Submitted:
02 September 2025
Posted:
02 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Population
2.2. Data Collection
2.3. Cognitive Function Assessment
2.4. Statistical Analysis
3. Result
3.1. Comparison of Baseline Characteristics Between Stable and Vulnerable Plaque Groups
3.2. Multivariate Logistic Regression Analysis of the Association Between Plaque Vulnerability and NMLR
| Variable | P | OR (95%CI) |
|---|---|---|
| Stroke | 0.014 | 2.50 (1.20 ~ 5.21) |
| Smoking | 0.992 | 0.99 (0.35 ~ 2.86) |
| Age | 0.234 | 0.97 (0.92 ~ 1.02) |
| TC | 0.070 | 0.60 (0.34 ~ 1.04) |
| Albumin | 0.443 | 0.94 (0.81 ~ 1.10) |
| Hemoglobin | 0.253 | 1.02 (0.99 ~ 1.04) |
| NMLR | <.001 | 4.51 (2.43 ~ 8.38) |
| MoCA | 0.015 | 0.84 (0.73 ~ 0.97) |
3.3. Multivariate Logistic Regression Analysis of the Association Between Cognitive Function and NMLR
| Variable | P | OR (95%CI) |
|---|---|---|
| Stroke | 0.034 | 2.59 (1.08 ~ 6.23) |
| Smoking | 0.603 | 0.78 (0.32 ~ 1.95) |
| Age | <.001 | 1.18 (1.10 ~ 1.25) |
| Albumin | 0.766 | 1.03 (0.86 ~ 1.23) |
| LDL | 0.712 | 1.09 (0.68 ~ 1.77) |
| NMLR | <.001 | 4.22 (1.98 ~ 8.96) |
3.4. Mediation Effect of Plaque Vulnerability in the Association Between NMLR and Cognitive Function
| Path | Relationship | P | β(95%CI) |
|---|---|---|---|
| NMLR→Vulnerability | Exposure→ Mediator | <.001 | 1.31(0.77~1.86) |
| NMLR→MoCA | Exposure→ Outcome | <.001 | -1.18(-1.68~-0.67) |
| Vulnerability→MoCA | Mediator→Outcome | <.001 | -1.44(-2.33~-0.64) |
| Effect | β(95%CI) | P | Mediation(%) |
|---|---|---|---|
| Indirect | -0.14(-0.28~-0.03) | <.001 | 10.58 |
| Direct | -1.19(-1.68~-0.70) | <.001 | 89.42 |
| Total | -1.33(-1.83~-0.83) | <.001 | 100 |
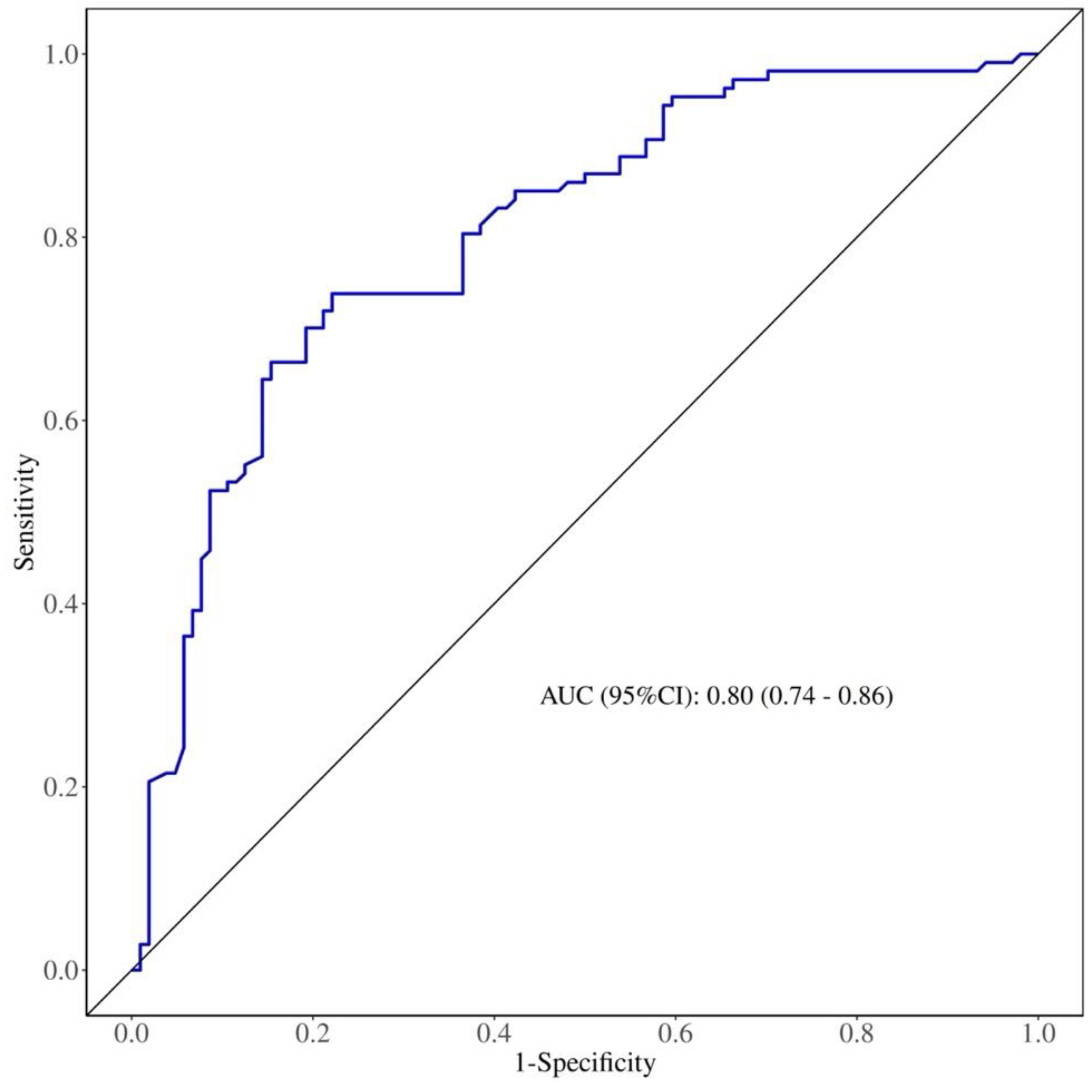
3.5. Predictive Value of NMLR for Carotid Plaque Stability in Patients with CAS.
| AUC | Cutoff | Sensitivity | Specificity | P | |
| NMLR | 0.80 | 2.525 | 77.9% | 73.8% | <0.001 |
4. Discussion
References
- Pelisek J, Eckstein HH, Zernecke A. Pathophysiological Mechanisms of Carotid Plaque Vulnerability: Impact on Ischemic Stroke. Arch Immunol Ther Exp. 2012;60(6):431-442. [CrossRef]
- Willey JZ, Pasterkamp G. The Role of the Vulnerable Carotid Plaque in Embolic Stroke of Unknown Source. Journal of the American College of Cardiology. 2022;79(22):2200-2202. [CrossRef]
- Barrett HE, Van Der Heiden K, Farrell E, Gijsen FJH, Akyildiz AC. Calcifications in atherosclerotic plaques and impact on plaque biomechanics. Journal of Biomechanics. 2019;87:1-12. [CrossRef]
- Cires-Drouet RS, Mozafarian M, Ali A, Sikdar S, Lal BK. Imaging of high-risk carotid plaques: Ultrasound. Seminars in Vascular Surgery. 2017;30(1):44-53. [CrossRef]
- Geovanini GR, Libby P. Atherosclerosis and inflammation: Overview and updates. Clinical Science. 2018;132(12):1243-1252. [CrossRef]
- Pang Y, Shao H, Yang Z, et al. The (Neutrophils + Monocyte)/Lymphocyte Ratio Is an Independent Prognostic Factor for Progression-Free Survival in Newly Diagnosed Multiple Myeloma Patients Treated With BCD Regimen. Front Oncol. 2020;10:1617. [CrossRef]
- Wang Y, Yuan M, Ma Y, et al. The Admission (Neutrophil+Monocyte)/Lymphocyte Ratio Is an Independent Predictor for In-Hospital Mortality in Patients With Acute Myocardial Infarction. Front Cardiovasc Med. 2022;9:870176. [CrossRef]
- Guo M, He W, Mao X, Luo Y, Zeng M. Association between ICU admission (neutrophil + monocyte)/lymphocyte ratio and 30-day mortality in patients with sepsis: a retrospective cohort study. BMC Infect Dis. 2023;23(1):697. [CrossRef]
- Li Y, Ge S, Liu J, et al. Peripheral Blood NMLR Can Predict 5-Year All-Cause Mortality in Patients with Chronic Obstructive Pulmonary Disease. COPD. 2025;Volume 20:95-105. [CrossRef]
- Syahna R, Amin MM, Camellia V, Effendy E, Yamamoto Z. Cognitive impairment and elevated neutrophil-to-lymphocyte ratio and monocyte-to-lymphocyte ratio in schizophrenia. East Asian Arch Psychiatry. 2025;35(1):16-20. [CrossRef]
- Yu J, Li J, Huang X. The beijing version of the montreal cognitive assessment as a brief screening tool for mild cognitive impairment: A community-based study. BMC Psychiatry. 2012;12(1):156. [CrossRef]
- Kounis NG, Koniari I, Plotas P, et al. Inflammation, Thrombosis, and Platelet-to-Lymphocyte Ratio in Acute Coronary Syndromes. Angiology. 2021;72(1):6-8. [CrossRef]
- Drechsler M, Megens RTA, Van Zandvoort M, Weber C, Soehnlein O. Hyperlipidemia-Triggered Neutrophilia Promotes Early Atherosclerosis. Circulation. 2010;122(18):1837-1845. [CrossRef]
- Schumski A, Ortega-Gómez A, Wichapong K, et al. Endotoxinemia Accelerates Atherosclerosis Through Electrostatic Charge–Mediated Monocyte Adhesion. Circulation. 2021;143(3):254-266. [CrossRef]
- Pieterse E, Rother N, Yanginlar C, Hilbrands LB, Van Der Vlag J. Neutrophils Discriminate between Lipopolysaccharides of Different Bacterial Sources and Selectively Release Neutrophil Extracellular Traps. Front Immunol. 2016;7. [CrossRef]
- Döring Y, Libby P, Soehnlein O. Neutrophil Extracellular Traps Participate in Cardiovascular Diseases: Recent Experimental and Clinical Insights. Circulation Research. 2020;126(9):1228-1241. [CrossRef]
- Maretti-Mira AC, Golden-Mason L, Salomon MP, Kaplan MJ, Rosen HR. Cholesterol-Induced M4-Like Macrophages Recruit Neutrophils and Induce NETosis. Front Immunol. 2021;12:671073. [CrossRef]
- Barrett TJ. Macrophages in Atherosclerosis Regression. ATVB. 2020;40(1):20-33. [CrossRef]
- Khoury MK, Yang H, Liu B. Macrophage Biology in Cardiovascular Diseases. ATVB. 2021;41(2). [CrossRef]
- Bäck M, Yurdagul A, Tabas I, Öörni K, Kovanen PT. Inflammation and its resolution in atherosclerosis: mediators and therapeutic opportunities. Nat Rev Cardiol. Published online March 7, 2019. [CrossRef]
- Ley K. Role of the adaptive immune system in atherosclerosis. Biochemical Society Transactions. 2020;48(5):2273-2281. [CrossRef]
- Van Duijn J, Kritikou E, Benne N, et al. CD8+ T-cells contribute to lesion stabilization in advanced atherosclerosis by limiting macrophage content and CD4+ T-cell responses. Cardiovascular Research. 2019;115(4):729-738. [CrossRef]
- Schäfer S, Zernecke A. CD8+ T Cells in Atherosclerosis. Cells. 2020;10(1):37. [CrossRef]
- Mangge H, Prüller F, Schnedl W, Renner W, Almer G. Beyond Macrophages and T Cells: B Cells and Immunoglobulins Determine the Fate of the Atherosclerotic Plaque. IJMS. 2020;21(11):4082. [CrossRef]
- Kumagai S, Amano T, Takashima H, et al. Impact of cigarette smoking on coronary plaque composition. Coronary Artery Disease. 2015;26(1):60-65. [CrossRef]
- Pan Z, Guo H, Wang Q, et al. Relationship between subclasses low-density lipoprotein and carotid plaque. Translational Neuroscience. 2022;13(1):30-37. [CrossRef]
- Scimeca M, Montanaro M, Cardellini M, et al. High sensitivity C-reactive protein increases the risk of carotid plaque instability in male dyslipidemic patients. Diagnostics. 2021;11(11):2117. [CrossRef]
- Ruan W, Wang M, Sun C, et al. Correlation between neutrophil-to-lymphocyte ratio and stability of carotid plaques. Clinical Neurology and Neurosurgery. 2022;212:107055. [CrossRef]
- Gisterå A, Hansson GK. The immunology of atherosclerosis. Nat Rev Nephrol. 2017;13(6):368-380. [CrossRef]
- Li T, Huang Y, Cai W, et al. Age-related cerebral small vessel disease and inflammaging. Cell Death Dis. 2020;11(10):932. [CrossRef]
- Rosenberg GA. Inflammation and white matter damage in vascular cognitive impairment. Stroke. 2009;40(3_suppl_1). [CrossRef]
- Krstic D, Knuesel I. Deciphering the mechanism underlying late-onset alzheimer disease. Nat Rev Neurol. 2013;9(1):25-34. [CrossRef]
- Aries ML, Hensley-McBain T. Neutrophils as a potential therapeutic target in alzheimer’s disease. Front Immunol. 2023;14:1123149. [CrossRef]
- Lee NT, Ong LK, Gyawali P, et al. Role of purinergic signalling in endothelial dysfunction and thrombo-inflammation in ischaemic stroke and cerebral small vessel disease. Biomolecules. 2021;11(7):994. [CrossRef]
- Hou JH, Ou YN, Xu W, et al. Association of peripheral immunity with cognition, neuroimaging, and alzheimer’s pathology. Alz Res Therapy. 2022;14(1):29. [CrossRef]
- Ponce DP, Salech F, SanMartin CD, et al. Increased susceptibility to oxidative death of lymphocytes from alzheimer patients correlates with dementia severity. Curr Alzheimer Res. 2014;11(9):892-898.
- Takasugi J, Miwa K, Watanabe Y, et al. Cortical cerebral microinfarcts on 3T magnetic resonance imaging in patients with carotid artery stenosis. Stroke. 2019;50(3):639-644. [CrossRef]
| Variable | Total (n = 211) | Stable (n = 104) |
Vulnerable (n = 107) |
Statistic | P | |
|---|---|---|---|---|---|---|
| Gender, n(%) | χ2=0.01 | 0.932 | ||||
| F | 37 (17.54) | 18 (17.31) | 19 (17.76) | |||
| M | 174 (82.46) | 86 (82.69) | 88 (82.24) | |||
| Smoking, n(%) | χ2=10.31 | 0.001 | ||||
| No | 153 (72.51) | 65 (62.50) | 88 (82.24) | |||
| Yes | 58 (27.49) | 39 (37.50) | 19 (17.76) | |||
| Stroke, n(%) | χ2=12.53 | <.001 | ||||
| No | 114 (54.03) | 69 (66.35) | 45 (42.06) | |||
| Yes | 97 (45.97) | 35 (33.65) | 62 (57.94) | |||
| Hypertension, n(%) | χ2=0.16 | 0.692 | ||||
| No | 45 (21.33) | 21 (20.19) | 24 (22.43) | |||
| Yes | 166 (78.67) | 83 (79.81) | 83 (77.57) | |||
| Diabetes, n(%) | χ2=2.97 | 0.085 | ||||
| No | 132 (62.56) | 59 (56.73) | 73 (68.22) | |||
| Yes | 79 (37.44) | 45 (43.27) | 34 (31.78) | |||
| CAD, n(%) | χ2=0.27 | 0.602 | ||||
| No | 189 (89.57) | 92 (88.46) | 97 (90.65) | |||
| Yes | 22 (10.43) | 12 (11.54) | 10 (9.35) | |||
| Age(Year) | 69.00 (61.00, 75.00) | 66.00 (59.00, 73.00) | 71.00 (65.00, 75.00) | Z=-3.53 | <.001 | |
| Albumin(g/L) | 39.60 (38.50, 41.10) | 39.70 (39.40, 41.30) | 39.60 (37.70, 40.70) | Z=-2.45 | 0.014 | |
| TC(mmol/L) | 3.71 (3.12, 4.22) | 3.84 (3.06, 4.38) | 3.53 (3.12, 4.03) | Z=-1.41 | 0.160 | |
| LDL(mmol/L) | 1.87 (1.56, 2.43) | 1.98 (1.52, 2.60) | 1.86 (1.62, 2.25) | Z=-0.93 | 0.352 | |
| CRP(mg/L) | 3.40 (2.40, 4.70) | 3.20 (2.40, 4.40) | 3.50 (2.50, 5.20) | Z=-1.21 | 0.228 | |
| Hemoglobin(g/L) | 135.00 (125.00, 145.50) | 137.00 (127.00, 148.00) | 134.00 (124.00, 140.00) | Z=-2.04 | 0.042 | |
| Fasting glucose(mmol/L) | 4.99 (4.42, 5.83) | 5.10 (4.60, 6.00) | 4.92 (4.31, 5.74) | Z=-1.74 | 0.082 | |
| Neutrophil count, (×109/L) | 3.70 (3.30, 4.10) | 3.50 (3.27, 4.10) | 3.80 (3.45, 4.20) | Z=-1.97 | 0.049 | |
| Lymphocyte count, (×109/L) | 1.60 (1.40, 2.05) | 1.90 (1.60, 2.40) | 1.40 (1.30, 1.70) | Z=-7.09 | <.001 | |
| Monocyte count, (×109/L) | 0.50 (0.40, 0.60) | 0.40 (0.30, 0.50) | 0.60 (0.50, 0.70) | Z=-7.56 | <.001 | |
| NMLR | 2.50 (1.95, 3.07) | 2.18 (1.56, 2.51) | 2.93 (2.47, 3.37) | Z=-7.61 | <.001 | |
| MoCA | 23.00 (20.00, 25.00) | 24.00 (23.00, 26.00) | 21.00 (19.00, 23.00) | Z=-6.80 | <.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




