Submitted:
26 August 2025
Posted:
27 August 2025
You are already at the latest version
Abstract
Background: Posterior epistaxis is a rare but potentially life-threatening condition that may be misdiagnosed as upper gastrointestinal bleeding due to similar clinical manifestations such as hematemesis. Case Presentation: We report a diagnostically challenging case of a 67-year-old woman presenting with hematemesis, initially suspected as gastrointestinal bleeding. Despite multiple interventions, the source of bleeding remained unclear until contrast-enhanced CT and otorhinolaryngology consultation revealed posterior nasal bleeding. The patient was successfully treated with endoscopic sphenopalatine artery ligation (ESPAL). Conclusion: This case highlights the importance of considering posterior epistaxis in the differential diagnosis of hematemesis and demonstrates the effectiveness of ESPAL in managing life-threatening epistaxis.
Keywords:
1. Introduction
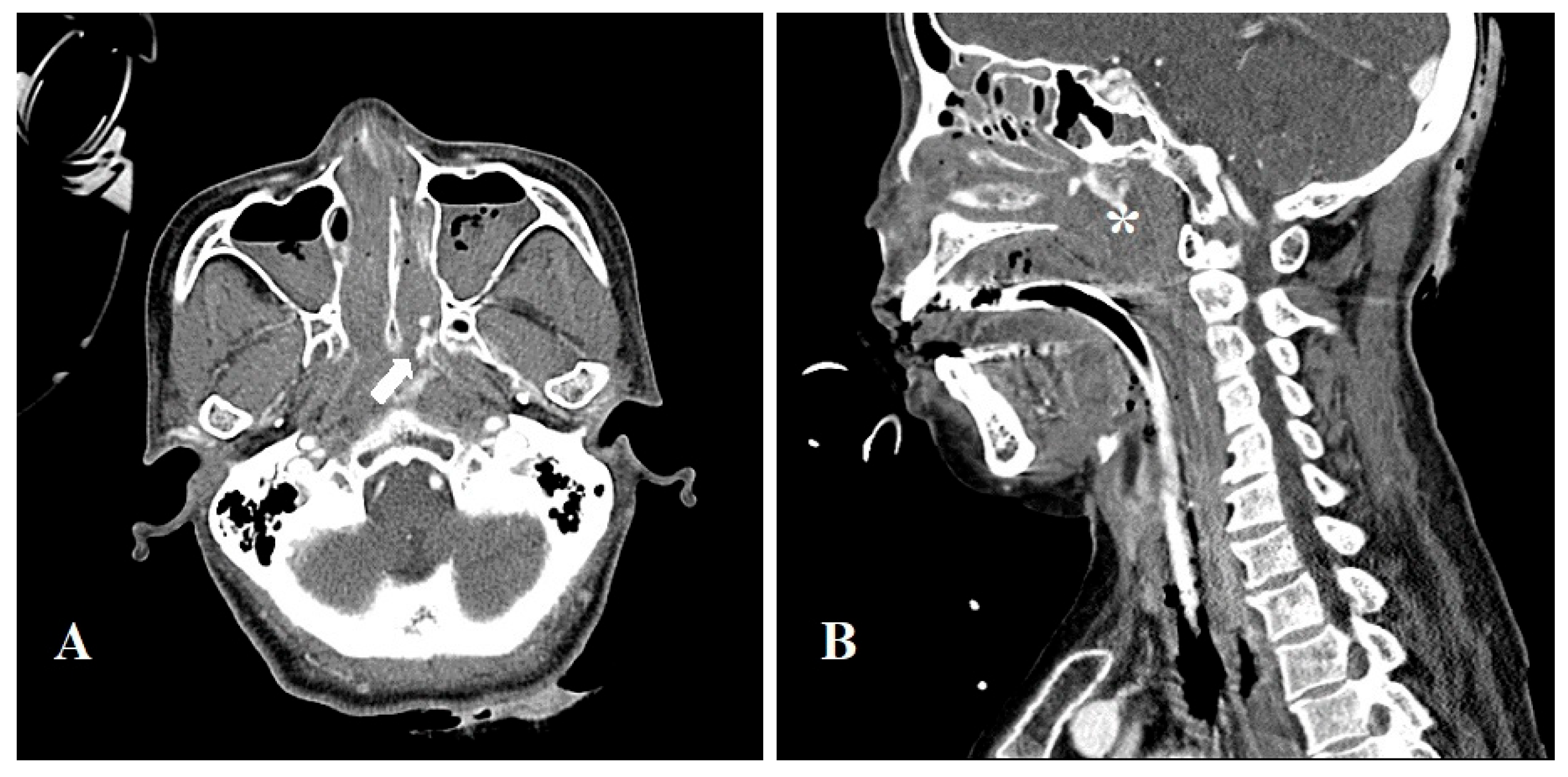
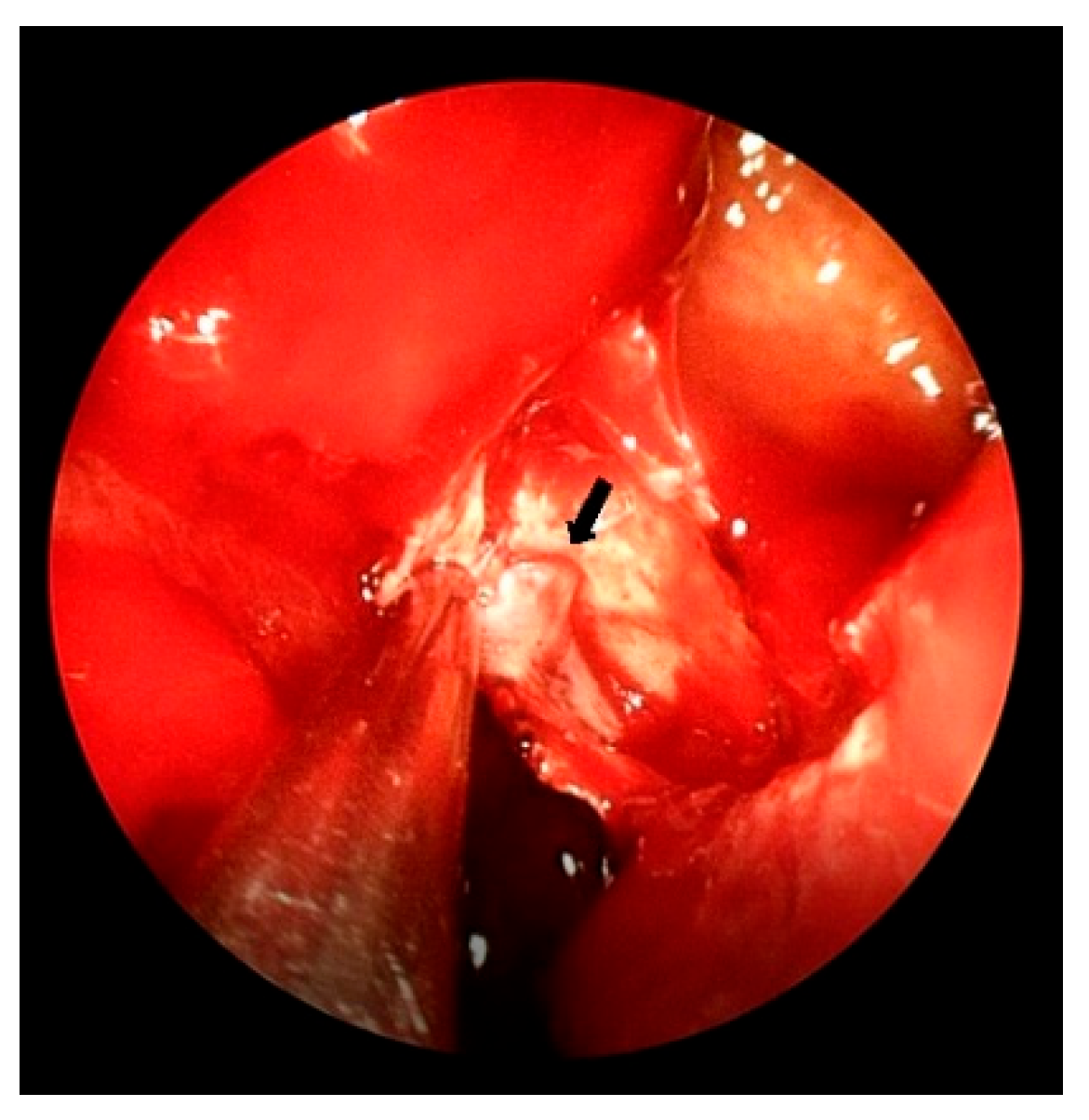
2. Case Presentation
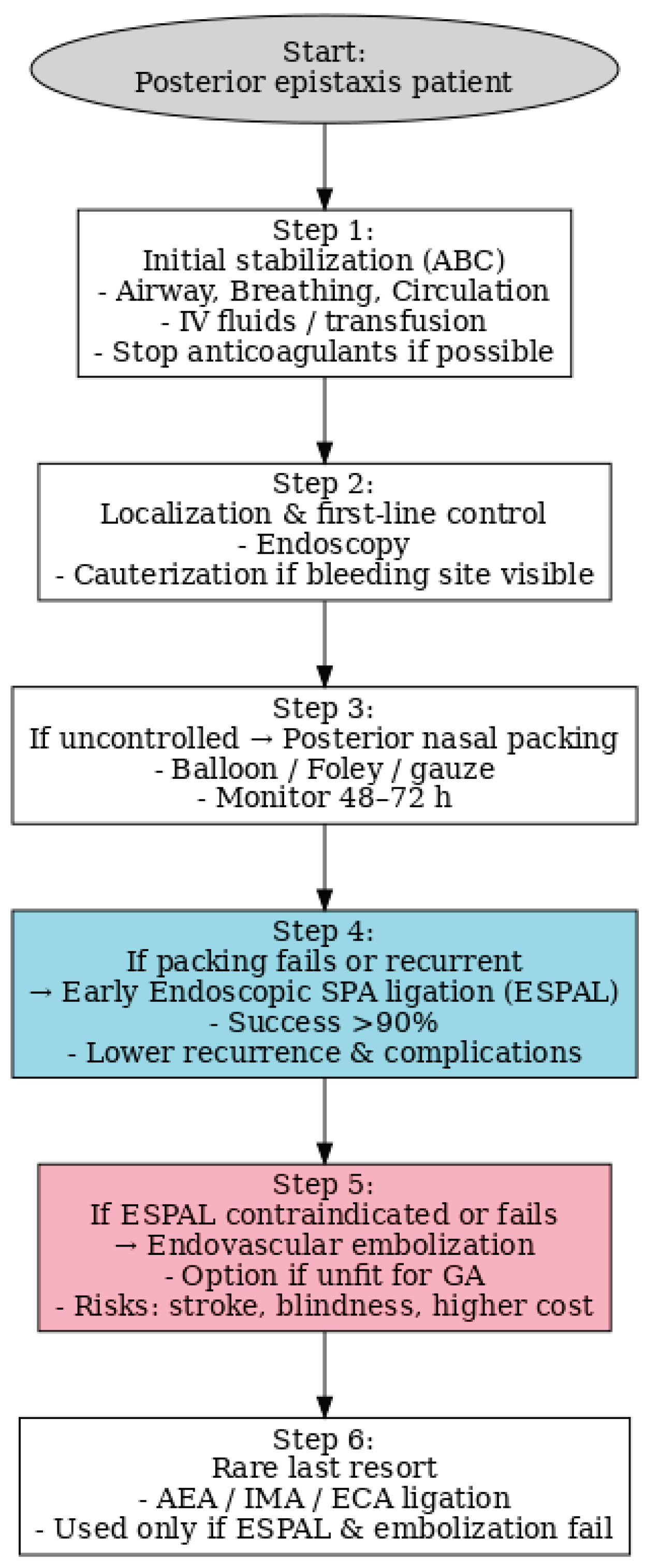
3. Discussion
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rudmik L, Smith TL. Management of intractable spontaneous epistaxis. Am J Rhinol Allergy. 2012;26(1):55-60. [CrossRef]
- Janapala RN, et al. Efficacy of topical tranexamic acid in epistaxis: a systematic review and meta-analysis. Am J Emerg Med. 2022;51:169-175. [CrossRef]
- Gifford, T.O.; Orlandi, R.R. Epistaxis. Otolaryngol Clin North America 2008, 41, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Pallin DJ, et al. Epidemiology of epistaxis in US emergency departments, 1992 to 2001. Ann Emerg Med. 2005;46(1):77-81. [CrossRef]
- Wolf S, et al. Epistaxis mimicking upper gastrointestinal bleeding. J Fam Pract. 1990. 1990.
- Rudmik L, Leung R. Cost-effectiveness analysis of endoscopic sphenopalatine artery ligation vs arterial embolization for intractable epistaxis. JAMA Otolaryngol Head Neck Surg. 2014;140(9):802-808. [CrossRef]
- Nouraei SAR, et al. Outcome of endoscopic sphenopalatine artery occlusion for intractable epistaxis. Laryngoscope. 2007;117(8):1452-1456. [CrossRef]
- Kindler, R.M.; Holzmann, D.; Landis, B.S.; Ditzen, B.; Soyka, M.B. The high rate of long-term recurrences and sequelae after epistaxis treatment. Auris Nasus Larynx 2016, 43, 412–417. [Google Scholar] [CrossRef] [PubMed]
- McDermott, A.M.; O’Cathain, E.; Carey, B.W.; O’Sullivan, P.; Sheahan, P. Sphenopalatine artery ligation for epistaxis: factors influencing outcome and impact of timing of surgery. Otolaryngol Head Neck Surg 2016, 154, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Barnes ML, Spielmann PM, White PS. Epistaxis: a contemporary evidence based approach. Otolaryngol Clin North Am. 2012;45(5):1005-1017. [CrossRef]
- Badran K, et al. Randomized controlled trial comparing Merocel® and RapidRhino® packing in the management of anterior epistaxis. Clin Otolaryngol. 2005;30(4):333-337. [CrossRef]
- Brescia G, et al. A novel nasal endoscopic approach for removing displaced dental implants from the maxillary sinus. Am J Otolaryngol. 2017;38(1):92-95. [CrossRef]
- Koskinas, I.; Timoleon, T.; Christos, G.; Georgios, C. Posterior epistaxis management: review of the literature and proposed guidelines of the hellenic rhinological-facial plastic surgery society. European Archives of Oto-Rhino-Laryngology, 2024, 281.4: 1613-1627.
- Villwock JA, Jones K. Recent trends in epistaxis management in the United States: 2008-2010. JAMA Otolaryngol Head Neck Surg. 2013;139(12):1279-1284. [CrossRef]
- Aref, A.; Bara, M.A.; Amal, M.S.; Anwar, Y.J.; Sami, D.J.; Kareem, I. Posterior epistaxis presenting as upper gastrointestinal bleeding: A case report. Cureus. 2025.
- Hutchison, Stephen M.W. M.B.; Finlayson, Niall D.C.M.B. Epistaxis as a cause of hematemesis and melena. Journal of clinical gastroenterology, 1987, 9.3: 283-285.
- Christensen, N.P.; Smith, D.S.; Barnwell, S.L.; Wax, M. Arterial embolization in the management of posterior epistaxis. Otolaryngol Head Neck Surg 2005, 133, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Chin-Lung, K.; Updates on the Management of Epistaxis. Clinical Medicine and Therapeutics (CMT), 2019,.
- Chaaban, M.R.; Zhang, D.; Resto, V.; Goodwin, J.S. Factors influencing recurrent emergency department visits for epistaxis in the elderly. Auris Nasus Larynx 2018, 45, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Min HJ, et al. Association between hypertension and epistaxis: systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2017;157(6):921-927. [CrossRef]
- Dispenza F, et al. Management of uncontrolled/recurrent epistaxis by ligation or cauterization of the sphenopalatine artery: a scoping review. Eur Arch Otorhinolaryngol. 2024;281(12):6229-6238. [CrossRef]
- Vizzuso, A.; Giraudo, A.; Martucci, M.; Baracchini, C.; Terranova, C.; Vitiello, C.; Cester, G.; Padoan, R.; Palumbo, V.; Masotto, B.; et al. Superselective Unilateral Embolization of the Sphenopalatine Artery for Severe Posterior Epistaxis: A Safe and Effective First-Line Treatment. J. Clin. Med. 2025, 14(14), 4864. [Google Scholar] [CrossRef] [PubMed]
- Budrovich, R.; Saetti, R. Microscopic and endoscopic ligature of the sphenopalatine artery. Laryngoscope 1992, 102, 1391–1394. [Google Scholar] [CrossRef] [PubMed]
- Nouraei, S.A.R.; Maani, T.; Hajioff, D.; Saleh, H.A.; Mackay, I.S. Outcome of endoscopic sphenopalatine artery occlusion for intractable epistaxis: a 10-year experience. Laryngoscope. 2007, 117(8), 1452–1456. [Google Scholar] [CrossRef] [PubMed]
- El N., K.; AbiChedid, J.; Safa, R.; Ballout, F.; Makki, M.; Chami, H.; Nasser, S.; Nader, M.E. Endovascular Embolization for Epistaxis: A Single Center Institutional Experience and Meta-Analysis. J. Clin. Med. 2023, 12(22), 6958. [Google Scholar] [CrossRef]
- Dispenza, F.; Maceri, A.; Martines, F.; Marchese, D.; Galletti, B.; Maniaci, A. Management of Uncontrolled/Recurrent Epistaxis by Ligation or Cauterization of the Sphenopalatine Artery: A Scoping Review. Eur. Arch. Otorhinolaryngol. 2024, 281(12), 6229–6238. [Google Scholar] [CrossRef] [PubMed]
- Traboulsi, H.; Alam, E.; Hadi, U. Changing trends in the management of epistaxis. Int J Otolaryngol 2015, 2015, 263987. [Google Scholar] [CrossRef] [PubMed]
- Chiu TW, McGarry GW. Prospective clinical study of bleeding sites in idiopathic adult posterior epistaxis. Otolaryngol Head Neck Surg. 2007;137(3):390-393. [CrossRef]
- Kucik CJ, Clenney T. Management of Epistaxis. Am Fam Physician. 2005; 71(2):305–311. https://www.aafp.org/pubs/afp/issues/2005/0115/p305.html.
- Soyka, M.B.; Nikolaou, G.; Rufibach, K.; Holzmann, D. On the effectiveness of treatment options in epistaxis: an analysis of 678 interventions. Rhinology 2011, 49, 474–478. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



