Submitted:
11 August 2025
Posted:
12 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. GLP-1 Physiology and Mechanism of Action of Its Analogues
2.1. Role of GLP-1 in Glycaemia
2.2. Role of GLP-1 in Body Weight
2.3. Role of GLP-1 in Lipid Profile
2.4. Role of GLP-1 in Inflammation
2.5. Role of GLP-1 in Blood Pressure (BP)
2.6. Role of GLP-1 in Heart Rate (HR)
2.7. Role of GLP-1 in Endothelial Function and Atherosclerosis
3. General Cardiovascular Effects of GLP-1RAs
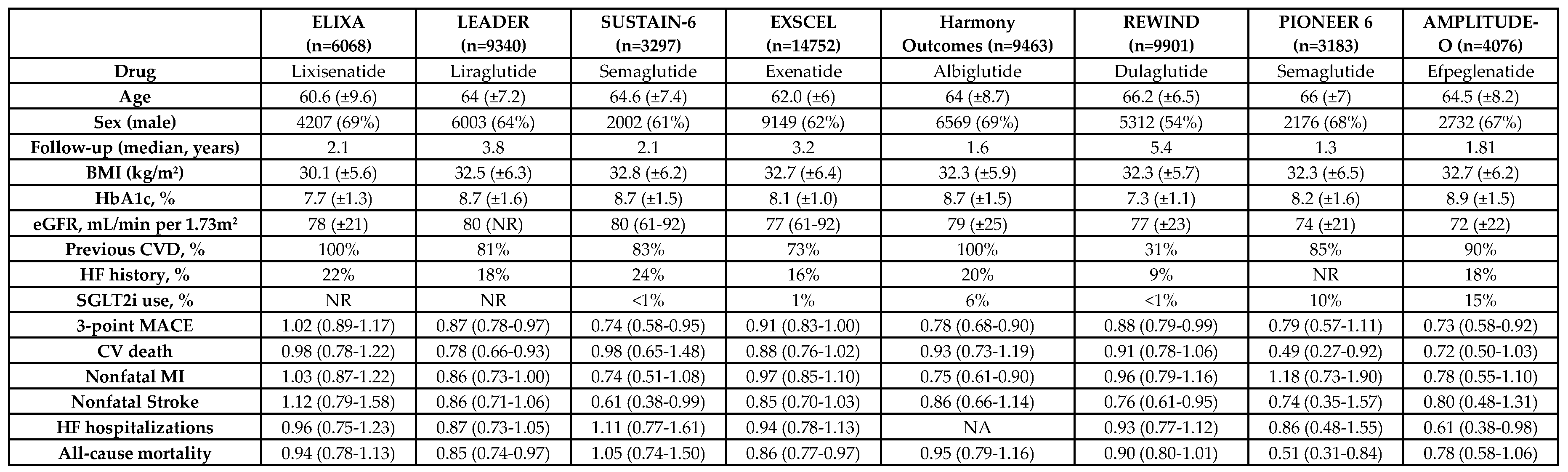
3.1. GLP-1RAs Cardiovascular Outcome Trials Results
4. GLP-1RAs and Chronic Heart Failure (HF)
4.1. GLP-1RAs and Heart Failure with Reduced Ejection Fraction (HFrEF)
4.2. GLP-1RAs and Heart Failure with Preserved Ejection Fraction (HFpEF)
5. Potential Mechanisms in Heart Failure
6. Gaps in Knowledge and Future Perspectives
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| BP | Blood Pressure |
| cAMP | Cyclic adenosine monophosphate |
| CI | Confidence Interval |
| CNS | Central Nervous System |
| CV | Cardiovascular |
| CVOT | Cardiovascular Outcome Trials |
| DPP-4 | Dipeptidyl-peptidase-4 |
| EAT | Epicardial Adipose Tissue |
| eGFR | Estimated Glomerular Filtration Rate |
| GIP | Glucose-dependent Insulinotropic Polypeptide |
| GLP-1 | Glucagon-like peptide -1 |
| GLP-1 RAs | Glucagon-like peptide-1 receptor agonists |
| HF | Heart Failure |
| HFrEF | Heart Failure with Reduced Ejection Fraction |
| HFpEF | Heart Failure with Preserved Ejection Fraction |
| HR | Heart Rate |
| KCCQ-CSS | Kansas City Cardiomyopathy Questionnaire – Clinical Summary Score |
| LVEF | Left Ventricle Ejection Fraction |
| MACE | Major Adverse Cardiovascular Events |
| RAAS | Renin-Angiotensin-Aldosteron System |
| RCT | Randomized Clinical Trials |
| T2DM | Type 2 Diabetes Mellitus |
| VT | Ventricular Tachycardia |
References
- McDonagh, T.A. et al., “2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure,” Sep. 21, 2021, Oxford University Press. [CrossRef]
- Khan, M.S.; Shahid, I.; Bennis, A.; Rakisheva, A.; Metra, M.; Butler, J., “Global epidemiology of heart failure,” Oct. 01, 2024, Nature Research. [CrossRef]
- Pop-Busui, R. et al., “Heart Failure: An Underappreciated Complication of Diabetes. A Consensus Report of the American Diabetes Association,” Diabetes Care, vol. 45, no. 7, pp. 1670–1690, Jul. 2022. [CrossRef]
- Campbell, P.; Krim, S.; Ventura, H., “Heart Failure and Diabetes The Epidemiology of Heart Failure in Diabetes Mellitus The Bi-directional Impact of Two Chronic Illnesses: Heart Failure and Diabetes-A review of the Epidemiology and Outcomes,” 2015. [Online]. Available: http://www.cdc.gov/diabetes/home.
- Guglin, M.; Lynch, K.; Krischer, J., “Heart failure as a risk factor for diabetes mellitus,” Cardiology (Switzerland), vol. 129, no. 2, pp. 84–92, Nov. 2014. [CrossRef]
- Wilcox, T.; De Block, C.; Schwartzbard, A.Z.; Newman, J.D., “Diabetic Agents, From Metformin to SGLT2 Inhibitors and GLP1 Receptor Agonists: JACC Focus Seminar,” Apr. 28, 2020, Elsevier Inc. [CrossRef]
- Heuvelman, V.D.; Van Raalte, D.H.; Smits, M.M.; Smits, M.M., “Cardiovascular effects of GLP-1 receptor agonists: from mechanistic studies in humans to clinical outcomes.,” Cardiovasc Res, vol. 116, no. 5, pp. 916–930, Apr. 2020. [CrossRef]
- Zheng, Z. et al., “Glucagon-like peptide-1 receptor: mechanisms and advances in therapy,” Signal Transduct Target Ther, vol. 9, no. 1, p. 234, Sep. 2024. [CrossRef]
- Bu, T.; Sun, Z.; Pan, Y.; Deng, X.; Yuan, G., “Glucagon-Like Peptide-1: New Regulator in Lipid Metabolism,” May 01, 2024, Korean Diabetes Association. [CrossRef]
- Sun, F. et al., “Effect of glucagon-like peptide-1 receptor agonists on lipid profiles among type 2 diabetes: A systematic review and network meta-analysis,” Jan. 01, 2015, Excerpta Medica Inc. [CrossRef]
- Hachuła, M.; Kosowski, M.; Ryl, S.; Basiak, M.; Okopień, B., “Impact of Glucagon-Like Peptide 1 Receptor Agonists on Biochemical Markers of the Initiation of Atherosclerotic Process,” Int J Mol Sci, vol. 25, no. 3, Feb. 2024. [CrossRef]
- Bendotti, G. et al., “The anti-inflammatory and immunological properties of GLP-1 Receptor Agonists,” Aug. 01, 2022, Academic Press. [CrossRef]
- Wilding, J.P.H. et al., “Once-Weekly Semaglutide in Adults with Overweight or Obesity,” New England Journal of Medicine, vol. 384, no. 11, pp. 989–1002, Mar. 2021. [CrossRef]
- Hullon, D.; Subeh, G.K.; Volkova, Y.; Janiec, K.; Trach, A.; Mnevets, R., “The role of glucagon-like peptide-1 receptor (GLP-1R) agonists in enhancing endothelial function: a potential avenue for improving heart failure with preserved ejection fraction (HFpEF),” Dec. 01, 2025, BioMed Central Ltd. [CrossRef]
- Bray, J.J.H. et al., “Glucagon-like peptide-1 receptor agonists improve biomarkers of inflammation and oxidative stress: A systematic review and meta-analysis of randomised controlled trials,” Diabetes Obes Metab, vol. 23, no. 8, pp. 1806–1822, Aug. 2021. [CrossRef]
- El Aziz, M.S.A.; Kahle, M.; Meier, J.J.; Nauck, M.A., “A meta-analysis comparing clinical effects of short- or long-acting GLP-1 receptor agonists versus insulin treatment from head-to-head studies in type 2 diabetic patients,” Diabetes Obes Metab, vol. 19, no. 2, pp. 216–227, Feb. 2017. [CrossRef]
- Pauza, A.G. et al., “GLP1R Attenuates Sympathetic Response to High Glucose via Carotid Body Inhibition,” Circ Res, vol. 130, no. 5, pp. 694–707, Mar. 2022. [CrossRef]
- Gaspari, T.; Welungoda, I.; Widdop, R.E.; Simpson, R.W.; Dear, A.E., “The GLP-1 receptor agonist liraglutide inhibits progression of vascular disease via effects on atherogenesis, plaque stability and endothelial function in an ApoE-/- mouse model,” Diab Vasc Dis Res, vol. 10, no. 4, pp. 353–360, Jul. 2013. [CrossRef]
- Kim, M. et al., “GLP-1 receptor activation and Epac2 link atrial natriuretic peptide secretion to control of blood pressure,” Nat Med, vol. 19, no. 5, pp. 567–575, May 2013. [CrossRef]
- Sun, F. et al., “Impact of GLP-1 receptor agonists on blood pressure, heart rate and hypertension among patients with type 2 diabetes: A systematic review and network meta-analysis,” Diabetes Res Clin Pract, vol. 110, no. 1, pp. 26–37, Oct. 2015. [CrossRef]
- Adler, A. et al., “Pharmacological blood pressure lowering for primary and secondary prevention of cardiovascular disease across different levels of blood pressure: an individual participant-level data meta-analysis,” The Lancet, vol. 397, no. 10285, pp. 1625–1636, May 2021. [CrossRef]
- Subaran, S.C. et al., “GLP-1 at physiological concentrations recruits skeletal and cardiac muscle microvasculature in healthy humans,” Clin Sci, vol. 127, no. 3, pp. 163–170, 2014. [CrossRef]
- Husain, M. et al., “Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes,” New England Journal of Medicine, vol. 381, no. 9, pp. 841–851, Aug. 2019. [CrossRef]
- Kaur, S.; Rose, R.A., “New insights into the effects of glucagon-like peptide-1 on heart rate and sinoatrial node function,” Cardiovasc Res, Oct. 2024. [CrossRef]
- Lorenz, M. et al., “Differential effects of glucagon-like peptide-1 receptor agonists on heart rate,” Jan. 13, 2017, BioMed Central Ltd. [CrossRef]
- Lau, K. et al., “Resting Heart Rate as an Important Predictor of Mortality and Morbidity in Ambulatory Patients With Heart Failure: A Systematic Review and Meta-Analysis,” Mar. 01, 2021, Elsevier B.V. [CrossRef]
- Pfeffer, M.A. et al., “Lixisenatide in Patients with Type 2 Diabetes and Acute Coronary Syndrome,” New England Journal of Medicine, vol. 373, no. 23, pp. 2247–2257, Dec. 2015. [CrossRef]
- Chen, W.R. et al., “Effects of liraglutide on reperfusion injury in patients with ST-segment-elevation myocardial infarction,” Circ Cardiovasc Imaging, vol. 9, no. 12, Dec. 2016. [CrossRef]
- Marso, S.P. et al., “Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes,” New England Journal of Medicine, vol. 375, no. 19, pp. 1834–1844, Nov. 2016. [CrossRef]
- Neves, J.S. et al., “Risk of adverse events with liraglutide in heart failure with reduced ejection fraction: A post hoc analysis of the FIGHT trial,” Obe Metab, vol. 2023, no. 1, pp. 189–197, 2022. [CrossRef]
- Alexander, Y. et al., “Endothelial function in cardiovascular medicine: A consensus paper of the European Society of Cardiology Working Groups on Atherosclerosis and Vascular Biology, Aorta and Peripheral Vascular Diseases, Coronary Pathophysiology and Microcirculation, and Thrombosis,” Jan. 01, 2021, Oxford University Press. [CrossRef]
- Wei, R. et al., “Exenatide exerts direct protective effects on endothelial cells through the AMPK/Akt/eNOS pathway in a GLP-1 receptor-dependent manner,” 2016. [CrossRef]
- Chai, W. et al., “Glucagon-like peptide 1 recruits microvasculature and increases glucose use in muscle via a nitric oxide-dependent mechanism,” Diabetes, vol. 61, no. 4, pp. 888–896, Apr. 2012. [CrossRef]
- Krasner, N.M.; Ido, Y.; Ruderman, N.B.; Cacicedo, J.M., “Glucagon-Like Peptide-1 (GLP-1) analog liraglutide inhibits endothelial cell inflammation through a calcium and AMPK dependent mechanism,” PLoS One, vol. 9, no. 5, May 2014. [CrossRef]
- Nyström, T. et al., “Effects of glucagon-like peptide-1 on endothelial function in type 2 diabetes patients with stable coronary artery disease,” Am J Physiol Endocrinol Metab, vol. 287, pp. 1209–1215, 2004. [CrossRef]
- Gejl, M. et al., “Exenatide alters myocardial glucose transport and uptake depending on insulin resistance and increases myocardial blood flow in patients with type 2 diabetes,” Journal of Clinical Endocrinology and Metabolism, vol. 97, no. 7, Jul. 2012. [CrossRef]
- Read, P.A.; Khan, F.Z.; Dutka, D.P., “Cardioprotection against ischaemia induced by dobutamine stress using glucagon-like peptide-1 in patients with coronary artery disease,” Heart, vol. 98, no. 5, pp. 408–413, Mar. 2012. [CrossRef]
- Turner, R., “Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33),” Lancet, vol. 352, no. 9131, pp. 837–853, Sep. 1998. [CrossRef]
- Stratton, I.M. et al., “Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study,” BMJ, vol. 321, pp. 405–412, Aug. 2000.
- Scirica, B.M. et al., “Saxagliptin and Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus,” New England Journal of Medicine, vol. 14, no. 3, pp. 1317–1343, 2013. [CrossRef]
- Page, R.L. et al., “Drugs that may cause or exacerbate heart failure,” Circulation, vol. 134, no. 6, pp. e32–e69, Aug. 2016. [CrossRef]
- Suh, S. et al., “Increased risk of hospitalization for heart failure with newly prescribed dipeptidyl peptidase-4 inhibitors and pioglitazone using the Korean Health Insurance claims database,” Diabetes Metab J, vol. 39, no. 3, pp. 247–252, 2015. [CrossRef]
- Marso, S.P. et al., “Liraglutide and cardiovascular outcomes in type 2 diabetes,” New England Journal of Medicina, vol. 375, no. 4, pp. 311–322, Jul. 2016. [CrossRef]
- Holman, R.R. et al., “Effects of Once-Weekly Exenatide on Cardiovascular Outcomes in Type 2 Diabetes,” New England Journal of Medicine, vol. 377, no. 13, pp. 1228–1239, Sep. 2017. [CrossRef]
- Hernandez, A.F. et al., “Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomised placebo-controlled trial,” The Lancet, vol. 392, no. 10157, pp. 1519–1529, Oct. 2018. [CrossRef]
- Gerstein, H.C. et al., “Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial,” The Lancet, vol. 394, no. 10193, pp. 121–130, Jul. 2019. [CrossRef]
- Gerstein, H.C. et al., “Cardiovascular and Renal Outcomes with Efpeglenatide in Type 2 Diabetes,” New England Journal of Medicine, vol. 385, no. 10, pp. 896–907, Sep. 2021. [CrossRef]
- Longato, E.; Di Camillo, B.; Sparacino, G.; Tramontan, L.; Avogaro, A.; Fadini, G.P., “Cardiovascular effectiveness of human-based vs. Exendin-based glucagon like peptide-1 receptor agonists: A retrospective study in patients with type 2 diabetes,” Eur J Prev Cardiol, vol. 28, no. 1, pp. 22–29, Jan. 2021. [CrossRef]
- Giugliano, D. et al., “GLP-1 receptor agonists and cardiorenal outcomes in type 2 diabetes: an updated meta-analysis of eight CVOTs,” Cardiovasc Diabetol, vol. 20, no. 1, Dec. 2021. [CrossRef]
- Kristensen, S.L. et al., “Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials,” Lancet Diabetes Endocrinol, vol. 7, no. 10, pp. 776–785, Oct. 2019. [CrossRef]
- Sattar, N. et al., “Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials,” Lancet Diabetes Endocrinol, vol. 9, no. 10, pp. 653–662, Oct. 2021. [CrossRef]
- Binu, A.J.; Kapoor, N., “Understanding Diabetic Cardiomyopathy: Insulin Resistance and Beyond,” Heart Int, vol. 18, no. 2, pp. 7–13, Oct. 2024. [CrossRef]
- Sokos, G.G.; Nikolaidis, L.A.; Mankad, S.; Elahi, D.; Shannon, R.P., “Glucagon-Like Peptide-1 Infusion Improves Left Ventricular Ejection Fraction and Functional Status in Patients With Chronic Heart Failure,” J Card Fail, vol. 12, no. 9, pp. 694–699, Dec. 2006. [CrossRef]
- Margulies, K.B. et al., “Effects of liraglutide on clinical stability among patients with advanced heart failure and reduced ejection fraction: A randomized clinical trial,” JAMA - Journal of the American Medical Association, vol. 316, no. 5, pp. 500–508, Aug. 2016. [CrossRef]
- Lepore, J.J. et al., “Effects of the Novel Long-Acting GLP-1 Agonist, Albiglutide, on Cardiac Function, Cardiac Metabolism, and Exercise Capacity in Patients With Chronic Heart Failure and Reduced Ejection Fraction,” JACC Heart Fail, vol. 4, no. 7, pp. 559–566, Jul. 2016. [CrossRef]
- Jorsal, A. et al., “Effect of liraglutide, a glucagon-like peptide-1 analogue, on left ventricular function in stable chronic heart failure patients with and without diabetes (LIVE)—a multicentre, double-blind, randomised, placebo-controlled trial,” Eur J Heart Fail, vol. 19, no. 1, pp. 69–77, Jan. 2017. [CrossRef]
- Kamel, R.; Leroy, J.; Vandecasteele, G.; Fischmeister, R., “Cyclic nucleotide phosphodiesterases as therapeutic targets in cardiac hypertrophy and heart failure,” Feb. 01, 2023, Nature Research. [CrossRef]
- Prausmüller, S. et al., “Obesity in heart failure with preserved ejection fraction with and without diabetes: risk factor or innocent bystander?,” Eur J Prev Cardiol, vol. 30, pp. 1247–1254, May 2023. [CrossRef]
- Powell-Wiley, T.M. et al., “Obesity and Cardiovascular Disease A Scientific Statement From the American Heart Association,” May 25, 2021, Lippincott Williams and Wilkins. [CrossRef]
- Lincoff, A.M. et al., “Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes,” New England Journal of Medicine, vol. 389, no. 24, pp. 2221–2232, Dec. 2023. [CrossRef]
- Kosiborod, M.N. et al., “Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity,” New England Journal of Medicine, vol. 389, no. 12, pp. 1069–1084, Sep. 2023. [CrossRef]
- Kosiborod, M.N. et al., “Semaglutide in Patients with Obesity-Related Heart Failure and Type 2 Diabetes,” New England Journal of Medicine, vol. 390, no. 15, pp. 1394–1407, Apr. 2024. [CrossRef]
- Shah, S.J. et al., “Semaglutide and diuretic use in obesity-related heart failure with preserved ejection fraction: a pooled analysis of the STEP-HFpEF and STEP-HFpEF-DM trials,” Eur Heart J, vol. 45, pp. 3254–3269, May 2024. [CrossRef]
- Pratley, R.E. et al., “Effects of Semaglutide on Heart Failure Outcomes in Diabetes and Chronic Kidney Disease in the FLOW Trial,” J Am Coll Cardiol, Oct. 2024. [CrossRef]
- Kosiborod, M.N. et al., “Semaglutide versus placebo in patients with heart failure and mildly reduced or preserved ejection fraction: a pooled analysis of the SELECT, FLOW, STEP-HFpEF, and STEP-HFpEF DM randomised trials,” The Lancet, vol. 404, no. 10456, pp. 949–961, Sep. 2024. [CrossRef]
- Packer, M. et al., “Tirzepatide for Heart Failure with Preserved Ejection Fraction and Obesity.,” N Engl J Med, Nov. 2024. [CrossRef]
- Siddiqui, H.F. et al., “The effect of GLP-1 receptor agonists on cardiac remodeling in heart failure patients with preserved and reduced ejection fraction: a systematic review and meta-analysis,” 2025, Springer. [CrossRef]
- Huixing, L.; Di, F.; Daoquan, P., “Effect of Glucagon-like Peptide-1 Receptor Agonists on Prognosis of Heart Failure and Cardiac Function: A Systematic Review and Meta-analysis of Randomized Controlled Trials,” Clin Ther, vol. 45, no. 1, pp. 17–30, Jan. 2023. [CrossRef]
- Avogaro, A.; Azzolina, D.; Gregori, D.; De Kreutzenberg, S.; Fadini, G.P.; Mannucci, E., “The effect of GLP-1 receptor agonists on N-terminal pro-brain natriuretic peptide. A scoping review and metanalysis,” Jun. 15, 2022, Elsevier Ireland Ltd. [CrossRef]
- Yagi, K. et al., “Diastolic cardiac function improvement by liraglutide is mainly body weight reduction dependent but independently contributes to B-type natriuretic peptide reduction in patients with type 2 diabetes with preserved ejection fraction,” J Diabetes Res, vol. 2021, 2021. [CrossRef]
- Wang, T.Y.; Yang, Q.; Cheng, X.Y.; Ding, J.C.; Hu, P.F., “Beyond weight loss: the potential of glucagon-like peptide-1 receptor agonists for treating heart failure with preserved ejection fraction,” Jan. 01, 2024, Springer. [CrossRef]
- Packer, M., “Epicardial Adipose Tissue May Mediate Deleterious Effects of Obesity and Inflammation on the Myocardium,” May 22, 2018, Elsevier USA. [CrossRef]
- Bray, J.J.H. et al., “Glucagon-like peptide-1 receptor agonists improve biomarkers of inflammation and oxidative stress: A systematic review and meta-analysis of randomised controlled trials,” Diabetes Obes Metab, vol. 23, no. 8, pp. 1806–1822, Aug. 2021. [CrossRef]
- Packer, M., “SGLT2 inhibitors: role in protective reprogramming of cardiac nutrient transport and metabolism,” Jul. 01, 2023, Nature Research. [CrossRef]
- Pandey, A.K. et al., “Mechanisms of benefits of sodium-glucose cotransporter 2 inhibitors in heart failure with preserved ejection fraction,” Oct. 01, 2023, Oxford University Press. [CrossRef]
- Gallo, G.; Volpe, M., “Potential Mechanisms of the Protective Effects of the Cardiometabolic Drugs Type-2 Sodium–Glucose Transporter Inhibitors and Glucagon-like Peptide-1 Receptor Agonists in Heart Failure,” Mar. 01, 2024, Multidisciplinary Digital Publishing Institute (MDPI). [CrossRef]
- Ng, A.C.T.; Delgado, V.; Borlaug, B.A.; Bax, J.J., “Diabesity: the combined burden of obesity and diabetes on heart disease and the role of imaging,” Apr. 01, 2021, Nature Research. [CrossRef]
- Chiang, C.-H. et al., “Glucagon-like peptide-1 Receptor Agonists and Gastrointestinal Adverse Events: a Systematic Review and Meta-analysis,” Gastroenterology, Jun. 2025. [CrossRef]
- Cai, C.X. et al., “Semaglutide and Nonarteritic Anterior Ischemic Optic Neuropathy,” JAMA Ophthalmol, 2025. [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



