Submitted:
25 August 2025
Posted:
25 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
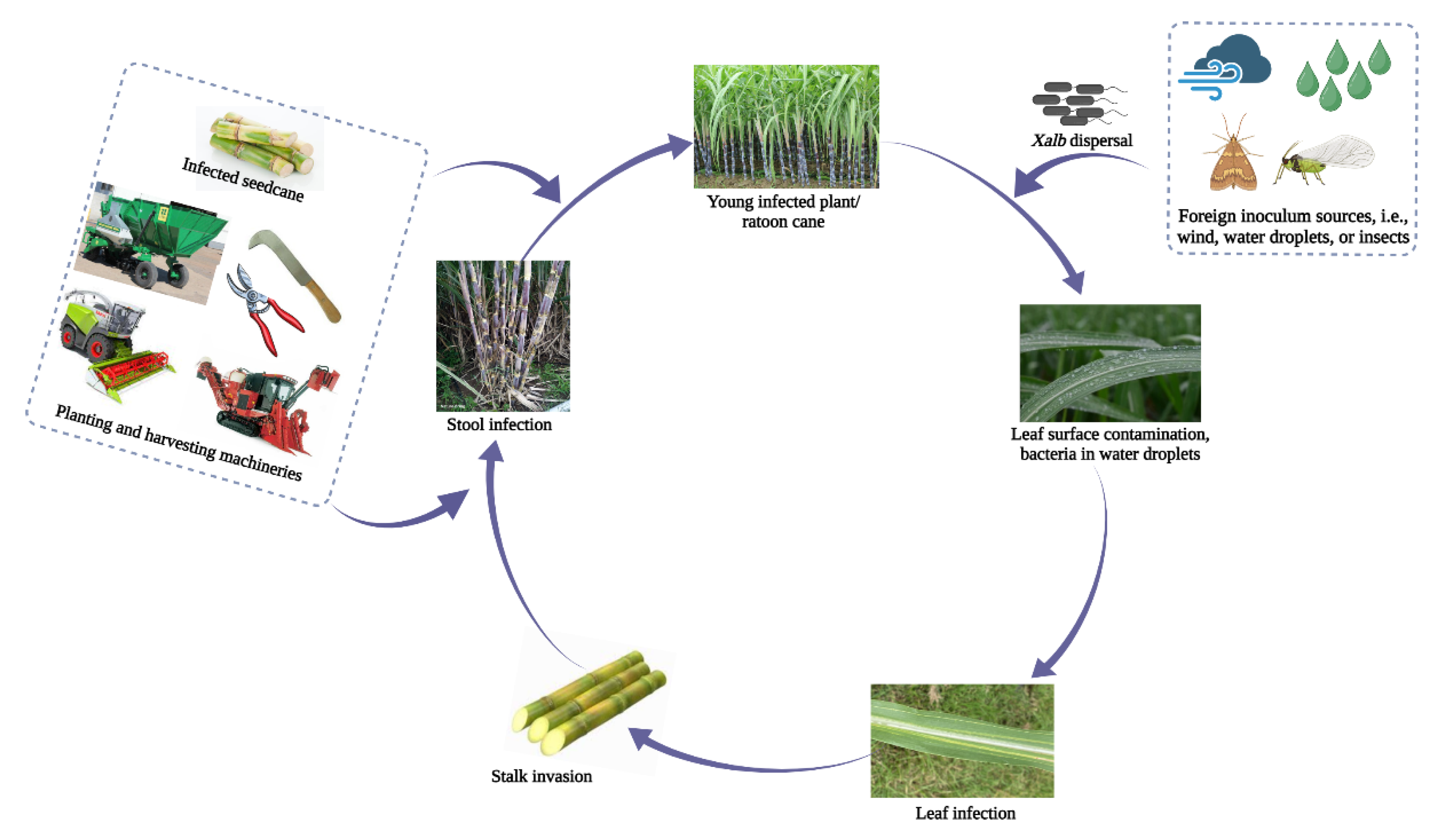
2. Infection Process and Transmission

3. Distribution and Economic Impact of LS
4. Current Management Practices for LS and Their Limitations
5. Traditional and Advanced Detection Methods for Xalb
6. Challenges in On-Farm Detection of LS
7. Conclusions and Future Directions
Funding
Conflicts of Interest
Acknowledgments
References
- Alvarez, A.M., Schenck, S., Benedict, A.A., 1996. Differentiation of Xanthomonas albilineans strains with monoclonal antibody reaction patterns and DNA fingerprints. Plant Pathol. 45, 358-366. [CrossRef]
- Arencibia, A.D., Vinagre, F., Estevez, Y., Bernal, A., Perez, J., Cavalcanti, J., et al., 2006. Gluconacetobacter diazotrophicus elicits a sugarcane defense response against a pathogenic bacterium Xanthomonas albilineans. Plant Signal. Behav. 1(5), 265-273. [CrossRef]
- Autrey, L.J.C., Saumtally, S., Dookun, A., Sullivan, S., Dhayan, S., 1992b. Aerial transmission of the leaf scald pathogen, Xanthomonas albilineans. Proc. 21st Int. Soc. Sugar Cane Technol. Congress, Bangkok, Thailand, 135-136.
- Autrey, L.J.C., Dookun, A., Saumtally, S., 1990. Improved serological methods for diagnosis of the leaf scald bacterium, Xanthomonas albilineans. Proc. Int. Soc. Sugar Cane Technol. 20, 704-713.
- Autrey, L.J.C., Saumtally, S., Dookun, A., Sullivan, S., Dhayan, S., 1995. Aerial transmission of leaf scald pathogen, Xanthomonas albilineans. Proc. Int. Soc. Sugar Cane Technol. 21, 508-526.
- Bakhtari, A., 2014. Managing odour sample degradation through on-site olfactometry and proper sample transportation and storage. Chem. Eng. Trans. 40, 163-168. [CrossRef]
- Birch, R., Bower, R., Elliott, A., Hansom, S., Basnayake, S., Zhang, L., 2000. Regulation of transgene expression: Progress towards practical development in sugarcane, and implications for other plant species. In: Proc. Int. Symp. Plant Genet. Eng., Havana, Cuba: Elsevier Science, 1999, pp. 1-12.
- Birch, R.G., Patil, S.S., 1987a. Correlation between albicidin production and chlorosis induction by Xanthomonas albilineans, the sugarcane pathogen. Physiol. Mol. Plant Pathol. 30, 199-206. [CrossRef]
- Birch, R.G., Patil, S.S., 1987b. Evidence that an albicidin-like phytotoxin induces chlorosis in sugarcane leaf scald disease by blocking plastid DNA replication. Physiol. Mol. Plant Pathol. 30, 207-214. [CrossRef]
- Birch, R.G., 2001. Xanthomonas albilineans and the antipathogenesis approach to disease control. Mol. Plant Pathol. 2, 1-11. [CrossRef]
- Blanch, M., Rodríguez, C.W., Legaz, M.E., Vicente, C., 2006. Modifications of sucrose crystallization by xanthans produced by Xanthomonas albilineans, a sugarcane pathogen. Sugar Tech 8, 255-259.
- Blanco, Y., Blanch, M.A., Piñón, D., Legaz, M.E., Vicente, C., 2005. Antagonism of Gluconacetobacter diazotrophicus against Xanthomonas albilineans studied in alginate-immobilized sugarcane stalk tissues. J. Biosci. Bioeng. 99(4), 366-371. [CrossRef]
- Blanco, Y., Legaz, M.E., Vicente, C., 2010. Gluconacetobacter diazotrophicus, a sugarcane endophyte, inhibits xanthan production by sugarcane-invading Xanthomonas albilineans. J. Plant Interact. 5, 241-248. [CrossRef]
- Cao, H., Shockey, J.M., 2012. Comparison of TaqMan and SYBR Green qPCR methods for quantitative gene expression in tung tree tissues. J. Agric. Food Chem. 60, 12296-12303. [CrossRef]
- Champoiseau, P., Rott, P., Daugrois, J.H., 2009. Epiphytic populations of Xanthomonas albilineans and subsequent sugarcane stalk infection are linked to rainfall in Guadeloupe. Plant Dis. 93, 339-346. [CrossRef]
- Champoiseau, P., Daugrois, J.H., Girard, J.C., Royer, M., Rott, P., 2006. Variation in albicidin biosynthesis genes and in pathogenicity of Xanthomonas albilineans, the sugarcane leaf scald pathogen. Phytopathology 96, 33-45. [CrossRef]
- Chatterjee, S., Almeida, R.P.P., Lindow, S., 2008. Living in two worlds: the plant and insect lifestyles of Xylella fastidiosa. Annu. Rev. Phytopathol. 46, 243-271. [CrossRef]
- Chen, C.T., Lin, C.P., Liang, Y.G., 1993. Leaf scald of sugarcane in Taiwan. Taiwan Sugar 40, 8-16.
- Chen, Y., Wang, Z., Liu, Y., Wang, X., Li, Y., Ma, P., Gu, B., Li, H., 2018. Recent advances in rapid pathogen detection method based on biosensors. Eur. J. Clin. Microbiol. Infect. Dis. 37, 1021-1037. [CrossRef]
- Comstock, J.C., Irey, M.S., 1992. Detection of the leaf scald pathogen, Xanthomonas albilineans, using tissue blot immunoassay, ELISA, and isolation techniques. Plant Dis. 76, 1033-1035.
- Comstock, J.C., Shine, J.M., 1992. Outbreak of leaf scald of sugarcane, caused by Xanthomonas albilineans, in Florida. Plant Dis. 76, 426. [CrossRef]
- Costet, L., Cunff, L., Royaert, S., Raboin, L.M., Hervouet, C., Toubi, L., et al., 2012. Haplotype structure around Bru1 reveals a narrow genetic basis for brown rust resistance in modern sugarcane cultivars. Theor. Appl. Genet. 125, 825-836. [CrossRef]
- Croft, B., Magarey, R., Whittle, P., 2000. Disease management. In: Manual of Cane Growing (eds Hogarth, D.M., Allsopp, P.G.). SRA, Brisbane.
- Croft, B.J., Cox, M., 2013. Procedures for the establishment and operation of approved seed plots. Sugar Research Australia Ltd. https://elibrary.sugarresearch.com.au/handle/11079/15325.
- Danhorn, T., Fuqua, C., 2007. Biofilm formation by plant-associated bacteria. Annu. Rev. Microbiol. 61, 401-422. [CrossRef]
- Daugrois, J.H., Dumont, V., Champoiseau, P., Costet, L., Boisne-Noc, R., Rott, P., 2003. Aerial contamination of sugarcane in Guadeloupe by two strains of Xanthomonas albilineans. Eur. J. Plant Pathol. 109, 445-458. [CrossRef]
- Daugrois, J.H., Boisne-Noc, R., Champoiseau, P., Rott, P., 2012. The revisited infection cycle of Xanthomonas albilineans, the causal agent of leaf scald of sugarcane. Funct. Plant Sci. Biotechnol. 6(2), 91-97.
- Davis, M.J., 1992. Increased incidence of leaf scald disease in Florida associated with a genetic variant of Xanthomonas albilineans. Sugar Y Azúcar 87(6), 34.
- Davis, M.J., Rott, P., Baudin, P., Dean, J.L., 1994. Evaluation of selective media and immunoassays for detection of Xanthomonas albilineans, causal agent of sugarcane leaf scald disease. Plant Dis. 78, 78-82. [CrossRef]
- Davis, M.J., Rott, P., Astua-Monge, G., 1998. Multiplex, nested, PCR for detection of Clavibacter xyli subsp. xyli and Xanthomonas albilineans. Phytopathology 88(9 Suppl), S20.
- Davis, M.J., Warmuth, C.J., Rott, P., Chatenet, M., Baudin, P., 1994. Worldwide genetic variation in Xanthomonas albilineans. ISSCT.
- Dias, D.V., Fernandez, E., Cunha, M.G., Pieretti, I., Hincapie, M., Roumagnac, P., Comstock, J.C., Rott, P., 2018. Comparison of loop-mediated isothermal amplification, polymerase chain reaction, and selective isolation assays for detection of Xanthomonas albilineans from sugarcane. Trop. Plant Pathol. 43, 351-359. [CrossRef]
- Dias, V.D., Carrer Filho, R., de Campos Dianese, É., da Cunha, M.G., 2019. Detection of sugarcane leaf scald from latent infections. Científica 47(1), 21-27. [CrossRef]
- Diaz, M., Peralta, E.L., Iglesia, A., Pazos, V., Carvajal, O., Perez, M.P., Giglioti, E.A., Gagliardi, P.R., Wendland, A., Camargo, L.E.A., 2001. Xanthomonas albilineans haplotype B responsible for a recent sugarcane leaf scald disease outbreak in Cuba. Plant Dis. 85, 334. [CrossRef]
- Duan, Y.Y., Zhang, Y.Q., Xu, Z.X., Lin, Y., Mao, L.R., Wang, W.H., Deng, Z.H., Huang, M.T., Gao, S.J., 2021. First report of Xanthomonas albilineans causing leaf scald on two chewing cane clones in Zhejiang province, China. Plant Dis. 105, 485. [CrossRef]
- Egan, B.T., Sturgess, O.W., 1980. Commercial control of leaf scald disease by thermotherapy and a clean seed program. Proc. Int. Soc. Sugar Cane Technol. 17, 1602–1606.
- Engvall, E., 2010. The ELISA, enzyme-linked immunosorbent assay. Clin. Chem. 56, 319–320. [CrossRef]
- Feldmann, P., Sapotille, J., Grédoire, P., Rott, P., 1994. Micropropagation of sugar cane. In: Teisson, C. (Ed.), In Vitro Culture of Tropical Plants. La Librairie du Cirad, Montpellier, France, pp. 15–17.
- Fischbach, J., Xander, N.C., Frohme, M., Glökler, J.F., 2015. Shining a light on LAMP assays—A comparison of LAMP visualization methods including the novel use of berberine. Biotechnol. Tech. 58, 189–194. [CrossRef]
- Flynn, J.L., Anderlini, T.A., 1990. Disease incidence and yield performance of tissue culture generated seedcane over the crop cycle in Louisiana. J. Am. Soc. Sugar Cane Technol. 10, 113.
- Fontaniella, B., Rodriguez, C.W., Piñón, D., Vicente, C., Legaz, M.E., 2002. Identification of xanthans isolated from sugarcane juices obtained from scalded plants infected by Xanthomonas albilineans. J. Chromatogr. B 770, 275–281. [CrossRef]
- Garces, F.F., Gutierrez, A., Hoy, J.W., 2014. Detection and quantification of Xanthomonas albilineans by qPCR and potential characterization of sugarcane resistance to leaf scald. Plant Dis. 98, 121–126. [CrossRef]
- García-Juárez, H.S., Ortiz-García, C.F., Salgado-García, S., Valdez-Balero, A., Silva-Rojas, H.V., Ovalle-Sáenz, W.R., 2015. Presence of Xanthomonas albilineans (Ashby) Dowson in sugarcane crops in La Chontalpa, Tabasco, México. Rev. Fitotec. Mex. 48, 397–404. [CrossRef]
- Ginzinger, D.G., 2002. Gene quantification using real-time quantitative PCR: An emerging technology hits the mainstream. Exp. Hematol. 30, 503–512. [CrossRef]
- Goto, M., Honda, E., Ogura, A., Nomoto, A., Hanaki, K., 2009. Colorimetric detection of loop mediated isothermal amplification reaction by using hydroxy naphthol blue. Biotechnol. Tech. 46, 167–172. [CrossRef]
- Grisham, M.P., Legendre, B.L., Comstock, J.C., 1994. First report of leaf scald, caused by Xanthomonas albilineans, of sugarcane in Louisiana. Plant Dis. 77, 537. [CrossRef]
- Hedman, J., Rådström, P., 2013. Overcoming inhibition in real-time diagnostic PCR. In: Wilks, M. (Ed.), PCR Detection of Microbial Pathogens. Methods in Molecular Biology, Vol. 943. Humana Press, Totowa, NJ, pp. 17–48.
- Honeycutt, R.J., Sobral, B.W., McClelland, M., 1995. tRNA intergenic spacers reveal polymorphisms diagnostic for Xanthomonas albilineans. Microbiology 141, 3229–3239. [CrossRef]
- Huang, Y., Li, W., 2016. Colored Atlas of Control on Diseases, Insect Pests and Weeds of Modern Sugarcane. China Agriculture Press, Beijing.
- Huerta-Lara, M., Cárdenas-Soriano, E., Rojas-Martínez, R.I., López-Olguín, J.F., Reyes-López, D., Bautista-Calles, J., Romero-Arenas, O., 2009. Vascular bundle occlusion as a measure of sugarcane resistance to Xanthomonas albilineans. Interciencia 34, 247–251.
- Irey, M.S., Comstock, J.C., 1991. Use of an enzyme-linked immunosorbent assay to detect leaf scald pathogen, Xanthomonas albilineans, in sugarcane. J. Am. Soc. Sugar Cane Technol. 11, 48–52.
- Irvine, J.E., Amador, M., Gallo, R.M.I., Riess, C.M., Comstock, J.C., 1993. First report of leaf scald, caused by Xanthomonas albilineans, of sugarcane in Mexico. Plant Dis. 77, 846. [CrossRef]
- Isakeit, T., Irvine, J.E., 1995. First report of leaf scald, caused by Xanthomonas albilineans, of sugarcane in Texas. Plant Dis. 79, 860. [CrossRef]
- Jackson, R.W., Johnson, L.J., Clarke, S.R., Arnold, D.L., 2011. Bacterial pathogen evolution: breaking news. Trends Genet. 27, 32–40. [CrossRef]
- Keymanesh, K., Soltani, S., Sardari, S., 2009. Application of antimicrobial peptides in agriculture and food industry. World J. Microbiol. Biotechnol. 25, 933–944. [CrossRef]
- Klett, P., Rott, P., 1994. Inoculum sources for the spread of leaf scald disease of sugarcane caused by Xanthomonas albilineans in Guadeloupe. J. Phytopathol. 142, 283–291. [CrossRef]
- Li, W.F., Huang, Y.K., 2012. Diagnosis, Detection and Control Technology of Modern Sugarcane Diseases. China Agriculture Press, Beijing.
- Lim, J.W., Ha, D., Lee, J., Lee, S.K., Kim, T., 2015. Review of micro/nanotechnologies for microbial biosensors. Front. Bioeng. Biotechnol. 3, 61. [CrossRef]
- Lin, L.H., Ntambo, M.S., Rott, P.C., Wang, Q.N., Lin, Y.H., Fu, H.Y., Gao, S.J., 2018. Molecular detection and prevalence of Xanthomonas albilineans, the causal agent of sugarcane leaf scald, in China. Crop Prot. 109, 17–23. [CrossRef]
- Lindeberg, M., 2012. Genome-enabled perspectives on the composition, evolution, and expression of virulence determinants in bacterial plant pathogens. Annu. Rev. Phytopathol. 50, 111–132. [CrossRef]
- Lopes, S.A., Damann, K., Grelen, L., 2001. Xanthomonas albilineans diversity and identification based on Rep-PCR fingerprints. Curr. Microbiol. 42, 155–159. [CrossRef]
- Maier, T.S., Kuhn, J., Müller, C., 2010. Proposal for field sampling of plants and processing in the lab for environmental metabolic fingerprinting. Plant Methods 6, 6. [CrossRef]
- Martin, J.P., 1938. Sugarcane Diseases in Hawaii. Hawaiian Sugar Planters Association, Hawaii, 295 pp.
- Martin, J.P., Carpenter, C.W., Weller, D.M., 1932. Leaf scald disease of sugar cane in Hawaii. Plan. Rec. 36, 145–196.
- Martin, J.P., Robinson, P.E., C.G., 1961. Leaf scald. In: Martin, J.P., Abbott, E.V., Hughes, C.G. (Eds.), Sugarcane Diseases of the World, Vol. 1. Elsevier, Amsterdam, pp. 79–107.
- Martinelli, F., Scalenghe, R., Davino, S., Panno, S., Scuderi, G., Ruisi, P., Villa, P., Stroppiana, D., Boschetti, M., Goulart, L.R., Cristina, E., Davis, C.E., Dandekar, A.M., 2015. Advanced methods of plant disease detection. A review. Agron. Sustain. Dev. 35, 1–25. [CrossRef]
- Mehdi, F., Cao, Z., Zhang, S., Gan, Y., Cai, W., Peng, L., Wu, Y., Wang, W., Yang, B., 2024. Factors affecting the production of sugarcane yield and sucrose accumulation: suggested potential biological solutions. Front. Plant Sci. 15, 1374228. [CrossRef]
- Mensi, I., Girard, J.C., Pieretti, I., Larbre, F., Roumagnac, P., Royer, M., Rott, P., 2013. First report of sugarcane leaf scald in Gabon caused by a highly virulent and aggressive strain of Xanthomonas albilineans. Plant Dis. 97, 988. [CrossRef]
- Mensi, I., Vernerey, M.-S., Gargani, D., Nicole, M., Rott, P., 2014. Breaking dogmas: the plant vascular pathogen Xanthomonas albilineans is able to invade non-vascular tissues despite its reduced genome. Open Biol. 4, 130116. [CrossRef]
- Nagamine, K., Hase, T., Notomi, T., 2002. Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 16, 223–229. [CrossRef]
- Ocenar, J., Arizala, D., Boluk, G., Dhakal, U., Gunarathne, S., Paudel, S., Dobhal, S., Arif, M., 2019. Development of a robust, field-deployable loop-mediated isothermal amplification (LAMP) assay for specific detection of potato pathogen Dickeya dianthicola targeting a unique genomic region. PLoS One 14, e0218868. [CrossRef]
- Ovalle, W., Comstock, J.C., Juarez, J., Soto, G., 1995. First report of leaf scald of sugarcane (Xanthomonas albilineans) in Guatemala. Plant Dis. 79, 212. [CrossRef]
- Pan, Y.B., Grisham, M., Burner, D., Legendre, B., Wei, Q., 1999. Development of polymerase chain reaction primers highly specific for Xanthomonas albilineans, the causal bacterium of sugarcane leaf scald disease. Plant Dis. 83, 218–222. [CrossRef]
- Pan, Y.B., Grisham, M., Burner, D., 1997. A polymerase chain reaction protocol for the detection of Xanthomonas albilineans, the causal agent of sugarcane leaf scald disease. Plant Dis. 81, 189–194. [CrossRef]
- Pieretti, I., et al., 2009. The complete genome of Xanthomonas albilineans provides new insights into the reductive genome evolution of the xylem-limited Xanthomonadaceae. BMC Genomics 10, 616. [CrossRef]
- Pieretti, I., et al., 2012. Genomic insights into strategies used by Xanthomonas albilineans with its reduced artillery to spread within sugarcane xylem vessels. BMC Genomics 13, 658. [CrossRef]
- Pieretti, I., Pesic, A., Petras, D., Royer, M., Süssmuth, R.D., Cociancich, S., 2015b. What makes Xanthomonas albilineans unique amongst xanthomonads? Front. Plant Sci. 6, 289. [CrossRef]
- Piñón, D., Casas, M., Blanch, M.A., Fontaniella, B., Blanco, Y., Vicente, C., et al., 2002. Gluconacetobacter diazotrophicus, a sugar cane endosymbiont, produces a bacteriocin against Xanthomonas albilineans, a sugar cane pathogen. Res. Microbiol. 153, 345–351. [CrossRef]
- Prasad, R., Bhattacharyya, A., Nguyen, Q.D., 2017. Nanotechnology in sustainable agriculture: recent developments, challenges, and perspectives. Front. Microbiol. 8, 1014. [CrossRef]
- Prasannakumar, M., Parivallal, P.B., Pramesh, D., Mahesh, H.B., Raj, E., 2021. LAMP-based foldable microdevice platform for the rapid detection of Magnaporthe oryzae and Sarocladium oryzae in rice seed. Sci. Rep. 11, 178. [CrossRef]
- Purcell, A.H., Hopkins, D.L., 1996. Fastidious xylem-limited bacterial plant pathogens. Annu. Rev. Phytopathol. 34, 131–151. [CrossRef]
- Rački, N., Dreo, T., Gutierrez-Aguirre, I., Blejec, A., Ravnikar, M., 2014. Reverse transcriptase droplet digital PCR shows high resilience to PCR inhibitors from plant, soil and water samples. Plant Methods 10, 42. [CrossRef]
- Rafiq, A., Ali, W.R., Asif, M., Ahmed, N., Khan, W.S., Mansoor, S., Bajwa, S.Z., Amin, I., 2021. Development of a LAMP assay using a portable device for the real-time detection of cotton leaf curl disease in field conditions. Biol. Methods Protoc. 6, bpab010. [CrossRef]
- Ricaud, C., Ryan, C.C., 1989. Leaf scald. In: Ricaud, C., Egan, B.T., Gillaspie, A.G., Hughes, C.G. (Eds.), Diseases of Sugarcane: Major Diseases. Elsevier, Amsterdam, pp. 39–58.
- Ricaud, C., Perombelon, M., 1964. Leaf scald. MSIRI Annu. Rep., Réduit, Mauritius, pp. 56–58.
- Rott, P., 1995. Leaf scald disease. In: Croft, B.J., Piggin, C.M., Wallis, E.S., Hogarth, D.M. (Eds.), Sugarcane Germplasm Conservation and Exchange. ACIAR Proceedings No. 67, Canberra, pp. 123–124.
- Rott, P., Arnaud, M., Baudin, P., 1986. Serological and lysotypical variability of Xanthomonas albilineans (Ashby) Dowson, causal agent of sugarcane leaf scald disease. J. Phytopathol. 116, 201–211. [CrossRef]
- Rott, P., Davis, M., 1995. Recent advances in research on variability of Xanthomonas albilineans, causal agent of sugarcane leaf scald disease. In: Proc. Int. Soc. Sugarcane Technol. Congr., Vol. 22, pp. 498–503.
- Rott, P., Davis, M.J., 2000. Leaf scald. In: Rott, P., Bailey, R.A., Comstock, J.C., Croft, B.J., Saumtally, A.S. (Eds.), A Guide to Sugarcane Diseases. CIRAD-ISSCT, Montpellier, France, pp. 38–44.
- Rott, P., Fleites, L., Marlow, G., Royer, M., Gabriel, D.W., 2011. Identification of new candidate pathogenicity factors in the xylem-invading pathogen Xanthomonas albilineans by transposon mutagenesis. Mol. Plant Microbe Interact. 24, 594–605. [CrossRef]
- Rott, P., Soupa, D., Brunet, Y., Feldmann, P., Letourmy, P., 1995. Leaf scald (Xanthomonas albilineans) incidence and its effect on yield in seven sugarcane cultivars in Guadeloupe. Plant Pathol. 44, 1075–1084. [CrossRef]
- Sandhu, H.S., Rott, P., Comstock, J.C., Gilbert, R.A., 2013. Diseases in sugarcane. Sugarcane Handbook. Agronomy Department, UF/IFAS Extension, University of Florida. Retrieved from: http://edis.ifas.ufl.edu/SC001.
- Saumtally, A., Dookun-Saumtally, A., Rao, G.P., Saumtally, A.S., Rott, P., 2004. Leaf scald of sugarcane: a disease of worldwide importance. In: Sugarcane Pathology: Bacterial and Nematode Diseases. New Hampshire: Science Publishers, pp. 65–76.
- Schori, M., Appel, M., Kitko, A., Showalter, A.M., 2013. Engineered DNA polymerase improves PCR results for plastid DNA. Appl. Plant Sci. 1, 1200519. [CrossRef]
- Shi, Y., Zhao, J.Y., Zhou, J.R., Ntambo, M.S., Xu, P.Y., Rott, P.C., Gao, S.J., 2021. Molecular detection and quantification of Xanthomonas albilineans in juice from symptomless sugarcane stalks using a real-time quantitative PCR assay. Plant Dis. 105, 3451–3458. [CrossRef]
- Tsai, C.C., Lin, C.P., Chen, C.T., 1990. Characterization of Xanthomonas albilineans (Ashby) Dowson, the causal agent of sugarcane leaf scald disease. Plant Protect. Bull. 32, 125–135.
- Umer, M., Aziz, N.B., Al Jabri, S., Bhuiyan, S.A., Shiddiky, M.J., 2021. Naked eye evaluation and quantitative detection of the sugarcane leaf scald pathogen, Xanthomonas albilineans, in sugarcane xylem sap. Crop Pasture Sci. 72, 361–371. [CrossRef]
- Urashima, A.S., Zavaglia, A.C., 2012. Comparison of two diagnostic methods for leaf scald (Xanthomonas albilineans) of sugarcane. Summa Phytopathol. 38, 155–158. [CrossRef]
- Viswanathan, R., Padmanaban, P., 2008. Handbook on Sugarcane Diseases and their Management. Coimbatore: Sugarcane Breeding Institute.
- Viswanathan, R., Ramesh, S.A., 2004. Isolation and production of antiserum to Xanthomonas albilineans and diagnosis of the pathogen through serological techniques. J. Mycol. Plant Pathol. 34, 797–800.
- Viswanathan, R., Padmanaban, P., Mohanraj, D., Nallathambi, P., 1997. Occurrence of leaf scald disease in Tamil Nadu. Indian Phytopathol. 50, 149.
- Walker, D.I.T., 1987. Breeding for resistance. In: Heinz, J.D. (Ed.), Sugarcane Improvement through Breeding. Elsevier Science Publishers B.V., Amsterdam, pp. 445–502.
- Wang, H.B., Xiao, N.Y., Wang, Y.J., Guo, J.L., Zhang, J.S., 2020. Establishment of a qualitative PCR assay for the detection of Xanthomonas albilineans (Ashby) Dowson in sugarcane. Crop Prot. 130, 105053. [CrossRef]
- Wang, Z.K., Comstock, J.C., Hatziloukas, E., Schaad, N.W., 1999. Comparison of PCR, BIO-PCR, DIA, ELISA and isolation on semiselective medium for detection of Xanthomonas albilineans, the causal agent of leaf scald of sugarcane. Plant Pathol. 48, 245–252. [CrossRef]
- Wiehe, P.O., 1951. Leaf scald and chlorotic streak—two sugarcane diseases occurring in British Guiana. Lecture to Br. Guiana Sugar Prod. Ass.
- Zhang, L., Birch, R.G., 1996. Biocontrol of sugar cane leaf scald disease by an isolate of Pantoea dispersa which detoxifies albicidin phytotoxins. Lett. Appl. Microbiol. 22, 132–136. [CrossRef]
- Zhang, R.Y., Shan, H.L., Li, W.F., Cang, X.Y., Wang, X.Y., Yin, J., Luo, Z.M., Huang, Y.K., 2017. First report of sugarcane leaf scald caused by Xanthomonas albilineans in the province of Guangxi, China. Plant Dis. 101, 1541. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



