1. Introduction
Gold-based therapies have been extensively studied for their potential to treat inflammatory and degenerative diseases, especially joint disorders like osteoarthritis and rheumatoid arthritis [
1]. Among these approaches, gold bead implantation (GBI) [
2,
3], gold microparticles (µGold) [
4,
5], and gold nanoparticles (AuNP) [
6] have emerged as prominent modalities, each with distinct properties influencing their clinical use. Both µGold and GBI interventions seek to harness gold’s anti-inflammatory properties and biocompatibility; however, their differences in size, delivery method, and therapeutic efficacy define their clinical applications [
2,
3,
4,
5]. Although AuNP is utilized in diagnostics, imaging, photothermal therapy, radiotherapy, and drug delivery [
6], it has not been reported for use in clinical treatment on its own.
GBI, traditionally used in permanent implantation procedures, has been employed in veterinary and human medicine, particularly in chronic pain management and degenerative joint conditions [
2,
3,
7,
8,
9]. Their localized presence provides long-term therapeutic pain relief and anti-inflammatory effects, but potential concerns remain regarding implant migration, localized fibrosis, and surgical risks associated with implantation [
10,
11]. Traditionally, GBI involves the surgical placement of small gold beads into specific anatomical locations, such as around artificial joints or acupuncture points to provide long-term pain relief and anti-inflammatory effects [
7,
8,
9].
In contrast, µGold offer a minimally invasive alternative, administered via intra-articular injection or targeted delivery systems, potentially reducing procedural risks while maintaining effective immune modulation and anti-inflammatory effects [
1,
4,
5]. µGold therapy involves the intra-articular injection of gold microparticles directly into the affected joint [
4,
5,
12,
13]. This minimally invasive technique aims to reduce inflammation and alleviate pain without the need for surgical implantation. Recent exploratory human studies have shown promising results using intra-articular injections of gold microparticles in patients with knee and hip osteoarthritis that led to significant pain reduction and improved joint function, with no reported adverse effects [
4,
12,
13]. These outcomes suggest that µGold may offer a safer and more effective alternative to traditional GBI.
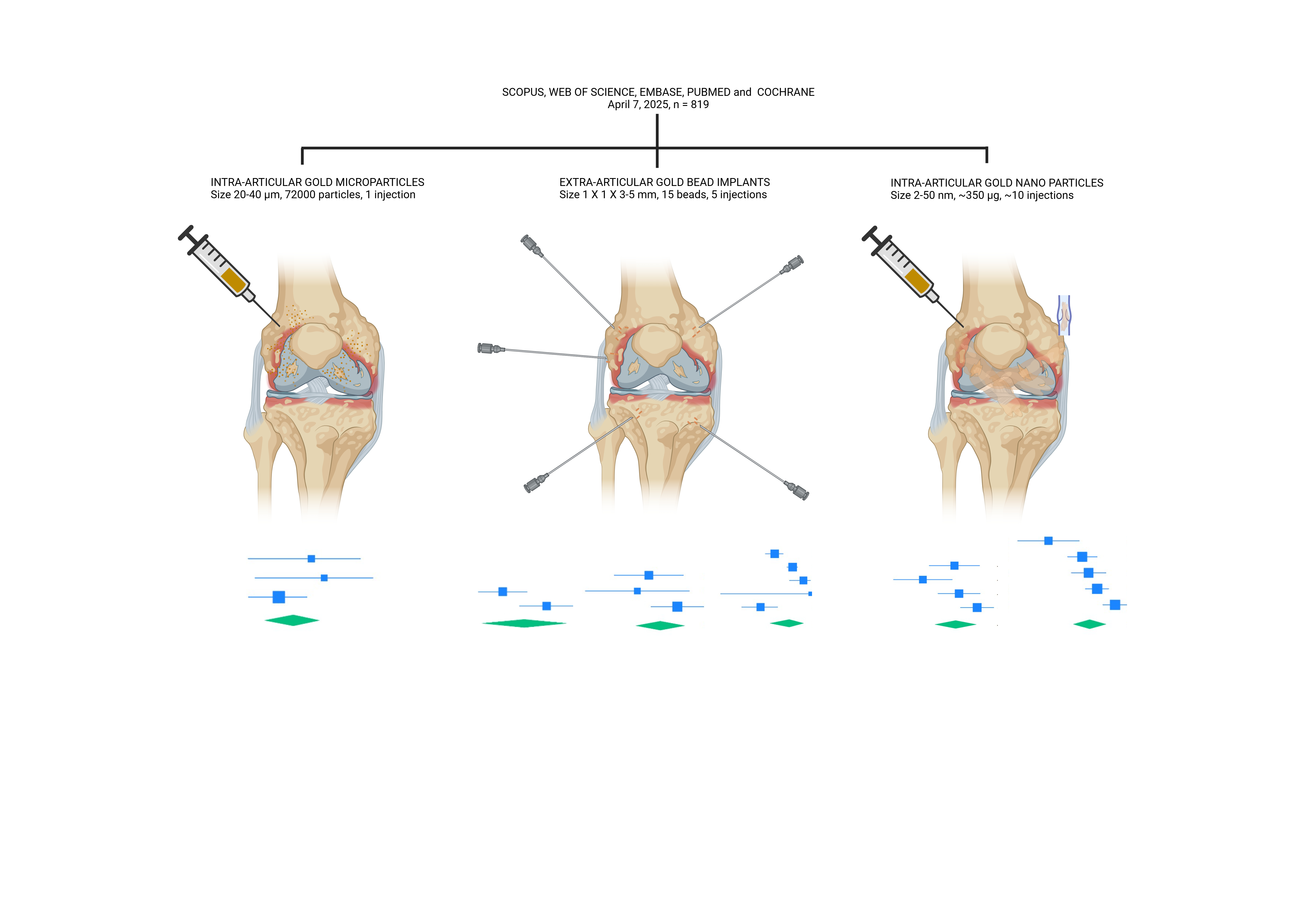
The effectiveness of gold-based local anti-inflammatory treatments is closely linked to the distribution of gold ions within the inflamed tissue [
14,
15]. The finding of a few mm spread of gold ions into the interstitial cellular fluid and cells from the gold surfaces [
14,
15] make it obvious why a multitude of gold particles, 72000 BMI gold particles in 2 ml suspension medium of synovial fluid or hyaluronic acid [
4,
12,
13] offers superior therapeutic outcomes compared to larger implants [
2,
3] or repeated injection of AuNP’s [
6,
16,
17]. The micron-sized particles compose of 99.99% pure gold further expand the available gold surface drastically compared to gold beads and substantially increase the amount of released gold ions. The tiny particles can be injected directly into joints without any side effects [
4,
12,
13]. Another advantage is that a thin 21G needle can be used to apply gold microparticles, making the treatment easier and almost painless compared to using a 14G needle and stylet to inject millimeter-sized gold beads [
2,
3,
7].
This study aims to investigate the research question of whether there is a spatial advantage among µGold, BMI, and AuNP. It is hypothesized that µGold therapy, which involves a minimally invasive single injection, is more effective than GBI and repeated injections of AuNP in treating joint diseases. The rationale behind this hypothesis is that having more gold implant particles present and stable in proximity to the inflamed tissue is expected to enhance the dissolucytotic and immunosuppressive effects, ultimately improving outcomes and reducing side effects.
2. Materials and Methods
2.1. Reporting and Registration
This report follows the PRISMA guidelines [
18]. The completed PRISMA checklist is provided in the
Supplementary Materials (S1). The protocol is registered in PROSPERO.
2.2. Information Sources and Search Strategy
This study identified human and animal studies of local injection of gold microparticles (µGold), gold bead implantation (GBI) and gold nanoparticles (AuNP) for osteoarthritis and other inflammatory joint disorders. Five databases were searched: Medline (PubMed), Embase, Cochrane, Scopus and Web of Science, without restriction on language or date of publication. The categories used were: (“gold microparticles” OR “gold implantation” OR “gold bead implantation” OR “gold nanoparticles”) AND ("osteoarthritis" OR "joint disease" OR "rheumatoid arthritis" OR "joint inflammation" OR "cartilage repair" OR "synovial inflammation" OR "degenerative joint disease") AND ("clinical trial" OR "human study" OR "animal study" OR "preclinical study" OR "in vivo" OR "in vitro") NOT ("gold dental implant" OR "gold stent" OR "gold alloy"). The search was conducted by SR and SK, Aalborg University, Denmark, on April 7, 2025, to investigate the effect of µGold, GBI and AuNP on osteoarthritis and other inflammatory joint disorders. Complete details of the electronic search strategies are provided in
Appendix A.
2.3. Study Selection
Duplicate entries were manually identified and removed by the authors, and the authors independently performed the screening of eligible studies in two steps based on the PRISMA Guidelines [
18]. In the first step, we screened all titles and abstracts according to the predefined inclusion and exclusion criteria. The inclusion criteria consisted of human and animal studies that investigate the effects of µGold, GBI, and AuNP on osteoarthritis and other inflammatory joint diseases, with quantifiable data (N, n1, n2, number improved, ratio, mean, SD, or SEM) required for each group. In cases of conflicts, the authors re-evaluated the title and abstract and reached a mutual consensus after discussion.
In the second step, the two reviewers individually read all full-text articles included in the previous step. In addition to the previously mentioned inclusion criteria, the final inclusion criteria were human and animal studies investigating the effects of µGold, GBI, and AuNP locally injected periarticular or intraarticular, specifically for measuring the number of subjects improved in randomized and cohort studies or the mean and SD of inflammatory markers in randomized studies. Additionally, for each study, the number of injections used is specified. Only studies reporting outcomes after a minimum of 8 weeks were included to ensure that the initial intention to treat, local and general anaesthesia had subsided, allowing for a clearer evaluation of early mid-stage to late-stage treatment outcomes. Reasons for exclusion were pre-specified as follows: 1) no in-vivo experiment, 2) no extractable or available data, 3) wrong route of administration, and 4) not µGold, GBI, or AuNP.
2.4. Quality Assessment
The quality of the included randomised human studies was assessed for bias by the Cochrane Collaboration’s tool for assessing risk of bias [
19], which includes an assessment of bias in: Random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. The authors conducted the quality assessment, and any disagreements that arose were resolved through discussion.
The quality of the included randomized animal studies was assessed by the Collaborative Approach to Meta-Analysis and Review of Animal Data from Experimental Studies (CAMARADES) checklist [
20,
21]. The tool includes 14 questions designed to identify potential biases in the study design. Scores range from 0 to 14, with a higher score reflecting a greater methodological quality of the study [
20,
22].
The quality of the included non-randomized studies was assessed for bias by the Cochrane Collaboration’s tool for assessing risk of bias [
23] version 2 of the ROBINS-I tool, launched on 22 November 2024 [
24], which includes an assessment of bias in seven domains: (1) bias due to confounding (control for confounders (different size of animals included, different joints treated, different causes of osteoarthritis) and control for postintervention variables (time varying, other treatment); (2) bias in classification of interventions (clearly defined intervention groups); (3) bias in selection of participants into the study (or into the analysis) (selection of participants into the study (or into the analysis) based on participant characteristics observed after the start of intervention); (4) bias due to deviations from intended interventions (deviations from the intended intervention beyond what would be expected in usual practice, and adheres to the assigned intervention regimen); (5) bias due to missing data (outcome data available for all, or nearly all, participants, and participants excluded due to missing data on other variables needed for the analysis); (6) bias arising from measurement of the outcome (outcome measure influenced by knowledge of the intervention received, outcome assessors aware of the intervention received by study participants, and systematic errors in measurement of the outcome related to the intervention received); (7) and bias in selection of the reported results (multiple outcome measurements, multiple analyses of the intervention-outcome relationship, and different subgroups). Each bias domain is evaluated as low, moderate, serious, or critical according to the ROBINS-I framework. The quality assessment was conducted by the authors, and any disagreements were resolved by discussion.
2.5. Data Extraction
For each eligible study, the following characteristics were extracted: year of publication, number of participants, type and formulation of gold used (e.g. µGold, GBI, AuNP), target condition (e.g., osteoarthritis or other inflammatory joint diseases), and route of administration (i.e., extra-articular or intra-articular injection). For randomized and cohort studies, we extracted data on the number of subjects who improved. For randomized studies with quantifiable outcomes, we extracted group-level data (N, n1, n2, number improved, proportion, mean, SD or SEM) on inflammatory markers.
2.6. Statistical Analysis
To conduct the analyses, the statistical software Stata MP 18.0 was utilized. We tested for statistical heterogeneity by including Tau-squared (τ²), H-squared (H²), and I-squared (I²). This approach provides a more comprehensive understanding of the variability among the results, allowing us to determine whether differences in study outcomes are likely due to actual differences in study variables rather than random variability. For randomized studies, we calculated the pooled standardized mean odds ratio (OR) with a 95% confidence interval (CI). The effect size measure for continuous data was determined using the inverse-variance method, considering both within-study and between-study variance. For incidence data, the log odds ratio was calculated using the same inverse-variance method, accounting for both types of variances. In the case of cohort studies, we used a new option in Stata MP 18.0 to calculate the Freeman-Tukey transformed proportions, also employing the inverse-variance method [
25,
26]
. The results are presented in forest and tunnel plots.
Generative artificial intelligence (GenAI) has not been used in this paper (e.g., to generate text, data, or graphics, or to assist in study design, data collection, analysis, or interpretation).
4. Discussion
The analysis of the study characteristics revealed that the µGold trials utilized a single injection, whereas the GBI and AuNP trials involved multiple injections. When calculating the total gold surface area that facilitates the diffusion of gold ions into the interstitial cellular fluid and cells from the gold surfaces [
14,
15], the area for this process, known as dissolucytosis [
4,
15,
41], was greater in the µGold studies compared to the BMI studies and the majority of AuNP studies.
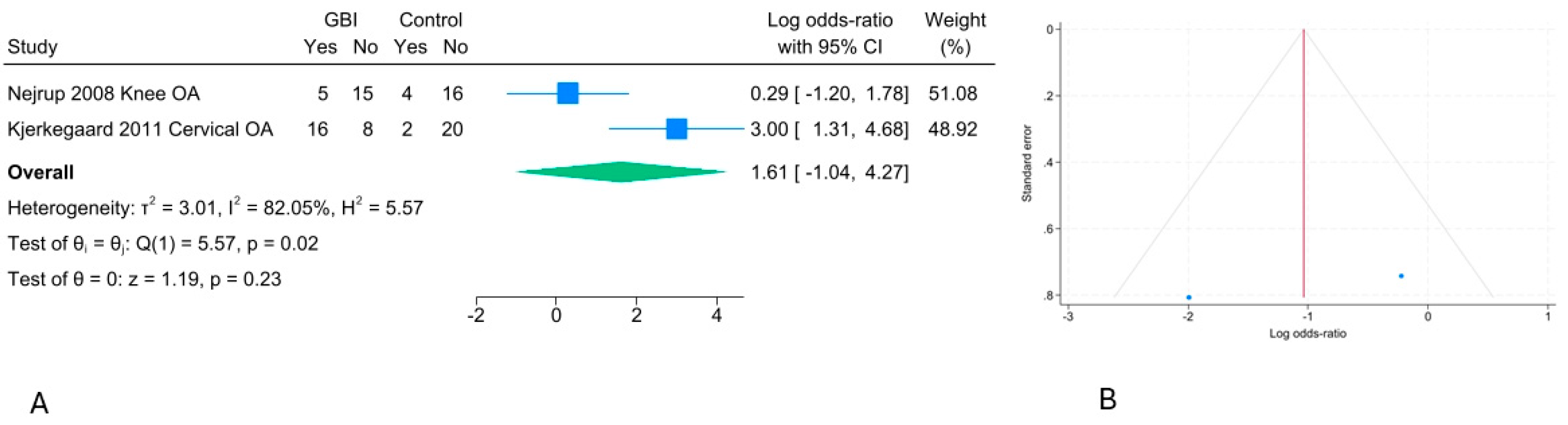
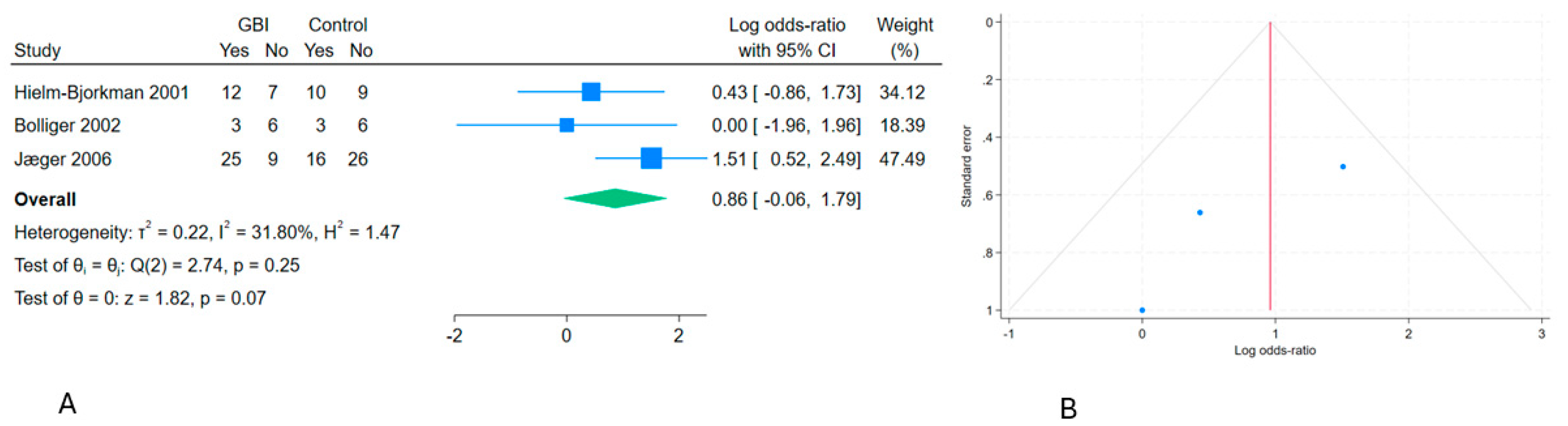
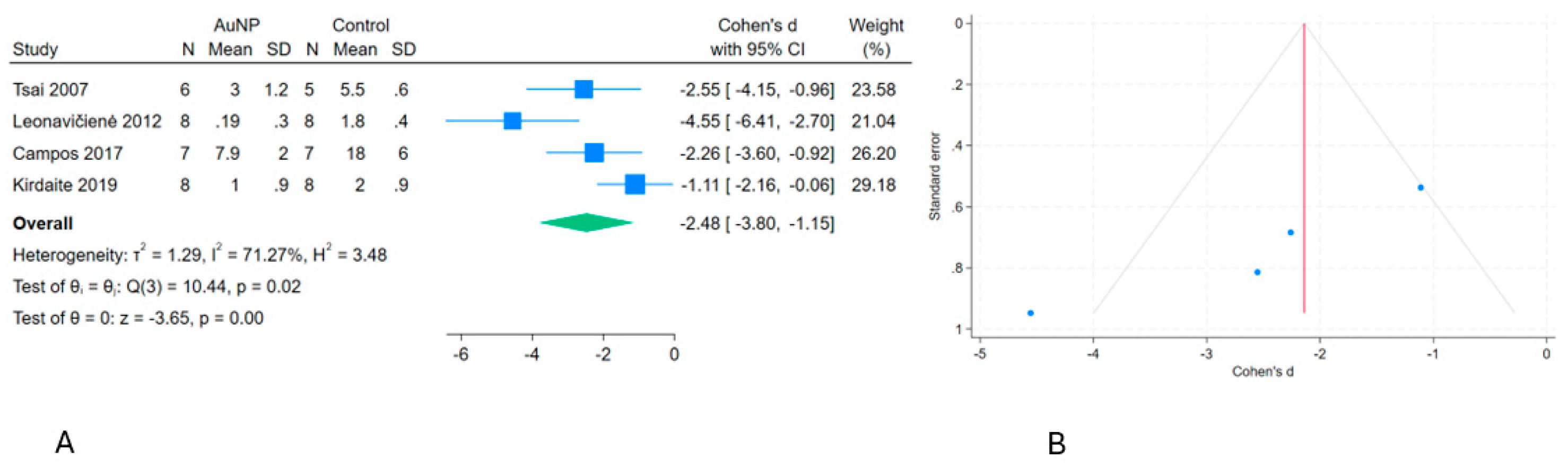
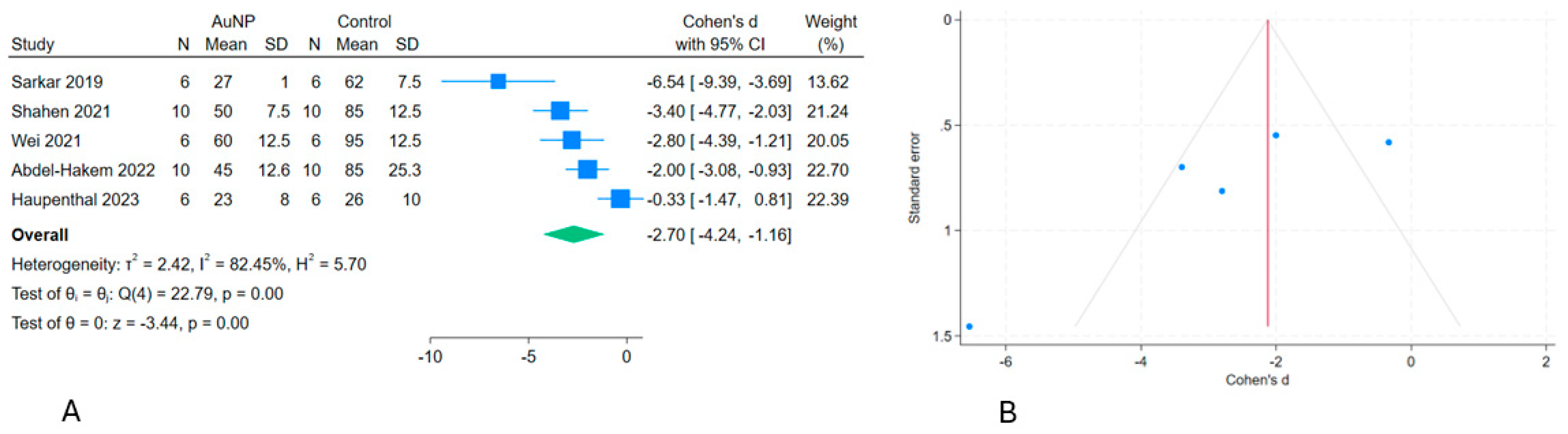
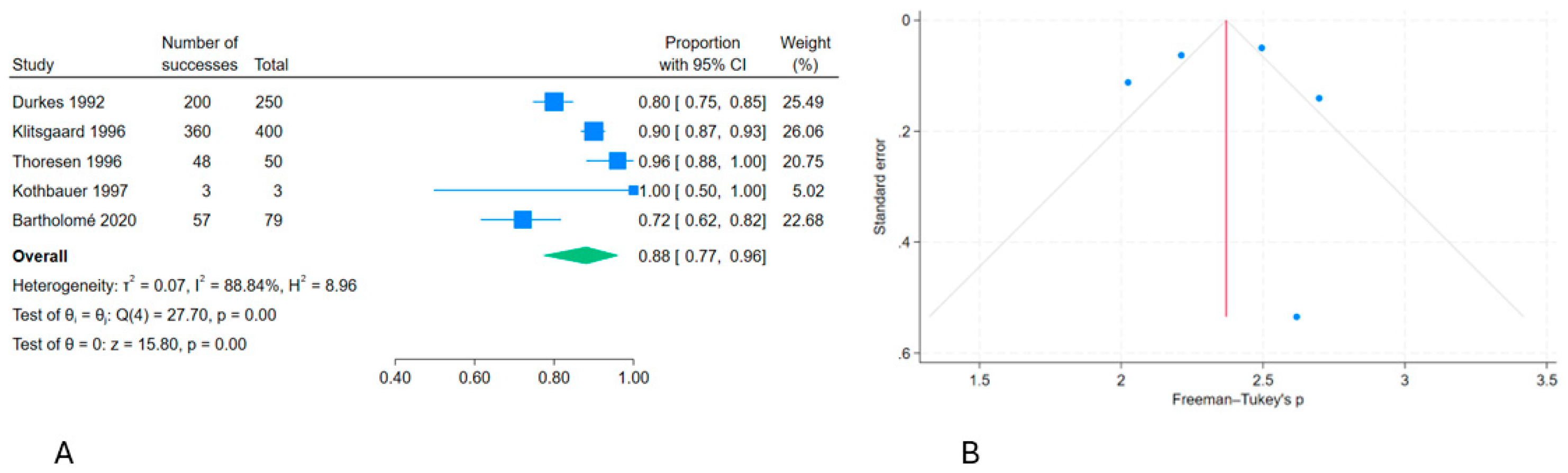
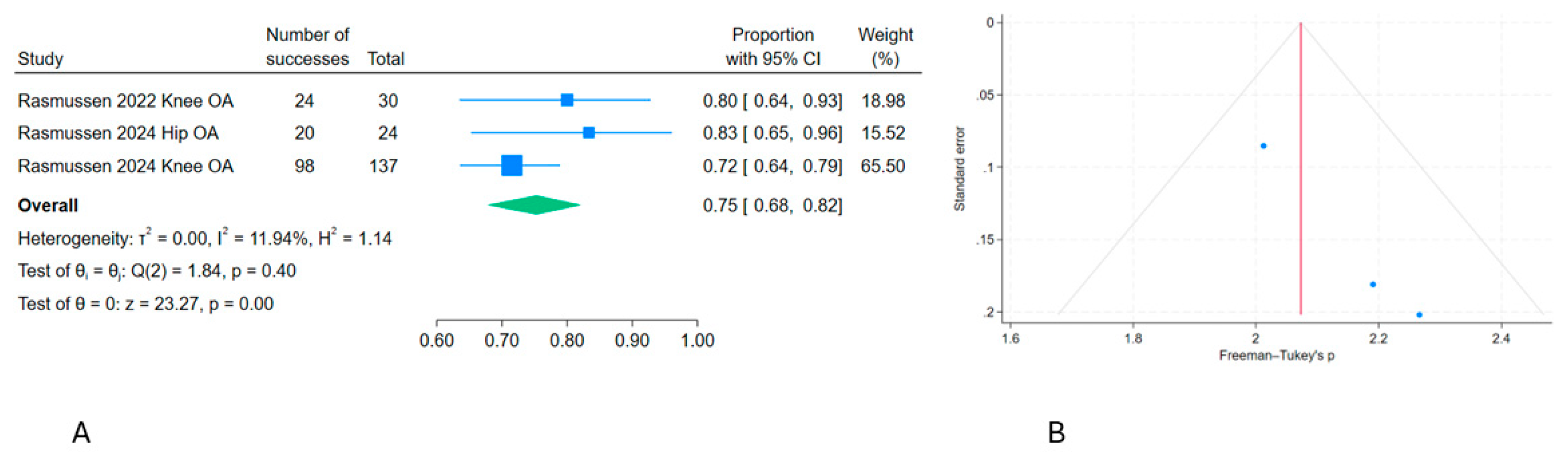
This systematic review is the first to evaluate the effects of extra-articular and intra-articular gold treatments for osteoarthritis (OA) in both human and animal trials. The meta-analyses revealed a significant effect of extra-articular GBI interventions in five animal cohort studies [
35,
36,
37,
38,
39] (
Figure 6). However, no significant effects were found in two human randomized trials and three animal randomized trials when comparing GBI to placebo [
2,
3,
7,
8,
9] (
Figure 2 and
Figure 3). Additionally, the meta-analysis indicated a significant effect of intra-articular gold nanoparticles (AuNP) on OA in nine animal randomized trials compared to placebo [
17,
27,
28,
29,
30,
31,
32,
33,
34] (
Figure 4 and
Figure 5). There was also a significant effect of intra-articular microgold (µGold) on OA in three human cohort studies [
4,
12,
13] (
Figure 7). The forest plots demonstrated a high degree of heterogeneity in four of the analyses (
Figure 2,
Figure 4,
Figure 5 and
Figure 6), while the funnel plots indicated some bias and low study precision. The meta-analysis and the examination of study characteristics support the hypothesis that µGold offers a spatial advantage compared to GBI and AuNP. The findings indicate that a single injection of µGold, its large surface area conducive to dissolucytosis, and the low heterogeneity observed across the µGold studies all bolster this hypothesis. However, some weaknesses include the limited number of studies in each category and the fact that the µGold studies are uncontrolled cohorts.
The use of gold to treat inflammatory diseases like rheumatoid arthritis became significant in the early 20th century and is well documented [
4,
42]. However, the used gold thiocompounds (i.e., sulfur-containing gold-based drugs such as auranofin) must be administered with care as they are toxic to the liver, kidney, and skin, and are replaced by more modern disease-modifying antirheumatic drugs [
43]. Later, it became evident that gold ions alter antigen-processing and reduce the cytokine expression of macrophages [
44]. With the introduction of GBI for treatment of animal OA using acupuncture points for the insertion in the 70-ties [
35,
45] it became interesting to investigate the effect of different gold therapies for OA [
4,
6,
16,
17,
27], based on the limited benefit of the intra-articular injections that is a major part of the treatment program for osteoarthritis, that include exercise, analgesics and dietary weight management [
46,
47]. The intra-articular treatment with corticosteroid and hyaluronic acid has limited benefits for 3-4 weeks [
46,
47], systematic reviews do not support viscosupplementation [
48], and there is no evidence for platelet-rich plasma [
49], stem cells [
50] or micro-fragmented adipose tissue [
51].
The effectiveness of intra-articular AuNP may be reduced by macrophages' ability to remove and scavenge nanoparticles [
28,
41,
52], which can explain the need for multiple injections. Intravenous [
53] and intraperitoneal [
54] injections and oral administration [
55] of AuNP can relieve OA symptoms in animal studies and demonstrate the macrophages’ ability to remove and transport the AuNP to all other tissues [
56], and indicate there is a systemic effect of AuNP. There is an accumulation of injected AuNP in the liver, spleen, and lungs [
56,
57] and granular degeneration is observed in these tissues [
56], which may be a risk. There is an intracellular release of gold ions from the AuNP that initiates the anti-inflammatory and other response [
14,
58]. Even if AuNPs are considered as highly compatible nano constructs, a potential toxicity mainly related to internalization modality has been demonstrated from the release of free gold ions in different cells and tissues [
59].
The macrophages are not able to remove or transport µGold and GBI and the gold microparticles and beads stay in place. Only cells close to the µGold and GBI become loaded with gold ions and imply that no gold is spread to other organs and may exclude any toxic effect [
14,
15,
41].
In the forest plots, we used the odds ratio for binomial outcomes and mean difference for continuous outcomes of controlled studies and Freeman-Tukey’s proportion for binomial outcomes of uncontrolled cohort studies. There is a risk of overemphasis of statistical significance when the confidence interval crosses the null line [
60]. It is crucial to examine individual studies when the pooled estimate indicates no overall effect, as shown in
Figure 2 and
Figure 3. This lack of effect may be due to study limitations, such as small sample sizes or insufficient duration, rather than a genuine absence of effect. Additionally, it is important to consider heterogeneity greater than 50%, as seen in
Figure 2,
Figure 4,
Figure 5 and
Figure 6; in these cases, the overall effect must be interpreted with caution [
61]. If negative or neutral studies are underrepresented, the forest plot may overestimate the treatment effect. In our analysis, four studies found no effect [
2,
7,
9,
34]. We employed a random effects model that assumes the study outcomes may vary.
The funnel plots indicated some bias in the studies and a lack of precision. The studies are to some degree asymmetrically distributed around the overall effect. Additionally, the small number of studies for each analysis may limit our ability to draw firm conclusions.
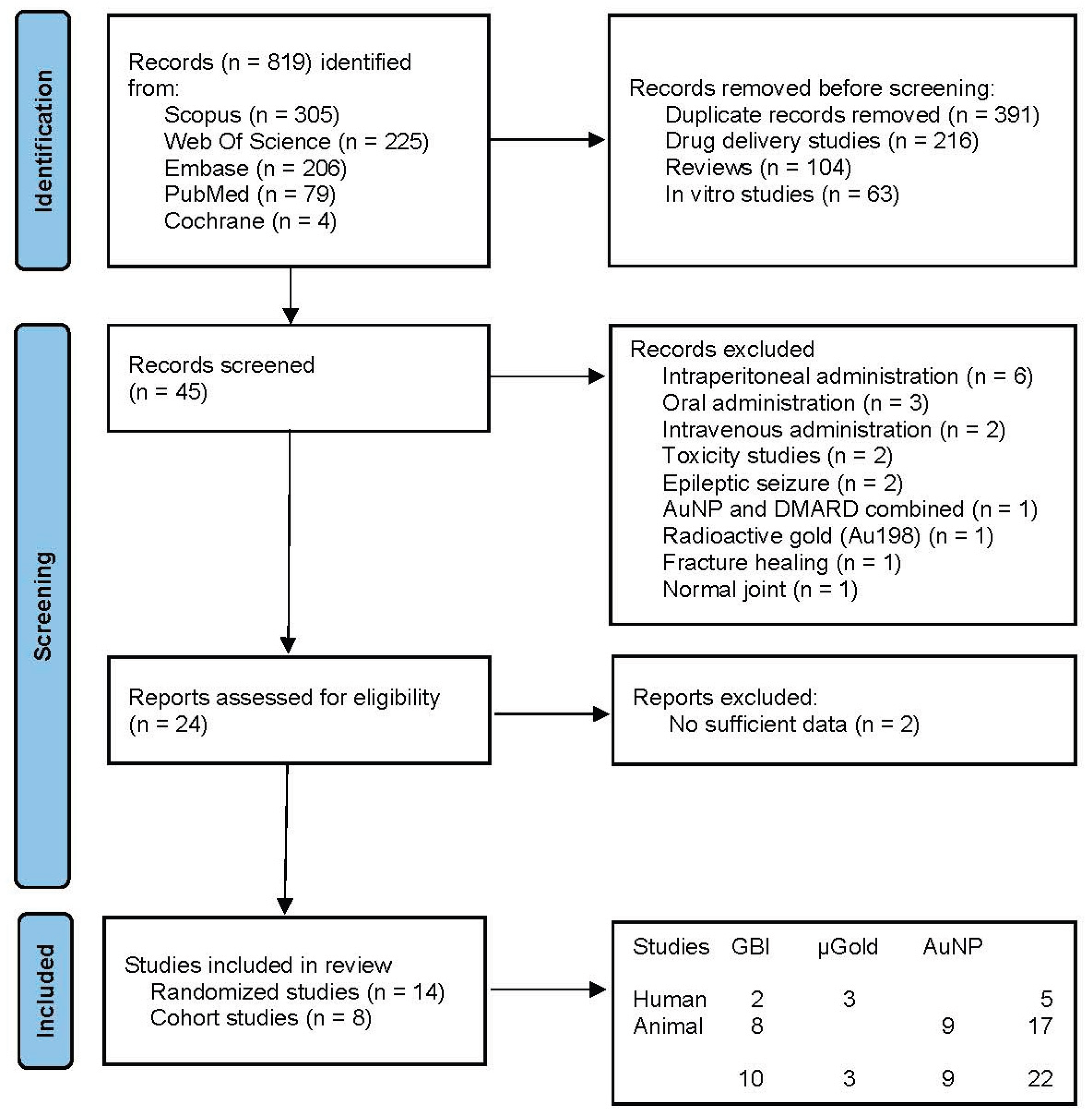
Figure 1.
PRISMA flow diagram.
Figure 1.
PRISMA flow diagram.
Figure 2.
Forest plot (A) and funnel plot (B) of pooled effect for number improved of GBI for human knee and cervical OA.
Figure 2.
Forest plot (A) and funnel plot (B) of pooled effect for number improved of GBI for human knee and cervical OA.
Figure 3.
Forest plot (A) and funnel plot (B) of pooled effect for number improved of GBI for dog hip OA.
Figure 3.
Forest plot (A) and funnel plot (B) of pooled effect for number improved of GBI for dog hip OA.
Figure 4.
Forest plot (A) and funnel plot (B) of pooled effect for osteoarthritic score of AuNP for small animal OA.
Figure 4.
Forest plot (A) and funnel plot (B) of pooled effect for osteoarthritic score of AuNP for small animal OA.
Figure 5.
Forest plot (A) and funnel plot (B) of pooled effect for TNF-Alfa of AuNP for small animal OA.
Figure 5.
Forest plot (A) and funnel plot (B) of pooled effect for TNF-Alfa of AuNP for small animal OA.
Figure 6.
Forest plot (A) and funnel plot (B) of pooled effect for number of successes of GBI for dog OA.
Figure 6.
Forest plot (A) and funnel plot (B) of pooled effect for number of successes of GBI for dog OA.
Figure 7.
Forest plot (A) and funnel plot (B) of pooled effect for number of successes of BMI for human OA.
Figure 7.
Forest plot (A) and funnel plot (B) of pooled effect for number of successes of BMI for human OA.
Table 1.
Study characteristics of two human randomized controlled trials of GBI for knee and cervical osteoarthritis. N = number of individuals; I = intervention group; C = control group; F-u = follow-up. Results in number improved.
Table 1.
Study characteristics of two human randomized controlled trials of GBI for knee and cervical osteoarthritis. N = number of individuals; I = intervention group; C = control group; F-u = follow-up. Results in number improved.
| Study |
N-I |
N-C |
Intervention |
Size/Weight/Area |
Control |
F-u |
Measure |
I |
C |
| Nejrup 2008 [2] |
20 |
20 |
Knee OA; 3 GBI at 5 points |
1x2.5mm/454mg/1.18cm2
|
Needle |
1 y |
Improved |
5 |
4 |
| Kjerkegaard 2011 [3] |
24 |
22 |
Cervical OA; 3 GBI at 2-3 lamina |
1x2.5mm/>364mg/>0.942cm2
|
Needle |
1 y |
Improved |
16 |
2 |
Table 2.
Study characteristics of 12 animal randomized controlled studies evaluating gold bead particles (GBI) and gold nanoparticles (AuNP) for osteoarthritis. N = number of individuals; I = intervention group; C = control group; F-u = follow-up; e.a. = extra articular; i.a. = intra articular; AuNP-P = AuNP conjugated with a protein; n.a. = not available. Results in number improved or in mean and SD.
Table 2.
Study characteristics of 12 animal randomized controlled studies evaluating gold bead particles (GBI) and gold nanoparticles (AuNP) for osteoarthritis. N = number of individuals; I = intervention group; C = control group; F-u = follow-up; e.a. = extra articular; i.a. = intra articular; AuNP-P = AuNP conjugated with a protein; n.a. = not available. Results in number improved or in mean and SD.
| Study |
Diagnosis |
N-I |
N-C |
Intervention |
Size/Weight/Area |
Control |
F-u |
Measure |
I |
C |
| Hielm-Bjorkman 2001 [7] |
Dog hip OA |
19 |
19 |
3 GBI x 3 e.a. |
1x2mm/273mg/0.71cm2
|
Needle |
24w |
Improved |
12 |
10 |
| Bolliger 2002 [9] |
Dog hip OA |
9 |
9 |
3 GBI x ≥ 3 e.a. |
1mm/>91mg/>0.282 cm2
|
Needle |
3 m |
Improved |
3 |
3 |
| Jæger 2006 [8] |
Dog hip OA |
36 |
42 |
3 GBI x 5 e.a. |
1x2mm/455mg/1.78cm2
|
Needle |
6 m |
Improved |
25 |
16 |
| Tsai 2007 [27] |
Rat ankle OA |
6 |
5 |
AuNP x 1, i.a. |
20nm/27µg/6.45cm2
|
Buffer |
9 d |
OA score 0-5 |
3(1.2) |
5.5(.6) |
| Leonavičienė 2012 [28] |
Rat ankle OA |
8 |
8 |
AuNP x 12 i.a. |
50nm/225.6µg/14.01cm2
|
Saline |
28 d |
OA score 0-3 |
.19(.3) |
1.8(.4) |
| Campos 2017 [29] |
Mice knee OA |
7 |
7 |
AuNP-P x 15 i.a. |
n.a. |
Saline |
7 w |
OA score 24 |
7.9(2) |
18.(6) |
| Kirdaite 2019 [30] |
Rat ankle OA |
8 |
8 |
AuNP x 7 i.a. |
50nm/131.6µg/8.18cm2
|
Saline |
10 d |
OA score 0-3 |
1 (.9) |
2 (.9) |
| Sarkar 2019 [17] |
Rat knee OA |
6 |
6 |
AuNP-P x 3 i.a. |
2.2nm/240µg/338.2cm2
|
Saline |
15 d |
TNF alfa |
27 (1) |
62(7.5) |
| Shahen 2021 [31] |
Rat ankle OA |
10 |
10 |
AuNP-P x 1 i.a. |
n.a. |
None |
42 d |
TNF alfa |
50(7.5) |
85(12.5) |
| Wei 2021 [32] |
Rat ankle OA |
6 |
6 |
AuNP-P x 1 i.a. |
11.4nm/500µg/135.9cm2
|
Saline |
n.a. |
TNF alfa |
60(12.5) |
95(12.5) |
| Abdel-Hakem 2022 [33] |
Rat ankle OA |
10 |
10 |
AuNP x1 i.a. |
25nm/6.5µg/0.81cm2
|
None |
35 d |
TNF alfa |
45(12.6) |
85(25.3) |
| Haupenthal 2023 [34] |
Rat knee OA |
6 |
6 |
AuNP x 2 i.a. |
20nm/687.5µg/106.8cm2
|
None |
60 d |
TNF alfa |
23(8) |
26(10) |
Table 3.
Study characteristics of 8 animal and human non-controlled studies of GBI and µGold for osteoarthritis. F-u. = follow-up; e.a. = extra articular; i.a. = intra articular; n.a. = not available.
Table 3.
Study characteristics of 8 animal and human non-controlled studies of GBI and µGold for osteoarthritis. F-u. = follow-up; e.a. = extra articular; i.a. = intra articular; n.a. = not available.
| Study |
Individuals and diagnosis |
Intervention |
Size/Weight/Area |
F-u |
Measure |
Results |
| Durkes 1992 [35] |
250 dogs, hip OA, dysplasia |
3 GBI at 3-16 point e.a. |
1x3mm/364mg/0.879cm2
|
>3 m |
Improved |
200 |
| Klitsgaard 1996 [36] |
400 dogs, hip OA or dysplasia |
3 GBI at 3 points e.a. |
1x2.5mm/341mg/0.848cm2
|
2 m |
Improved |
360 |
| Thoresen 1996 [37] |
50 dogs, hip OA and dysplasia |
1 GBI at 1 point e.a. |
n.a. |
1 m |
Improved |
48 |
| Kothbauer 1997 [38] |
3 dogs, hip OA and dysplasia |
3 GBI at 3 points e.a. |
1x2mm/272mg0.707cm2
|
>1 y |
Improved |
3 |
| Bartholomé 2020 [39] |
79 dogs, hip, elbow, spine OA |
3 GBI at 3-5 points e.a. |
1x3-5mm/728mg/1.70cm2
|
2 m |
Improved |
57 |
| Rasmussen 2022 [4] |
30 patients with knee OA |
µGold 20 mg i.a. |
20-40nm/20mg/2.05cm2
|
2 y |
Improved |
24 |
| Rasmussen 2024 [13] |
24 patients with hip OA |
µGold 20 mg + HA i.a. |
20-40nm/20mg/2.05cm2
|
2 y |
Improved |
20 |
| Rasmussen 2024 [12] |
137 patients with knee OA |
µGold 20 mg + HA i.a. |
20-40nm/20mg/2.05cm2
|
2 y |
Improved |
98 |
Table 4.
Risk of bias in 2 human randomized controlled studies investigating extraarticular gold bead implant (GBI) for osteoarthritis using the Cochrane Collaboration’s tool for assessing risk of bias. Random sequence generation (randomization), allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias.
Table 4.
Risk of bias in 2 human randomized controlled studies investigating extraarticular gold bead implant (GBI) for osteoarthritis using the Cochrane Collaboration’s tool for assessing risk of bias. Random sequence generation (randomization), allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias.
| Id |
Randomization |
Allocation |
Blinding |
Assessment |
Data |
Reporting |
Other |
| Nejrup 2008 [2] |
Low |
Low |
Low |
Low |
Low |
Low |
Low |
| Kjerkegaard 2011 [3] |
Low |
Low |
Low |
Low |
Low |
Low |
Unclear |
Table 5.
Risk of bias in 12 animal RCT’s investigating gold for osteoarthritis, arthritic joint disease or hip dysplasia using the CAMARADES checklist. Studies fulfilling the criteria of: (1) peer reviewed publication; (2) control of temperature; (3) random allocation to treatment or control; (4) allocation concealment; (5) blinded assessment of outcome; (6) use of anesthetic without significant intrinsic neuroprotective activity; (7) animal model (osteoarthritis, arthritis and/or hip dysplasia); (8) sample size calculation; (9) compliance with animal welfare regulations; (10) statement of potential conflict of interests; (11) physiological monitoring; (12) prespecified in- and exclusion criteria; (13) reporting animals excluded from analysis; (14) and reporting of study funding.
Table 5.
Risk of bias in 12 animal RCT’s investigating gold for osteoarthritis, arthritic joint disease or hip dysplasia using the CAMARADES checklist. Studies fulfilling the criteria of: (1) peer reviewed publication; (2) control of temperature; (3) random allocation to treatment or control; (4) allocation concealment; (5) blinded assessment of outcome; (6) use of anesthetic without significant intrinsic neuroprotective activity; (7) animal model (osteoarthritis, arthritis and/or hip dysplasia); (8) sample size calculation; (9) compliance with animal welfare regulations; (10) statement of potential conflict of interests; (11) physiological monitoring; (12) prespecified in- and exclusion criteria; (13) reporting animals excluded from analysis; (14) and reporting of study funding.
| Id |
|
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
12 |
13 |
14 |
Score |
| Hielm-Bjorkman 2001 [7] |
Dog, GBI |
x |
x |
x |
x |
x |
x |
x |
x |
|
|
x |
x |
x |
x |
12 |
| Bolliger 2002 [9] |
Dog, GBI |
x |
|
x |
x |
x |
x |
x |
|
|
|
|
|
x |
|
7 |
| Jæger 2006 [8] |
Dog, GBI |
x |
|
x |
x |
x |
x |
x |
x |
x |
|
x |
x |
x |
x |
12 |
| Tsai 2007 [27] |
Rat, AuNP |
x |
|
|
|
|
|
x |
|
|
|
|
|
|
|
2 |
| Leonavičienė 2012 [28] |
Rat, AuNP |
x |
x |
|
|
x |
|
x |
|
x |
x |
x |
|
|
x |
8 |
| Campos 2017 [29] |
Mice, AuNP |
x |
x |
|
|
x |
|
x |
|
x |
|
x |
|
|
x |
7 |
| Kirdaite 2019 [30] |
Rat, AuNP |
x |
|
x |
x |
|
|
x |
|
x |
x |
|
|
|
|
6 |
| Sarkar 2019 [17] |
Rat, AuNP |
x |
x |
|
|
|
|
x |
|
x |
x |
x |
|
|
x |
7 |
| Shahen 2021 [31] |
Rat, AuNP |
x |
x |
|
|
|
|
x |
|
x |
x |
x |
|
|
|
6 |
| Wei 2021 [32] |
Rat, AuNP |
x |
|
|
|
|
|
|
|
x |
x |
|
|
|
x |
4 |
| Abdel-Hakem 2022 [33] |
Rat, AuNP |
x |
x |
|
|
|
|
x |
|
x |
x |
|
|
|
x |
6 |
| Haupenthal 2023 [34] |
Rat, AuNP |
x |
x |
x |
x |
x |
|
x |
x |
|
x |
x |
|
|
x |
10 |
Table 6.
Risk of bias in 8 non-randomized cohort studies investigating gold for osteoarthritis, arthritic joint disease or hip dysplasia using the ROBINS-I V2 tool. Studies fulfilling the criteria of: (1) bias due to confounding; (2) bias in classification of interventions; (3) bias in selection of participants into the study (or into the analysis); (4) bias due to deviations from intended interventions; (5) bias due to missing data; (6) bias arising from measurement of the outcome; (7) and bias in selection of the reported results. Each bias domain is evaluated as low (L), moderate (M), serious (S) or critical (C).
Table 6.
Risk of bias in 8 non-randomized cohort studies investigating gold for osteoarthritis, arthritic joint disease or hip dysplasia using the ROBINS-I V2 tool. Studies fulfilling the criteria of: (1) bias due to confounding; (2) bias in classification of interventions; (3) bias in selection of participants into the study (or into the analysis); (4) bias due to deviations from intended interventions; (5) bias due to missing data; (6) bias arising from measurement of the outcome; (7) and bias in selection of the reported results. Each bias domain is evaluated as low (L), moderate (M), serious (S) or critical (C).
| Id |
|
1 |
2 |
3 |
4 |
5 |
6 |
7 |
| Durkes 1992 [35] |
Dog, GBI |
S |
L |
C |
L |
C |
C |
C |
| Klitsgaard 1996 [36] |
Dog, GBI |
S |
L |
C |
L |
C |
C |
C |
| Thoresen 1996 [37] |
Dog, GBI |
C |
L |
C |
C |
C |
C |
C |
| Kothbauer 1997 [38] |
Dog, GBI |
C |
L |
C |
C |
C |
C |
C |
| Bartholomé 2020 [39] |
Dog, GBI |
S |
L |
M |
L |
L |
S |
S |
| Rasmussen 2022 [4] |
Human, µGold |
L |
L |
L |
L |
L |
M |
M |
| Rasmussen 2024 [13] |
Human, µGold |
M |
L |
L |
L |
L |
M |
M |
| Rasmussen 2024 [12] |
Human, µGold |
M |
L |
L |
L |
L |
M |
M |