Submitted:
27 August 2025
Posted:
29 August 2025
You are already at the latest version
Abstract
Keywords:
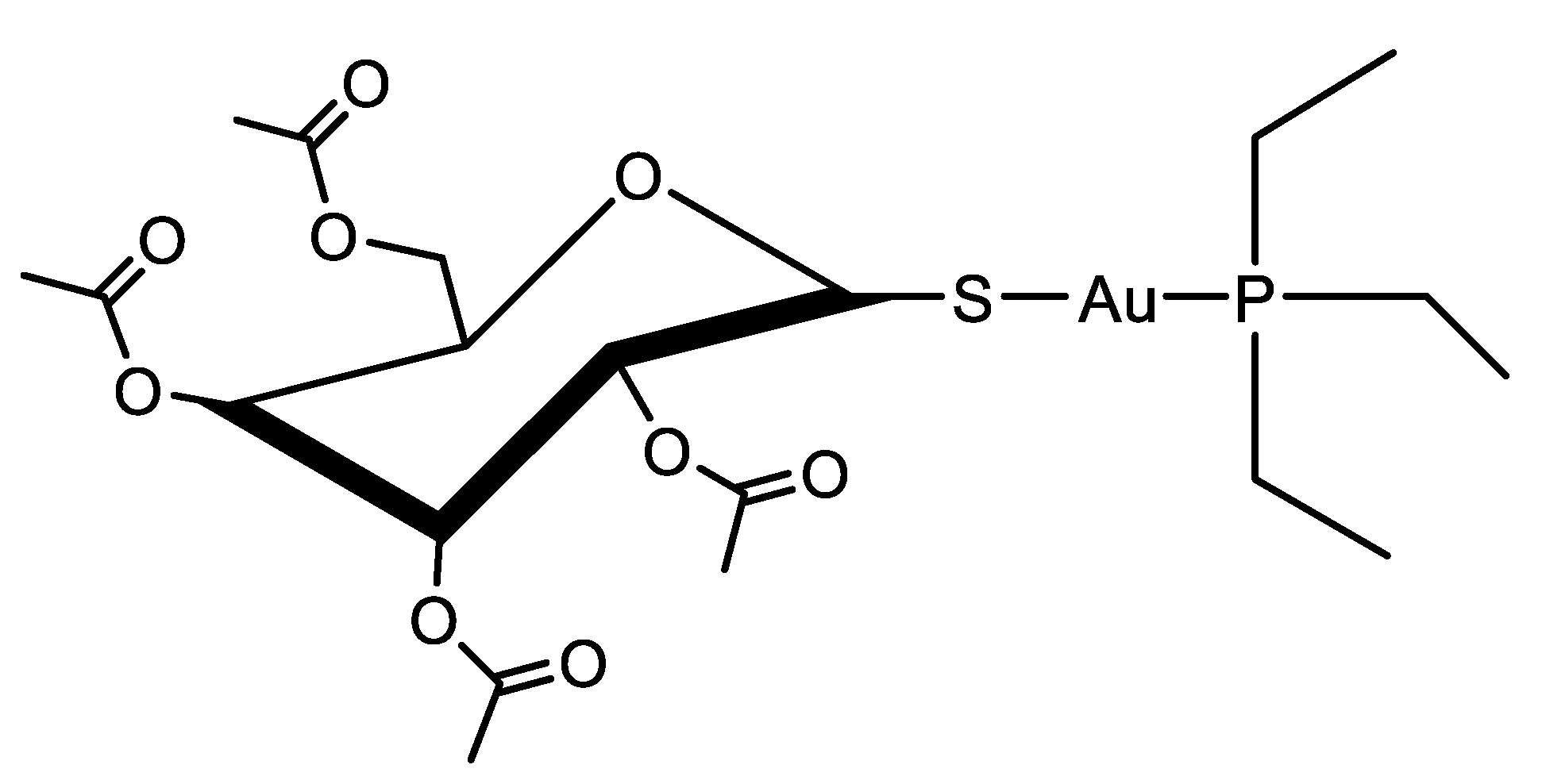
1. Introduction
2. Auranofin-based Monotherapy for Cancer Management
NCT01747798. Auranofin in Treating Patients with Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer https://clinicaltrials.gov/study/NCT01747798
NCT01419691. Phase I and II Study of AF in Chronic Lymphocytic Leukemia (CLL) https://clinicaltrials.gov/study/NCT01419691
NCT02063698. Auranofin in Decreasing Pain in Patients with Paclitaxel-Induced Pain Syndrome https://clinicaltrials.gov/study/NCT02063698
3. Combinatory Regimens for Cancer Management Involving Auranofin
NCT01737502. Sirolimus and Auranofin in Treating Patients with Advanced or Recurrent Non-Small Cell Lung Cancer or Small Cell Lung Cancer https://clinicaltrials.gov/study/NCT01737502
Results and Conclusions. Although the clinical trial NCT01737502 has been completed, as of the latest update on March 25, 2024, no results have been posted.NCT03456700. Auranofin and Sirolimus in Treating Participants with Ovarian Cancer https://clinicaltrials.gov/study/NCT03456700
NCT02770378. A Proof-of-concept Clinical Trial Assessing the Safety of the Coordinated Undermining of Survival Paths by 9 Repurposed Drugs Combined with Metronomic Temozolomide (CUSP9v3 Treatment Protocol) for Recurrent Glioblastoma https://clinicaltrials.gov/study/NCT02770378
4. Auranofin in Non-Oncological Diseases
NCT02089048. Auranofin pharmacokinetic (PK) Following Oral Dose Administration https://clinicaltrials.gov/study/NCT02089048
NCT02736968. Auranofin for Giardia Protozoa and Entamoeba histolytica http://clinicaltrials.gov/study/NCT02736968
NCT02961829. Multi Interventional Study Exploring HIV-1 Residual Replication: a Step Towards HIV-1 Eradication and Sterilizing Cure https://clinicaltrials.gov/study/NCT02961829
5. Conclusions and Future Perspectives
Author Contributions
Funding
References
- Yamashita, M. Auranofin: Past to Present, and Repurposing. Int. Immunopharmacol. 2021, 101, 108272. [Google Scholar] [CrossRef]
- Betts, K.A.; Griffith, J.; Ganguli, A.; Li, N.; Douglas, K.; Wu, E.Q. Economic Burden and Treatment Patterns of Cycling between Conventional Synthetic Disease-Modifying Antirheumatic Drugs among Biologic-Treated Patients with Rheumatoid Arthritis. Clin. Ther. 2016, 38, 1205–1216. [Google Scholar] [CrossRef] [PubMed]
- Marzo, T.; Messori, L. A Role for Metal-Based Drugs in Fighting COVID-19 Infection? The Case of Auranofin. ACS Med. Chem. Lett. 2020, 11, 1067–1068. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lu, Y.; Xu, Z.; Ma, X.; Chen, X.; Liu, W. Repurposing of the Gold Drug Auranofin and a Review of Its Derivatives as Antibacterial Therapeutics. Drug Discov. Today 2022, 27, 1961–1973. [Google Scholar] [CrossRef] [PubMed]
- Gamberi, T.; Chiappetta, G.; Fiaschi, T.; Modesti, A.; Sorbi, F.; Magherini, F. Upgrade of an Old Drug: Auranofin in Innovative Cancer Therapies to Overcome Drug Resistance and to Increase Drug Effectiveness. Med. Res. Rev. 2022, 42, 1111–1146. [Google Scholar] [CrossRef]
- Capparelli, E. V.; Bricker-Ford, R.; Rogers, M.J.; McKerrow, J.H.; Reed, S.L. Phase I Clinical Trial Results of Auranofin, a Novel Antiparasitic Agent. Antimicrob. Agents Chemother. 2017, 61. [Google Scholar] [CrossRef]
- Zhang, X.; Selvaraju, K.; Saei, A.A.; D’Arcy, P.; Zubarev, R.A.; Arnér, E.S.; Linder, S. Repurposing of Auranofin: Thioredoxin Reductase Remains a Primary Target of the Drug. Biochimie 2019, 162, 46–54. [Google Scholar] [CrossRef]
- Bjørklund, G.; Zou, L.; Wang, J.; Chasapis, C.T.; Peana, M. Thioredoxin Reductase as a Pharmacological Target. Pharmacol. Res. 2021, 174, 105854. [Google Scholar] [CrossRef]
- Bindoli, A.; Rigobello, M.P.; Scutari, G.; Gabbiani, C.; Casini, A.; Messori, L. Thioredoxin Reductase: A Target for Gold Compounds Acting as Potential Anticancer Drugs. Coord. Chem. Rev. 2009, 253, 1692–1707. [Google Scholar] [CrossRef]
- Marzano, C.; Gandin, V.; Folda, A.; Scutari, G.; Bindoli, A.; Rigobello, M.P. Inhibition of Thioredoxin Reductase by Auranofin Induces Apoptosis in Cisplatin-Resistant Human Ovarian Cancer Cells. Free Radic. Biol. Med. 2007, 42, 872–881. [Google Scholar] [CrossRef]
- Jackson-Rosario, S.; Cowart, D.; Myers, A.; Tarrien, R.; Levine, R.L.; Scott, R.A.; Self, W.T. Auranofin Disrupts Selenium Metabolism in Clostridium Difficile by Forming a Stable Au-Se Adduct. J. Biol. Inorg. Chem. 2009, 14, 507–519. [Google Scholar] [CrossRef]
- Arnér, E.S.J.; Holmgren, A. The Thioredoxin System in Cancer. Semin. Cancer Biol. 2006, 16, 420–426. [Google Scholar] [CrossRef]
- Jia, J.J.; Geng, W.S.; Wang, Z.Q.; Chen, L.; Zeng, X.S. The Role of Thioredoxin System in Cancer: Strategy for Cancer Therapy. Cancer Chemother. Pharmacol. 2019, 84, 453–470. [Google Scholar] [CrossRef]
- Gencheva, R.; Arner, E.S.J. Thioredoxin Reductase Inhibition for Cancer Therapy. Annu. Rev. Pharmacol. Toxicol. 2021, 62, 177–196. [Google Scholar] [CrossRef]
- Berek, J.S.; Renz, M.; Kehoe, S.; Kumar, L.; Friedlander, M. Cancer of the Ovary, Fallopian Tube, and Peritoneum: 2021 Update. Int. J. Gynecol. Obstet. 2021, 155, 61–85. [Google Scholar] [CrossRef]
- Ding, D.N.; Xie, L.Z.; Shen, Y.; Li, J.; Guo, Y.; Fu, Y.; Liu, F.Y.; Han, F.J. Insights into the Role of Oxidative Stress in Ovarian Cancer. Oxid. Med. Cell. Longev. 2021, 2021, 8388258. [Google Scholar] [CrossRef]
- Abdalbari, F.H.; Telleria, C.M. The Gold Complex Auranofin: New Perspectives for Cancer Therapy. Discov. Oncol. 2021, 12, 1–18. [Google Scholar] [CrossRef]
- Momenimovahed, Z.; Mazidimoradi, A.; Allahqoli, L.; Salehiniya, H. The Role of CA-125 in the Management of Ovarian Cancer: A Systematic Review. Cancer Rep. 2025, 8, e70142. [Google Scholar] [CrossRef] [PubMed]
- Ghia, P.; Ferreri, A.M.; Galigaris-Cappio, F. Chronic Lymphocytic Leukemia. Crit. Rev. Oncol. Hematol. 2007, 64, 234–246. [Google Scholar] [CrossRef] [PubMed]
- Fiskus, W.; Saba, N.; Shen, M.; Ghias, M.; Liu, J.; Gupta, S. Das; Chauhan, L.; Rao, R.; Gunewardena, S.; Schorno, K.; et al. Auranofin Induces Lethal Oxidative and Endoplasmic Reticulum Stress and Exerts Potent Preclinical Activity against Chronic Lymphocytic Leukemia. Cancer Res. 2014, 74, 2520–2532. [Google Scholar] [CrossRef]
- Saba, N.; Shen, M.; Ghias, M.; Farooqui, M.; Austin, C.; Schorno, K.; Weir, S.; Bhalla, K.; Wiestner, A. The Gold Compound Auranofin Induces Oxidative Stress and Apoptosis in Primary CLL Cells Independent of Classic Prognostic Markers and the Protective Effect of the Tissue Microenvironment. Blood 2012, 120, 865–865. [Google Scholar] [CrossRef]
- Blodgett, R.C.; Pietrusko, R.G. Long-Term Efficacy and Safety of Auranofin: A Review of Clinical Experience. Scand. J. Rheumatol. 1987, 16, 67–78. [Google Scholar] [CrossRef]
- Hleuhel, M.H.; Ben-Dali, Y.; Da Cunha-Bang, C.; Brieghel, C.; Clasen-Linde, E.; Niemann, C.U.; Andersen, M.A. Risk Factors Associated with Richter’s Transformation in Patients with Chronic Lymphocytic Leukaemia: Protocol for a Retrospective Population-Based Cohort Study. BMJ Open 2019, 9, e023566. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Quispe, C.; Patra, J.K.; Singh, Y.D.; Panda, M.K.; Das, G.; Adetunji, C.O.; Michael, O.S.; Sytar, O.; Polito, L.; et al. Paclitaxel: Application in Modern Oncology and Nanomedicine-Based Cancer Therapy. Oxid. Med. Cell. Longev. 2021, 2021, 3687700. [Google Scholar] [CrossRef]
- Alqahtani, F.Y.; Aleanizy, F.S.; El Tahir, E.; Alkahtani, H.M.; AlQuadeib, B.T. Paclitaxel. In Profiles of Drug Substances, Excipients and Related Methodology; Academic Press Inc., 2019; Vol. 44, pp. 205–238 ISBN 9780128171653.
- Yan, X.; Maixner, D.W.; Yadav, R.; Gao, M.; Li, P.; Bartlett, M.G.; Weng, H.R. Paclitaxel Induces Acute Pain via Directly Activating Toll like Receptor 4. Mol. Pain 2015, 11. [Google Scholar] [CrossRef]
- Da̧bek, J.; Kułach, A.; Ga̧sior, Z. Nuclear Factor Kappa-Light-Chain-Enhancer of Activated B Cells (NF-ΚB): A New Potential Therapeutic Target in Atherosclerosis? Pharmacol. Reports 2010, 62, 778–783. [Google Scholar] [CrossRef]
- Chen, W.H.; Chan, K.S.; Gan, T.J.; Chen, C.; Lakshminarayanan, M.; Revicki, D.A. Validation of the Modified Brief Pain Inventory-Exploratory Form in Surgery Patients. Health Outcomes Res. Med. 2010, 1, e17–e28. [Google Scholar] [CrossRef]
- Rehan, M. An Anti-Cancer Drug Candidate OSI-027 and Its Analog as Inhibitors of MTOR: Computational Insights Into the Inhibitory Mechanisms. J. Cell. Biochem. 2017, 118, 4558–4567. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Chen, J.; Yu, Y.; Wu, F.; Shen, X.; Qiu, C.; Zhang, T.; Hong, L.; Zheng, P.; Shao, R.; et al. Compensatory Combination of MTOR and TrxR Inhibitors to Cause Oxidative Stress and Regression of Tumors. Theranostics 2021, 11, 4335–4350. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hill, K.S.; Fields, A.P. PKCi Maintains a Tumor-Initiating Cell Phenotype That Is Required for Ovarian Tumorigenesis. Mol. Cancer Res. 2013, 11, 1624–1635. [Google Scholar] [CrossRef]
- Tyagi, K.; Roy, A.; Mandal, S. Protein Kinase C Iota Promotes Glycolysis via PI3K/AKT/MTOR Signalling in High Grade Serous Ovarian Cancer. Mol. Biol. Rep. 2024, 51, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, Q.T.; Gittleman, H.; Truitt, G.; Boscia, A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011-2015. Neuro. Oncol. 2018, 20, iv1–iv86. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Jezierzański, M.; Nafalska, N.; Stopyra, M.; Furgoł, T.; Miciak, M.; Kabut, J.; Gisterek-Grocholska, I. Temozolomide (TMZ) in the Treatment of Glioblastoma Multiforme—A Literature Review and Clinical Outcomes. Curr. Oncol. 2024, 31, 3994–4002. [Google Scholar] [CrossRef]
- Pendergrass, K.; Hargreaves, R.; Petty, K.J.; Carides, A.D.; Evans, J.K.; Horgan, K.J. Aprepitant: An Oral NK1 Antagonist for the Prevention of Nausea and Vomiting Induced by Highly Emetogenic Chemotherapy. Drugs of Today 2004, 40, 853–863. [Google Scholar] [CrossRef]
- Elewa, H.F.; Hilali, R.; Hess, D.C.; Machado, L.S.; Fagan, S.C. Minocycline for Short-Term Neuroprotection. Pharmacotherapy 2006, 26, 515–521. [Google Scholar] [CrossRef]
- Caminear, M.W.; Harrington, B.S.; Kamdar, R.D.; Kruhlak, M.J.; Annunziata, C.M. Disulfiram Transcends ALDH Inhibitory Activity When Targeting Ovarian Cancer Tumor-Initiating Cells. Front. Oncol. 2022, 12, 762820. [Google Scholar] [CrossRef]
- Rosas, C.; Sinning, M.; Ferreira, A.; Fuenzalida, M.; Lemus, D. Celecoxib Decreases Growth and Angiogenesis and Promotes Apoptosis in a Tumor Cell Line Resistant to Chemotherapy. Biol. Res. 2014, 47, 1–9. [Google Scholar] [CrossRef]
- Fayyaz, S.; Atia-Tul-Wahab; Irshad, R. ; Siddiqui, R.A.; Choudhary, M.I. Antidepressant Sertraline Hydrochloride Inhibits the Growth of HER2+ AU565 Breast Cancer Cell Line through Induction of Apoptosis and Cell Cycle Arrest. Anticancer. Agents Med. Chem. 2024, 24, 1038–1046. [Google Scholar] [CrossRef]
- Wysocki, P.J.; Kwiatkowska, E.P.; Kazimierczak, U.; Suchorska, W.; Kowalczyk, D.W.; Mackiewicz, A. Captopril, an Angiotensin-Converting Enzyme Inhibitor, Promotes Growth of Immunogenic Tumors in Mice. Clin. Cancer Res. 2006, 12, 4095–4102. [Google Scholar] [CrossRef]
- Tsai, Y.C.; Tsai, T.F. Itraconazole in the Treatment of Nonfungal Cutaneous Diseases: A Review. Dermatol. Ther. (Heidelb). 2019, 9, 271–280. [Google Scholar] [CrossRef]
- Laurence, J.; Elhadad, S.; Gostynska, S.; Yu, Z.; Terry, H.; Varshney, R.; Fung, K.M.; Choi, M.E.; Ahamed, J. HIV Protease Inhibitor Ritonavir Induces Renal Fibrosis and Dysfunction: Role of Platelet-Derived TGF-SS1 and Intervention via Antioxidant Pathways. AIDS 2020, 34, 989–1000. [Google Scholar] [CrossRef]
- Halatsch, M.E.; Kast, R.E.; Karpel-Massler, G.; Mayer, B.; Zolk, O.; Schmitz, B.; Scheuerle, A.; Maier, L.; Bullinger, L.; Mayer-Steinacker, R.; et al. A Phase Ib/IIa Trial of 9 Repurposed Drugs Combined with Temozolomide for the Treatment of Recurrent Glioblastoma: CUSP9v3. Neuro-Oncology Adv. 2021, 3. [Google Scholar] [CrossRef]
- Diaz, R.S.; Shytaj, I.L.; Giron, L.B.; Obermaier, B.; della Libera, E.; Galinskas, J.; Dias, D.; Hunter, J.; Janini, M.; Gosuen, G.; et al. Potential Impact of the Antirheumatic Agent Auranofin on Proviral HIV-1 DNA in Individuals under Intensified Antiretroviral Therapy: Results from a Randomised Clinical Trial. Int. J. Antimicrob. Agents 2019, 54, 592–600. [Google Scholar] [CrossRef]
- Coscione, F.; Zineddu, S.; Vitali, V.; Fondi, M.; Messori, L.; Perrin, E. The Many Lives of Auranofin: How an Old Anti-Rheumatic Agent May Become a Promising Antimicrobial Drug. Antibiotics 2024, 13, 652. [Google Scholar] [CrossRef] [PubMed]
- Wassmann, C.; Hellberg, A.; Tannich, E.; Bruchhaus, I. Metronidazole Resistance in the Protozoan Parasite Entamoeba Histolytica Is Associated with Increased Expression of Iron-Containing Superoxide Dismutase and Peroxiredoxin and Decreased Expression of Ferredoxin 1 and Flavin Reductase. J. Biol. Chem. 1999, 274, 26051–26056. [Google Scholar] [CrossRef] [PubMed]
- Upcroft, P.; Upcroft, J.A. Drug Targets and Mechanisms of Resistance in the Anaerobic Protozoa. Clin. Microbiol. Rev. 2001, 14, 150–164. [Google Scholar] [CrossRef]
- Debnath, A.; Parsonage, D.; Andrade, R.M.; He, C.; Cobo, E.R.; Hirata, K.; Chen, S.; García-Rivera, G.; Orozco, E.; Martínez, M.B.; et al. A High-Throughput Drug Screen for Entamoeba Histolytica Identifies a New Lead and Target. Nat. Med. 2012, 18, 956–960. [Google Scholar] [CrossRef]
- Tejman-Yarden, N.; Miyamoto, Y.; Leitsch, D.; Santini, J.; Debnath, A.; Gut, J.; McKerrow, J.H.; Reed, S.L.; Eckmann, L. A Reprofiled Drug, Auranofin, Is Effective against Metronidazole-Resistant Giardia Lamblia. Antimicrob. Agents Chemother. 2013, 57, 2029–2035. [Google Scholar] [CrossRef]
- Ocholaid, E.A.; Karanja, D.M.S.; Elliott, S.J. The Impact of Neglected Tropical Diseases (Ntds) on Health and Wellbeing in Sub-Saharan Africa (Ssa): A Case Study of Kenya. PLoS Negl. Trop. Dis. 2021, 15, 1–19. [Google Scholar] [CrossRef]
- Ilari, A.; Baiocco, P.; Messori, L.; Fiorillo, A.; Boffi, A.; Gramiccia, M.; Di Muccio, T.; Colotti, G. A Gold-Containing Drug against Parasitic Polyamine Metabolism: The X-Ray Structure of Trypanothione Reductase from Leishmania Infantum in Complex with Auranofin Reveals a Dual Mechanism of Enzyme Inhibition. Amino Acids 2012, 42, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Auclair, M.; Guénantin, A.C.; Fellahi, S.; Garcia, M.; Capeau, J. HIV Antiretroviral Drugs, Dolutegravir, Maraviroc and Ritonavir-Boosted Atazanavir Use Different Pathways to Affect Inflammation, Senescence and Insulin Sensitivity in Human Coronary Endothelial Cells. PLoS One 2020, 15, e0226924. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, K.; Chavez, L.; Hakre, S.; Calvanese, V.; Verdin, E. Reactivation of Latent HIV by Histone Deacetylase Inhibitors. Trends Microbiol. 2013, 21, 277–285. [Google Scholar] [CrossRef]
- de Almeida Baptista, M.V.; da Silva, L.T.; Samer, S.; Oshiro, T.M.; Shytaj, I.L.; Giron, L.B.; Pena, N.M.; Cruz, N.; Gosuen, G.C.; Ferreira, P.R.A.; et al. Immunogenicity of Personalized Dendritic-Cell Therapy in HIV-1 Infected Individuals under Suppressive Antiretroviral Treatment: Interim Analysis from a Phase II Clinical Trial. AIDS Res. Ther. 2022, 19, 1–15. [Google Scholar] [CrossRef]
- Cirri, D.; Chiaverini, L.; Pratesi, A.; Marzo, T. Is the Next Cisplatin Already in Our Laboratory? https://doi.org/10.1080/02603594.2022.2152016 2022, 1–14. [CrossRef]
- Chiaverini, L.; Leo, R. Di; Famlonga, L.; Pacini, M.; Baglini, E.; Barresi, E.; Peana, M.F.; Tolbatov, I.; Marrone, A.; Mendola, D. La; et al. The Metal(Loid)s’ Dilemma. What’s the next Step for a New Era of Inorganic Molecules in Medicine? Metallomics 2025, 17, 13. [Google Scholar] [CrossRef]
- Cirri, D.; Bartoli, F.; Pratesi, A.; Baglini, E.; Barresi, E.; Marzo, T. Strategies for the Improvement of Metal-Based Chemotherapeutic Treatments. Biomedicines 2021, 9, 504. [Google Scholar] [CrossRef] [PubMed]
| ClinicalTrials.gov Identifier | Indication | Sponsor | Statusa | Phaseb | Resultsc |
| NCT01747798 | Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer | Mayo Clinic | C(2019) | Early 1 (0) | N/A |
| NCT01419691 | Chronic Lymphocytic Leukemia (CLL) | University of Kansas Medical Center | C(2016) | 2 | N/A |
| NCT02063698 | Paclitaxel-Induced Pain Syndrome | Mayo Clinic | C(2019) | 2 | Y |
| NCT01737502 | Lung cancer | Mayo Clinic | C(2024) | 1/2 | N/A |
| NCT03456700 | Ovarian Cancer | Mayo Clinic | T(2025) | 2 | Y |
| NCT02770378 | Glioblastoma | University of Ulm | C(2021) | 1/2 | N/A |
| NCT02089048 | Amoebiasis | National Institute of Allergy and Infectious Diseases (NIAID) | C(2017) | 1 | N/A |
| NCT02736968 | amoebiasis or giardiasis | National Institute of Allergy and Infectious Diseases (NIAID) | C(2023) | 2 | Y |
| NCT02961829 | HIV | Federal University of São Paulo | C(2020) | N/A | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




