Submitted:
05 August 2025
Posted:
07 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. From the Angle of Structures
2.1. The Soup
2.2. Sensory Afferents
2.2.1. Nociceptors
- Peripheral Sensitization
- Substance P (SP)
- Somatostatin (STT)
- Histamine (HIST)
- Endocannabinoids (eCBs)
- Endogenous Opioids
- Nerve Growth Factor (NGF)
- Brain-derived Neurotrophic Factor (BDNF)
- Acetylcholine (ACh)
2.2.2. Proprioceptors
2.3. Spinal Presynapic Inhibition (PSI)
- Inputs/Outputs
- GABA or not GABA?
- The Case for GABA
- GABAA Receptors as Presynaptic Modulators
- Presynaptic Inhibition (PSI) of Nociceptive Afferents
- The Case for Alternatives
- Comments
2.4. Spinal Cord
- Wide-dynamic-range (WDR) Neurons
- Anti-nociception by Corticosteroids
- Dopamine (DA) Modulation
- Noradrenergic (NA) Modulation
- Serotonergic (5-HT) Modulation
- Substance P (SP)
- Calcitonin Gene-related Peptide (CGRP)
- Cholecystokinin (CCK)
- Somatostatin (STT)
- Galanin (GAL)
- Endocannabinoids (eCBs)
- Endogenous Opioids
- Nerve Growth Factor (NGF)
- Nerve Growth Factor (NGF) in Inflammation
- NGF/TrkA-induced Sprouting and Neuroma Formation
- Interleukin-6 (IL-6)
- Brain-derived Neurotrophic Factor (BDNF)
- Cholinergic (ACh) Modulation
- Spinal DH Neurons Expressing Neurokinin-1 (NK1) and GABAB Receptors
- NK1 and STT2A Receptors in DH Lamina I
- Glutamate
- Kainate
- AMPA and NMDA at Spinal Level
2.5. The PAG-Triad Connection
2.5.1. Peri-Aqueductal Gray (PAG)
- Orexin (ORX)
- Dopamine (DA)
- Substance P (SP)
- Calcitonin Gene-related Peptide (CGRP)
- Endocannabinoids (eCBs)
- Opioids
- Nitric Oxide (NO)
- Brain-derived Neurotrophic Fator (BDNF)
- Glutamate
- GABA
2.5.2. Rostral Ventro-Medial MEDULLA (RVM)
- Dopamine (DA)
- Noradrenaline (NA)
- Serotonergic (5-HT) RVM Cells
- Dopamine (DA), Noradrenaline (NA) and Serotonin (5-HT) Interactions
- Opioids
- Substance P (SP)
- Cholecystokinin (CCK)
- Neurotensin (NT)
- Endocannabinoids (eCBs)
- Endogenous Opioids
- Cholinergic (ACh) Signaling in Brainstem Nuclei
- Glutamate
- GABA
- Co-expression of GABA and Opioid Receptors
2.5.3. Caudal Ventro-Lateral Medulla (CVLM)
- Noradrenaline (NA) and Angiotensin II
- Noradrenergic (NA) A5 Cell Group
- Serotonin (5-HT)
- Endogenous Opioids
2.5.4. Dorsal Reticular Nucleus (DReN)
- Noradrenaline (NA)
- Endogenous Opioids
- Glutamate
- GABA
- Case Report: Acute Thoracic Pain Syndrome with Central Dysregulation of Descending Pain Modulation
2.6. Nucleus Tractus Solitarii (NTS)
- Galanin (GAL)
- POMC Neuron Effects
- NMDA
2.7. Parabrachial Nucleus (PBN)
- Adrenoceptors
- Noradrenaline (NA) Effects from cNTS
- Endogenous Opioids
- Synaptic Transmission
- GABAB Receptors
2.8. Hypothalamic and Midbrain Dopamine (DA) Neurons
2.8.1. Hypothalamic Dopamine (DA) Cell Cluster
2.8.2. Midbrain Dopamine (DA) System
- Meso-limbic DA System
- Pain Modulation
- CNS Dopamine (DA) Receptors
- Dopamine (DA) in Nociceptors and Spinal Cord
- Dopamine (DA) Effects on Spinal Nociceptive Reflexes
- Dopamine (DA) in Nucleus Accumbens (NAc)
- Dopamine (DA) and Opioids in Prefrontal Cortex (PFC)
- Dopamine (DA) in Pain: Anti-nociception or Motivational Salience?
- Case Report: Dopamine-mediated Yawning-Fatigue syndrome
2.9. Brainstem Noradrenergic (NA) Cell Groups
- Noradrenaline (NA) Sources
- Spinal Actions
2.10. Hypothalamus (HYP)
- Descending Pain Modulation
2.10.1. Hypothalamo-Pituitary-Adrenal (HPA) Axis
Paraventricular Nucleus (PVN)
- Hypothalamo-pituitary-adrenal (HPA) Axis
- Corticotropin-releasing Hormone (CRH)
- Extra-hypothalamic Corticotropin-releasing Hormone (CRH) Neurons
- Corticotropin-releasing Hormone (CRH) in CeA
- Corticosteroids
Oxytocin (OXT) Neurons
- Pain Modulation
- Oxytocin (OXT) Effects in Sensory Afferents
- Oxytocin (OXT) Actions in the Dorsal Horn (DH)
- Oxytocin (OXT) Actions in other Central Structures
- Oxytocin (OXT) in PFC
- Interaction between Oxytocin (OXT) and Dopamine (DA)
- Interaction between Oxytocin (OXT) and Serotonin (5-HT)
Vasopressin (AVP) Neurons
- Pain Modulation
- Vasopressin (AVP) Effects on Behavior
- Peripheral Oxytocin (OXT) and Vasopressin (AVP) Effects
- Vasopressin (AVP) Effects in Dorsal Horn (DH)
- Vasopressin (AVP) Effects on Supraspinal Structures
Orexin (ORX) Neurons
Hypothalamic Neurons Producing Other Neuromodulators
- Cholecystokinin (CCK)
- Opioids
2.10.2. Hypothalamo-Pituitary-Gonadal (HPG) Axis
- Relation between Hypothalamo-pituitary-adrenal (HPA) Axis and Hypothalamo-pituitary-gonadal (HPG) Axis
- Female Gonadal Hormones
- Receptors
- Androgens
- Testosterone
- Dehydroepiandrosterone (DHEA)
- Androgen Effects on Meso-cortico-limbic System
2.10.3. Hypothalamo-Pituitary-Thyroid (HPT) Axis
- Thyrotropin-releasing Hormone (TRH) in Dorsal Horn (DH)
- Case Report: Acute Monoarthritis-Induced Pain Syndrome with Hypothalamic Activation
2.11. Basal Ganglia (BG)
2.11.1. Striatum
- Dopamine (DA)
- Serotonin (5-HT)
- Substance P (SP)
- Acetylcholine (ACh)
- Glutamate
- Adenosine
2.11.2. Nucleus Accumbens (NAc)
- Calcitonin Gene-related Peptide (CGRP)
- Substance P (SP)
- Neuropeptide Y (NPY)
- Opioids
- Interplay between Opioids and Dopamine (DA)
- Acetylcholine (ACh)
- Glutamate
- N-acetyl-aspartyl-glutamate
- GABA
2.11.3. Subthalamic Nucleus (STN)
- Noradrenaline (NA)
2.12. Amygdala (AMY)
- Corticotropin-releasing Hormone (CRH)
- Oxytocin (OXT) and Vasopressin (AVP)
- Calcitonin Gene-related Peptide (CGRP)
- Dopamine (DA)
- Noradrenaline (NA)
- Serotonin (5-HT)
- Somatostatin (STT)
- Neuropeptide S (NPS)
- Endogenous Opioids
- Acetylcholine (ACh)
- Glutamate
- GABA
- Pain and itch
2.13. Cerebral Cortex
2.13.1. Somatosensory Cortices
- Acetylcholine (ACh)
- Opioids in S1
- Glutamate
- GABA
2.13.2. Motor Cortex
2.13.3. Prefrontal Cortex (PFC)
- Oxytocin (OXT) in Paraventricular (PVN)-PFC Circuit
- Dopamine (DA)
- Somatostatin (STT)
- Opioids
- Ionotropic Glutamate Receptors
Medial Prefrontal Cortex (mPFC)
- Dopamine (DA)
- Opioids
- GABA
Anterior Cingulate Cortex (ACC)
- Opioids
- Glutamate
Dorso-lateral Prefrontal Cortex (dlPFC)
Ventro-lateral Orbito-frontal Cortex (vlOFC)
- Dopamine (DA) Effects
- Adrenaline (Adr)
- Opioids
- Glutamate and GABA
- Sm-vlOFC-PAG Pathway
Hippocampus (HIPP)
- Dentate Gyrus (DG)
- Case Report: Acute Trigeminal Neuralgia Triggered by Minor Facial Trauma
3. From the Angle of Substances
3.1. Substance P (SP)
- Substance P (SP) in Multiple Brain Regions
- Modulation of Ion Channels
3.2. Adenosine
3.3. Histamine (HIST)
3.4. Melanocortin
- Melanocortin in Sensory Afferents
- Melanocortin in Spinal Cord
- Melanocortin in Descending Pain-modulatory System
- Melanocortin Sex Specificity
3.5. Somatostatin (STT)
- Anti-nociceptive Somatostatin (STT) Effects
- Somatostatin (STT) in Spinal Cord
- Somatostatin (STT) in Peri-aqueductal Gray (PAG) and Amygdala (AMY)
- Somatostatin (STT) in Amygdala (AMY) and Hippocampus (HIPP)
- Somatostatin (STT) in Central Nucleus of Amygdala (CeA)
- Somatostatin (STT) in Prefrontal Cortex (PFC)
- Somatostatin (STT) in Primary Somatosensory Cortex (S1)
3.6. Neuropeptide Y (NPY)
- Neuropeptide Y (NPY) in Dorsal Horn (DH)
- Neuropeptide Y (NPY) in Peri-aqueductal Gray (PAG)
- Neuropeptide Y (NPY) in Rostral Ventro-medial Medulla (RVM)
- Neuropeptide Y (NPY) in Parabrachial Nucleus (PBN)
- Neuropeptide Y (NPY) in Hypothalamus (HYP)
- Neuropeptide Y (NPY) in Amygdala (AMY)
- Neuropeptide Y (NPY) in Nucleus Accumbens (NAc)
- Neuropeptide Y (NPY) in Bed Nucleus of the Stria Terminalis (BNST)
3.7. Neuropeptide S (NPS)
- Neuropeptide S (NPS) in Amygdala (AMY)
3.8. Cholecystokinin (CCK)
- Cholecystokinin (CCK) and Opioids
3.9. Calcitonin Gene-Related Peptide (CGRP)
- Pro-inflammatory Effects
- Calcitonin Gene-related Peptide (CGRP) in Central Nucleus of Amygdala (CeA)
- Case Report: Acute Migraine with CGRP-Linked Neurogenic Inflammation
3.10. Galanin (GAL)
- Galanin (GAL) in Dorsal Horn (DH)
- Galanin (GAL) in Amygdala (AMY)
- Galanin (GAL) in Anterior Cingulate Cortex (ACC)
3.11. Neurotensin (NT)
- Neurotensin (NT) in Spinal Cord
3.12. Endocannabinoids (eCBs)
- Pain Modulation
- Endocannabinoids (eCBs) in the PAG-RVM Axis
- Effects on Ion Channels
- Case Report: Acute Cannabinoid-Responsive Pain Syndrome Following Distal Radius Fracture
3.13. Endogenous Opioids
- Analgesia?
- Nucleus Tractus Solitarii (NTS) POMC Neuron Effects
- Effects on Ion Channels
3.14. Neurotrophins
3.14.1. Nerve Growth Factor (NGF)
3.14.2. Brain-Derived Neurotrophic Factor (BDNF)
- Pain Modulation
- Brain-derived Neurotrophic Factor (BDNF) in Amygdala (AMY)
- Brain-derived Neurotrophic Factor (BDNF) in Hippocampus (HIPP)
- Brain-derived Neurotrophic Factor (BDNF) in Prefrontal Cortex (PFC)
3.15. Neurotransmitters
3.15.1. Acetylcholine (ACh)
- Populations of Cholinergic (ACh) Projection Neurons
- Pain Perception and Modulation
- Acetylcholine (ACh) and GABA
- Acetylcholine (ACh) in Spinal Cord
3.15.2. Glutamate and Its Receptors
- Glutamate in Spinal Cord
Ionotropic Glutamate Receptors
- N-methyl-D-aspartate Receptors (NMDARs)
Metabotropic Glutamate Receptors (mGluRs)
- Metabotropic Glutamate Receptors (mGluRs) in Thalamus (THAL)
- Metabotropic Glutamate Receptors (mGluRs) in Striatum
- Sex Differences
3.15.3. Glycine and Its Receptors
3.15.4. GABA and Its Receptors
GABAA Receptors (GABAARs)
- GABA in Sensory Afferents
- GABA in Spinal Cord
- GABAARs in Central Nucleus of Amygdala (CeA)
GABAB Receptors (GABABRs)
- GABAB Receptors (GABABRs) in Sensory Afferents and Spinal Cord
- GABAB Receptors (GABABRs) in Thalamus (THAL)
4. Clinical Syndromes
- Postoperative Pain
- Acute Neuropathic Pain
- Acute Musculoskeletal Pain
- Acute Visceral Pain
- Headache Syndromes
- Case-Based Integration
- Summary
5. Concluding Remarks
Funding
Ethics Approval and Consent to Participate
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Adke, A.P.; Khan, A.; Ahn, H.-S.; Becker, J.J.; Wilson, T.D.; Valdivia, S.; Sugimura, Y.K.; Martinez Gonzalez, S.; Carrasquillo, Y. Cell-Type Specificity of Neuronal Excitability and Morphology in the Central Amygdala. eNeuro 2021, 8, ENEURO.0402-20.2020. [Google Scholar] [CrossRef] [PubMed]
- Allen, H.N.; Bobnar, H.J.; Kolber, B.J. Left and right hemispheric lateralization of the amygdala in pain. Prog Neurobiol 2021, 196, 1018913. [Google Scholar] [CrossRef]
- Althammer, F.; Eliava, M.; Grinevich, V. Central and peripheral release of oxytocin: Relevance of neuroendocrine and neurotransmitter actions for physiology and behavior. Handb Clin Neurol 2021, 180, 25–44. [Google Scholar] [PubMed]
- Amaral, D.G.; Campbell, M.J. Transmitter systems in the primate dentate gyrus. Hum Neurobiol 1986, 5, 169–180. [Google Scholar]
- Ambriz-Tututi, M.; Palomero-Rivero, M.; Ramirez-López, F.; Millán-Aldaco, D.; Drucker-Colin, A.R. Role of glutamate receptors in the dorsal reticular nucleus in formalin-induced secondary allodynia. Eur J Neurosci 2013, 38, 3008–3017. [Google Scholar] [CrossRef]
- Anyetei-Anum, C.S.; Roggero, V.R.; Allison, L.A. Thyroid hormone receptor localization in target tissues. J Endocrinol 2018, 237, R19–R34. [Google Scholar] [CrossRef] [PubMed]
- Asan, E.; Steinke, M.; Lesch, K.-P. Serotonergic innervation of the amygdala: Targets, receptors, and implications for stress and anxiety. Histochem Cell Biol 2013, 139, 785–813. [Google Scholar] [CrossRef]
- Baba, K.; Kawasaki, M.; Nishimura, H.; Suzuki, H.; Matsuura, T.; Ikeda, N.; Fujitani, T.; Yamanaka, Y.; Tsukamoto, M.; Ohnishi, N.; et al. Upregulation of the hypothalamo-neurohypophysial system and activation of vasopressin neurones attenuates hyperalgesia in a neuropathic pain model rat. Sci Rep 2022, 12, 13046. [Google Scholar] [CrossRef]
- Bagley, E.E.; Ingram, S.L. Endogenous opioid peptides in the descending pain modulatory circuit. Neuropharmacol 2020, 73, 108131. [Google Scholar] [CrossRef]
- Bannister, K.; Dickenson, A.H. What do monoamines do in pain modulation? Curr Opin Support Palliat Care 2016, 10, 143–148. [Google Scholar] [CrossRef]
- Barceló, A.C.; Filippini, B.; Pazo, J.H. The striatum and pain modulation. Cell Mol Neurobiol 2012, 32, 1–12. [Google Scholar] [CrossRef]
- Bardoni, R. Role of presynaptic glutamate receptors in pain transmission at the spinal cord level. Curr Neuropharmacol 2013, 11, 477–483. [Google Scholar] [CrossRef]
- Bardoni, R. Serotonergic modulation of nociceptive circuits in spinal cord dorsal horn. Curr Neuropharmacol 2019, 17, 1133–1145. [Google Scholar] [CrossRef] [PubMed]
- Barker, P.A.; Mantyh, P.; Arendt-Nielsen, L.; Viktrup, L.; Tive, L. Nerve growth factor signaling and its contribution to pain. J Pain Res 2020, 13, 1223–1241. [Google Scholar] [CrossRef]
- Belforte, J.E.; Pazo, J.H. Striatal inhibition of nociceptive responses evoked in trigeminal sensory neurons by tooth pulp stimulation. J Neurophysiol 2005, 93, 1730–1741. [Google Scholar] [CrossRef]
- Bellasio, S.; Nicolussi, E.; Bertorelli, R.; Reggiani, A. Melanocortin receptor agonists and antagonists modulate nociceptive sensitivity in the mouse formalin test. Eur J Pharmacol 2003, 482, 127–132. [Google Scholar] [CrossRef]
- Benarroch, E.E. Pain-autonomic interactions. Neurol. Sci. 2006, 27 (Suppl. 2), S130–S133. [Google Scholar] [CrossRef]
- Björklund, M.; Radovanovic, S.; Ljubisavljevic, M.; Windhorst, U.; Johansson, H. Muscle stretch-induced modulation of noxiously activated dorsal horn neurons of feline spinal cord. Neurosci Res 2004, 48, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Blumenkopf, B. Neuroschemistry of the dorsal horn. Appl Neurophysiol 1988, 51, 89–103. [Google Scholar] [PubMed]
- Boccella, S.; Marabese, I.; Guida, F.; Luongo, L.; Maione, S.; Palazzo, E. The modulation of pain by metabotropic glutamate receptors 7 and 8 in the dorsal striatum. Curr Neuropharmacol 2020, 18, 34–50. [Google Scholar] [CrossRef]
- Boll, S.; Almeida de Minas, A.C.; Raftogianni, A.; Herpertz, S.C.; Grinevich, V. Oxytocin and pain perception: From animal models to human research. Neuroscience 2018, 387, 149–161. [Google Scholar] [CrossRef]
- Bonsi, P.; Cuomo, D.; Martella, G.; Madeo, G.; Schirinzi, T.; Puglisi, F.; Ponterio, G.; Pisani, A. Centrality of striatal cholinergic transmission in basal ganglia function. Front Neuroanat 2011, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, C.A.; Ingram, S.L. Cannabinoids in the descending pain modulatory circuit: Role in inflammation. Pharmacol Ther 2020, 209, 107495. [Google Scholar] [CrossRef] [PubMed]
- Bourgoin, S.; Benoliel, J.J.; Collin, E.; Mauborgne, A.; Pohl, M.; Hamon, M.; Cesselin, F. Opioidergic control of the spinal release of neuropeptides. Possible significance for the analgesic effects of opioids. Fundam Clin Pharmacol 1994, 8, 307–321. [Google Scholar] [PubMed]
- Bourinet, E.; Altier, C.; Hildebrand, M.E.; Trang, T.; Salter, M.W.; Zamponi, G.W. Calcium-permeable ion channels in pain signals. Physiol Rev 2014, 94, 81–140. [Google Scholar] [CrossRef]
- Bowers, M.E.; Choi, D.C.; Ressler, K.J. Neuropeptide regulation of fear and anxiety: Implications of cholecystokinin, endogenous opioids, and neuropeptide Y. Physiol Behav 2012, 107, 699–710. [Google Scholar] [CrossRef]
- Boyce, V.S.; Mendell, L.M. Neurotrophins and spinal circuit function. Front Neural Circuits 2014, 8, 59. [Google Scholar] [CrossRef]
- Brockway, D.F.; Crowley, N.A. Turning the tides on neuropsychiatric diseases: The role of peptides in the prefrontal cortex. Front Behav Neurosci 2020, 14, 588400. [Google Scholar] [CrossRef]
- Brodal, A. Neurological Anatomy. In Relation to Clinical Medicine, 3rd ed.; Oxford University Press: New York, NY, USA, 1981. [Google Scholar]
- Budai, D.; Khasabov, S.G.; Mantyh, P.W.; Simone, D.A. NK-1 receptors modulate the excitability of ON cells in the rostral ventromedial medulla. J Neurophysiol 2007, 97, 1388–1395. [Google Scholar] [CrossRef]
- Buesa, I.; Aira, Z.; Azkue, J.J. Regulation of mociceptive plasticity threshold and DARPP-32 phosphorylation in spinal dorsal horn neurons by convergent dopamine and glutamate inputs. PLoS ONE 2016, 11, e0162416. [Google Scholar] [CrossRef]
- Buhler, A.V.; Proudfit, H.K.; Gebhart, G.F. Neurotensin-produced antinociception in the rostral ventromedial medulla is partially mediated by spinal cord norepinephrine. Pain 2008, 135, 280–290. [Google Scholar] [CrossRef]
- Burston, J.J.; Woodhams, S.G. Endocannabinoid system and pain: An introduction. Proc Nutr Soc 2014, 73, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Capogna, M. GABAergic cell type diversity in the basolateral amygdala. Curr Opin Neurobiol 2014, 26, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Cappoli, N.; Tabolacci, E.; Aceto, P.; Dello Russo, C. The emerging role of the BDNF-TrkB signaling pathway in the modulation of pain perception. J Neuroimmunol 2020, 349, 577406. [Google Scholar] [CrossRef] [PubMed]
- Carlton, S.M. Nociceptive primary afferents: They have a mind of their own. J Physiol 2014, 592, 3403–3411. [Google Scholar] [CrossRef] [PubMed]
- Carlton, S.M.; Du, J.; Zhou, S.; Coggeshall, R.E. Tonic control of peripheral cutaneous nociceptors by somatostatin receptors. J Neurosci 2001, 21, 4042–4049. [Google Scholar] [CrossRef]
- Carter, C.S.; Kenkel, W.M.; MacLean, E.L.; Wilson, S.R.; Perkeybile, A.M.; Yee, J.R.; Ferris, C.F.; Nazarloo, H.P.; Porges, S.W.; Davis, J.M.; et al. Is oxytocin “Nature’s Medicine”? Pharmacol Rev 2020, 72, 829–861. [Google Scholar] [CrossRef]
- Castro, A.R.; Pinto, M.; Lima, D.; Tavares, I. Nociceptive spinal neurons expressing NK1 and GABAB receptors are located in lamina I. Brain Res 2004, 1003, 77–85. [Google Scholar] [CrossRef]
- Cesselin, F. Opioid and anti-opioid peptides. Fundam Clin Pharmacol 1995, 9, 409–433. [Google Scholar] [CrossRef]
- Chang, C.-T.; Jiang, B.-Y.; Chen, C.-C. Ion channels involved in substance P-mediated nociception and antinociception. Int J Mol Sci 2019, 20, 1596. [Google Scholar] [CrossRef]
- Chen, J.; Cheuk, I.W.Y.; Shin, V.Y.; Kwong, A. Acetylcholine receptors: Key players in cancer development. Surg Oncol 2019, 31, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, W.; Tan, T.; Han, H.; Dong, Z. GABA(A) Receptors in the central nucleus of the amygdala are involved in pain- and itch-related responses. J Pain 2016, 17, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Shao, C.; Zhou, H.; Ma, R.; Jiang, P.; Yang, K. Differential sensitivity of presynaptic and postsynaptic GABAB receptors in rat ventrolateral periaqueductal gray. Neuroreport 2017, 28, 1221–1224. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-H.; Lien, C.-C.; Chen, C.-C. Neuronal basis for pain-like and anxiety-like behaviors in the central nucleus of the amygdala. Pain 2022, 163, e463–e475. [Google Scholar] [CrossRef]
- Chieng, B.; Christie, M.J. Somatostatin and nociceptin inhibit neurons in the central nucleus of amygdala that project to the periaqueductal grey. Neuropharmacology 2010, 59, 425–430. [Google Scholar] [CrossRef]
- Chudler, E.H.; Dong, W.K. The role of the basal ganglia in nociception and pain. Pain 1995, 60, 3–38. [Google Scholar] [CrossRef]
- Colucci-D’Amato, L.; Speranza, L.; Volpicelli, F. Neurotrophic factor BDNF, Physiological functions and therapeutic potential in depression, neurodegeneration and brain cancer. Int J Mol Sci 2020, 21, 7777. [Google Scholar] [CrossRef]
- Comitato, A.; Bardoni, R. Presynaptic inhibition of pain and touch in the spinal cord: From receptors to circuits. Int J Mol Sci 2021, 22, 414. [Google Scholar] [CrossRef]
- Corder, G.; Castro, D.C.; Bruchas, M.R.; Scherrer, G. Endogenous and exogenous opioids in pain. Annu Rev Neurosci 2018, 41, 453–473. [Google Scholar] [CrossRef]
- Cortes-Altamirano, J.L.; Olmos-Hernández, A.; Jaime, H.B.; Carrillo-Mora, P.; Bandala, C.; Reyes-Long, S.; Alfaro-Rodriguez, A. Review: 5-HT1, 5-HT2, 5-HT3 and 5-HT7 receptors and their role in the modulation of pain response in the central nervous system. Curr Neuropharmacol 2018, 16, 210–221. [Google Scholar] [CrossRef]
- Costigan, M.; Scholz, J.; Woolf, C.J. Neuropathic pain: A maladaptive response of the nervous system to damage. Annu Rev Neurosci 2009, 32, 1–32. [Google Scholar] [CrossRef]
- Côté, M.-P.; Murray, L.M.; Knikou, M. Spinal control of locomotion: Individual neurons, their circuits and functions. Front Physiol 2018, 9, 784. [Google Scholar] [CrossRef]
- Cui, L.; Liang, L.; Abdus-Saboor, I.; Olson, W.; Fleming, M.S.; Ma, M.; Tao, Y.-X.; Luo, W. Identification of early RET+ deep dorsal spinal cord interneurons in gating pain. Neuron 2016, 91, 1137–1153. [Google Scholar] [CrossRef]
- Cullinan, W.E. Stress response. In Encyclopedia of Neuroscience; Binder, M.D., Hirokawa, N., Windhorst, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 3861–3865. [Google Scholar]
- Curatolo, M. Is central sensitization relevant in acute low back pain? Eur J Pain 2023, 27, 457–458. [Google Scholar] [CrossRef]
- Dai, D.; Li, W.; Chen, A.; Gao, X.-F.; Xiong, L. Lateral habenula and its potential roles in pain and related behaviors. ACS Chem Neurosci 2022, 13, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Davey, R.A.; Grossmann, M. Androgen receptor structure, function and biology: From bench to bedside. Clin Biochem Rev 2016, 37, 3–15. [Google Scholar] [PubMed]
- De Felice, M.; Ossipov, M.H. Cortical and subcortical modulation of pain. Pain Manag 2016, 6, 111–120. [Google Scholar] [CrossRef]
- Delaney, A.J.; Crane, J.W. Presynaptic GABAB receptors reduce transmission at parabrachial synapses in the lateral central amygdala by inhibiting N-type calcium channels. Sci Rep 2016, 6, 19255. [Google Scholar] [CrossRef]
- Delaney, A.J.; Crane, J.W.; Sah, P. Noradrenaline modulates transmission at a central synapse by a presynaptic mechanism. Neuron 2007, 56, 880–982. [Google Scholar] [CrossRef] [PubMed]
- DeLong, M.R.; Wichmann, T. Circuits and circuit disorders of the basal ganglia. Arch Neurol 2007, 64, 20–24. [Google Scholar] [CrossRef]
- Dembo, T.; Braz, J.M.; Hamel, K.A.; Kuhn, J.A.; Basbaum, A.I. Primary afferent-derived BDNF contributes minimally to the processing of pain and itch. eNeuro 2018, 5, ENEURO.0402-18.2018. [Google Scholar] [CrossRef]
- de Novellis, V.; Mariani, L.; Palazzo, E.; Vita, D.; Marabese, I.; Scafuro, M.; Rossi, F.; Maione, S. Periaqueductal grey CB1 cannabinoid and metabotropic glutamate subtype 5 receptors modulate changes in rostral ventromedial medulla neuronal activities induced by subcutaneous formalin in the rat. Neuroscience 2005, 134, 269–281. [Google Scholar] [CrossRef]
- Devesa, I.; Ferrer-Montiel, A. Neurotrophins, endocannabinoids and thermo-transient receptor potential: A threesome in pain signalling. Eur J Neurosci 2014, 39, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Diaz Brinton, R.; Thompson, R.F.; Foy, M.R.; Baudry, M.; Wang, J.; Finch, C.E.; Morgan, T.E.; Pike, C.J.; Mack, W.J.; Stanczyk, Z.; et al. Progesterone receptors: Form and function in brain. Front Neuroendocrinol 2008, 29, 313–339. [Google Scholar] [CrossRef] [PubMed]
- Dibaj, P.; Safavi-Abbasi, E.; Asadollahi, E. In vivo spectrally unmixed multi-photon imaging of longitudinal axon-glia changes in injured spinal white matter. Neurosci Lett 2024, 841, 137959. [Google Scholar] [CrossRef] [PubMed]
- Dibaj, P.; Windhorst, U. Motor-control notions in health and disease (what controls motor control). Preprints 2024, 2024031799. [Google Scholar] [CrossRef]
- Dibaj, P.; Windhorst, U. Muscle fatigue in health and disease. Preprints 2024, 2024091515. [Google Scholar] [CrossRef]
- Dibaj, P.; Brockmann, K.; Gärtner, J. Dopamine-mediated yawning-fatigue syndrome with specific recurrent initiation and responsiveness to opioids. JAMA Neurol 2020, 77, 254. [Google Scholar] [CrossRef] [PubMed]
- Dibaj, P.; Seeger, D.; Gärtner, J.; Petzke, F. Follow-up of a case of dopamine-mediated yawning-fatigue-syndrome responsive to opioids, successful desensitization via graded activity treatment. Neurol Int 2021, 13, 79–84. [Google Scholar] [CrossRef]
- Ding, S.; Zhou, F.-M. Serotonin regulation of subthalamic neurons. Rev Neurosci 2014, 25, 605–619. [Google Scholar] [CrossRef]
- Doan, L.; Manders, T.; Wang, J. Neuroplasticity underlying the comorbidity of pain and depression. Neural Plast 2015, 2015, 504691. [Google Scholar] [CrossRef]
- Dobner, P.R. Neurotensin and pain modulation. Peptides 2006, 27, 2405–2414. [Google Scholar] [CrossRef]
- Donadon, M.F.; Martin-Santos, R.; de Lima Osório, F. The associations between oxytocin and trauma in humans: A systematic review. Front Pharmacol 2018, 9, 154. [Google Scholar] [CrossRef]
- Du, X.; Hao, H.; Yang, Y.; Huang, S.; Wang, C.; Gigout, S.; Ramli, R.; Li, X.; Jaworska, E.; Edwards, I.; et al. Local GABAergic signaling within sensory ganglia controls peripheral nociceptive transmission. J Clin Invest 2017, 127, 1741–1756. [Google Scholar] [CrossRef]
- Du, Y.; Zhao, Y.; Zhang, A.; Li, Z.; Wei, C.; Zheng, Q.; Qiao, Y.; Liu, Y.; Ren, W.; Han, J.; et al. The role of the mu opioid receptors of the medial prefrontal cortex in the modulation of analgesia induced by acute restraint stress in male mice. Int J Mol Sci 2024, 25, 9774. [Google Scholar] [CrossRef] [PubMed]
- Duan, B.; Cheng, L.; Bourane, S.; Britz, O.; Padilla, C.; Garcia-Campmany, L.; Krashes, M.; Knowlton, W.; Velasquez, T.; Ren, X.; et al. Identification of spinal circuits transmitting and gating mechanical pain. Cell 2014, 159, 1417–1432. [Google Scholar] [CrossRef]
- Dubin, A.E.; Patapoutian, A. Nociceptors: The sensors of the pain pathway. J Clin Invest 2010, 120, 3760–3772. [Google Scholar] [CrossRef] [PubMed]
- Dussán-Sarria, J.A.; Jardim da Silva, N.R.; Deitos, A.; Stefani, L.C.; Laste, G.; de Souza, A.; Torres, I.L.S.; Fregni, F.; Caumo, W. Higher cortical facilitation and serum BDNF are associated with increased sensitivity to heat pain and reduced endogenous pain inhibition in healthy males. Pain Med 2018, 19, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Edin, B.B.; Abbs, J.H. Finger movement responses of cutaneous mechanoreceptors in the dorsal skin of the human hand. J Neurophysiol 1991, 65, 657–670. [Google Scholar] [CrossRef]
- Edvinsson, L. Calcitonin gene-related peptide (CGRP) and the pathophysiology of headache: Therapeutic implications. CNS Drugs 2001, 15, 745–753. [Google Scholar] [CrossRef]
- Ekman, E.F.; Koman, L.A. Acute pain following musculoskeletal injuries and orthopaedic surgery: Machanisms and management. Instr Course Lect 2005, 54, 21–33. [Google Scholar] [CrossRef]
- Elde, R.; Hökfelt, T. Localization of hypophysiotropic peptides and other biologically active peptides within the brain. Annu Rev Physiol 1979, 41, 587–602. [Google Scholar] [CrossRef]
- Esmaeili, M.-H.; Reise, Z.; Ezzatpanah, S.; Haghparast, A. Role of orexin-2 and CB1 receptors within the periaqueductal gray matter in lateral hypothalamic-induced antinociception in rats. Behav Pharmacol 2017, 28, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Fabian, C.B.; Seney, M.L.; Joffe, M.E. Sex differences and hormonal regulation of metabotropic glutamate receptor synaptic plasticity. Int Rev Neurobiol 2023, 168, 311–347. [Google Scholar]
- Feng, Y.-P.; Wang, J.; Dong, Y.-L.; Wang, Y.-Y.; Li, Y.-Q. The roles of neurotensin and its analogues in pain. Curr Pharm Des 2015, 21, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Ferdousi, M.; Finn, D.P. Stress-induced modulation of pain: Role of the endogenous opioid system. Prog Brain Res 2018, 239, 121–177. [Google Scholar] [PubMed]
- Fernández-Ruiz, J.; Lastres-Becker, I.; Cabranes, A.; González, S.; Ramos, J.A. Endocannabinoids and basal ganglia functionality. Prostaglandins Leukot Essent Fatty Acids 2002, 66, 257–267. [Google Scholar] [CrossRef]
- Ferrari, L.F.; Pei, J.; Zickella, M.; Rey, C.; Zickella, J.; Ramirez, A.; Taylor, N.E. D2 Receptors in the periaqueductal gray/dorsal raphe modulate peripheral inflammatory hyperalgesia via the rostral ventral medulla. Neuroscience 2021, 463, 159–173. [Google Scholar] [CrossRef]
- Ferré, S.; Sarasola, L.I.; Quiroz, C.; Ciruela, F. Presynaptic adenosine receptor heteromers as key modulators of glutamatergic and dopaminergic neurotransmission in the striatum. Neuropharmacology 2023, 223, 109329. [Google Scholar] [CrossRef]
- Finn, D.P.; Haroutounian, S.; Hohmann, A.G.; Krane, E.; Soliman, N.; Rice, A.S. (2021) Cannabinoids, the endocannabinoid system, and pain: A review of preclinical studies. Pain 2021, 162 (Suppl. 1), S5–S25. [Google Scholar] [CrossRef]
- Finnerup, N.B.; Kuner, R.; Jensen, T.S. Neuropathic pain: From mechanisms to treatment. Physiol Rev 2021, 101, 259–301. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.; Clemente, J.T.; Tambeli, C.H. The protective role of testosterone in the development of temporomandibular joint pain. J Pain 2007, 8, 437–442. [Google Scholar] [CrossRef]
- Follansbee, T.; Domocos, D.; Nguyen, E.; Nguyen, A.; Bountouvas, A.; Velasquez, L.; Carstens, M.I.; Takanami, K.; Ross, S.E.; Carstens, E. Inhibition of itch by neurokinin 1 receptor (Tacr1) -expressing ON cells in the rostral ventromedial medulla in mice. Elife 2022, 11, e69626. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Rodrigues, D.; Almeida, A.; Pinto-Ribeiro, F. A new gal in town: A systematic review of the role of galanin and its receptors in experimental pain. Cells 2022, 11, 839. [Google Scholar] [CrossRef]
- François, A.; Low, S.A.; Sypek, E.I.; Christensen, A.J.; Sotoudeh, C.; Beier, K.T.; Ramakrishnan, C.; Ritola, K.D.; Sharif-Naeini, R.; Deisseroth, K.; et al. Brainstem-spinal cord inhibitory circuit for mechanical pain modulation by GABA and enkephalins. Neuron 2017, 93, 822–839.e6. [Google Scholar] [CrossRef]
- Fürst, S. Transmitters involved in antinociception in the spinal cord. Brain Res Bull 1999, 48, 129–141. [Google Scholar] [CrossRef]
- Fong, H.; Zheng, J.; Kurrasch, D. The structural and functional complexity of the integrative hypothalamus. Science 2023, 382, 388–394. [Google Scholar] [CrossRef]
- Fundytus, M.E. Glutamate receptors and nociception: Implications for the drug treatment of pain. CNS Drugs 2001, 15, 29–58. [Google Scholar] [CrossRef]
- Gamal-Eltrabily, M.; Manzano-García, A. Role of central oxytocin and dopamine systems in nociception and their possible interactions: Suggested hypotheses. Rev Neurosci 2018, 29, 377–386. [Google Scholar] [CrossRef]
- Gamal-Eltrabily, M.; Márquez-Morales, C.; Martínez-Lorenzana, G.; González-Hernández, A.; Condés-Lara, M. Cortical modulation of nociception. Neuroscience 2021, 458, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Esteves, F.O.; McWilliam, P.N.; Batten, T.F. Substance P (NK1) and somatostatin (sst2A) receptor immunoreactivity in NTS-projecting rat dorsal horn neurones activated by nociceptive afferent input. J Chem Neuroanat 2004, 27, 251–266. [Google Scholar] [CrossRef]
- Garcia-Recio, S.; Gascón, P. Biological and pharmacological aspects of the NK1-receptor. Biomed Res Int 2015, 2015, 495704. [Google Scholar] [CrossRef]
- Gear, R.W.; Levine, J.D. Nucleus accumbens facilitates nociception. Exp Neurol 2011, 229, 502–506. [Google Scholar] [CrossRef]
- Gilron, I.; Dickenson, A.H. Emerging drugs fpr neuropathic pain. Expert Opin Emerg Drugs 2014, 19, 329–341. [Google Scholar] [CrossRef]
- Goadsby, P.J.; Holland, P.R.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of migraine: A disorder of sensory processing. Physiol Rev 2017, 97, 553–622. [Google Scholar] [CrossRef]
- Godínez-Chaparro, B.; Martínez-Lorenzana, G.; Rodríguez-Jiménez, R.; Manzano-García, A.; Rojas-Piloni, G.; Condés-Lara, M.; González-Hernández, A. The potential role of serotonergic mechanisms in the spinal oxytocin-induced antinociception. Neuropeptides 2016, 60, 51–60. [Google Scholar] [CrossRef]
- Gomtsian, L.; Bannister, K.; Eyde, N.; Robles, D.; Dickenson, A.H.; Porreca, K.; Navratilova, E. Morphine effects within the rodent anterior cingulate cortex and rostral ventromedial medulla reveal separable modulation of affective and sensory qualities of acute or chronic pain. Pain 2018, 159, 2512–2521. [Google Scholar] [CrossRef] [PubMed]
- Gonkowski, S.; Rytel, L. Somatostatin as an active substance in the mammalian enteric nervous system. Int J Mol Sci 2019, 20, 4461. [Google Scholar] [CrossRef] [PubMed]
- González-Hernández, A.; Manzano-García, A.; Martínez-Lorenzana, G.; Tello-García, I.A.; Carranza, M.; Arámburo, C.; Condés-Lara, M. Peripheral oxytocin receptors inhibit the nociceptive input signal to spinal dorsal horn wide-dynamic-range neurons. Pain 2017, 58, 2117–2128. [Google Scholar] [CrossRef]
- Goodin, B.R.; Ness, T.J.; Robbins, M.T. Oxytocin—A multifunctional analgesic for chronic deep tissue pain. Curr Pharm Des 2015, 21, 906–913. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Wessendorf, M. Endomorphin-2-immunoreactive fibers selectively appose serotonergic neuronal somata in the rostral ventral medial medulla. J Comp Neurol 2007, 502, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Greenwell, T.N.; Martin-Schild, S.; Inglis, F.M.; Zadina, J.E. Colocalization and shared distribution of endomorphins with substance P, calcitonin gene-related peptide, gamma-aminobutyric acid, and the mu opioid receptor. J Comp Neurol 2007, 503, 319–333. [Google Scholar] [CrossRef]
- Gregory, N.S.; Gautam, M.; Benson, C.J.; Sluka, K.A. Acid sensing ion channel 1a (ASIC1a) mediates sctivity-induced pain by modulation of heteromeric ASIC channel kinetics. Neurosci 2018, 386, 166–174. [Google Scholar] [CrossRef]
- Groenewegen, H.J. The basal ganglia and motor control. Neural Plast 2003, 10, 107–120. [Google Scholar] [CrossRef]
- Guindon, J.; Hohmann, A.G. The endocannabinoid system and pain. CNS Neurol Disord Drug Targets 2009, 8, 403–421. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Hu, J. Spinal presynaptic inhibition in pain control. Neuroscience 2014, 283, 95–106. [Google Scholar] [CrossRef]
- Gupta, K.; Harvima, I.T. Mast cell-neural interactions contribute to pain and itch. Immunol Rev 2018, 282, 168–187. [Google Scholar] [CrossRef]
- Haber, S.N. Corticostriatal circuitry. Dialogues Clin Neurosci 2016, 18, 7–21. [Google Scholar] [CrossRef]
- Haber, S.N.; Fudge, J.L. The primate substantia nigra and VTA: Integrative circuitry and function. Crit Rev Neurobiol 1997, 11, 323–342. [Google Scholar] [CrossRef]
- Hagelberg, N.; Jääkeläinen, S.K.; Martikainen, I.K.; Mansikka, H.; Forssell, H.; Scheinin, H.; Hietala, J.; Pertovaara, A. Striatal dopamine D2 receptors in modulation of pain in humans: A review. Eur J Pharmacol 2004, 500, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Haleem, D.J. Targeting serotonin1A receptors for treating chronic pain and depression. Curr Neuropharmacol 2019, 17, 1098–1108. [Google Scholar] [CrossRef]
- Hambrecht-Wiedbusch, V.S.; Mitchell, M.F.; Firn, K.A.; Baghdoyan, H.A.; Lydic, R. Benzodiazepine site agonists differentially alter acetylcholine release in rat amygdala. Anesth Analg 2014, 118, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Hamon, M.; Blier, P. Monoamine neurocircuitry in depression and strategies for new treatments. Prog Neuropsychopharmacol Biol Psychiatry 2013, 45, 54–63. [Google Scholar] [CrossRef]
- Han, D.-J.; He, Z.-G.; Yang, H. Melanocortin-4 receptor in subthalamic nucleus is involved in the modulation of nociception. Am J Clin Exp Immunol 2018, 7, 76–80. [Google Scholar]
- Harris, H.N.; Peng, Y.B. Evidence and explanation for the involvement of the nucleus accumbens in pain processing. Neural Regen Res 2020, 15, 597–605. [Google Scholar] [CrossRef]
- Hashemi, M.; Karami, M.; Zarrindast, M. The regulatory role of nitric oxide in morphine-induced analgesia in the descending path of pain from the dorsal hippocampus to the dorsolateral periaqueductal gray. Eur J Pain 2022, 26, 888–901. [Google Scholar] [CrossRef]
- Haws, C.M.; Heinricher, M.M.; Fields, H.L. Alpha-adrenergic receptor agonists, but not antagonists, alter the tail-flick latency when microinjected into the rostral ventromedial medulla of the lightly anesthetized rat. Brain Res 1990, 533, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Hay, D.L.; Garelja, M.L.; Poyner, D.R.; Walker, C.S. Update on the pharmacology of calcitonin/CGRP family of peptides: IUPHAR Review 25. Br J Pharmacol 2018, 175, 3–17. [Google Scholar] [CrossRef]
- Hebb, A.L.O.; Poulin, J.-F.; Roach, S.P.; Zacharko, R.M.; Drolet, G. Cholecystokinin and endogenous opioid peptides: Interactive influence on pain, cognition, and emotion. Prog Neuropsychopharmacol Biol Psychiatry 2005, 29, 1225–1238. [Google Scholar] [CrossRef] [PubMed]
- Heinricher, M.M.; Morgan, M.M.; Fields, H.L. Direct and indirect actions of morphine on medullary neurons that modulate nociception. Neuroscience 1992, 48, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Heinricher, M.M.; Tavares, I.; Leith, J.L.; Lumb, B.M. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev 2009, 60, 214–225. [Google Scholar] [CrossRef]
- Helmreich, D.L.; Parfitt, D.B.; Lu, X.-Y.; Akil, H.; Watson, S.J. Relation between the hypothalamic-pituitary-thyroid (HPT) axis and the hypothalamic-pituitary-adrenal (HPA) axis during repeated stress. Neuroendocrinology 2005, 81, 183–192. [Google Scholar] [CrossRef]
- Herrero, M.-T.; Barcia, C.; Navarro, J.M. Functional anatomy of thalamus and basal ganglia. Childs Nerv Syst 2002, 18, 386–404. [Google Scholar] [CrossRef]
- Heuermann, R.J.; Gereau, R.W. Inhibitory effects of dopamine agonists on pain-responsive neurons in the central nucleus of the amygdala. J Neurophysiol. 2025, 133, 1947–1954. [Google Scholar] [CrossRef]
- Hillard, C.J. Stress regulates endocannabinoid-CB1 receptor signaling. Semin Immunol 2014, 26, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J. Endocannabinoids and the endocrine system in health and disease. Handb Exp Pharmacol 2015, 231, 317–339. [Google Scholar] [PubMed]
- Hillard, C.J.; Beatka, M.; Sarvaideo, J. Endocannabinoid signaling and the hypothalamic-pituitary-adrenal axis. Compr Physiol 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Hochman, S.; Shreckengost, J.; Kimura, H.; Quevedo, J. Presynaptic inhibition of primary afferents by depolarization: Observations supporting nontraditional mechanisms. Ann NY Acad Sci 2010, 1198, 140–152. [Google Scholar] [CrossRef]
- Hökfelt, T.; Barde, S.; Xu, Z.-Q.D.; Kuteeva, E.; Rüegg, J.; Le Maitre, E.; Risling, M.; Kehr, J.; Ihnatko, R.; Theodorsson, E.; et al. Neuropeptide and small transmitter coexistence: Fundamental studies and relevance to mental illness. Front Neural Circuits 2018, 12, 106. [Google Scholar] [CrossRef]
- Holden, J.E.; Pizzi, J.A.; Jeong, Y. An NK1 receptor antagonist microinjected into the periaqueductal gray blocks lateral hypothalamic-induced antinociception in rats. Neurosci Lett 2009, 453, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Holsboer, F.; Ising, M. Stress hormone regulation: Biological role and translation into therapy. Annu Rev Psychol 2010, 61, 81–109. [Google Scholar] [CrossRef] [PubMed]
- Hossaini, M.; Duraku, L.S.; Saraç, Ç.; Jongen, J.L.M.; Holstege, J.C. Differential distribution of activated spinal neurons containing glycine and/or GABA and expressing c-fos in acute and chronic pain models. Pain 2010, 151, 356–365. [Google Scholar] [CrossRef]
- Hough, L.B.; Rice, F.L. H3 receptors and pain modulation: Peripheral, spinal, and brain interactions. J Pharmacol Exp Ther 2011, 336, 30–37. [Google Scholar] [CrossRef]
- Hu, R.; Li, Y.-J.; Li, X.-H. An overview of non-neural sources of calcitonin gene-related peptide. Curr Med Chem 2016, 23, 763–773. [Google Scholar] [CrossRef]
- Huang, J.; Polgar, E.; Solinsky, H.J.; Mishra, S.K.; Tseng, P.-Y.; Iwagaki, N.; Boyle, K.A.; Dickie, A.C.; Kriegbaum, M.C.; Wildner, H.; et al. Circuit dissection of the role of somatostatin in itch and pain. Nat Neurosci 2018, 21, 707–716. [Google Scholar] [CrossRef]
- Huang, J.; Xu, F.; Yang, L.; Tuolihong, L.; Wang, X.; Du, Z.; Zhang, Y.; Yin, X.; Li, Y.; Lu, K.; et al. Involvement of the GABAergic system in PTSD and its therapeutic significance. Front Mol Neurosci 2023, 16, 1052288. [Google Scholar] [CrossRef]
- Huang, S.; Borgland, S.L.; Zamponi, G.W. Dopaminergic modulation of pain signals in the medial prefrontal cortex: Challenges and perspectives. Neurosci Lett 2019, 702, 71–76. [Google Scholar] [CrossRef]
- Huang, W.-J.; Chen, W.-W.; Zhang, X. Endocannabinoid system: Role in depression, reward and pain control (Review). Mol Med Rep 2016, 14, 2899–2903. [Google Scholar] [CrossRef]
- Humes, C.; Sic, A.; Knezevic, N.N. Substance P’s impact on chronic pain and psychiatric conditions—A narrative review. Int J Mol Sci 2024, 25, 5905. [Google Scholar] [CrossRef] [PubMed]
- Humphries, M.D.; Prescott, T.J. The ventral basal ganglia, a selection mechanism at the crossroads of space, strategy, and reward. Prog Neurobiol 2010, 90, 385–417. [Google Scholar] [CrossRef] [PubMed]
- Jean, A. The nucleus tractus solitarius: Neuroanatomic, neurochemical and functional aspects. Arch Int Physiol Biochim Biophys 1991, 99, A3-52. (In French) [Google Scholar]
- Ji, Y.; Onwukwe, C.; Smith, J.; Laub, H.; Posa, L.; Keller, A.; Masri, R.; Cramer, N. Noradrenergic input from nucleus of the solitary tract regulates parabrachial activity in mice. eNeuro 2023, 10, ENEURO.0412-22.2023. [Google Scholar] [CrossRef]
- Jia, T.; Wang, Y.-D.; Chen, J.; Zhang, X.; Cao, J.-L.; Xiao, C.; Zhou, C. A nigro-subthalamo-parabrachial pathway modulates pain-like behaviors. Nat Commun 2022, 13, 7756. [Google Scholar] [CrossRef]
- Jones, S.L. Descending noradrenergic influences on pain. Prog Brain Res 1991, 88, 381–394. [Google Scholar]
- Joseph-Bravo, P.; Jaimes-Hoy, L.; Charli, J.-L. Regulation of TRH neurons and energy homeostasis-related signals under stress. J Endocrinol 2015, 224, R139–R159. [Google Scholar] [CrossRef]
- Joyce, M.K.P.; Uchendu, S.; Arnsten, A.F.T. Stress and inflammation target dorsolateral prefrontal cortex function: Neural mechanisms underlying weakened cognitive control. Biol Psychiatry 2025, 97, 359–371. [Google Scholar] [CrossRef]
- Juif, P.-E.; Poisbeau, P. Neurohormonal effects of oxytocin and vasopressin receptor agonists on spinal pain processing in male rats. Pain 2013, 154, 1449–1456. [Google Scholar] [CrossRef] [PubMed]
- Julia, V.; Buéno, L. Tachykininergic mediation of viscerosensitive responses to acute inflammation in rats: Role of CGRP. Am J Physiol 1997, 272 Pt 1, G141–G146. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.J.; Jo, S.-H.; Lee, S.; Oh, E.; Kim, M.-S.; Nam, W.D.; Oh, S.B. Effects of somatostatin on the responses of rostrally projecting spinal dorsal horn neurons to noxious stimuli in cats. Korean J Physiol Pharmacol 2008, 12, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Jurek, B.; Neumann, I.D. The oxytocin receptor: From intracellular signaling to behavior. Physiol Rev 2018, 98, 1805–1908. [Google Scholar] [CrossRef]
- Kalezic, I.; Bugaychenko, L.A.; Kostyukov, A.I.; Pilyavskii, A.I.; Ljubisavljevic, M.; Windhorst, U.; Johansson, H. Fatigue-related depression of the feline monosynaptic gastrocnemius-soleus reflex. J. Physiol. 2004, 556, 283–296. [Google Scholar] [CrossRef]
- Kaneko, T.; Oura, A.; Imai, Y.; Kusumoto-Yoshida, I.; Kanekura, T.; Okuno, H.; Kuwaki, T.; Kashiwadani, H. Orexin neurons play contrasting roles in itch and pain neural processing via projecting to the periaqueductal gray. Commun Biol 2024, 7, 290. [Google Scholar] [CrossRef]
- Kang, X.; Tang, H.; Liu, Y.; Yuan, Y.; Wang, M. Research progress on the mechanism of orexin in pain regulation in different brain regions. Open Life Sci 2021, 16, 46–52. [Google Scholar] [CrossRef]
- Kaur, G.; Singh, N.; Jaggi, A.S. Mast cells in neuropathic pain: An increasing spectrum of their involvement in pathophysiology. Rev Neurosci 2017, 28, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Kautz, M.; Charney, D.S.; Murrough, J.W. Neuropeptide Y, resilience, and PTSD therapeutics. Neurosci Lett 2017, 649, 164–169. [Google Scholar] [CrossRef]
- Kelly, E.A.; Fudge, J.L. The neuroanatomic complexity of the CRF and DA systems and their interface: What we still don’t know. Neurosci Biobehav Rev 2018, 90, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Mishina, M.; Kokubo, R.; Nakajima, T.; Moromoto, D.; Isu, T.; Kobayashi, S.; Teramoto, A. Ketamine for acute neuropathic pain in patients with spinal cord injury. J Clin Neurosci 2013, 20, 804–807. [Google Scholar] [CrossRef]
- Klein, M.O.; Battagello, D.S.; Cardoso, A.R.; Hauser, D.N.; Bittencourt, J.C.; Correa, R.G. Dopamine: Functions, signaling, and association with neurological diseases. Cell Mol Neurobiol 2018, 39, 31–59. [Google Scholar] [CrossRef]
- Knox, D. The role of basal forebrain cholinergic neurons in fear and extinction memory. Neurobiol Learn Mem 2016, 133, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, T.; Sasaki, M.; Sanai, K.; Kuwahata, H.; Sakurada, C.; Tsuzuki, M.; Iwata, Y.; Sakurada, S.; Sakurada, T. Intrathecal substance P augments morphine-induced antinociception: Possible relevance in the production of substance P N-terminal fragments. Peptides 2009, 30, 1689–1696. [Google Scholar] [CrossRef]
- Kondo, T.; Oshima, T.; Obata, K.; Sakurai, J.; Knowles, C.H.; Matsumoto, T.; Noguchi, K.; Miwa, H. Role of transient receptor potential A1 in gastric nociception. Digestion 2010, 82, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Kormos, V.; Gaszner, B. Role of neuropeptides in anxiety, stress, and depression: From animals to humans. Neuropeptides 2013, 47, 401–419. [Google Scholar] [CrossRef] [PubMed]
- Kumamoto, E. Cellular mechanisms for antinociception produced by oxytocin and orexins in the rat spinal lamina II—Comparison with those of other endogenous pain modulators. Pharmaceuticals 2019, 12, 136. [Google Scholar] [CrossRef]
- Kummer, K.K.; Mitrić, M.; Kalpachidou, T.; Kress, M. The medial prefrontal cortex as a central hub for mental comorbidities associated with chronic pain. Int J Mol Sci 2020, 21, 3440. [Google Scholar] [CrossRef]
- Kuner, R.; Kuner, T. Cellular circuits in the brain and their modulation in acute and chronic pain. Physiol Rev 2021, 101, 213–258. [Google Scholar] [CrossRef]
- Kvetnansky, R.; Sabban, E.L.; Palkovits, M. Catecholaminergic systems in stress: Structural and molecular genetic approaches. Physiol Rev 2009, 89, 535–606. [Google Scholar] [CrossRef]
- Laing, I.; Todd, A.J.; Heizmann, C.W.; Schmidt, H.H. Subpopulations of GABAergic neurons in laminae I-III of rat spinal dorsal horn defined by coexistence with classical transmitters, peptides, nitric oxide synthase or parvalbumin. Neuroscience 1994, 61, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Lamotte, G.; Shouman, K.; Benarroch, E.E. Stress and central autonomic network. Auton Neurosci 2021, 235, 102870. [Google Scholar] [CrossRef]
- Laste, G.; Ripoll Rozinsky, J.; Caumo, W.; da Silva Torres, I.L. Short- but not long-term melatonin administration reduces central levels of brain-derived neurotrophic factor in rats with inflammatory pain. Neuroimmunomodulation 2015, 22, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J Pain 2009, 10, 895–926. [Google Scholar] [CrossRef]
- LaVigne, J.E.; Alles, S.R.A. CCK2 receptors in chronic pain. Neurobiol Pain 2022, 11, 100092. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Chen, C.-C. Role of proprioceptors in chronic musculoskeletal pain. Exp Physiol 2023, 109, 45–54. [Google Scholar] [CrossRef]
- Lee, C.-H.; Lin, J.-H.; Lin, S.-H.; Chang, C.-T.; Wu, Y.-W.; Bewick, G.; Banks, R.W.; Gründer, S.; Hochgeschwender, U.; Chen, C.-C. A role for proprioceptors in sngception. Sci Adv 2025, 11, eabc5219. [Google Scholar] [CrossRef] [PubMed]
- Le Merrier, J.; Becker, J.A.J.; Befort, K.; Kiefer, B.L. Reward processing by the opioid system in the brain. Physiol Rev 2009, 89, 1379–1412. [Google Scholar] [CrossRef]
- Levine, J.D.; Fields, H.L.; Basbaum, A.I. Peptides and the primary afferent nociceptor. J Neurocsci 1993, 13, 2273–2286. [Google Scholar] [CrossRef]
- Lewin, G.R.; Lu, Y.; Park, T.J. A plethora of painful molecules. Curr Opin Neurobiol 2004, 14, 443–449. [Google Scholar] [CrossRef]
- Lewin, G.R.; Nykjaer, A. Pro-neutrophins, sortilin, and nociception. Eur J Neurosci 2014, 39, 363–374. [Google Scholar] [CrossRef]
- Li, C.; Sugam, J.A.; Lowery-Gionta, E.G.; McElligott, Z.-A.; McCall, N.M.; Lopez, A.J.; McKlveen, J.M.; Pleil, K.E.; Kash, T.L. Mu opioid receptor modulation of dopamine neurons in the periaqueductal gray/dorsal raphe: A role in regulation of pain. Neuropsychopharmacology 2016, 41, 2122–2132. [Google Scholar] [CrossRef]
- Li, G.; Shao, C.; Chen, Q.; Wang, Q.; Yang, K. Accumulated GABA activates presynaptic GABAB receptors and inhibits both excitatory and inhibitory synaptic transmission in rat midbrain periaqueductal gray. Neuroreport 2017, 28, 313–318. [Google Scholar] [CrossRef]
- Li, S.-Y.; Huo, M.-L.; Wu, X.-Y.; Huang, Y.-Q.; Wang, L.; Zhang, X.; Jiang, Y.-M.; Zhang, M.-L.; Wang, L.-L.; Yu, L.-C. Involvement of galanin and galanin receptor 1 in nociceptive modulation in the central nucleus of amygdala in normal and neuropathic rats. Sci Rep 2017, 7, 153173. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, J.-J.; Yu, L.-C. Anti-nociceptive effect of neuropeptide Y in the nucleus accumbens of rats: An involvement of opioid receptors in the effect. Brain Res 2002, 940, 69–78. [Google Scholar] [CrossRef]
- Liu, J.; Ren, Y.; Li, G.; Liu, Z.-L.; Liu, R.; Tong, Y.; Zhang, L.; Yang, K. GABAB receptors resist acute desensitization in both postsynaptic and presynaptic compartments of periaqueductal gray neurons. Neurosci Lett 2013, 543, 146–151. [Google Scholar] [CrossRef]
- Liu, J.A.; Yu, J.; Cheung, C.W. Immune actions on the peripheral nervous system in pain. Int J Mol Sci 2021, 22, 1448. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, A.; Bair-Marshall, C.; Xu, H.; Jee, H.J.; Zhu, E.; Sun, M.; Zhang, Q.; Lefevre, A.; Chen, Z.S.; et al. Oxytocin promotes prefrontal population activity via the PVN-PFC pathway to regulate pain. Neuron 2023, 111, 1795–1811.e7. [Google Scholar] [CrossRef] [PubMed]
- Lopes, E.F.; West, A.M.; Locke, J.L.; Holleran, K.; Adrian, L.A.; Dawes, M.H.; Curry, A.M.; McKelvey, H.A.; Martin, T.; Jones, S.R. Morphine-induced antinociception is potentiated and dopamine elevations are inhibited by the biased kappa opioid receptor agonist triazole 1.1. ACS Chem Neurosci 2025, 16, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Lopes, P.S.S.; Pinheiro Campos, A.C.; Talamoni Fonoff, E.; Giorgetti Britto, L.R.; Lima Pagano, R. Motor cortex and pain control: Exploring the descending relay analgesic pathways and spinal nociceptive neurons in healthy conscious rats. Behav Brain Funct 2019, 15, 5. [Google Scholar] [CrossRef]
- Louis-Gray, K.; Tupal, S.; Premkuma, L.S. TRPV1, A common denominator mediating antinociceptive and antiemetic effects of cannabinoids. Int J Mol Sci 2022, 23, 10016. [Google Scholar] [CrossRef]
- Lu, Y.; Doroshenko, M.; Lauzakis, J.; Kanjiya, M.P.; Rebecchi, M.J.; Kaczocha, M.; Puopolo, M. Presynaptic inhibition of primary nociceptive signals to dorsal horn lamina I neurons by dopamine. J Neurosci 2018, 38, 8809–8821. [Google Scholar] [CrossRef]
- Luo, J.; Feng, J.; Liu, S.; Walters, E.T.; Hu, H. Molecular and cellular mechanisms that initiate pain and itch. Cell Mo Life Sci 2015, 72, 3201–3223. [Google Scholar] [CrossRef]
- Lyubashina, O.A.; Sivachenko, I.B.; Sushkevich, B.M.; Busygina, I.I. Opposing effects of 5-HT1A receptor agonist buspirone on supraspinal abdominal pain transmission in normal and visceral hypersensitive rats. J Neurosci Res 2023, 101, 1555–1571. [Google Scholar] [CrossRef]
- Maletic, V.; Raison, C.L. Neurobiology of depression, fibromyalgia and neuropathic pain. Front Biosci 2009, 14, 5291–5338. [Google Scholar] [CrossRef] [PubMed]
- Mantyh, P.W.; Koltzenburg, M.; Mendell, L.M.; Tive, L.; Shelton, D.L. Antagonism of nerve growth factor-TrkA signaling and the relief of pain. Anesthesiology 2011, 115, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Manzano-García, A.; González-Hernández, A.; Tello-García, A.; Martínez-Lorenzana, G.; Condés-Lara, M. The role of peripheral vasopressin 1A and oxytocin receptors on the subcutaneous vasopressin antinociceptive effects. Eur J Pain 2018, 22, 511–526. [Google Scholar] [CrossRef]
- Marques-Lopes, J.; Pinho, D.; Albino-Texeira, A.; Tavares, I. The hyperalgesic effects induced by the injection of angiotensin II into the caudal ventrolateral medulla are mediated by the pontine A5 noradrenergic cell group. Brain Res 2010, 1325, 41–52. [Google Scholar] [CrossRef]
- Marques-Lopes, J.; Martins, I.; Pinho, D.; Morato, M.; Wilson, S.P.; Albino-Texeira, A.; Tavares, I. Decrease in the expression of N-methyl-D-aspartate receptors in the nucleus tractus solitarii induces antinociception and increases blood pressure. J Neurosci Res 2012, 90, 356–366. [Google Scholar] [CrossRef]
- Martínez-Lorenzana, G.; Palma-Tirado, L.; Cifuentes-Diaz, C.; González-Hernández, A.; Condés-Lara, M. Ultrastructural evidence for oxytocin and oxytocin receptor at the spinal dorsal horn: Mechanism of nociception modulation. Neuroscience 2021, 475, 117–126. [Google Scholar] [CrossRef]
- Martins, I.; Costa-Araújo, S.; Fadel, J.; Wilson, S.P.; Lima, D.; Tavares, I. Reversal of neuropathic pain by HSV-1-mediated decrease of noradrenaline in a pain facilitatory area of the brain. Pain 2010, 151, 137–145. [Google Scholar] [CrossRef]
- Martins, I.; de Vries, M.G.; Teixeira-Pinto, A.; Fadel, J.; Wilson, S.P.; Westerink, B.H.C.; Tavares, I. Noradrenaline increases pain facilitation from the brain during inflammatory pain. Neuropharmacology 2013, 71, 299–307. [Google Scholar] [CrossRef]
- Martins, I.; Tavares, I. Reticular formation and pain: The past and the future. Front Neuroanat 2017, 11, 51. [Google Scholar] [CrossRef]
- Mavani, G.P.; DeVita, M.V.; Michelis, M.F. A review of the nonpressor and nonantidiuretic actions of the hormone vasopressin. Front Med 2015, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Mazzitelli, M.; Palazzo, E.; Maione, S.; Neugebauer, V. Group II metabotropic glutamate receptors: Role in pain mechanisms and pain modulation. Front Mol Neurosci 2018, 11, 383. [Google Scholar] [CrossRef]
- McEwen, B.S.; Kalia, M. The role of corticosteroids and stress in chronic pain conditions. Metabolism 2010, 59 (Suppl. 1), S9–S15. [Google Scholar] [CrossRef] [PubMed]
- McNearney, T.A.; Westlund, K.N. Pluripotential GluN1 (NMDA NR1): Functional significance in cellular nuclei in pain/nociception. Int J Mol Sci 2023, 24, 13196. [Google Scholar] [CrossRef]
- McPherson, K.B.; Ingram, S.L. Cellular and circuit diversity determines the impact of endogenous opioids in the descending pain modulatory pathway. Front Syst Neurosci 2022, 16, 963812. [Google Scholar] [CrossRef]
- Merighi, A. The histology, physiology, neurochemistry and circuitry of the substantia gelatinosa Rolandi (lamina II) in mammalian spinal cord. Prog Neurobiol 2018, 169, 91–134. [Google Scholar] [CrossRef]
- Merighi, A. Brain-derived neurotrophic factor, nociception, and pain. Biomolecules 2024, 14, 539. [Google Scholar] [CrossRef]
- Merighi, A.; Salio, C.; Ghirri, A.; Lossi, L.; Ferrini, F. BDNF as a pain modulator. Prog Neurobiol 2008, 85, 297–317. [Google Scholar] [CrossRef]
- Meyer, P.J.; Morgan, M.M.; Kozell, L.B.; Ingram, S.L. Contribution of dopamine receptors to periaqueductal gray-mediated antinociception. Psychopharmacology 2009, 204, 531–540. [Google Scholar] [CrossRef]
- Micale, V.; Drago, F. Endocannabinoid system, stress and HPA axis. Eur J Pharmacol 2018, 834, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Micioni Di Bonaventura, E.; Botticelli, L.; Del Bello, F.; Giorgioni, G.; Piergentili, A.; Quaglia, W.; Romano, A.; Gaetani, S.; Micioni Di Bonaventura, M.V.; Cifani, C. Investigating the role of the central melanocortin system in stress and stress-related disorders. Pharmacol Res 2022, 185, 106521. [Google Scholar] [CrossRef] [PubMed]
- Milligan, A.L.; Szabo-Pardi, T.A.; Burton, M.D. Cannabinoid receptor type 1 and its role as an analgesic: An opioid alternative? J Dual Diagn 2019, 16, 106–119. [Google Scholar] [CrossRef]
- Millón, C.; Flores-Burgess, A.; Narváez, M.; Borroto-Escuela, D.O.; Gago, B.; Santin, L.; Castilla-Ortega, E.; Narváez, J.A.; Fuxe, K.; Díaz-Cabiale, Z. The neuropeptides Galanin and Galanin(1-15) in depression-like behaviours. Neuropeptides 2017, 64, 39–45. [Google Scholar] [CrossRef]
- Mills, E.P.; Keay, K.A.; Henderson, L.A. Brainstem pain-modulation circuitry and its plasticity in neuropathic pain: Insights from human brain imaging investigations. Front Pain Res 2021, 2, 705345. [Google Scholar] [CrossRef]
- Mitsi, V.; Zachariou, V. Modulation of pain, nociception, and analgesia by the brain reward center. Neuroscience 2016, 338, 81–92. [Google Scholar] [CrossRef]
- Mizumura, K.; Murase, S. Role of nerve growth factor in pain. Handb Exp Pharmacol 2015, 227, 57–77. [Google Scholar] [PubMed]
- Moalem, G.; Tracey, D.J. Immune and inflammatory mechanisms in neuropathic pain. Brain Res Rev 2006, 51, 240–264. [Google Scholar] [CrossRef] [PubMed]
- Mogil, J.S.; Wilson, S.G.; Chesler, E.J.; Rankin, A.L.; Nemmani, K.V.S.; Lariviere, W.R.; Groce, M.K.; Wallace, M.R.; Kaplan, L.; Staud, R.; et al. The melanocortin-1 receptor gene mediates female-specific mechanisms of analgesia in mice and humans. Proc Natl Acad Sci USA 2003, 100, 4867–4872. [Google Scholar] [CrossRef]
- Morales, M.; Margolis, E.B. Ventral tegmental area: Cellular heterogeneity, connectivity and behaviour. Nat Rev Neurosci 2017, 18, 73–85. [Google Scholar] [CrossRef]
- Morena, M.; Patel, S.; Bains, J.S.; Hill, M.N. Neurobiological interactions between stress and the endocannabinoid system. Neuropsychopharmacology 2016, 41, 80–102. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.M.; Whittier, K.L.; Hegarty, D.M.; Aicher, S.A. Periaqueductal gray neurons project to spinally projecting GABAergic neurons in the rostral ventromedial medulla. Pain 2008, 140, 376–386. [Google Scholar] [CrossRef]
- Muñoz, A.; Lopez-Lopez, A.; Labandeira, C.M.; Labandeira-Garcia, J.L. Interactions between the serotonergic and other neurotransmitter systems in the basal ganglia: Role in Parkinson’s Disease and adverse effects of L-DOPA. Front Neuroanat 2020, 14, 26. [Google Scholar] [CrossRef]
- Muñoz, M.; Coveñas, R. Involvement of substance P and the NK-1 receptor in human pathology. Amino Acids 2014, 46, 1727–1750. [Google Scholar] [CrossRef]
- Mussetto, V.; Teuchmann, H.L.; Heinke, B.; Trofimova, L.; Sandkühler, J.; Drdla-Schutting, R.; Hogri, R. Opioids induce bidirectional synaptic plasticity in a brainstem pain center in the rat. J Pain 2023, 24, 1664–1680. [Google Scholar] [CrossRef] [PubMed]
- Mutolo, D.; Cinelli, E.; Bongianni, F.; Pantaleo, T. Inhibitory control of the cough reflex by galanin receptors in the caudal nucleus tractus solitarii of the rabbit. Am J Physiol Regul Integr Comp Physiol 2014, 307, R1358-67. [Google Scholar] [CrossRef] [PubMed]
- Nadrigny, F.; Le Meur, K.; Schomburg, E.D.; Safavi-Abbasi, S.; Dibaj, P. Two-photon laser-scanning microscopy for single and repetitive imaging of dorsal and lateral spinal white matter in vivo. Physiol Res 2017, 66, 531–537. [Google Scholar] [CrossRef]
- Nakamura, Y.; Fukushige, R.; Watanabe, K.; Kishida, Y.; Hisaoka-Nakashima, K.; Nakata, Y.; Morioka, N. Continuous infusion of substance P inhibits acute, but not subacute, inflammatory pain induced by complete Freund’s adjuvant. Biochem Biophys Res Commun 2020, 533, 971–975. [Google Scholar] [CrossRef]
- Naser, P.V.; Kuner, R. Molecular, cellular and circuit basis of cholinergic modulation of pain. Neuroscience 2018, 387, 135–148. [Google Scholar] [CrossRef]
- Navratilova, E.; Qu, C.; Ji, G.; Neugebauer, V.; Guerrero, M.; Rosen, H.; Roberts, E.; Porreca, F. Opposing effects on descending control of nociception by µ and κ opioid receptors in the anterior cingulate cortex. Anesthesiology 2024, 40, 272–283. [Google Scholar] [CrossRef]
- Nelson, A.B.; Kreitzer, A.C. Reassessing models of basal ganglia function and dysfunction. Annu Rev Neurosci. 2014, 37, 117–135. [Google Scholar] [CrossRef]
- Nelson, T.S.; Allen, H.A.; Khanna, R. Neuropeptide Y and pain: Insights from brain research. ACS Pharmacol Transl Sci 2024, 7, 3718–3728. [Google Scholar] [CrossRef]
- Nelson, T.S.; Taylor, B.K. Targeting spinal neuropeptide Y1 receptor-expressing interneurons to alleviate chronic pain and itch. Prog Neurobiol 2021, 196, 101894. [Google Scholar] [CrossRef] [PubMed]
- Neubert, M.J.; Kincaid, W.; Heinricher, M.M. Nociceptive facilitating neurons in the rostral ventromedial medulla. Pain 2004, 110, 158–165. [Google Scholar] [CrossRef]
- Neugebauer, V. Amygdala pain mechanisms. Handb Exp Pharmacol 2015, 227, 261–284. [Google Scholar] [PubMed]
- Neugebauer, V.; Galhardo, V.; Maione, S.; Mackey, S.C. Forebrain pain mechanisms. Brain Res Rev 2009, 60, 226–242. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, V.; Mazzitelli, M.; Cragg, B.; Ji, G.; Navratilova, E.; Porreca, F. Amygdala, neuropeptides, and chronic pain-related affective behaviors. Neuropharmacology 2020, 170, 108052. [Google Scholar] [CrossRef]
- Neugebauer, V.; Presto, P.; Yakhnitsa, V.; Antenucci, N.; Mendoza, B.; Ji, G. Pain-related cortico-limbic plasticity and opioid signaling. Neuropharmacology 2023, 231, 109510. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, E.; Grajales-Reyes, J.G.; Gereau, R.W., 4th; Ross, S.E. Cell type-specific dissection of sensory pathways involved in descending modulation. Trends Neurosci 2023, 46, 539–550. [Google Scholar] [CrossRef]
- Nicol, G.D.; Vasko, M.R. Unraveling the story of NGF-mediated sensitization of nociceptive sensory neurons: ON or OFF the Trks? Mol Interv 2007, 7, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.B. Human spinal motor control. Annu Rev Neurosci 2016, 39, 81–101. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, H.; Kawasaki, M.; Matsuura, T.; Suzuki, H.; Motojima, Y.; Baba, K.; Ohnishi, H.; Yamanaka, Y.; Fujuitani, T.; Yoshimura, M.; et al. Acute mono-arthritis activates the neurohypophysial system and hypothalamo-pituitary adrenal axis in rats. Front Endocrinol 2020, 11, 43. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, T.; Gyermek, L.; Lee, C.; Kawasaki-Yatsugi, S.; Yamaguchi, T. Analgesic interaction between intrathecal midazolam and glutamate receptor antagonists on thermal-induced pain in rats. Anesthesiology 1999, 91, 531–537. [Google Scholar] [CrossRef]
- Olivier, B. Serotonin: A never-ending story. Eur J Pharmacol 2015, 753, 2–18. [Google Scholar] [CrossRef]
- Ong, W.Y.; Stohler, C.S.; Herr, D.R. Role of the prefrontal cortex in pain processing. Mol Neurobiol 2019, 56, 1137–1166. [Google Scholar] [CrossRef]
- O’Neill, A.; Lirk, P. Multimodal analgesia. Anesthesiol Clin 2022, 40, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Osório, C.; Probert, T.; Jones, E.; Young, A.H.; Robbins, I. Adapting to stress: Understanding the neurobiology of resilience. Behav Med 2017, 43, 307–322. [Google Scholar] [CrossRef]
- Ossipov, M.H.; Dussor, G.O.; Porreca, F. Central modulation of pain. J Clin Invest 2010, 120, 3779–3787. [Google Scholar] [CrossRef] [PubMed]
- Ossipov, M.H.; Morimura, K.; Porreca, F. Descending pain modulation and chronification of pain. Curr Opin Support Palliat Care 2014, 8, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Oyola, M.G.; Handa, R.J. Hypothalamic-pituitary-adrenal and hypothalamic-pituitary-gonadal axes: Sex differences in regulation of stress responsivity. Stress 2017, 20, 476–494. [Google Scholar] [CrossRef] [PubMed]
- Pagliusi, M.; Gomes, F.V. The role of the rostral ventromedial medulla in stress responses. Brain Sci 2023, 13, 776. [Google Scholar] [CrossRef]
- Palazzo, E.; Luongo, L.; de Novellis, V.; Rossi, F.; Maione, S. The role of cannabinoid receptors in the descending modulation of pain. Pharmaceuticals 2010, 3, 2661–2673. [Google Scholar] [CrossRef]
- Pan, H.-L.; Wu, Z.-Z.; Zhou, H.-Y.; Chen, S.-R.; Zhang, H.-M.; Li, D.-P. Modulation of pain transmission by G-protein-coupled receptors. Pharmacol Ther 2007, 117, 141–161. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.-C.; Song, Y.-T.; Liu, C.; Xiang, H.-B.; Lu, C.-J. Melanocortin-4 receptor expression in the rostral ventromedial medulla involved in modulation of nociception in transgenic mice. J Huazhong Univ Sci Technolog Med Sci 2013, 33, 195–198. [Google Scholar] [CrossRef]
- Pape, H.-C.; Jüngling, K.; Seidenbecher, T.; Lesting, J.; Reinscheid, R.K. Neuropeptide S: A transmitter system in the brain regulating fear and anxiety. Neuropharmacology 2010, 58, 29–34. [Google Scholar] [CrossRef]
- Patte-Mensah, C.; Meyer, L.; Taleb, O.; Mensah-Nyagan, A.G. Potential role of allopregnanolone for a safe and effective therapy of neuropathic pain. Prog Neurobiol 2014, 113, 70–78. [Google Scholar] [CrossRef]
- Patra, P.H.; Tench, B.; Hitrec, T.; Hohnes, F.; Drake, R.; Cerritelli, S.; Spanswick, D.; Pickering, A.E. Pro-opiomelanocortin neurons in the nucleus of the solitary tract mediate endorphinergic endogenous analgesia in mice. Pain 2023, 164, 1051–1066. [Google Scholar] [CrossRef]
- Peng, B.; Jiao, Y.; Zhang, Y.; Li, S.; Chen, S.; Xu, S.; Gao, P.; Fan, Y.; Yu, W. Bulbospinal nociceptive ON and OFF cells related neural circuits and transmitters. Front Pharmacol 2023, 14, 1159753. [Google Scholar] [CrossRef]
- Peng, H.-Y.; Chen, G.-D.; Lee, S.-D.; Lai, C.-Y.; Chiu, C.-H.; Cheng, Y.-L.; Chang, Y.-S.; Hsieh, M.-C.; Tung, K.-C.; Lin, T.-B. Neuroactive steroids inhibit spinal reflex potentiation by selectively enhancing specific spinal GABA(A) receptor subtypes. Pain 2009, 143, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.; Goudet, C. Emerging trends in pain modulation by metabotropic glutamate receptors. Front Mol Neurosci 2019, 11, 464. [Google Scholar] [CrossRef] [PubMed]
- Pertovaara, A. NA pain modulation. Prog Neurobiol 2006, 80, 53–83. [Google Scholar] [CrossRef]
- Pertovaara, A. The NA pain regulation system: A potential target for pain therapy. Eur J Pharmacol 2013, 716, 2–7. [Google Scholar] [CrossRef]
- Pertovaara, A.; Bravo, R.; Herdegen, T. Induction and suppression of immediate-early genes in the rat brain by a selective alpha-2-adrenoceptor agonist and antagonist following noxious peripheral stimulation. Neuroscience 1993, 54, 117–126. [Google Scholar] [CrossRef]
- Pezet, S.; Cunningham, J.; Patel, J.; Grist, J.; Gavazzi, I.; Lever, I.J.; Malcangio, M. BDNF modulates sensory neuron synaptic activity by a facilitation of GABA transmission in the dorsal horn. Mol Cell Neurosci 2002, 21, 51–62. [Google Scholar] [CrossRef]
- Pezet, S.; McMahon, S.B. Neurotrophins: Mediators and modulators of pain. Annu Rev Neurosci 2006, 29, 507–538. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C. Brain-derived neurotrophic factor, depression, and physical activity: Making the Neuroplastic Connection. Neural Plast 2017, 2017, 7260130. [Google Scholar] [CrossRef]
- Pinto, M.; Castro, A.R.; Tshudy, F.; Wilson, S.P.; Lima, D.; Tavares, I. Opioids modulate pain facilitation from the dorsal reticular nucleus. Mol Cell Neurosci 2008, 39, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.; Sousa, M.; Lima, D.; Tavares, I. Participation of mu-opioid, GABA(B), and NK1 receptors of major pain control medullary areas in pathways targeting the rat spinal cord: Implications for descending modulation of nociceptive transmission. J Comp Neurol 2008, 510, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Pitsillou, E.; Bresnehan, S.M.; Kagarakis, E.A.; Wijoyo, S.J.; Liang, J.; Hung, A.; Karagiannis, T.C. The cellular and molecular basis of major depressive disorder: Towards a unified model for understanding clinical depression. Mol Biol Rep 2020, 47, 753–770. [Google Scholar] [CrossRef]
- Ploski, J.E.; Vaidya, V.A. The neurocircuitry of posttraumatic stress disorder and major depression: Insights into overlapping and distinct circuit dysfunction—A tribute to Ron Duman. Biol Psychiatry 2021, 90, 109–117. [Google Scholar] [CrossRef]
- Plumb, A.N.; Lesnak, J.B.; Rasmussen, L.; Sluka, K.A. Female specific interactions of serotonin and testosterone in the rostral ventromedial medulla after activity-induced muscle pain. J Pain 2025, 26, 104723. [Google Scholar] [CrossRef]
- Poe, G.R.; Foote, S.; Eschenko, O.; Johansen, J.P.; Bouret, S.; Aston-Jones, G.; Harley, C.W.; Manahan-Vaughan, D.; Weinshenker, D.; Valentino, R.; et al. Locus coeruleus: A new look at the blue spot. Nat Rev Neurosci 2020, 21, 644–659. [Google Scholar] [CrossRef]
- Pogatzki-Zahn, E.M.; Segelcke, D.; Schug, S.A. Postoperative pain—From mechanisms to treatment. Pain Rep 2017, 2, e588. [Google Scholar] [CrossRef] [PubMed]
- Poisbeau, P.; Grinevich, V.; Charlet, A. Oxytocin signaling in pain: Cellular, circuit, system, and behavioral levels. Curr Top Behav Neurosci 2018, 35, 193–211. [Google Scholar] [PubMed]
- Poisbeau, P.; Keller, A.F.; Aouad, M.; Kamoun, N.; Groyer, G.; Schuhmacher, M. Analgesic strategies aimed at stimulating the endogenous production of allopregnanolone. Front Cell Neurosci 2014, 8, 174. [Google Scholar] [CrossRef] [PubMed]
- Popescu AT, Saghyan AA, Paré D NMDA-dependent facilitation of corticostriatal plasticity by the amygdala. Proc Natl Acad Sci USA 2007, 104, 341–346. [CrossRef]
- Puopolo, M. The hypothalamic-spinal dopaminergic system: A target for pain modulation. Neural Regen Res 2019, 14, 925–930. [Google Scholar] [CrossRef]
- Qian, K.; Zhou, J.; Xiong, J.; Wang, Q.; Chen, L.; Zhuang, T.; Jin, J.; Zhang, G.; Hao, C.; Huang, L.; et al. Discovery of a novel Kv7.2/7.3 channels agonist for the treatment of neuropathic pain. Eur J Med Chem 2024, 280, 116953. [Google Scholar] [CrossRef]
- Quevedo, J.N. Presynaptic inhibition. In Encyclopedia of Neuroscience; Binder, M.D., Hirokawa, N., Windhorst, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 3266–3270. [Google Scholar]
- Raggenbass, M. Overview of cellular electrophysiological actions of vasopressin. Eur J Pharmacol 2008, 583, 243–254. [Google Scholar] [CrossRef]
- Rahimi, K.; Sajedianfard, J.; Owji, A.A. The effect of intracerebroventricular injection of CGRP on pain behavioral responses and monoamines concentrations in the periaqueductal gray area in rat. Iran J Basic Med Sci 2018, 21, 395–399. [Google Scholar]
- Rana, T.; Behl, T.; Sehgal, A.; Singh, S.; Sharma, N.; Abdeen, A.; Ibrahim, S.F.; Mani, V.; Iqbal, M.S.; Bhatia, S.; et al. Exploring the role of neuropeptides in depression and anxiety. Prog Neuropsychopharmacol Biol Psychiatry 2022, 114, 110478. [Google Scholar] [CrossRef]
- Rashvand, M.; Khajavi, A.; Parviz, M.; Hasanein, P.; Keshavarz, M. GABAA receptors are involved in the analgesic effects of morphine microinjected into the central nucleus of the amygdala. Clin Exp Pharmacol Physiol 2014, 41, 338–344. [Google Scholar] [CrossRef]
- Razavi, B.M.; Hosseinzadeh, H. A review of the role of orexin system in pain modulation. Biomed Pharmacother 2017, 90, 187–193. [Google Scholar] [CrossRef]
- Rea, K.; Roche, M.; Finn, D.P. Supraspinal modulation of pain by cannabinoids: The role of GABA and glutamate. Br J Pharmacol 2007, 152, 633–648. [Google Scholar] [CrossRef]
- Ren, S.; Wang, S.; Lv, S.; Gao, J.; Mao, Y.; Liu, Y.; Xie, Q.; Zhang, T.; Zhao, L.; Shi, J. The nociceptive inputs of the paraventricular hypothalamic nucleus in formalin stimulated mice. Neurosci Lett 2024, 841, 137948. [Google Scholar] [CrossRef]
- Ribeiro-da-Silva, A.; De Koninck, Y.; Cuello, A.D.; Henry, J.L. Enkephalin-immunoreactive nociceptive neurons in the cat spinal cord. Neuroreport 1992, 3, 25–58. [Google Scholar] [CrossRef]
- Roberts JW Cholecystokinin and pain. Anesth Prog 1986, 33, 87–90.
- Rogoz, K.; Andersen, H.H.; Lagerström, M.C.; Kullander, K. Multimodal use of calcitonin gene-related peptide and substance P in itch and acute pain uncovered by the elimination of vesicular glutamate transporter 2 from transient receptor potential cation channel subfamily V member 1 neurons. J Neurosci 2014, 34, 14055–14068. [Google Scholar] [CrossRef]
- Rojas-Piloni, G.; Mejía-Rodríguez, R.; Martínez-Lorenzana, G.; Condés-Lara, M. Oxytocin, but not vassopressin, modulates nociceptive responses in dorsal horn neurons. Neurosci Lett 2010, 476, 32–35. [Google Scholar]
- Rosen, J.B.; Schulkin, J. Hyperexcitability: From normal fear to pathological anxiety and trauma. Front Syst Neurosci 2022, 16, 727054. [Google Scholar] [CrossRef] [PubMed]
- Roth, R.H.; Ding, J.B. Cortico-basal ganglia plasticity in motor learning. Neuron 2024, 112, 2486–2502. [Google Scholar] [CrossRef]
- Roussy, G.; Dansereau, M.-A.; Baudisson, S.; Ezzoubaa, F.; Belleville, K.; Beaudet, N.; Martinez, J.; Richelson, E.; Sarret, P. Evidence for a role of NTS2 receptors in the modulation of tonic pain sensitivity. Mol Pain 2009, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P. In search of lost presynaptic inhibition. Exp Brain Res 2009, 196, 139–151. [Google Scholar] [CrossRef]
- Rudomin, P.; Schmidt, R.F. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res 1999, 129, 1–37. [Google Scholar] [CrossRef]
- Sabban, E.L.; Alaluf, L.G.; Serova, L.I. Potential of neuropeptide Y for preventing or treating post-traumatic stress disorder. Neuropeptides 2016, 56, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Salgado, S.; Kaplitt, M.G. The nucleus accumbens: A comprehensive review. Stereotact Funct Neurosurg 2015, 93, 75–93. [Google Scholar] [CrossRef] [PubMed]
- Salt, T.E.; Jones, H.E.; Copeland, C.S.; Sillito, A.M. Function of mGlu1 receptors in the modulation of nociceptive processing in the thalamus. Neuropharmacology 2014, 79, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, M.; Pawlowski, S.A.; Barthas, F.; Yalcin, I.; Kaufling, J.; Dardente, H.; Zachariou, V.; Dileone, J.; Barrot, M.; Veinante, P. BDNF parabrachio-amygdaloid pathway in morphine-induced analgesia. Int J Neuropsychopharmacol 2013, 16, 1649–1660. [Google Scholar] [CrossRef]
- Savadipour, A.; Palmer, D.; Ely, E.V.; Collins, K.H.; Garcia-Castorena, J.M.; Harissa, Z.; Kim, Y.S.; Oestrich, A.; Qu, F.; Rashidi, N.; et al. The role of PIEZO ion channels in the musculoskeletal system. Am J Physiol Cell Physiol 2023, 324, C728–C740. [Google Scholar] [CrossRef]
- Sawchenko, P.E.; Swanson, L.W. Immunohistochemical identification of neurons in the paraventricular nucleus of the hypothalamus that project to the medulla or to the spinal cord in the rat. J Comp Neurol 1982, 205, 260–272. [Google Scholar] [CrossRef]
- Sawynok, J. Adenosine receptor activation and nociception. Eur J Pharmacol 1998, 347, 1–11. [Google Scholar] [CrossRef]
- Schmeltzer, S.N.; Herman, J.P.; Sah, R. Neuropeptide Y (NPY) and Posttraumatic Stress Disorder (PTSD): A translational update. Exp Neurol 2016, 284 Pt B, 196–210. [Google Scholar] [CrossRef]
- Schomburg, E.D.; Dibaj, P.; Steffens, H. Differentiation between Aδ and C fibre evoked nociceptive reflexes by TTX resistance and opioid sensitivity in the cat. Neurosci Res 2011, 69, 241–245. [Google Scholar] [CrossRef]
- Schomburg, E.D.; Dibaj, P.; Steffens, H. Role of L-DOPA in spinal nociceptive reflex activity: Higher sensitivity of Aδ versus C fibre-evoked nociceptive reflexes to L-DOPA. Physiol Res 2011, 60, 701–703. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, E.D.; Kalezic, I.; Dibaj, P.; Steffens, H. Reflex transmission to lumbar α-motoneurones in the mouse similar and different to those in the cat. Neurosci Res 2013, 76, 133–140. [Google Scholar] [CrossRef]
- Schomburg, E.D.; Steffens, H.; Dibaj, P.; Sears, T.A. Major contribution of Aδ-fibres to increased reflex transmission in the feline spinal cord during acute muscle inflammation. Neurosci Res 2012, 72, 155–162. [Google Scholar] [CrossRef]
- Schomburg, E.D.; Steffens, H.; Pilyavskii, A.I.; Maisky, V.A.; Brück, W.; Dibaj, P.; Sears, T.A. Long lasting activity of nociceptive muscular afferents facilitates bilateral flexion pattern in the feline spinal cord. Neurosci Res 2015, 95, 51–58. [Google Scholar] [CrossRef]
- Schou, W.S.; Ashina, S.; Amin, F.M.; Goadsby, P.J.; Ashina, M. Calcitonin gene-related peptide and pain: A systematic review. J Headache Pain 2017, 18, 34. [Google Scholar] [CrossRef]
- Schumacher, M.; Mattern, C.; Ghoumari, A.; Oudinet, J.P.; Liere, P.; Labombarda, F.; Sitruk-Ware, R.; De Nicola, A.F.; Guennoun, R. Revisiting the roles of progesterone and allopregnanolone in the nervous system: Resurgence of the progesterone receptors. Prog Neurobiol 2014, 113, 6–39. [Google Scholar] [CrossRef] [PubMed]
- Selye, H. Anaesthetic effects of steroid hormones. Proc Soc Exp Biol 1941, 46, 116–121. [Google Scholar] [CrossRef]
- Semba, K.; Fibiger, H.C.; Vincent, S.R. Neurotransmitters in the mammalian striatum: Neuronal circuits and heterogeneity. Can J Neurol Sci 1987, 14 (Suppl. 3), 386–394. [Google Scholar] [CrossRef]
- Seminowicz, D.A.; Moayedi, M. The dorsolateral prefrontal cortex in acute and chronic pain. Pain 2017, 18, 1027–1035. [Google Scholar] [CrossRef]
- Sexton, J.E.; Vernon, J.; Wood, J.N. TRPs and pain. Handb Exp Pharmacol 2014, 223, 873–897. [Google Scholar] [PubMed]
- Shenoy, S.S.; Lui, F. Biochemistry, Endogenous opioids. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Shono, K.; Yamamoto, T. Orexin-2 receptors inhibit primary afferent fiber-evoked responses of ventral roots in the neonatal rat isolated spinal cord. Brain Res 2008, 1218, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Soares Potes, C.; Neto, F.L.; Castro-Lopes, J.M. Inhibition of pain behavior by GABA(B) receptors in the thalamic ventrobasal complex: Effect on normal rats subjected to the formalin test of nociception. Brain Res 2006, 1115, 37–47. [Google Scholar] [CrossRef]
- Spooner, M.-F.; Robichaud, P.; Carrier, J.C.; Marchand, S. Endogenous pain modulation during the formalin test in estrogen receptor beta knockout mice. Neuroscience 2007, 150, 675–680. [Google Scholar] [CrossRef]
- Spuz, C.A.; Tomaszycki, M.L.; Borszez, G.S. N-methyl-D-aspartate receptor agonism and antagonism within the amygdaloid central nucleus suppresses pain affect: Differential contribution of the ventrolateral periaqueductal gray. J Pain 2014, 15, 1305–1318. [Google Scholar] [CrossRef]
- Stanton, C.H.; Holmes, A.J.; Chang, S.W.C.; Joorman, J. From stress to anhedonia: Molecular processes through functional circuits. Trends Neurosci 2019, 42, 23–42. [Google Scholar] [CrossRef]
- Stebbins, C.L.; Ortiz-Acevedo, A.; Hill, J.M. Spinal vasopressin modulates the reflex cardiovascular response to static contraction. J Appl Physiol 1985 1992, 72, 731–738. [Google Scholar] [CrossRef]
- Sze, Y.; Brunton, P.J. Sex, stress and steroids. Eur J Neurosci 2020, 52, 2487–2515. [Google Scholar] [CrossRef]
- Takayanagi, Y.; Onaka, T. Roles of oxytocin in stress responses, allostasis and resilience. Int J Mol Sci 2021, 23, 150. [Google Scholar] [CrossRef]
- Tanaka, M.; Matsumoto, Y.; Murakami, T.; Ibata, Y. The origins of catecholaminergic innervation in the rostral ventromedial medulla oblongata of the rat. Neurosci Lett 1996, 207, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Yamada, S.; Watanabe, Y. The role of neuropeptide Y in the nucleus accumbens. Int J Mol Sci 2021, 22, 7287. [Google Scholar] [CrossRef]
- Tanaka, M.; Yoshida, M.; Emoto, H.; Ishii, H. Noradrenaline systems in the hypothalamus, amygdala and locus coeruleus are involved in the provocation of anxiety: Basic studies. Eur J Pharmacol 2000, 405, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.-S.; Qu, C.-L.; Huo, F.-Q. The thalamic nucleus. Prog Neurobiol 2009, 89, 383–389. [Google Scholar] [CrossRef]
- Tasker, J.G.; Chen, C.; Fisher, M.O.; Fu, X.; Rainville, J.R.; Weiss, G.L. Endocannabinoid regulation of neuroendocrine systems. Int Rev Neurobiol 2015, 125, 163–201. [Google Scholar] [PubMed]
- Tavares, I.; Costa-Pereira, J.T.; Martins, I. Monoaminergic and opioidergic modulation of brainstem circuits: New insights into the clinical challenges of pain treatment? Front Pain Res 2021, 2, 696515. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.W.; Becker, S.; Schweinhardt, P.; Cahill, C. Mesolimbic dopamine signaling in acute and chronic pain: Implications for motivation, analgesia, and addiction. Pain 2016, 157, 1194–1198. [Google Scholar] [CrossRef]
- Thompson, J.M.; Neugebauer, V. Cortico-limbic pain mechanisms. Neurosci Lett 2019, 702, 15–23. [Google Scholar] [CrossRef]
- Thurston, C.L.; Culhane, E.S.; Suberg, S.N.; Carstens, E.; Watkins, L.R. Antinociception vs motor effects of intrathecal vasopressin as measured by four pain tests. Brain Res 1988, 463, 1–11. [Google Scholar] [CrossRef]
- Timmers I, Quaedflieg CWEM, Hsu C, Heathcote LC, Rovnaghi CR, Simons LE The interaction between stress and chronic pain through the lens of threat learning. Neurosci Biobehav Rev 2019, 107, 641–655. [CrossRef]
- Tewari, A.; Jog, R.; Jog, M.S. The striatum and subthalamic nucleus as independent and collaborative structures in motor control. Front Syst Neurosci 2016, 10, 17. [Google Scholar] [CrossRef]
- Tobiansky, D.J.; Wallin-Miller, K.G.; Floresco, S.B.; Wood, R.I.; Soma, K.K. Androgen regulation of the mesocorticolimbic system and executive function. Front Endocrinol 2018, 9, 279. [Google Scholar] [CrossRef] [PubMed]
- Todd AJSpike, R.C. The localization of classical transmitters and neuropeptides within neurons in laminae I-III of the mammalian spinal dorsal horn. Prog Neurobiol 1993, 41, 609–645. [Google Scholar] [CrossRef]
- Toufexis, D.; Rivarola, M.A.; Lara, H.; Viau, V. Stress and the reproductive axis. J Neuroendocrinol 2014, 26, 573–586. [Google Scholar] [CrossRef]
- Tracey, I.; Mantyh, P.W. The cerebral signature for pain perception and its modulation. Neuron 2007, 55, 377–391. [Google Scholar] [CrossRef]
- Tran, M.; Kuhn, J.A.; Bráz, J.M.; Basbaum, A.I. Neuronal aromatase expression in pain processing regions of the medullary and spinal cord dorsal horn. J Comp Neurol 2017, 525, 3414–3428. [Google Scholar] [CrossRef]
- Treister, R.; Pud, D.; Eisenberg, E. The dopamine agonist apomorphine enhances conditioned pain modulation in healthy humans. Neurosci Lett 2013, 548, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Urban, M.O.; Coutinho, S.V.; Gebhart, G.F. Biphasic modulation of visceral nociception by neurotensin in rat rostral ventromedial medulla. J Pharmacol Exp Ther 1999, 290, 207–213. [Google Scholar] [CrossRef]
- Valentino, R.J.; Van Bockstaele, E. Endogenous opioids: The downside of opposing stress. Neurobiol Stress 2015, 1, 23–32. [Google Scholar] [CrossRef]
- Vazifeshenas, A.; Seddighfar, M.; Haghparast, A. Interaction of opioid and D2-like dopamine receptors in the nucleus accumbens modulate acute pain-related behaviors. Behav Brain Res 2025, 489, 115614. [Google Scholar] [CrossRef] [PubMed]
- Viisanen, H.; Lilius, T.O.; Sagalajev, B.; Rauhala, P.; Kalso, E.; Pertovaara, A. Neurophysiological response properties of medullary pain-control neurons following chronic treatment with morphine or oxycodone: Modulation by acute ketamine. J Neurophysiol 2020, 124, 790–801. [Google Scholar] [CrossRef]
- Vogt, B.A. Cingulate cortex in the three limbic subsystems. Handb Clin Neurol 2019, 166, 39–51. [Google Scholar]
- Vrtačnik, P.; Ostanek, B.; Mencej-Bedrač, S.; Marc, J. The many faces of estrogen signaling. Biochem Med 2014, 24, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Wagner, K.M.; Roeder, Z.; Desrochers, K.; Buhler, A.V.; Heinricher, M.M.; Cleary, D.R. The dorsomedial hypothalamus mediates stress-induced hyperalgesia and is the source of the pronociceptive peptide cholecystokinin in the rostral ventromedial medulla. Neuroscience 2013, 238, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.M.; Magnuson, D.J.; Gray, T.S. Organization of amygdaloid projections to brainstem dopaminergic, noradrenergic, and adrenergic cell groups in the rat. Brain Res Bull 1992, 28, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Tawfik, V.L.; Corder, G.; Low, S.A.; François, A.; Basbaum, A.I.; Scherrer, G. Functional Divergence of Delta and Mu Opioid Receptor Organization in CNS Pain Circuits. Neuron 2018, 98, 90–108.e5. [Google Scholar] [CrossRef]
- Wang, N.; Wang, J.-Y.; Luo, F. Corticofugal outputs facilitate acute, but inhibit chronic pain in rats. Pain 2009, 142, 108–115. [Google Scholar] [CrossRef]
- Wang, S.; Lim, J.; Joseph, J.; Wang, S.; Wei, F.; Ro, J.Y.; Chung, M. Spontaneous and bite-evoked muscle pain are mediated by a common nociceptive pathway with differential contribution by TRPV1. J Pain 2017, 18, 1333–1345. [Google Scholar] [CrossRef]
- Wang, X.Q.; Mokhtari, T.; Zeng, Y.X.; Yue, L.P.; Hu, L. The distinct functions of the dopaminergic receptors on pain modulation: A narrative review. Neural Plast 2021, 2021, 6682275. [Google Scholar] [CrossRef]
- Watanabe, M.; Narita, M. Brain reward circuit and pain. Adv Exp Med Biol 2018, 1099, 201–210. [Google Scholar]
- Watanabe, M.; Sugiura, Y.; Sugiyama, E.; Narita, M.; Navratilova, E.; Kondo, T.; Uchiyama, N.; Yamanaka, A.; Kuzumaki, N.; Porreca, F.; et al. Extracellular N-acetylaspartylglutamate released in the nucleus accumbens modulates the pain sensation: Analysis using a microdialysis/mass spectrometry integrated system. Mol Pain 2018, 14, 1744806918754934. [Google Scholar] [CrossRef]
- Watanabe, Y.; Ikegaya, Y.; Saito, H.; Abe, K. Roles of GABAA, NMDA and muscarinic receptors in induction of long-term potentiation in the medial and lateral amygdala in vitro. Neurosci Res 1995, 21, 317–322. [Google Scholar] [CrossRef]
- Watkins, L.R.; Suberg, S.N.; Thurston, C.L.; Culhane, E.S. Role of spinal cord neuropeptides in pain sensitivity and analgesia: Thyrotropin releasing hormone and vasopressin. Brain Res 1986, 362, 308–317. [Google Scholar] [CrossRef]
- Wei, H.-H.; Yuan, X.-S.; Chen, Z.-K.; Chen, P.-P.; Xiang, Z.; Qu, W.-M.; Li, R.-X.; Zhou, G.-M.; Huang, Z.-L. Presynaptic inputs to vasopressin neurons in the hypothalamic supraoptic nucleus and paraventricular nucleus in mice. Exp Neurol 2021, 343, 113784. [Google Scholar] [CrossRef]
- Weisskopf, M.G.; LeDoux, J.E. Distinct populations of NMDA receptors at subcortical and cortical inputs to principal cells of the lateral amygdala. J Neurophysiol 1999, 81, 930–934. [Google Scholar] [CrossRef] [PubMed]
- West, S.J.; Bannister, K.; Dickenson, A.H.; Bennett, D.L. Circuitry and plasticity of the dorsal horn—toward a better understanding of neuropathic pain. Neuroscience 2015, 300, 254–275. [Google Scholar] [CrossRef]
- Wichmann, T.; DeLong, M.R. Deep brain stimulation for movement disorders of basal ganglia origin: Restoring function or functionality? Neurotherapeutics 2016, 13, 264–283. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.R.; Maher, L.; Morgan, M.M. Repeated cannabinoid injections into the rat periaqueductal gray enhance subsequent morphine antinociception. Neuropharmacology 2008, 55, 1219–1225. [Google Scholar] [CrossRef] [PubMed]
- Windhorst, U. Muscle proprioceptive feedback and spinal networks. Brain Res Bull 2007, 73, 155–202. [Google Scholar] [CrossRef]
- Windhorst, U. Spinal cord circuits: Models and reality. Neurophysiology 2021, 53, 142–222. [Google Scholar] [CrossRef]
- Windhorst, U.; Dibaj, P. Plastic spinal motor circuits in health and disease. J Integr Neurosci 2023, 22, 167. [Google Scholar] [CrossRef]
- Windhorst, U.; Dibaj, P. Nociception and acute pain: Ascending functional Structure and descending modulation. Preprints 2025, 2025050743. [Google Scholar] [CrossRef]
- Wood, P.B. Role of central dopamine in pain and analgesia. Expert Rev Neurother 2008, 8, 781–797. [Google Scholar] [CrossRef]
- Woodhams, S.G.; Chapman, V.; Finn, D.P.; Hohmann, A.G.; Neugebauer, V. The cannabinoid system and pain. Neuropharmacol 2017, 124, 105–120. [Google Scholar] [CrossRef]
- Woolf, C.J.; Ma, Q. Nociceptors—Noxious stimulus detectors. Neuron 2007, 55, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Woolf, N.J.; Butcher, L.L. Cholinergic systems in the rat brain: III. Projections from the pontomesencephalic tegmentum to the thalamus, tectum, basal ganglia, and basal forebrain. Brain Res Bull 1986, 16, 603–637. [Google Scholar] [CrossRef]
- Wu, L.-J.; Ko, S.W.; Zhuo, M. Kainate receptors and pain: From dorsal root ganglion to the anterior cingulate cortex. Curr Pharm Des 2007, 13, 1597–1605. [Google Scholar] [CrossRef]
- Xie, Y.-F.; Huo, F.-Q.; Tang, J.-S. Cerebral cortex modulation of pain. Acta Pharmacol Sin 2009, 30, 31–41. [Google Scholar] [CrossRef]
- Xu, M.; Kim, C.J.; Neubert, M.J.; Heinricher, M.M. NMDA receptor-mediated activation of medullary pro-nociceptive neurons is required for secondary thermal hyperalgesia. Pain 2007, 127, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Jin, L.; Wang, L.; Tang, Y.; Wu, H.; Chen, Q.; Sun, L. The role of gonadal hormones in regulating opioid antinociception. Ann Med 2024, 56, 2329259. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-J.; Hökfelt, T.; Wiesenfeld-Hallin, Z. Galanin and spinal pain mechanisms: Past, present, and future. Exp Suppl 2010, 102, 39–50. [Google Scholar]
- Yamamoto, T.; Nozaki-Taguchi, N.; Chiba, T. Analgesic effect of intrathecally administered orexin-A in the rat formalin test and in the rat hot plate test. Br J Pharmacol 2002, 137, 170–176. [Google Scholar] [CrossRef]
- Yang, L.-N.; Chen, K.; Yin, X.-P.; Liu, D.; Zhu, L.-Q. The comprehensive neural mechanism of oxytocin in analgesia. Curr Neuropharmacol 2022, 20, 147–157. [Google Scholar] [CrossRef]
- Yang, Q.; Wu, Z.-Z.; Li, X.; Li, Z.-W.; Wei, J.-B.; Hu, Q.-S. Modulation by oxytocin of ATP-activated currents in rat dorsal root ganglion neurons. Neuropharmacology 2002, 43, 910–916. [Google Scholar] [CrossRef]
- Yang, S.; Chang, M.C. Chronic pain: Structural and functional changes in brain structures and associated negative affective states. Int J Mol Sci 2019, 20, 3130. [Google Scholar] [CrossRef]
- Ye, D.-W.; Liu, C.; Liu, T.-T.; Tian, X.-B.; Xiang, H.-B. Motor cortex-periaqueductal gray-spinal cord neuronal circuitry may involve in modulation of nociception: A virally mediated transsynaptic tracing study in spinally transected transgenic mouse model. PLoS ONE 2014, 9, e89486. [Google Scholar] [CrossRef] [PubMed]
- Yetnikoff, L.; Laezzi, H.N.; Reichard, R.A.; Zahm, D.S. An update on the connections of the ventral mesencephalic dopaminergic complex. Neuroscience 2014, 282, 23–48. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.-B.; Wu, H.-H.; Dong, Y.-L.; Zhang, T.; Wang, J.; Zhang, Y.; Wei, Y.-Y.; Lu, Y.-C.; Wu, S.-X.; Wang, W.; et al. Neurochemical properties of BDNF-containing neurons projecting to rostral ventromedial medulla in the ventrolateral periaqueductal gray. Front Neural Circuits 2014, 8, 137. [Google Scholar] [CrossRef] [PubMed]
- Yip, J.; Chahl, L.A. Distribution of Fos-like immunoreactivity in guinea-pig brain following administration of the neurokinin-1 receptor agonist, [SAR9,MET(O2)11]substance P. Neuroscience 1999, 94, 663–673. [Google Scholar] [CrossRef]
- Yirmiya, R.; Ben-Eliyahu, S.; Shavit, Y.; Marek, P.; Liebeskind, J.C. Stimulation of the hypothalamic paraventricular nucleus produces analgesia not mediated by vasopressin or endogenous opioids. Brain Res 1990, 537, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.S.; Hwang, I.K.; Lee, I.S.; Suh, J.G.; Shin, J.W.; Kang, T.C.; Oh, Y.S.; Won, M.H. Galanin-immunoreactive cells and their relation to calcitonin gene-related peptide-, substance P- and somatostatin-immunoreactive cells in rat lumbar dorsal root ganglia. Anat Histol Embryol 2003, 32, 110–115. [Google Scholar] [CrossRef]
- Yoshimura, M.; Furue, H. Mechanisms for the anti-nociceptive actions of the descending noradrenergic and serotonergic systems in the spinal cord. J Pharmacol Sci 2006, 101, 107–117. [Google Scholar] [CrossRef]
- Yu, L.-C.; Hou, J.-F.; Fu, F.-H.; Zhang, Y.-X. Roles of calcitonin gene-related peptide and its receptors in pain-related behavioral responses in the central nervous system. Neurosci Biobehav Rev 2009, 33, 1185–1191. [Google Scholar] [CrossRef] [PubMed]
- Zamfir, M.; Sharif, B.; Locke, S.; Ehrlich, A.T.; Ochandarena, N.E.; Scherrer, G.; Ribeiro-da-Silva, A.; Kieffer, B.L.; Séguéla, P. Distinct and sex-specific expression of mu opioid receptors in anterior cingulate and somatosensory S1 cortical areas. Pain 2023, 164, 703–716. [Google Scholar] [CrossRef] [PubMed]
- Zeilhofer, H.U.; Werynska, K.; Gingras, J.; Yévenes, G.E. Glycine receptors in spinal nociceptive control—An update. Biomolecules 2021, 11, 846. [Google Scholar] [CrossRef] [PubMed]
- Zeilhofer, H.U.; Wildner, H.; Yévenes, G.E. Fast synaptic inhibition in spinal sensory processing and pain control. Physiol Rev 2012, 92, 193–235. [Google Scholar] [CrossRef]
- Zeng, F.; Zhang, Q.; Liu, Y.; Sun, G.; Li, A.; Talay, R.S.; Wang, J. AMPAkines potentiate the corticostriatal pathway to reduce acute and chronic pain. Mol Brain 2021, 14, 45. [Google Scholar] [CrossRef]
- Zhang, M.-L.; Wang, H.-B.; Fu, F.-H.; Yu, L.-C. Involvement of galanin and galanin receptor 2 in nociceptive modulation in anterior cingulate cortex of normal rats and rats with mononeuropathy. Sci Rep 2017, 7, 45930. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, D.; Li, H.; Sun, Y.; Jiang, X.; Gong, S.; Qian, Z.; Tao, J. Melanocortin type 4 receptor-mediated inhibition of A-type K+ current enhances sensory neuronal excitability and mechanical pain sensitivity in rats. J Biol Chem 2019, 294, 5496–5507. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Tao, B.; Lv, Q.; Lui, S.; He, L. Gray matter and regional brain activity abnormalities in subclinical hypothyroidism. Front Endocrinol 2021, 12, 582519. [Google Scholar] [CrossRef]
- Zheng, H.; Lim, J.Y.; Kim, Y.; Jung, S.T.; Hwang, S.W. The role of oxytocin, vasopressin, and their receptors at nociceptors in peripheral pain modulation. Front Neuroendocrinol 2021, 63, 100942. [Google Scholar] [CrossRef]
- Zheng, H.; Lim, J.Y.; Seong, J.Y.; Hwang, S.W. The role of corticotropin-releasing hormone at peripheral nociceptors: Implications for pain modulation. Biomedicines 2020, 8, 623. [Google Scholar] [CrossRef]
- Zhu, Q.; Ge, F.; Li, X.; Deng, H.-S.; Xu, M.; Bu, T.; Li, J.; Wang, Y.; Shan, Y.; Ge, R.-S.; et al. Dehydroepiandrosterone antagonizes pain stress-induced suppression of testosterone production in male rats. Front Pharmacol 2018, 9, 322. [Google Scholar] [CrossRef]
- Zhuo, M. Long-term plasticity of NMDA GluN2B (NR2B) receptor in anterior cingulate cortical synapses. Mol Pain 2024, 20, 17448069241230258. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, A.L.; Kovatsis, E.M.; Pozsgai, R.Y.; Tasnim, A.; Zhang, Q.; Ginty, D.D. Distinct modes of presynaptic inhibition of cutaneous afferents and their functions in behavior. Neuron 2019, 102, 420–434.e8. [Google Scholar] [CrossRef] [PubMed]
- Zubieta, J.-K.; Stohler, C.S. Neurobiological mechanisms of placebo responses. Ann N Y Acad Sci 2009, 1156, 198–210. [Google Scholar] [CrossRef] [PubMed]
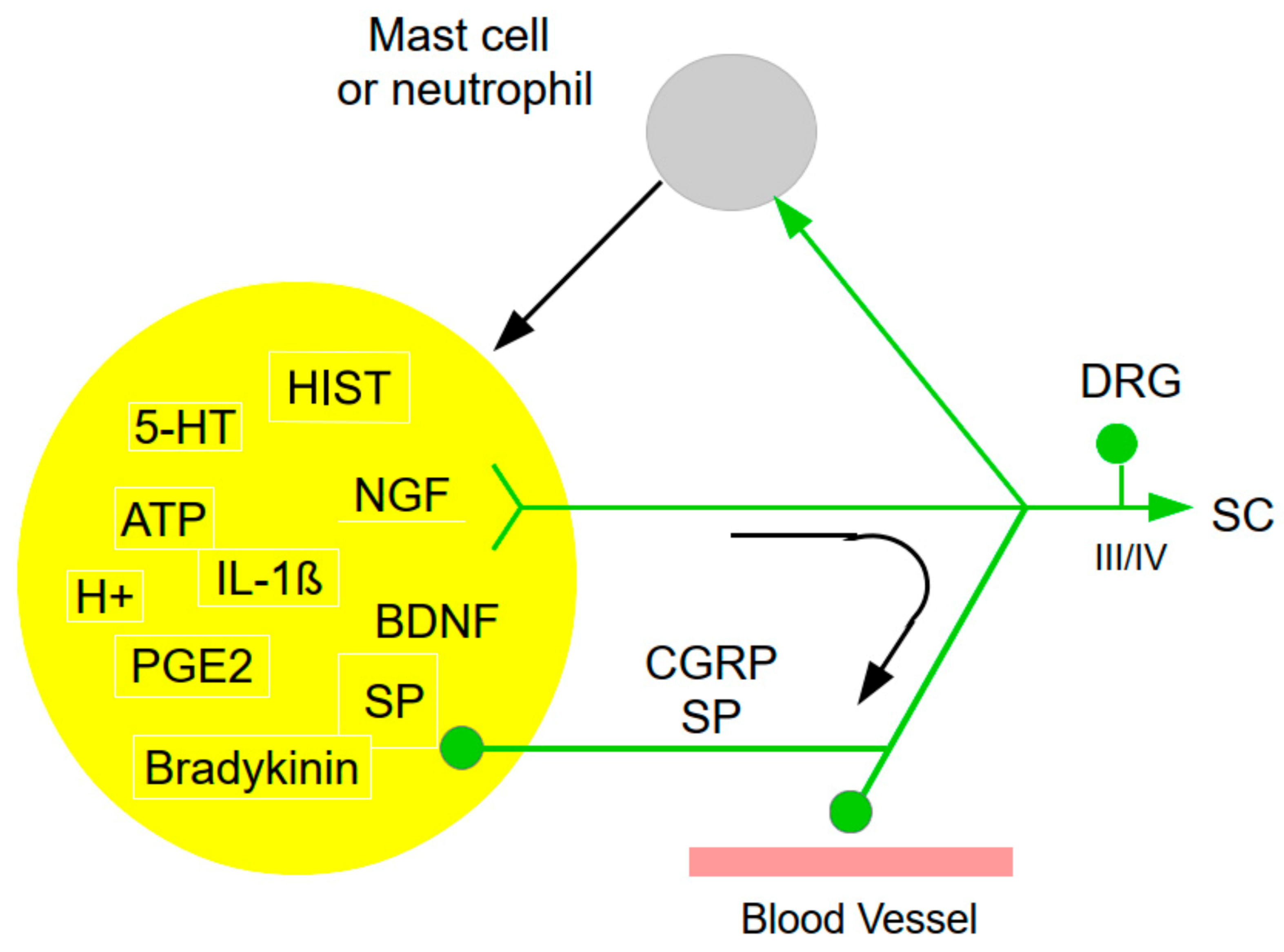
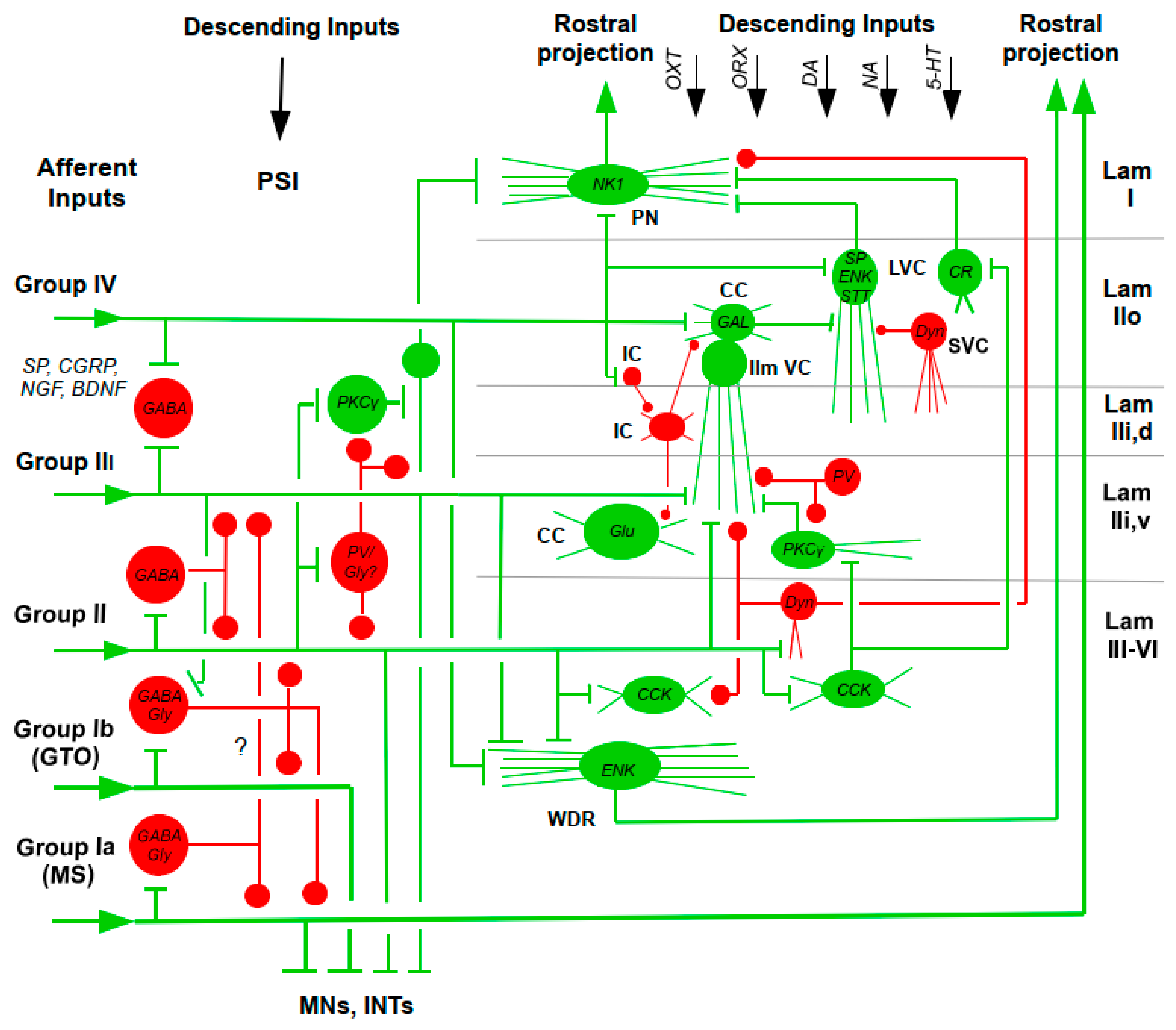
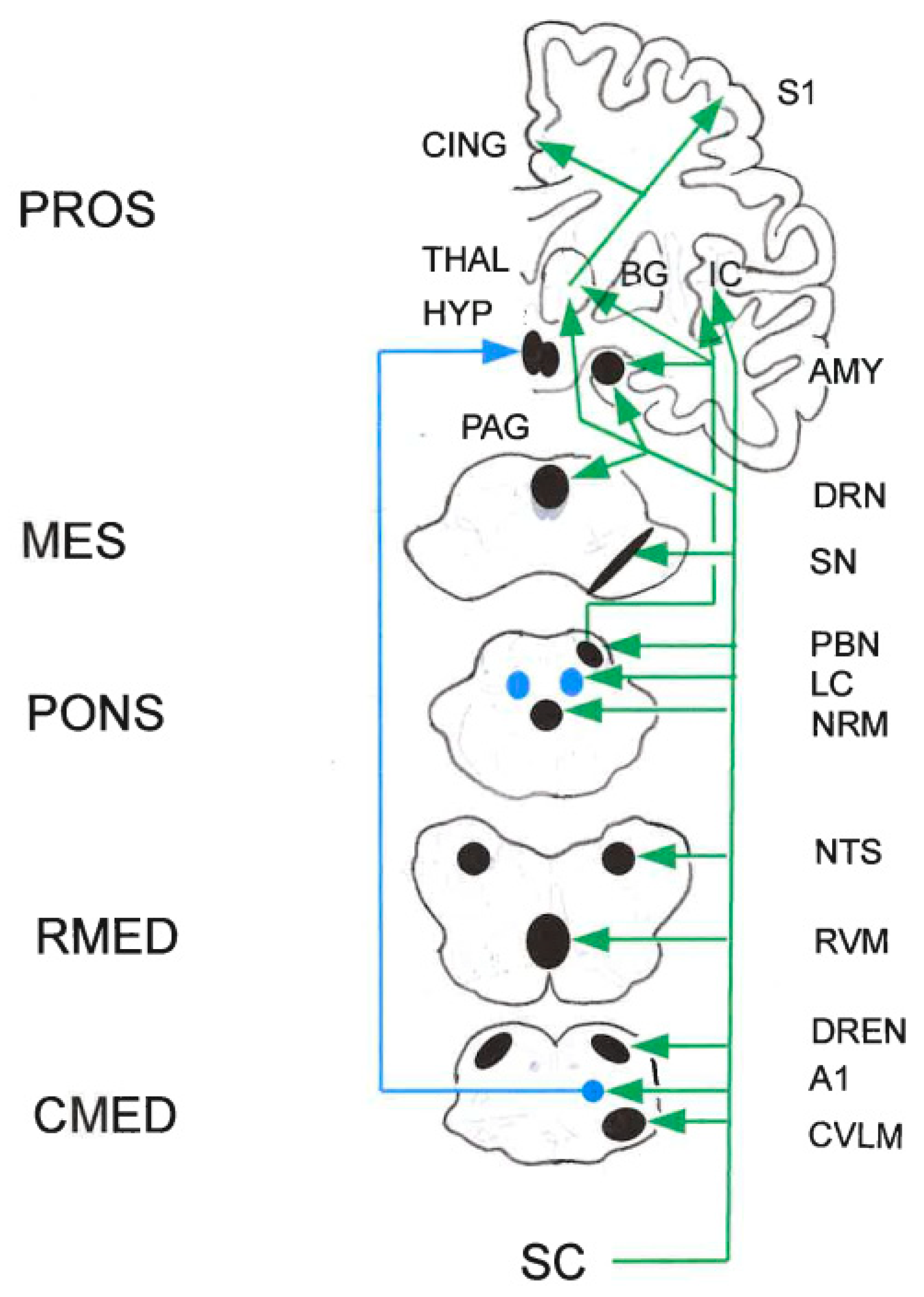
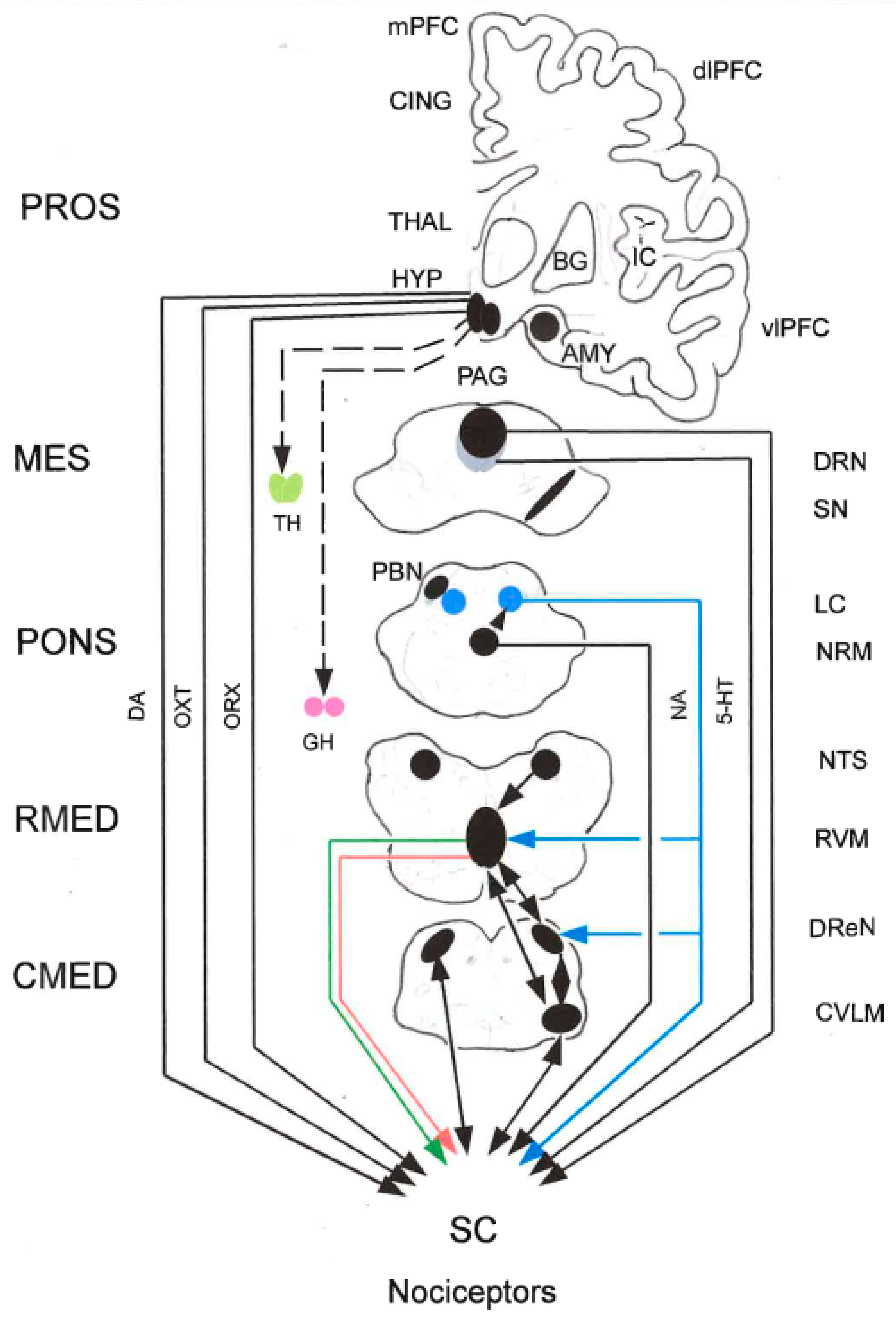
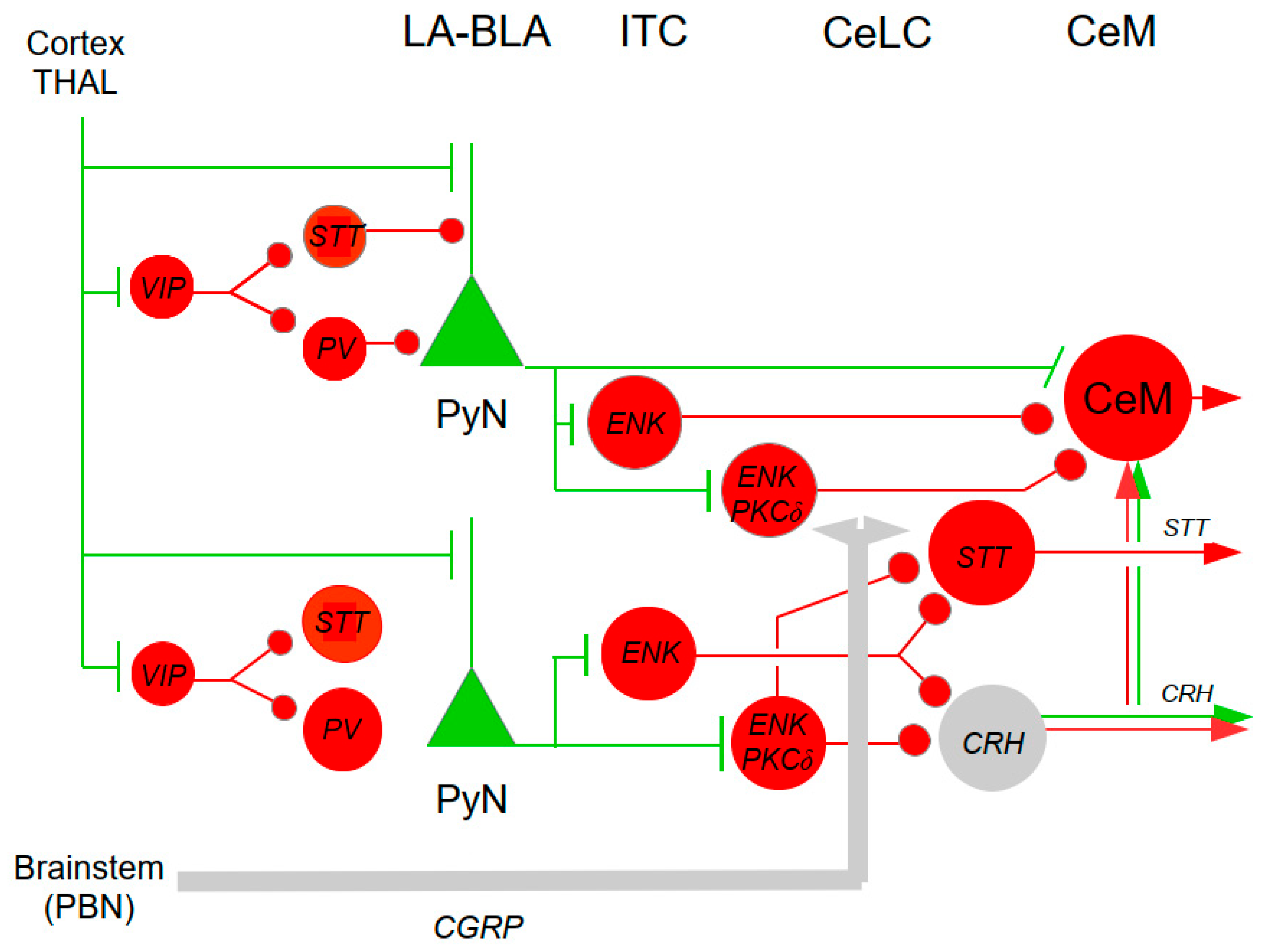
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



