Submitted:
06 August 2025
Posted:
07 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Ethics Statement
Viruses and Vaccines
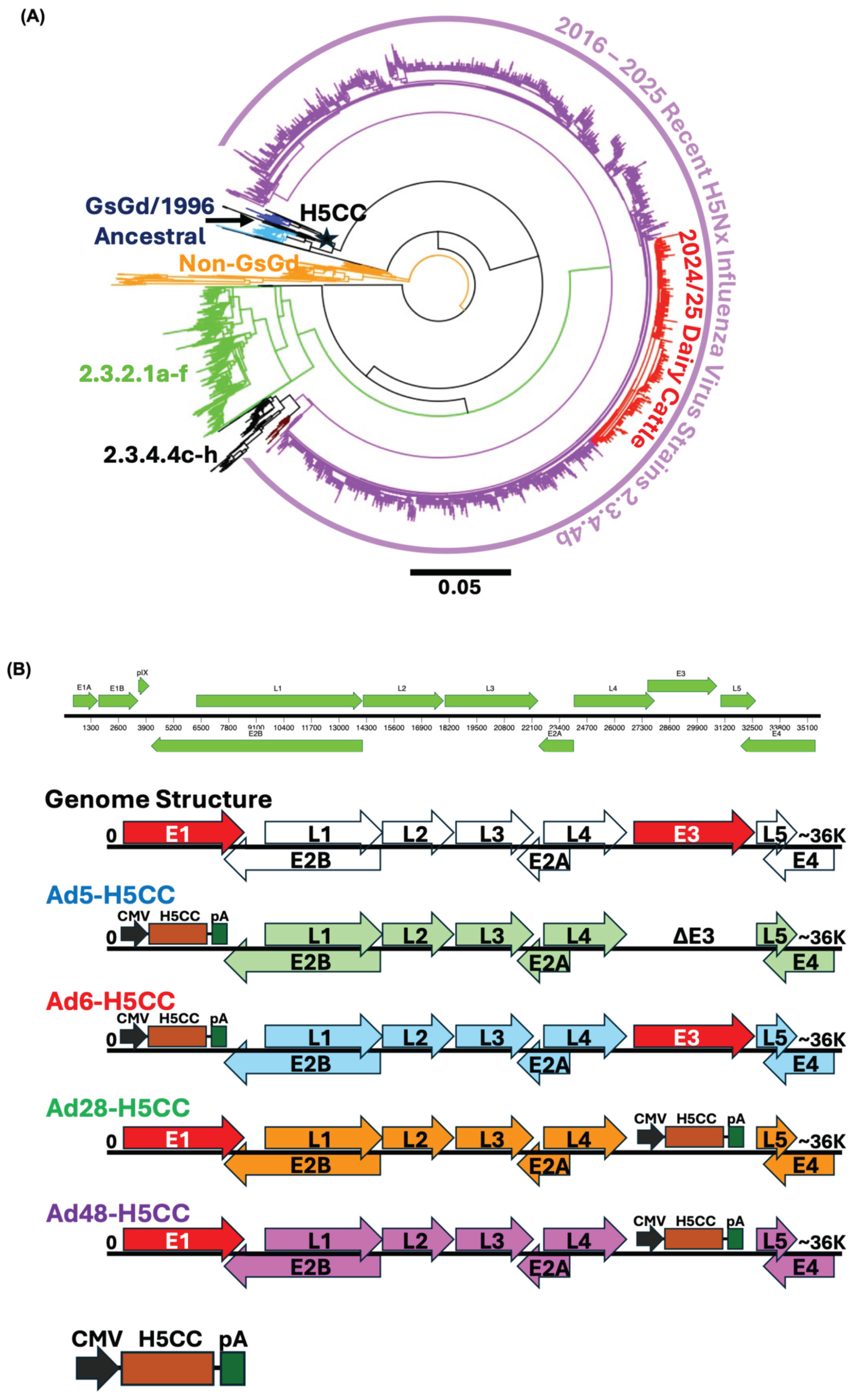
Centralized Genes Construction and Adenovirus Vaccines
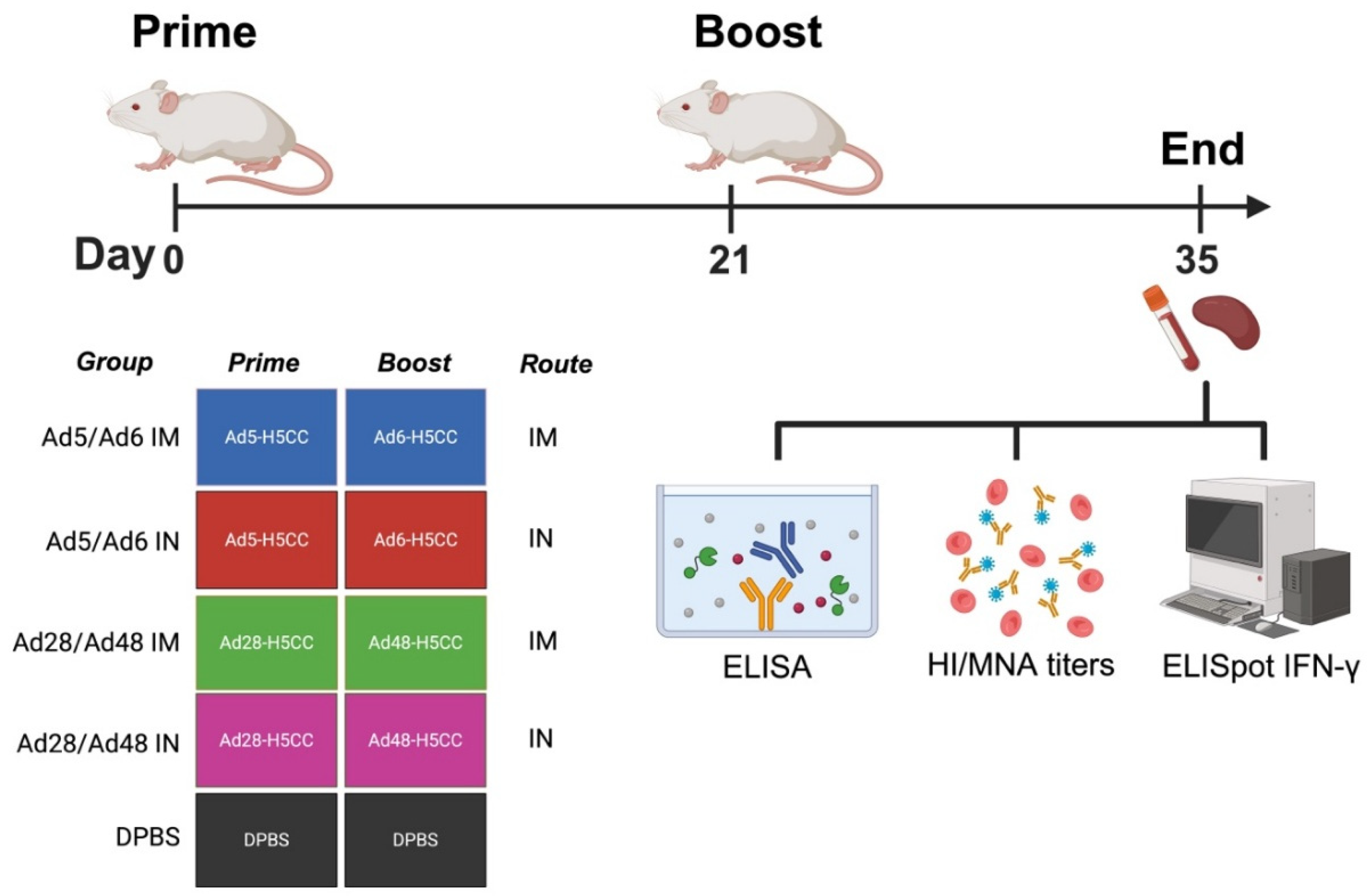
Immune Correlates Study in Mice
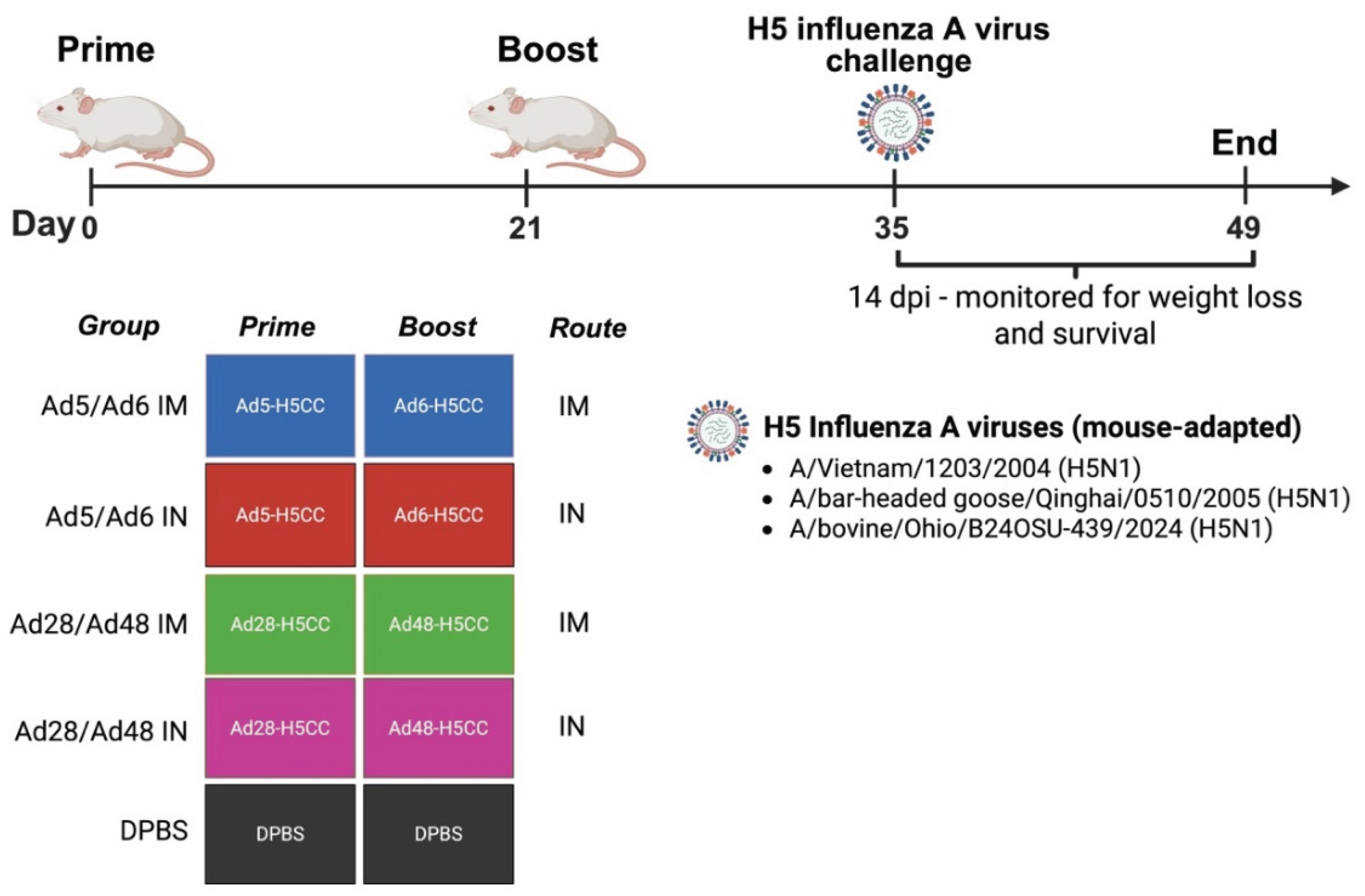
Protective Efficacy Against H5 Influenza A Viruses Challenge in Mice
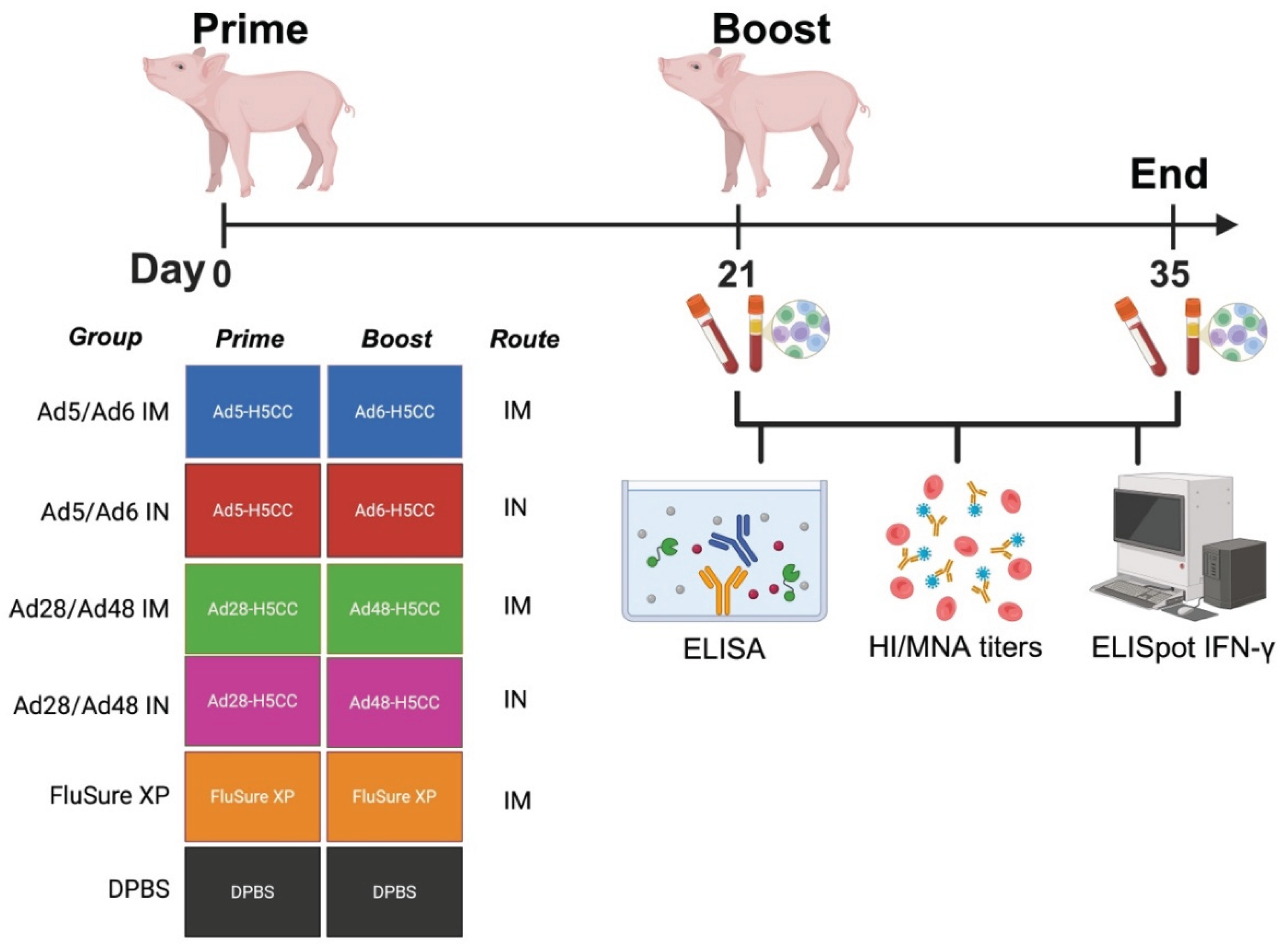
Immune Correlates Study in Swine
Hemagglutination Inhibition (HI) Assay
Microneutralization Assay (MNA)
Antibody Responses as Measured by ELISA
Enzyme-Linked Immunospot (ELISpot) Assay
Statistical Analysis
3. Results
3.1. Construction and Quality Control for Ad-H5CC Vaccines
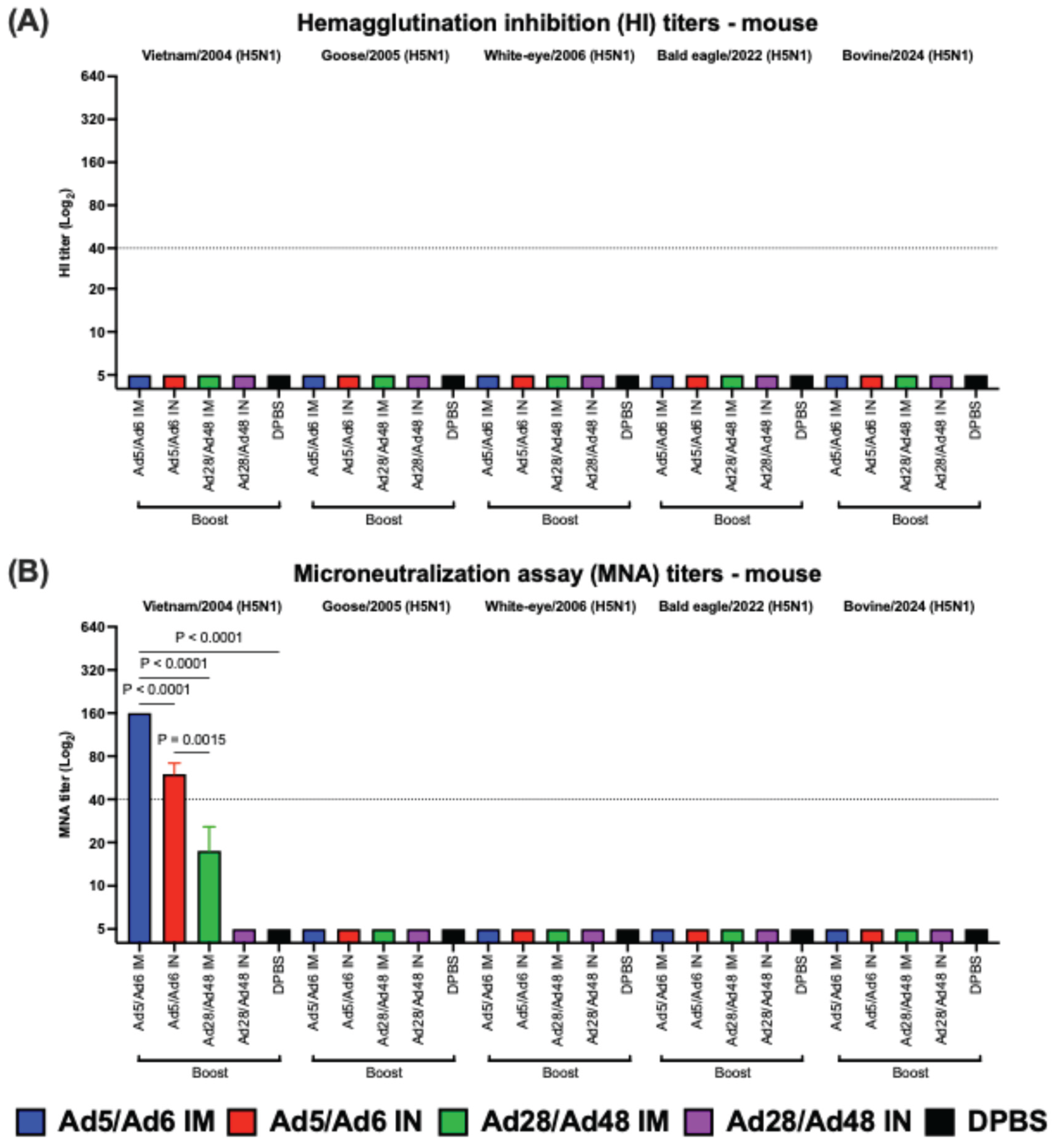
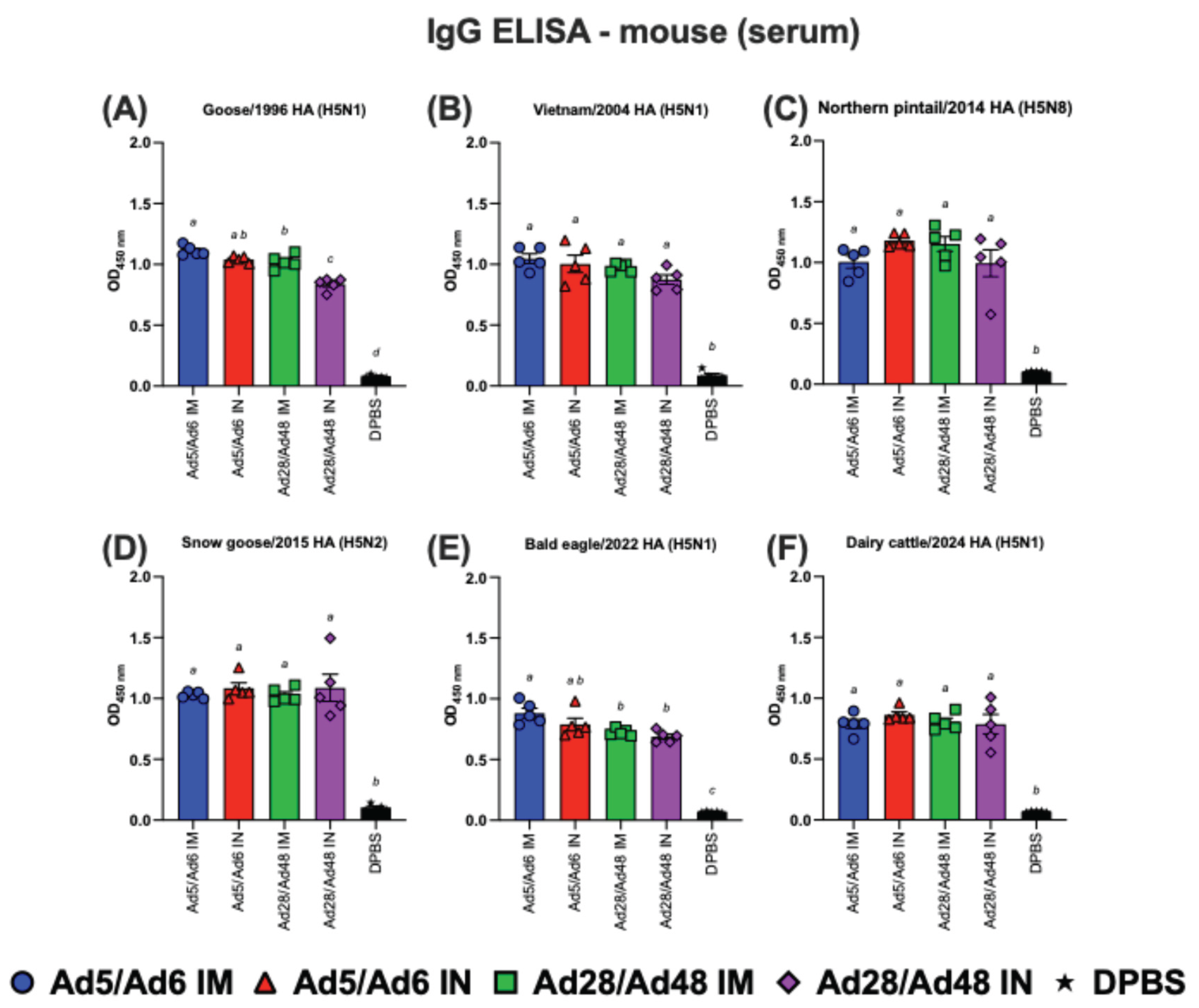
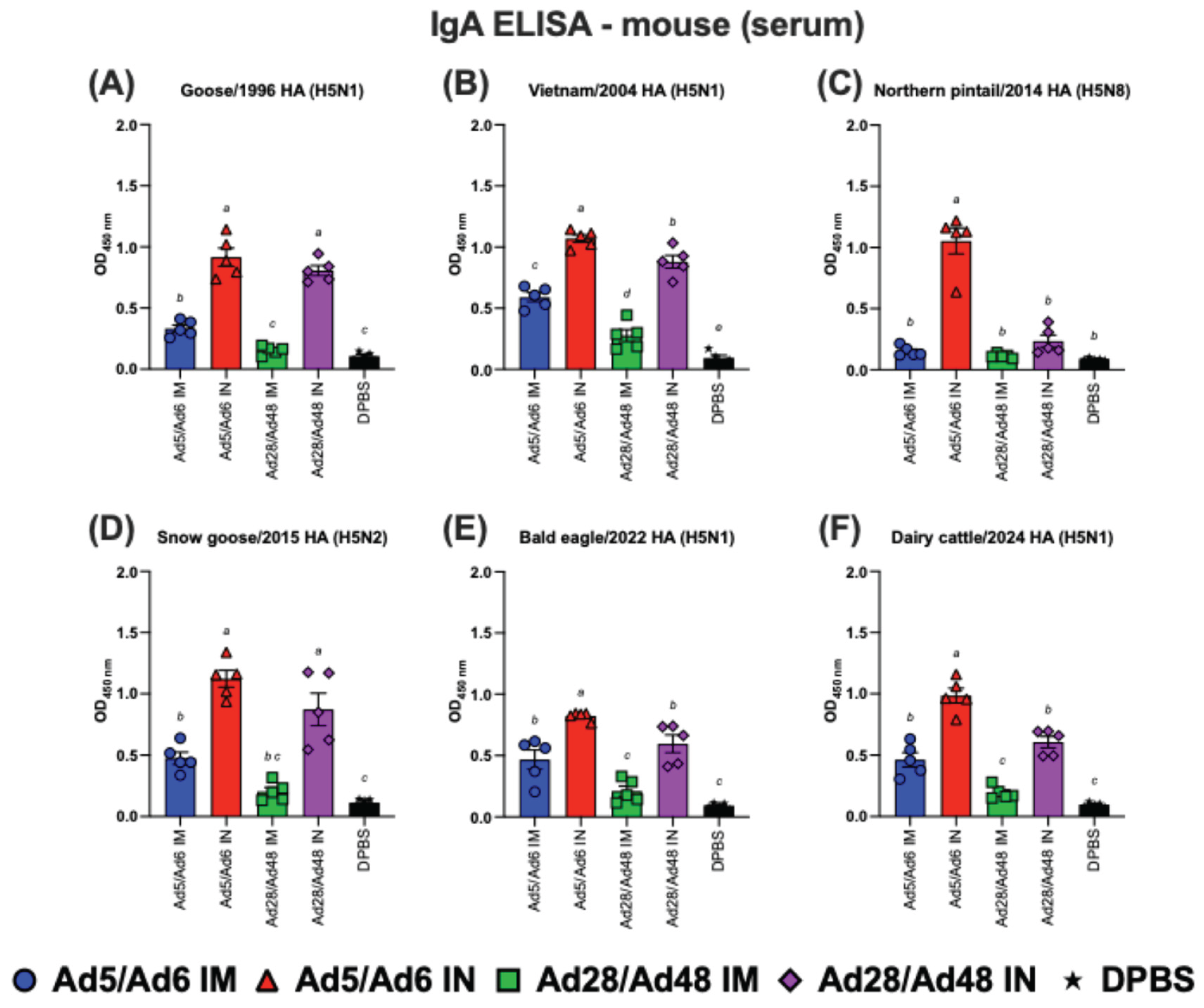
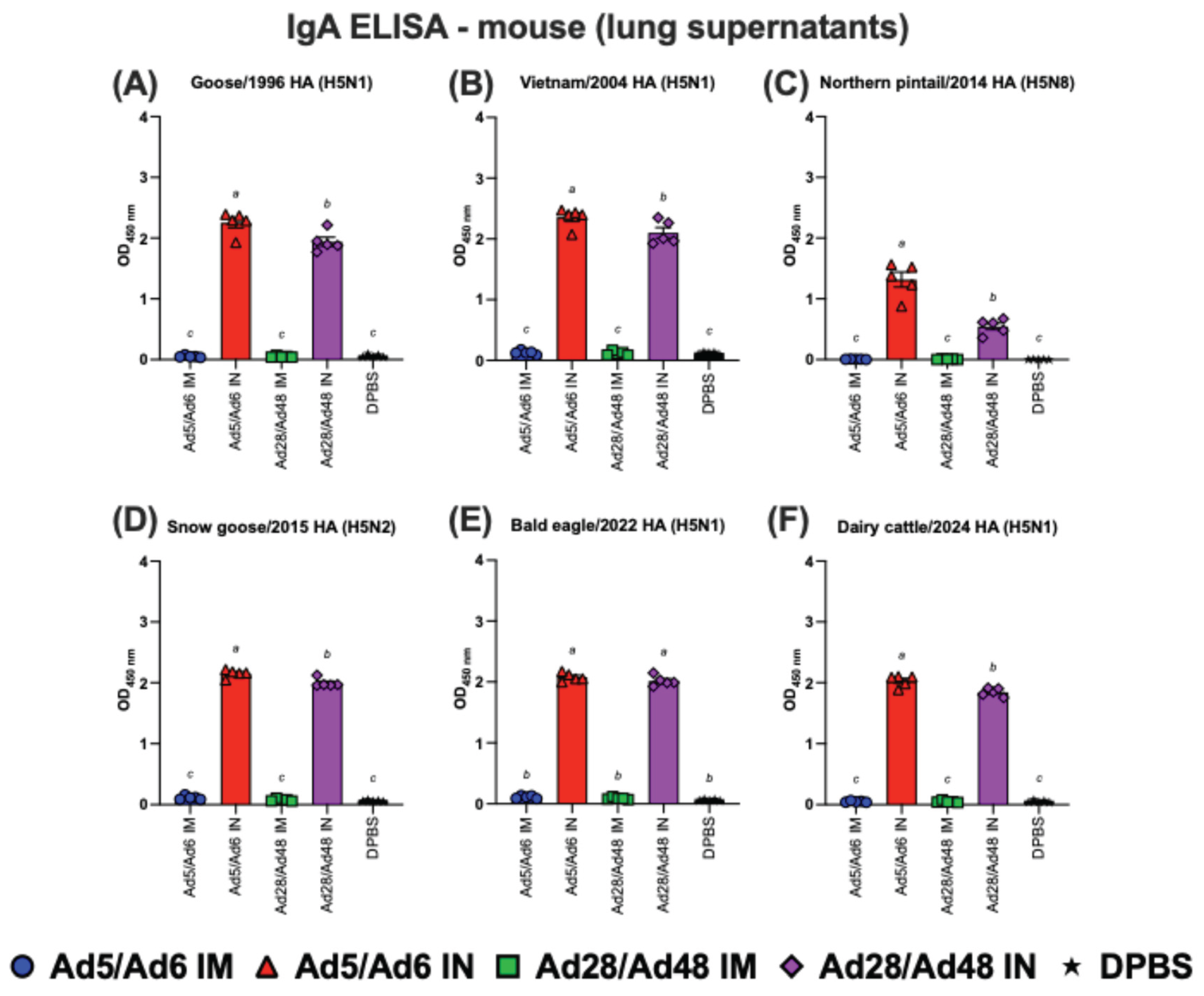
3.2. Systemic and Mucosal Immune Responses in Vaccinated Mice
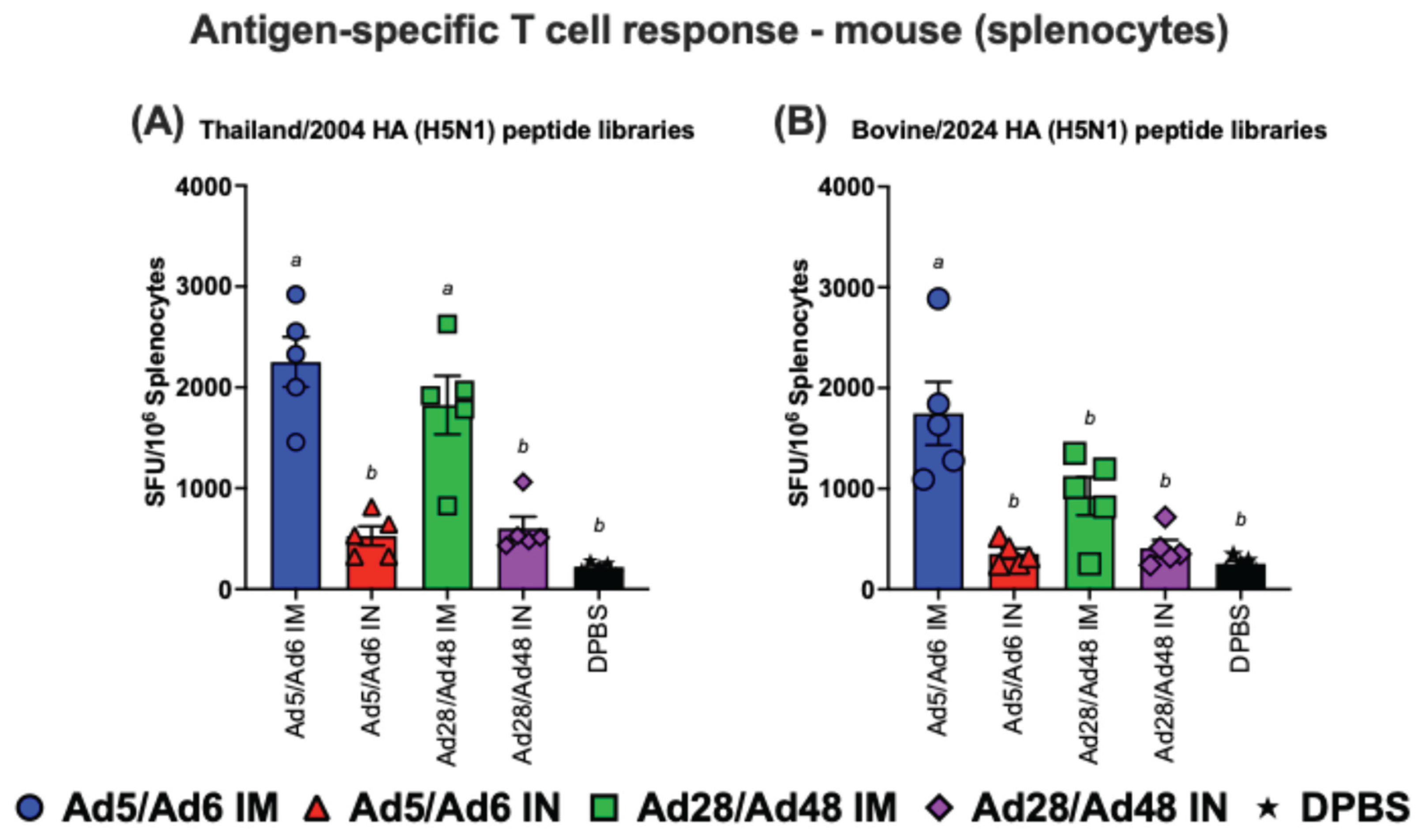
3.3. Cross-Reactive T Cell Responses of Immunized Mice Against H5-HA Peptide Libraries
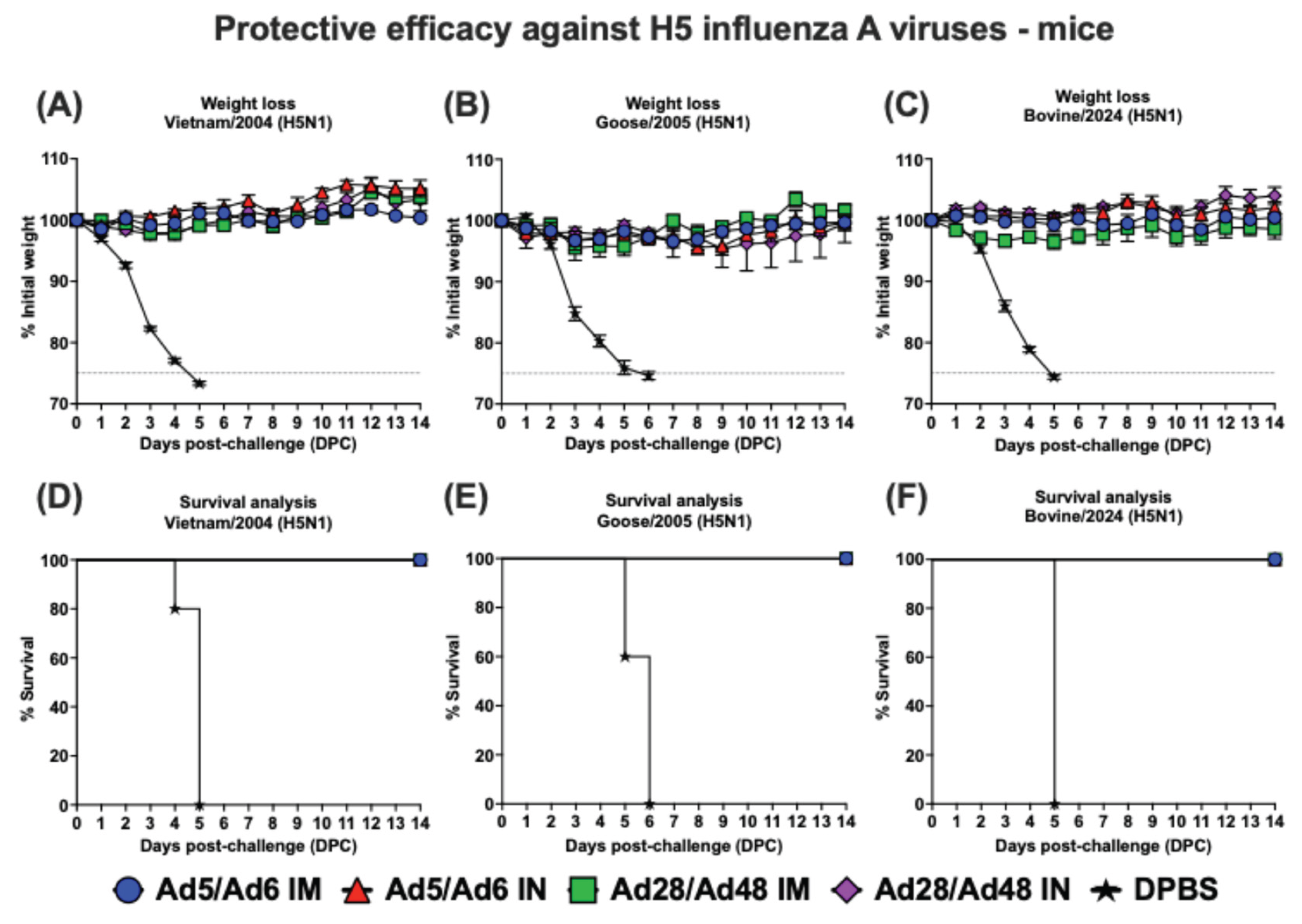
3.4. Protective Efficacy Against H5 Influenza A Virus Challenge in Mice
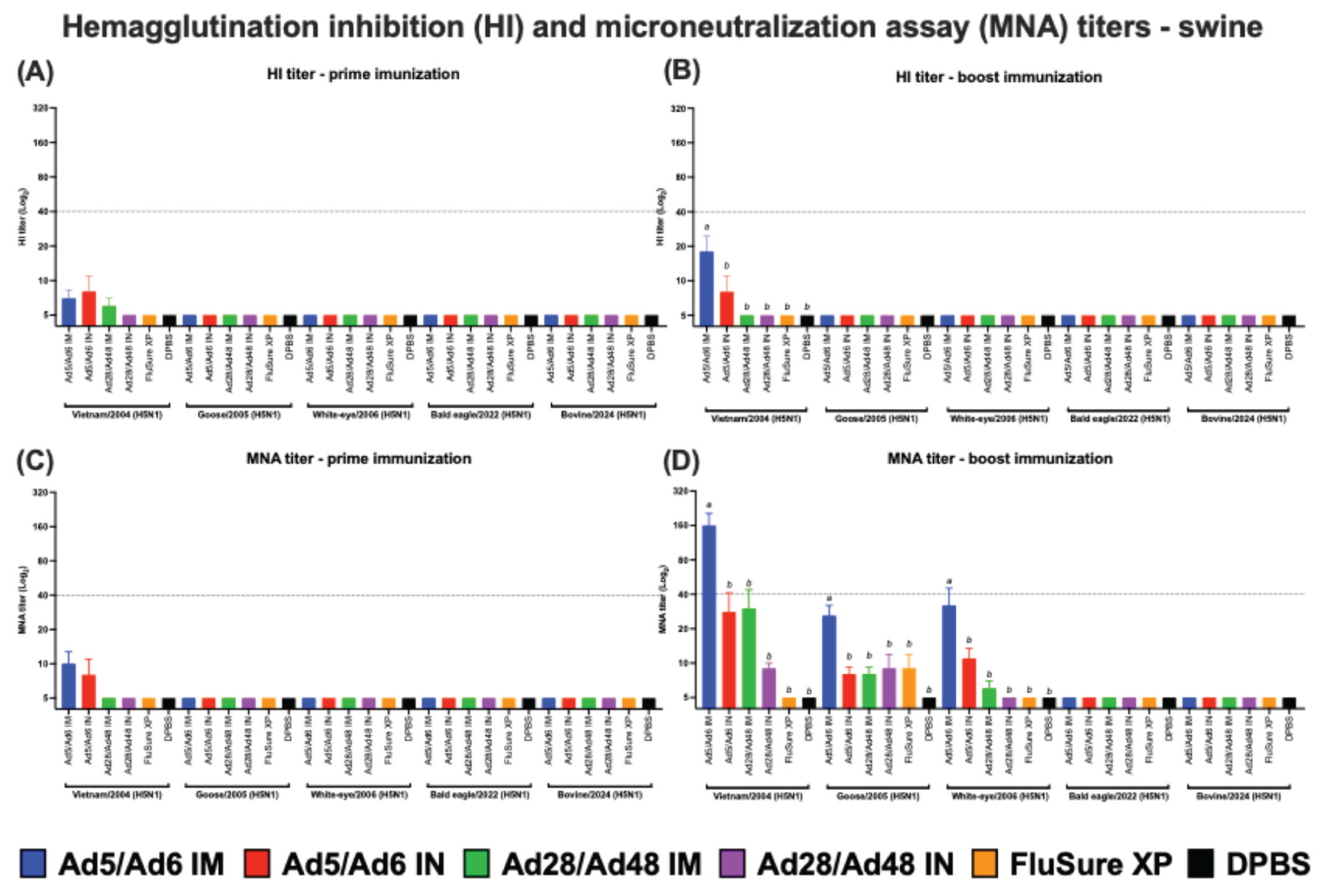
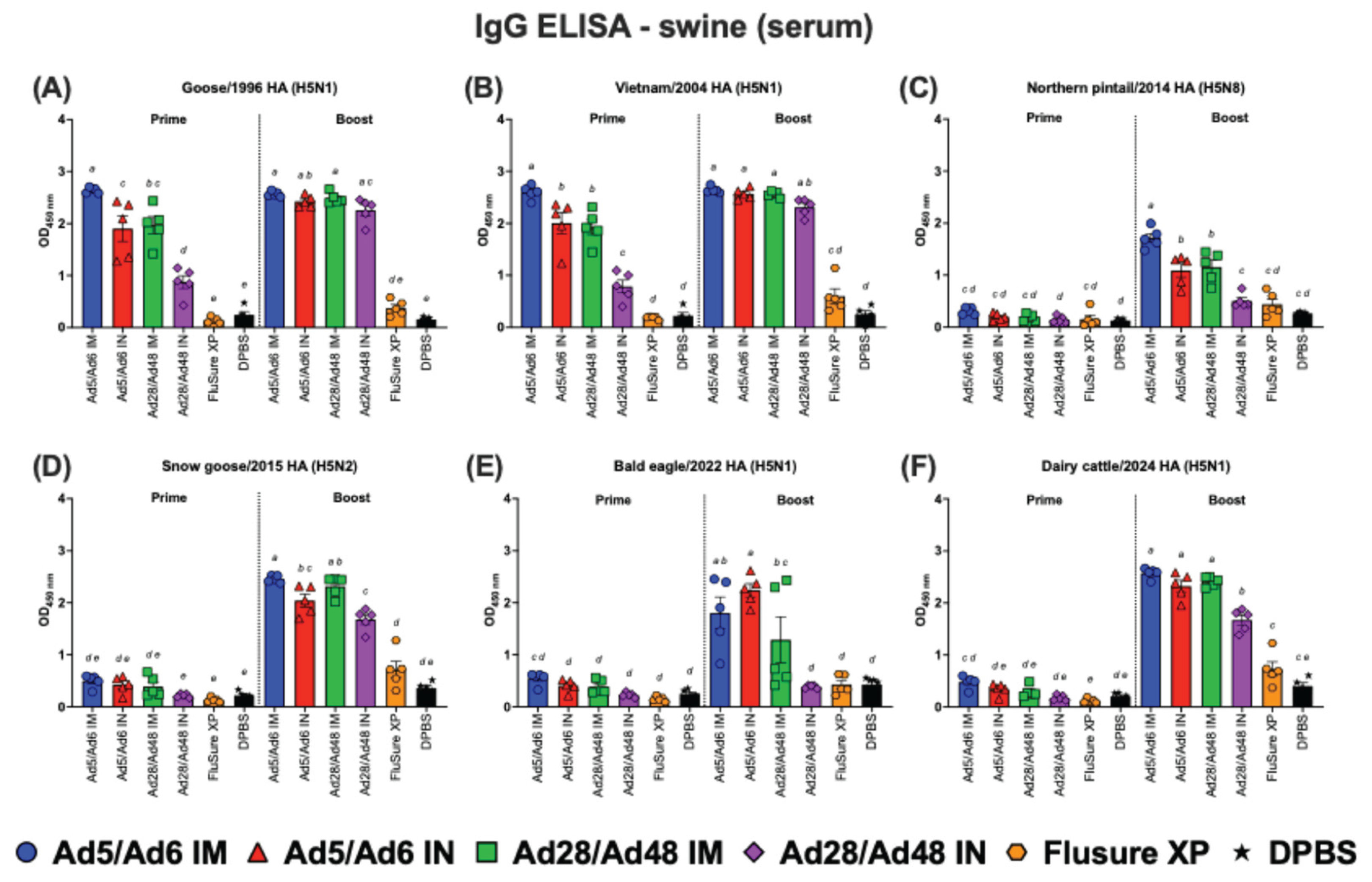
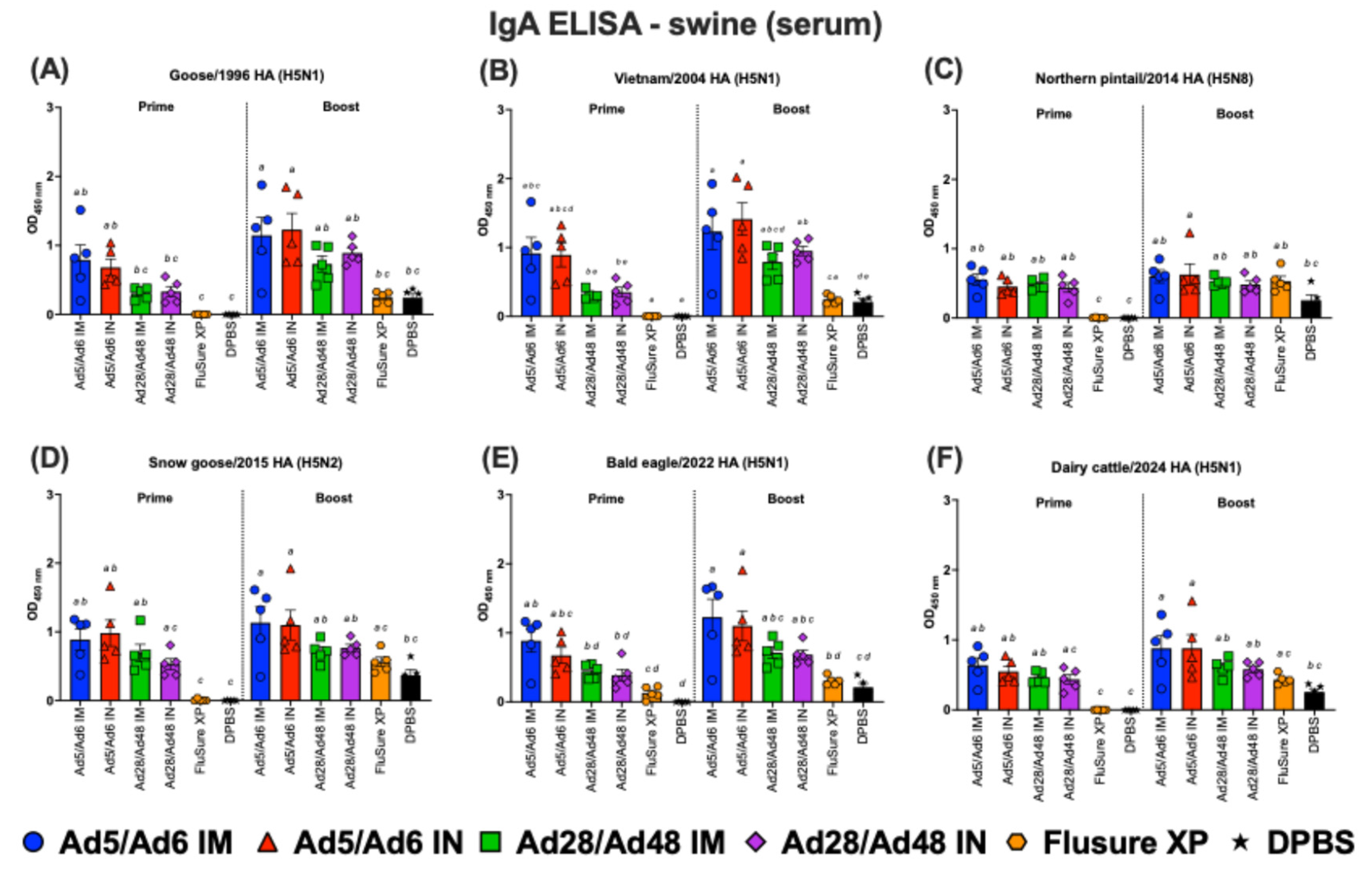
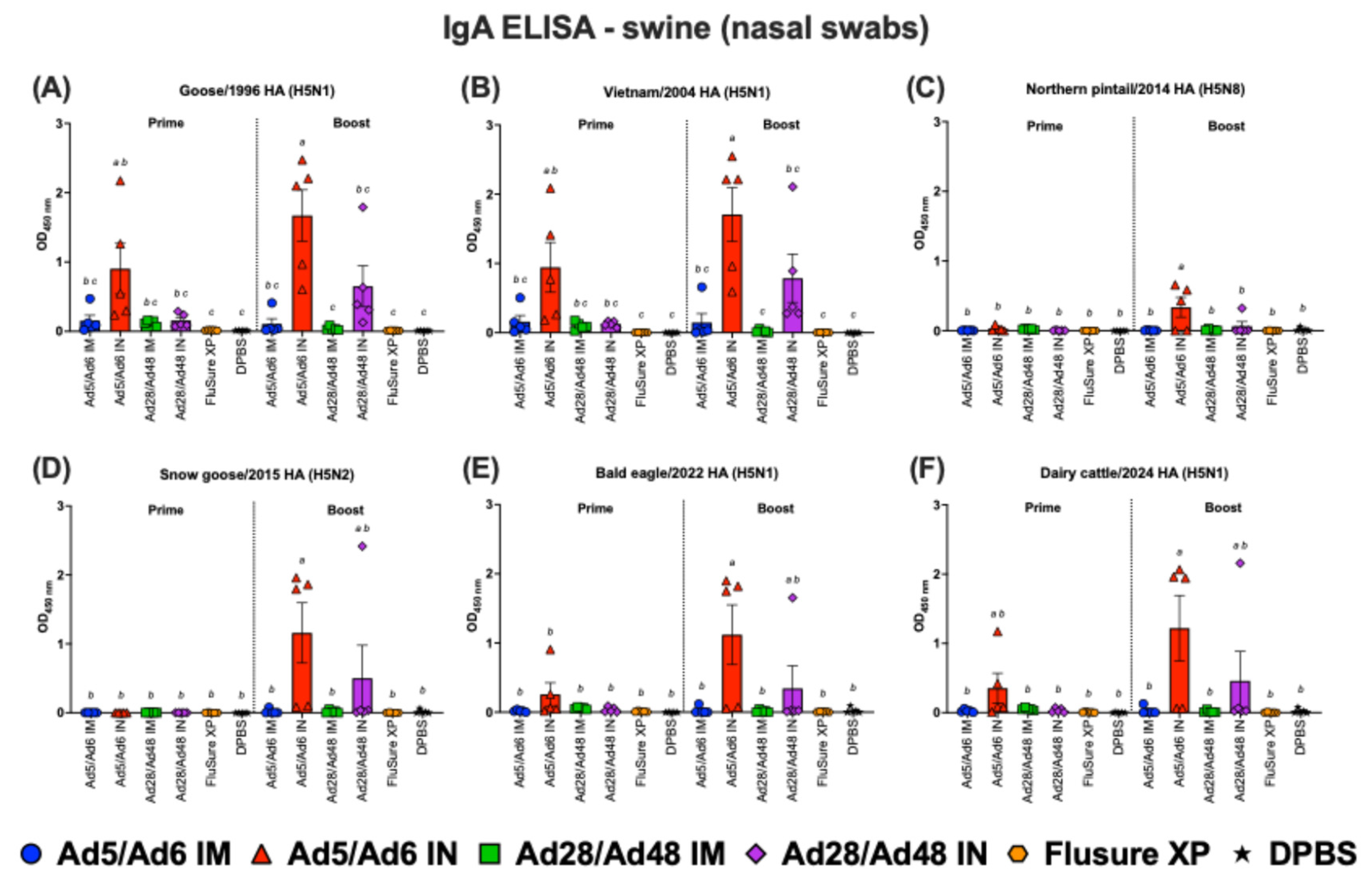
3.2. Systemic and Mucosal Immune Responses in Vaccinated Swine
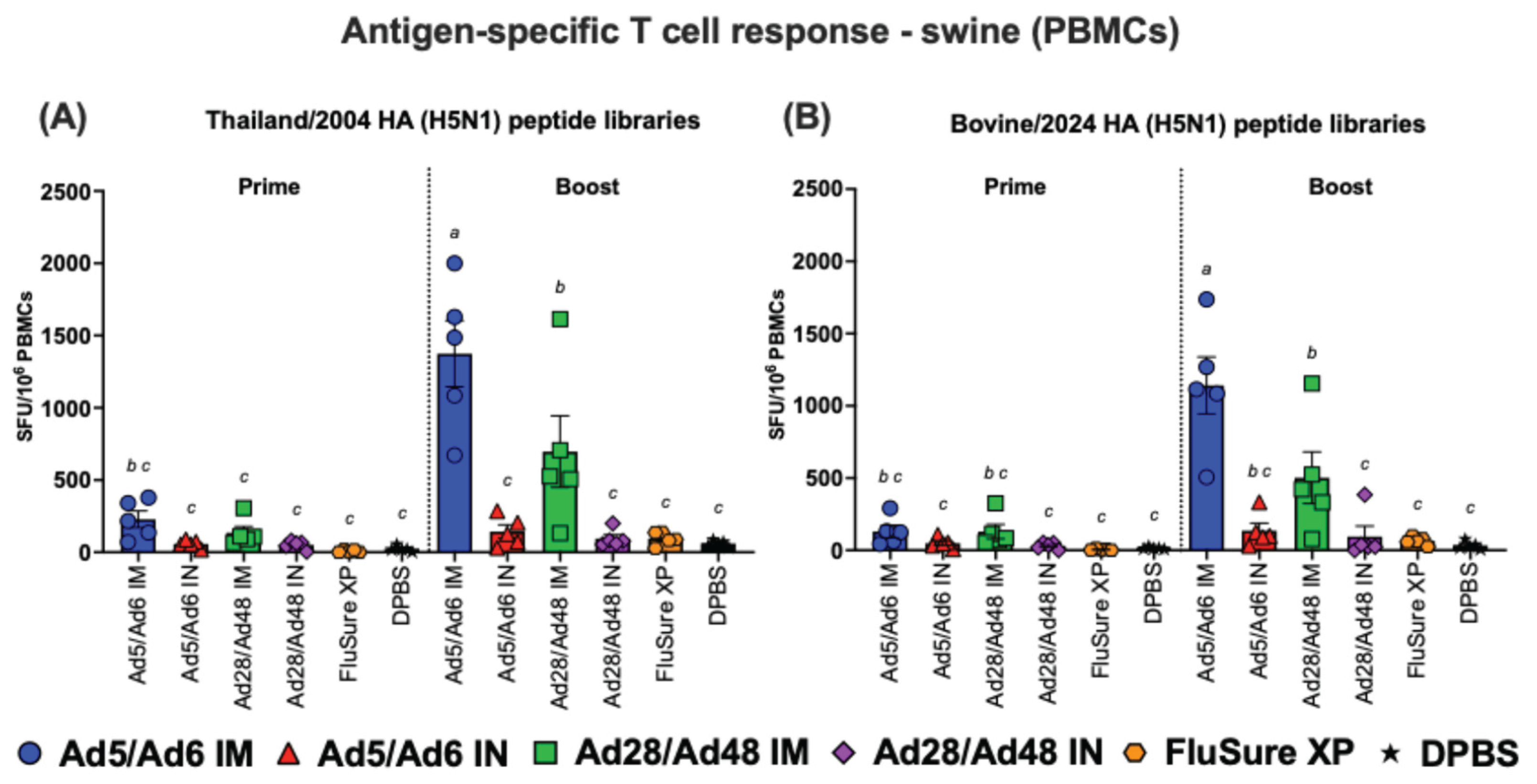
3.3. Cross-Reactive T Cell Responses of Immunized Mice Against H5-HA Peptide Libraries
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Bruno, A.; de Mora, D.; Olmedo, M.; Garces, J.; Marzal, A.; Garcia-Bereguiain, M.A. Highly pathogenic avian influenza A (H5N1) virus outbreaks in South America in 2022-2024: a comprehensive review of an ongoing panzootic. Virology 2025, 610, 110602. [CrossRef]
- Lycett, S.J.; Pohlmann, A.; Staubach, C.; Caliendo, V.; Woolhouse, M.; Beer, M.; Kuiken, T.; Global Consortium for, H.N.; Related Influenza, V. Genesis and spread of multiple reassortants during the 2016/2017 H5 avian influenza epidemic in Eurasia. Proc Natl Acad Sci U S A 2020, 117, 20814-20825. [CrossRef]
- Bi, Y.; Chen, J.; Zhang, Z.; Li, M.; Cai, T.; Sharshov, K.; Susloparov, I.; Shestopalov, A.; Wong, G.; He, Y.; et al. Highly pathogenic avian influenza H5N1 Clade 2.3.2.1c virus in migratory birds, 2014-2015. Virol Sin 2016, 31, 300-305. [CrossRef]
- Lee, D.H.; Torchetti, M.K.; Hicks, J.; Killian, M.L.; Bahl, J.; Pantin-Jackwood, M.; Swayne, D.E. Transmission Dynamics of Highly Pathogenic Avian Influenza Virus A(H5Nx) Clade 2.3.4.4, North America, 2014-2015. Emerg Infect Dis 2018, 24, 1840-1848. [CrossRef]
- Yang, J.; Zhang, C.; Yuan, Y.; Sun, J.; Lu, L.; Sun, H.; Sun, H.; Chu, D.; Qin, S.; Chen, J.; et al. Novel Avian Influenza Virus (H5N1) Clade 2.3.4.4b Reassortants in Migratory Birds, China. Emerg Infect Dis 2023, 29, 1244-1249. [CrossRef]
- Guilfoyle, K.; Mirolo, M.; de Waal, L.; van Amerongen, G.; van der Net, G.; Stork, T.; Lombardo, M.S.; Baumgartner, W.; Bjarnason, A.; Johannsdottir, H.B.; et al. Highly Pathogenic Avian Influenza Virus A/H5N1 subclade 2.3.4.4 b isolated from a European grey seal (Halichoerus grypus) is highly virulent in ferrets. J Infect Dis 2025. [CrossRef]
- Naraharisetti, R.; Weinberg, M.; Stoddard, B.; Stobierski, M.G.; Dodd, K.A.; Wineland, N.; Beal, M.; Morse, J.; Hatter, S.; Sledge, D.; et al. Highly Pathogenic Avian Influenza A(H5N1) Virus Infection of Indoor Domestic Cats Within Dairy Industry Worker Households - Michigan, May 2024. MMWR Morb Mortal Wkly Rep 2025, 74, 61-65. [CrossRef]
- Himsworth, C.G.; Caleta, J.M.; Jassem, A.N.; Yang, K.C.; Zlosnik, J.E.A.; Tyson, J.R.; Wilson, L.; Kuchinski, K.S.; Giacinti, J.; Willie, M.; et al. Highly Pathogenic Avian Influenza A(H5N1) in Wild Birds and a Human, British Columbia, Canada, 2024. Emerg Infect Dis 2025, 31, 1216-1221. [CrossRef]
- Rodriguez, Z.; Picasso-Risso, C.; O’Connor, A.; Ruegg, P.L. Hot topic: Epidemiological and clinical aspects of highly pathogenic avian influenza H5N1 in dairy cattle. JDS Commun 2024, 5, S8-S12. [CrossRef]
- Kamel, M.; Aleya, S.; Almagharbeh, W.T.; Aleya, L.; Abdel-Daim, M.M. The emergence of highly pathogenic avian influenza H5N1 in dairy cattle: implications for public health, animal health, and pandemic preparedness. Eur J Clin Microbiol Infect Dis 2025. [CrossRef]
- Vincent, A.; Awada, L.; Brown, I.; Chen, H.; Claes, F.; Dauphin, G.; Donis, R.; Culhane, M.; Hamilton, K.; Lewis, N.; et al. Review of influenza A virus in swine worldwide: a call for increased surveillance and research. Zoonoses Public Health 2014, 61, 4-17. [CrossRef]
- Ito, T.; Couceiro, J.N.; Kelm, S.; Baum, L.G.; Krauss, S.; Castrucci, M.R.; Donatelli, I.; Kida, H.; Paulson, J.C.; Webster, R.G.; et al. Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J Virol 1998, 72, 7367-7373. [CrossRef]
- Nelli, R.K.; Kuchipudi, S.V.; White, G.A.; Perez, B.B.; Dunham, S.P.; Chang, K.C. Comparative distribution of human and avian type sialic acid influenza receptors in the pig. BMC Vet Res 2010, 6, 4. [CrossRef]
- Arruda, B.; Baker, A.L.V.; Buckley, A.; Anderson, T.K.; Torchetti, M.; Bergeson, N.H.; Killian, M.L.; Lantz, K. Divergent Pathogenesis and Transmission of Highly Pathogenic Avian Influenza A(H5N1) in Swine. Emerg Infect Dis 2024, 30, 738-751. [CrossRef]
- Kwon, T.; Trujillo, J.D.; Carossino, M.; Lyoo, E.L.; McDowell, C.D.; Cool, K.; Matias-Ferreyra, F.S.; Jeevan, T.; Morozov, I.; Gaudreault, N.N.; et al. Pigs are highly susceptible to but do not transmit mink-derived highly pathogenic avian influenza virus H5N1 clade 2.3.4.4b. Emerg Microbes Infect 2024, 13, 2353292. [CrossRef]
- Root, J.J.; Porter, S.M.; Lenoch, J.B.; Ellis, J.W.; Bosco-Lauth, A.M. Susceptibilities and viral shedding of peridomestic wildlife infected with clade 2.3.4.4b highly pathogenic avian influenza virus (H5N1). Virology 2024, 600, 110231. [CrossRef]
- Abente, E.J.; Rajao, D.S.; Santos, J.; Kaplan, B.S.; Nicholson, T.L.; Brockmeier, S.L.; Gauger, P.C.; Perez, D.R.; Vincent, A.L. Comparison of Adjuvanted-Whole Inactivated Virus and Live-Attenuated Virus Vaccines against Challenge with Contemporary, Antigenically Distinct H3N2 Influenza A Viruses. J Virol 2018, 92. [CrossRef]
- Rajao, D.S.; Chen, H.; Perez, D.R.; Sandbulte, M.R.; Gauger, P.C.; Loving, C.L.; Shanks, G.D.; Vincent, A. Vaccine-associated enhanced respiratory disease is influenced by haemagglutinin and neuraminidase in whole inactivated influenza virus vaccines. J Gen Virol 2016, 97, 1489-1499. [CrossRef]
- Gauger, P.C.; Loving, C.L.; Khurana, S.; Lorusso, A.; Perez, D.R.; Kehrli, M.E., Jr.; Roth, J.A.; Golding, H.; Vincent, A.L. Live attenuated influenza A virus vaccine protects against A(H1N1)pdm09 heterologous challenge without vaccine associated enhanced respiratory disease. Virology 2014, 471-473, 93-104. [CrossRef]
- Gauger, P.C.; Vincent, A.L.; Loving, C.L.; Henningson, J.N.; Lager, K.M.; Janke, B.H.; Kehrli, M.E., Jr.; Roth, J.A. Kinetics of lung lesion development and pro-inflammatory cytokine response in pigs with vaccine-associated enhanced respiratory disease induced by challenge with pandemic (2009) A/H1N1 influenza virus. Vet Pathol 2012, 49, 900-912. [CrossRef]
- Abbink, P.; Lemckert, A.A.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. Journal of virology 2007, 81, 4654-4663.
- Lasaro, M.O.; Ertl, H.C. New insights on adenovirus as vaccine vectors. Mol Ther 2009, 17, 1333-1339.
- Weaver, E.A. Vaccines within vaccines: the use of adenovirus types 4 and 7 as influenza vaccine vectors. Human vaccines & immunotherapeutics 2014, 10, 544-556.
- Weaver, E.A.; Nehete, P.N.; Nehete, B.P.; Yang, G.; Buchl, S.J.; Hanley, P.W.; Palmer, D.; Montefiori, D.C.; Ferrari, G.; Ng, P.; et al. Comparison of Systemic and Mucosal Immunization with Helper-Dependent Adenoviruses for Vaccination against Mucosal Challenge with SHIV. PloS one 2013, in press.
- Weaver, E.A.; Rubrum, A.M.; Webby, R.J.; Barry, M.A. Protection against divergent influenza H1N1 virus by a centralized influenza hemagglutinin. PloS one 2011, 6, e18314. [CrossRef]
- Harro, C.; Sun, X.; Stek, J.E.; Leavitt, R.Y.; Mehrotra, D.V.; Wang, F.; Bett, A.J.; Casimiro, D.R.; Shiver, J.W.; DiNubile, M.J.; et al. Safety and immunogenicity of the Merck adenovirus serotype 5 (MRKAd5) and MRKAd6 human immunodeficiency virus type 1 trigene vaccines alone and in combination in healthy adults. Clin Vaccine Immunol 2009, 16, 1285-1292. [CrossRef]
- Camacho, Z.T.; Turner, M.A.; Barry, M.A.; Weaver, E.A. CD46-mediated transduction of a species D adenovirus vaccine improves mucosal vaccine efficacy. Human gene therapy 2014, 25, 364-374. [CrossRef]
- Lingel, A.; Bullard, B.L.; Weaver, E.A. Efficacy of an Adenoviral Vectored Multivalent Centralized Influenza Vaccine. Scientific Reports 2017, 7, 14912. [CrossRef]
- Webby, R.J.; Weaver, E.A. Centralized Consensus Hemagglutinin Genes Induce Protective Immunity against H1, H3 and H5 Influenza Viruses. PloS one 2015, 10, e0140702. [CrossRef]
- Weaver, E.A.; Barry, M.A. Low seroprevalent species D adenovirus vectors as influenza vaccines. PloS one 2013, 8, e73313. [CrossRef]
- Petro-Turnquist, E.M.; Madapong, A.; Pekarek, M.; Steffen, D.; Weaver, E.A. Epitope-optimized vaccine elicits enduring immunity against swine influenza A virus. Nat Commun 2025, 16, 4046. [CrossRef]
- Ma, W.; Kahn, R.E.; Richt, J.A. The pig as a mixing vessel for influenza viruses: Human and veterinary implications. J Mol Genet Med 2008, 3, 158-166.
- Rose, N.; Herve, S.; Eveno, E.; Barbier, N.; Eono, F.; Dorenlor, V.; Andraud, M.; Camsusou, C.; Madec, F.; Simon, G. Dynamics of influenza A virus infections in permanently infected pig farms: evidence of recurrent infections, circulation of several swine influenza viruses and reassortment events. Vet Res 2013, 44, 72. [CrossRef]
- Smith, G.J.; Vijaykrishna, D.; Bahl, J.; Lycett, S.J.; Worobey, M.; Pybus, O.G.; Ma, S.K.; Cheung, C.L.; Raghwani, J.; Bhatt, S.; et al. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature 2009, 459, 1122-1125. [CrossRef]
- Kaplan, B.S.; Torchetti, M.K.; Lager, K.M.; Webby, R.J.; Vincent, A.L. Absence of clinical disease and contact transmission of HPAI H5NX clade 2.3.4.4 from North America in experimentally infected pigs. Influenza Other Respir Viruses 2017, 11, 464-470. [CrossRef]
- Larsen, D.L.; Karasin, A.; Zuckermann, F.; Olsen, C.W. Systemic and mucosal immune responses to H1N1 influenza virus infection in pigs. Vet Microbiol 2000, 74, 117-131. [CrossRef]
- Nelson, M.I.; Viboud, C.; Vincent, A.L.; Culhane, M.R.; Detmer, S.E.; Wentworth, D.E.; Rambaut, A.; Suchard, M.A.; Holmes, E.C.; Lemey, P. Global migration of influenza A viruses in swine. Nat Commun 2015, 6, 6696. [CrossRef]
- Bullard, B.L.; DeBeauchamp, J.; Pekarek, M.J.; Petro-Turnquist, E.; Vogel, P.; Webby, R.J.; Weaver, E.A. An epitope-optimized human H3N2 influenza vaccine induces broadly protective immunity in mice and ferrets. NPJ Vaccines 2022, 7, 65. [CrossRef]
- Barouch, D.H.; Pau, M.G.; Custers, J.H.; Koudstaal, W.; Kostense, S.; Havenga, M.J.; Truitt, D.M.; Sumida, S.M.; Kishko, M.G.; Arthur, J.C.; et al. Immunogenicity of recombinant adenovirus serotype 35 vaccine in the presence of pre-existing anti-Ad5 immunity. J Immunol 2004, 172, 6290-6297. [CrossRef]
- Zhang, J. Advances and future challenges in recombinant adenoviral vectored H5N1 influenza vaccines. Viruses 2012, 4, 2711-2735. [CrossRef]
- Verschoor, C.P.; Singh, P.; Russell, M.L.; Bowdish, D.M.; Brewer, A.; Cyr, L.; Ward, B.J.; Loeb, M. Microneutralization assay titres correlate with protection against seasonal influenza H1N1 and H3N2 in children. PLoS One 2015, 10, e0131531. [CrossRef]
- Creanga, A.; Gillespie, R.A.; Fisher, B.E.; Andrews, S.F.; Lederhofer, J.; Yap, C.; Hatch, L.; Stephens, T.; Tsybovsky, Y.; Crank, M.C.; et al. A comprehensive influenza reporter virus panel for high-throughput deep profiling of neutralizing antibodies. Nat Commun 2021, 12, 1722. [CrossRef]
- Hu, Z.; Zhao, J.; Zhao, Y.; Fan, X.; Hu, J.; Shi, L.; Wang, X.; Liu, X.; Hu, S.; Gu, M.; et al. Hemagglutinin-Specific Non-neutralizing Antibody Is Essential for Protection Provided by Inactivated and Viral-Vectored H7N9 Avian Influenza Vaccines in Chickens. Front Vet Sci 2019, 6, 482. [CrossRef]
- Linnik, J.; Syedbasha, M.; Hollenstein, Y.; Halter, J.; Egli, A.; Stelling, J. Model-based inference of neutralizing antibody avidities against influenza virus. PLoS Pathog 2022, 18, e1010243. [CrossRef]
- Waldock, J.; Remarque, E.J.; Zheng, L.; Ho, S.; Hoschler, K.; Neumann, B.; Sediri-Schon, H.; Trombetta, C.M.; Montomoli, E.; Marchi, S.; et al. Haemagglutination inhibition and virus microneutralisation serology assays: use of harmonised protocols and biological standards in seasonal influenza serology testing and their impact on inter-laboratory variation and assay correlation: A FLUCOP collaborative study. Front Immunol 2023, 14, 1155552. [CrossRef]
- Gu, C.; Zeng, X.; Song, Y.; Li, Y.; Liu, L.; Kawaoka, Y.; Zhao, D.; Chen, H. Glycosylation and an amino acid insertion in the head of hemagglutinin independently affect the antigenic properties of H5N1 avian influenza viruses. Sci China Life Sci 2019, 62, 76-83. [CrossRef]
- Kaufmann, L.; Syedbasha, M.; Vogt, D.; Hollenstein, Y.; Hartmann, J.; Linnik, J.E.; Egli, A. An Optimized Hemagglutination Inhibition (HI) Assay to Quantify Influenza-specific Antibody Titers. J Vis Exp 2017. [CrossRef]
- Bibby, D.C.; Savanovic, M.; Zhang, J.; Torelli, A.; Jeeninga, R.E.; Gagnon, L.; Harris, S.L. Interlaboratory Reproducibility of Standardized Hemagglutination Inhibition Assays. mSphere 2022, 7, e0095321. [CrossRef]
- Zhou, F.; Hansen, L.; Pedersen, G.; Grodeland, G.; Cox, R. Matrix M Adjuvanted H5N1 Vaccine Elicits Broadly Neutralizing Antibodies and Neuraminidase Inhibiting Antibodies in Humans That Correlate With In Vivo Protection. Front Immunol 2021, 12, 747774. [CrossRef]
- Monto, A.S.; Petrie, J.G.; Cross, R.T.; Johnson, E.; Liu, M.; Zhong, W.; Levine, M.; Katz, J.M.; Ohmit, S.E. Antibody to Influenza Virus Neuraminidase: An Independent Correlate of Protection. J Infect Dis 2015, 212, 1191-1199. [CrossRef]
- Holzer, B.; Martini, V.; Edmans, M.; Tchilian, E. T and B Cell Immune Responses to Influenza Viruses in Pigs. Front Immunol 2019, 10, 98. [CrossRef]
- Braucher, D.R.; Henningson, J.N.; Loving, C.L.; Vincent, A.L.; Kim, E.; Steitz, J.; Gambotto, A.A.; Kehrli, M.E., Jr. Intranasal vaccination with replication-defective adenovirus type 5 encoding influenza virus hemagglutinin elicits protective immunity to homologous challenge and partial protection to heterologous challenge in pigs. Clin Vaccine Immunol 2012, 19, 1722-1729. [CrossRef]
- van Ginkel, F.W.; Tang, D.C.; Gulley, S.L.; Toro, H. Induction of mucosal immunity in the avian Harderian gland with a replication-deficient Ad5 vector expressing avian influenza H5 hemagglutinin. Dev Comp Immunol 2009, 33, 28-34. [CrossRef]
- Fausther-Bovendo, H.; Kobinger, G.P. Pre-existing immunity against Ad vectors: humoral, cellular, and innate response, what’s important? Hum Vaccin Immunother 2014, 10, 2875-2884. [CrossRef]
- Tatsis, N.; Ertl, H.C. Adenoviruses as vaccine vectors. Mol Ther 2004, 10, 616-629. [CrossRef]
- Wesley, R.D.; Lager, K.M. Overcoming maternal antibody interference by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of swine influenza virus. Vet Microbiol 2006, 118, 67-75. [CrossRef]
- Cox, R.J.; Brokstad, K.A. Not just antibodies: B cells and T cells mediate immunity to COVID-19. Nat Rev Immunol 2020, 20, 581-582. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



