Submitted:
21 August 2025
Posted:
01 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Epidemiology of Keratoacanthoma
3. Etiology
4. Clinical Manifestations
4. Dermoscopy
5. Reflectance Confocal Microscopy (RCM) and Line-Field Confocal Optical Coherence Tomography (LC-OCT)
6. Histology of Keratoacanthoma
7. Human Papillomavirus and Carcinogenesis
7. Therapy
8. Anti-HPV Vaccines
8. Materials and Methods
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| KA | Keratoacanthoma |
| SCC | Squamous Cell Carcinoma |
| RCM | Reflectance Confocal Microscopy |
| LC-OCT | Line-Field Confocal Optical Coherence Tomography |
| HPV | Human Papillomavirus |
| UV | Ultraviolet |
| MTS | Muir-Torre Syndrome |
| MSSE | Multiple Self-healing Squamous Epithelioma |
| MFKWZ | Multiple Familial KA of Witten and Zak |
| XP | Xeroderma Pigmentosum |
| IP | Incontinentia Pigmenti |
| EV | Epidermodysplasia Verruciformis |
| GEKA | Generalized Eruptive Keratoacanthoma |
| HR | High-Risk |
| LR | Low-Risk |
| NER | Nucleotide Excision Repair |
| E6AP | E6-Associated Protein |
| pRb | Retinoblastoma protein |
| RT | Radiotherapy |
| PDT | Photodynamic therapy |
| 5-FU | 5-Fluorouracil |
| EGFR | Epidermal Growth Factor Receptor |
References
- Robertson, S.J.; Bashir, S.J.; Pichert, G.; Robson, A.; Whittaker, S. Severe exacerbation of multiple self-healing squamous epithelioma (Ferguson–Smith disease) with radiotherapy, which was successfully treated with acitretin. Clin Exp Dermatol 2010, 35(4), e100–2. [Google Scholar] [CrossRef]
- Kwiek, B.; Schwartz, R.A. Keratoacanthoma (KA): An update and review. J Am Acad Dermatol 2016, 74(6), 1220–33. [Google Scholar] [CrossRef]
- Miedzinski, F.; Kozakiewicz, J. Keratoacanthoma centrifugum--a special variety of keratoacanthoma. Hautarzt 1962, 13, 348–52. [Google Scholar] [PubMed]
- Ramselaar, C.G.; Ruitenberg, E.J.; Kruizinga, W. Regression of induced keratoacanthomas in anagen (hair growth phase) skin grafts in mice. Cancer Res 1980, 40(5), 1668–73. [Google Scholar] [PubMed]
- Zito, G.; Saotome, I.; Liu, Z.; Ferro, E.G.; Sun, T.Y.; Nguyen, D.X.; Bilguvar, K.; Ko, C.J.; Greco, V. Spontaneous tumour regression in keratoacanthomas is driven by Wnt/retinoic acid signalling cross-talk. Nat Commun 2014, 5, 3543. [Google Scholar] [CrossRef]
- Hu, W.; Cook, T.; Oh, C.W.; Penneys, N.S. Expression of the cyclin-dependent kinase inhibitor p27 in keratoacanthoma. J Am Acad Dermatol 2000, 42(3), 473–5. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Viros, A.; Milagre, C.; Trunzer, K.; Bollag, G.; Spleiss, O.; Reis-Filho, J.S.; Kong, X.; Koya, R.C.; Flaherty, K.T.; Chapman, P.B.; Kim, M.J.; Hayward, R.; Martin, M.; Yang, H.; Wang, Q.; Hilton, H.; Hang, J.S.; Noe, J.; Lambros, M.; Geyer, F.; Dhomen, N.; Niculescu-Duvaz, I.; Zambon, A.; Niculescu-Duvaz, D.; Preece, N.; Robert, L.; Otte, N.J.; Mok, S.; Kee, D.; Ma, Y.; Zhang, C.; Habets, G.; Burton, E.A.; Wong, B.; Nguyen, H.; Kockx, M.; Andries, L.; Lestini, B.; Nolop, K.B.; Lee, R.J.; Joe, A.K.; Troy, J.L.; Gonzalez, R.; Hutson, T.E.; Puzanov, I.; Chmielowski, B.; Springer, C.J.; McArthur, G.A.; Sosman, J.A.; Lo, R.S.; Ribas, A.; Marais, R. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med 2012, 366(3), 207–15. [Google Scholar] [CrossRef]
- Corominas, M.; Kamino, H.; Leon, J.; Pellicer, A. Oncogene activation in human benign tumors of the skin (keratoacanthomas): is HRAS involved in differentiation as well as proliferation? Proc Natl Acad Sci USA 1989, 86(16), 6372–6. [Google Scholar] [CrossRef]
- Karampinis, E.; Kostopoulou, C.; Toli, O.; Marinos, L.; Papadimitriou, G.; Roussaki Schulze, A.V.; Zafiriou, E. Multiple Keratoacanthoma-like Syndromes: Case Report and Literature Review. Medicina (Kaunas) 2024, 60(3), 371. [Google Scholar] [CrossRef]
- Dobre, A.; Nedelcu, R.I.; Turcu, G.; Brinzea, A.; Struna, I.; Tudorache, G.; Ali, A.; Hulea, I.; Balasescu, E.; Fertig, T.E.; Gherghiceanu, M.; Harwood, C.; Ion, D.A.; Forsea, A.M. Multiple Keratoacanthomas Associated with Genetic Syndromes: Narrative Review and Proposal of a Diagnostic Algorithm. Am J Clin Dermatol 2025, 26(1), 45–59. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Garbe, C.; Dessinioti, C.; Lebbe, C.; van Akkooi, A.; Bataille, V.; Bastholt, L.; Dreno, B.; Dummer, R.; Fargnoli, M.C.; Forsea, A.M.; Harwood, C.A.; Hauschild, A.; Hoeller, C.; Kandolf-Sekulovic, L.; Kaufmann, R.; Kelleners-Smeets, N.W.; Lallas, A.; Leiter, U.; Malvehy, J.; Del Marmol, V.; Moreno-Ramirez, D.; Pellacani, G.; Peris, K.; Saiag, P.; Tagliaferri, L.; Trakatelli, M.; Ioannides, D.; Vieira, R.; Zalaudek, I.; Arenberger, P.; Eggermont, A.M.M.; Röcken, M.; Grob, J.J.; Lorigan, P.; EADO; EDF; ESTRO; UEMS; EADV; and EORTC. European consensus-based interdisciplinary guideline for invasive cutaneous squamous cell carcinoma: Part 2. Treatment-Update 2023. Eur J Cancer; 2023; 193, p. 113252. [Google Scholar]
- Cohen, P.R.; Schulze, K.E.; Teller, C.F.; Nelson, B.R. Intralesional methotrexate for keratoacanthoma of the nose. Skinmed 2005, 4(6), 393–5. [Google Scholar]
- Kiss, N.; Avci, P.; Bánvölgyi, A.; Lőrincz, K.; Szakonyi, J.; Gyöngyösi, N.; Fésűs, L; Lee, G; Wikonkál, N. Intralesional therapy for the treatment of keratoacanthoma. Dermatol Ther. 2019, 32(3), e12872. [Google Scholar] [CrossRef]
- Sullivan, J.J. Keratoacanthoma: the Australian experience. Australas J Dermatol 1997, 38, S36–39. [Google Scholar]
- Reizner, G.T.; Chuang, T.Y.; Elpern, D.J.; Stone, J.L.; Farmer, E.R. Basal cell carcinoma and keratoacanthoma in Hawaiians: an incidence report. J Am Acad Dermatol 1993, 29 5 Pt 1, 780–2. [Google Scholar] [CrossRef]
- Vergilis-Kalner, I.J.; Kriseman, Y.; Goldberg, L.H. Keratoacanthomas: overview and comparison between Houston and minneapolis experiences. J Drugs Dermatol 2010, 9(2), 117–21. [Google Scholar]
- Fania, L.; Didona, D.; Di Pietro, F.R.; Verkhovskaia, S.; Morese, R.; Paolino, G.; Donati, M.; Ricci, F.; Coco, V.; Ricci, F.; Candi, E.; Abeni, D.; Dellambra, E. Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines 2021, 9(2), 171. [Google Scholar] [CrossRef]
- Goldberg, L.H.; Silapunt, S.; Beyrau, K.K.; Peterson, S.R.; Friedman, P.M.; Alam, M. Keratoacanthoma as a postoperative complication of skin cancer excision. J Am Acad Dermatol 2004, 50(5), 753–8. [Google Scholar] [CrossRef]
- Gleich, T.; Chiticariu, E.; Huber, M.; Hohl, D. Keratoacanthoma: a distinct entity? Exp Dermatol 2016, 25(2), 85–91. [Google Scholar] [CrossRef]
- Ambur, A.; Clark, A.; Nathoo, R. An Updated Review of the Therapeutic Management of Keratoacanthomas. J Clin Aesthet Dermatol 2022, 15(11), 30–6. [Google Scholar]
- Joshi, S.; De Angelis, P.M.; Zucknick, M.; Schjølberg, A.R.; Andersen, S.N.; Clausen, O.P.F. Role of the Wnt signaling pathway in keratoacanthoma. Cancer Rep (Hoboken) 2020, 3(2), e1219. [Google Scholar] [CrossRef]
- Zito, P.M.; Scharf, R. Keratoacanthoma. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 [accessed online 2025 Apr 27]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK499931/.
- Ilaria, P.; Nevena, S.; Ersilia, T.; Federica, T.; Felice, F.; Agnieszka Ewa, D.; Francesco, F.; Concetta, P. Generalized Eruptive Keratoacanthoma (GEKA) after Pfizer mRNABNT162b2 (Comirnaty®) COVID-19 Vaccination Successfully Treated with Cemiplimab. Viruses. 2024, 16(8), 1260. [Google Scholar] [CrossRef]
- Yumeen, S.; Robinson-Bostom, L.; Firoz, E.F. Solitary Eruptive Keratoacanthoma Developing at Site of COVID-19 Vaccine Injection. R I Med J 2023, 106(11), 49–51. [Google Scholar]
- Mitri, F.; Hartschuh, W.; Toberer, F. Multiple Keratoacanthomas after a Recent Tattoo: A Case Report. Case Rep Dermatol 2021, 13(1), 23–7. [Google Scholar] [CrossRef]
- Brongo, S.; Moccia, L.S.; Nunziata, V.; D’Andrea, F. Keratoacanthoma arising after site injection infection of cosmetic collagen filler. Int J Surg Case Rep 2013, 4(4), 429–31. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.H.; Hong, S.P.; Kim, J.; Kim, S.W. Solitary Keratoacanthoma at the Recipient Site of a Full-Thickness Skin Graft: A Case Report and Review of the Literature. Arch Plast Surg 2023, 50(1), 59–62. [Google Scholar] [CrossRef]
- Fradet, M.; Sibaud, V.; Tournier, E.; Lamant, L.; Boulinguez, S.; Brun, A.; Pages, C.; Meyer, N. Multiple Keratoacanthoma-like Lesions in a Patient Treated with Pembrolizumab. Acta Derm Venereol 2019, 99(13), 1301–2. [Google Scholar] [CrossRef]
- Li, S.; Ye, X.; Li, X.; Yang, Y. Case Report: Multiple cutaneous keratoacanthoma-like lesions in a colorectal cancer patient treated with sintilimab. Front Immunol 2025, 16, 1535220. [Google Scholar] [CrossRef]
- Frances, L.; Guijarro, J.; Marin, I.; Leiva-Salinas, M. del C, Bouret, A.M. Multiple eruptive keratoacanthomas associated with leflunomide. Dermatol Online J 2013, 19(7), 18968. [Google Scholar] [CrossRef]
- Dobre, A.; Nedelcu, R.I.; Turcu, G.; Brinzea, A.; Struna, I.; Tudorache, G.; Ali, A.; Hulea, I.; Balasescu, E.; Fertig, T.E.; Gherghiceanu, M.; Harwood, C.; Ion, D.A.; Forsea, A.M. Multiple Keratoacanthomas Associated with Genetic Syndromes: Narrative Review and Proposal of a Diagnostic Algorithm. Am J Clin Dermatol 2025, 26(1), 45–59. [Google Scholar] [CrossRef]
- Neagu, N.; Dianzani, C.; Venuti, A.; Bonin, S.; Voidăzan, S.; Zalaudek, I.; Conforti, C. The role of HPV in keratinocyte skin cancer development: A systematic review. J Eur Acad Dermatol Venereol 2023, 37(1), 40–6. [Google Scholar] [CrossRef]
- Ljubojevic, S.; Skerlev, M. HPV-associated diseases. Clin Dermatol 2014, 32(2), 227–34. [Google Scholar] [CrossRef]
- Conforti, C.; Paolini, F.; Venuti, A.; Dianzani, C.; Zalaudek, I. The detection rate of human papillomavirus in well-differentiated squamous cell carcinoma and keratoacanthoma: is there new evidence for a viral pathogenesis of keratoacanthoma? Br J Dermatol 2019, 181(6), 1309–11. [Google Scholar] [CrossRef]
- McLaughlin-Drubin, M.E. . Human papillomaviruses and non-melanoma skin cancer. Semin Oncol 2015, 42, 284–290. [Google Scholar] [CrossRef]
- Arroyo Mühr, L.S.; Hultin, E.; Bzhalava, D.; Eklund, C.; Lagheden, C.; Ekström, J.; Johansson, H.; Forslund, O.; Dillner, J. Human papillomavirus type 197 is commonly present in skin tumors. Int J Cancer 2015, 136(11), 2546–55. [Google Scholar]
- Eversole, L.R.; Leider, A.S.; Alexander, G. Intraoral and labial keratoacanthoma. Oral Surg Oral Med Oral Pathol 1982, 54(6), 663–7. [Google Scholar]
- Cristalli, M.P. ; Marini, R; La Monaca, G.; Vitolo, D.; Pompa, G.; Annibali, S. Double localization of keratoacantho-ma on the cutaneous and mucosal sides of the lower lip: report of a case. Oral Implantol (Rome) 2013, 6, 94–98. [Google Scholar]
- Janette, A.; Pecaro, B.; Lonergan, M.; Lingen, M.W. Solitary intraoral keratoacanthoma: report of a case. J Oral Maxillofac Surg 1996, 54(8), 1026–30. [Google Scholar]
- Habel, G; O’Regan, B. ; Eissing, A.; Khoury, F.; Donath, K. Intra-oral keratoacanthoma: an eruptive variant and review of the literature. Br Dent J 1991, 170(9), 336–9. [Google Scholar] [CrossRef]
- Whyte, A.M.; Hansen, L.S.; Lee, C. The intraoral keratoacanthoma: a diagnostic problem. Br J Oral Maxillofac Surg 1986, 24(6), 438–41. [Google Scholar] [CrossRef]
- Heidenreich, R.K.; Gongloff, R.K.; Wescott, W.B. A solitary, exophytic, crateriform lesion on the mandibular retromolar gingiva. J Am Dent Assoc 1986, 112(3), 377–9. [Google Scholar]
- Svirsky, J.A.; Freedman, P.D.; Lumerman, H. Solitary intraoral keratoacanthoma. Oral Surg Oral Med Oral Pathol 1977, 43(1), 116–22. [Google Scholar] [PubMed]
- Zegarelli, D.J. Solitary intraoral keratoacanthoma. Oral Surg Oral Med Oral Pathol 1975, 40(6), 785–8. [Google Scholar] [PubMed]
- Divers, A.K.; Correale, D.; Lee, J.B. Keratoacanthoma centrifugum marginatum: a diagnostic and therapeutic challenge. Cutis 2004, 73(4), 257–62. [Google Scholar]
- Wu, T.P.; Miller, K.; Cohen, D.E.; Stein, J.A. Keratoacanthomas arising in association with prurigo nodules in pruritic, actinically damaged skin. J Am Acad Dermatol 2013, 69(3), 426–30. [Google Scholar]
- Goudie, D.R.; D’Alessandro, M.; Merriman, B.; Lee, H.; Szeverényi, I.; Avery, S.; O'Connor, B.D.; Nelson, S.F.; Coats, S.E.; Stewart, A.; Christie, L.; Pichert, G.; Friedel, J.; Hayes, I.; Burrows, N.; Whittaker, S.; Gerdes, A.M.; Broesby-Olsen, S.; Ferguson-Smith, M.A.; Verma, C.; Lunny, D.P.; Reversade, B.; Lane, E.B. Multiple self-healing squamous epithelioma is caused by a disease-specific spectrum of mutations in TGFBR1. Nat Genet 2011, 43(4), 365–9. [Google Scholar]
- Grzybowski, M. A case of peculiar generalized epithelial tumours of the skin. Br J Dermatol Syph 1950, 62, 310–313. [Google Scholar]
- Anzalone, C.L.; Cohen, P.R. Generalized eruptive keratoacanthomas of Grzybowski. Int J Dermatol 2014, 53(2), 131–6. [Google Scholar]
- Agarwal, M.; Chander, R.; Karmakar, S.; Walia, R. Multiple familial keratoacanthoma of Witten and Zak - A report of three siblings. Dermatology 1999, 198(4), 396–9. [Google Scholar]
- Boateng, B.; Hornstein, O.P.; von den Driesch, P.; Kiesewetter, F. [Multiple keratoacanthomas (Witten-Zak type) in prurigo simplex subacuta]. Hautarzt 1995, 46(2), 114–7. [Google Scholar]
- Rosendahl, C.; Cameron, A.; Argenziano, G.; Zalaudek, I.; Tschandl, P.; Kittler, H. Dermoscopy of squamous cell carcinoma and keratoacanthoma. Arch Dermatol 2012, 148(12), 1386–92. [Google Scholar] [PubMed]
- Zalaudek, I.; Giacomel, J.; Schmid, K.; Bondino, S.; Rosendahl, C.; Cavicchini, S.; Tourlaki, A.; Gasparini, S.; Bourne, P.; Keir, J.; Kittler, H.; Eibenschutz, L.; Catricalà, C.; Argenziano, G. Dermatoscopy of facial actinic keratosis, intraepidermal carcinoma, and invasive squamous cell carcinoma: a progression model. J Am Acad Dermatol 2012, 66(4), 589–97. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Wang, H.; Zheng, L.; Guo, Z.; Fan, X.; Gao, M. The first report of diagnosing of keratoacanthoma in Chinese Han patients using dermoscopy and reflectance confocal microscopy. Skin Res Technol 2021, 27(3), 422–7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.P.; Peppelman, M.; Hoogedoorn, L.; Van Erp, P.E.J.; Gerritsen, M.J.P. The current role of in vivo reflectance confocal microscopy within the continuum of actinic keratosis and squamous cell carcinoma: a systematic review. Eur J Dermatol 2016, 26(6), 549–65. [Google Scholar] [PubMed]
- Latriglia, F.; Ogien, J.; Tavernier, C.; Fischman, S.; Suppa, M.; Perrot, J.L.; Dubois, A. Line-Field Confocal Optical Coherence Tomography (LC-OCT) for Skin Imaging in Dermatology. Life (Basel) 2023, 13(12), 2268. [Google Scholar]
- Cinotti, E.; Brunetti, T.; Cartocci, A.; Tognetti, L.; Suppa, M.; Malvehy, J.; Perez-Anker, J.; Puig, S.; Perrot, J.L.; Rubegni, P. Diagnostic Accuracy of Line-Field Confocal Optical Coherence Tomography for the Diagnosis of Skin Carcinomas. Diagnostics (Basel) 2023, 13(3), 361. [Google Scholar] [PubMed]
- Schwartz, R.A. Keratoacanthoma: a clinico-pathologic enigma. Dermatol Surg 2004, 30 2 Pt 2, 326–33. [Google Scholar]
- Ogita, A.; Ansai, S.I.; Misago, N.; Anan, T.; Fukumoto, T.; Saeki, H. Histopathological diagnosis of epithelial crateriform tumors: Keratoacanthoma and other epithelial crateriform tumors. J Dermatol 2016, 43(11), 1321–31. [Google Scholar]
- Takai, T. Advances in histopathological diagnosis of keratoacanthoma. J Dermatol 2017, 44(3), 304–14. [Google Scholar] [CrossRef]
- Cribier, B.; Asch, P.; Grosshans, E. Differentiating squamous cell carcinoma from keratoacanthoma using histopathological criteria. Is it possible? A study of 296 cases. Dermatology 1999, 199(3), 208–12. [Google Scholar]
- Savage, J.A.; Maize, J.C. Keratoacanthoma clinical behavior: a systematic review. Am J Dermatopathol 2014, 36(5), 422–9. [Google Scholar] [CrossRef]
- Kossard, S.; Tan, K.B.; Choy, C. Keratoacanthoma and infundibulocystic squamous cell carcinoma. Am J Dermatopathol 2008, 30(2), 127–34. [Google Scholar] [PubMed]
- Weedon, D.D.; Malo, J.; Brooks, D.; Williamson, R. Squamous cell carcinoma arising in keratoacanthoma: a neglected phenomenon in the elderly. Am J Dermatopathol 2010, 32(5), 423–6. [Google Scholar] [PubMed]
- Piscioli, F.; Boi, S.; Zumiani, G.; Cristofolini, M. A gigantic, metastasizing keratoacanthoma. Report of a case and discussion on classification. Am J Dermatopathol 1984, 6(2), 123–9. [Google Scholar] [CrossRef]
- Nelson, C.W.; Mirabello, L. Human papillomavirus genomics: Understanding carcinogenicity. Tumour Virus Research 2023, 15, 200258. [Google Scholar] [CrossRef] [PubMed]
- Skelin, J.; Tomaić, V. Comparative analysis of alpha and beta HPV E6 oncoproteins: Insights into functional distinctions and divergent mechanisms of pathogenesis. Viruses 2023, 15(11), 2253. [Google Scholar] [CrossRef]
- Doorbar, J.; Quint, W.; Banks, L.; Bravo, I.G.; Stoler, M.; Broker, T.R.; Stanley, M.A. The biology and life-cycle of human papillomaviruses. Vaccine 2012, 30 (Suppl 5), F55–70. [Google Scholar] [CrossRef]
- Buck, C.B.; Cheng, N.; Thompson, C.D.; Lowy, D.R.; Steven, A.C.; Schiller, J.T.; et al. Arrangement of L2 within the papillomavirus capsid. J. Virol 2008, 82(11), 5190–7. [Google Scholar]
- Gheit, T. Mucosal and Cutaneous Human Papillomavirus Infections and Cancer Biology. Front Oncol 2019, 9, 355. [Google Scholar] [CrossRef]
- Schiffman, M.; Doorbar, J.; Wentzensen, N.; De Sanjosé, S.; Fakhry, C.; Monk, B.J.; Stanley, M.A.; Franceschi, S. Carcinogenic human papillomavirus infection. Nat. Rev. Dis. Primers 2016, 2(1), 16086. [Google Scholar] [CrossRef]
- Müller, M.; Prescott, E.L.; Wasson, C.W.; Macdonald, A. Human papillomavirus E5 oncoprotein: Function and potential target for antiviral therapeutics. Future Virol 2015, 10(1), 27–39. [Google Scholar] [CrossRef]
- Pešut, E.; Đukić, A.; Lulić, L.; Skelin, J.; Šimić, I.; Milutin Gašperov, N.; Tomaić, V.; Sabol, I.; Grce, M. Human Papillomaviruses-Associated Cancers: An Update of Current Knowledge. Viruses. 2021, 13(11), 2234. [Google Scholar] [CrossRef] [PubMed]
- Messa, L.; Loregian, A. HPV-induced cancers: preclinical therapeutic advancements. Expert Opin Investig Drugs 2022, 31(1), 79–93. [Google Scholar] [CrossRef] [PubMed]
- Egawa, N.; Egawa, K.; Griffin, H.; Doorbar, J. Human Papillomaviruses; Epithelial Tropisms, and the Development of Neoplasia. Viruses. 2015, 7(7), 3863–90. [Google Scholar] [CrossRef]
- Tampa, M.; Mitran, C.I.; Mitran, M.I.; Nicolae, I.; Dumitru, A.; Matei, C.; Manolescu, L.; Popa, G.L.; Caruntu, C.; Georgescu, S.R. The Role of Beta HPV Types and HPV-Associated Inflammatory Processes in Cutaneous Squamous Cell Carcinoma. J Immunol Research 2020, 2020, 1–10. [Google Scholar] [CrossRef]
- Orth, G. Genetics of Epidermodysplasia Verruciformis: Insights into Host Defense against Papillomaviruses. Semin Immunol 2006, 18, 362–74. [Google Scholar] [CrossRef]
- Quint, K.D.; Genders, R.E.; de Koning, M.N.; Borgogna, C.; Gariglio, M.; Bouwes Bavinck, J.N.; Doorbar, J.; Feltkamp, M.C.W. Human Beta-Papillomavirus Infection and Keratinocyte Carcinomas. J Pathol 2015, 235, 342–54. [Google Scholar] [CrossRef]
- Hufbauer, M.; Akgül, B. Molecular Mechanisms of Human Papillomavirus Induced Skin Carcinogenesis. Viruses 2017, 9, 187. [Google Scholar] [CrossRef]
- Schuch, A.P.; Moreno, N.C.; Schuch, N.J.; Menck, C.F.M.; Garcia, C.C.M. Sunlight Damage to Cellular DNA: Focus on Oxidatively Generated Lesions. Free Radic Biol Med 2017, 107, 110–24. [Google Scholar] [CrossRef]
- Douki, T.; von Koschembahr, A.; Cadet, J. Insight in DNA Repair of UV-Induced Pyrimidine Dimers by Chromatographic Methods. Photochem Photobiol 2017, 93, 207–15. [Google Scholar] [CrossRef]
- Brash, D.E.; Ziegler, A.; Jonason, A.S.; Simon, J.A.; Kunala, S.; Leffell, D.J. Sunlight and Sunburn in Human Skin Cancer: p53, Apoptosis, and Tumor Promotion. J Investig Dermatol Symp Proc 1996, 1, 136–42. [Google Scholar] [PubMed]
- Wahl, G.M.; Carr, A.M. The Evolution of Diverse Biological Responses to DNA Damage: Insights from Yeast and p53. Nat Cell Biol 2001, 3, E277–86. [Google Scholar] [CrossRef]
- el-Deiry, W.S.; Harper, J.W.; O’Connor, P.M.; Velculescu, V.E.; Canman, C.E.; Jackman, J.; Pietenpol, J.A.; Burrell, M.; Hill, D.E.; Wang, Y.; Wiman, K.G.; Mercer, W.E.; Kastan, M.B.; Kohn, K.W.; Elledge, S.J.; Kinzler, K.W.; Vogelstein, B. WAF1/CIP1 Is Induced in p53-Mediated G1 Arrest and Apoptosis. Cancer Res 1994, 54, 1169–74. [Google Scholar] [PubMed]
- Scheffner, M.; Werness, B.A.; Huibregtse, J.M.; Levine, A.J.; Howley, P.M. The E6 Oncoprotein Encoded by Human Papillomavirus Types 16 and 18 Promotes the Degradation of p53. Cell 1990, 63, 1129–36. [Google Scholar] [CrossRef] [PubMed]
- Hengstermann, A.; Linares, L.K.; Ciechanover, A.; Whitaker, N.J.; Scheffner, M. Complete Switch from Mdm2 to Human Papillomavirus E6-Mediated Degradation of p53 in Cervical Cancer Cells. Proc Natl Acad Sci USA 2001, 98, 1218–23. [Google Scholar] [CrossRef]
- Li, S.; Hong, X.; Wei, Z.; Xie, M.; Li, W.; Liu, G.; Guo, H.; Yang, J.; Wei, W.; Zhang, S. Ubiquitination of the HPV Oncoprotein E6 Is Critical for E6/E6AP-Mediated p53 Degradation. Front Microbiol 2019, 10, 2483. [Google Scholar] [CrossRef]
- Martinez-Zapien, D.; Ruiz, F.X.; Poirson, J.; Mitschler, A.; Ramirez, J.; Forster, A.; Cousido-Siah, A.; Masson, M.; Vande Pol, S.; Podjarny, A.; Travé, G.; Zanier, K. Structure of the E6/E6AP/p53 Complex Required for HPV-Mediated Degradation of p53. Nature 2016, 529, 541–5. [Google Scholar] [CrossRef]
- Muench, P.; Probst, S.; Schuetz, J.; Leiprecht, N.; Busch, M.; Wesselborg, S.; Stubenrauch, F.; Iftner, T. Cutaneous Papillomavirus E6 Proteins Must Interact with p300 and Block p53-Mediated Apoptosis for Cellular Immortalization and Tumorigenesis. Cancer Res 2010, 70, 6913–24. [Google Scholar] [CrossRef]
- Muschik, D.; Braspenning-Wesch, I.; Stockfleth, E.; Rösl, F.; Hofmann, T.G.; Nindl, I. Cutaneous HPV23 E6 Prevents p53 Phosphorylation through Interaction with HIPK2. PLoS ONE 2011, 6, e27655. [Google Scholar] [CrossRef]
- Minoni, L.; Romero-Medina, M.C.; Venuti, A.; Sirand, C.; Robitaille, A.; Altamura, G.; Tommasino, M.; Accardi, R. Transforming Properties of Beta-3 Human Papillomavirus E6 and E7 Proteins. mSphere 2020, 5, e00398. [Google Scholar] [CrossRef] [PubMed]
- Cornet, I.; Bouvard, V.; Campo, M.S.; Thomas, M.; Banks, L.; Gissmann, L.; Tommasino, M. Comparative Analysis of Transforming Properties of E6 and E7 from Different Beta Human Papillomavirus Types. J Virol 2012, 86, 2366–70. [Google Scholar] [CrossRef]
- White, E.A.; Kramer, R.E.; Tan, M.J.A.; Hayes, S.D.; Harper, J.W.; Howley, P.M. Comprehensive Analysis of Host Cellular Interactions with Human Papillomavirus E6 Proteins Identifies New E6 Binding Partners and Reflects Viral Diversity. J Virol 2012, 86, 13174–86. [Google Scholar] [PubMed]
- Accardi, R.; Dong, W.; Smet, A.; Cui, R.; Hautefeuille, A.; Gabet, A.; Tommasino, M. Skin Human Papillomavirus Type 38 Alters p53 Functions by Accumulation of ΔNp73. EMBO Rep 2006, 7, 334–40. [Google Scholar] [CrossRef] [PubMed]
- Di, C.; Yang, L.; Zhang, H.; Ma, X.; Zhang, X.; Sun, C.; Zhu, H. Mechanisms, Function and Clinical Applications of ΔNp73. Cell Cycle 2013, 12, 1861–7. [Google Scholar] [CrossRef]
- Accardi, R.; Scalise, M.; Gheit, T.; Hussain, I.; Yue, J.; Carreira, C.; Tommasino, M. IκB Kinase β Promotes Cell Survival by Antagonizing p53 Functions through ΔNp73α Phosphorylation and Stabilization. Mol Cell Biol 2011, 31, 2210–26. [Google Scholar] [CrossRef] [PubMed]
- Peña-Blanco, A.; García-Sáez, A.J. Bax, Bak and Beyond — Mitochondrial Performance in Apoptosis. FEBS J 2018, 285, 416–31. [Google Scholar] [CrossRef]
- Jackson, S.; Harwood, C.; Thomas, M.; Banks, L.; Storey, A. Role of Bak in UV-Induced Apoptosis in Skin Cancer and Abrogation by HPV E6 Proteins. Genes Dev 2000, 14, 3065–73. [Google Scholar] [CrossRef]
- Thomas, M.; Banks, L. Inhibition of Bak-Induced Apoptosis by HPV-18 E6. Oncogene 1998, 17, 2943–54. [Google Scholar] [CrossRef]
- Magal, S.S.; Jackman, A.; Ish-Shalom, S.; Botzer, L.E.; Gonen, P.; Schlegel, R.; Shai, A. Downregulation of Bax mRNA Expression and Protein Stability by the E6 Protein of Human Papillomavirus 16. J. Gen. Virol. 2005, 86, 611–21. [Google Scholar] [CrossRef]
- Lee, J.O.; Russo, A.A.; Pavletich, N.P. Structure of the Retinoblastoma Tumour-Suppressor Pocket Domain Bound to a Peptide from HPV E7. Nature 1998, 391, 859–65. [Google Scholar] [CrossRef]
- Davies, R.; Hicks, R.; Crook, T.; Morris, J.; Vousden, K. Human Papillomavirus Type 16 E7 Associates with a Histone H1 Kinase and with p107 through Sequences Necessary for Transformation. J Virol 1993, 67, 2521–28. [Google Scholar]
- Zhang, B.; Chen, W.; Roman, A. The E7 Proteins of Low- and High-Risk Human Papillomaviruses Share the Ability to Target the pRB Family Member p130 for Degradation. Proc Natl Acad Sci USA 2006, 103, 437–42. [Google Scholar] [CrossRef]
- Münger, K.; Werness, B.A.; Dyson, N.; Phelps, W.C.; Harlow, E.; Howley, P.M. Complex Formation of Human Papillomavirus E7 Proteins with the Retinoblastoma Tumor Suppressor Gene Product. EMBO J 1989, 8, 4099–105. [Google Scholar] [CrossRef] [PubMed]
- Szalmás, A.; Tomaić, V.; Basukala, O.; Massimi, P.; Mittal, S.; Kónya, J.; Banks, L. The PTPN14 Tumor Suppressor Is a Degradation Target of Human Papillomavirus E7. J Virol 2017, 91, e00057–17. [Google Scholar] [CrossRef]
- Hatterschide, J.; Bohidar, A.E.; Grace, M.; Nulton, T.J.; Kim, H.W.; Windle, B.; Morgan, I.M. PTPN14 Degradation by High-Risk Human Papillomavirus E7 Limits Keratinocyte Differentiation and Contributes to HPV-Mediated Oncogenesis. Proc Natl Acad Sci USA 2019, 116, 7033–42. [Google Scholar] [CrossRef]
- Lee, H.S.; Kim, M.W.; Jin, K.S.; Shin, H.C.; Kim, W.K.; Lee, S.C.; Park, J.H. Molecular Analysis of the Interaction between Human PTPN21 and the Oncoprotein E7 from Human Papillomavirus Genotype 18. Mol Cells 2021, 44, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Trejo-Cerro, O.; Massimi, P.; Broniarczyk, J.; Myers, M.; Banks, L. Repression of Memo1, a Novel Target of Human Papillomavirus Type 16 E7, Increases Cell Proliferation in Cervical Cancer Cells. J Virol 2022, 96. [Google Scholar] [CrossRef] [PubMed]
- Romero-Masters, J.C.; Lambert, P.F.; Munger, K. Molecular Mechanisms of MmuPV1 E6 and E7 and Implications for Human Disease. Viruses 2022, 14. [Google Scholar] [CrossRef]
- Giarrè, M.; Caldeira, S.; Malanchi, I.; Ciccolini, F.; Leão, M.J.; Tommasino, M. Induction of pRb Degradation by the Human Papillomavirus Type 16 E7 Protein Is Essential To Efficiently Overcome p16^INK4a^-Imposed G1 Cell Cycle Arrest. J Virol 2001, 75(10), 4705–12. [Google Scholar] [CrossRef]
- Caldeira, S.; Zehbe, I.; Accardi, R.; Malanchi, I.; Dong, W.; Giarrè, M.; Tommasino, M. The E6 and E7 Proteins of the Cutaneous Human Papillomavirus Type 38 Display Transforming Properties. J Virol 2003, 77(3), 2195–206. [Google Scholar] [CrossRef]
- Wallace, N.A.; Robinson, K.; Howie, H.L.; Galloway, D.A. HPV 5 and 8 E6 Abrogate ATR Activity Resulting in Increased Persistence of UVB Induced DNA Damage. PLoS Pathog 2012, 8(7), e1002807. [Google Scholar] [CrossRef]
- Tran, D.C.; Li, S.; Henry, S.; Wood, D.J.; Chang, A.L.S. An 18-year retrospective study on the outcomes of keratoacanthomas with different treatment modalities at a single academic centre. Br J Dermatol 2017, 177(6), 1749–51. [Google Scholar] [CrossRef]
- Caccialanza, M.; Sopelana, N. Radiation therapy of keratoacanthomas: results in 55 patients. Int J Radiat Oncol Biol Phys 1989, 16(2), 475–7. [Google Scholar] [CrossRef] [PubMed]
- Donahue, B.; Cooper, J.S.; Rush, S. Treatment of aggressive keratoacanthomas by radiotherapy. J Am Acad Dermatol 1990, 23 3 Pt 1, 489–93. [Google Scholar] [CrossRef]
- Shimm, D.S.; Duttenhaver, J.R.; Doucette, J.; Wang, C.C. Radiation therapy of keratoacanthoma. Int J Radiat Oncol Biol Phys 1983, 9(5), 759–61. [Google Scholar] [CrossRef]
- Shaw, J.C.; Storrs, F.J.; Everts, E. Multiple keratoacanthomas after megavoltage radiation therapy. J Am Acad Dermatol 1990, 23 5 Pt 2, 1009–11. [Google Scholar] [CrossRef]
- Kuflik, E.G. Cryosurgery for cutaneous malignancy: An update. Dermatol Surg 1997, 23(11), 1081–87. [Google Scholar] [CrossRef]
- Gogia, R.; Grekin, R.C.; Shinkai, K. Eruptive self-resolving keratoacanthomas developing after treatment with photodynamic therapy and microdermabrasion. Dermatol Surg 2013, 39, 1717–20. [Google Scholar] [CrossRef]
- Neumann, R.A.; Knobler, R.M. Argon laser treatment of small keratoacanthomas in difficult locations. Int J Dermatol 1990, 29(10), 733–6. [Google Scholar] [CrossRef]
- Yoshioka, A.; Tanaka, S.; Hiraoka, O.; Koyama, Y.; Hirota, Y.; Ayusawa, D.; Nishimoto, I. Deoxyribonucleoside triphosphate imbalance. 5-Fluorodeoxyuridine-induced DNA double strand breaks in mouse FM3A cells and the mechanism of cell death. J Biol Chem 1987, 262, 8235–41. [Google Scholar] [CrossRef] [PubMed]
- Thompson, B.J.; Ravits, M.; Silvers, D.N. Clinical efficacy of short contact topical 5-Fluorouracil in the treatment of keratoacanthomas: a retrospective analysis. J Clin Aesthet Dermatol 2014, 7(11), 35–7. [Google Scholar]
- Goette, D.K.; Odom, R.B. Successful treatment of keratoacanthoma with intralesional fluorouracil. J Am Acad Dermatol 1980, 2(3), 212–6. [Google Scholar] [CrossRef] [PubMed]
- Thiele, J.J.; Ziemer, M.; Fuchs, S.; Elsner, P. Combined 5-fluorouracil and Er:YAG laser treatment in a case of recurrent giant keratoacanthoma of the lower leg. Dermatol Surg 2004, 30 12 Pt 2, 1556–60. [Google Scholar]
- Jeon, H.C.; Choi, M.; Paik, S.H.; Ahn, C.H.; Park, H.S.; Cho, K.H. Treatment of keratoacanthoma with 5% imiquimod cream and review of the previous report. Ann Dermatol 2011, 23(3), 357–61. [Google Scholar] [CrossRef]
- Gavric, G.; Lekic, B.; Milinkovic Sreckovic, M.; Bosic, M.; Zivanovic, D. Keratoacanthoma centrifugum marginatum associated with mechanical trauma: Response to acitretin—A case report and review of the literature. Dermatol Ther 2020, 33(3), e13397. [Google Scholar] [CrossRef]
- Mizuta, H.; Takahashi, A.; Mori, T.; Namikawa, K.; Nakano, E.; Muto, Y.; et al. Efficacy of oral retinoids for keratoacanthoma centrifugum marginatum. Dermatol Ther 2020, 33(3), e13291. [Google Scholar] [CrossRef]
- Zhang, S.; Han, D.; Wang, T.; Liu, Y. Multiple keratoacanthoma and oral lichen planus successfully treated with systemic retinoids and review of multiple keratoacanthoma associated with lichen planus. Int J Dermatol 2018, 57, 1125–7. [Google Scholar] [CrossRef]
- Reid, D.C.; Guitart, J.; Agulnik, M.; Lacouture, M.E. Treatment of multiple keratoacanthomas with erlotinib. Int. J. Clin. Oncol. 2010, 15, 413–5. [Google Scholar] [CrossRef] [PubMed]
- Pogoda, C.S.; Roden, R.B.S.; Garcea, R.L. Immunizing against Anogenital Cancer: HPV Vaccines. PLoS Pathog 2016, 12, e1005587. [Google Scholar] [CrossRef]
- Pouyanfard, S.; Spagnoli, G.; Bulli, L.; Balz, K.; Yang, F.; Odenwald, C.; et al. Minor Capsid Protein L2 Polytope Induces Broad Protection against Oncogenic and Mucosal Human Papillomaviruses. J Virol 2018, 92, e01930–17. [Google Scholar] [CrossRef]
- Seitz, H.; Ribeiro-Müller, L.; Canali, E.; Bolchi, A.; Tommasino, M.; Ottonello, S.; et al. Robust In Vitro and In Vivo Neutralization against Multiple High-Risk HPV Types Induced by a Thermostable Thioredoxin-L2 Vaccine. Cancer Prev Res 2015, 8, 932–41. [Google Scholar] [CrossRef] [PubMed]
- German Cancer Research Center. PANHPVAX, Study of a New HPV Vaccine in Healthy Volunteers (PANHPVAX). 2023 [cited 2025 Aug 8]. Available from: https://clinicaltrials.gov/study/NCT05208710?intr=Cyclic.
- Mariz, F.C.; Balz, K.; Dittrich, M.; Zhang, Y.; Yang, F.; Zhao, X.; et al. A broadly protective vaccine against cutaneous human papillomaviruses. NPJ Vaccines 2022, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Bar-Ilan, E.; Bar, J.; Baniel, A.; Slodownik, D.; Artzi, O.; Samuelov, L.; et al. Intralesional human papillomavirus vaccine for the treatment of recalcitrant cutaneous warts. J Dermatol 2023, 50, 1373–80. [Google Scholar] [CrossRef] [PubMed]
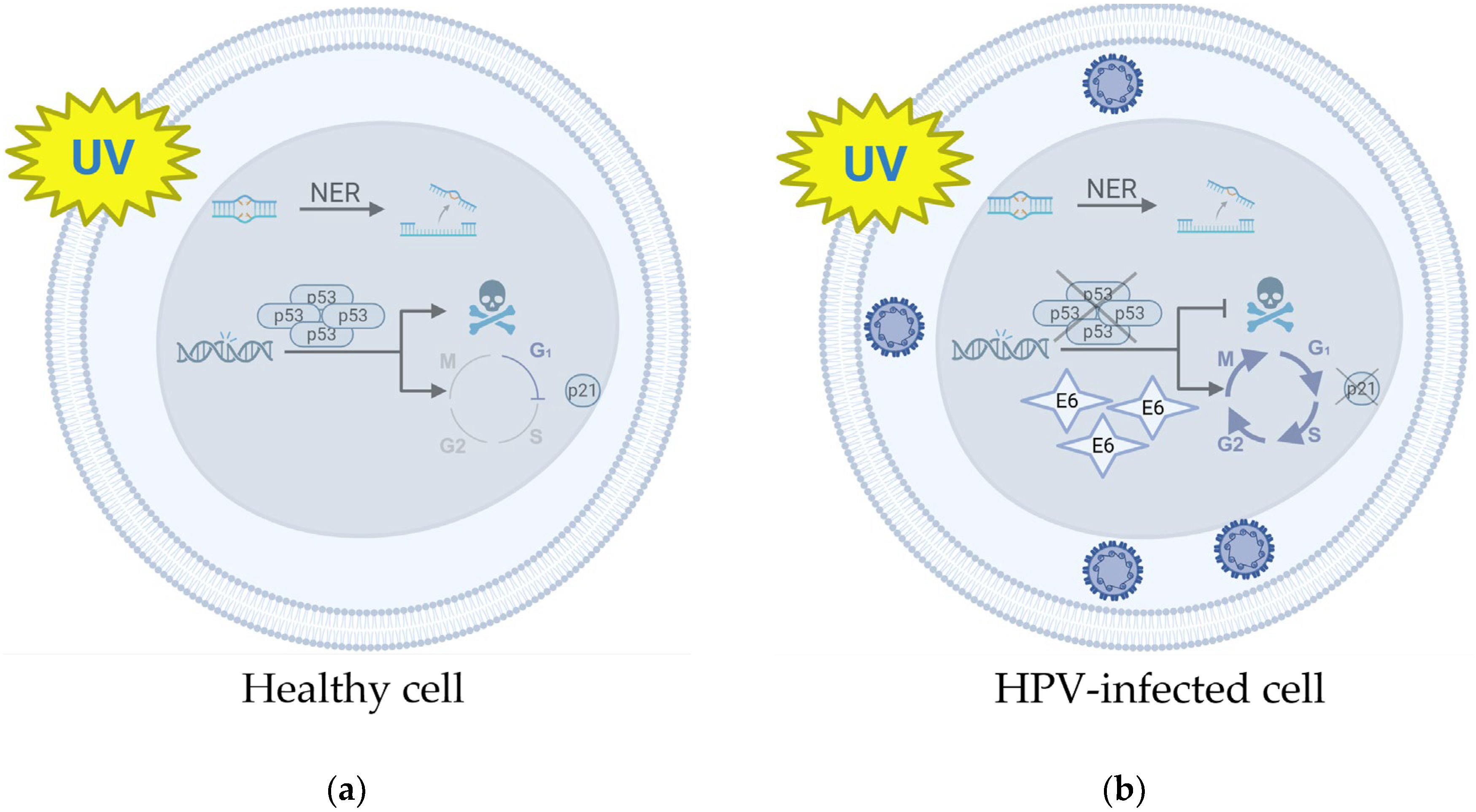
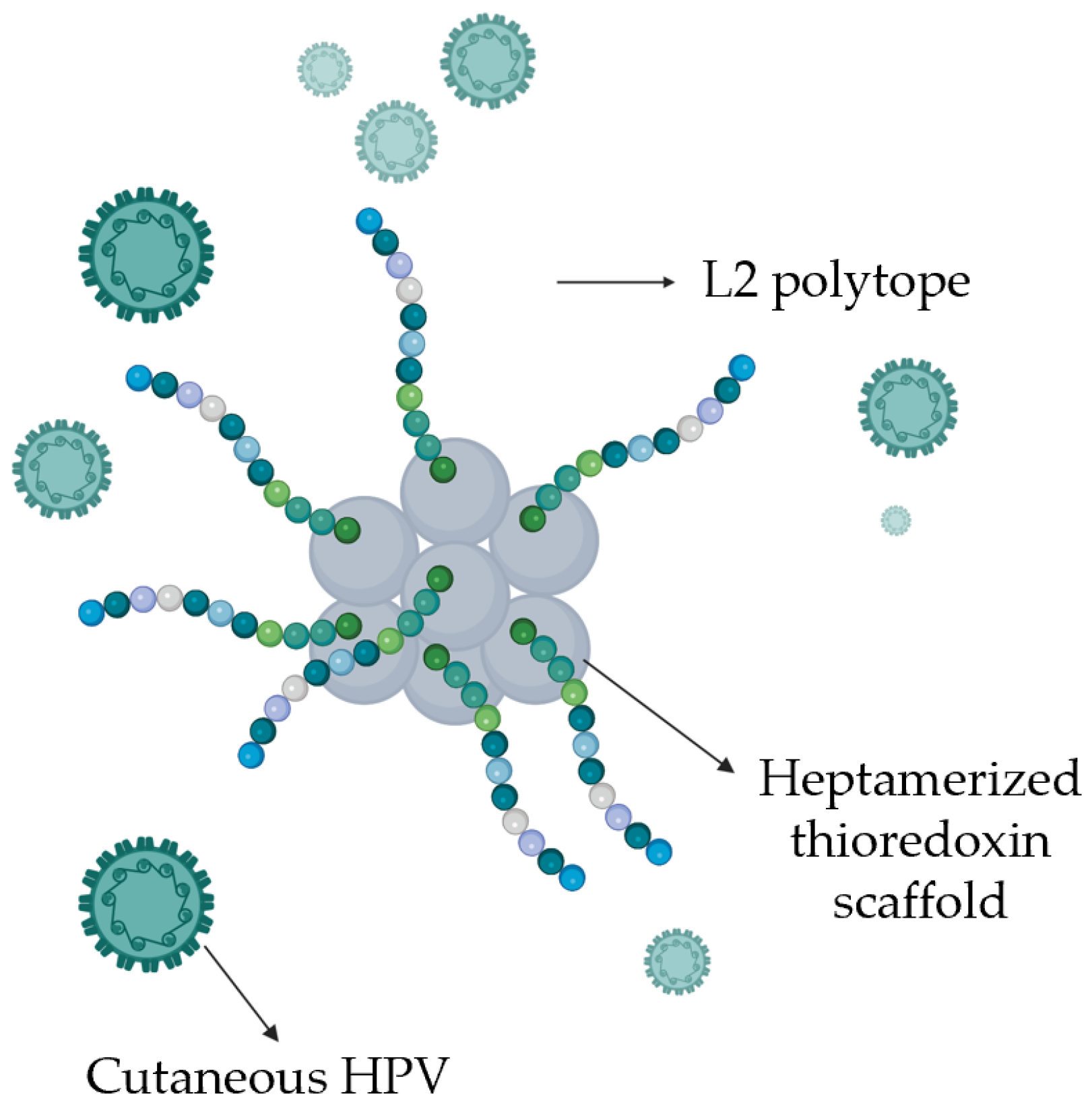
| Therapy type | Description | Mechanism | Effectiveness | ||
|---|---|---|---|---|---|
 surgical |
Complete surgical removal | Surgical removal with histopathological control | Gold-standard, but 8% recurrence | ||
 physical |
Radiotherapy | Targeted destruction of lesions via | Energy | Effective, but rarely used | |
| Cryotherapy | Cold | Effective (~87%), but not widely studied | |||
| PDT1 | Locally activated drug | Limited available data | |||
| Argon laser | Laser | Effective for solitary KA2 | |||
 topical |
Local application of | 5-FU3 | Inhibition of DNA synthesis | Very effective, first-line treatment | |
| Imiquimod | Local immune response activation | Effective | |||
 systemic |
Retinoids | Control of proliferation, differentiation, and immune response | Valuable option for generalized eruptive KA2 | ||
| Erlotinib | EGFR4 inhibition | Still limited experience | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




