Submitted:
06 August 2025
Posted:
20 August 2025
You are already at the latest version
Abstract
Background/Objectives: Manual cardiothoracic ratio (CTR) measurement from chest X-rays remains widely used but is time-consuming and error-prone, particularly in low-resource clinical settings. This study presents a CPU-based deep learning model for automated CTR estimation designed for use in Sub-Saharan Africa. Methods: A U-Net segmentation model was trained on 3,000 anonymized chest radiographs to extract heart and thoracic contours. The trained model was deployed via a lightweight desktop application optimized for inference on standard CPUs. Results: The system achieved a mean absolute error (MAE) of 0.019 and an R2 value of 0.91 when compared to expert manual CTR measurements. Usability testing with local radiologists and radiographers revealed strong acceptance, highlighting the tool’s diagnostic and educational value. Conclusions: The findings suggest that accurate and efficient AI-based CTR estimation can be performed without GPU support, offering an accessible and cost-effective diagnostic aid for low- and middle-income healthcare systems.
Keywords:
1. Introduction
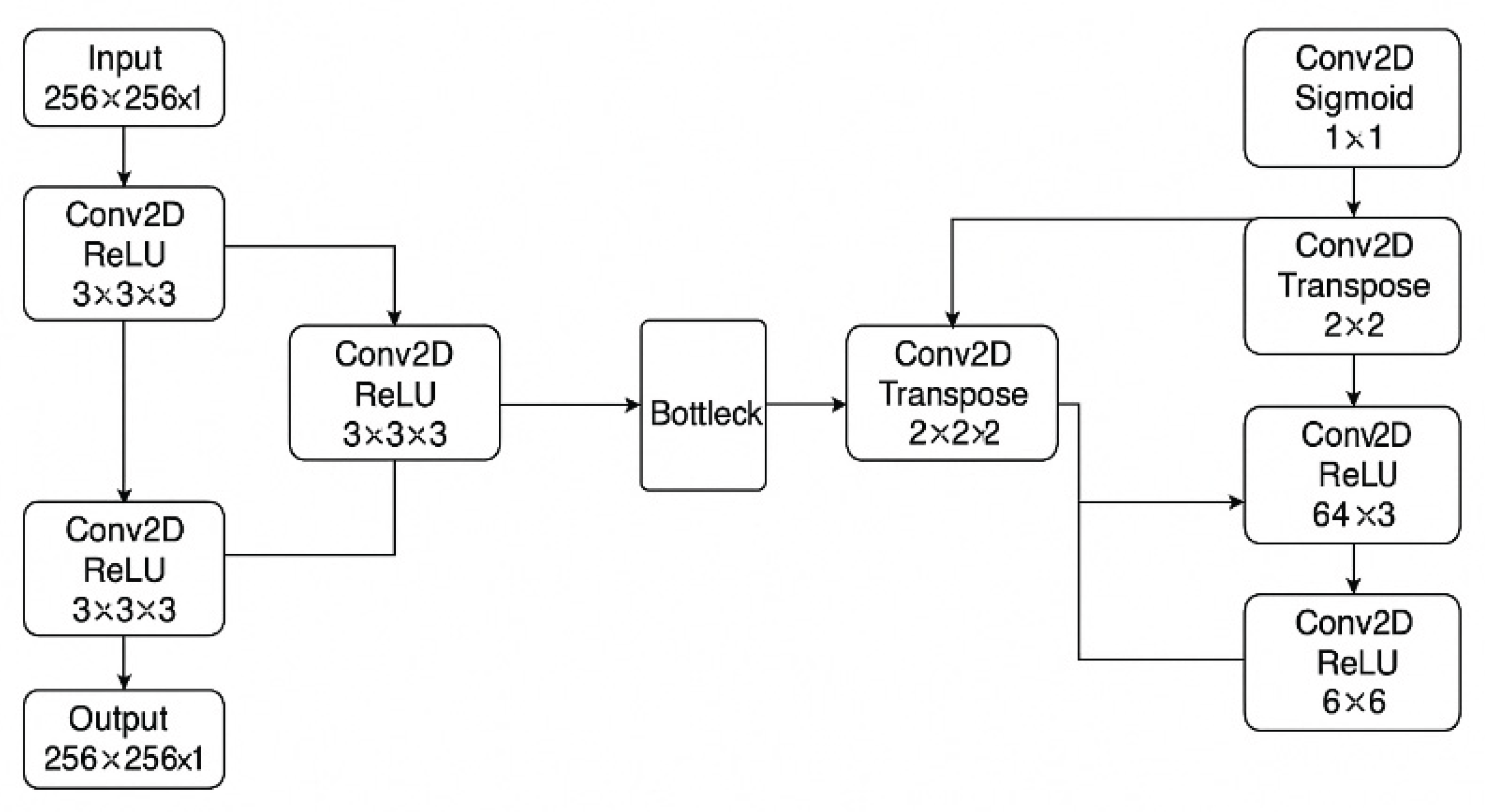
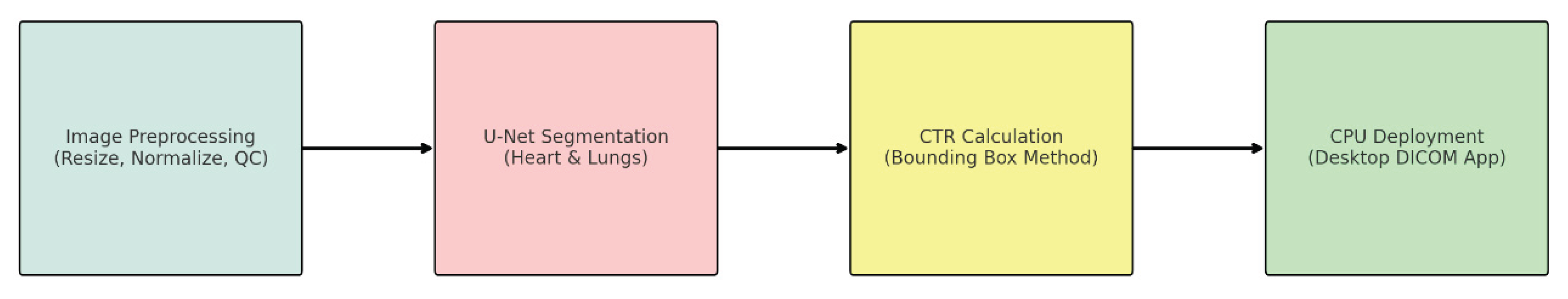
2. Materials and Methods
2.1. Study Design and Setting
2.2. Data Collection and Preprocessing
- Adequate inspiration, defined by visualization of at least nine posterior ribs;
- Proper patient positioning, indicated by symmetrical clavicles and centrally aligned spinous processes;
- Absence of significant rotation or artefacts that could affect accurate cardiothoracic ratio (CTR) estimation.
- Annotation workload limitations: Due to the labor-intensive nature of manual segmentation and limited expert availability, only 3000 images could be fully annotated and validated within the study timeline.
- Quality assurance filtering: Some images were excluded due to subtle artefacts, poor contrast, or suboptimal positioning that could compromise segmentation quality.
- Radiologist validation prioritization: The final 3000 images represent the highest-quality cases that passed expert validation, ensuring a reliable dataset for model development.
2.3. Model Development
| Model | Training Phase | Accuracy (%) | Recall (%) | Specificity (%) | AUC-ROC | Notes |
|---|---|---|---|---|---|---|
| Heart Segmentation | Initial (15 epochs) | 95.85 | 78.54 | — | 0.8847 | Baseline model |
| Fine-tuned (2×20 epochs) | 98.54 | 91.42 | — | 0.9310 | Selective fine-tuning on challenging cases | |
| Lung Segmentation | Initial | 71.18 | 88.52 | — | 0.7879 | Baseline model |
| Final (20 epochs) | 97.66 | 97.10 | 98.06 | 0.9758 | Improved with fine-tuning and data augmentation |
2.4. Cardiothoracic Ratio Calculation
2.5. Model Evaluation Metrics
2.6. Deployment and Prototype Application
2.7. Data and Code Availability
2.8. Generative AI Disclosure
2.9. Ethical Approval
3. Results
3.1. Dataset Characteristics
3.2. Segmentation Performance
-
Dice Similarity Coefficient (DSC):
- ○
- Heart segmentation: 0.87 ± 0.04
- ○
- Lung segmentation: 0.93 ± 0.02
-
Intersection over Union (IoU):
- ○
- Heart: 0.79 ± 0.05
- ○
- Lungs: 0.88 ± 0.03
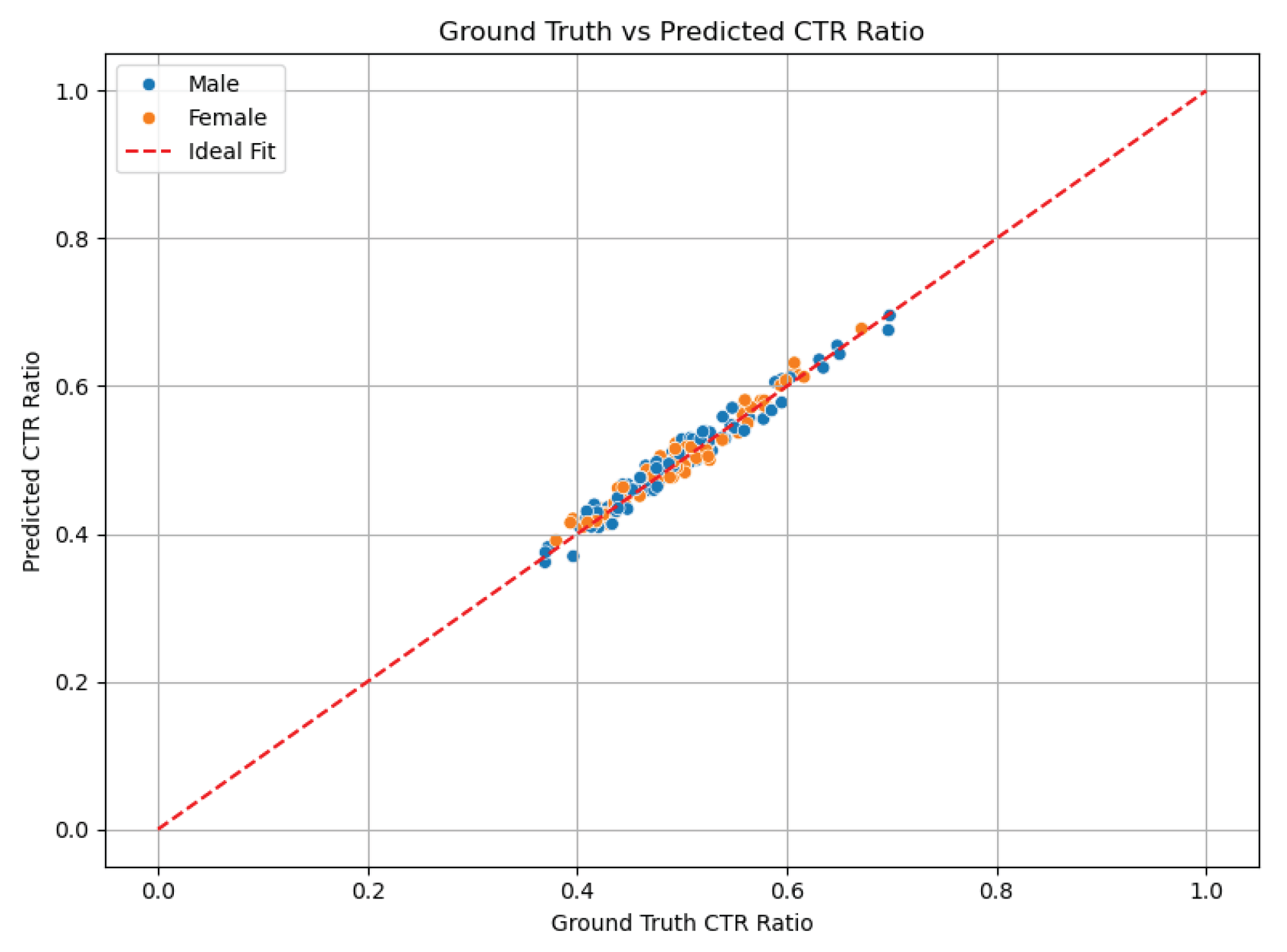
3.3. CTR Prediction Accuracy
- Pearson correlation coefficient (r): 0.91, p < 0.001 — demonstrating a strong positive linear relationship between AI and manual measurements.
- Mean absolute error (MAE): 0.025
-
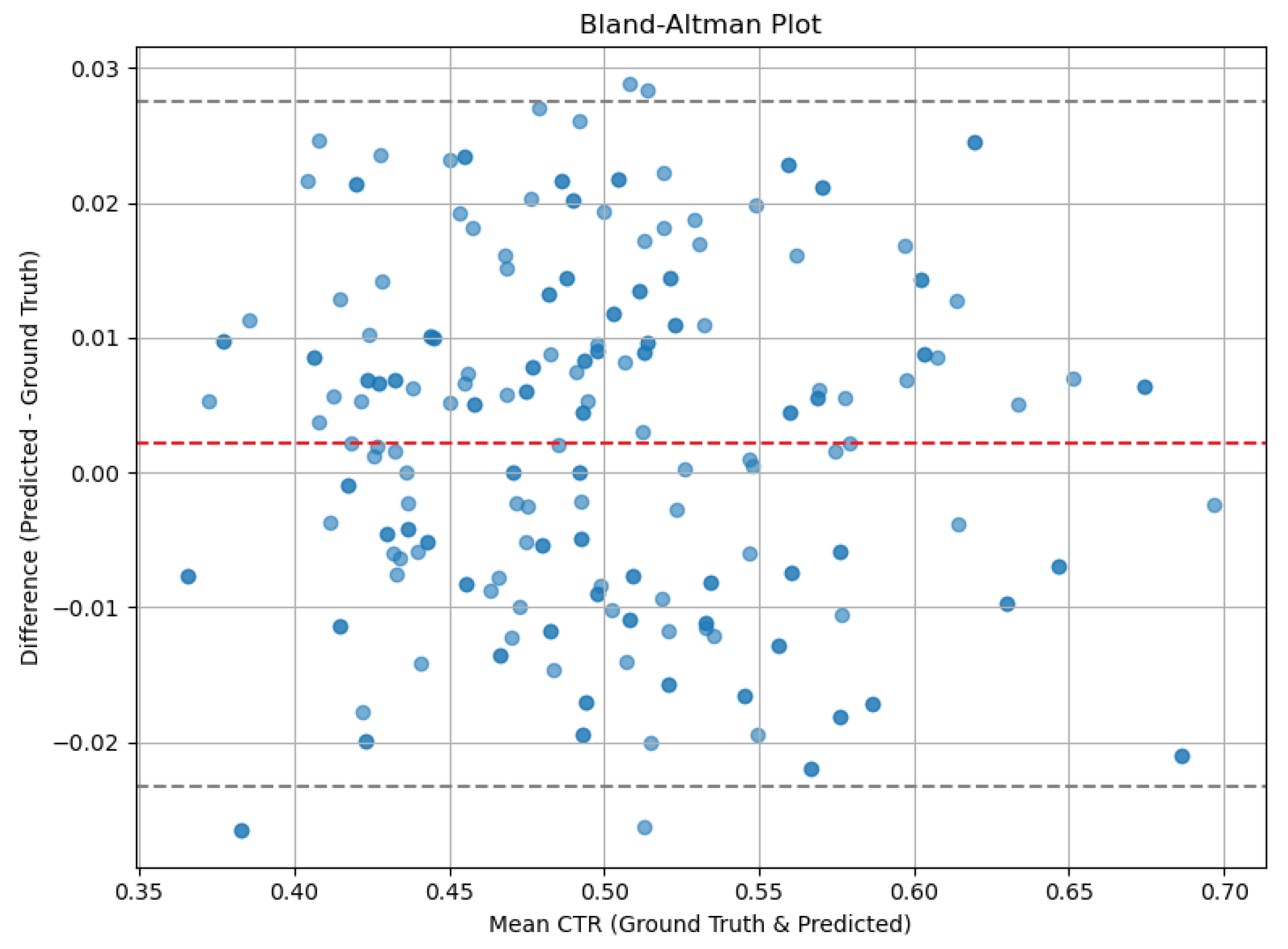
Bland–Altman analysis:
- ○
- Mean difference (bias): −0.01
- ○
- 95% limits of agreement: −0.06 to +0.04
3.4. Clinical Threshold Detection
- Sensitivity: 90.2%
- Specificity: 94.7%
- Accuracy: 92.4%
3.5. Prototype Application Demonstration
3.6. Expert Evaluation of the Demonstration Interface
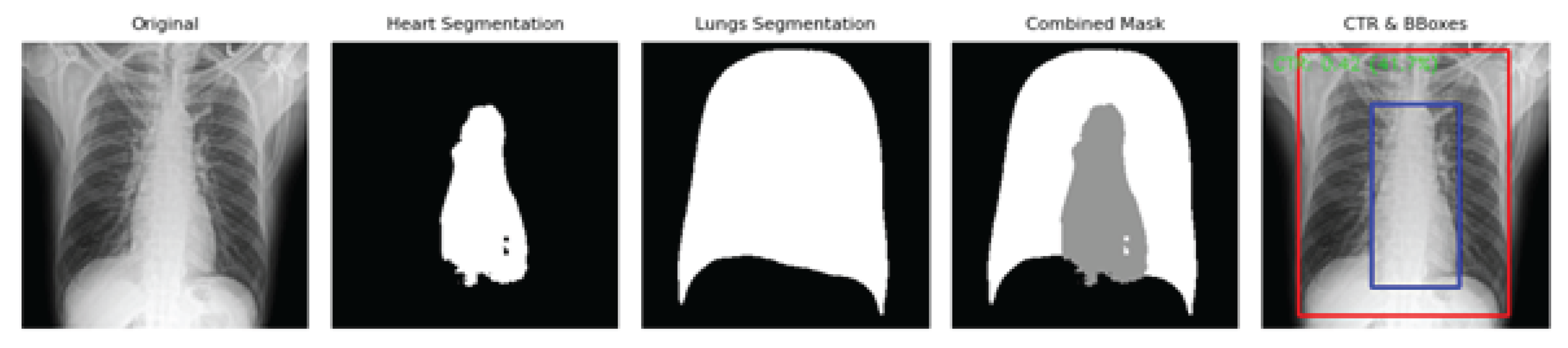
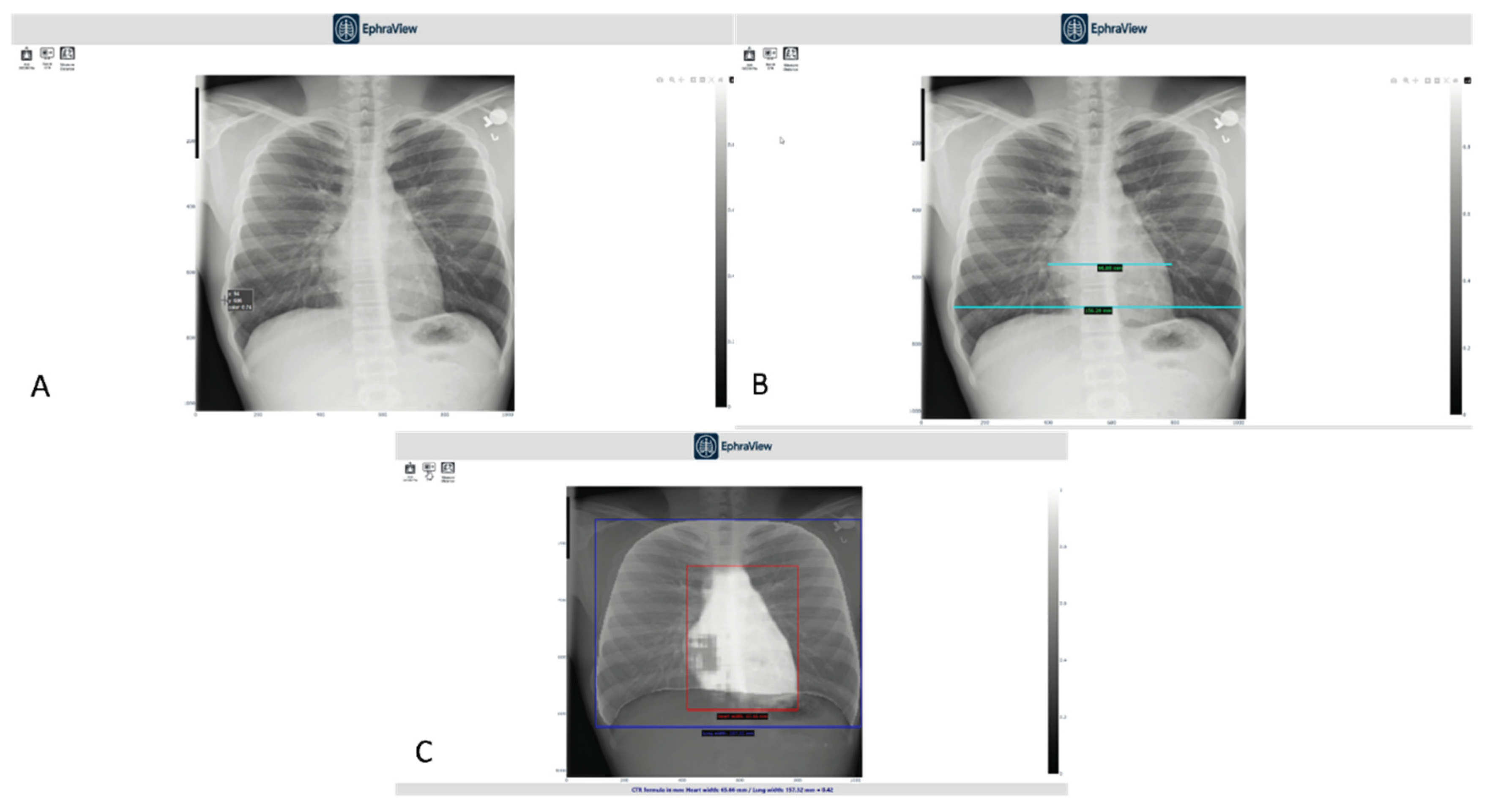
- Panel A shows the manual CTR measurement using traditional PACS tools.
- Panel B demonstrates thoracic width measurement with standard digital calipers, used by experts as a reference.
- Panel C displays the AI-generated overlay with bounding boxes: red for heart width and blue for thoracic width, alongside an automatically computed CTR.
- A – Manual measurement interface using standard PACS tools for cardiac width estimation.
- B – Manual thoracic width measurement using digital calipers, performed by a radiologist for reference CTR computation.
- C – AI-generated segmentation overlay with bounding boxes for heart (red) and thorax (blue), including automatically computed CTR displayed at the bottom.
| Evaluation Item | % Positive (n) | % Neutral (n) | Mean Rating (1–5) |
|---|---|---|---|
| Demonstration was clear and easy to follow | 89% (8) | 11% (1) | 4.6 |
| Manual measurement reflected good radiographic practice | 78% (7) | 22% (2) | 4.3 |
| Interface was user-friendly and precise | 78% (7) | 22% (2) | 4.2 |
| AI estimation was accurate compared to manual | 67% (6) | 33% (3) | 4.1 |
| AI segmentation of heart and lungs was acceptable | 78% (7) | 22% (2) | 4.3 |
| AI measurement is clinically useful in its current form | 67% (6) | 33% (3) | 4.1 |
| Application is valuable for clinical or educational settings | 89% (8) | 11% (1) | 4.5 |
| Recommendation for further development or pilot testing | 100% (9) | 0% (0) | 4.8 |
4. Discussion
4.1. Limitations and Future Work
- Larger, multi-center datasets across varied demographic and technical settings
- Expansion to mobile-based deployment
- Integration into PACS and teleradiology platforms
- Prospective clinical trials to evaluate diagnostic impact
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| AUC | Area Under the Curve |
| CNN | Convolutional Neural Network |
| CTR | Cardiothoracic Ratio |
| DICOM | Digital Imaging and Communications in Medicine |
| DL | Deep Learning |
| IRB | Institutional Review Board |
| PA | Posteroanterior |
| ROC | Receiver Operating Characteristic |
| U-Net | U-shaped Convolutional Network |
| X-ray | X-radiation |
References
- Truszkiewicz, K.; Poręba, R.; Gać, P. Radiological cardiothoracic ratio in evidence based medicine. J. Clin. Med. 2021, 10, 2016. [CrossRef]
- Grotenhuis, H.B.; Zhou, C.; Tomlinson, G.; Isaac, K.V.; Seed, M.; Grosse-Wortmann, L.; Yoo, S.J. Cardiothoracic ratio on chest radiograph in pediatric heart disease: correlation with heart volumes at magnetic resonance imaging. Pediatr. Radiol. 2015, 45, 1616–1623. [CrossRef]
- Sahin, H.; Chowdhry, D.N.; Olsen, A.; Nemer, O.; Wahl, L. Is there any diagnostic value of anteroposterior chest radiography in predicting cardiac chamber enlargement? Int. J. Cardiovasc. Imaging 2019, 35, 195–206. [CrossRef]
- Anjuna, R.; Paulius, S.; Manuel, G.G.; Audra, B.; Jurate, N.; Monika, R. Diagnostic value of cardiothoracic ratio in patients with non-ischemic cardiomyopathy: comparison to cardiovascular magnetic resonance imaging. Curr. Probl. Diagn. Radiol. 2024, 53, 353–358. [CrossRef]
- Chamveha, I.; Promwiset, T.; Tongdee, T.; Saiviroonporn, P.; Chaisangmongkon, W. Automated cardiothoracic ratio calculation and cardiomegaly detection using deep learning approach. arXiv 2020, arXiv:2002.07468. [CrossRef]
- Gupte, T.; Niljikar, M.; Gawali, M.; Kulkarni, V.; Kharat, A.; Pant, A. Deep learning models for calculation of cardiothoracic ratio from chest radiographs for assisted diagnosis of cardiomegaly. arXiv 2021, arXiv:2101.07606. [CrossRef]
- Ajmera, P.; Kharat, A.; Gupte, T.; Pant, R.; Kulkarni, V.; Duddalwar, V.; Lamghare, P. Observer performance evaluation of the feasibility of a deep learning model to detect cardiomegaly on chest radiographs. Acta Radiol. Open 2022, 11, 20584601221107345. [CrossRef]
- Chou, H.-H.; Lin, J.-Y.; Shen, G.-T.; Huang, C.-Y. Validation of an Automated Cardiothoracic Ratio Calculation for Hemodialysis Patients. Diagnostics 2023, 13, 1376. [CrossRef]
- Wang, X.; Peng, Y.; Lu, L.; Lu, Z.; Bagheri, M.; Summers, R.M. ChestX-ray8: Hospital-scale chest X-ray database and benchmarks on weakly-supervised classification and localization of common thorax diseases. Proc. IEEE Conf. Comput. Vis. Pattern Recognit. 2017, 2017, 3462–3471. [CrossRef]
- Ajmera, P.; Kharat, A.; Gupte, T.; Pant, R.; Kulkarni, V.; Duddalwar, V.; Lamghare, P. Observer performance evaluation of the feasibility of a deep learning model to detect cardiomegaly on chest radiographs. Acta Radiol. Open 2022, 11, 20584601221107345. [CrossRef]
- Tang, Y.-X.; Tang, Y.-B.; Peng, Y.; Yan, K.; Harrison, A.P.; Bagheri, M.; Summers, R.M. Automated abnormality classification of chest radiographs using deep convolutional neural networks. NPJ Digit. Med. 2020, 3, 70. [CrossRef]
- Arora, R.; Banerjee, I.; Soni, A.; Sivasankaran, S.; Aggarwal, S.; Mahajan, V.; Arora, A. Developing AI for low-resource settings: a case study on a real-time offline model for tuberculosis detection. BMJ Health Care Inform. 2022, 29, e100496. [CrossRef]
- Ayeni, F.A.; Adedeji, A.T.; Agwuna, E.S.; Omoregbe, N.; Ojo, O.; Fakunle, O.O. Assessing the readiness of low-resource hospitals for artificial intelligence integration in Sub-Saharan Africa. Health Technol. 2023, 13, 241–251. [CrossRef]
- Kazeminia, S.; Khaki, M.; Asadi, A.; Jolfaei, A.; Ghassemi, N.; Parsaei, H.; Rajaraman, S. Deep learning for medical image analysis in low-resource settings: a review. Comput. Biol. Med. 2023, 158, 106874. [CrossRef]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. In Medical Image Computing and Computer-Assisted Intervention (MICCAI); Springer: Cham, Switzerland, 2015; Vol. 9351, pp. 234–241. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




