Submitted:
29 August 2025
Posted:
02 September 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction: Bridging Anti-Aging, Regeneration, and Oncology Through Senescence
2. Results
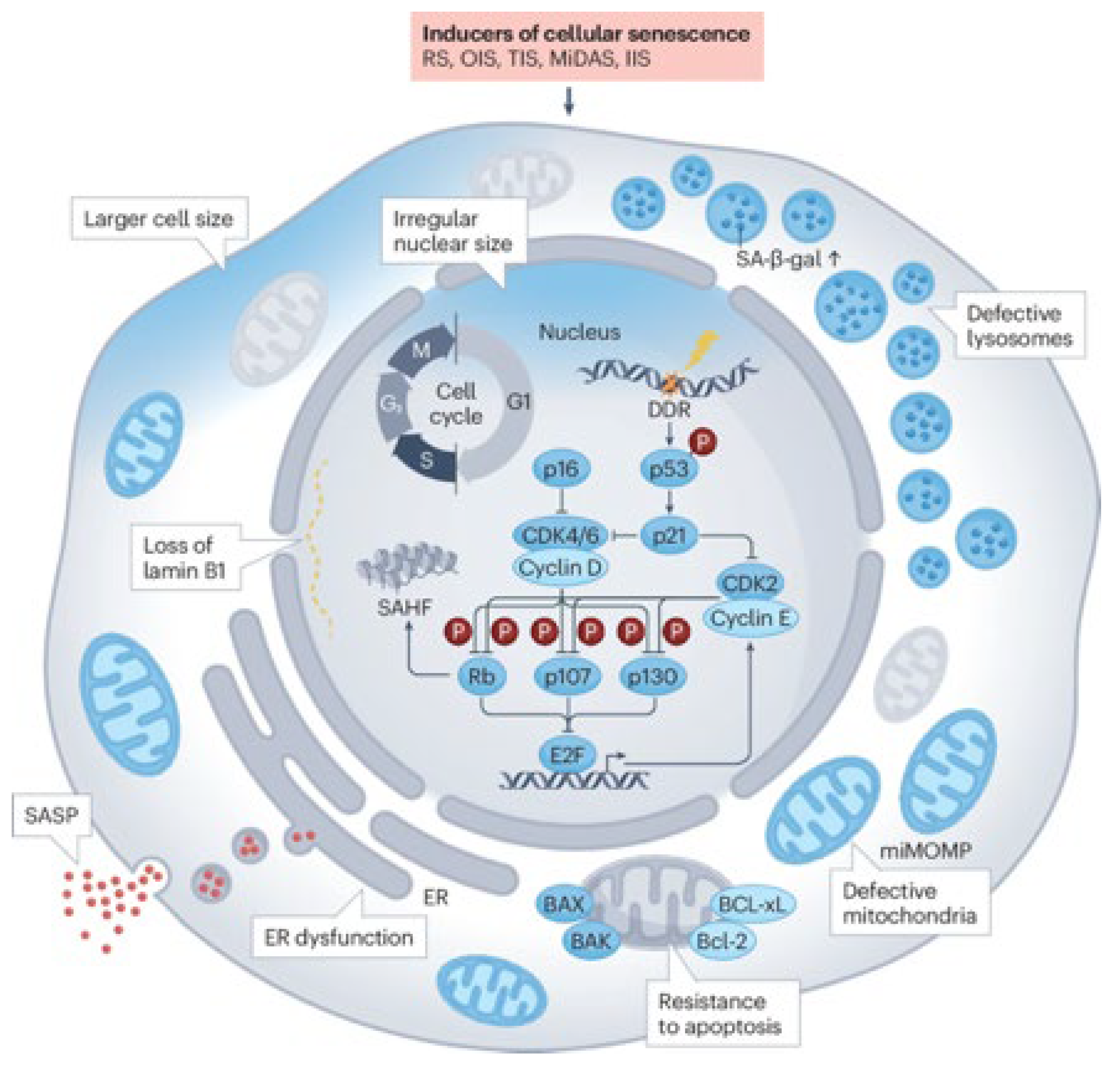
2.1. The Fundamental Role of Senescence: A Shared Biological Mechanism
2.1.1. Senescence Modulation in Dermatology, Anti-Aging and Regenerative Aesthetics: A Different Approach to Rejuvenation
2.1.2. Senescence Modulation in Oncology: A Multi-level Therapeutic Target
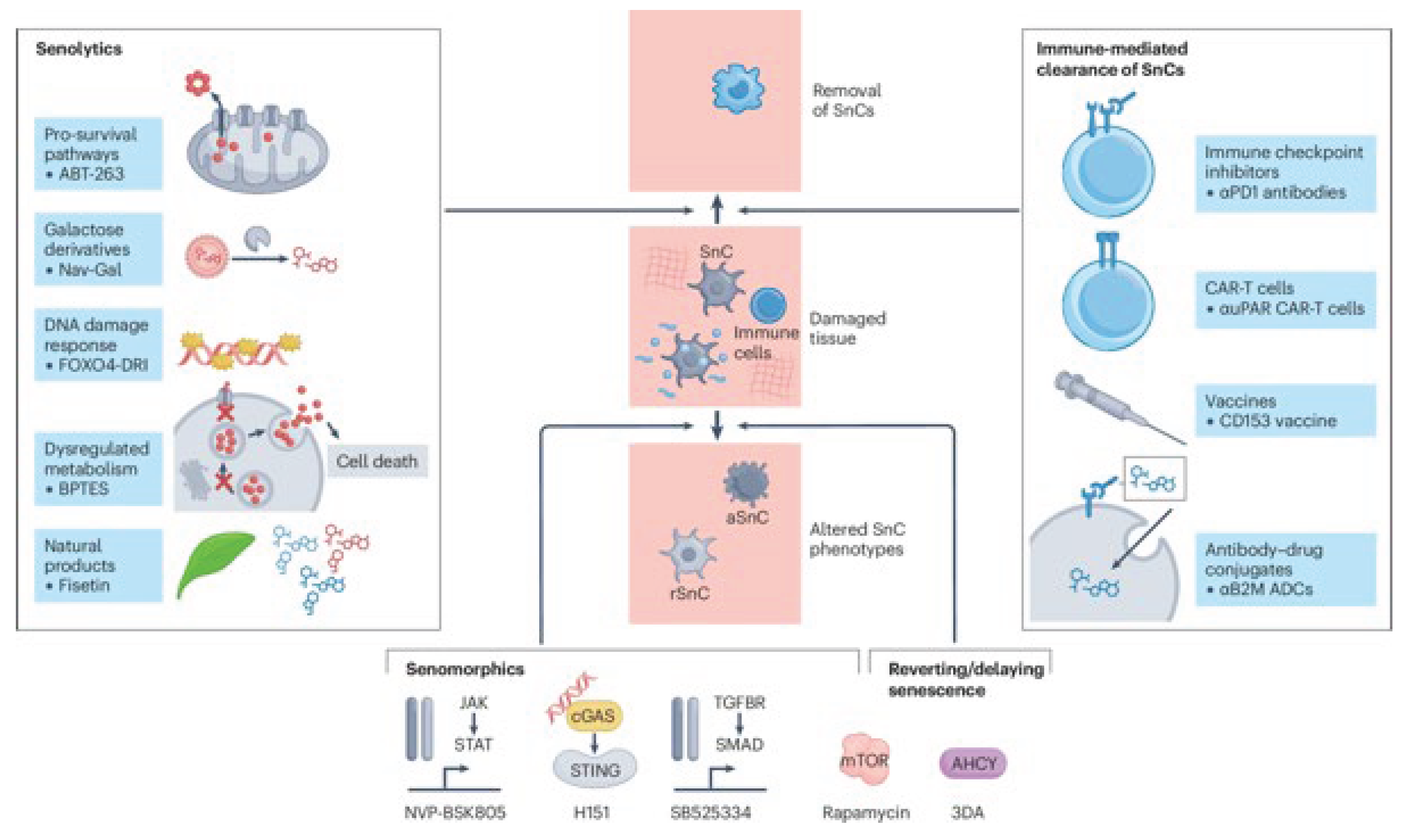
2.2. Targeting Senescence: Therapeutic Agents and Approaches
3. Discussion
4. Materials and Methods
4.1. Literature Search and Selection
4.2. Inclusion and Exclusion Criteria
4.3. Data Extraction and Synthesis
- The fundamental biological mechanisms of cellular senescence.
- The dual roles of the SASP in aging and oncology.
- Specific examples of senotherapeutic agents (senolytics and senomorphics).
- Applications of senescence modulation in anti-aging and regenerative aesthetics.
- Applications of senescence modulation in various oncology therapy modalities (e.g., chemotherapy, immunotherapy, nucleic acid-based therapies).
- Emerging therapeutic targets and future directions.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3DA | 3-deazaadenosine |
| ADCs | Antibody-drug conjugates |
| AHCY | S-adenosylhomocysteine hydrolase |
| AMPK | 5' adenosine monophosphate-activated protein kinase |
| aSnc | Activated senescent cell |
| BAX | Bcl-2-associated X protein |
| Bcl-2 | B-cell lymphoma 2 |
| BCL-xL | B-cell lymphoma-extra large |
| CAR | Chimeric antigen receptor |
| CD153 vaccine | Cluster of Differentiation 153 vaccine |
| CDK | Cyclin-dependent kinase |
| cGAS | Cyclic GMP-AMP synthase |
| CML | Chronic myeloid leukemia |
| D+Q | Dasatinib, a tyrosine kinase inhibitor, with quercetin, a naturally occurring flavonoid |
| DDR | DNA damage response |
| DNA | Deoxyribonucleic acid |
| E2F | E2 promoter-binding factor |
| ER | Endoplasmic reticulum |
| FAP-alpha | Fibroblast Activation Protein-alpha |
| HNSCC | Head and neck squamous cell carcinoma |
| IIS | Inflammation-induced Senescence |
| JAKs | Janus kinases |
| mAbs | Monoclonal antibodies |
| MIDAS | Mitochondria-induced Senescence |
| miMOMP | Mitochondrial outer membrane permeabilization |
| miRNA | MicroRNA |
| mRNA | Messenger RNA |
| MSCs | Mesenchymal stem cells |
| mTOR | Mechanistic target of rapamycin |
| NSCLC | Non-small cell lung cancer |
| OIS | Oncogene-induced Senescence |
| PDRN | Polydeoxyribonucleotide |
| PN | Polynucleotide |
| Rb | Retinoblastoma protein |
| RS | Replicative senescence |
| rSnC | Resident senescent cell |
| SA-β-gal | Senescence-Associated β-galactosidase |
| SAHF | Senescence-associated heterochromatin foci |
| SASP | Senescence-associated secretory phenotype |
| siRNA | Small interfering RNA |
| SMAD | Mothers against decapentaplegic homolog |
| SnC | Senescent cell |
| STAT | Signal Transducer and Activator of Transcription |
| STING | Stimulator of Interferon Genes |
| TGFBR | Transforming growth factor beta receptor |
| TIS | Therapy-induced Senescence |
| uPAR | Urokinase plasminogen activator receptor |
| αB2M ADCs | Anti-beta-2-microglobulin Antibody-Drug Conjugates |
| αPD1 antibodies | Anti-Programmed Death-1 antibodies |
| αuPAR CAR-T cells | Urokinase plasminogen activator receptor Chimeric Antigen Receptor T-cells |
References
- Guo, J.; Huang, X.; Dou, L.; Yan, M.; Shen, T.; Tang, W.; Li, J. , Aging and aging-related diseases: From molecular mechanisms to interventions and treatments. Signal Transduction and Targeted Therapy 2022, 7, 391. [Google Scholar] [CrossRef]
- Wang, B.; Han, J.; Elisseeff, J.H.; Demaria, M. , The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol 2024, 25, 958–978. [Google Scholar] [CrossRef]
- Liao, Z.; Yeo, H.L.; Wong, S.W.; Zhao, Y. , Cellular senescence: Mechanisms and therapeutic potential. Biomedicines 2021, 9. [Google Scholar] [CrossRef]
- McHugh, D.; Gil, J. , Senescence and aging: Causes, consequences, and therapeutic avenues. J Cell Biol 2018, 217, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.A.; Wang, B.; Demaria, M. , Senescence and cancer - role and therapeutic opportunities. Nat Rev Clin Oncol 2022, 19, 619–636. [Google Scholar] [CrossRef] [PubMed]
- Freund, A.; Patil, C.K.; Campisi, J. , p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. Embo j 2011, 30, 1536–1548. [Google Scholar] [CrossRef] [PubMed]
- Herranz, N.; Gallage, S.; Mellone, M.; Wuestefeld, T.; Klotz, S.; Hanley, C.J.; Raguz, S.; Acosta, J.C.; Innes, A.J.; Banito, A.; et al. mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype. Nat Cell Biol 2015, 17, 1205–1217. [Google Scholar] [CrossRef]
- Laberge, R.M.; Sun, Y.; Orjalo, A.V.; Patil, C.K.; Freund, A.; Zhou, L.; Curran, S.C.; Davalos, A.R.; Wilson-Edell, K.A.; Liu, S.; et al. MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol 2015, 17, 1049–1061. [Google Scholar] [CrossRef]
- Chien, Y.; Scuoppo, C.; Wang, X.; Fang, X.; Balgley, B.; Bolden, J.E.; Premsrirut, P.; Luo, W.; Chicas, A.; Lee, C.S.; et al. Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev 2011, 25, 2125–2136. [Google Scholar] [CrossRef]
- Kuilman, T.; Michaloglou, C.; Vredeveld, L.C.; Douma, S.; van Doorn, R.; Desmet, C.J.; Aarden, L.A.; Mooi, W.J.; Peeper, D.S. , Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 2008, 133, 1019–1031. [Google Scholar] [CrossRef]
- Deng, G.; Xu, C.; Mo, D. , Identification and mechanistic insights of cell senescence-related genes in psoriasis. PeerJ 2025, 13, e18818. [Google Scholar] [CrossRef]
- Rider, P.; Carmi, Y.; Cohen, I. , Biologics for Targeting Inflammatory Cytokines, Clinical Uses, and Limitations. Int J Cell Biol 2016, 2016, 9259646. [Google Scholar] [CrossRef]
- Thau, H.; Gerjol, B.P.; Hahn, K.; von Gudenberg, R.W.; Knoedler, L.; Stallcup, K.; Emmert, M.Y.; Buhl, T.; Wyles, S.P.; Tchkonia, T.; et al. Senescence as a molecular target in skin aging and disease. Ageing Research Reviews 2025, 105, 102686. [Google Scholar] [CrossRef] [PubMed]
- Hussein, R.S.; Bin Dayel, S.; Abahussein, O.; El-Sherbiny, A.A. , Influences on Skin and Intrinsic Aging: Biological, Environmental, and Therapeutic Insights. J Cosmet Dermatol 2025, 24, e16688. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Wu, J.; Feng, J.; Cheng, H. , Cellular Senescence and Anti-Aging Strategies in Aesthetic Medicine: A Bibliometric Analysis and Brief Review. Clin Cosmet Investig Dermatol 2024, 17, 2243–2259. [Google Scholar] [CrossRef] [PubMed]
- Chin, T.; Lee, X.E.; Ng, P.Y.; Lee, Y.; Dreesen, O. , The role of cellular senescence in skin aging and age-related skin pathologies. Front Physiol 2023, 14, 1297637. [Google Scholar] [CrossRef]
- Mansfield, L.; Ramponi, V.; Gupta, K.; Stevenson, T.; Mathew, A.B.; Barinda, A.J.; Herbstein, F.; Morsli, S. , Emerging insights in senescence: Pathways from preclinical models to therapeutic innovations. npj Aging 2024, 10, 53. [Google Scholar] [CrossRef]
- Thompson, E.L.; Pitcher, L.E.; Niedernhofer, L.J.; Robbins, P.D. , Targeting cellular senescence with senotherapeutics: Development of new approaches for skin care. Plast Reconstr Surg 2022, 150, 12s–19s. [Google Scholar] [CrossRef]
- Lueangarun, S.; Visutjindaporn, P.; Parcharoen, Y.; Jamparuang, P.; Tempark, T. , A systematic review and meta-analysis of randomized controlled trials of United States Food and Drug Administration-approved, home-use, low-level light/laser therapy devices for pattern hair loss: Device design and technology. J Clin Aesthet Dermatol 2021, 14, E64–e75. [Google Scholar]
- Heidari Beigvand, H.; Razzaghi, M.; Rostami-Nejad, M.; Rezaei-Tavirani, M.; Safari, S.; Rezaei-Tavirani, M.; Mansouri, V.; Heidari, M.H. , Assessment of laser effects on skin rejuvenation. J Lasers Med Sci 2020, 11, 212–219. [Google Scholar] [CrossRef]
- Akinbiyi, T.; Othman, S.; Familusi, O.; Calvert, C.; Card, E.B.; Percec, I. , Better results in facial rejuvenation with fillers. Plast Reconstr Surg Glob Open 2020, 8, e2763. [Google Scholar] [CrossRef] [PubMed]
- Lampridou, S.; Bassett, S.; Cavallini, M.; Christopoulos, G. , The effectiveness of polynucleotides in esthetic medicine: A systematic review. J Cosmet Dermatol 2025, 24, e16721. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Wang, G.; Zhou, F.; Gong, L.; Zhang, J.; Qi, L.; Cui, H. , Polydeoxyribonucleotide: A promising skin anti-aging agent. Chinese Journal of Plastic and Reconstructive Surgery 2022, 4, 187–193. [Google Scholar] [CrossRef]
- Li, X.; Zhang, D.; Yu, Y.; Wang, L.; Zhao, M. , Umbilical cord-derived mesenchymal stem cell secretome promotes skin regeneration and rejuvenation: From mechanism to therapeutics. Cell Proliferation 2024, 57, e13586. [Google Scholar] [CrossRef]
- Zonari, A.; Brace, L.E.; Al-Katib, K.; Porto, W.F.; Foyt, D.; Guiang, M.; Cruz, E.A.O.; Marshall, B.; Gentz, M.; Guimarães, G.R.; et al. Senotherapeutic peptide treatment reduces biological age and senescence burden in human skin models. NPJ Aging 2023, 9, 10. [Google Scholar] [CrossRef]
- Zhang, L.; Pitcher, L.E.; Prahalad, V.; Niedernhofer, L.J.; Robbins, P.D. , Targeting cellular senescence with senotherapeutics: Senolytics and senomorphics. Febs j 2023, 290, 1362–1383. [Google Scholar] [CrossRef]
- Battram, A.M.; Bachiller, M.; Martín-Antonio, B. , Senescence in the Development and Response to Cancer with Immunotherapy: A Double-Edged Sword. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Prasanna, P.G.; Citrin, D.E.; Hildesheim, J.; Ahmed, M.M.; Venkatachalam, S.; Riscuta, G.; Xi, D.; Zheng, G.; Deursen, J.V.; Goronzy, J.; et al. Therapy-induced senescence: Opportunities to improve anticancer therapy. J Natl Cancer Inst 2021, 113, 1285–1298. [Google Scholar] [CrossRef]
- Zingoni, A.; Antonangeli, F.; Sozzani, S.; Santoni, A.; Cippitelli, M.; Soriani, A. , The senescence journey in cancer immunoediting. Mol Cancer 2024, 23, 68. [Google Scholar] [CrossRef]
- Zhang, L.; Pitcher, L.E.; Yousefzadeh, M.J.; Niedernhofer, L.J.; Robbins, P.D.; Zhu, Y. , Cellular senescence: A key therapeutic target in aging and diseases. J Clin Invest 2022, 132. [Google Scholar] [CrossRef]
- Shiravand, Y.; Khodadadi, F.; Kashani, S.M.A.; Hosseini-Fard, S.R.; Hosseini, S.; Sadeghirad, H.; Ladwa, R.; O'Byrne, K.; Kulasinghe, A. , Immune checkpoint inhibitors in cancer therapy. Curr Oncol 2022, 29, 3044–3060. [Google Scholar] [CrossRef]
- Byrd, J.C.; Waselenko, J.K.; Maneatis, T.J.; Murphy, T.; Ward, F.T.; Monahan, B.P.; Sipe, M.A.; Donegan, S.; White, C.A. Rituximab therapy in hematologic malignancy patients with circulating blood tumor cells: Association with increased infusion-related side effects and rapid blood tumor clearance. J Clin Oncol 1999, 17, 791–795. [Google Scholar] [CrossRef]
- Yi, M.; Li, T.; Niu, M.; Zhang, H.; Wu, Y.; Wu, K.; Dai, Z. , Targeting cytokine and chemokine signaling pathways for cancer therapy. Signal Transduction and Targeted Therapy 2024, 9, 176. [Google Scholar] [CrossRef]
- Grothey, A.; Blay, J.Y.; Pavlakis, N.; Yoshino, T.; Bruix, J. , Evolving role of regorafenib for the treatment of advanced cancers. Cancer Treat Rev 2020, 86, 101993. [Google Scholar] [CrossRef]
- Zhu, Y.; Tchkonia, T.; Pirtskhalava, T.; Gower, A.C.; Ding, H.; Giorgadze, N.; Palmer, A.K.; Ikeno, Y.; Hubbard, G.B.; Lenburg, M.; et al. The Achilles' heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell 2015, 14, 644–658. [Google Scholar] [CrossRef] [PubMed]
- Yousefzadeh, M.J.; Zhu, Y.; McGowan, S.J.; Angelini, L.; Fuhrmann-Stroissnigg, H.; Xu, M.; Ling, Y.Y.; Melos, K.I.; Pirtskhalava, T.; Inman, C.L.; et al. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine 2018, 36, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; DiPersio, J.F. , ReCARving the future: Bridging CAR T-cell therapy gaps with synthetic biology, engineering, and economic insights. Front Immunol 2024, 15, 1432799. [Google Scholar] [CrossRef] [PubMed]
- Amor, C.; Feucht, J.; Leibold, J.; Ho, Y.J.; Zhu, C.; Alonso-Curbelo, D.; Mansilla-Soto, J.; Boyer, J.A.; Li, X.; Giavridis, T.; et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 2020, 583, 127–132. [Google Scholar] [CrossRef]
- Jacobs, W.; Khalifeh, M.; Koot, M.; Palacio-Castañeda, V.; van Oostrum, J.; Ansems, M.; Verdurmen, W.P.R.; Brock, R. , RNA-based logic for selective protein expression in senescent cells. The International Journal of Biochemistry & Cell Biology 2024, 174, 106636. [Google Scholar]
- Ratti, M.; Lampis, A.; Ghidini, M.; Salati, M.; Mirchev, M.B.; Valeri, N.; Hahne, J.C. , MicroRNAs (miRNAs) and Long Non-Coding RNAs (lncRNAs) as New Tools for Cancer Therapy: First Steps from Bench to Bedside. Target Oncol 2020, 15, 261–278. [Google Scholar] [CrossRef]
- Czajkowski, K.; Herbet, M.; Murias, M.; Piątkowska-Chmiel, I. , Senolytics: Charting a new course or enhancing existing anti-tumor therapies? Cell Oncol (Dordr) 2025, 48, 351–371. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Tchkonia, T.; Pirtskhalava, T.; Gower, A.C.; Ding, H.; Giorgadze, N.; Palmer, A.K.; Ikeno, Y.; Hubbard, G.B.; Lenburg, M.; et al. The Achilles’ heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell 2015, 14, 644–658. [Google Scholar] [CrossRef]
- Liu, H.; Xu, Q.; Wufuer, H.; Li, Z.; Sun, R.; Jiang, Z.; Dou, X.; Fu, Q.; Campisi, J.; Sun, Y. , Rutin is a potent senomorphic agent to target senescent cells and can improve chemotherapeutic efficacy. Aging Cell 2024, 23, e13921. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Liu, H.; Wu, H. , Cellular senescence in health, disease, and lens aging. In Pharmaceuticals, 2025; Vol. 18.
- Khansari, N.; Shakiba, Y.; Mahmoudi, M. , Chronic inflammation and oxidative stress as a major cause of age-related diseases and cancer. Recent Pat Inflamm Allergy Drug Discov 2009, 3, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Ostojic, A.; Vrhovac, R.; Verstovsek, S. , Ruxolitinib: A new JAK1/2 inhibitor that offers promising options for treatment of myelofibrosis. Future Oncol 2011, 7, 1035–1043. [Google Scholar] [CrossRef]
- Lamming, D.W. , Inhibition of the mechanistic target of rapamycin (mTOR)-rapamycin and beyond. Cold Spring Harb Perspect Med 2016, 6. [Google Scholar] [CrossRef]
- Blagosklonny, M.V. , Rapamycin extends life- and health span because it slows aging. Aging (Albany NY) 2013, 5, 592–598. [Google Scholar] [CrossRef]
- Baroja-Mazo, A.; Revilla-Nuin, B.; Ramírez, P.; Pons, J.A. , Immunosuppressive potency of mechanistic target of rapamycin inhibitors in solid-organ transplantation. World J Transplant 2016, 6, 183–192. [Google Scholar] [CrossRef]
- Fu, T.E.; Zhou, Z. , Senescent cells as a target for anti-aging interventions: From senolytics to immune therapies. J Transl Int Med 2025, 13, 33–47. [Google Scholar] [CrossRef]
- McHugh, D.; Duran, I.; Gil, J. , Senescence as a therapeutic target in cancer and age-related diseases. Nat Rev Drug Discov 2025, 24, 57–71. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



