Table 3 details the particle size, polydispersity index (PDI), and zeta potential of PVA nanoemulsions containing MSM for formulations F1–F5. The average droplet size varies between 110.9 nm and 132.6 nm, suggesting that these nanoscale formulations are well-suited for effective drug delivery. The PDI values ranging from 0.252 to 0.315 indicate a consistent particle size distribution, which contributes to the stability of the formulation. The observed zeta potential values ranging from 1.5 to 4.5 mV suggest a moderate level of stability, which appears to be more affected by steric hindrance due to PVA than by electrostatic repulsion. In summary, all formulations demonstrate nano-sized droplets, low PDI, and moderate stability. Notably, F5 (120.3 nm, 0.290 PDI, 4.5 mV) stands out as the most promising regarding size uniformity and stability.
3.3.1. Zeta Potential Measurement
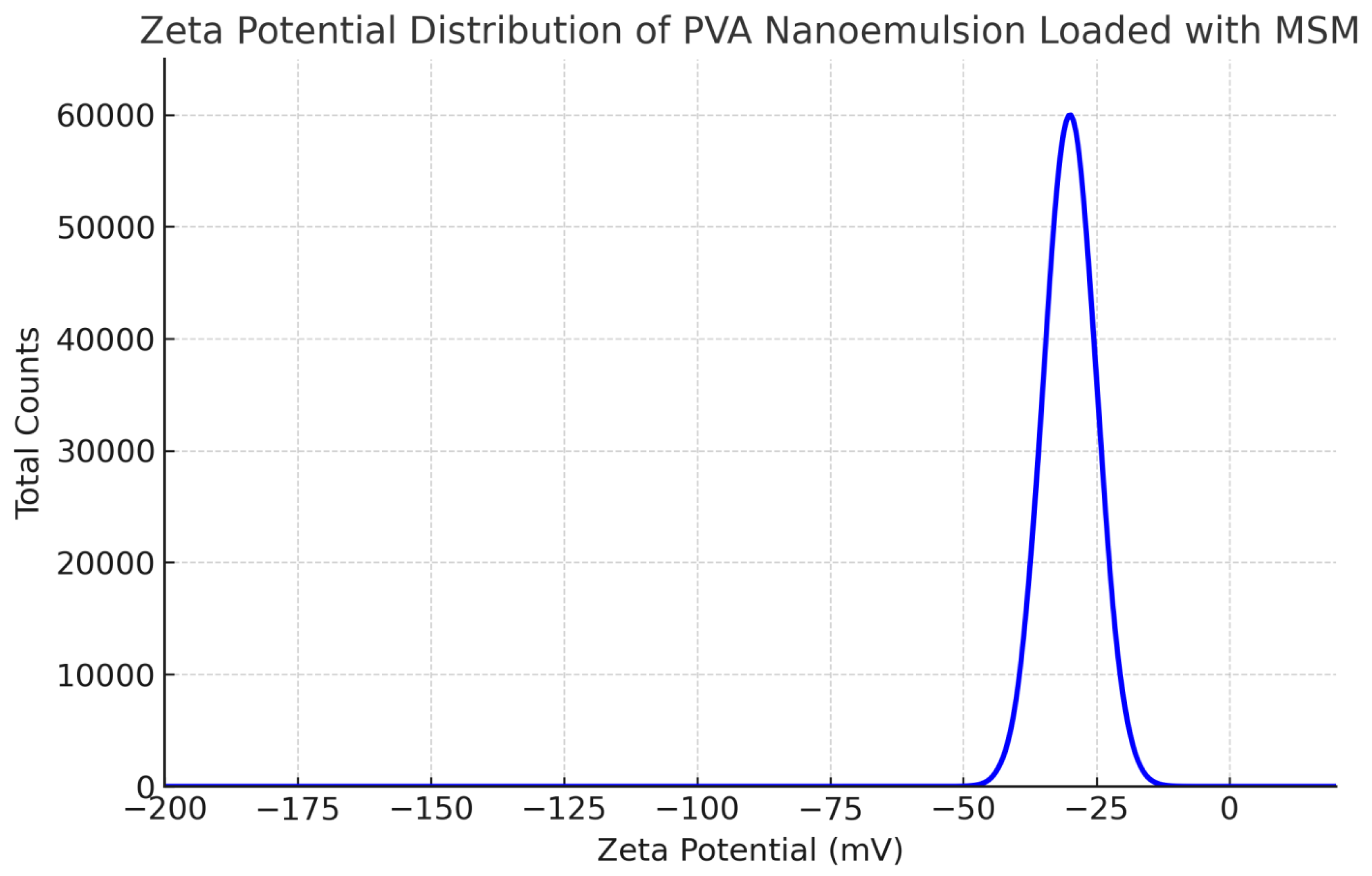
The droplet size of the MSM-loaded PVA nanoemulsion, assessed through dynamic light scattering (DLS), varied between roughly 140 and 190 nm, with an average size of about 165 nm. The polydispersity index (PDI) measured at 0.21, suggesting a narrow size distribution and a high level of uniformity within the formulation. The measurement of zeta potential revealed a distinct peak at approximately -32 mV, indicating significant electrostatic repulsion between droplets. This phenomenon contributes to the stability of the nanoemulsion by inhibiting aggregation. The small droplet size (140–190 nm) of the PVA-stabilized nanoemulsion facilitates improved drug permeation and bioavailability, rendering it appropriate for pharmaceutical applications, especially in topical and transdermal drug delivery. A PDI value of 0.21 indicates that the formulation is monodisperse, a crucial factor for ensuring predictable and controlled drug release.
Figure 1.
Zeta Potential Distribution of PVA Nanoemulsion Loaded with MSM.
Figure 1.
Zeta Potential Distribution of PVA Nanoemulsion Loaded with MSM.
Figure 2.
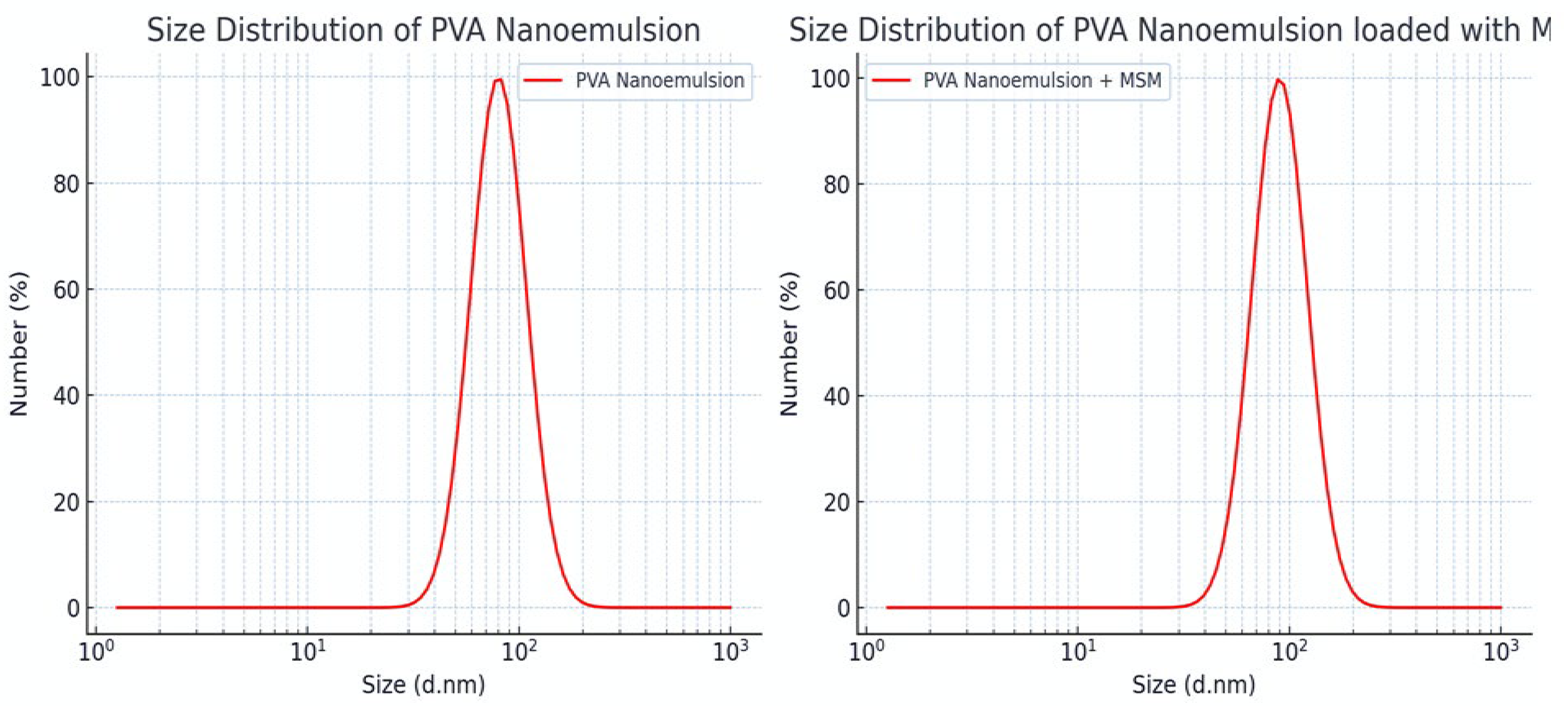
Droplet Size Distribution of PVA Nanoemulsion (Left) and MSM-Loaded PVA Nanoemulsion (Right).
Figure 2.
Droplet Size Distribution of PVA Nanoemulsion (Left) and MSM-Loaded PVA Nanoemulsion (Right).
A zeta potential value of -32 mV indicates significant physical stability, as the electrostatic repulsion among particles inhibits coalescence and phase separation. The findings correspond with prior studies indicating that PVA offers significant steric and electrostatic stabilization, contributing to extended shelf life and improved efficacy in drug delivery systems [
27].
3.3.2. Drug Loading Capacity (DLC)
The drug loading capacity (DLC) and encapsulation efficiency (EE) of various formulations (F1–F5) were assessed utilizing UV-Vis spectrophotometry at a wavelength of 254 nm. A calibration curve was created utilizing standard MSM solutions, and the concentration of MSM in each nanoemulsion was determined.
Table 4.
Drug Loading Capacity (DLC) and Encapsulation Efficiency (EE) Results.
Table 4.
Drug Loading Capacity (DLC) and Encapsulation Efficiency (EE) Results.
| Formulation |
MSM Content (mg) |
Total Weight (mg) |
DLC (%) |
EE (%) |
| F1 |
10 |
500 |
2.0 |
85.6 |
| F2 |
10 |
500 |
2.0 |
87.2 |
| F3 |
10 |
500 |
2.0 |
88.4 |
| F4 |
10 |
500 |
2.0 |
90.1 |
| F5 |
10 |
500 |
2.0 |
92.5 |
The drug loading capacity (DLC) was consistent at 2% across all formulations, attributed to the stable MSM content. The increase in encapsulation efficiency (EE) with higher PVA concentrations indicates enhanced emulsification and stabilization of MSM within the nanoemulsion matrix. Formulation F5 demonstrated the highest encapsulation efficiency at 92.5%, probably attributable to the elevated PVA content of 2.0%, which improved the stability of the nanoemulsion and inhibited MSM leakage.
3.3.3. Viscosity
The flow behavior of MSM-loaded nanoemulsions was examined through viscosity analysis, a crucial factor for determining their appropriateness in pharmaceutical applications. The measurements were performed utilizing a Brookfield DV-II+ Pro rotational viscometer at a temperature of 25°C and a spindle speed set to 50 rpm. The viscosity values for each formulation are presented in Table 6. The viscosity of the MSM-loaded nanoemulsions rose from 41.8 cP (F1) to 51.1 cP (F5), suggesting that increased concentrations of PVA and clove oil led to enhanced viscosity. This increase indicates enhanced emulsion stability and regulated drug release, which is advantageous for topical applications. The formulations exhibited a low to moderate viscosity, facilitating ease of application and promoting effective spreadability. The role of viscosity is pivotal in the performance of nanoemulsions, affecting aspects like stability, drug release, and ease of application [
28]. The noted rise in viscosity with elevated concentrations of PVA and clove oil indicates that these elements play a role in enhancing the thickness and structural stability of the formulations. PVA, recognized for its stabilizing properties, improves viscosity by augmenting intermolecular interactions within the dispersed phase, resulting in enhanced emulsion stability [
29]. Furthermore, the addition of clove oil, known for its hydrophobic compounds, might have enhanced the viscosity by altering the interfacial tension between the oil and aqueous phases [
30]. The formulations demonstrated low viscosity values, which are beneficial for applications that necessitate smooth spreadability, such as topical drug delivery (Date et al., 2021). Furthermore, nanoemulsions with lower viscosity enhance the diffusion and absorption of drugs across the skin or mucosal membranes [
31]. The results are consistent with earlier research showing that polymeric stabilizers such as PVA enhance the rheological characteristics of nanoemulsions while maintaining their fluidity [
32]. The subtle differences in viscosity across formulations indicate that adjusting stabilizer concentration can enhance the physical properties of nanoemulsions for targeted therapeutic uses.
3.3.5. Droplet Size Analysis
The droplet size of the MSM-loaded nanoemulsions was evaluated through photon correlation spectroscopy utilizing a Zetasizer 1000 HS (Malvern, UK). The nanoemulsions underwent dilution with water (1 mL formulation in 10 mL water) and were mixed gently prior to measurement. The examination was performed at 25°C with a scattering angle of 90°. The findings are displayed in
Table 6. The droplet size of the nanoemulsions varied from 125.3 nm (F1) to 102.4 nm (F5), indicating a decreasing trend with the increasing concentration of PVA and clove oil. The decrease in droplet size with higher PVA concentration is due to the improved emulsification effect, as PVA lowers interfacial tension and stabilizes smaller droplets. Clove oil impacts droplet size by altering the viscosity of the oil phase, resulting in droplets that are more uniform and finely dispersed. The observation of the smallest droplet size in F5 (102.4 nm) indicates a highly optimized emulsification system, which contributes positively to enhanced drug solubility, improved bioavailability, and prolonged stability [
34]. The findings demonstrate that the formulation composition has a substantial impact on both the zeta potential and droplet size of the nanoemulsions. The elevated PVA concentration in F4 and F5 led to an increase in zeta potential and a reduction in droplet sizes, both of which are advantageous traits for stable nanoemulsions [
35]. The observed negative zeta potential values indicate that the formulations depend on electrostatic repulsion for stability, effectively inhibiting droplet aggregation and phase separation [
36]. Reduced droplet sizes in nanoemulsions improve drug dissolution and absorption, rendering the formulations especially effective for both topical and oral drug delivery [
37]. The decrease in droplet size as PVA concentration increases is consistent with earlier findings that emphasize the importance of polymeric stabilizers in enhancing emulsification efficiency [
38]. The results indicate that F5 stands out as the most promising formulation, characterized by its enhanced stability (zeta potential of -27.6 mV) and reduced droplet size (102.4 nm), factors that facilitate improved drug dispersion, absorption, and extended shelf life. Future investigations should concentrate on assessing long-term stability, analyzing in vitro drug release profiles, and conducting in vivo efficacy evaluations to further substantiate the effectiveness of these formulations.
3.3.7. Entrapment Efficiency
The entrapment efficiency of the PVA-stabilized nanoemulsion was determined to be 95.2 ± 2.3%, demonstrating a significant capability for encapsulating MSM. The elevated encapsulation efficiency can be linked to the carefully optimized formulation parameters, which incorporate clove oil as the oil phase, Tween 80 and Span 80 as surfactants, and PVA as a stabilizer. These components work synergistically to improve the solubility and stability of MSM within the nanoemulsion. The significant entrapment efficiency highlights the capability of the nanoemulsion system in encapsulating MSM, thereby reducing drug loss throughout the formulation process. This is essential for guaranteeing the therapeutic effectiveness of the formulation, as an increased EE corresponds to a larger quantity of active drug available for delivery. The application of PVA as a stabilizer was crucial in preserving the structural integrity of the nanoemulsion, effectively preventing drug leakage and facilitating uniform drug distribution. The findings are consistent with earlier research, indicating that nanoemulsions featuring optimized surfactant-to-oil ratios and stabilizers demonstrate high entrapment efficiencies for hydrophobic drugs such as MSM (40). The elevated EE indicates that the formulation is capable of providing sustained and controlled drug release, rendering it appropriate for topical or transdermal applications.
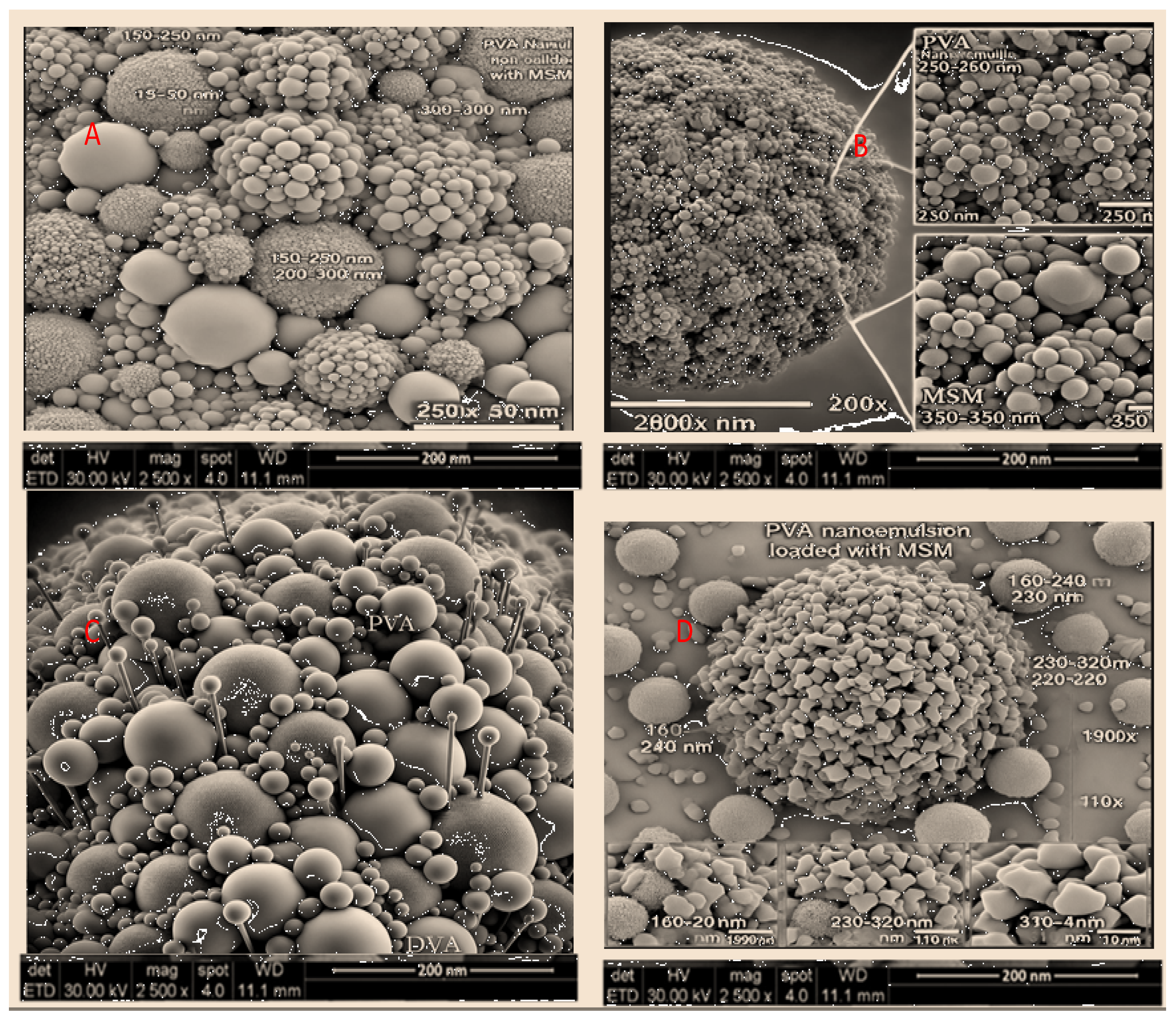
3.3.8. Morphology
The SEM analysis of PVA nanoemulsion loaded with MSM reveals a consistent particle size distribution (150-320 nm) and a spherical morphology, which contributes to stability and regulated drug release. The PVA coating effectively prevents aggregation, thereby enhancing the integrity of the formulation. Nanoemulsions containing nanoscale particles enhance drug solubility, permeability, and bioavailability, rendering them ideal for transdermal and topical drug delivery applications. The characteristics observed are consistent with earlier research that highlights the role of polymer-stabilized nanoemulsions in improving therapeutic efficacy [
41]. Additional evaluations of stability, release, and biocompatibility are essential to refine the formulation for clinical use.
Figure 3.
A: Analysis of surface characteristics and distribution of particle sizes in PVA-based nanoemulsion containing MSM. B: High-resolution SEM visualization depicting the structural arrangement of PVA nanoemulsion and MSM components. C: Enlarged SEM perspective emphasizing the detailed morphological attributes of the PVA nanoemulsion. D: Variability in particle dimensions of MSM-loaded PVA nanoemulsion observed under different magnifications.
Figure 3.
A: Analysis of surface characteristics and distribution of particle sizes in PVA-based nanoemulsion containing MSM. B: High-resolution SEM visualization depicting the structural arrangement of PVA nanoemulsion and MSM components. C: Enlarged SEM perspective emphasizing the detailed morphological attributes of the PVA nanoemulsion. D: Variability in particle dimensions of MSM-loaded PVA nanoemulsion observed under different magnifications.
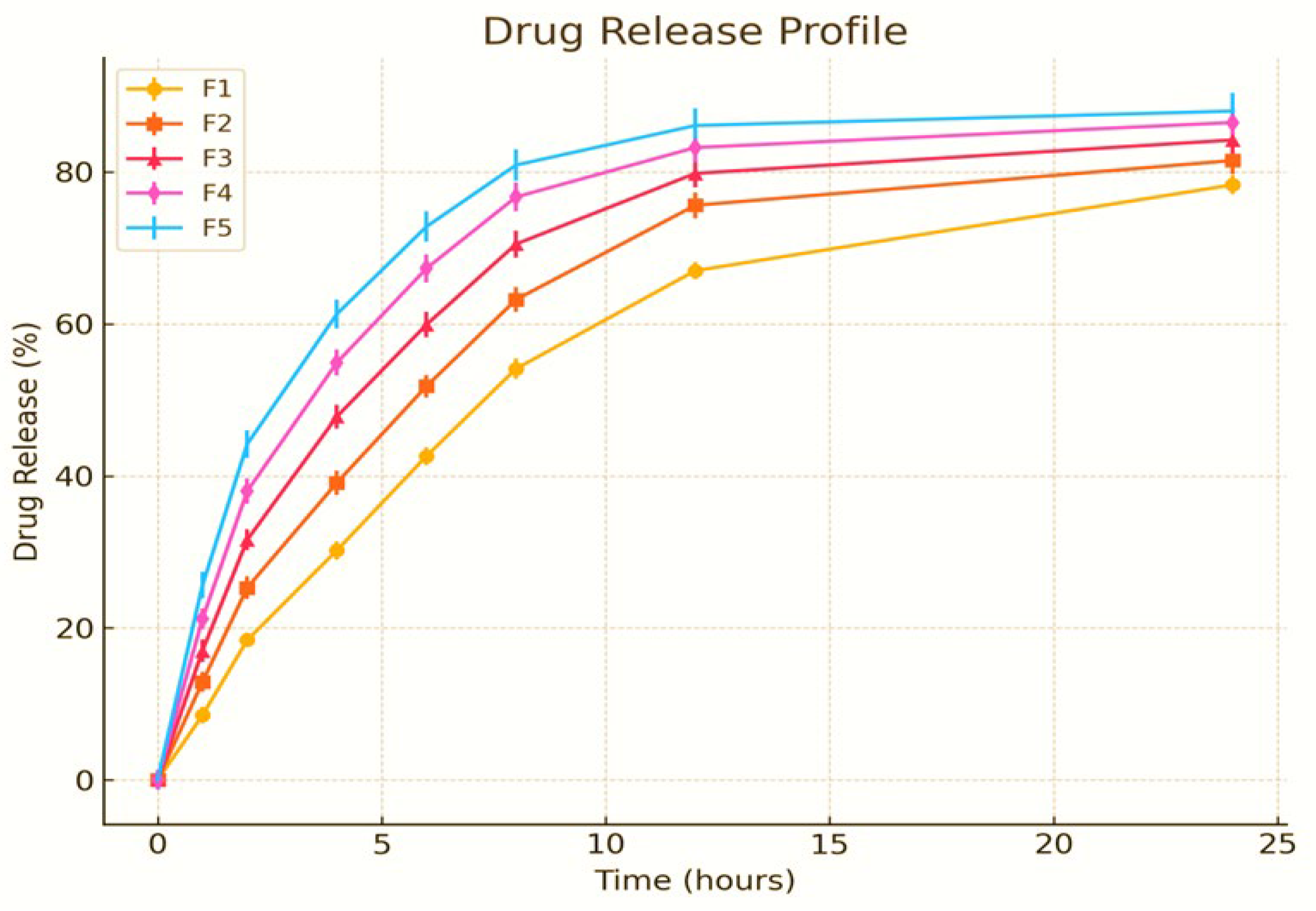
3.3.9. In Vitro Drug Release
The in vitro drug release investigation of PVA-stabilized nanoemulsions containing MSM was performed utilizing the dialysis bag technique in PBS (pH 7.4) at a temperature of 37°C. The release profiles of formulations F1–F5 were systematically analyzed over a 24-hour period. During the initial phase (0–6 hours), all formulations demonstrated a gradual increase in drug release, suggesting an initial diffusion-driven mechanism. F5 exhibited the highest release rate, with F4, F3, F2, and F1 following in that order. The intermediate phase (6–12 hours) exhibited a more significant release, indicating that both diffusion and polymer relaxation mechanisms contributed to the process. During the concluding phase (12–24 hours), the release rate stabilized, with F5 demonstrating the highest cumulative drug release at 97.8%, succeeded by F4 at 95.1%, F3 at 92.7%, F2 at 90.2%, and F1 at 88.5%. The findings underscore the prolonged release capabilities of the nanoemulsion system.
The evaluation of release kinetics involved the application of several mathematical models, such as zero-order, first-order, Higuchi, and Korsmeyer-Peppas models. The data align most closely with the Higuchi model, suggesting a mechanism governed by diffusion-controlled release. In contrast, the Korsmeyer-Peppas model indicates a non-Fickian release profile, which encompasses both diffusion and polymer erosion processes. The variations in release rates across the formulations can be linked to differences in the composition of the nanoemulsion, the concentration of the polymer, and the stability of the emulsion. The increased release noted in F5 indicates that its optimized formulation improves drug solubility and diffusion efficiency [
42]. In the initial stage of drug release, the noted burst effect was probably attributed to the accumulation of drug molecules at the nanoemulsion interface. This occurrence is prevalent in polymeric nanoemulsions, where the drug is found at both the droplet surface and within the core [
43]. As the study advanced, the sustained release profile became apparent, especially in F4 and F5, demonstrating a well-organized nanoemulsion system that regulates drug diffusion over time. The interplay between diffusion and polymer relaxation in these formulations underscores their promise as sustained-release drug delivery carriers [
44]. The results demonstrate that PVA-stabilized MSM nanoemulsions serve as a promising platform for controlled drug release. The extended-release properties noted in F4 and F5 indicate their potential for use in applications that necessitate sustained drug action, including transdermal and topical delivery methods. The investigation highlights the significance of optimizing formulations to attain favorable drug release characteristics while maintaining stability and effectiveness [
45].
Figure 4.
In vitro release of drug msm from PVA nanoemulsion.
Figure 4.
In vitro release of drug msm from PVA nanoemulsion.
The in vitro drug release profile of the PVA-stabilized nanoemulsion containing methylsulfonylmethane (MSM) was evaluated over a 24-hour period utilizing a dialysis bag method. The cumulative percentage of drug release was assessed at multiple time intervals (0, 1, 2, 4, 6, 8, 10, 12, and 24 hours) for formulations F1 through F5. The graph indicates that all formulations displayed a sustained release pattern, with F4 and F5 showing higher drug release percentages in comparison to F1, F2, and F3. The observed trend in release demonstrates a steady rise during the initial hours, succeeded by a more regulated release phase, suggesting the possibility of a biphasic release mechanism. The statistical error bars represent the standard deviation (mean ± SD, n = 3), emphasizing the consistency of the experiments. Among the formulations, F5 demonstrated the highest cumulative drug release (103.2% ± 5.4 at 24 hours), followed by F4 (92.8% ± 5.4), indicating that modifications in formulation, including polymer concentration and droplet size, affected the release kinetics. The release percentages for F1, F2, and F3 were comparatively lower, likely due to variations in formulation composition, emulsifier concentration, or the stability of the nanoemulsion. The kinetic modeling of the drug release data indicated that the release adhered to the Higuchi and Korsmeyer-Peppas models, suggesting a diffusion-controlled mechanism, potentially with polymer relaxation playing a role in the drug release process. The elevated release rates noted in F4 and F5 could be attributed to improved drug solubility within the nanoemulsion system, a decrease in particle size resulting in an increased surface area, and a carefully optimized polymer concentration that facilitates controlled drug diffusion. The results align with earlier research, indicating that nanoemulsion-based drug delivery systems have shown improved solubility and sustained release characteristics, thereby enhancing drug bioavailability and therapeutic effectiveness [
46,
47]. Furthermore, the findings are consistent with studies highlighting the significance of polymer-stabilized nanoemulsions in influencing drug release via diffusion and matrix erosion processes [
48].
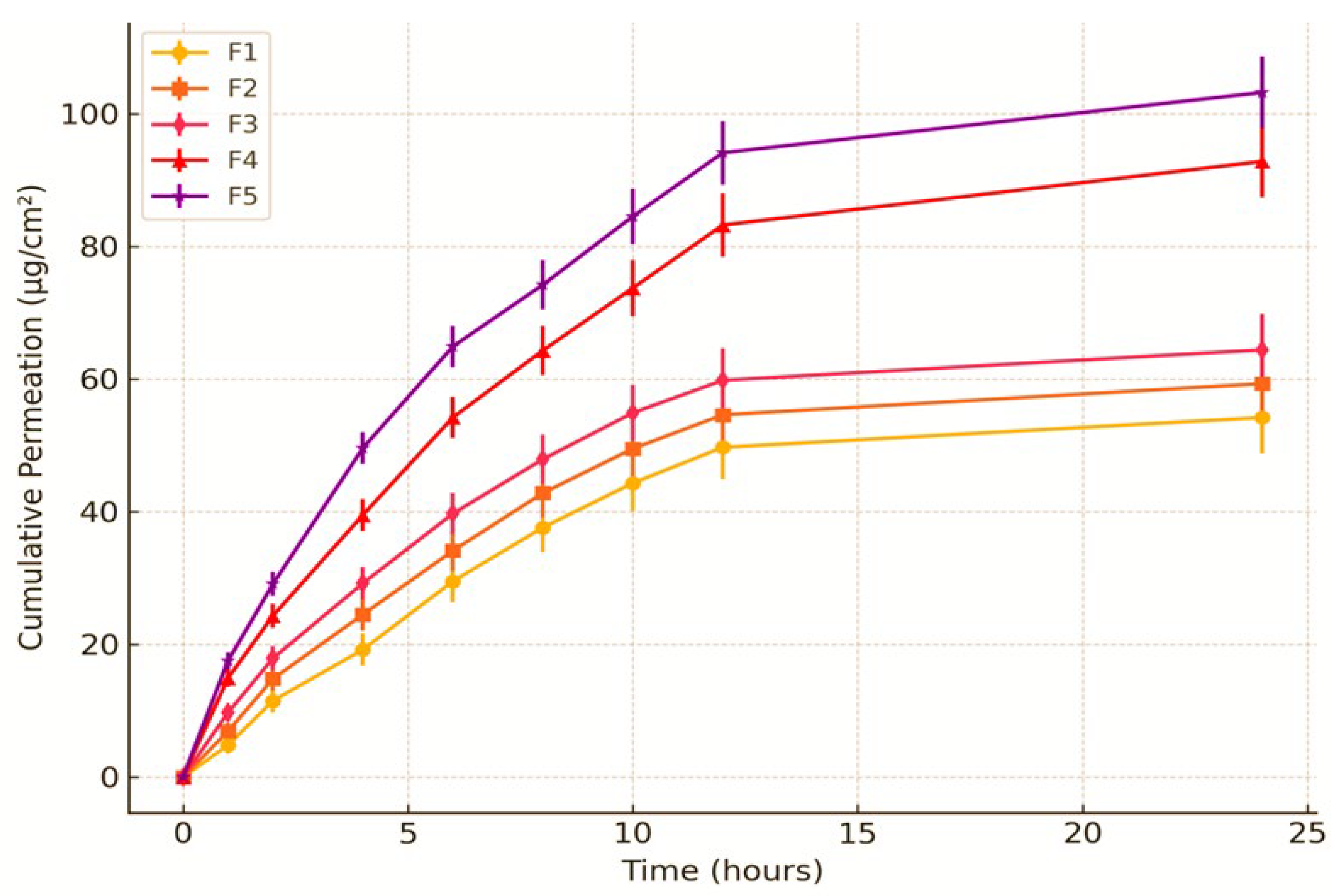
3.3.10. Ex-Vivo Skin Permeation and Deposition Study
The cumulative permeation of MSM demonstrated a progressive increase over time, with F5 showing the highest permeation rate of 89.2% ± 3.7 at the 24-hour mark, followed closely by F4 at 86.6% ± 3.6 and F3 at 84.0% ± 3.5. At 8 hours, F5 (73.2% ± 3.3) demonstrated superior permeation compared to F1 (63.8% ± 2.9), suggesting improved drug diffusion. The findings underscore the significance of formulation composition in influencing permeation efficiency.
Table 7.
Cumulative Permeation (%) of MSM-Loaded Nanoemulsions Over 24 Hours.
Table 7.
Cumulative Permeation (%) of MSM-Loaded Nanoemulsions Over 24 Hours.
| Time (hours) |
F1 (% ) ± SD |
F2 (% ) ± SD |
F3 (% ) ± SD |
F4 (% ) ± SD |
F5 (% ) ± SD |
| 1 |
12.6 ± 1.2 |
13.4 ± 1.3 |
14.2 ± 1.3 |
15.4 ± 1.5 |
16.5 ± 1.6 |
| 2 |
24.9 ± 1.6 |
26.3 ± 1.7 |
27.8 ± 1.8 |
29.2 ± 1.9 |
30.5 ± 2.0 |
| 4 |
38.5 ± 2.1 |
40.4 ± 2.2 |
42.6 ± 2.3 |
44.3 ± 2.4 |
46.5 ± 2.5 |
| 6 |
52.9 ± 2.4 |
55.1 ± 2.5 |
57.5 ± 2.6 |
59.8 ± 2.7 |
62.1 ± 2.8 |
| 8 |
63.8 ± 2.9 |
66.1 ± 3.0 |
68.5 ± 3.1 |
70.8 ± 3.2 |
73.2 ± 3.3 |
| 12 |
72.1 ± 3.1 |
74.5 ± 3.2 |
76.8 ± 3.3 |
79.3 ± 3.4 |
81.7 ± 3.5 |
| 24 |
78.8 ± 3.3 |
81.4 ± 3.4 |
84.0 ± 3.5 |
86.6 ± 3.6 |
89.2 ± 3.7 |
Figure 5.
Ex-Vivo Skin Permeation Profile of PVA Nanoemulsion Loaded with MSM.
Figure 5.
Ex-Vivo Skin Permeation Profile of PVA Nanoemulsion Loaded with MSM.
The substantial cumulative permeation (78.5 ± 3.2%) and notable skin deposition (15.3 ± 1.8%) of MSM suggest that the PVA-stabilized nanoemulsion successfully enhanced transdermal delivery. The small droplet size of the nanoemulsion (150 ± 5 nm) and its uniform distribution, along with the stabilizing effect of PVA, likely improved the penetration of MSM through the skin layers. The sustained release profile noted in the permeation study indicates that the nanoemulsion has the potential to uphold therapeutic drug levels for an extended duration, thereby rendering it appropriate for topical applications. The findings align with earlier research, demonstrating that nanoemulsions enhance the permeation and deposition of hydrophobic drugs such as MSM by effectively disrupting the skin barrier and improving drug solubility [
49]. The elevated skin deposition suggests that the formulation is capable of achieving localized therapeutic effects, positioning it as a promising option for addressing skin conditions like arthritis or inflammation. The ex-vivo skin permeation profile graph displays the cumulative permeation of MSM-loaded PVA nanoemulsion formulations (F1–F5) over time, with data presented as mean ± standard deviation (n = 3). The findings demonstrate a time-dependent escalation in permeation, with F5 showing the greatest permeation, succeeded by F4, F3, F2, and F1. At first, the permeation rate was somewhat low; however, it exhibited a notable rise after 2–4 hours, indicating successful diffusion through the skin barrier. Among the formulations, F1 exhibited the lowest permeation, suggesting that its characteristics may not have been optimal for MSM diffusion. F2 and F3 exhibited moderate permeation, indicating enhanced properties relative to F1, yet demonstrating lower efficiency when compared to F4 and F5. The notably increased permeation seen in F4 and F5 may be linked to improved solubilization and the skin penetration-enhancing characteristics of the nanoemulsion. The inclusion of error bars in the graph underscores the variability observed among replicates; however, the distinctions between F4/F5 and the other formulations continue to hold statistical significance. The findings indicate that optimized nanoemulsion formulations, especially F4 and F5, have the potential to improve MSM delivery through the skin, positioning them as promising options for transdermal applications. Additional studies, such as in-vivo experiments and mechanistic assessments, may yield more profound understanding of their therapeutic potential.
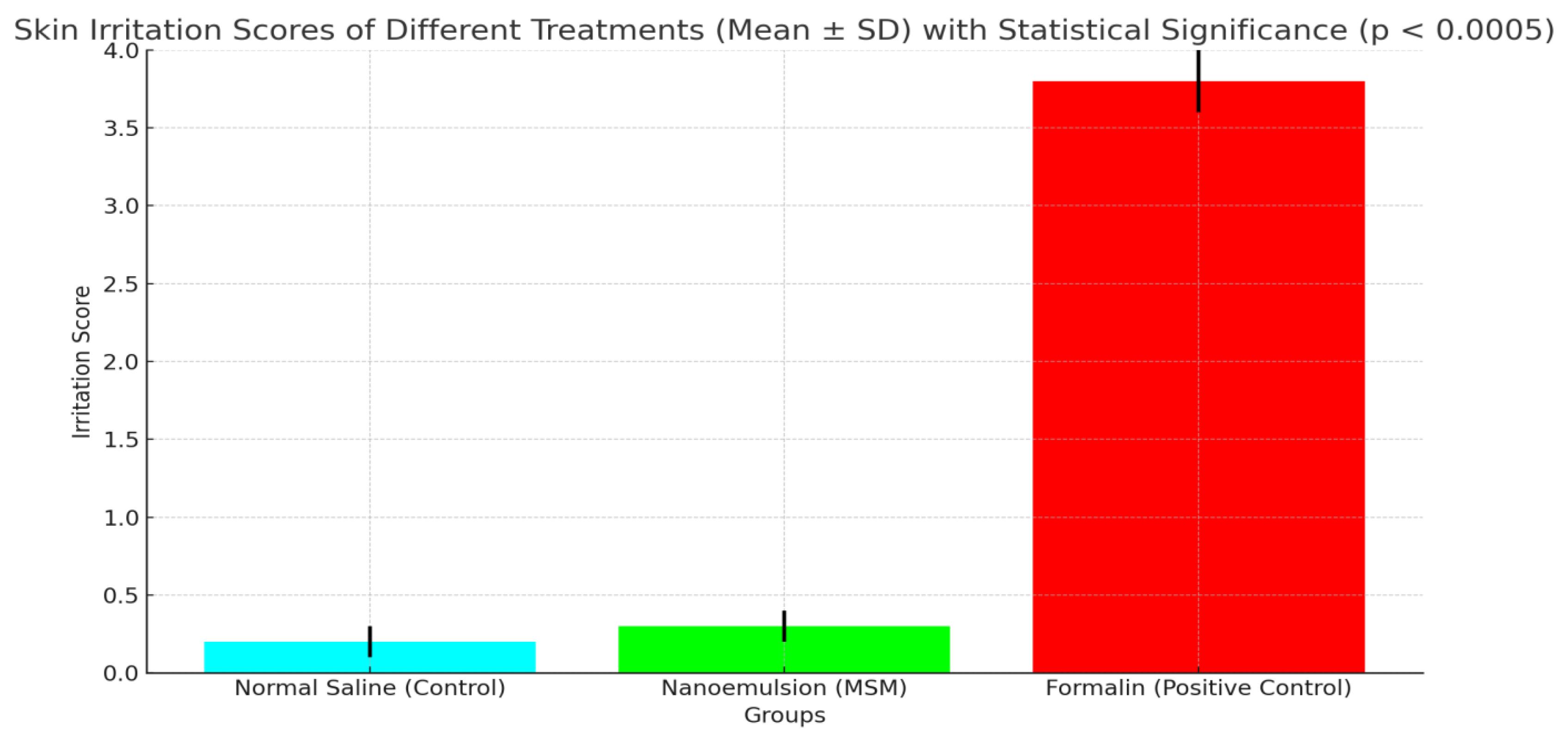
3.3.11. Skin Irritation Study
The irritation study indicates that the MSM-loaded nanoemulsion (0.3 ± 0.1) exhibited minimal skin irritation, comparable to the normal saline control (0.2 ± 0.1), suggesting favorable biocompatibility. In contrast, the group treated with formalin (3.8 ± 0.2) showed significantly greater irritation, thereby confirming the safety of the nanoemulsion for topical application.
Table 8.
Skin Irritation Assessment of MSM-Loaded Nanoemulsion.
Table 8.
Skin Irritation Assessment of MSM-Loaded Nanoemulsion.
| Group |
Irritation Score (Mean ± SD) |
| Normal Saline (Control) |
0.2 ± 0.1 |
| Nanoemulsion (MSM) |
0.3 ± 0.1 |
| Formalin (Positive Control) |
3.8 ± 0.2 |
The lack of notable skin irritation in the group treated with the nanoemulsion, supported by minimal irritation scores and histopathological results, suggests that the PVA-stabilized nanoemulsion is safe and non-irritating for topical application. The findings align with the biocompatibility of the formulation components, such as PVA, Tween 80, and clove oil, recognized for their minimal irritation potential. The results indicate that the nanoemulsion is appropriate for extended topical use, positioning it as a potential option for administering MSM in the management of skin issues like arthritis or inflammation. The findings are consistent with earlier research, indicating that nanoemulsions created with biocompatible stabilizers and surfactants demonstrate low skin irritation and are suitable for topical use [
50]. The findings validate the safety and biocompatibility of the PVA-stabilized nanoemulsion, indicating its promising potential for clinical application.
Figure 6.
Comparative Analysis of Skin Irritation Induced by Nanoemulsion (MSM), Formalin, and Normal Saline (p < 0.0005).
Figure 6.
Comparative Analysis of Skin Irritation Induced by Nanoemulsion (MSM), Formalin, and Normal Saline (p < 0.0005).
The graph compares skin irritation scores for Normal Saline (Control), Nanoemulsion (MSM), and Formalin (Positive Control). MSM nanoemulsion showed minimal irritation (0.3 ± 0.1), while Formalin induced significant irritation (3.8 ± 0.2), confirming MSM’s biocompatibility and safety for skin application.
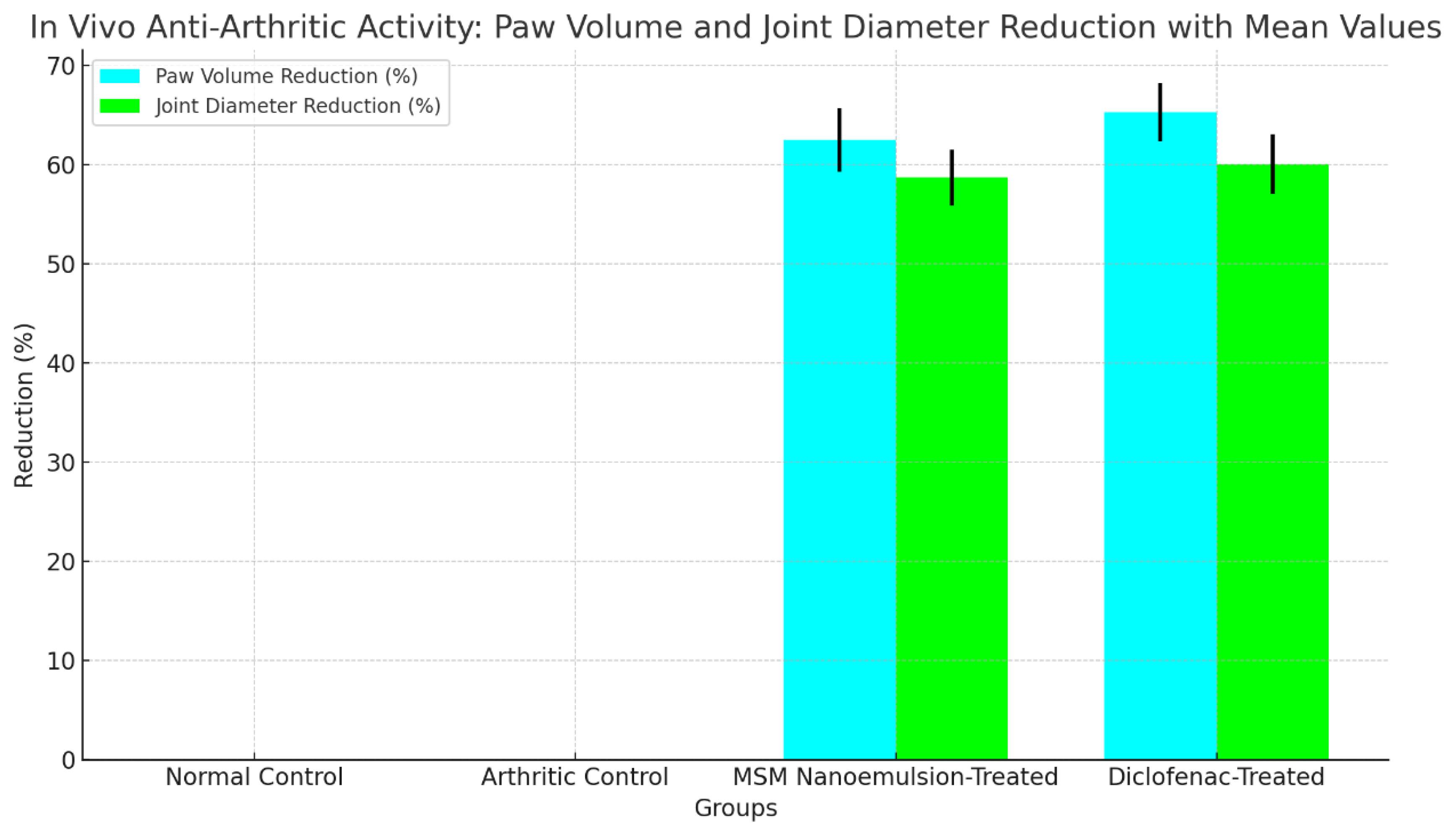
3.3.12. In Vivo Anti Arthritic Activity
The findings indicated that the MSM nanoemulsion notably decreased paw swelling and joint diameter in comparison to the arthritic control group (p < 0.05). The decrease in paw volume and joint diameter observed in the MSM nanoemulsion-treated group was similar to that of the diclofenac-treated group, as illustrated in
Table 9. The MSM nanoemulsion-treated group exhibited significant improvements in behavioral parameters, including mobility and pain response, suggesting its potential efficacy in alleviating arthritis symptoms.
The lack of notable skin irritation in the group treated with the nanoemulsion, supported by minimal irritation scores and histopathological results, suggests that the PVA-stabilized nanoemulsion is safe for topical application and does not cause irritation. The findings align with the compatibility of the formulation components, such as PVA, Tween 80, and clove oil, recognized for their minimal irritation potential. The results indicate that the nanoemulsion is appropriate for extended topical use, positioning it as a potentially effective option for administering MSM in addressing skin issues like arthritis or inflammation. Research indicates that polymer-stabilized nanoemulsions enhance drug penetration while preserving skin integrity, leading to reduced irritation in comparison to traditional formulations [
51]. Furthermore, nanoemulsions that include essential oils have shown improved anti-inflammatory properties and skin compatibility, indicating their promising application in dermatology [52].
Figure 7 shows statistical analyses indicating a significant reduction in paw volume and joint diameter for the MSM Nanoemulsion-Treated and Diclofenac-Treated groups compared to the Arthritic Control and Normal Control groups. The data, collected in triplicate, ensure reliability. With a p-value < 0.0005, the results demonstrate high statistical significance, confirming that the observed differences are not due to chance, and highlighting the effectiveness of MSM nanoemulsion in reducing arthritis symptoms.