Submitted:
27 August 2025
Posted:
27 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
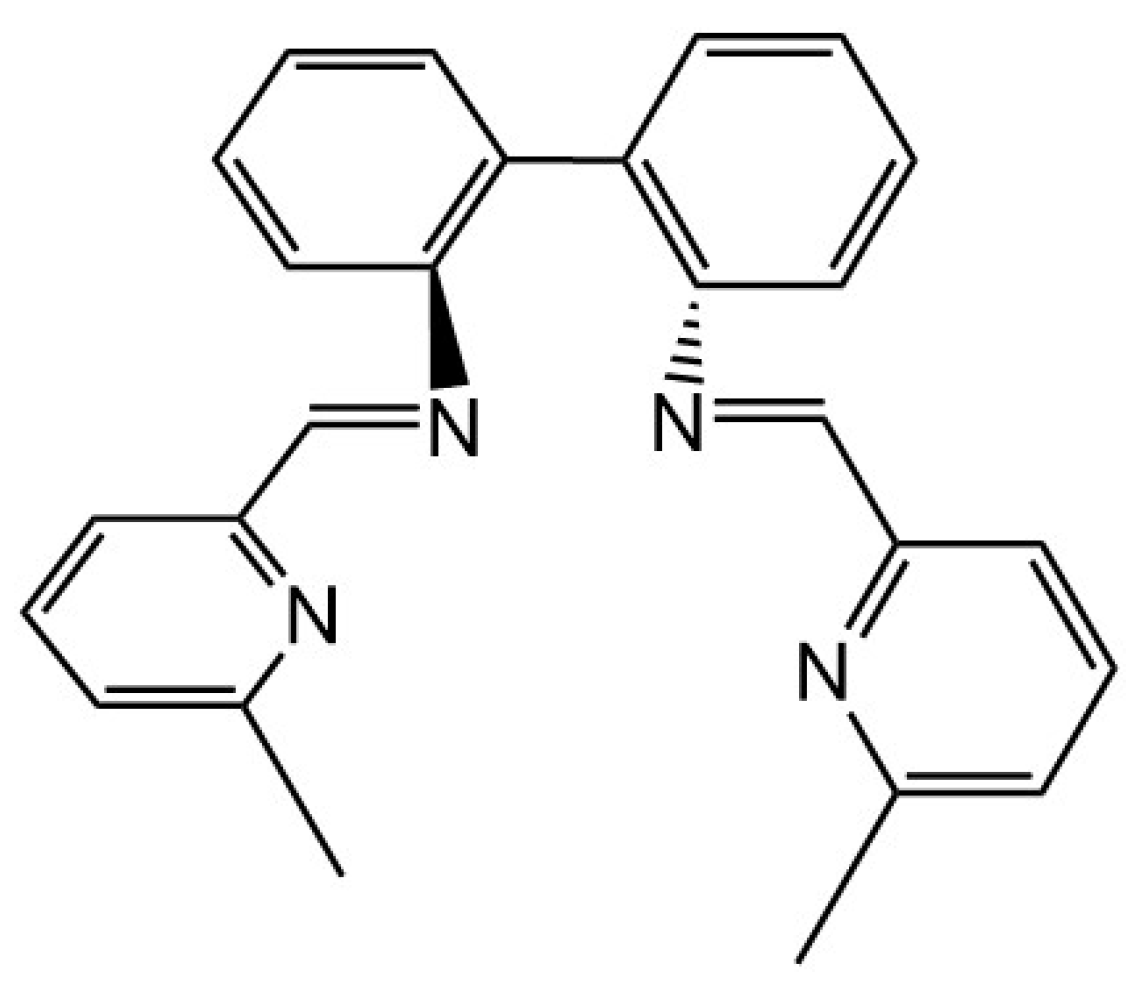
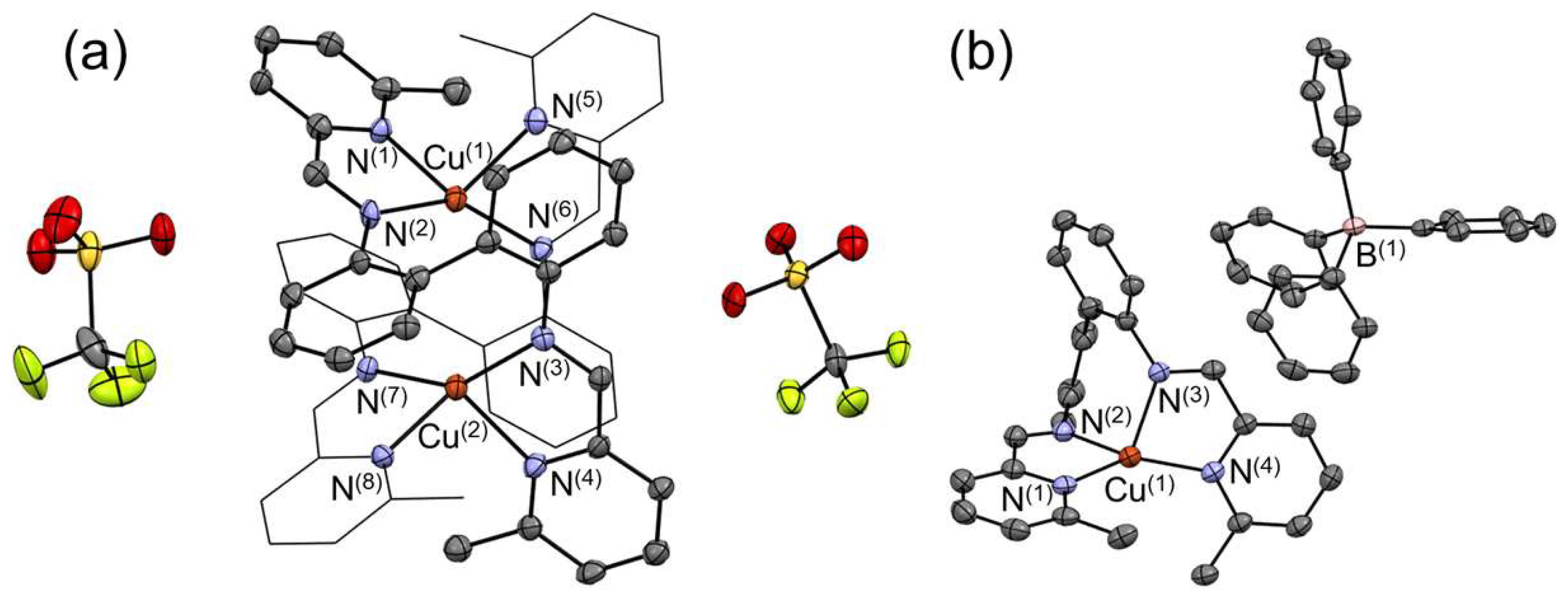
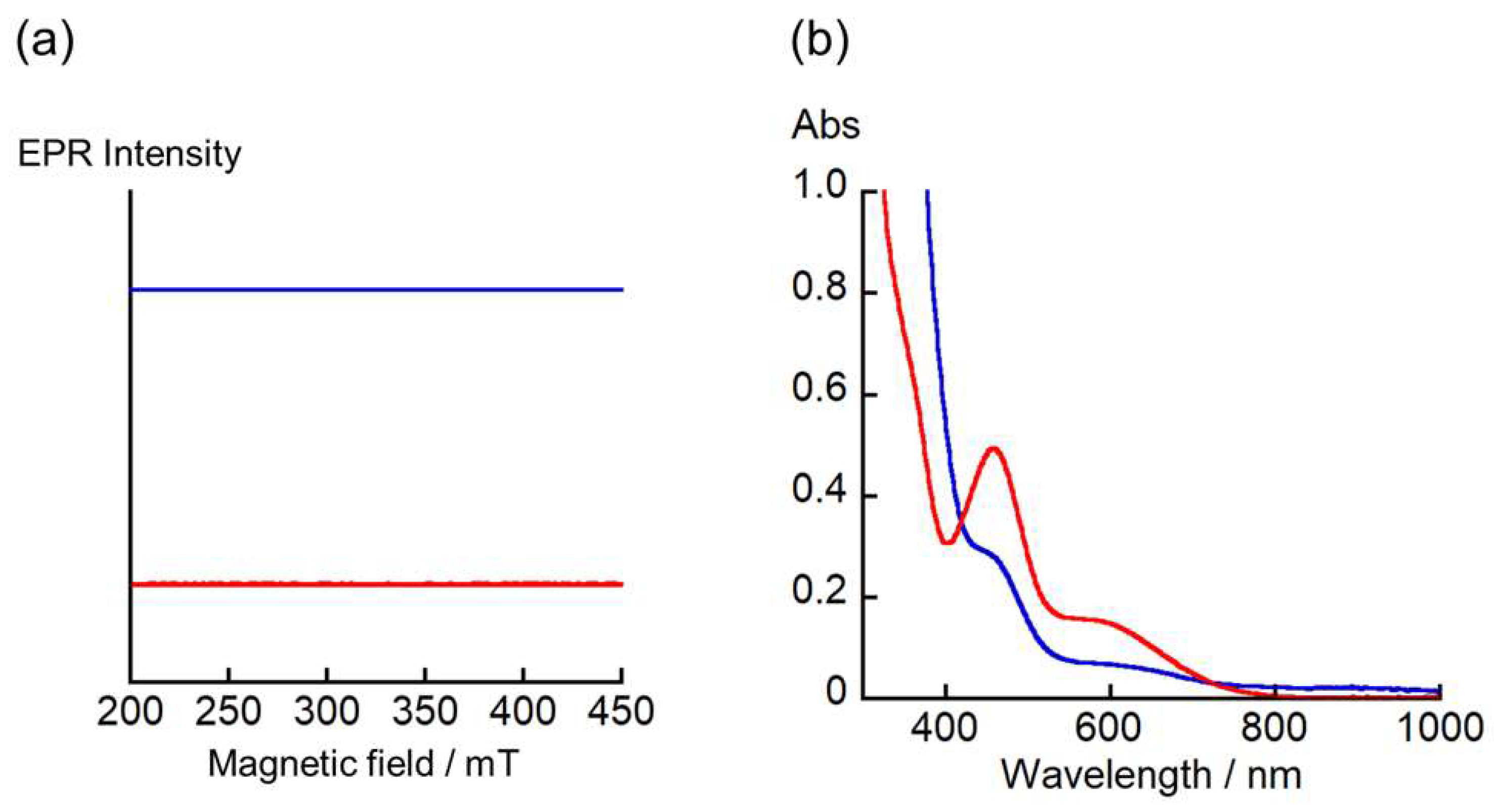
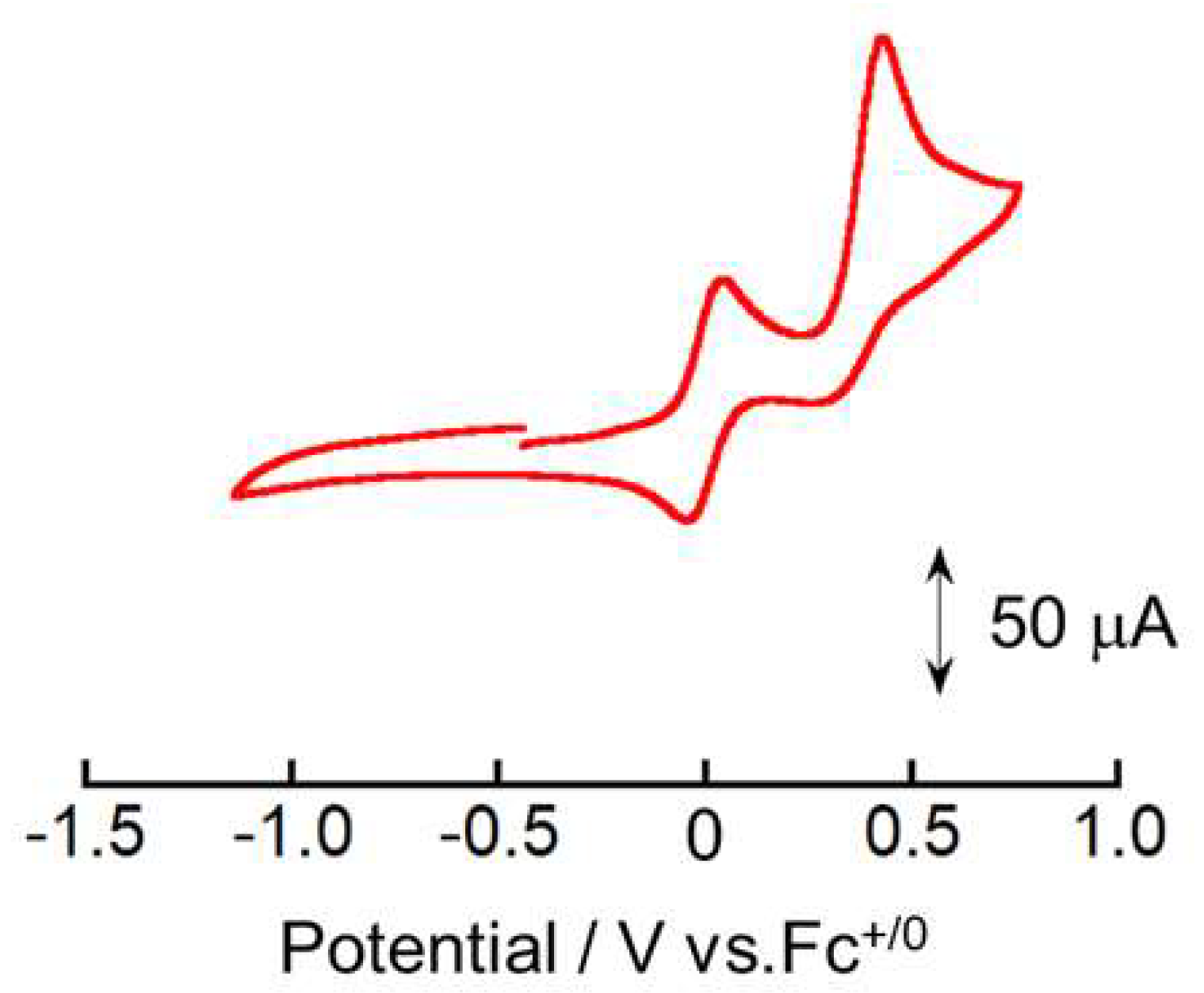
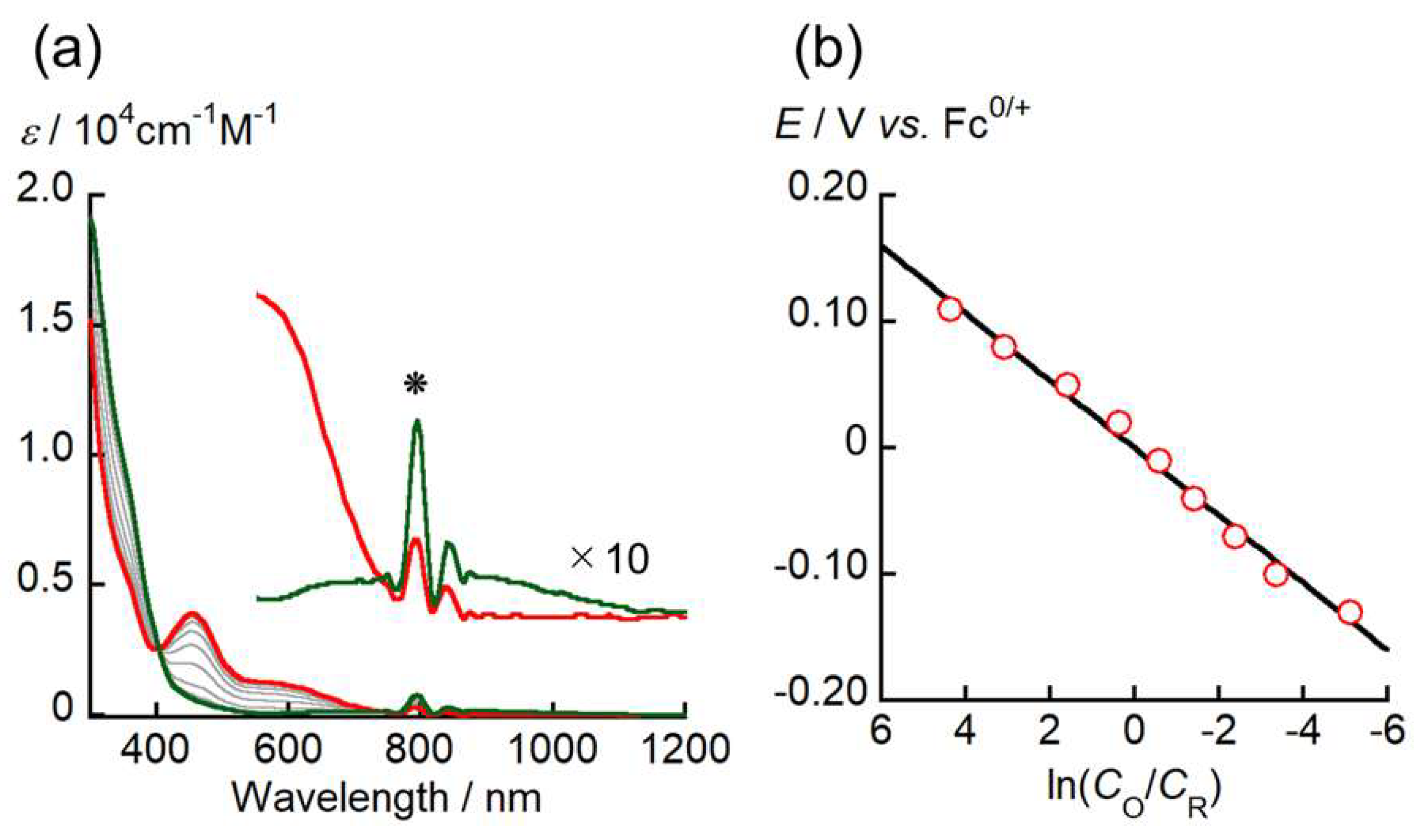
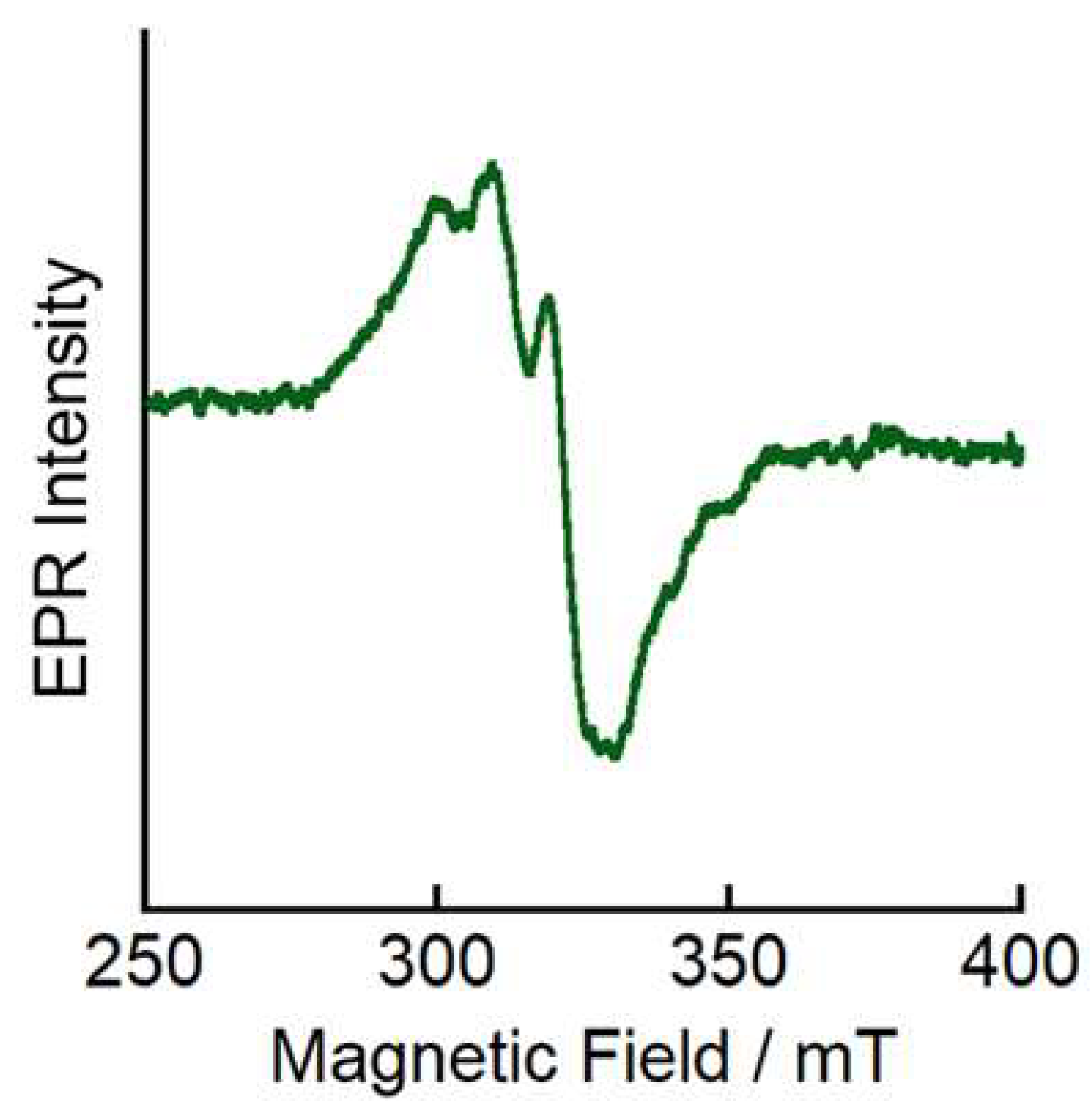
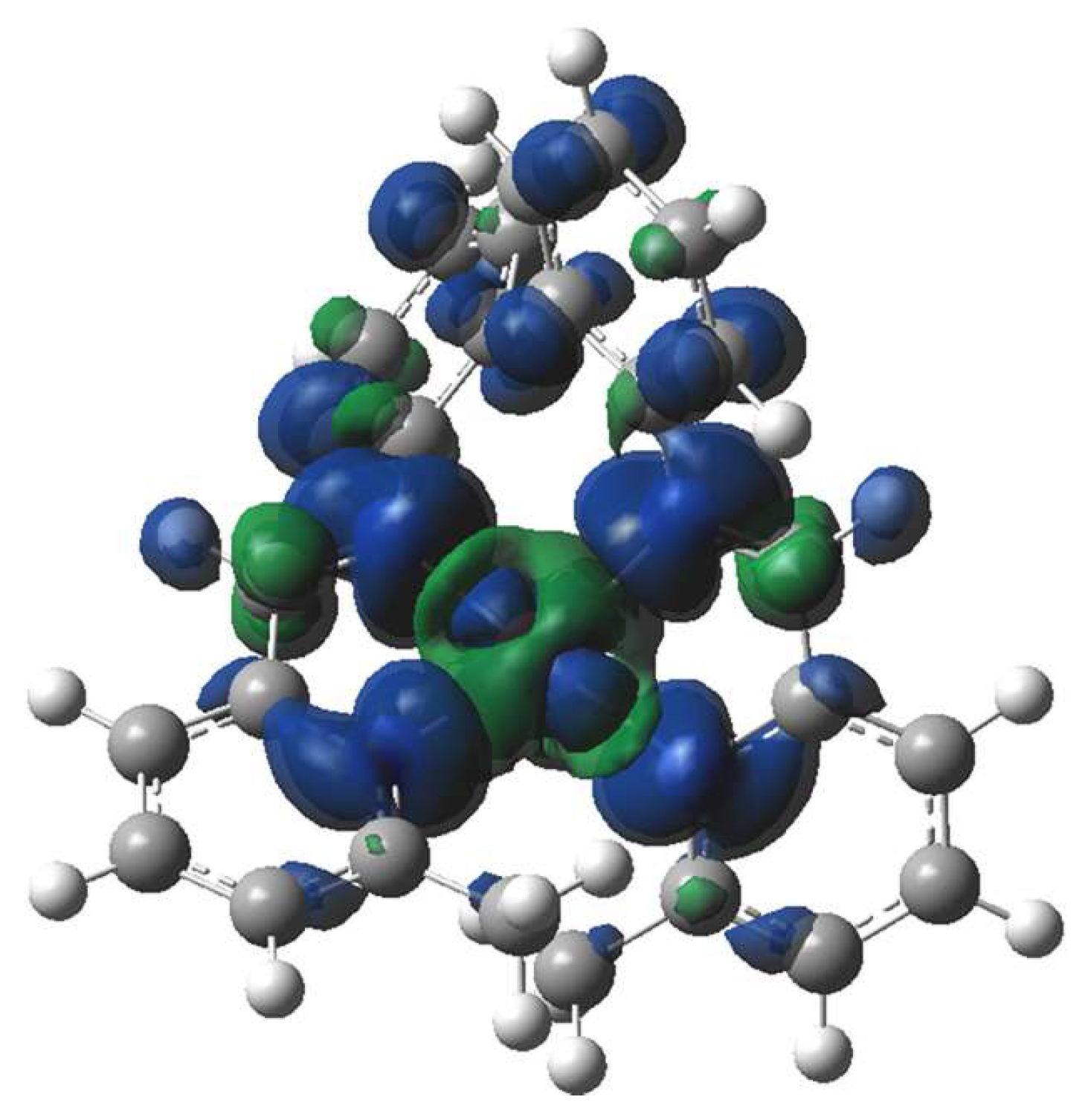
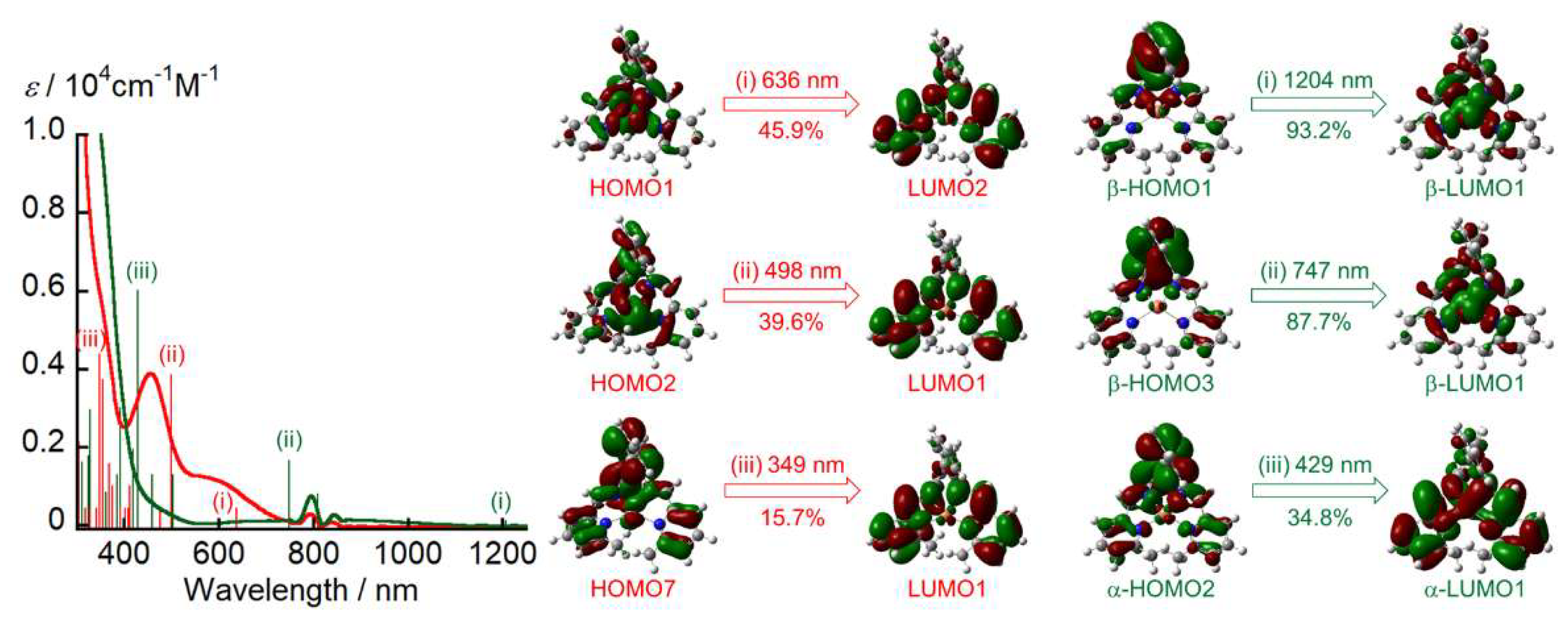
2. Results and Discussion
3. Materials and Methods
Materials
Synthesis of CuI Complexes
Physical Measurements
Crystallographic Analysis
Theoretical Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Solomon, E.I.; Heppner, D.E.; Johnston, E.M.; Ginsbach, J.W.; Cirera, J.; Qayyum, M.; Kieber-Emmons, M.T.; Kjaergaard, C.H.; Hadt, R.G.; Tian, L. Copper active sites in biology. Chem. Rev. 2014, 114, 3659–3853. [Google Scholar] [CrossRef]
- Solomon, E.I.; Szilagyi, R.K.; George, S.D.; Basumallick, L. Electronic structures of metal sites in proteins and models: Contributions to function in blue copper proteins. Chem. Rev. 2004, 104, 419–458. [Google Scholar] [CrossRef]
- Whittaker, W.J. Free radical catalysis by galactose oxidase. Chem. Rev. 2003, 103, 2347–2363. [Google Scholar] [CrossRef]
- Takeyama, T.; Shimazaki, Y. Diversity of oxidation state in copper complexes with phenolate ligands. Dalton Trans. 2024, 53, 3911–3929. [Google Scholar] [CrossRef]
- Kitagawa, S.; Munakata, M.; Higashi, A. Autoreduction of copper(II) complexes of 6,6′-diakyl-2,2′-bipyridine and characterization of their copper(I) complexes. Inorg. Chim. Acta 1984, 84, 79–84. [Google Scholar] [CrossRef]
- Malachowski, M.R.; Adams, M.; Elia, N.; Rheingold, A.L.; Kelly, R.S. Enforcing geometrical constraints on metal complexes using biphenyl-based ligands: Spontaneous reduction of copper(II) by sulfur-containing ligands. J. Chem. Soc. Dalton Trans. 1999, 2177–2182. [Google Scholar] [CrossRef]
- Su, C.Y.; Liao, S.; Wanner, M.; Fiedler, J.; Zhang, C.; Kang, B.S.; Kaim, W. The copper(I)/copper(II) transition in complexes with 8-alkylthioquinoline based multidentate ligands. Dalton Trans. 2003, 189–202. [Google Scholar] [CrossRef]
- Noda, K.; Sasaki, T.; Iwatsuki, S.; Kashiwabara, K.; Suzuki, T.; Takagi, H.D. Syntheses and first structural analyses of Cu(I)–PS complexes with bidentate 1,1-diphenyl-1-phospha-4-thiapentane (mtdpp) and quadridentate 5,9-diphenyl-5,9-diphospha-2,12-dithiatridecane (2,3,2-SPPS): Successful synthetic route for monomeric [Cu(mtdpp)2]BF4 and dimeric [Cu2(2,3,2-SPPS)2](BF4)2. Inorg. Chim. Acta. 2004, 357, 526–532. [Google Scholar] [CrossRef]
- Kumari, S.; Muthuramalingam, S.; Dhara, A.K.; Singh, U.P.; Mayilmurugan, R.; Ghosh, K. Cu(I) complexes obtained via spontaneous reduction of Cu(II) complexes supported by designed bidentate ligands: Bioinspired Cu(I) based catalysts for aromatic hydroxylation. Dalton Trans. 2020, 49, 13829–13839. [Google Scholar] [CrossRef]
- Fedorova, O.A.; Shepel, N.E.; Tokarev, S.D.; Lukovskaya, E.V.; Sotnikova, Y.A.; Moiseeva, A.A.; Aleo, A.D.; Fages, F.; Maurel, F.; Fedorov, Y.V. New J. Chem. 2019, 43, 2817–2827. [CrossRef]
- Morimoto, Y.; Inoue, K.; Itoh, S. Reactivity of copper(I) complexes supported by tripodal nitrogen-containing tetradentate ligands toward gaseous diatomic molecules, NO, CO and O2. Dalton Trans. 2025, 54, 5327–5333. [Google Scholar] [CrossRef]
- Mikata, Y.; Akedo, M.; Hamamoto, E.; Yoshida, S.; Shoji, S.; Ohsedo, Y.; Matsuo, T.; Storr, T.; Funahashi, Y. Structural and electrochemical properties of mononuclear copper(II) complexes with pentadentate ethylenediamine-based ligands with pyridine/quinoline/isoquinoline/quinoxaline binding sites. Dalton Trans. 2024, 53, 16716–16732. [Google Scholar] [CrossRef]
- Mekhail, M.A.; Smith, K.J.; Freire, D.M.; Pota, K.; Nguyen, N.; Burnett, M.E.; Green, K.N. Increased efficiency of a functional SOD mimic achieved with pyridine modification on a pyclen-based copper(II) complex. Inorg. Chem. 2023, 62, 5415–5425. [Google Scholar] [CrossRef] [PubMed]
- Gagne, R.R.; Kreh, R.P.; Dodge, J.A.; Marsh, R.E.; McCool, M. Synthesis and structure of (N,N,N’,N’-tetrakis(2-pyridylmethyl)ethylenediamine)dicopper(I) and its dicarbonyl adduct. Inorg. Chem. 1982, 21, 254–261. [Google Scholar] [CrossRef]
- Siebe, L.; Butenuth, C.; Stammler, A.; Bögge, H.; Walleck, S.; Glaser, T. Generation and reactivity of μ-1,2-peroxo CuIICuII and bis-μ-oxo CuIIICuIII species and catalytic hydroxylation of benzene to phenol with hydrogen peroxide. Inorg. Chem. 2024, 63, 2627–2639. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.X.; Kaderli, S.; Costas, M.; Kim, E.; Neuhold, Y.-M.; Karlin, K.D.; Zuberbühler, A.D. Copper(I)–dioxygen reactivity of [(L)CuI]+ (L = tris(2-pyridylmethyl)amine): Kinetic/thermodynamic and spectroscopic studies concerning the formation of Cu–O2 and Cu2–O2 adducts as a function of solvent medium and 4-pyridyl ligand substituent variations. Inorg. Chem. 2003, 42, 1807–1824. [Google Scholar] [CrossRef]
- Tapia, M.; Pahari, S.K.; Das, S.; Khan, F.S.T.; Hematian, S. Dimerization in TMPA-based copper(i) complexes: Implications for redox kinetics and thermodynamics. Inorg. Chem. 2025, 64, 12416–12422. [Google Scholar] [CrossRef]
- Debnath, S.; Laxmi, S.; McCubbin Stepanic, O.; Quek, S.Y.; van Gastel, M.; DeBeer, S.; Krämer, T.; England, J. A four-coordinate end-on superoxocopper(ii) complex: Probing the link between coordination number and reactivity. J. Am. Chem. Soc. 2024, 146, 23704–23716. [Google Scholar] [CrossRef]
- Langerman, M.; Hetterscheid, D.G.H. Fast oxygen reduction catalyzed by a copper(ii) tris(2-pyridylmethyl)amine complex through a stepwise mechanism. Angew. Chem., Int. Ed. 2019, 58, 12974–12978. [Google Scholar] [CrossRef]
- Wijeratne, G.B.; Hematian, S.; Siegler, M.A.; Karlin, K.D. Copper(I)/NO(g) reductive coupling producing a trans-hyponitrite bridged dicopper(II) complex: Redox reversal giving copper(I)/NO(g) disproportionation. J. Am. Chem. Soc. 2017, 139, 13276–13279. [Google Scholar] [CrossRef]
- Amendola, V.; Fabbrizzi, L.; Gianelli, L.; Maggi, C.; Mangano, C.; Pallavicini, P.; Zema, M. Electrochemical assembling/disassembling of helicates with hysteresis. Inorg. Chem. 2001, 40, 3579–3587. [Google Scholar] [CrossRef]
- Takeyama, T.; Suzuki, T.; Kikuchi, M.; Kobayashi, M.; Oshita, H.; Kawashima, K.; Mori, S.; Abe, H.; Hoshino, N.; Iwatsuki, S.; Shimazaki, Y. Solid state characterization of one- and two-electron oxidized CuII-salen complexes with para-substituents: Geometric structure-magnetic property relationship. Eur. J. Inorg. Chem. 2021, 2021, 4133. [Google Scholar] [CrossRef]
- Kunert, R.; Philouze, C.; Berthiol, F.; Jarjayes, O.; Storr, T.; Thomas, F. ; Distorted copper(II) radicals with sterically hindered salens: Electronic structure and aerobic oxidation of alcohols. Dalton Trans. 2020, 49, 12990–13002. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; DuBois, J.L.; Hedman, B.; Hodgson, K.O.; Stack, T.D.P. Catalytic galactose oxidase models: Biomimetic Cu(II)-phenoxyl-radical reactivity. Science 1998, 279, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Adams, H.; Bailey, N.A.; Dwyer, M.J.S.; Fenton, D.E.; Hellier, P.C.; Hempstead, P.D.; Latour, J.M. Synthesis and crystal structure of a first-generation model for the trinuclear copper site in ascorbate oxidase and of a dinuclear silver precursor. J. Chem. Soc., Dalton Trans. 1993, 1207–1216. [Google Scholar] [CrossRef]
- Kitajima, N.; Fujisawa, K.; Morooka, Y. Tetrahedral copper(II) complexes supported by a hindered pyrazolylborate formation of the thiolato complex, which closely mimics the spectroscopic characteristics of blue copper proteins. J. Am. Chem. Soc. 1990, 112, 3210–3212. [Google Scholar] [CrossRef]
- Shimizu, I.; Morimoto, Y.; Faltermeier, D.; Kerscher, M.; Paria, S.; Abe, T.; Sugimoto, H.; Fujieda, N.; Asano, K.; Suzuki, T.; Comba, P.; Itoh, S. Tetrahedral copper(II) complexes with a labile coordination site supported by a tris-tetramethylguanidinato ligand. Inorg. Chem. 2017, 56, 9634–9645. [Google Scholar] [CrossRef]
- Shima, Y.; Suzuki, T.; Abe, H.; Yajima, T.; Mori, S.; Shimazaki, Y. Non-innocent redox behavior of CuII–p-dimethylaminophenolate complexes: Formation and characterization of the CuI–phenoxyl radical species. Chem. Commun. 2022, 58, 6401–6404. [Google Scholar] [CrossRef]
- Takeyama, T.; Tsushima, S.; Takao, K. Effects of substituents on the molecular structure and redox behavior of uranyl(V/VI) complexes with N3O2-donating Schiff base ligands. Inorg. Chem. 2021, 60, 11435–11449. [Google Scholar] [CrossRef]
- Takeyama, T.; Tsushima, S.; Takao, K. Utility of redox-active ligands for reversible multi-electron transfer in uranyl(VI) complexes. Inorg. Chem. Front. 2023, 10, 4028–4044. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Gaussian 16, Revision C.02, Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; Li, X.; Caricato, M.; Marenich, A.V.; Bloino, J.; Janesko, B.G.; Gomperts, R.; Mennucci, B.; Hratchian, H.P.; Ortiz, J.V.; Izmaylov, A.F.; Sonnenberg, J.L.; Williams-Young, D.; Ding, F.; Lipparini, F.; Egidi, F.; Goings, J.; Peng, B.; Petrone, A.; Henderson, T.; Ranasinghe, D.; Zakrzewski, V.G.; Gao, J.; Rega, N.; Zheng, G.; Liang, W.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Throssell, K.; Montgomery, J.A., Jr.; Peralta, J.E.; Ogliaro, F.; Bearpark, M.J.; Heyd, J.J.; Brothers, E.N.; Kudin, K.N.; Staroverov, V.N.; Keith, T.A.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.P.; Burant, J.C.; Iyengar, S.S.; Tomasi, J.; Cossi, M.; Millam, J.M.; Klene, M.; Adamo, C.; Cammi, R.; Ochterski, J.W.; Martin, R.L.; Morokuma, K.; Farkas, O.; Foresman, J.B.; Fox, D.J.; Gaussian, Inc., Wallingford CT, 2019.
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef]
- Dennington, R.; Keith, T.A.; Millam, J.M. GaussView, Version 6; Semichem Inc: Shawnee Mission, KS, 2016.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



