Submitted:
27 August 2025
Posted:
28 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
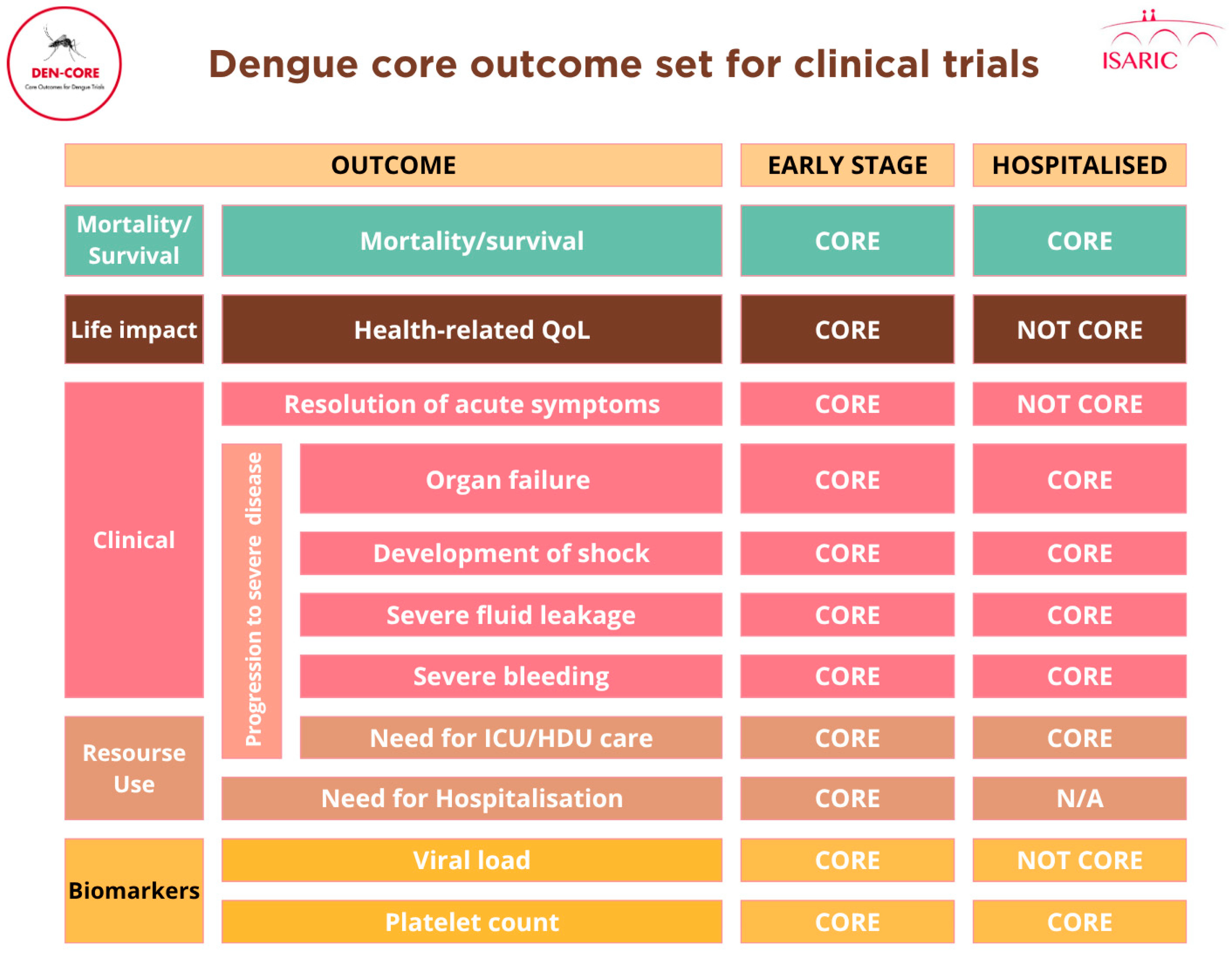
- ‘early stage’ of the disease corresponding to the febrile or viraemic phase, prior to severe manifestations and the vast majority of patients are managed in outpatient settings.
- ‘hospitalised’ stage referred to the late or critical phase of the disease, when patients have developed complications.
- Outcomes specific to trials that recruit individuals admitted to high-dependency units (HDU) or intensive care units (ICU) only were reviewed separately under the “ICU/HDU”.
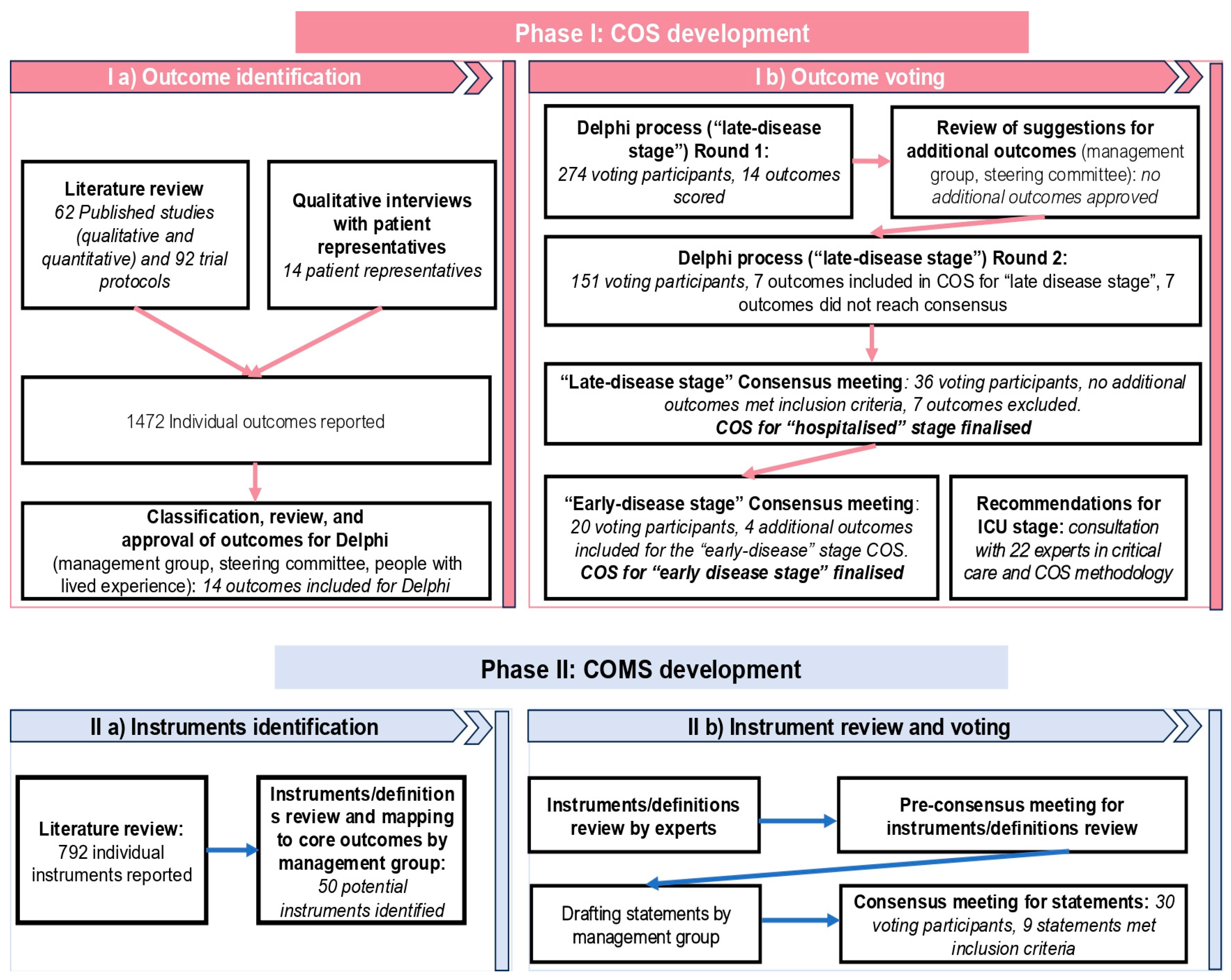
Phase I: COS Development
Phase Ia: Outcome Identification
Phase Ib: Outcome Voting
Data Analysis
Results
Phase I: Core Outcome Set development
Phase Ia: Outcome identification
Phase Ib: Outcome voting
| “Hospitalised” stage COS | “Early stage” COS | COMS | Results | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Outcome | %Stakeholders voting 7-9 in R2 online Delphi | %Stakeholders voting 7-9 in consensus meeting | % Stakeholders voting 7-9 in consensus meeting | % Stakeholders voting for outcome measurement instruments during consensus workshop | |||||
| P | HCP / RS with lived experience | HCP / RS without lived experience | P | HCP/RS | |||||
| Mortality | 94% | 92% | 95% | NA | NA | NA | 100% | Outcome included in the COS both for “hospitalised” and “early-stage” dengue. Agreement has been reached on measurement instrument | |
| Health-related Quality of life | 82% | 49% | 58% | - | 33% | 84% | NA | Outcome excluded for the “hospitalised” dengue COS following consensus meeting. Outcome included in the COS for “early-stage” dengue. OMI were not discussed at the consensus workshop. Work ongoing | |
| Time to recovery | 82% | 65% | 79% | 60% | 64% | NA | NA |
Outcome not included in the COS |
|
| Resolution of acute symptoms | Fatigue | 65% | 30% | 38% | 25% | 20% | 85% | NA | Individual outcomes were excluded for the “hospitalised” dengue COS following consensus meeting. Outcome “Resolution of acute symptoms” included in the COS for “early-stage” dengue. OMI were not discussed at the consensus workshop. Work ongoing |
| Fever | 82% | 43% | 48% | 25% | 42% | ||||
| Pain | 69% | 41% | 37% | - | 13% | ||||
| Severe gastrointestinal symptoms | 88% | 68% | 69% | 40% | 37% | ||||
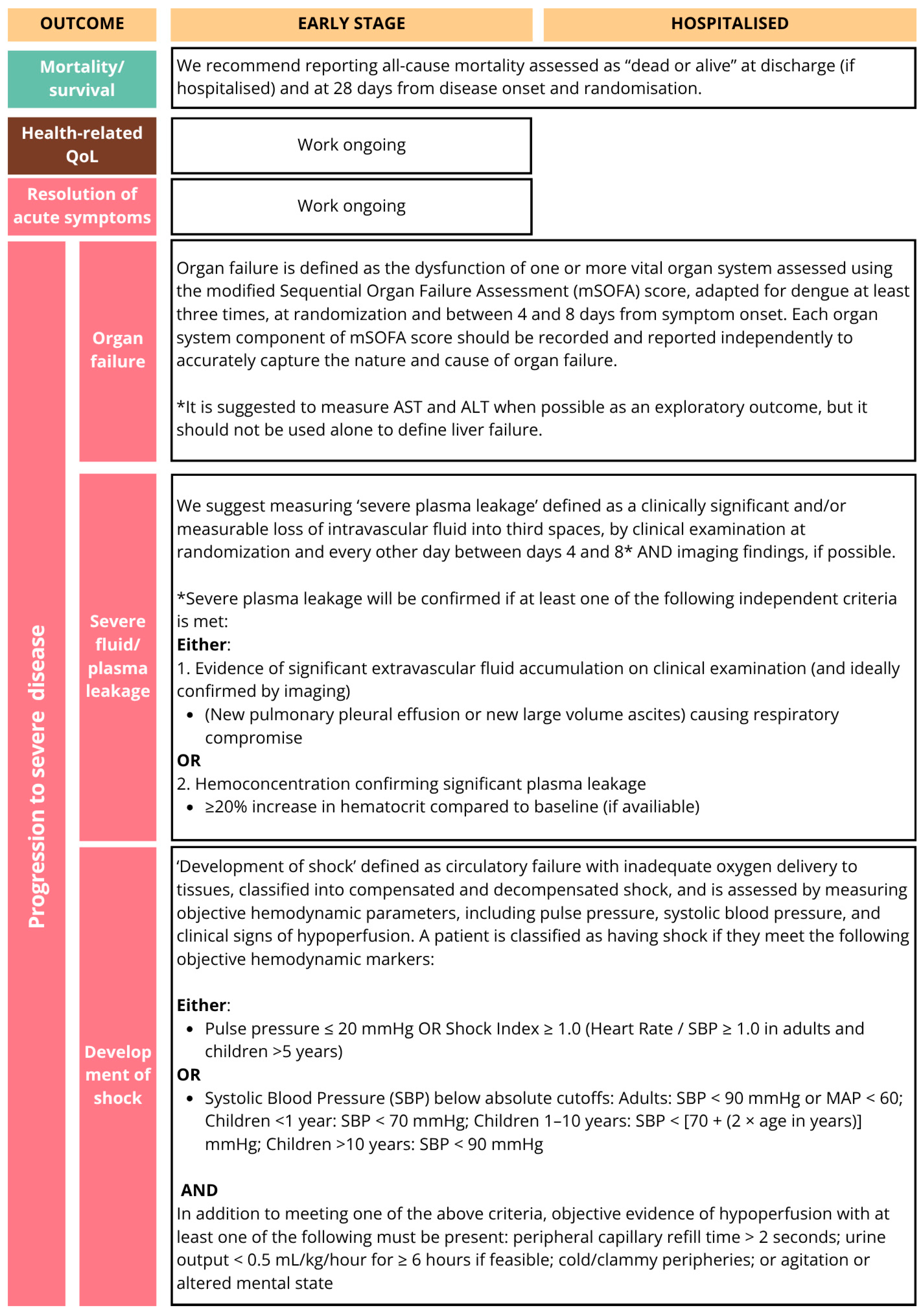
| Progression to severe disease | Organ failure | 100% | 94% | 96% | NA | NA | NA | 83% | Outcomes included in the COS following Delphi survey both for “hospitalised” and “early-stage” dengue. Agreement has been reached on measurement instrument |
| Severe fluid/plasma leakage | 100% | 95% | 96% | NA | NA | NA | 84% | ||
| Development of shock | 94% | 97% | 95% | NA | NA | NA | 92% | ||
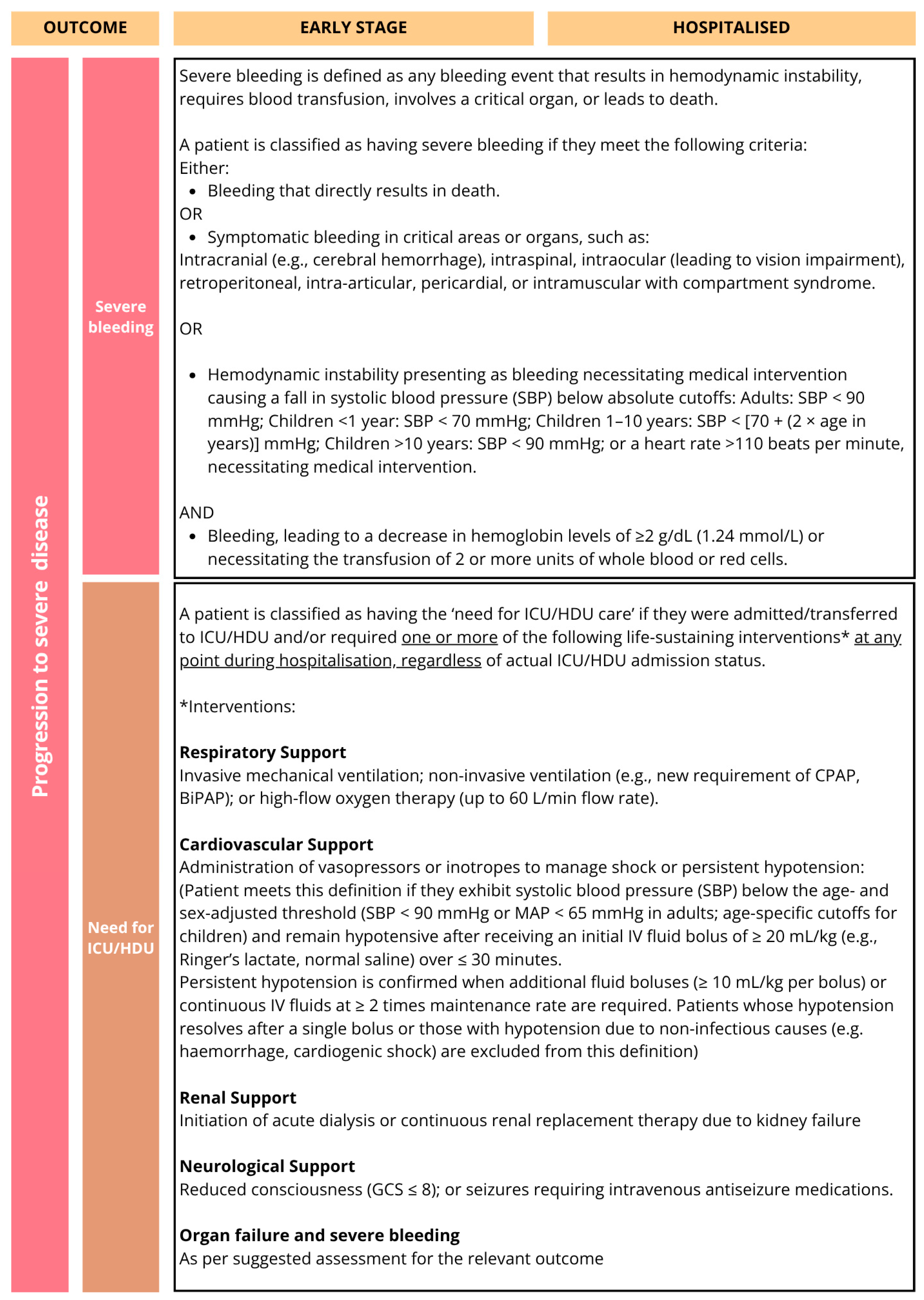
| Severe bleeding | 100% | 92% | 95% | NA | NA | NA | 79% | ||
| Need for ICU/HDU care | 94% | 95% | 92% | NA | NA | NA | 100% | ||
| Need for hospitalisation | NA | NA | NA | NA | NA | 84% | 83% | Outcome “Need for hospitalisation” was added to COS for “early stage” prior to the consensus meeting following core team discussions. Agreement has been reached on measurement instrument | |
| Platelet count | 94% | 73% | 73% | NA | NA | NA | 92% | Outcome included in the COS following Delphi survey both for “hospitalised” and “early-stage” dengue. Agreement has been reached on measurement instrument | |
| Viral load | 71% | 53% | 49% | 33% | 60% | 85% | 93% | Outcome excluded for “hospitalised” COS. Outcome included in the COS for “early-stage” dengue. Agreement has been reached on measurement instrument | |
| COS, core outcome set; HCP, healthcare professionals; OMI, outcome measurement instrument; P, patients; R, researchers. Box 1. Recommendation for ICU dengue clinical trials* We recommend using the agreed-upon DEN-CORE COS in clinical trials involving mixed populations (hospitalised/early-stage and patients requiring ICU/HDU care), where applicable. For dengue clinical trials aiming at assessing efficacy of interventions restricted to patients in HDU/ICU settings only, we suggest using one of the existing ICU-specific COS listed in appendix 1 p 73 Selection of the most appropriate COS should depend on the specific needs of the trial. We strongly encourage use of available critical care COS to ensure consistency across trials. | |||||||||
| Core Outcome | Suitability as Primary Endpoint* | Rationale |
|---|---|---|
| Mortality | High (may be hindered by low prevalence) |
The most objective and robust outcome. Rare in most trial populations; requires large sample sizes. |
| Organ failure | High | Infrequent in general trial populations; better as a safety or severity indicator. |
| Development of shock | High | Clinically meaningful, treatment-sensitive, and recognised as a key severe disease indicator. |
| Health-related quality of life | Moderate (if validated PROMs are available) |
Applicable for phase III-IV trials of early stage, but requires culturally appropriate, validated tools, which are not yet agreed upon. |
| Resolution of acute symptoms | Moderate | Patient-relevant, responsive to treatment, and feasible to measure across phases of disease. Refinement of symptoms to be included, as it is still ongoing |
| Severe bleeding | Moderate | Relevant but less frequent and variable across settings; may require detailed adjudication. |
| Severe plasma leakage | Moderate | Specific endpoint in pathophysiology of dengue; useful in both hospital and research settings. Difficult to measure robustly. |
| Need for ICU/HDU admission | Moderate | Heavily influenced by health system capacity and thresholds for escalation of care. |
| Need for hospitalisation | Moderate | Pragmatic endpoint reflecting clinical worsening; feasible, observable, and patient-centred. |
| Platelet count | Low | Important biological marker but lacks direct clinical relevance as a standalone effectiveness/efficacy outcome. |
| Viral load | Low | More suitable for mechanistic/early-phase studies; does not directly reflect patient benefit. Lack of direct correlation of effect on viral load on clinical benefit currently precludes viral load as a standalone efficacy outcome for regulatory approval. While viral load is currently not considered a valid surrogate of clinical efficacy, regulators recommend to collect viral load data in pivotal clinical trials to potentially establish a correlation of clinical efficacy. |
Phase II: Core Outcome Measurement Set Development
Phase IIa: Instrument Identification
Phase IIb: Instruments Review and Voting

Discussion
Search Strategy and Selection Criteria
Funding
Acknowledgments
Declaration of AI and AI-assisted technologies in the writing process
Contributions
Disclaimer/Publisher’s Note
Declaration of Interests
References
- WHO. Dengue and severe dengue 23 April 2024 [11.06.2025]. Available online: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue.
- Control) EECfDPa. Dengue worldwide overview 2025 [updated 2025-03-03T09:17:53+0100. Available online: https://www.ecdc.europa.eu/en/dengue-monthly.
- Ryan, S.J.; Carlson, C.J.; Mordecai, E.A.; Johnson, L.R. Global expansion and redistribution of Aedes-borne virus transmission risk with climate change. PLOS Neglected Tropical Diseases 2019, 13, e0007213. [Google Scholar] [CrossRef] [PubMed]
- Nakase, T.; Giovanetti, M.; Obolski, U.; Lourenço, J. Population at risk of dengue virus transmission has increased due to coupled climate factors and population growth. Communications Earth & Environment 2024, 5, 475. [Google Scholar] [CrossRef]
- Shepard, D.S.; Undurraga, E.A.; Halasa, Y.A.; Stanaway, J.D. The global economic burden of dengue: a systematic analysis. Lancet Infect Dis. 2016, 16, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Williamson, P.R.; Altman, D.G.; Bagley, H.; Barnes, K.L.; Blazeby, J.M.; Brookes, S.T.; et al. The COMET Handbook: version 1.0. Trials 2017, 18 (Suppl. 3), 280. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, J.J.; Clarke, M.; Williamson, P.R. A methodological approach for assessing the uptake of core outcome sets using ClinicalTrials.gov: findings from a review of randomised controlled trials of rheumatoid arthritis. BMJ 2017, 357, j2262. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, J.J.; Davis, K.; Altman, D.G.; Blazeby, J.M.; Clarke, M.; Tunis, S.; et al. Core Outcome Set-STAndards for Development: The COS-STAD recommendations. PLoS Med. 2017, 14, e1002447. [Google Scholar] [CrossRef] [PubMed]
- Dodd, S.; Clarke, M.; Becker, L.; Mavergames, C.; Fish, R.; Williamson, P.R. A taxonomy has been developed for outcomes in medical research to help improve knowledge discovery. J Clin Epidemiol. 2018, 96, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control: New Edition. WHO Guidelines Approved by the Guidelines Review Committee. Geneva 2009.
- Tomashek, K.M.; Wills, B.; See Lum, L.C.; Thomas, L.; Durbin, A.; Leo, Y.S.; et al. Development of standard clinical endpoints for use in dengue interventional trials. PLoS Negl Trop Dis. 2018, 12, e0006497. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Zambrano, M.; Torres-Hernandez, D.; Murillo-Ortiz, M.A.; Hurtado, I.C.; Davalos, D.M.; Cantor, E.; et al. Different Clinical Severity and Outcomes in a Cohort of Patients With Dengue With Warning Signs in an Endemic Latin American City. Open Forum Infect Dis. 2025, 12, ofaf227. [Google Scholar] [CrossRef] [PubMed]
- The Lancet P. A good enough measure. The Lancet Psychiatry 2020, 7, 825. [CrossRef] [PubMed]
- Krause, K.R.; Chung, S.; Adewuya, A.O.; Albano, A.M.; Babins-Wagner, R.; Birkinshaw, L.; et al. International consensus on a standard set of outcome measures for child and youth anxiety, depression, obsessive-compulsive disorder, and post-traumatic stress disorder. The Lancet Psychiatry 2021, 8, 76–86. [Google Scholar] [CrossRef] [PubMed]
| ‘Late stage’ | ‘Early stage’ | |||
|---|---|---|---|---|
| Delphi R 1 (n=291) |
Delphi R 2 (n=160) |
Consensus meeting (n=38) | Consensus meeting (n=23) | |
| Stakeholder group n (%) | ||||
| People with lived experience of dengue and family members/carers | 48 | 17 | 5 |
20 |
| Healthcare professionals/researchers with lived experience of dengue | 64 | 37 | ||
| Healthcare professionals/researchers without lived experience of dengue | 162 | 97 | 31 | |
| Representatives of other dengue stakeholders including funding agencies, governmental and non-governmental bodies, industry, regulatory authorities, etc. | 17 | 9 | 2 | 3 |
| Gender | ||||
| Female | 153 (53%) | 78 (49%) | - | - |
| Male | 135 (46%) | 82 (51%) | - | - |
| Non-Binary | - | - | ||
| Prefer not to answer | 3 (1%) | - | - | |
| Unknown† | 38 | 23 | ||
| Country of residence | ||||
| Argentina | 1 (0.3%) | 1 (0.6%) | - | - |
| Australia | 2 (0.7%) | 1 (0.6%) | 1 (2,6%) | - |
| Bangladesh | 2 (0.7%) | 1 (0.6%) | - | |
| Bolivia | 3 (1.0%) | 2 (1.3%) | 2 (5,3%) | - |
| Brazil | 14 (4.8%) | 10 (6.3%) | 1 (2,6%) | 2 (9%) |
| Cambodia | 1 (0.3%) | 1 (0.6%) | - | - |
| Cameroon | 1 (0.3%) | - | - | |
| China | 1 (0.3%) | 1 (0.6%) | - | 1 (4%) |
| Colombia | 40 (14%) | 13 (8.1%) | 4 (10,5%) | 5 (22%) |
| Egypt | 5 (1.7%) | 3 (1.9%) | - | - |
| Gambia | 1 (0.3%) | - | - | - |
| Germany | 1 (0.3%) | - | - | - |
| Ghana | 2 (0.7%) | 1 (0.6%) | - | - |
| Honduras | 3 (1.0%) | 2 (1.3%) | - | - |
| India | 10 (3.4%) | 4 (2.5%) | 1 (2,6%) | - |
| Indonesia | 17 (5.8%) | 12 (7.5%) | 2 (5,3%) | - |
| Japan | 2 (0.7%) | 2 (1.3%) | - | - |
| Jordan | 1 (0.3%) | - | - | - |
| Lao People's Democratic Republic | 2 (0.7%) | - | - | - |
| Malaysia | 21 (7.2%) | 13 (8.1%) | 5 (13%) | 1 (4%) |
| Nepal | 7 (2.4%) | 4 (2.5%) | 1 (2,6%) | 2 (9%) |
| Netherlands | 1 (0.3%) | - | - | - |
| Pakistan | 10 (3.4%) | 5 (3.1%) | 1 (2,6%) | - |
| Peru | 26 (8.9%) | 7 (4.4%) | 1 (2,6%) | - |
| Philippines | 13 (4.5%) | 7 (4.4%) | 1 (2,6%) | - |
| Qatar | 1 (0.3%) | 1 (0.6%) | - | |
| Singapore | 22 (7.6%) | 17 (11%) | 2 (5,3%) | - |
| Sri Lanka | 21 (7.2%) | 15 (9.4%) | 4 (10,5%) | 1 (4%) |
| Sudan | 1 (0.3%) | 1 (0.6%) | ||
| Switzerland | 4 (1.4%) | 4 (2.5%) | 2 (5,3%) | 2 (9%) |
| Syrian Arab Republic | 1 (0.3%) | 1 (0.6%) | - | |
| Thailand | 1 (0.6%) | - | ||
| United Kingdom of Great Britain and Northern Ireland | 8 (2.7%) | 5 (3.1%) | - | 1 (4%) |
| United States of America | 6 (2.1%) | 4 (2.5%) | 4 (10,5%) | 3 (13%) |
| Venezuela | 1 (0.3%) | 1 (0.6%) | ||
| Viet Nam | 35 (12%) | 20 (13%) | 6 (16%) | 5 (22%) |
| Age group | ||||
| 18-29 | 50 (17%) | 17 (11%) | - | - |
| 30-39 | 81 (28%) | 45 (28%) | - | - |
| 40-49 | 80 (27%) | 48 (30%) | - | - |
| 50-59 | 48 (16%) | 30 (19%) | - | - |
| 60-69 | 29 (10.0%) | 18 (11%) | - | - |
| 70-79 | 3 (1.0%) | 2 (1.3%) | - | - |
| Unknown† | 38 | 23 | ||
| †Not all percentages add up to 100% owing to rounding. Some participants did not specify their gender, ancestry, location or age group | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




