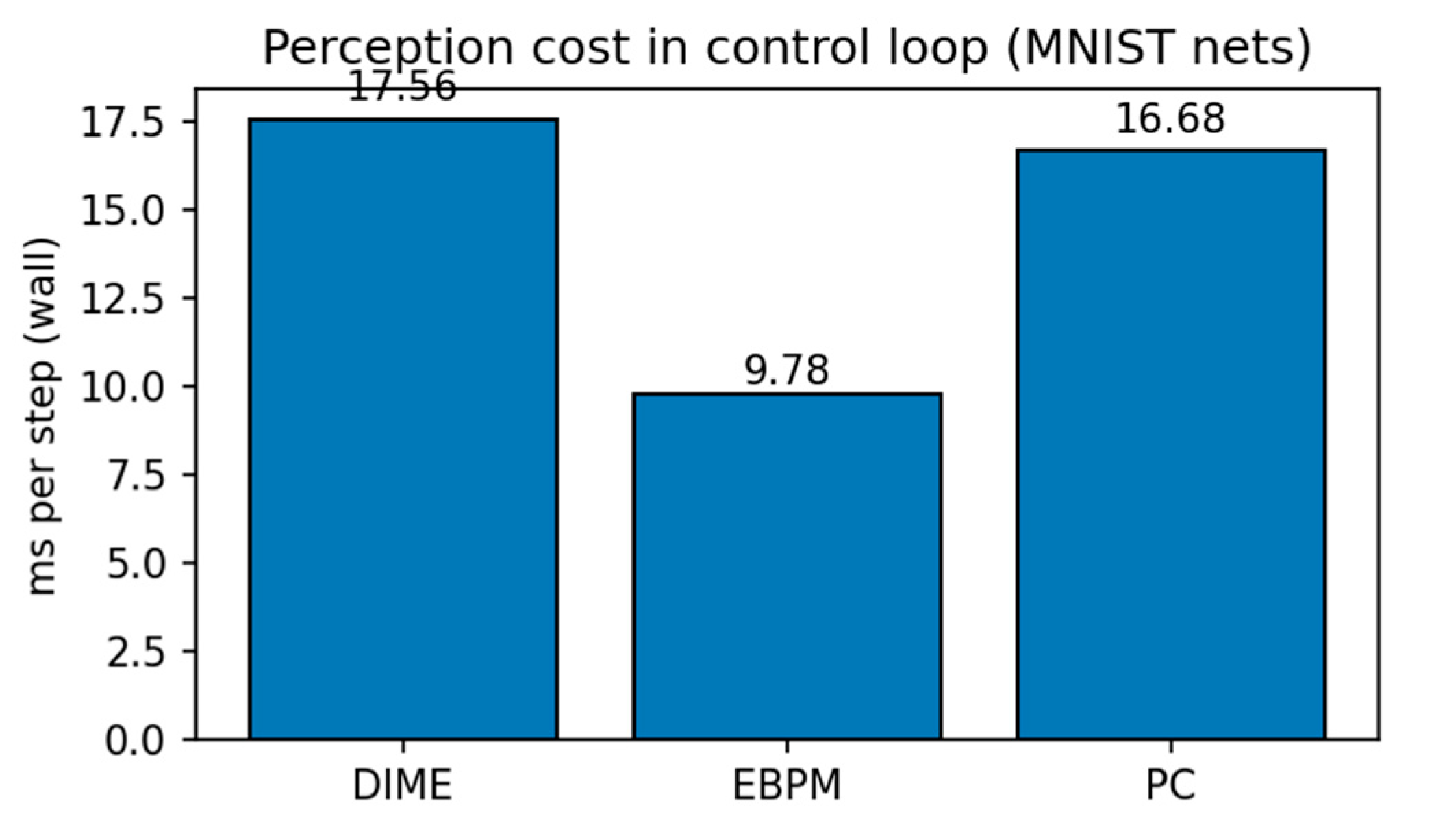
| results_csv/pybullet_perception_metrics.csv | demo perception ms/step EBPM vs PC vs DIME |
References
- K. Friston. The free-energy principle: a unified brain theory? Nat. Rev. Neurosci. 2010, 11, 127–138. [Google Scholar] [CrossRef]
- R. P. N. Rao and D. H. Ballard. Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nat. Neurosci. 1999, 2, 79–87. [Google Scholar] [CrossRef]
- R. Kanai, Y. Komura, S. Shipp, and K. Friston. Cerebral hierarchies: predictive processing, precision and the pulvinar. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140169. [Google Scholar] [CrossRef]
- G. Buzsáki. Neural Syntax: Cell Assemblies, Synapsembles, and Readers. Neuron 2010, 68, 362–385. [Google Scholar] [CrossRef]
- J. Hawkins and S. Ahmad. Why Neurons Have Thousands of Synapses, a Theory of Sequence Memory in Neocortex. Front. Neural Circuits 2016, 10. [CrossRef]
- H. Markram, W. H. Markram, W. Gerstner, and P. J. Sjöström. Spike-Timing-Dependent Plasticity: A Comprehensive Overview. Front. Synaptic Neurosci. 2012, 4. [Google Scholar] [CrossRef]
- J. J. Hopfield. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl. Acad. Sci. 1982, 79, 2554–2558. [Google Scholar] [CrossRef] [PubMed]
- J. L. McClelland, B. L. McNaughton, and R. C. O’Reilly. Why there are complementary learning systems in the hippocampus and neocortex: Insights from the successes and failures of connectionist models of learning and memory. Psychol. Rev. 1995, 102, 419–457. [Google Scholar] [CrossRef]
- T. J. Teyler and P. DiScenna. The hippocampal memory indexing theory. Behav. Neurosci. 1986, 100, 147–154. [Google Scholar] [CrossRef]
- E. T. Rolls. The mechanisms for pattern completion and pattern separation in the hippocampus. Front. Syst. Neurosci. 2013, 7. [Google Scholar] [CrossRef]
- R. M. Nosofsky. Attention, similarity, and the identification–categorization relationship. J. Exp. Psychol. Gen. 1986, 115, 39–57. [Google Scholar] [CrossRef]
- G. D. Logan. Toward an instance theory of automatization. Psychol. Rev. 1988, 95, 492–527. [Google Scholar] [CrossRef]
- A. Biederman. Recognition-by-components: A theory of human image understanding. Psychol. Rev. 1987, 94, 115–147. [Google Scholar] [CrossRef]
- P. Kanerva, Sparse distributed memory. Cambridge, Mass: MIT Press, 1988.
- I. Graves, G. Wayne, and I. Danihelka. Neural Turing Machines. 2014, arXiv. [CrossRef]
- A. Graves et al.. Hybrid computing using a neural network with dynamic external memory. Nature 2016, 538, 471–476. [Google Scholar] [CrossRef] [PubMed]
- A. Blundell et al.. Model-Free Episodic Control. 2016, arXiv. arXiv. [CrossRef]
- J. Snell, K. J. Snell, K. Swersky, and R. S. Zemel. Prototypical Networks for Few-shot Learning. 2017, arXiv. [CrossRef]
- I. Clark. Whatever next? Predictive brains, situated agents, and the future of cognitive science. Behav. Brain Sci. 2013, 36, 181–204. [Google Scholar] [CrossRef] [PubMed]
- A. M. Bastos, W. M. Usrey, R. A. Adams, G. R. Mangun, P. Fries, and K. J. Friston. Canonical Microcircuits for Predictive Coding. Neuron 2012, 76, 695–711. [Google Scholar] [CrossRef]
- A. A. Olshausen and D. J. Field. Emergence of simple-cell receptive field properties by learning a sparse code for natural images. Nature 1996, 381, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Y. Bengio, A. Courville, and P. Vincent. Representation Learning: A Review and New Perspectives. IEEE Trans. Pattern Anal. Mach. Intell. 2013, 35, 1798–1828. [Google Scholar] [CrossRef]
- L. Itti and C. Koch. Computational modelling of visual attention. Nat. Rev. Neurosci. 2001, 2, 194–203. [Google Scholar] [CrossRef]
- G. Aston-Jones and J. D. Cohen. AN INTEGRATIVE THEORY OF LOCUS COERULEUS-NOREPINEPHRINE FUNCTION: Adaptive Gain and Optimal Performance. Annu. Rev. Neurosci. 2005, 28, 403–450. [Google Scholar] [CrossRef]
- S. Dehaene, F. Meyniel, C. Wacongne, L. Wang, and C. Pallier. The Neural Representation of Sequences: From Transition Probabilities to Algebraic Patterns and Linguistic Trees. Neuron 2015, 88, 2–19. [Google Scholar] [CrossRef] [PubMed]
- R. J. Douglas and K. A. C. Martin. Recurrent neuronal circuits in the neocortex. Curr. Biol. 2007, 17, R496–R500. [Google Scholar] [CrossRef] [PubMed]
- G. G. Ambrus. Shared neural codes of recognition memory. Sci. Rep. 2024, 14, 15846. [Google Scholar] [CrossRef]
- W. Gerstner, W. M. Kistler, R. Naud, and L. Paninski, Neuronal Dynamics: From Single Neurons to Networks and Models of Cognition, 1st ed. Cambridge University Press, 2014. [CrossRef]
- J. St. B. T. Evans and K. E. Stanovich. Dual-Process Theories of Higher Cognition: Advancing the Debate. Perspect. Psychol. Sci. 2013, 8, 223–241. [Google Scholar] [CrossRef]
- U. Hasson, J. Chen, and C. J. Honey. Hierarchical process memory: memory as an integral component of information processing. Trends Cogn. Sci. 2015, 19, 304–313, June. [Google Scholar] [CrossRef]
- A. Summerfield and F. P. De Lange. Expectation in perceptual decision making: neural and computational mechanisms. Nat. Rev. Neurosci. 2014, 15, 745–756. [Google Scholar] [CrossRef]
- P. Kok, J. F. M. Jehee, and F. P. de Lange. Less Is More: Expectation Sharpens Representations in the Primary Visual Cortex. Neuron 2012, 75, 265–270. [Google Scholar] [CrossRef]
- S. J. Kiebel, J. Daunizeau, and K. J. Friston. A Hierarchy of Time-Scales and the Brain. PLoS Comput. Biol. 2008, 4, e1000209. [Google Scholar] [CrossRef]
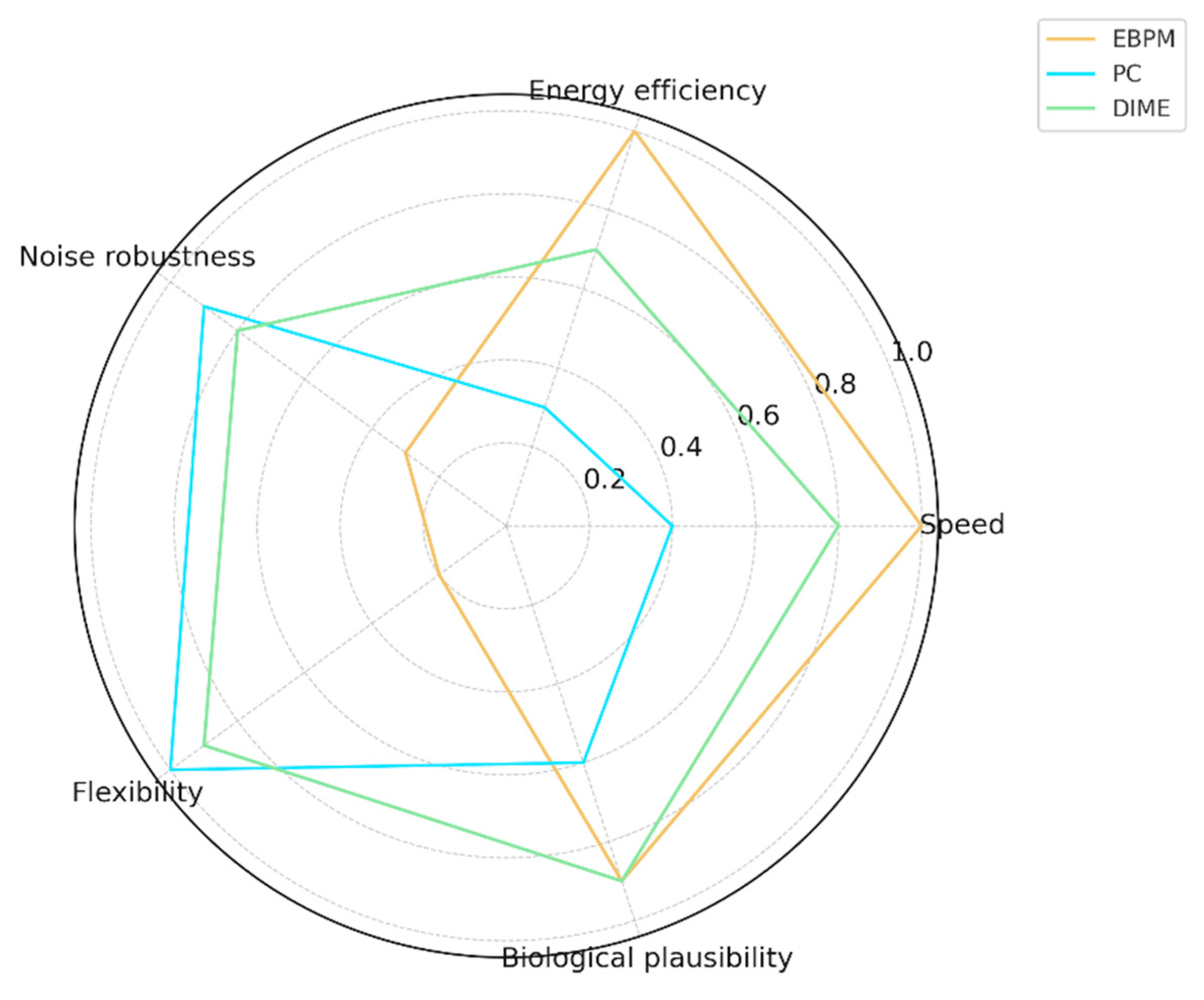
| Aspect | EBPM | PC | DIME |
|---|---|---|---|
| Speed | High | Medium | High |
| Energy Efficiency | High | Low | Medium–High |
| Noise Robustness | Medium-Low | High | High |
| Flexibility | Low | High | High |
| Biological Plausibility | High | Medium | High |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




