Submitted:
15 August 2025
Posted:
20 August 2025
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
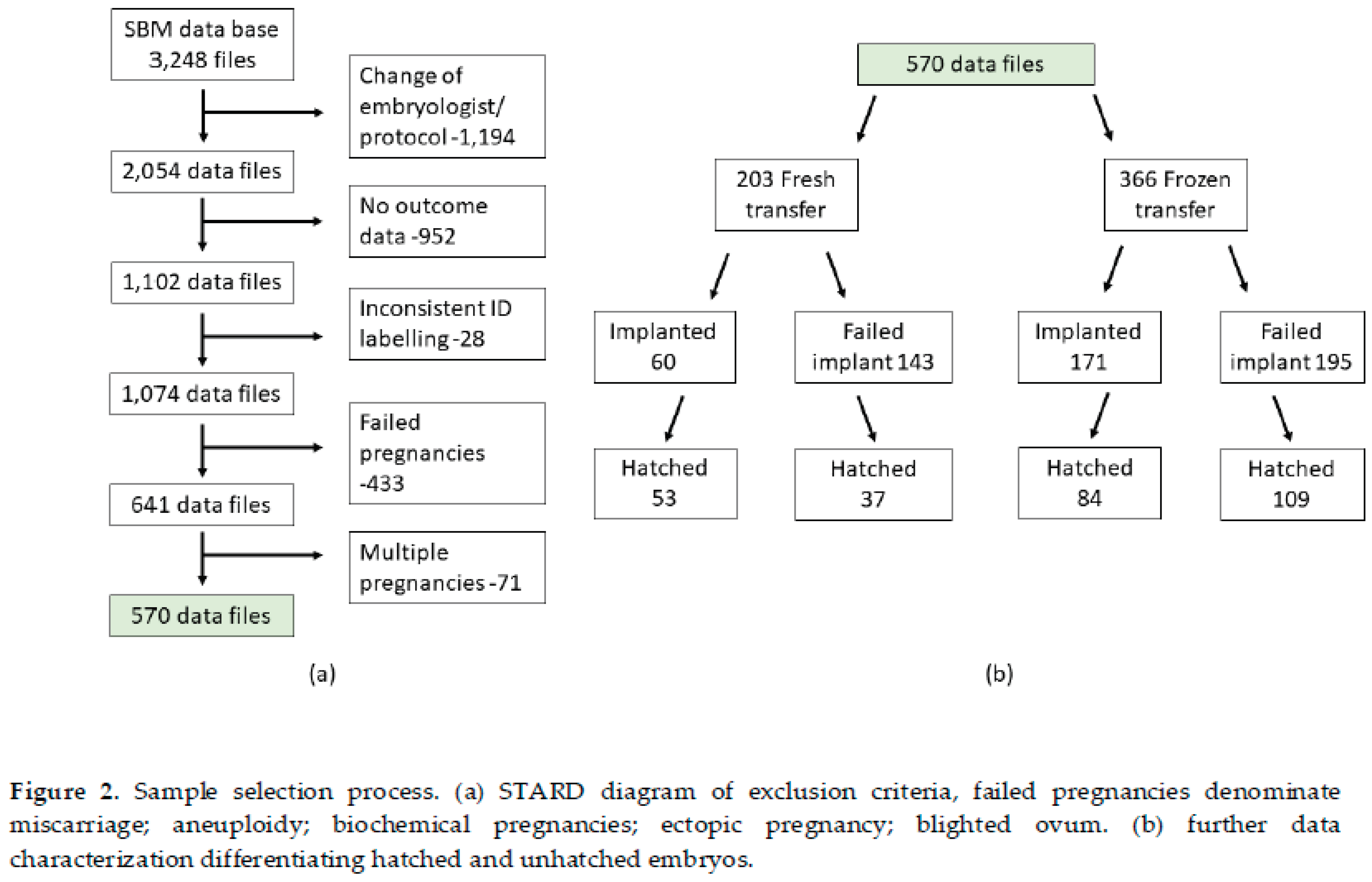
2.1. Samples
2.2. MALDI-ToF Mass Spectrometry
2.3. Bioinformatics
2.4. Data Modelling
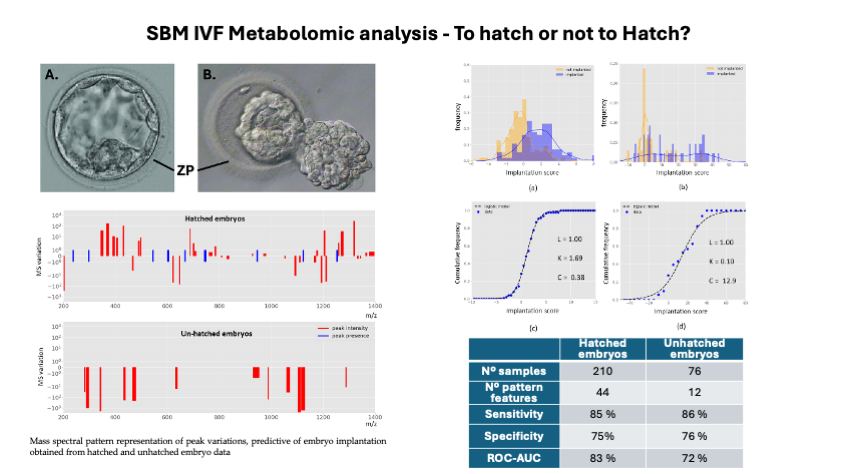
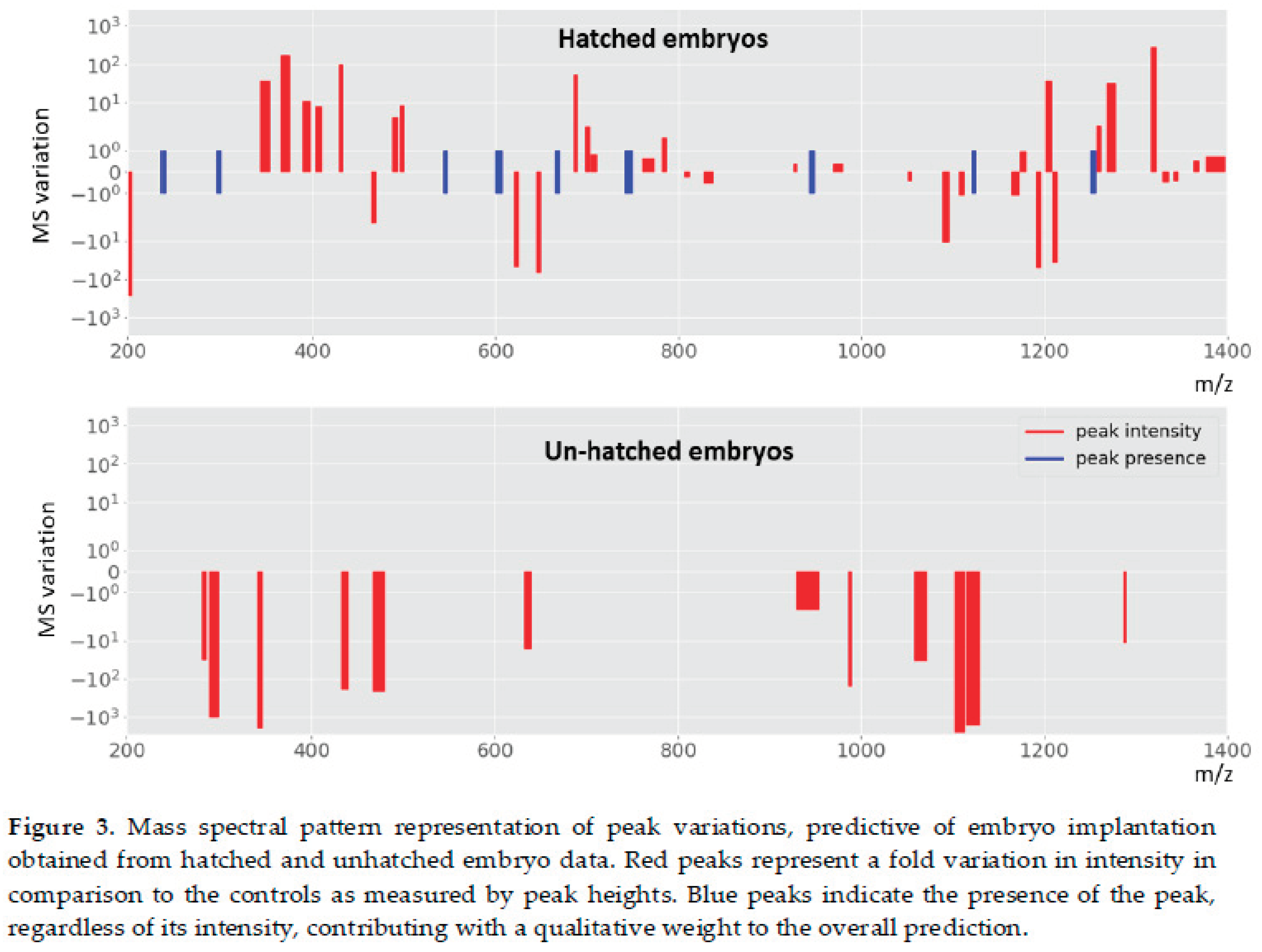
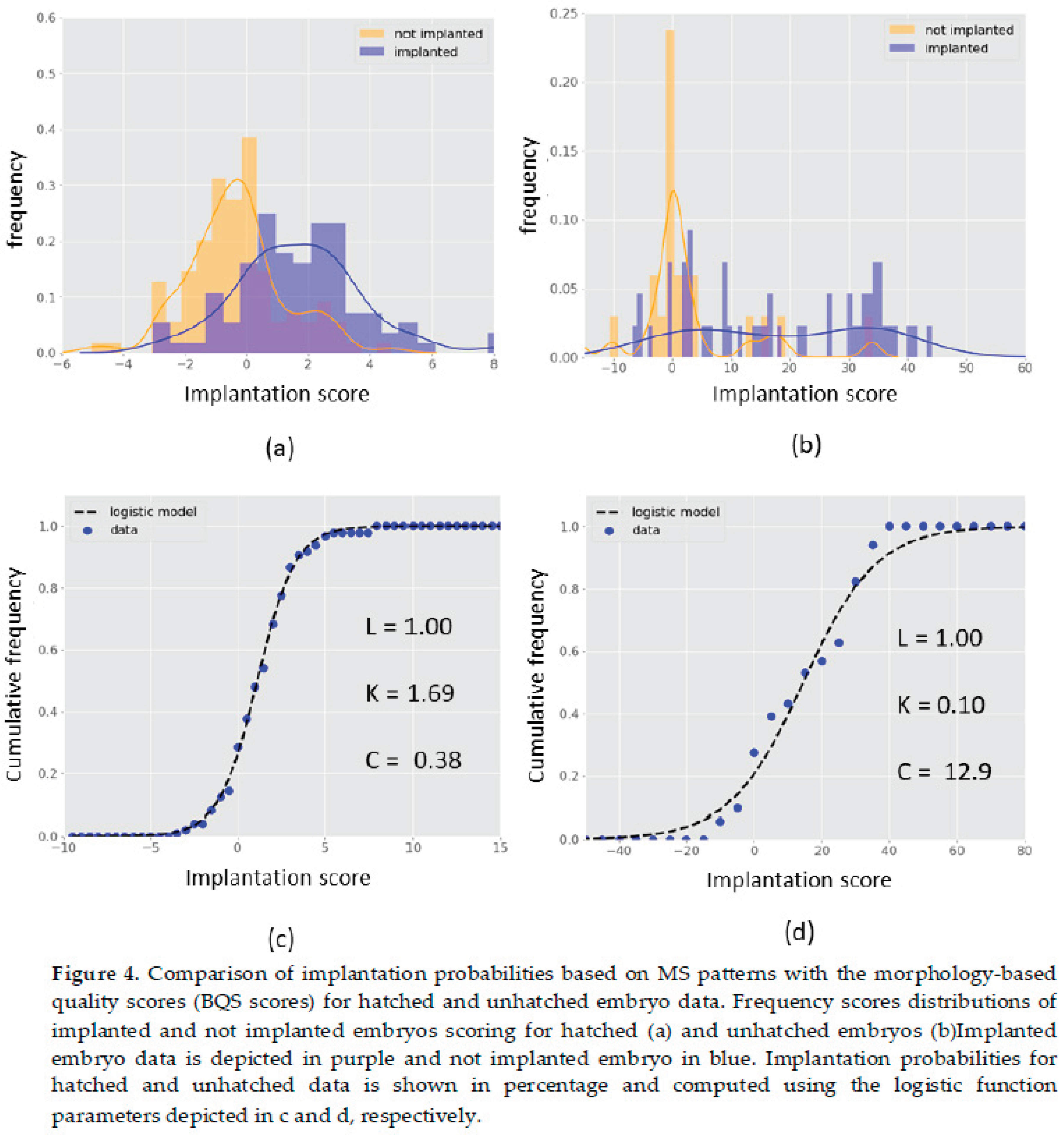
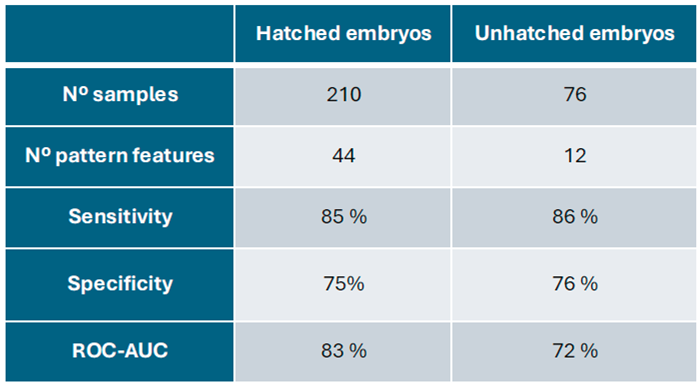
3. Results
4. Discussion
Conflicts of Interest
References
- Roberts, S.A.; Wilkinson, J.; Vail, A.; Brison, D.R. Does PGT-A improve assisted reproduction treatment success rates: what can the UK Register data tell us? J. Assist. Reprod. Genet. 2022, 39, 2547–2554. [Google Scholar] [CrossRef]
- The use of preimplantation genetic testing for aneuploidy: a committee opinion. Fertil. Steril. 2024, 122, 421–434. [CrossRef]
- Zmuidinaite, R.; Sharara, F.I.; Iles, R.K. Current Advancements in Noninvasive Profiling of the Embryo Culture Media Secretome. Int. J. Mol. Sci. 2021, 22, 2513. [Google Scholar] [CrossRef]
- F. I. Sharara, S.A. F. I. Sharara, S.A. Butler, R.J. Pais, R. Zmuidinaite, S. Keshavarez, R.K. Iles “BESST, a Non-Invasive compuational Tool for Embryo selection using mas spectral profiling of embryo culture media. EMJ Repro Health, 5(1):59-60, 2019 https://www.emjreviews.com/reproductive-health/abstract/besst-a-non-invasive-computational-tool-for-embryo-selection-using-mass-spectral-profiling-of-embryo-culture-media/.
- Tesarik, J. Noninvasive Biomarkers of Human Embryo Developmental Potential. Int. J. Mol. Sci. 2025, 26, 4928. [Google Scholar] [CrossRef]
- Simopoulou, M.; Sfakianoudis, K.; Rapani, A.; Giannelou, P.; Anifandis, G.; Bolaris, S.; Pantou, A.; Lambropoulou, M.; Pappas, A.; Deligeoroglou, E.; et al. Considerations Regarding Embryo Culture Conditions: From Media to Epigenetics. Vivo 2018, 32, 451–460. [Google Scholar] [CrossRef]
- Gardner, D.K.; Kelley, R.L. Impact of the IVF laboratory environment on human preimplantation embryo phenotype. J. Dev. Orig. Health Dis. 2017, 8, 418–435. [Google Scholar] [CrossRef] [PubMed]
- E. S. Litscher and P. M. Wassarman, “Zona pellucida proteins, fibrils, and matrix,” Annu. Rev. Biochem., vol. 89, pp. 695–715, 2020.
- Sathananthan, H.; Gunasheela, S.; Menezes, J. Mechanics of human blastocyst hatching in vitro. Reprod. Biomed. Online 2003, 7, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Malter, H.; Fehilly, C.; Wright, G.; Elsner, C.; Kort, H.; Massey, J. Implantation of embryos after partial opening of oocyte zona pellucida to facilitate sperm penetration, Lancet 1988, 332, 162. [Google Scholar] [CrossRef]
- Alteri, A.; Viganò, P.; Abu Maizar, A.; Jovine, L.; Giacomini, E.; Rubino, P. Revisiting embryo assisted hatching approaches: a systematic review of the current protocols. J. Assist. Reprod. Genet. 2018, 35, 367–391. [Google Scholar] [CrossRef]
- Seif, M.; Edi-Osagie, E.; Farquhar, C.; Hooper, L.; Blake, D.; McGinlay, P.; Seif, M. Assisted hatching on assisted conception (IVF & ICSI). Cochrane Database Syst. Rev. 2005, 4. [Google Scholar]
- Lacey, L.; Hassan, S.; Franik, S.; Seif, M.W.; Akhtar, M.A. ; Cochrane Gynaecology and Fertility Group Assisted hatching on assisted conception (in vitro fertilisation (IVF) and intracytoplasmic sperm injection (ICSI)). Cochrane Database Syst. Rev. 2021, 2021, CD001894. [Google Scholar] [CrossRef]
- Hammadeh, M.E.; Fischer-Hammadeh, C.; Ali, K.R. Assisted hatching in assisted reproduction: a state of the art. J. Assist. Reprod. Genet. 2010, 28, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Iles, R.K.; Sharara, F.I.; Zmuidinaite, R.; Abdo, G.; Keshavarz, S.; Butler, S.A. Secretome profile selection of optimal IVF embryos by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. J. Assist. Reprod. Genet. 2019, 36, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Gorry, P.A. General least-squares smoothing and differentiation by the convolution (Savitzky-Golay) method. Anal. Chem. 1990, 62, 570–573. [Google Scholar] [CrossRef]
- Pais, R.J.; Sharara, F.; Zmuidinaite, R.; Butler, S.; Keshavarz, S.; Iles, R. Bioinformatic identification of euploid and aneuploid embryo secretome signatures in IVF culture media based on MALDI-ToF mass spectrometry. J. Assist. Reprod. Genet. 2020, 37, 2189–2198. [Google Scholar] [CrossRef]
- Rehman, K.S.; Bukulmez, O.; Langley, M.; Carr, B.R.; Nackley, A.C.; Doody, K.M.; Doody, K.J. Late stages of embryo progression are a much better predictor of clinical pregnancy than early cleavage in intracytoplasmic sperm injection and in vitro fertilization cycles with blastocyst-stage transfer. Fertil. Steril. 2007, 87, 1041–1052. [Google Scholar] [CrossRef]
- Almagor, M.; Levin, Y.; Amiran, R.H.; Fieldust, S.; Harir, Y.; Or, Y.; Shoham, Z. Spontaneous in vitro hatching of the human blastocyst: the proteomics of initially hatching cells. Vitr. Cell. Dev. Biol. - Anim. 2020, 56, 859–865. [Google Scholar] [CrossRef]
- Paulson, R.J. Outcome of in vitro fertilization cycles with preimplantation genetic testing for aneuploidies: let’s be honest with one another. Fertil. Steril. 2019, 112, 1013–1014. [Google Scholar] [CrossRef]
- G. L. Schattman, “Preimplantation genetic testing for aneuploidy: It’s déjà vu all over again!,” Fertil. Steril., vol. 112, no. 6, pp. 1046–1047, 2019.
- Pagliardini, L.; Viganò, P.; Alteri, A.; Corti, L.; Somigliana, E.; Papaleo, E. Shooting STAR: reinterpreting the data from the 'Single Embryo TrAnsfeR of Euploid Embryo' randomized clinical trial. Reprod. Biomed. Online 2020, 40, 475–478. [Google Scholar] [CrossRef]
- Sanchez, T.; Seidler, E.A.; Gardner, D.K.; Needleman, D.; Sakkas, D. Will noninvasive methods surpass invasive for assessing gametes and embryos? Fertil. Steril. 2017, 108, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Abreu, C.M.; Thomas, V.; Knaggs, P.; Bunkheila, A.; Cruz, A.; Teixeira, S.R.; Alpuim, P.; Francis, L.W.; Gebril, A.; Ibrahim, A.; et al. Non-invasive molecular assessment of human embryo development and implantation potential. Biosens. Bioelectron. 2020, 157, 112144. [Google Scholar] [CrossRef]
- Cimadomo, D.; Fernandez, L.S.; Soscia, D.; Fabozzi, G.; Benini, F.; Cesana, A.; Canto, M.B.D.; Maggiulli, R.; Muzzì, S.; Scarica, C.; et al. Inter-centre reliability in embryo grading across several IVF clinics is limited: implications for embryo selection. Reprod. Biomed. Online 2022, 44, 39–48. [Google Scholar] [CrossRef]
- Syrkasheva, A.G.; Dolgushina, N.V.; Romanov, A.Y.; Burmenskaya, O.V.; Makarova, N.P.; Ibragimova, E.O.; Kalinina, E.A.; Sukhikh, G.T. Cell and genetic predictors of human blastocyst hatching success in assisted reproduction. Zygote 2017, 25, 631–636. [Google Scholar] [CrossRef]
- Shafei, R.A.; Syrkasheva, A.G.; Romanov, A.Y.; Makarova, N.P.; Dolgushina, N.V.; Semenova, M.L. Blastocyst hatching in humans. Russ. J. Dev. Biol. 2017, 48, 5–15. [Google Scholar] [CrossRef]
- L. Lefièvre, S. J. Conner, A. Salpekar, O. Olufowobi, P. Ashton, B. Pavlovic, W. Lenton, M. Afnan, I. A. Brewis, and M. Monk, “Four zona pellucida glycoproteins are expressed in the human,” Hum. Reprod., vol. 19, no. 7, pp. 1580–1586, 2004.
- Cohen, J.; Alikani, M.; Trowbridge, J.; Rosenwaks, Z. Implantation enhancement by selective assisted hatching using zona drilling of human embryos with poor prognosis. Hum. Reprod. 1992, 7, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Van Soom, A.; Wrathall, A.E.; Herrler, A.; Nauwynck, H.J. Is the zona pellucida an efficient barrier to viral infection? Reprod. Fertil. Dev. 2009, 22, 21–31. [Google Scholar] [CrossRef]
- Sellens, M.H.; Jenkinson, E.J. Permeability of the mouse zona pellucida to immunoglobulin. Reproduction 1975, 42, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Soupart, P.; Noyes, R.W. Sialic acid as a component of the zona pellucida of the mammalian ovum. Reproduction 1964, 8, 251–253. [Google Scholar] [CrossRef]
- Butler, S.A.; Luttoo, J.; Freire, M.O.T.; Abban, T.K.; Borrelli, P.T.A.; Iles, R.K. Human Chorionic Gonadotropin (hCG) in the Secretome of Cultured Embryos. Reprod. Sci. 2013, 20, 1038–1045. [Google Scholar] [CrossRef]
- Feichtinger, M.; Vaccari, E.; Carli, L.; Wallner, E.; Mädel, U.; Figl, K.; Palini, S.; Feichtinger, W. Non-invasive preimplantation genetic screening using array comparative genomic hybridization on spent culture media: a proof-of-concept pilot study. Reprod. Biomed. Online 2017, 34, 583–589. [Google Scholar] [CrossRef]
- Pais, R.J.; Iles, R.K.; Zmuidinaite, R. MALDI-ToF Mass spectra phenomic analysis for human disease diagnosis enabled by cutting-edge data processing pipelines and bioinformatics tools. Curr. Med. Chem. 2020, 27, 1–14. [Google Scholar] [CrossRef] [PubMed]
- E. Vargas, F. J. Esteban, and S. Altmäe, “Computational Approaches in Reproductomics,” in Reproductomics, Elsevier, 2018, pp. 347–383.
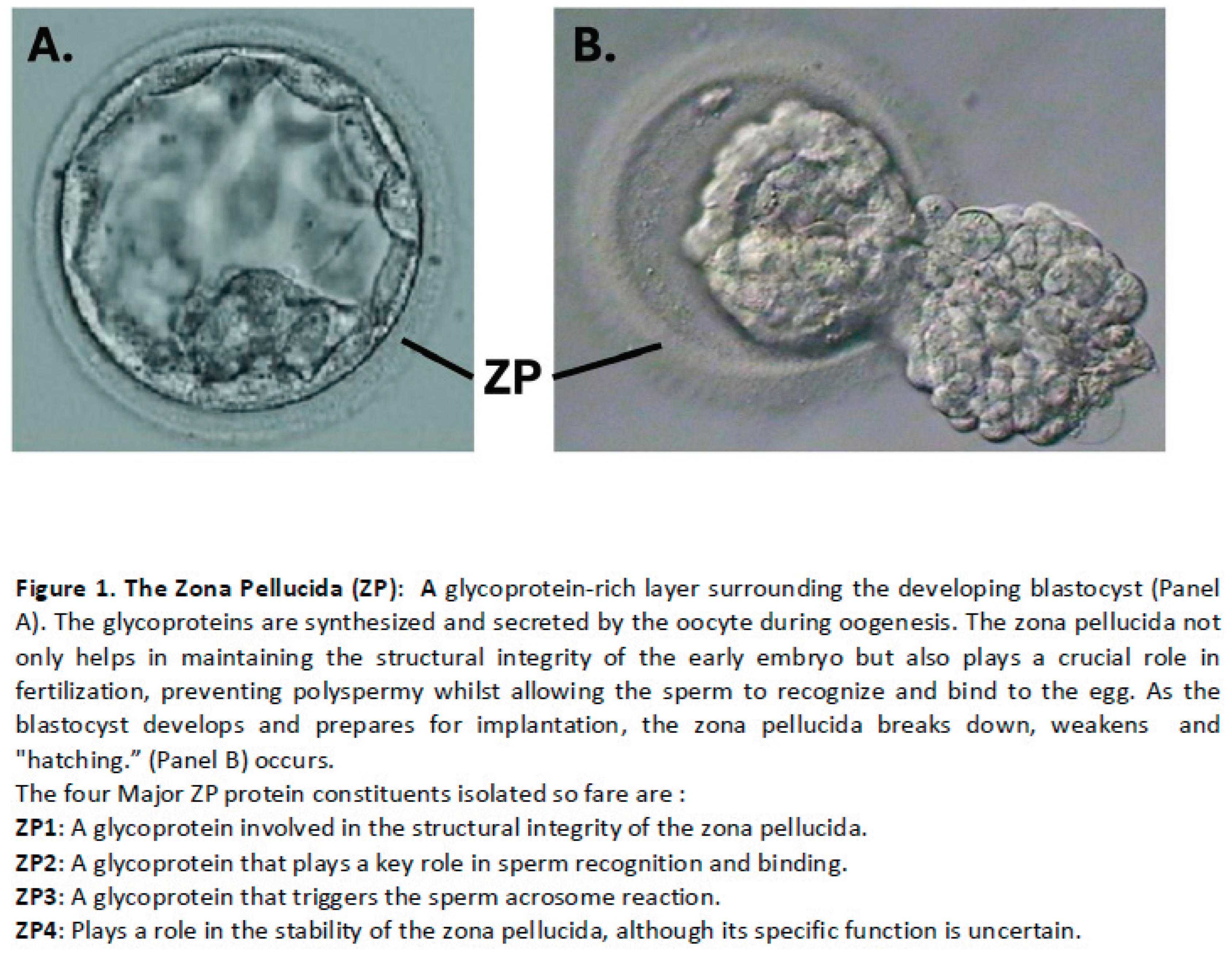
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




