Submitted:
16 August 2025
Posted:
20 August 2025
Read the latest preprint version here
Abstract

Keywords:
Fundamentals and Formula of Decision Curve Analysis
Interpreting a Decision Curve
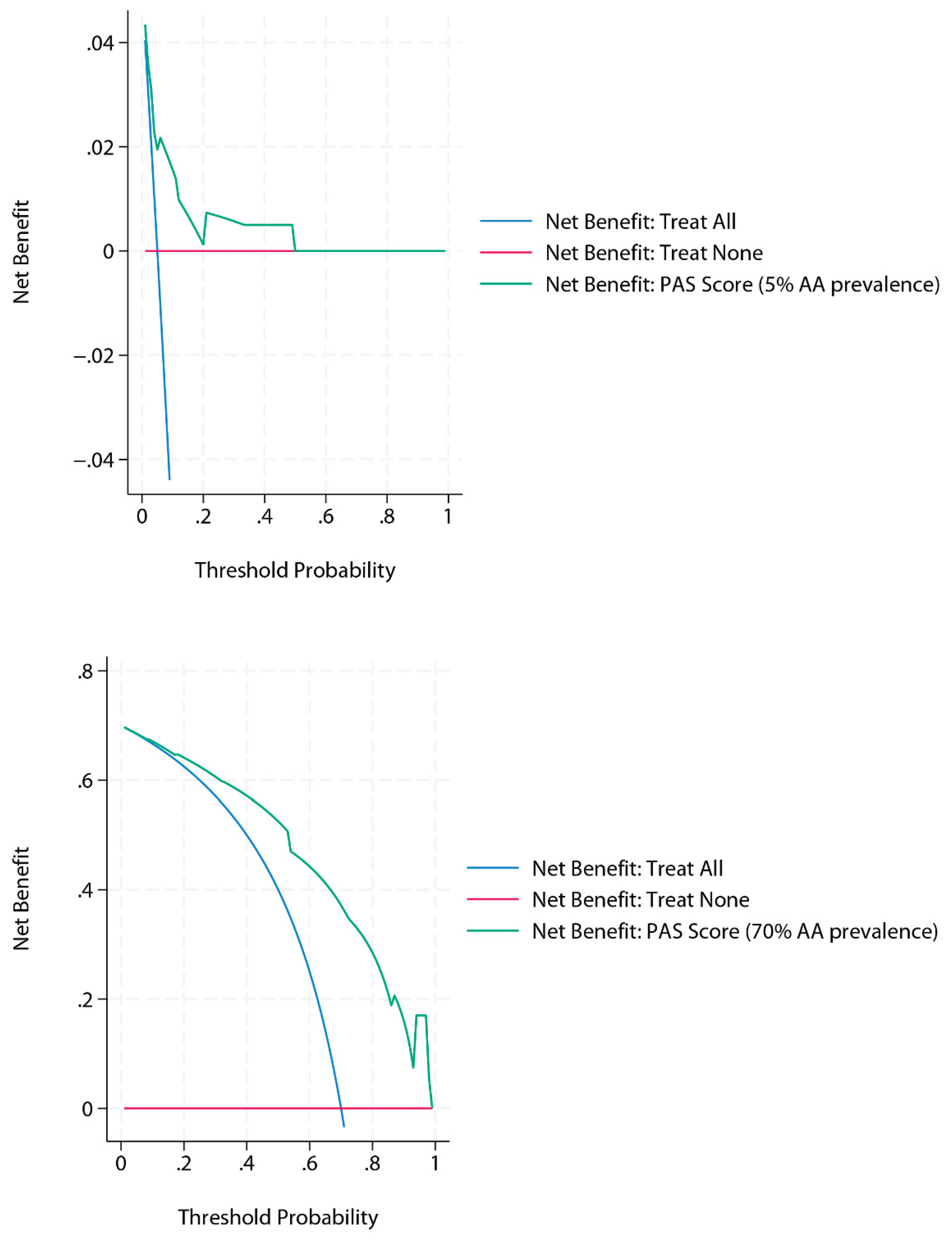
DCA and Prevalence
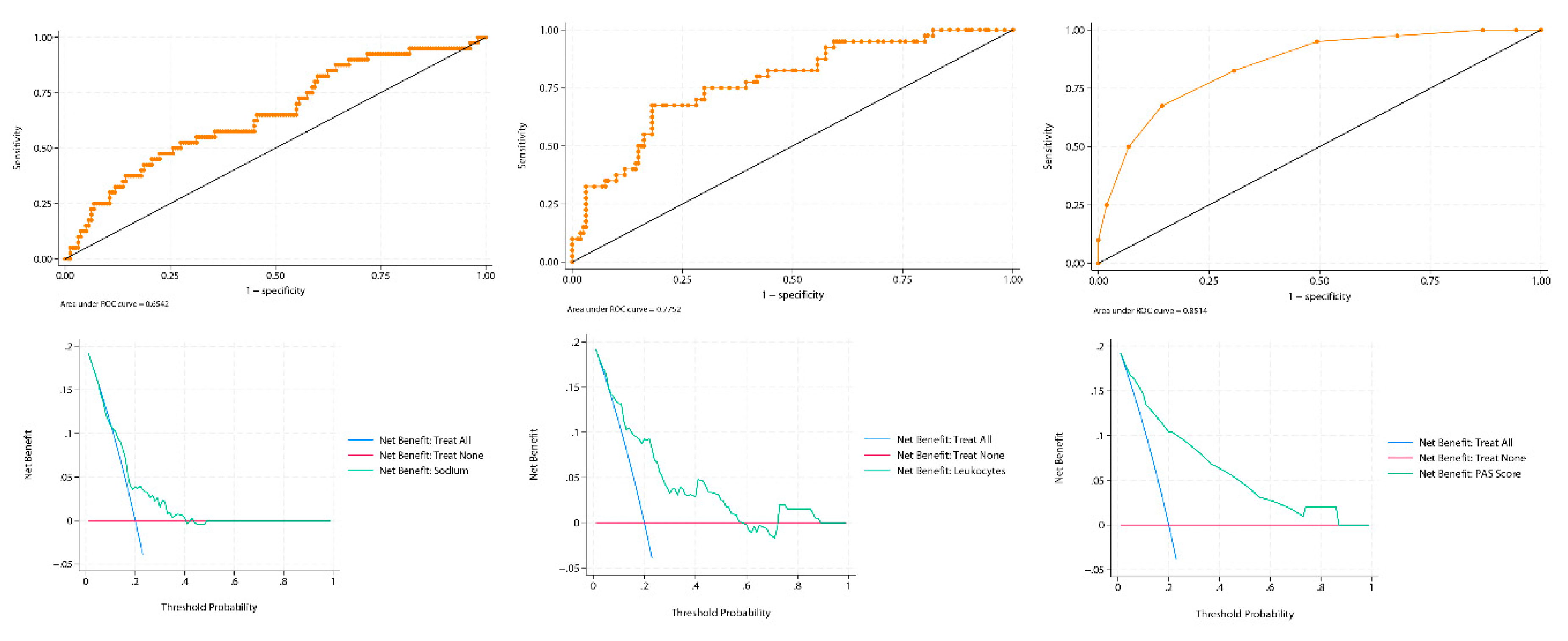
An Applied Example
Strengths and Limitations of DCA
Special Considerations: Overfitting, Binary Predictors, and Calibration
Conclusions
Supplementary Materials
Original Work
AI Use Disclosure
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Ethical Statement
References
- Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making. 2006 Nov-Dec;26(6):565-74. [CrossRef] [PubMed]
- Vickers A J, Van Calster B, Steyerberg E W. Net benefit approaches to the evaluation of prediction models, molecular markers, and diagnostic tests BMJ 2016; 352 :i6. [CrossRef]
- Bhatt M, Joseph L, Ducharme FM, Dougherty G, McGillivray D. Prospective validation of the pediatric appendicitis score in a Canadian pediatric emergency department. Acad Emerg Med. 2009 Jul;16(7):591-6. [CrossRef] [PubMed]
- Kottakis G, Bekiaridou K, Roupakias S, Pavlides O, Gogoulis I, Kosteletos S, Dionysis TN, Marantos A, Kambouri K. The Role of Hyponatremia in Identifying Complicated Cases of Acute Appendicitis in the Pediatric Population. Diagnostics (Basel). 2025 May 30;15(11):1384. [CrossRef] [PubMed]
- Duman L, Karaibrahimoğlu A, Büyükyavuz Bİ, Savaş MÇ. Diagnostic Value of Monocyte-to-Lymphocyte Ratio Against Other Biomarkers in Children With Appendicitis. Pediatr Emerg Care. 2022 Feb 1;38(2):e739-e742. [CrossRef] [PubMed]
- Vickers AJ, Cronin AM, Elkin EB, Gonen M. Extensions to decision curve analysis, a novel method for evaluating diagnostic tests, prediction models and molecular markers. BMC Med Inform Decis Mak. 2008 Nov 26;8:53. [CrossRef] [PubMed]
- Van Calster B, Wynants L, Verbeek JFM, Verbakel JY, Christodoulou E, Vickers AJ, Roobol MJ, Steyerberg EW. Reporting and Interpreting Decision Curve Analysis: A Guide for Investigators. Eur Urol. 2018 Dec;74(6):796-804. [CrossRef] [PubMed]
- Kerr KF, Brown MD, Zhu K, Janes H. Assessing the Clinical Impact of Risk Prediction Models With Decision Curves: Guidance for Correct Interpretation and Appropriate Use. J Clin Oncol. 2016 Jul 20;34(21):2534-40. [CrossRef] [PubMed]
- Steyerberg EW, Vickers AJ, Cook NR, Gerds T, Gonen M, Obuchowski N, Pencina MJ, Kattan MW. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology. 2010 Jan;21(1):128-38. [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



