Submitted:
22 August 2025
Posted:
25 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Impact and Response to Diabetes Mellitus
1.2. 5-ALA and Its Potential Role
1.3. Specific Hypothesis and Study Objectives
2. Materials and Methods
2.1. Study Design
2.2. Inclusion Criteria
- Age between 20 and 75 years and considered to be in general good health (aside from having diabetes) as determined by the results of their medical history, physical examinations, and laboratory assessments.
- HbA1c levels between 6.5% and 10% (inclusive), which is uncontrolled, despite the use of one or more oral hypoglycemic agents
- Body Mass Index (BMI) ≤ 40 kg/m2 (Six patients exceeded the defined BMI limit of ≤ 40 kg/m2 )
- Sitting blood pressure (BP)≤160/100 mmHg
- Ophthalmological exam within normal limits as judged by the investigator. If abnormal findings are observed, they must be evaluated as not clinically significant
- Female subjects who are not pregnant or breastfeeding. If of childbearing potential, they should agree to use an acceptable form of birth control
2.3. Exclusion Criteria
- Liver dysfunction, defined as liver function test values ≥1.5 times the normal upper limit
- Renal dysfunction, defined as blood urea nitrogen and/or serum creatinine values ≥ 1.5 times the upper limit of normal and/or estimated Glomerular Filtration Rate (eGFR) < 30 ml/min/1.73 m2
- History of any life-threatening disease, cardiovascular disease, viral hepatitis, porphyria, or hemochromatosis
- Allergy to 5-ALA phosphate, SFC, or any other component of the study product
- Use of insulin for the management of serum glucose
- Hypoglycemic event within the previous 3 months, defined as serum glucose levels less than 70 mg/dl (3.89 mmol/l)
- History of sickle cell anemia
2.4. Doses
2.5. The Standard of Care Used for Diabetes Patients
- Monotherapy: metformin
- Dual Therapy: metformin + sulfonylurea
- Triple Therapy: metformin + sulfonylurea + DPP-4 Inhibitor
2.6. Statistical Analysis
3. Results
- Pre-study: 16 weeks before treatment initiation (t0–16 weeks)
- Baseline: At the start of treatment (t0)
- Follow-up: 16 weeks after treatment initiation (t0+16 weeks)
- Post-study: 16 weeks after the end of treatment (t0+32 weeks)
- Monotherapy: Treated with one antidiabetic drug, mainly metformin
- Dual Therapy: Treated with two drugs, typically metformin and sulfonylurea
- Triple Therapy: Treated with three drugs, most commonly metformin, sulfonylurea, and a DPP-4 inhibitor
3.1. Demographics
3.2. Adverse Events and Safety
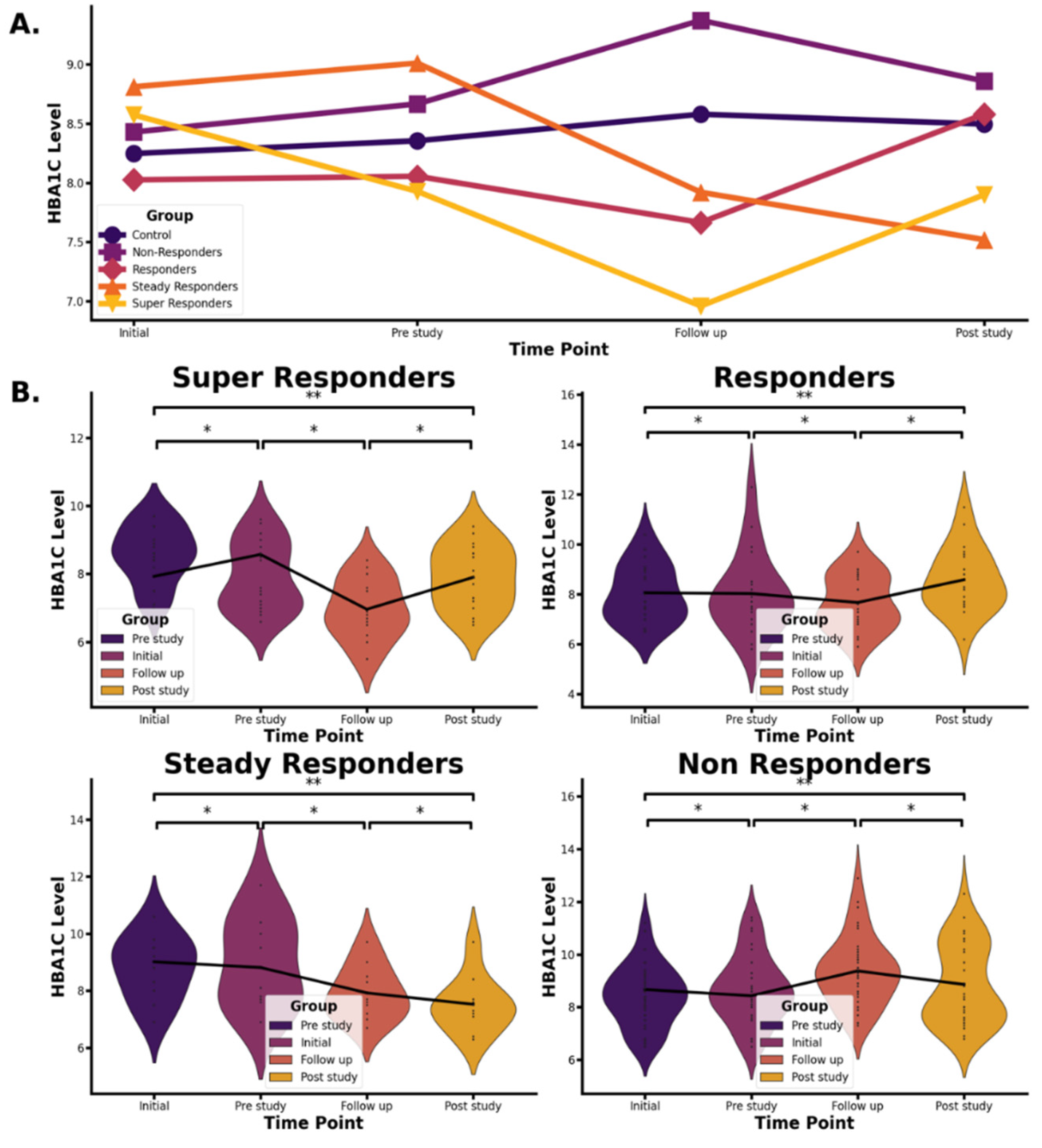
3.3. Efficacy: Patient Response to 5-ALA Phosphate /SFC
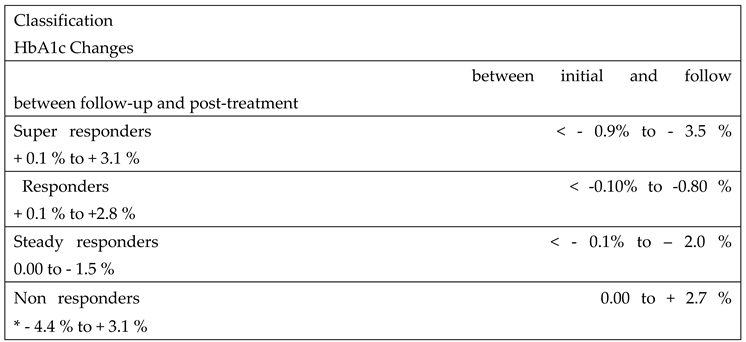
- Super Responders: These patients exhibited a marked and statistically significant reduction in HbA1c levels four weeks after initiating treatment. However, this effect diminished following the discontinuation of 5-ALA phosphate/SFC, with HbA1c levels rising significantly during the post-treatment phase.
- Responders: This group showed a moderate reduction in HbA1c levels four weeks after starting treatment. Like the super responders, their HbA1c levels increased after stopping treatment, indicating a temporary benefit during 5-ALA phosphate/SFC treatment.
- Steady Responders: Patients in this group experienced a sustained reduction in HbA1c after 16 weeks of treatment, which persisted even four weeks after discontinuation of 5-ALA phosphate/SFC, suggesting a more prolonged therapeutic effect.
- Non-Responders: These individuals showed no improvement or an increase in HbA1c levels throughout the study period, indicating no measurable response to the 5-ALA phosphate/SFC treatment.
3.4. Efficacy: Age and Gender Variability in Response to 5-ALA Phosphate/SFC Treatment
3.5. HbA1c Changes in Patients Receiving Monotherapy, Dual Therapy, and Triple Therapy With and Without 5-ALA Phosphate/SFC
- Monotherapy: Treated with one antidiabetic drug, mainly metformin
- Dual Therapy: Treated with two drugs, typically metformin and sulfonylurea
- Triple Therapy: Treated with three drugs, most commonly metformin, sulfonylurea, and a DPP-4 inhibitor
3.6. Changes in Weight and BMI
3.7. Laboratory Blood Analysis
4. Discussions
4.1. Safety and Effects of 5-ALA/SFC on Liver and Kidney Functions
4.2. Efficacy: Overall Patient Response to 5-ALA Phosphate/SFC
- 7 patients responded to insulin with a reduction in HbA1c,
- 1 patient showed no change,
- 3 patients exhibited further increases in HbA1c, indicating limited or no response even with intensified therapy.
4.3. Efficacy: Overall Patient Response to 5-ALA Phosphate/SFC
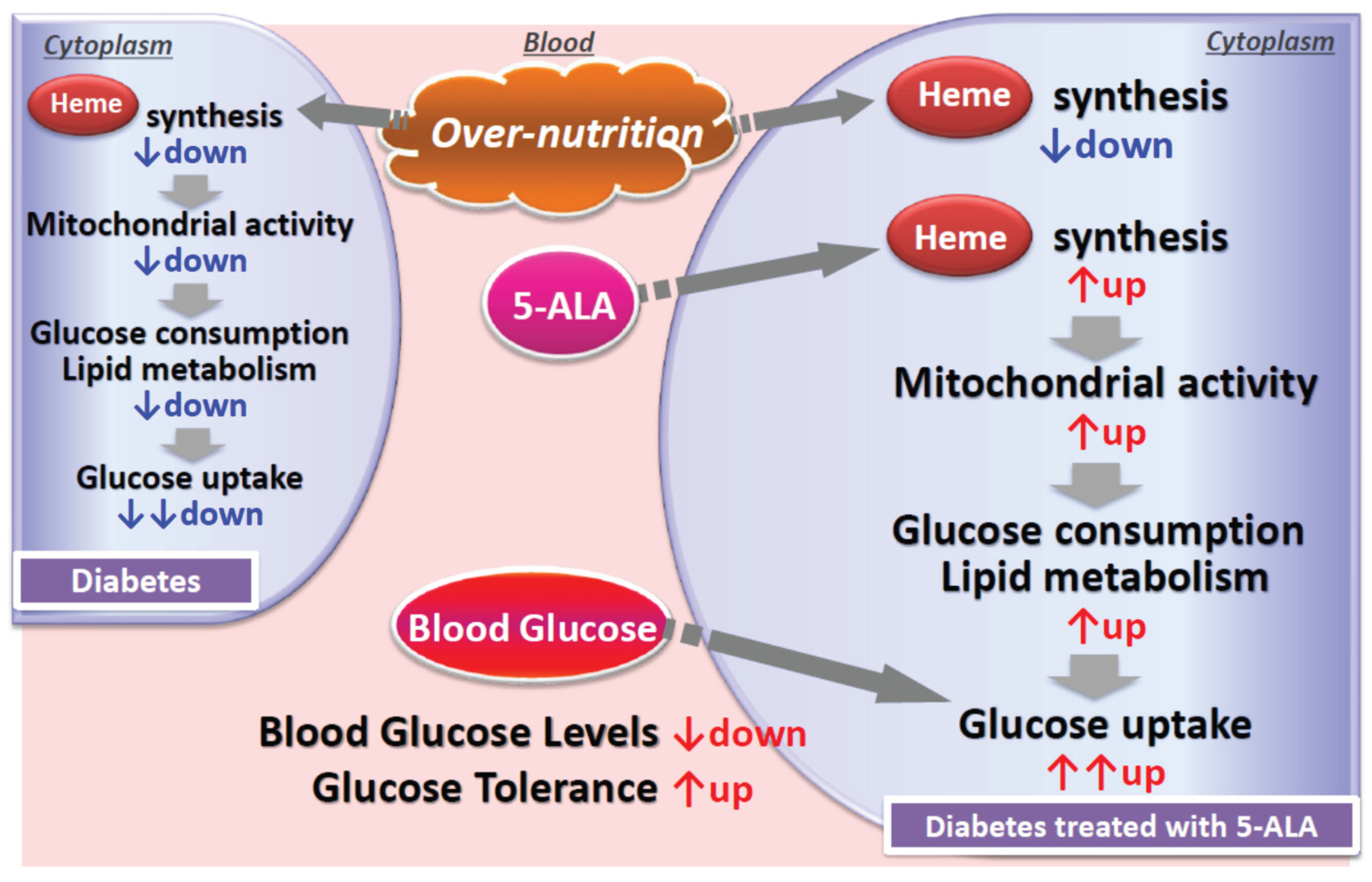
4.4. Proposed Mechanism of Action and Response Variability to 5-ALA/SFC
4.5. Effect of Hypoglycemic Agents on Reducing HbA1c
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- World Health Organization. Global Report on Diabetes. World Health Organization, 2016. https://www.who.int/publications/i/item/9789241565257.
- Diabetes Atlas. 10th ed., International Diabetes Federation Diabetes Atlas 10th ed, 2021. https://diabetesatlas.org/idfawp/resource files/2021/07/IDF_Atlas_10th_Edition_2021.pdf.
- Bahrain Ministry of Health website, Our services, Diabetes. https://www.moh.gov.bh/Services/Diabetes?lang=en.
- NICE, National Institute for Health and Care Excellence. “Type 2 Diabetes in Adults: Management.” NICE Guidelines, 28 Dec. 2015, pp. 9. https://www.nice.org.uk/guidance/ng28/resources/2019-surveillance-of-diabetes-nice-guidelines-ng17-ng18-ng19-and-ng28-pdf-8862045321157.
- Hirst, J. A., A. J. Farmer, R. Ali, N. W. Roberts, and R. J. Stevens. “Quantifying the Effect of Metformin Treatment and Dose on Glycemic Control.” Diabetes Care, 2012, vol. 35, no. 2, pp. 446-454. https://pubmed.ncbi.nlm.nih.gov/22275444/. [CrossRef]
- Phung, O. J., J. M. Scholle, M. Talwar, and C. I. Coleman. “Effect of Noninsulin Antidiabetic Drugs Added to Metformin Therapy on Glycemic Control, Weight Gain, and Hypoglycemia in Type 2 Diabetes.” JAMA, 2010, vol. 303, no.14, pp. 1410-1418. https://pubmed.ncbi.nlm.nih.gov/20388897/. [CrossRef]
- The Look AHEAD Research Group. “Long Term Effects of a Lifestyle Intervention on Weight and Cardiovascular Risk Factors in Individuals with Type 2 Diabetes: Four Year Results of the Look AHEAD Trial.” Arch Med Res., 2010, vol. 170, no. 17, pp. 1566-1575. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3084497/. [CrossRef]
- Ishizuka M, Abe F, Sano Y, Takahashi K, Inoue K, Nakajima M, Kohda T, Komatsu N, Ogura S, Tanaka T. Novel development of 5-aminolevulinic acid (ALA) in cancer diagnoses and therapy. Int Immunopharmacol. 2011 Mar;11(3):358-65. PMID: 21144919. [CrossRef]
- Kim JA, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res. 2008 Feb 29;102(4):401-14. https://www.ahajournals.org/doi/10.1161/circresaha.107.165472. [CrossRef]
- KuryataO, Oleh Akimov O, Mykola Riabushko M, Kostenko H, Kostenko V, Mishchenko A, Nazarenko S, Solovyova N, Vitalii Kostenko V. Therapeutic potential of 5-aminolevulinic acid in metabolic disorders: Current insights and future directions. iScience 2024 Nov 26;27(12):111477. [CrossRef]
- Sato, T., T. Yasuzawa, A. Uesaka, et al. “Type 2 Diabetic Conditions in Otsuka Long-Evans Tokushima Fatty Rats Are Ameliorated by 5-Aminolevulinic Acid.” Nutr Res., vol. 34, no. 6, 2014, pp. 544-551. https://pubmed.ncbi.nlm.nih.gov/25026922/. [CrossRef]
- Saitoh, S., S. Okano, H. Nohara, H. Nakano, N. Shirasawa, A. Naito, M. Yamamoto, V. P. Kelly, K. Takahashi, T. Tanaka, M. Nakajima, and O. Nakajima. “5-Aminolevulinic Acid (ALA) Deficiency Causes Impaired Glucose Tolerance and Insulin Resistance Coincident with an Attenuation of Mitochondrial Function in Aged Mice.” PLoS One, 2018, vol. 13, no. 1, p. e0189593. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5783358/. [CrossRef]
- Al-Saber, F., W. Aldosari, M. Alselaiti, et al. “The Safety and Tolerability of 5-Aminolevulinic Acid Phosphate with Sodium Ferrous Citrate in Patients with Type 2 Diabetes Mellitus in Bahrain.” J Diabetes Res., 2016, pp. 1-10. https://pubmed.ncbi.nlm.nih.gov/27738640/. [CrossRef]
- Yamashita, N., A. Watanabe, H. Kondo, S. Kawata, T. Tanaka, and M. Nakajima. “Safety Test of a Supplement, 5-Aminolevulinic Acid Phosphate with Sodium Ferrous Citrate, in Diabetic Patients Treated with Oral Hypoglycemic Agents.” Food Funct Health Dis., 2014, vol. 4, no. 9, pp. 415-428. https://www.ffhdj.com/index.php/ffhd/article/view/151. [CrossRef]
- Stummer, W., A. Novotny, H. Stepp, C. Goetz, K. Bise, and H. J. Reulen. “Fluorescence-Guided Resection of Glioblastoma Multiforme Utilizing 5-ALA-Induced Porphyrins: A Prospective Study in 52 Consecutive Patients.” J Neurosurg., 2000, vol. 93, no. 6, pp. 1003-1013. https://pubmed.ncbi.nlm.nih.gov/11117842/. [CrossRef]
- Taoka R, Fukuhara H, Miyake M, Kobayashi K, Ikeda A, Kanao K, et al. Effect of extending the period from oral administration of 5-aminolevulinicacid hydrochloride to photodynamic diagnosis during transurethral resection for non-muscle invasive bladder cancer on diagnostic accuracy and safety: a single-arm multicenter phase III trial. Int J Clin Oncol. 2024;1–11. https://doi.org/10.1007/s10147-024-02654-5. [CrossRef]
- Rodriguez, B. L., J. D. Curb, J. Davis, T. Shintani, M. H. Perez, N. Apau-Ludlum, et al. “Use of the Dietary Supplement 5-Aminolevulinic Acid (5-ALA) and Its Relationship with Glucose Levels and Hemoglobin A1C among Individuals with Prediabetes.” Clin Transl Sci., 2012, vol. 5, no. 4, pp. 314-320. https://pubmed.ncbi.nlm.nih.gov/22883608/. [CrossRef]
- Kawamura K, Matsushima, H, Sakai, H, Iwashima, A, Nakamura S, Kojima T, Sasaki S, Shigenaga T, Natsume I, Sasaki T, Ohsaki Y, Iwanaga K, Nishi K, Mitsuishi Y, Taniguchi H, Sato K, Yamauchi M, Nakajima M, Takahashi K. A Randomized Phase 2 Study of 5-Aminolevulinic Acid Hydrochloride and Sodium Ferrous Citrate for the Prevention of Nephrotoxicity Induced by Cisplatin-Based Chemotherapy of Lung Cancer. Oncology 2022;100:620–632. [CrossRef]
- Alalawi, N. S., & Allani, N. (2024). Promoting Sustainability in Bahraini Primary Health Care Centers. In Nia, H. A., & Rahbarianyazd, R. (Eds.), Innovative Approaches to Cultural Heritage and Sustainable Urban Development: Integrating Tradition and Modernity, (pp. 1-17) Cinius Yayınları. [CrossRef]
- Rehani, P. R., H. Iftikhar, M. Nakajima, T. Tanaka, Z. Jabbar, and R. N. Rehani. “Safety and Mode of Action of Diabetes Medications in Comparison with 5-Aminolevulinic Acid (5-ALA).” J Diabetes Res., 2019, vol 2019, p. 4267357. https://www.hindawi.com/journals/jdr/2019/4267357/. [CrossRef]
| Gender | n | Minimum Age/years |
Maximum Age/years |
Mean Age/years |
Std. Deviation |
| Male | 59 | 31 | 73 | 53.6 | 9.0202 |
| Female | 41 | 36 | 77* | 51.4 | 9.7051 |
| Overall | 100 | 31 | 77 | 52.7 | 9.3681 |
| Subject number | AE |
| 2 | Nausea |
| 15 | Flatulence |
| 16 | Tremor |
| 2 | Abdominal pain |
| 2 | Diarrhea |
| 19 | Body ache |
 |
| Group | Pre study (t0-16 weeks) |
Initial (t0) | Follow up (t0+16 weeks) |
Post study ( t0+32 weeks) |
| Super Responders | 7.93 ± 1.01 [7.46, 8.40] |
8.57 ± 0.86 [8.17, 8.97] |
6.96 ± 0.88 [6.55, 7.37] |
7.90 ± 0.92 [7.48, 8.32] |
| Responders | 8.05 ± 1.59 [7.36, 8.75] |
8.03 ± 1.16 [7.52, 8.53] |
7.67 ± 1.09 [7.19, 8.14] |
8.58 ± 1.29 [8.01, 9.15] |
| Steady Responders | 9.01 ± 1.59 [8.02, 10.00] |
8.81 ± 1.13 [8.11, 9.51] |
7.92 ± 0.94 [7.34, 8.50] |
7.52 ± 0.98 [6.91, 8.13] |
| Non-Responders | 8.67 ± 1.29 [8.26, 9.07] |
8.43 ± 1.16 [8.06, 8.80] |
9.37 ± 1.31 [8.95, 9.79] |
8.86 ± 1.52 [8.38, 9.34] |
| Control | 8.35 ± 1.15 [8.08, 8.63] |
8.25 ± 1.07 [7.99, 8.51] |
8.58 ± 1.11 [8.31, 8.85] |
8.50 ± 1.36 [8.17, 8.82] |
| Group | n | Pre study vs Initial |
Initial vs Follow up |
Follow up vs Post study |
Pre study vs Post study |
| Super Responders | 18 | 0.64 ± 1.29 [0.05, 1.24] |
-1.61 ± 0.68 [-1.93, -1.30] |
0.94 ± 0.75 [0.59, 1.29] |
-0.03 ± 1.28 [-0.62, 0.56] |
| Responders | 20 | -0.03 ± 1.52 [-0.70, 0.64] |
-0.36 ± 0.25 [-0.47, -0.25] |
0.92 ± 0.78 [0.57, 1.26] |
0.53 ± 1.61 [-0.18, 1.23] |
| Steady Responders | 10 | -0.20 ± 1.82 [-1.33, 0.93] |
-0.89 ± 0.67 [-1.31, -0.47] |
-0.40 ± 0.45 [-0.68, -0.12] |
-1.49 ± 1.58 [-2.47, -0.51] |
| Non-Responders | 38 | -0.24 ± 0.83 [-0.50, 0.03] |
0.94 ± 0.72 [0.71, 1.17] |
-0.51 ± 1.33 [-0.94, -0.09] |
0.19 ± 1.44 [-0.27, 0.65] |
| Control | 66 | -0.11 ± 1.13 [-0.38, 0.17] |
0.33 ± 0.79 [0.14, 0.52] |
-0.08 ± 1.09 [-0.35, 0.18] |
0.14 ± 1.36 [-0.19, 0.47] |
| Type of treatment | Mono | Dual | Triple | |||
| Time point | Initial | Follow up | Initial | Follow up | Initial | Follow up |
| With 5 ALA /SFC | 7.29 | 6.81 | 8.53 | 7.56 | 8.81 | 7.74 |
| control | 7.33 | 6.71 | 7.99 | 7.30 | 8.41 | 7.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




