Submitted:
15 August 2025
Posted:
18 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. ABO Blood Group System
3. ABO-Incompatible Kidney Transplantation (ABOi KTx)
4. Antibody Titer Assessment
4.1. DTT Treatment:
4.2. 2-Mercaptoethanol (2-ME):
4.3. Heat Inactivation:
4.4. Enzymatic Digestion:
4.5. Immunoaffinity-Based Removal:
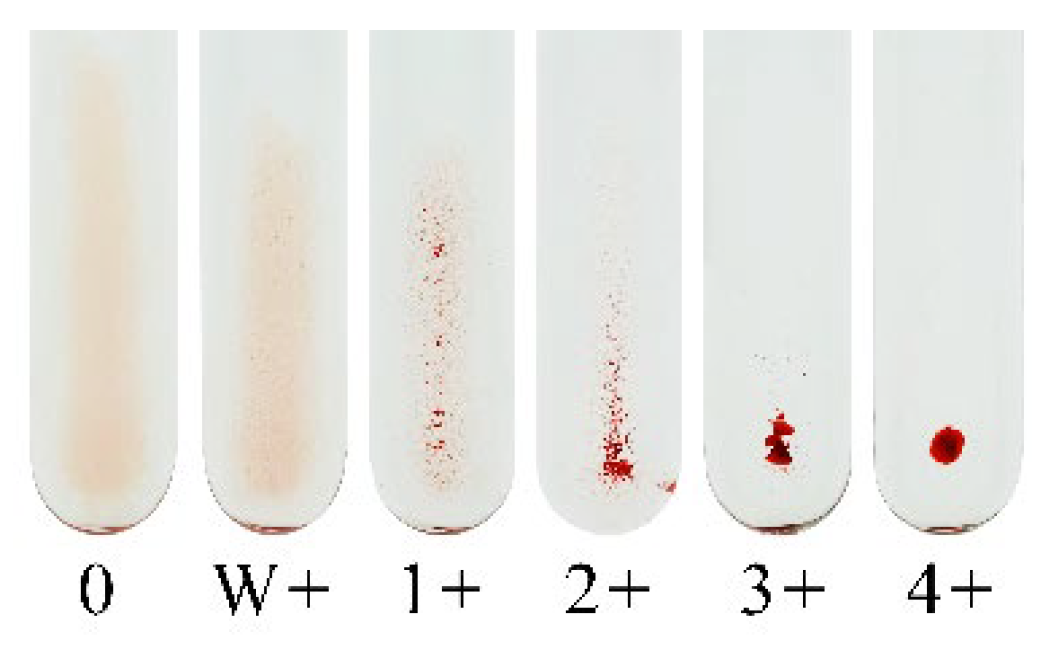
5. Conventional Tube Method (CTM)
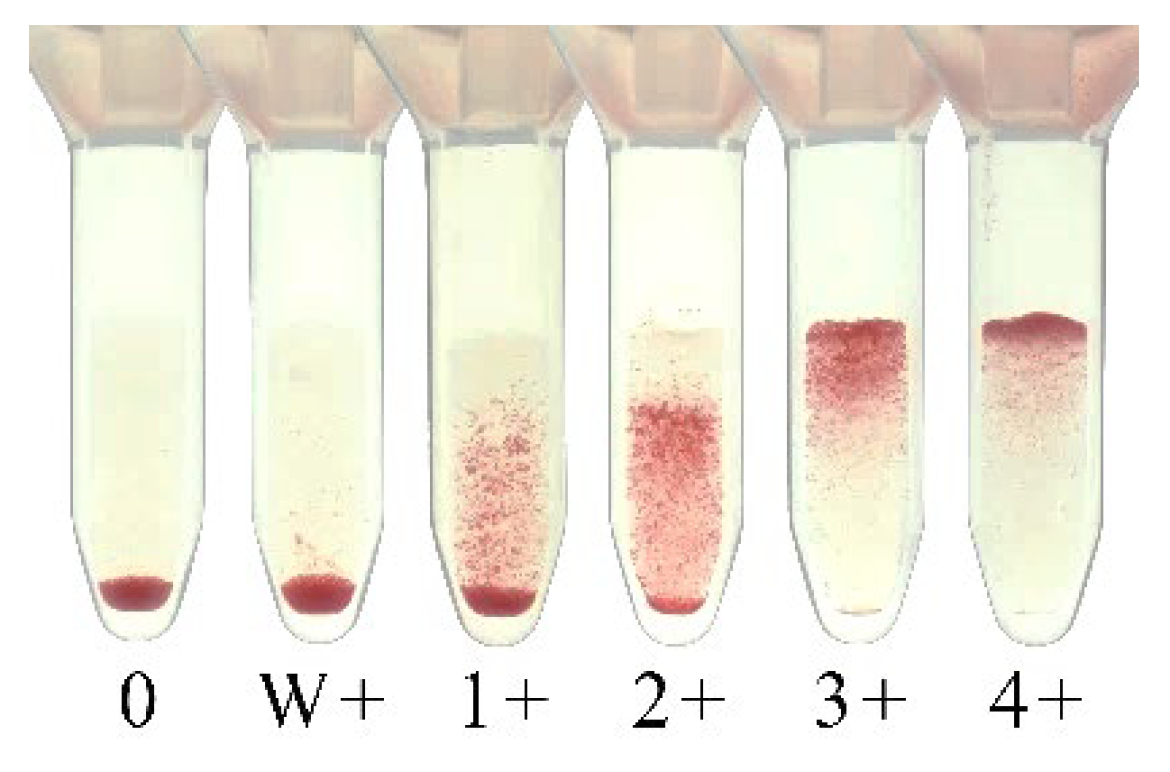
6. Column Agglutination Technique (CAT)
7. Flow Cytometry Method (FCM)
8. Enzyme-Linked Immunosorbent Assay (ELISA)-Based Method
9. ABO Subtype Antibody Assessment (Glycan Microarray and Multiplex Bead Immunoassay)
10. CD31-ABO Microarray
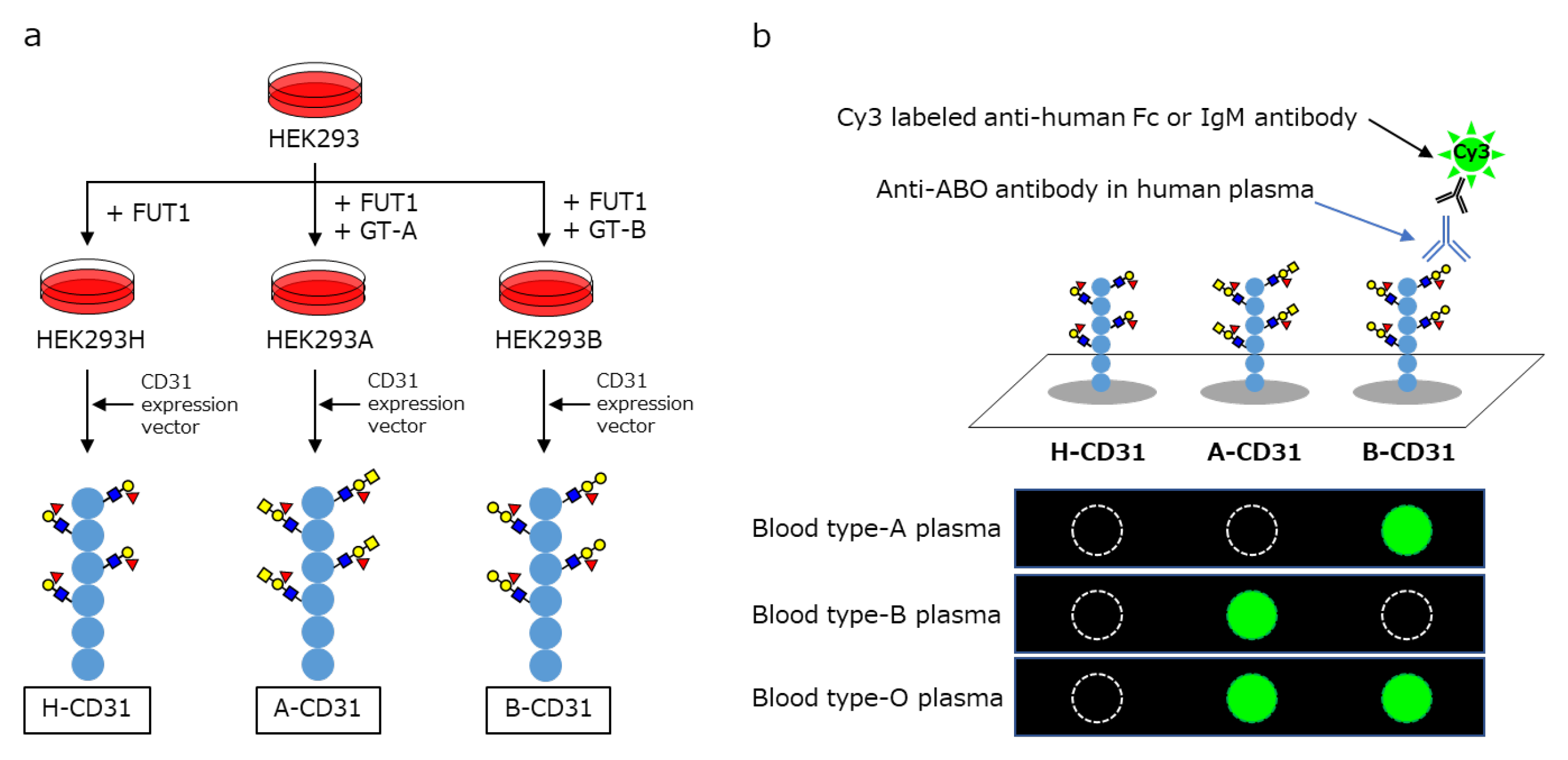
11. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABOi | ABO incompatible |
| ABMR | Antibody-mediated rejection |
| CAT | Column agglutination technique |
| CTM | Conventional tube method |
| DTT | Dithiothreitol |
| ELISA | Enzyme-linked immunosorbent assay |
| FCM | Flow cytometry method |
| HLA | Human leukocyte antigen |
| MFI | Mean fluorescence intensity |
| KTx | Kidney transplantation |
| RBC | Red blood cell |
| 2-ME | 2-Mercaptoethanol |
References
- Landsteiner K. Ueber Agglutinationserscheinungen normalen menschlichen Blutes. Wien Klin Wochschr 1901;14:1132-34.
- Mollison PL. Blood transfusion in clinical medicine. 5th ed. Oxford: Blackwell Scientific Publications; 1972.
- Springer GF, Tegtmeyer H. Apparent violation of “Landsteiner’s law.” Klin Wochenschr 1974;52:295-7.
- Rawson AJ, Abelson NM. Studies of blood groups antibodies. III. Observations on the physicochemical properties of isohemagglutinins and isohemolysins. J Immunol 1960;85:636-9.
- Brouwers HA, Overbeeke MA, Gemke RJ, Maas CJ, van Leeuwen EF, Engelfriet CP. Sensitive methods for determining subclasses of IgG anti-A and anti-B in sera of blood-group-O women with a blood-group-A or -B child. Br J Haematol 1987;66:267-70.
- Springer GF, Horton RE. Blood group isoantibody stimulation in man by feeding blood group-active bacteria. J Clin Invest 1969;48:1280-91. [CrossRef]
- Tasaki M, Yoshida Y, Miyamoto M, Nameta M, Cuellar LM, Xu B, Zhang Y, Yaoita E, Nakagawa Y, Saito K, Yamamoto T, Takahashi K. Identification and characterization of major proteins carrying ABO blood group antigens in the human kidney. Transplantation. 2009 Apr 27;87(8):1125-33. [CrossRef]
- HUME DM, MERRILL JP, MILLER BF, THORN GW. Experiences with renal homotransplantation in the human: report of nine cases. J Clin Invest. 1955 Feb;34(2):327-82. [CrossRef]
- STARZL TE, MARCHIORO TL, HOLMES JH, HERMANN G, BRITTAIN RS, STONINGTON OH, TALMAGE DW, WADDELL WR. RENAL HOMOGRAFTS IN PATIENTS WITH MAJOR DONOR-RECIPIENT BLOOD GROUP INCOMPATIBILITIES. Surgery. 1964 Feb;55:195-200.
- Wilbrandt R, Tung KS, Deodhar SD, Nakamoto S, Kolff WJ. ABO blood group incompatibility in human renal homotransplantation. Am J Clin Pathol. 1969 Jan;51(1):15-23. [CrossRef]
- Alexandre GP, De Bruyere M, Squifflet JP, Moriau M, Latinne D, Pirson Y. Human ABO-incompatible living donor renal homografts. Neth J Med. 1985;28(6):231-4.
- Reding R, Squifflet JP, Pirson Y, Jamart J, De Bruyère M, Moriau M, Latinne D, Carlier M, Alexandre GP. Living-related and unrelated donor kidney transplantation: comparison between ABO-compatible and incompatible grafts. Transplant Proc. 1987 Feb;19(1 Pt 2):1511-3.
- Alexandre GPJ, Latinne D, Gianello P, Squifflet JP. Preformed cytotoxic antibodies and ABO-incompatible grafts. Clin Transplantation 1991: (Spec. issue) 5: 583–594. [CrossRef]
- Takahashi K, Tanabe K, Ooba S, Yagisawa T, Nakazawa H, Teraoka S, Hayasaka Y, Kawaguchi H, Ito K, Toma H, et al. Prophylactic use of a new immunosuppressive agent, deoxyspergualin, in patients with kidney transplantation from ABO-incompatible or preformed antibody-positive donors. Transplant Proc. 1991 Feb;23(1 Pt 2):1078-82.
- Kawaguchi H, Hattori M, Ito K, Takahashi K, Yagisawa T, Agishi T, Ota K. A successful ABO blood type incompatible kidney transplantation in a child. Transpl Int. 1991 Apr;4(1):63-4.
- Ota K, Takahashi K, Agishi T, Sonda T, Oka T, Ueda S, Amemiya H, Shiramizu T, Okazaki H, Akiyama N, Hasegawa A, Kawamura T, Takagi H, Ueno A. Multicentre trial of ABO-incompatible kidney transplantation. Japanese Biosynsorb ABO-incompatible kidney transplant study group. Transpl Int. 1992;5 Suppl 1:S40-3.
- Takahashi K, Saito K, Takahara S, Okuyama A, Tanabe K, Toma H, Uchida K, Hasegawa A, Yoshimura N, Kamiryo Y; Japanese ABO-Incompatible Kidney Transplantation Committee. Excellent long-term outcome of ABO-incompatible living donor kidney transplantation in Japan. Am J Transplant. 2004 Jul;4(7):1089-96.
- Saito K, Nakagawa Y, Suwa M, Kumagai N, Tanikawa T, Nishiyama T, Ueno M, Gejyo F, Nishi S, Takahashi K. Pinpoint targeted immunosuppression: anti-CD20/MMF desensitization with anti-CD25 in successful ABO-incompatible kidney transplantation without splenectomy. Xenotransplantation. 2006 Mar;13(2):111-7. [CrossRef]
- Takahashi K. Recent findings in ABO-incompatible kidney transplantation: classification and therapeutic strategy for acute antibody-mediated rejection due to ABO-blood-group-related antigens during the critical period preceding the establishment of accommodation. Clin Exp Nephrol. 2007 Jun;11(2):128-141. [CrossRef]
- AuBuchon JP, de Wildt-Eggen J, Dumont LJ; Biomedical Excellence for Safer Transfusion Collaborative; Transfusion Medicine Resource Committee of the College of American Pathologists. Reducing the variation in performance of antibody titrations. Arch Pathol Lab Med. 2008 Jul;132(7):1194-201.
- Kang SJ, Lim YA, Baik SY. Comparison of ABO antibody titers on the basis of the antibody detection method used. Ann Lab Med 2014;34 (4):300–6. [CrossRef]
- AuBuchon JP, de Wildt-Eggen J, Dumont LJ. Reducing the variation in performance of antibody titrations. Vox Sang 2008;95:57-65. [CrossRef]
- Kobayashi T, Saito K. A series of surveys on assay for anti-A/B antibody by Japanese ABO-incompatible Transplantation Committee. Xenotransplantation. 2006 Mar;13(2):136-40. [CrossRef]
- Tanabe K. Interinstitutional variation in the measurement of anti-A/B antibodies: the Japanese ABO-Incompatible Transplantation Committee survey. Transplantation. 2007 Dec 27;84(12 Suppl):S13-6. [CrossRef]
- Park ES, Jo KI, Shin JW, Park R, Choi TY, Bang HI, Chai GR, Yun SG. Comparison of total and IgG ABO antibody titers in healthy individuals by using tube and column agglutination techniques. Ann Lab Med. 2014 May;34(3):223-9. [CrossRef]
- Zhou S, Liu Q, Fang M, Shen S, Wang R, Jing Y, Wang M. Evaluation of different methods for antibody titer determination in ABO-incompatible kidney transplantation. Transl Androl Urol. 2025 Mar 30;14(3):540-552. [CrossRef]
- Nayak S, Makroo RN, Prakash B, Chandra T, Agrawal S, Chowdhry M, Mohapatra A. Comparative Evaluation of Five Different Methods of Anti-ABO Antibody Titration: An Aid for ABO-Incompatible Organ Transplants. Ther Apher Dial. 2019 Feb;23(1):86-91. [CrossRef]
- Yung GP, Valli PV, Starke A, Mueller RJ, Fehr T, Cesar-Ozpamir M, Schanz U, Weber M, Wüthrich RP, Seebach JD, Stussi G. Flow cytometric measurement of ABO antibodies in ABO-incompatible living donor kidney transplantation. Transplantation. 2007 Dec 27;84(12 Suppl):S20-3. [CrossRef]
- Stussi G, Huggel K, Lutz HU, Schanz U, Rieben R, Seebach JD. Isotype-specific detection of ABO blood group antibodies using a novel flow cytometric method. Br J Haematol. 2005 Sep;130(6):954-63. [CrossRef]
- Spalter SH, Kaveri SV, Bonnin E, Mani JC, Cartron JP, Kazatchkine MD. Normal human serum contains natural antibodies reactive with autologous ABO blood group antigens. Blood. 1999 Jun 15;93(12):4418-24.
- Satoh A, Kawagishi N, Minegishi M, Takahashi H, Akamatsu Y, Doi H, Satomi S. Development of a novel ELISA for detection of anti-A and anti-B antibodies in recipients of ABO-incompatible living donor liver grafts. Tohoku J Exp Med. 2007 Apr;211(4):359-67. [CrossRef]
- Clausen H, Hakomori S. ABH and related histo-blood group antigens; immunochemical differences in carrier isotypes and their distribution. Vox Sang. 1989;56(1):1-20. [CrossRef]
- Meloncelli PJ, Lowary TL. Synthesis of ABO histo-blood group type I and II antigens. Carbohydr Res. 2010 Nov 2;345(16):2305-22. [CrossRef]
- Meloncelli PJ, West LJ, Lowary TL. Synthesis and NMR studies on the ABO histo-blood group antigens: synthesis of type III and IV structures and NMR characterization of type I-VI antigens. Carbohydr Res. 2011 Sep 6;346(12):1406-26. [CrossRef]
- Jeyakanthan M, Meloncelli PJ, Zou L, Lowary TL, Larsen I, Maier S, Tao K, Rusch J, Chinnock R, Shaw N, Burch M, Beddows K, Addonizio L, Zuckerman W, Pahl E, Rutledge J, Kanter KR, Cairo CW, Buriak JM, Ross D, Rebeyka I, West LJ. ABH-Glycan Microarray Characterizes ABO Subtype Antibodies: Fine Specificity of Immune Tolerance After ABO-Incompatible Transplantation. Am J Transplant. 2016 May;16(5):1548-58. [CrossRef]
- Bentall A, Jeyakanthan M, Braitch M, Cairo CW, Lowary TL, Maier S, Halpin A, Motyka B, Zou L, West LJ, Ball S. Characterization of ABH-subtype donor-specific antibodies in ABO-A-incompatible kidney transplantation. Am J Transplant. 2021 Nov;21(11):3649-3662. [CrossRef]
- Halpin AM, Murphey C, Motyka B, Li C, Pearcey J, Ellis M, Dijke IE, Urschel S, Bingaman A, Van Ong T, Kapturczak M, Lowary TL, Cairo CW, West LJ. Multiplex bead immunoassay in ABO-A2-incompatible kidney transplantation. Am J Transplant. 2025 Apr 10:S1600-6135(25)00174-1. [CrossRef]
- Koshino K, Okamoto M, Sakai K, Suzuki T, Nobori S, Matsuyama M, Ushigome H, Okajima H, Yoshimura N. The excellent outcomes of ABO-incompatible kidney transplantation with high titer (>×2048) using anti-CD20 and anti-CD25 antibody without splenectomy: two case reports. Transplant Proc. 2011 Jul-Aug;43(6):2379-82. [CrossRef]
- Kim H, Choe W, Shin S, Kim YH, Han DJ, Park SK, Kwon SW, Ko DH. ABO-incompatible kidney transplantation can be successfully conducted by monitoring IgM isoagglutinin titers during desensitization. Transfusion. 2020 Mar;60(3):598-606. [CrossRef]
- Tobian AA, Shirey RS, Montgomery RA, Cai W, Haas M, Ness PM, King KE. ABO antibody titer and risk of antibody-mediated rejection in ABO-incompatible renal transplantation. Am J Transplant. 2010 May;10(5):1247-53. [CrossRef]
- Tasaki M, Saito K, Nakagawa Y, Imai N, Ito Y, Yoshida Y, Ikeda M, Ishikawa S, Narita I, Takahashi K, Tomita Y. Analysis of the prevalence of systemic de novo thrombotic microangiopathy after ABO-incompatible kidney transplantation and the associated risk factors. Int J Urol. 2019 Dec;26(12):1128-1137. [CrossRef]
- Tasaki M, Yoshida Y, Miyamoto M, Nameta M, Cuellar LM, Xu B, Zhang Y, Yaoita E, Nakagawa Y, Saito K, Yamamoto T, Takahashi K. Identification and characterization of major proteins carrying ABO blood group antigens in the human kidney. Transplantation. 2009 Apr 27;87(8):1125-33. [CrossRef]
- Tasaki M, Tateno H, Sato T, Tomioka A, Kaji H, Narimatsu H, Saito K, Nakagawa Y, Aoki T, Kamimura M, Ushiki T, Okada M, Miwa Y, Hotta K, Yoshida Y, Takahashi K, Tomita Y. A Novel Method of CD31-Combined ABO Carbohydrate Antigen Microarray Predicts Acute Antibody-Mediated Rejection in ABO-Incompatible Kidney Transplantation. Transpl Int. 2022 Mar 23;35:10248. [CrossRef]
- Tasaki M, Tateno H, Sato T, Narimatsu H, Saito K, Nakagawa Y, Aoki T, Kamimura M, Ushiki T, Takahashi K, Tomita Y. Hyporesponsiveness against donor’s ABO antigens of renal grafts after ABO-incompatible kidney transplantation. Clin Exp Nephrol. 2023 Jan;27(1):89-95. [CrossRef]
| Method | Need for IgM Removal |
Cost | Target Antigen | Objectivity | Advantages | Disadvantages |
|---|---|---|---|---|---|---|
| Conventional Tube Method (CTM) | Yes (recommended) | Low | Native ABO antigens on RBCs | Low (subjective) |
Widely used; low cost; simple | Subjective; inter-observer variability; poor reproducibility |
| Column Agglutination Technique (CAT) | Yes (recommended) | Moderate | Native ABO antigens on RBCs | Moderate | More standardized than CTM | Still partially subjective; limited standardization |
| Flow Cytometry (FCM) | No | Moderate | ABO antigens on Chemically-fixed RBCs | High | Quantitative; sensitive; can detect low-level antibodies | Requires specialized equipment and training; Limited clinical validation |
| ELISA | No | High | Synthetic ABO oligosaccharides or glycoproteins | High | Quantitative; adaptable | Limited clinical validation |
| ABO Glycan Microarray | No | High | Synthetic A/B oligosaccharide subtypes (I–VI) |
High | Subtype-specific analysis; quantitative | Technically complex; not widely available |
| CD31-ABO Microarray | No | High | Recombinant CD31 with ABO antigens (endothelial-like) | High | Endothelial-specific ABO detection | Not yet standard; high cost; requires recombinant protein |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




