Submitted:
18 August 2025
Posted:
19 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
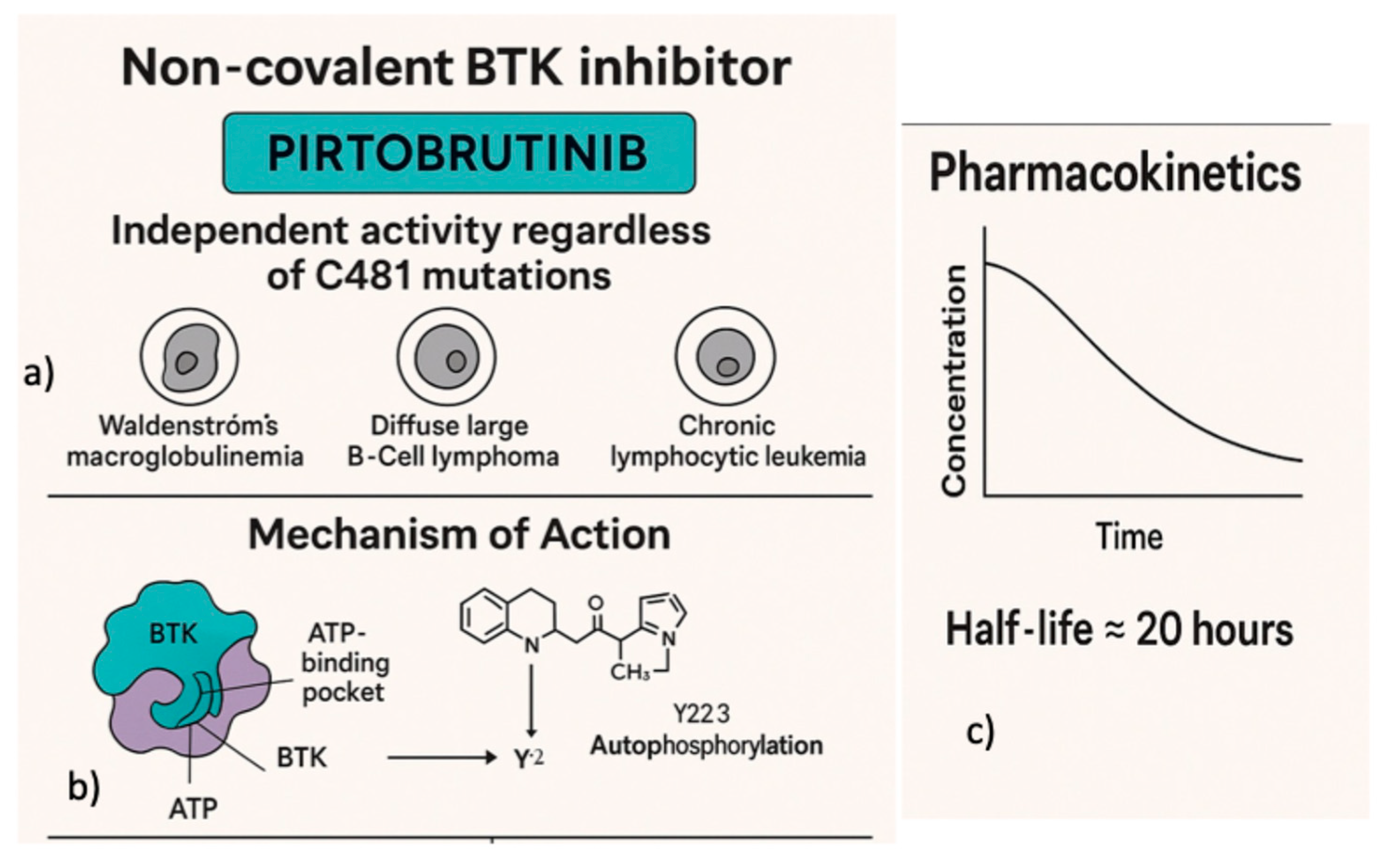
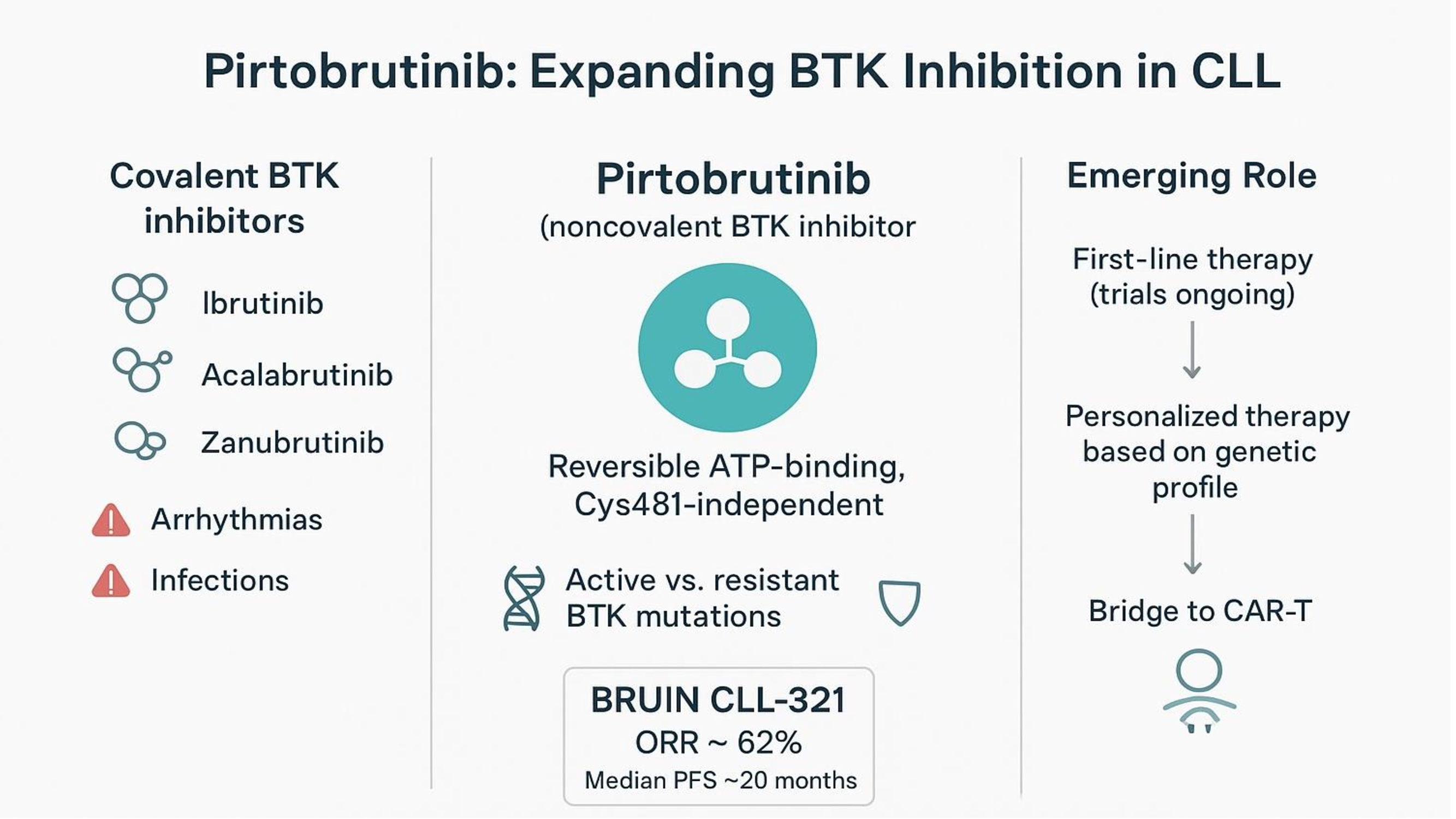
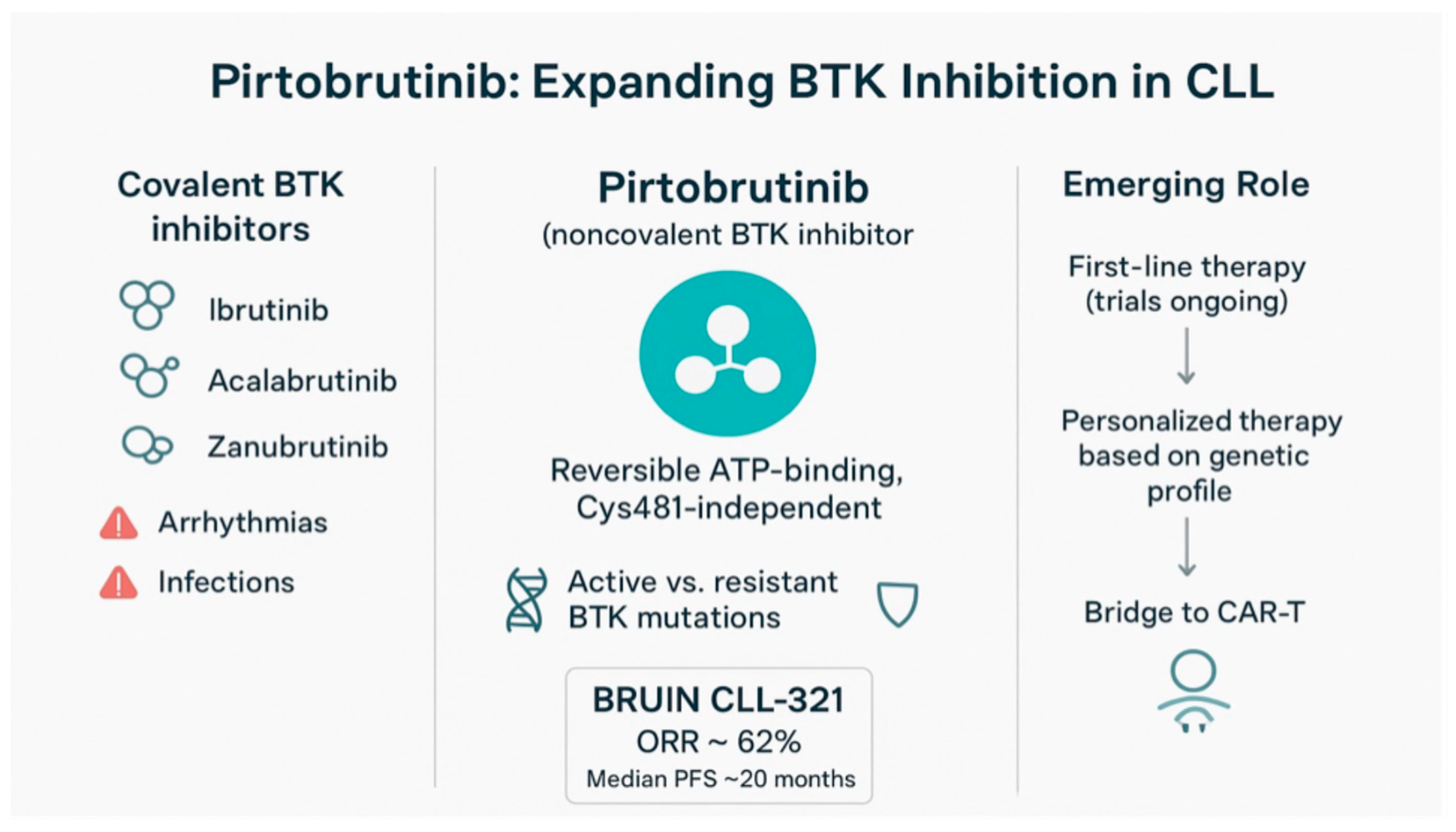
2. Pirtobrutinib Mechanism-of-Action and Pharmacology
3. Early Clinical Studies
4. The Phase 3 Trial BRUIN CLL-321
5. Pirtobrutinib in Patients in RICHTER Transformation
6. Pirtobrutinib in Fixed-Duration Regimens
7. Pirtobrutinib Resistance and the Strategic Integration in CLL Management
8. Improving BTKi-Safety with Pirtobrutinib
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- O’Brien S, Furman RR, Coutre SE, Sharman JP, Burger JA, Blum KA, Grant B, Richards DA, Coleman M, Wierda WG, et al. Ibrutinib as Initial Therapy for Elderly Patients With Chronic Lymphocytic Leukaemia or Small Lymphocytic Lymphoma: An Open-Label, Multicentre, Phase 1b/2 Trial,” Lancet Oncol. 2014 Jan;15(1):48-58. [CrossRef]
- Burger JA, Tedeschi A, Barr PM, Robak T, Owen C, Ghia P, Bairey O, Hillmen P, Bartlett NL, Li J, et al. Ibrutinib as Initial Therapy for Patients With Chronic Lymphocytic Leukemia, N Engl J Med. 2015 Dec 17;373(25):2425-2437.
- Burger JA, Barr PM, Robak T Prof, Owen C, Tedeschi A, Sarma A, Patten PE, Grosicki S, McCarthy H Dr, Offner F, et al. Final analysis of the RESONATE-2 study: up to 10 years of follow-up of first-line ibrutinib treatment for CLL/SLL. Blood. 2025 Jul 30:blood.2024028205. Online ahead of print. [CrossRef]
- Shanafelt, T.D.; Wang, X.V.; Kay, N.E.; Hanson, C.A.; O’Brien, S.; Barrientos, J.; Jelinek, D.F.; Braggio, E.; Leis, J.F.; Zhang, C.C.; et al. Ibrutinib- Rituximab or Chemoimmunotherapy for Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2019, 381, 432–443.
- Woyach, J.A.; Ruppert, A.S.; Heerema, N.A.; Zhao, W.; Booth, A.M.; Ding, W.; Bartlett, N.L.; Brander, D.M.; Barr, P.M.; Rogers, K.A.; et al. Ibrutinib Regimens versus Chemoimmunotherapy in Older Patients with Untreated CLL. N. Engl. J. Med. 2018, 379, 2517–2528. [CrossRef]
- Moreno, C.; Greil, R.; Demirkan, F.; Tedeschi, A.; Anz, B.; Larratt, L.; Simkovic, M.; Samoilova, O.; Novak, J.; Ben-Yehuda, D.; et al. Ibrutinib plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): A multicenter, randomized, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 43–56.
- Munir, T.; Brown, J.R.; O’Brien, S.; Barrientos, J.C.; Barr, P.M.; Reddy, N.M.; Coutre, S.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; et al. Final analysis from RESONATE: Up to six years of follow-up on ibrutinib in patients with previously treated chronic lymphocytic leukemia or small lymphocytic lymphoma. Am. J. Hematol. 2019, 94, 1353–1363. [CrossRef]
- Sharman JP, Egyed M, Jurczak W, Skarbnik A, Pagel JM, Flinn IW, Kamdar M, Munir T, Walewska R, Corbett G, et al. Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet 2020 Apr 18;395(10232):1278-1291.
- Shadman M, Munir T, Robak T, Brown JR, Kahl BS, Ghia P, Giannopoulos K, Šimkovič M, Österborg A, Laurenti L, et al. Zanubrutinib Versus Bendamustine and Rituximab in Patients With Treatment-Naïve Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma: Median 5-Year Follow-Up of SEQUOIA. J Clin Oncol 2025 Mar;43(7):780-787.
- Tam C, Thompson PA. BTK inhibitors in CLL: second-generation drugs and beyond.Blood Adv. 2024 May 14;8(9):2300-2309. [CrossRef]
- Huntington SF, de Nigris E, Puckett J, Kamal-Bahl S, Farooqui M, Ryland K, Sarpong E, Leng S, Yang X, Doshi JA. Ibrutinib discontinuation and associated factors in a real-world national sample of elderly Medicare beneficiaries with chronic lymphocytic leukemia. Leuk Lymphoma[ 2023 Dec;64(14):2286-2295.
- Lipsky, A.; Lamanna, N. Managing toxicities of Bruton tyrosine kinase inhibitors. Am. Soc. Hematol. Educ. Program 2020, 2020, 336–345.
- Dickerson, T.; Wiczer, T.; Waller, A.; Philippon, J.; Porter, K.; Haddad, D.; Guha, A.; Rogers, K.A.; Bhat, S.; Byrd, J.C.; et al. Hypertension and incident cardiovascular events following ibrutinib initiation. Blood 2019, 134, 1919–1928. [CrossRef]
- Molica S, Allsup D, Giannarelli D. Prevalence of BTK and PLCG2 Mutations in CLL Patients With Disease Progression on BTK Inhibitor Therapy: A Meta-Analysis. Am J Hematol. 2025 Feb;100(2):334-337.
- Byrd, J.C.; Hillmen, P.; Ghia, P.; Kater, A.P.; Chanan-Khan, A.; Furman, R.R.; O’Brien, S.; Yenerel, M.N.; Illés, A.; Kay, N.; et al. Acalabrutinib Versus Ibrutinib in Previously Treated Chronic Lymphocytic Leukemia: Results of the First Randomized Phase III Trial. J. Clin. Oncol. 2021, 39, 3441–3452. [CrossRef]
- Brown, J.R.; Eichhorst, B.; Hillmen, P.; Jurczak, W.; Kaźmierczak, M.; Lamanna, N.; O’Brien, S.M.; Tam, C.S.; Qiu, L.; Zhou, K.; et al. Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2023, 388, 319–332. [CrossRef]
- Pan Z, Scheerens H, Li SJ, et al. Discovery of selective irreversible inhibitors for Bruton’s tyrosine kinase. Chem Med Chem. 2007;2(1):58-61.
- Byrd JC, Furman RR, Coutre SE, et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N Engl J Med. 2013;369(1):32-42.
- Guo Y, Liu Y, Hu N, et al. Discovery of zanubrutinib (BGB-3111), a novel, potent, and selective covalent inhibitor of Bruton’s tyrosine kinase. J Med Chem. 2019;62(17):7923-7940.
- Alu A, Lei H, Han X, Wei Y, Wei X. BTK inhibitors in the treatment of hematological malignancies and inflammatory diseases: mechanisms and clinical studies.J Hematol Oncol. 2022 Oct 1;15(1):138. [CrossRef]
- Gruessner C, Wiestner A, Sun C. Resistance mechanisms and approach to chronic lymphocytic leukemia after BTK inhibitor therapy. Leuk Lymphoma 2025 Jul;66(7):1176-1188.
- Woyach JA, Ruppert AS, Guinn D, et al. BTK(C481S)-mediated resistance to ibrutinib in chronic lymphocytic leukemia. J Clin Oncol. 2017;35(13):1437-1443.
- Molica S, Allsup D. Bruton’s Tyrosine Kinase (BTK) Mutations in Chronic Lymphocytic Leukemia (CLL): A Clinical View. Mediterr J Hematol Infect Dis. 2025 Jul 1;17(1):e2025053.
- Gomez EB, Ebata K, Randeria HS, Rosendahl MS, Cedervall EP, Morales TH, Hanson LM, Brown NE, Gong X, Stephens J, et al. Preclinical characterization of pirtobrutinib, a highly selective, noncovalent (reversible) BTK inhibitor. Blood. 2023 Jul 6;142(1):62-72.
- Bravo-Gonzalez A, Alasfour M, Soong D, Noy J, Pongas G. Advances in Targeted Therapy: Addressing Resistance to BTK Inhibition in B-Cell Lymphoid Malignancies. Cancers (Basel). 2024 Oct 10;16(20):3434.
- Thompson PA, Tam CS. Pirtobrutinib: a new hope for patients with BTK inhibitor-refractory lymphoproliferative disorders.Blood. 2023 Jun 29;141(26):3137-3142. [CrossRef]
- Tam CS, Balendran S, Blombery P. Novel mechanisms of resistance in CLL: variant BTK mutations in second-generation and noncovalent BTK inhibitors. Blood. 2025 Mar 6;145(10):1005-1009.
- Naeem A, Utro F, Wang Q, Cha J, Vihinen M, Martindale S, Zhou Y, Ren Y, Tyekucheva S, Kim AS, et al. Pirtobrutinib targets BTK C481S in ibrutinib-resistant CLL but second-site BTK mutations lead to resistance. Blood Adv. 2023 May 9;7(9):1929-1943. [CrossRef]
- Aslan B, Kismali G, Iles LR, Manyam GC, Ayres ML, Chen LS, Gagea M, Bertilaccio MTS, Wierda WG, Gandhi V. Pirtobrutinib inhibits wild-type and mutant Bruton’s tyrosine kinase-mediated signaling in chronic lymphocytic leukemia. Blood Cancer J. 2022 May 20;12(5):80. [CrossRef]
- Mato AR, Shah NN, Jurczak W, Cheah CY, Pagel JM, Woyach JA, Fakhri B, Eyre TA, Lamanna N, Patel MR, et al. Pirtobrutinib in relapsed or refractory B-cell malignancies (BRUIN): a phase 1/2 study. Lancet. 2021 Mar 6;397(10277):892-901.
- Mato AR, Woyach JA, Brown JR, Ghia P, Patel K, Eyre TA, Munir T, Lech-Maranda E, Lamanna N, Tam CS, et al. Pirtobrutinib after a Covalent BTK Inhibitor in Chronic Lymphocytic Leukemia. N Engl J Med. 2023 Jul 6;389(1):33-44.
- Shah NN, Wang M, Roeker LE, Patel K, Woyach JA, Wierda WG, Ujjani CS, Eyre TA, Zinzani PL, Alencar AJ,et al Pirtobrutinib monotherapy in Bruton tyrosine kinase inhibitor-intolerant patients with B-cell malignancies: results of the phase I/II BRUIN trial Haematologica. 2025 Jan 1;110(1):92-102.
- Al-Sawaf O, Jen MH, Hess LM, Zhang J, Goebel B, Pagel JM, Abhyankar S, Davids MS, Eyre TA. Pirtobrutinib versus venetoclax in covalent Bruton tyrosine kinase inhibitor-pretreated chronic lymphocytic leukemia: a matching-adjusted indirect comparison.Haematologica. 2024 Jun 1;109(6):1866-1873. [CrossRef]
- Rogers KA. Choosing between CAR T-cell therapy and pirtobrutinib in double-refractory CLL. Clin Adv Hematol Oncol. 2024 Dec;22(10):494-496.
- Jain N, Eyre TA, Winfree KB, Bhandari NR, Khanal M, Sugihara T, Chen Y, Abada P, Patel K. Real-world outcomes after discontinuation of covalent BTK inhibitor-based therapy in patients with chronic lymphocytic leukemia/small lymphocytic lymphoma. Leuk Lymphoma. 2025 Aug;66(8):1400-1412.
- Kater AP, Harrup R, Kipps TJ, Eichhorst B, Owen CJ, Assouline S, Lamanna N, Robak T, de la Serna J, Jaeger U, et al. The MURANO study: final analysis and retreatment/crossover substudy results of VenR for patients with relapsed/refractory CLL. Blood. 2025 Jun 5;145(23):2733-2745.
- Huntington SF, de Nigris E, Puckett J, Kamal-Bahl S, Farooqui M, Ryland K, Sarpong E, Leng S, Yang X, Doshi JA. Ibrutinib discontinuation and associated factors in a real-world national sample of elderly Medicare beneficiaries with chronic lymphocytic leukemia. Leuk Lymphoma. 2023 Dec;64(14):2286-2295.
- Sharman JP, Munir T, Grosicki S, Roeker LE, Burke JM, Chen CI, Grzasko N, Follows G, Mátrai Z, Sanna A, et al. Phase III Trial of Pirtobrutinib Versus Idelalisib/Rituximab or Bendamustine/Rituximab in Covalent Bruton Tyrosine Kinase Inhibitor-Pretreated Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (BRUIN CLL-321). J Clin Oncol. 2025 Aug;43(22):2538-2549.
- Delgado, A.; Guddati, A.K. Clinical endpoints in oncology—A primer. Am. J. Cancer Res. 2021, 11, 1121–1131.
- Molica S. Redefining efficacy and safety endpoints for chronic lymphocytic leukemia in the era of targeted therapy. Expert Rev Hematol. 2023 Jul-Dec;16(11):803-806. [CrossRef]
- Thompson MC, Bhat SA, Jurczak W, Patel K, Shah NN, Woyach JA, Coombs CC, Eyre TA, Danecki M, Dlugosz-Danecka M, et al. Outcomes of Therapies Following Discontinuation of Non-Covalent Bruton’s Tyrosine Kinase Inhibitors for Patients with Chronic Lymphocytic Leukemia and Richter Transformation: Results from an International, Multicenter Study. Blood (2024) 144 (Supplement 1): 1870.
- Wierda WG, Shah NN, Cheah CY, Lewis D, Hoffmann MS, Coombs CC, Lamanna N, Ma S, Jagadeesh D, Munir T, et al. Pirtobrutinib, a highly selective, non-covalent (reversible) BTK inhibitor in patients with B-cell malignancies: analysis of the Richter transformation subgroup from the multicentre, open-label, phase 1/2 BRUIN study. Lancet Haematol. 2024 Sep;11(9):e682-e692. [CrossRef]
- Eyre TA. Richter transformation-is there light at the end of this tunnel?Hematology Am Soc Hematol Educ Program. 2023 Dec 8;2023(1):427-432.
- Molica S. Navigating the gap between guidelines and practical challenges in selecting first-line therapy for chronic lymphocytic leukemia. Expert Rev Hematol. 2025 Jan-Mar;18(3):195-200.
- Roeker LE, Woyach JA, Cheah CY, Coombs CC, Shah NN, Wierda WG, Patel MR, Lamanna N, Tsai DE, Nair B, et al. Fixed-duration pirtobrutinib plus venetoclax with or without rituximab in relapsed/refractory CLL: the phase 1b BRUIN trial. Blood. 2024 Sep 26;144(13):1374-1386. [CrossRef]
- Qi J, Endres S, Yosifov DY, Tausch E, Dheenadayalan RP, Gao X, Müller A, Schneider C, Mertens D, Gierschik P, et al. Acquired BTK mutations associated with resistance to noncovalent BTK inhibitors. Blood Adv. 2023 Oct 10;7(19):5698-5702.
- Brown J, Desikan S, Nguyen B, Won H, Tantawy SI, McNeely S, Marella N, Ebata K, Woyach JA, Patel K, et al. Genomic evolution and resistance during pirtobrutinib therapy in covalent BTK-inhibitor (cBTKi) pretreated chronic lymphocytic leukemia patients: updated analysis from the BRUIN study [abstract]. Blood. 2023;142(suppl 1):326.
- Gandhi V, Tantawy S, Aslan B, Manyam G, Iles L, Timofeeva N, Singh N, Jain N, Ferrajoli A, Thompson P, et al. Pharmacological profiling in CLL patients during pirtobrutinib therapy and disease progression. Res Sq [Preprint]. 2025 Mar 31:rs.3.rs-6249480. [Version 1]. [CrossRef]
- Shadman M, Davids MS.How I treat patients with CLL after prior treatment with a covalent BTK inhibitor and a BCL-2 inhibitor. Blood. 2025 Jul 29:blood.2024025482. Online ahead of print. PMID: 40729699. [CrossRef]
- Molica S, Allsup D Chronic Lymphocytic Leukemia Care and Beyond: Navigating the Needs of Long-Term Survivors. Cancers (Basel). 2025 Jan 2;17(1):119. [CrossRef]
- Lamanna N, Tam CS, Woyach JA, Alencar AJ, Palomba ML, Zinzani PL, Flinn IW, Fakhri B, Cohen JB, Kontos A, et al. Evaluation of bleeding risk in patients who received pirtobrutinib in the presence or absence of antithrombotic therapy. EJHaem. 2024 Sep 27;5(5):929-939. [CrossRef]
- Forconi F, Moss P. Perturbation of the normal immune system in patients with CLL.Blood. 2015 Jul 30;126(5):573-581.
- Shah PV, Gladstone DE. Covalent and Non-Covalent BTK Inhibition in Chronic Lymphocytic Leukemia Treatment. Curr Treat Options Oncol. 2025 Jul 18. [CrossRef]
- Molica S. Defining treatment success in chronic lymphocytic leukemia: exploring surrogate markers, comorbidities, and patient-centered endpoints. Expert Rev Hematol. 2024 Jul;17(7):279-285.
- Montoya S, Bourcier J, Noviski M, Lu H, Thompson MC, Chirino A, Jahn J, Sondhi AK, Gajewski S, Tan YSM, et al. Kinase-impaired BTK mutations are susceptible to clinical-stage BTK and IKZF1/3 degrader NX-2127. Science. 2024 Feb 2;383(6682):eadi5798.
- Shadman M, Davids MS How I treat patients with CLL after prior treatment with a covalent BTK inhibitor and a BCL-2 inhibitor. Blood. 2025 Jul 29:blood.2024025482. Online ahead of print. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



