Submitted:
04 August 2025
Posted:
05 August 2025
You are already at the latest version
Abstract
Keywords:
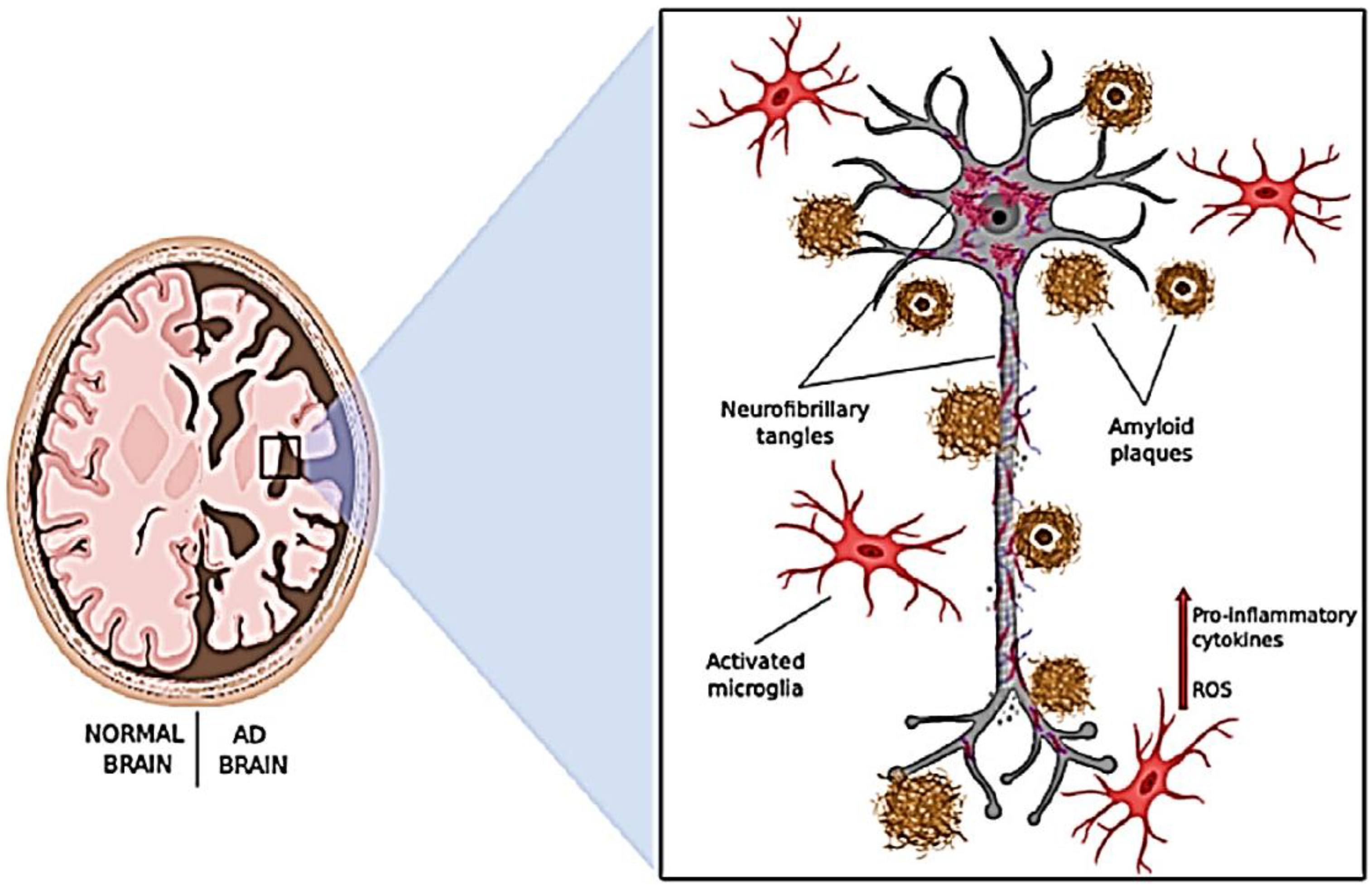
1. Introduction
2. Significance
3. Hypothesis
4. Aims and Objectives
- Evaluate the effectiveness of curcumin, glycitein, kaempferol, squalene, astaxanthin, and β-sitosterol individually against amyloid beta and tau using in silico methods.
- Investigate the synergistic potential of MTCC design by combining identified compounds using merging, fusion, and linking strategies.
- Analyze the interactions between MTCC designs and amyloid beta, tau proteins from the Protein Data Bank (PDB), focusing on binding kinetics and structural modulations.
- Prioritize the most promising MTCC candidates based on their predicted efficacy, safety, and pharmacokinetic profiles, using in silico pharmacodynamics modeling and simulation methods.
- Compare the efficacy of MTCC designs against amyloid beta aggregation and tau pathology, benchmarking results with individual compound assessments.
- Identify potential challenges and limitations of MTCC design and application in AD, paving the way for further advancements and refinements of these promising therapeutic strategies.
5. Methodology
5.1. Compound Selection: Collection and Preparation of Ligand Structures
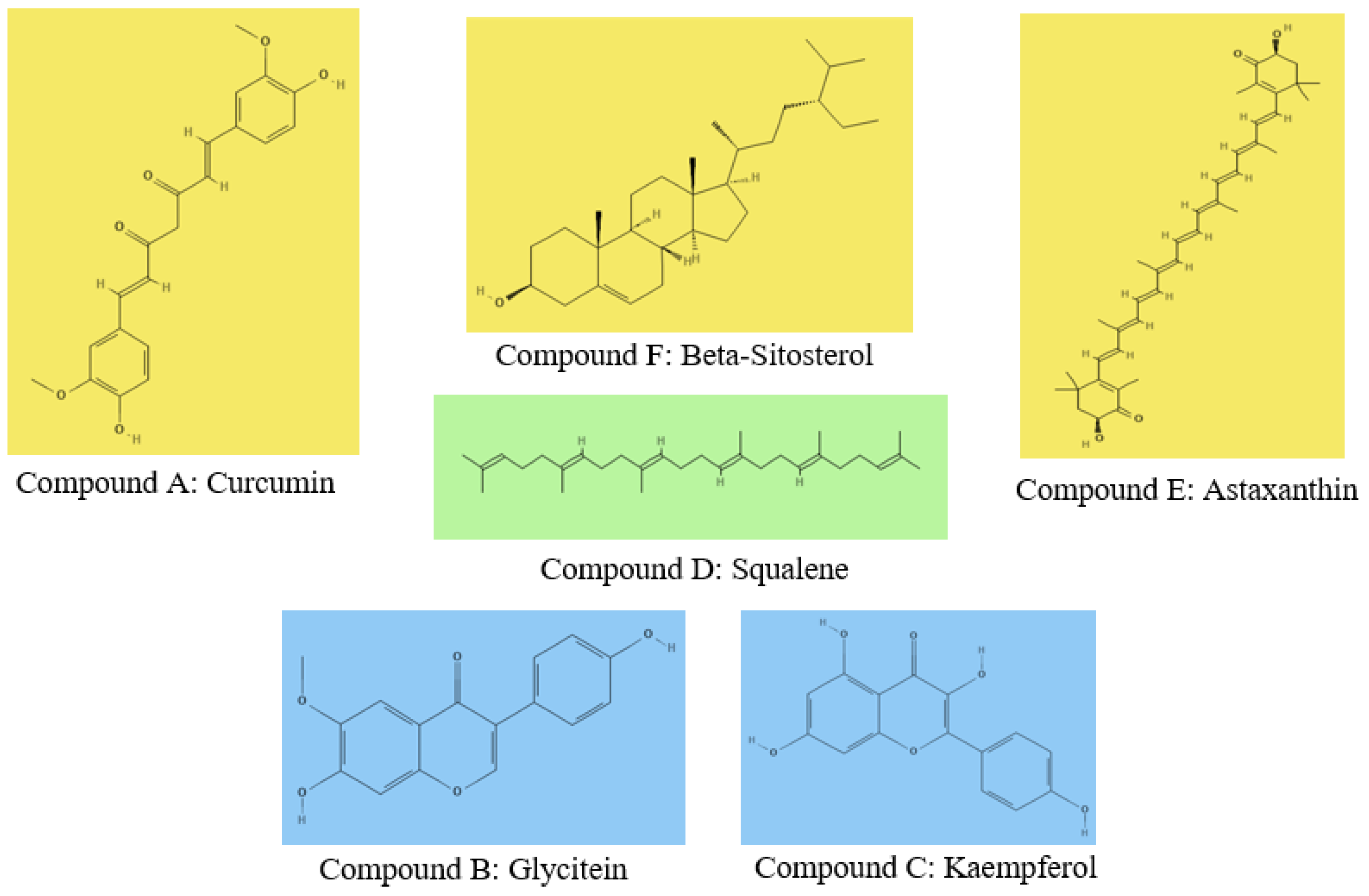
5.2. Target Identification: Collection and Preparation of 3D Structure
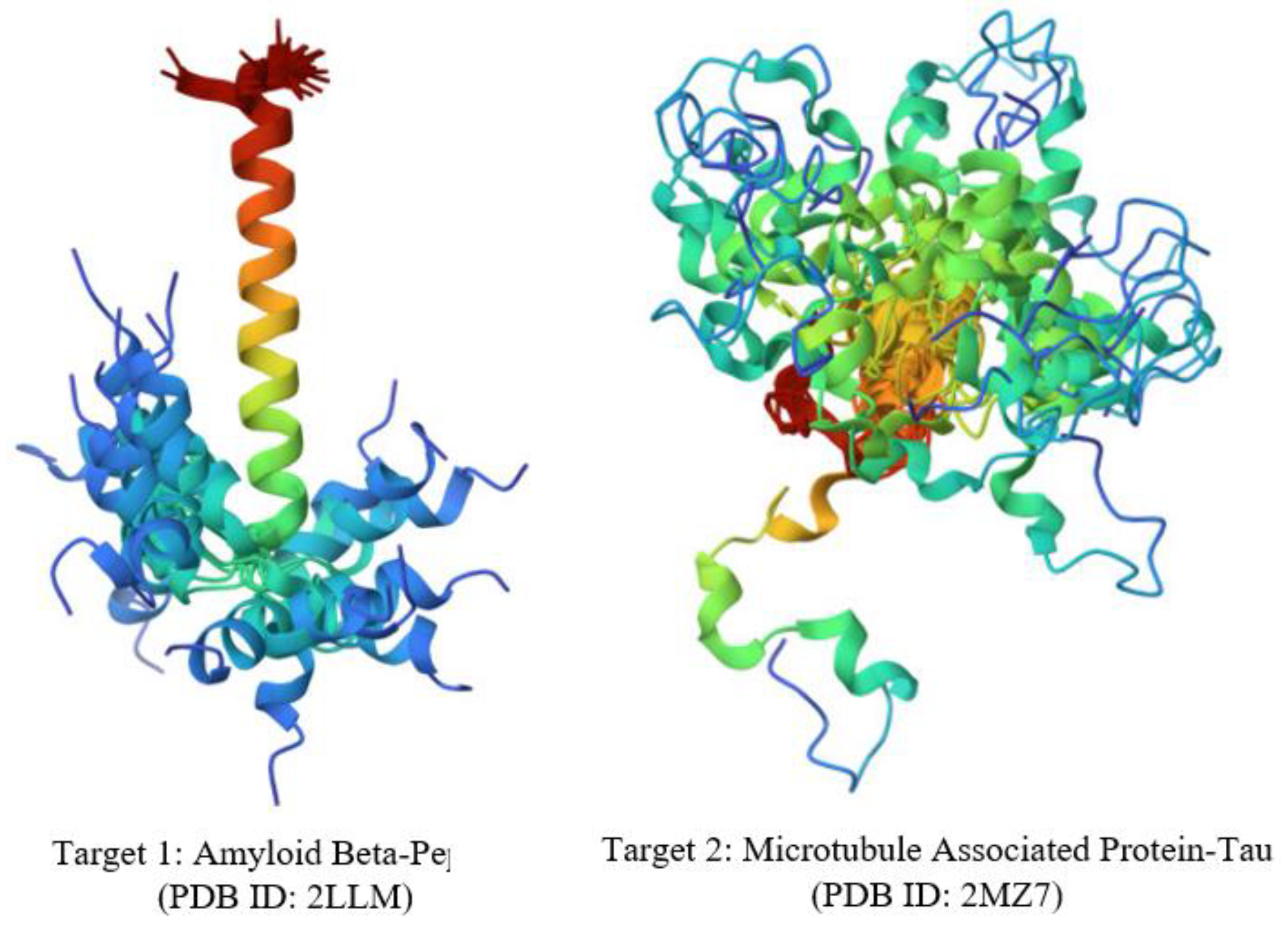
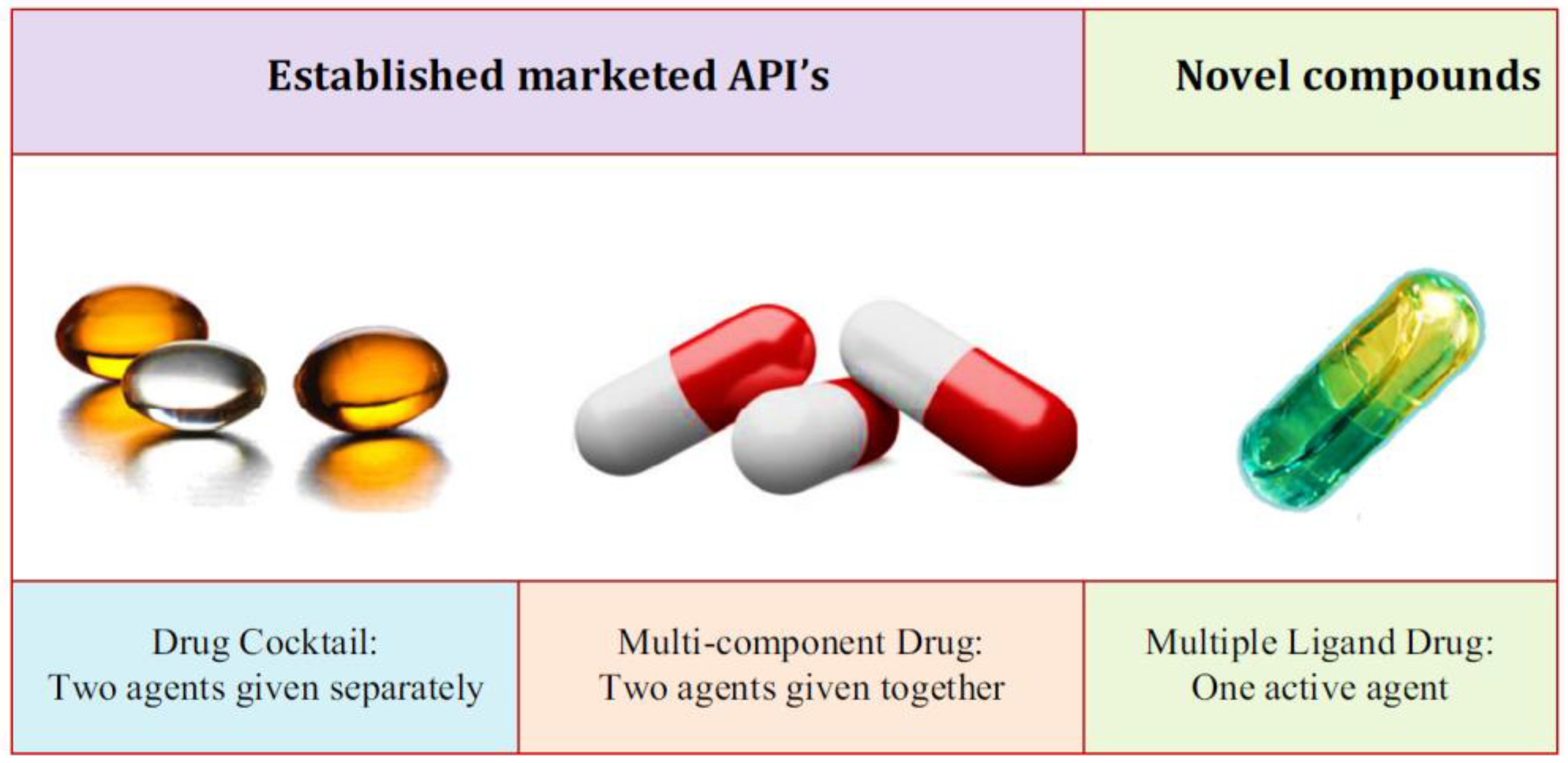
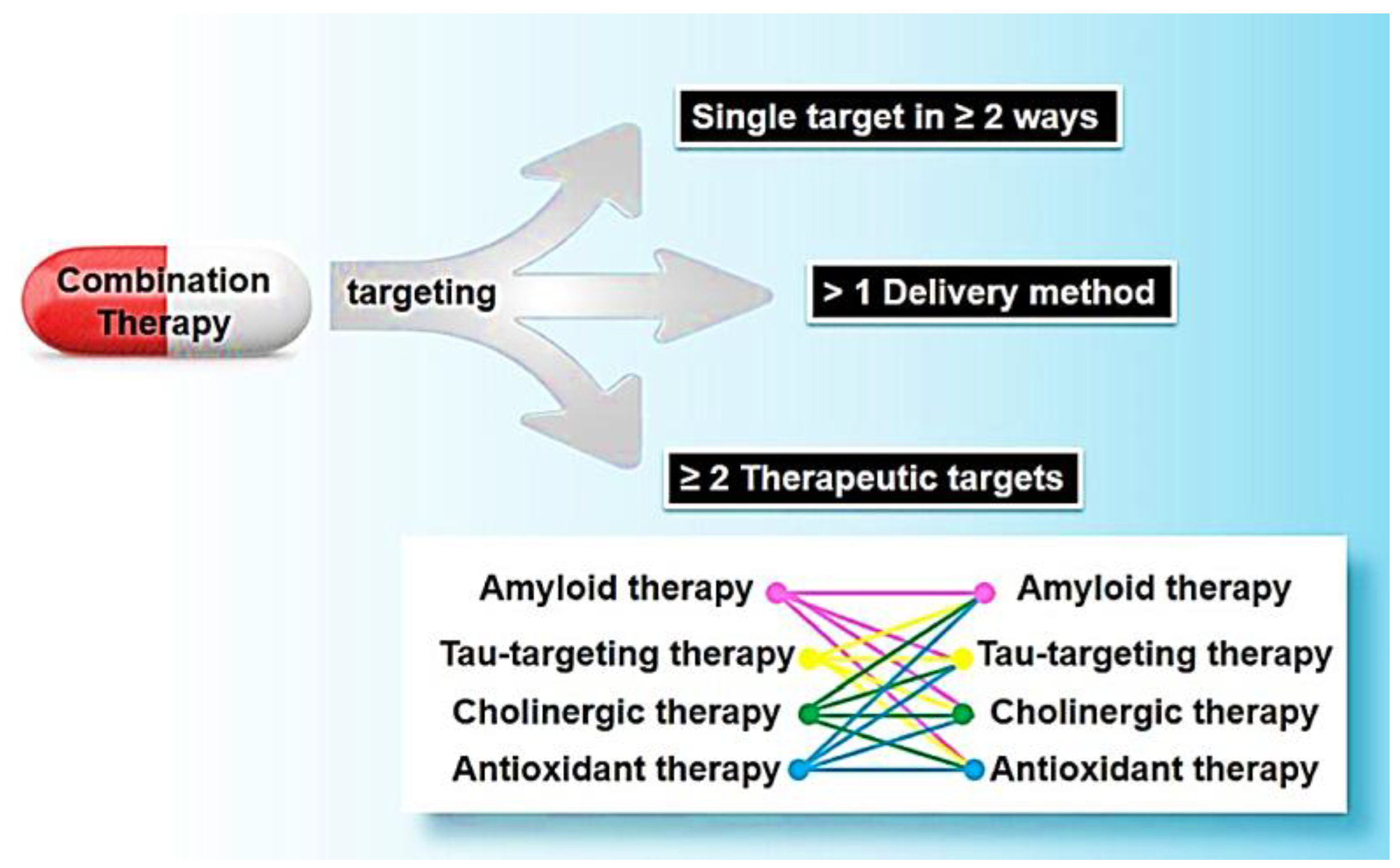
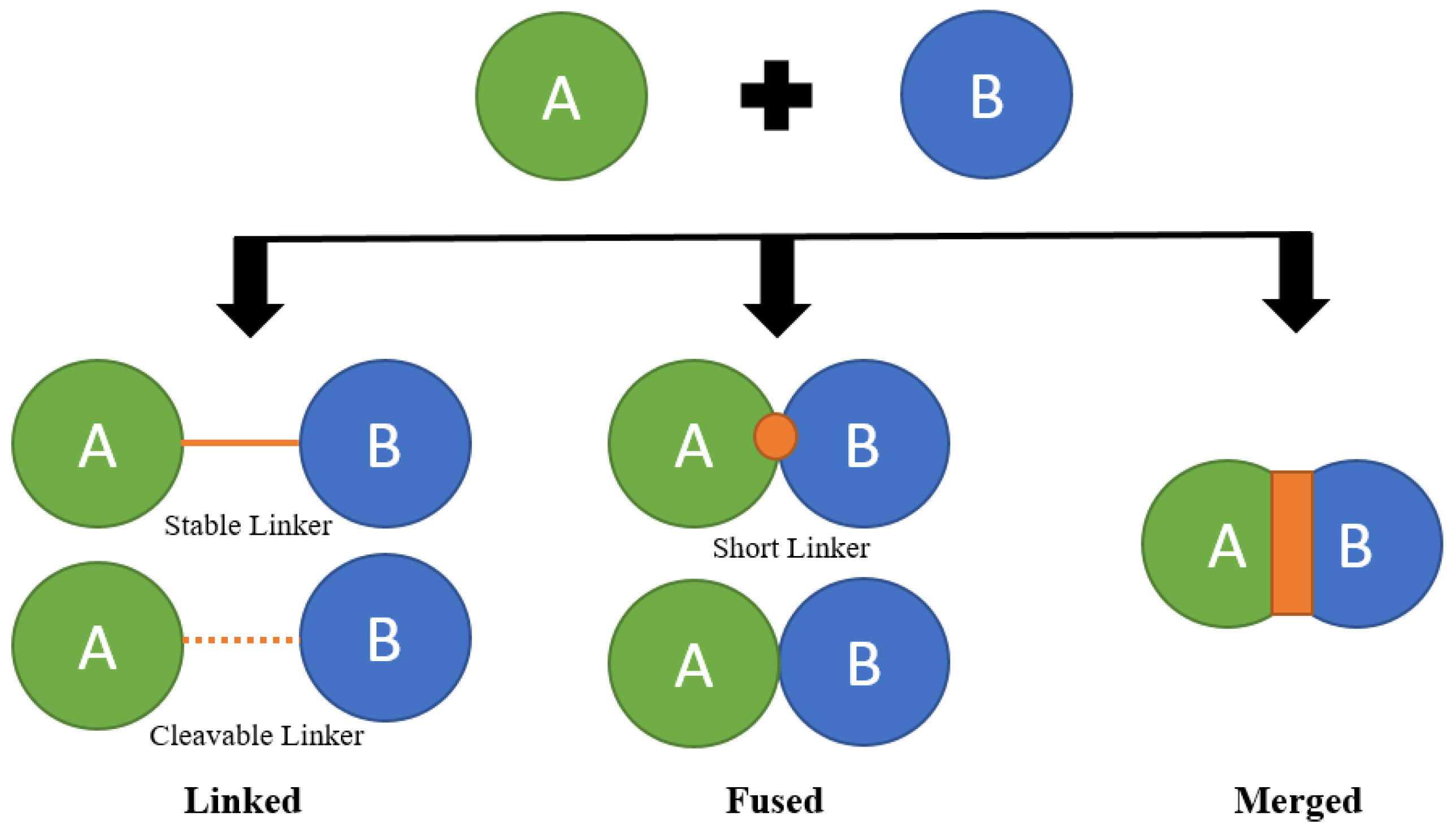
5.3. Approaches for Combination: Strategies and Formation
5.4. Molecular Docking:
5.5. Molecular Dynamics Simulation:
5.6. Pharmacokinetics Analysis:
6. Data Analysis
7. Expected Results
8. Conclusion
9. Future Prospects
Funding
Conflict of Interest Statement
References
- Salloway, S.; Farlow, M.; McDade, E.; Clifford, D.B.; Wang, G.; Llibre-Guerra, J.J.; Hitchcock, J.M.; Mills, S.L.; Santacruz, A.M.; Aschenbrenner, A.J.; et al. A trial of gantenerumab or solanezumab in dominantly inherited Alzheimer’s disease. Nat. Med. 2021, 27, 1187–1196. [Google Scholar] [CrossRef]
- Nguyen, T.T.D.; Vo, T.K.; Vo, V.G. Advances in developing therapeutic strategies for Alzheimer's disease. Biomed. Pharmacother. 2021, 139, 111623. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H.; Li, R.; Sterling, K.; Song, W. Amyloid β-based therapy for Alzheimer’s disease: challenges, successes and future. Signal Transduct Target Ther. 2023, 8, 1–26. [Google Scholar]
- Liu, L.; Zhao, S.; Chen, H.; Wang, A. A new machine learning method for identifying Alzheimer's disease. Simul. Model. Pr. Theory 2020, 99. [Google Scholar] [CrossRef]
- Cummings, J.; Lee, G.; Zhong, K.; Fonseca, J.; Taghva, K. Alzheimer’s disease drug development pipeline: 2021. Alzheimer’s Dement Transl Res Clin Interv. 2021, 7, 1–24. [Google Scholar]
- Jahn, H. Memory loss in alzheimer’s disease. Dialogues Clin Neurosci. 2013, 15, 445–454. [Google Scholar]
- Schachter, A.S.; Davis, K.L. Alzheimer’s disease. 2022, 2000.
- Abeysinghe, A.A.D.T.; Deshapriya, R.D.U.S.; Udawatte, C. Alzheimer's disease; a review of the pathophysiological basis and therapeutic interventions. Life Sci. 2020, 256, 117996. [Google Scholar] [CrossRef]
- Bellenguez, C.; Grenier-Boley, B.; Lambert, J.C. Genetics of Alzheimer’s disease: where we are, and where we are going. Curr Opin Neurobiol. 2020, 61, 40–48. [Google Scholar]
- Santiago, J.A.; Potashkin, J.A. The Impact of Disease Comorbidities in Alzheimer’s Disease. Front Aging Neurosci. 2021, 13, 1–13. [Google Scholar]
- Das, N.; Raymick, J.; Sarkar, S. Role of metals in Alzheimer’s disease. Metab. Brain Dis. 2021, 36, 1627–1639. [Google Scholar] [CrossRef]
- Rabbito, A.; Dulewicz, M.; Kulczyńska-Przybik, A.; Mroczko, B. Biochemical Markers in Alzheimer’s Disease. Int. J. Mol. Sci. 2020, 21, 1989. [Google Scholar] [CrossRef]
- Blaikie, L.; Kay, G.; Maciel, P.; Lin, P.K.T. Experimental modelling of Alzheimer's disease for therapeutic screening. Eur. J. Med. Chem. Rep. 2022, 5. [Google Scholar] [CrossRef]
- Thoe, E.S.; Fauzi, A.; Tang, Y.Q.; Chamyuang, S.; Chia, A.Y.Y. A review on advances of treatment modalities for Alzheimer's disease. Life Sci. 2021, 276, 119129. [Google Scholar] [CrossRef]
- Fan, L.; Mao, C.; Hu, X.; Zhang, S.; Yang, Z.; Hu, Z.; et al. New Insights Into the Pathogenesis of Alzheimer’s Disease. Front Neurol. 2020, 10, 1–12. [Google Scholar]
- Martins, M.M.; Branco, P.S.; Ferreira, L.M. Enhancing the Therapeutic Effect in Alzheimer's Disease Drugs: The role of Polypharmacology and Cholinesterase inhibitors. ChemistrySelect 2023, 8. [Google Scholar] [CrossRef]
- Abdolmaleki, A.; Ghasemi, J.B. Dual-acting of Hybrid Compounds - A New Dawn in the Discovery of Multi-target Drugs: Lead Generation Approaches. Curr. Top. Med. Chem. 2016, 17, 1096–1114. [Google Scholar] [CrossRef]
- Kim, S.; Nam, Y.; Shin, S.J.; Prajapati, R.; Shin, S.M.; Kim, M.-J.; Kim, H.S.; Leem, S.H.; Kim, T.-J.; Park, Y.H.; et al. Dual modulators of aggregation and dissociation of amyloid beta and tau: In vitro, in vivo, and in silico studies of Uncaria rhynchophylla and its bioactive components. Biomed. Pharmacother. 2022, 156, 113865. [Google Scholar] [CrossRef]
- Kumar, A.; Tiwari, A.; Sharma, A. Changing Paradigm from one Target one Ligand Towards Multi-target Directed Ligand Design for Key Drug Targets of Alzheimer Disease: An Important Role of In Silico Methods in Multi-target Directed Ligands Design. Curr. Neuropharmacol. 2018, 16, 726–739. [Google Scholar] [CrossRef]
- Job, N.; Thimmakondu, V.S.; Thirumoorthy, K. In Silico Drug Design and Analysis of Dual Amyloid-Beta and Tau Protein-Aggregation Inhibitors for Alzheimer’s Disease Treatment. Molecules 2023, 28, 1388. [Google Scholar] [CrossRef]
- Kou, X.; Song, L.; Wang, Y.; Yu, Q.; Ju, H.; Yang, A.; Shen, R. Design, synthesis and anti-Alzheimer's disease activity study of xanthone derivatives based on multi-target strategy. Bioorganic Med. Chem. Lett. 2020, 30, 126927. [Google Scholar] [CrossRef]
- Sang, Z.; Wang, K.; Shi, J.; Liu, W.; Cheng, X.; Zhu, G.; Wang, Y.; Zhao, Y.; Qiao, Z.; Wu, A.; et al. The development of advanced structural framework as multi-target-directed ligands for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2020, 192, 112180. [Google Scholar] [CrossRef]
- Neto, L.R.d.S.; Moreira-Filho, J.T.; Neves, B.J.; Maidana, R.L.B.R.; Guimarães, A.C.R.; Furnham, N.; Andrade, C.H.; Silva, F.P. In silico Strategies to Support Fragment-to-Lead Optimization in Drug Discovery. Front. Chem. 2020, 8, 93. [Google Scholar] [CrossRef]
- Caldwell, G.W. In silicotools used for compound selection during target-based drug discovery and development. Expert Opin. Drug Discov. 2015, 10, 901–923. [Google Scholar] [CrossRef]
- Athar, T.; Al Balushi, K.; Alam Khan, S. Recent advances on drug development and emerging therapeutic agents for Alzheimer’s disease. Mol. Biol. Rep. 2021, 48, 5629–5645. [Google Scholar] [CrossRef]
- Gulisano, W.; Maugeri, D.; Baltrons, M.A.; Fà, M.; Amato, A.; Palmeri, A.; et al. Role of Amyloid-β and Tau Proteins in Alzheimer’s Disease: Confuting the Amyloid Cascade. J Alzheimer’s Dis. 2018, 64, S611–31. [Google Scholar]
- Makhaeva, G.F.; Kovaleva, N.V.; Boltneva, N.P.; Lushchekina, S.V.; Rudakova, E.V.; Stupina, T.S.; Terentiev, A.A.; Serkov, I.V.; Proshin, A.N.; Radchenko, E.V.; et al. Conjugates of tacrine and 1,2,4-thiadiazole derivatives as new potential multifunctional agents for Alzheimer’s disease treatment: Synthesis, quantum-chemical characterization, molecular docking, and biological evaluation. Bioorganic Chem. 2020, 94, 103387. [Google Scholar] [CrossRef]
- Sagar, S.R.; Singh, D.P.; Das, R.D.; Panchal, N.B.; Sudarsanam, V.; Nivsarkar, M.; Vasu, K.K. Pharmacological investigation of quinoxaline-bisthiazoles as multitarget-directed ligands for the treatment of Alzheimer’s disease. Bioorganic Chem. 2019, 89, 102992. [Google Scholar] [CrossRef]
- Najafi, Z.; Mahdavi, M.; Saeedi, M.; Karimpour-Razkenari, E.; Edraki, N.; Sharifzadeh, M.; Khanavi, M.; Akbarzadeh, T. Novel tacrine-coumarin hybrids linked to 1,2,3-triazole as anti-Alzheimer’s compounds: In vitro and in vivo biological evaluation and docking study. Bioorganic Chem. 2019, 83, 303–316. [Google Scholar] [CrossRef]
- Kareti, S.R.; P, S. In Silico Molecular Docking Analysis of Potential Anti-Alzheimer's Compounds Present in Chloroform Extract of Carissa carandas Leaf Using Gas Chromatography MS/MS. Curr. Ther. Res. 2020, 93, 100615. [Google Scholar] [CrossRef]
- Alam, A.; Tamkeen, N.; Imam, N.; Farooqui, A.; Ahmed, M.M.; Tazyeen, S.; et al. Pharmacokinetic and molecular docking studies of plant-derived natural compounds to exploring potential anti-Alzheimer activity. Silico Approach Sustain Agric. 2018, 217–38. Available from: https://link.springer.com/chapter/10.1007/978-981-13-0347-0_13.
- Vanaja, D.; Yellamma, K. Molecular Docking Studies on Evolvulus Alsinoides Compounds Against TAU Protein in Alzheimer’s Disease. Int. J. Sci. Res. 2012, 3, 21–24. [Google Scholar] [CrossRef]
- Sato, R.; Vohra, S.; Yamamoto, S.; Suzuki, K.; Pavel, K.; Shulga, S.; Blume, Y.; Kurita, N. Specific interactions between tau protein and curcumin derivatives: Molecular docking and ab initio molecular orbital simulations. J. Mol. Graph. Model. 2020, 98, 107611. [Google Scholar] [CrossRef]
- Praveen, K.; Yellamma, K. Insilco studies on astaxanthin derivatives against Tau protein - A novel approach to design anti-Alzheimers drug. 2017, 8, 226–31.
- Tibon, N.S.; Ng, C.H.; Cheong, S.L. Current progress in antimalarial pharmacotherapy and multi-target drug discovery. Eur. J. Med. Chem. 2020, 188, 111983. [Google Scholar] [CrossRef]
- Arrué, L.; Cigna-Méndez, A.; Barbosa, T.; Borrego-Muñoz, P.; Struve-Villalobos, S.; Oviedo, V.; Martínez-García, C.; Sepúlveda-Lara, A.; Millán, N.; Montesinos, J.C.E.M.; et al. New Drug Design Avenues Targeting Alzheimer’s Disease by Pharmacoinformatics-Aided Tools. Pharmaceutics 2022, 14, 1914. [Google Scholar] [CrossRef]
- Umar, T.; Meena, R.; Mustehasan; Kumar, P.; Khan, A.A. Recent updates in the development of small molecules as potential clinical candidates for Alzheimer's disease: A review. Chem. Biol. Drug Des. 2022, 100, 674–681. [Google Scholar] [CrossRef]
- Kukkarasapalli, P.; Kuna, Y. Docking Studies on Ache and Tau Proteins with Marine Bioactive Compound Squalene, A New Approach to Design Anti-Alzheimer’s Drug Targets. Int. J. Pharm. Sci. Rev. Res. 2021, 70. [Google Scholar] [CrossRef]
- Mantile, F.; Trovato, M.; Santoni, A.; Barba, P.; Ottonello, S.; De Berardinis, P.; et al. Alum and squalene-oil-in-water emulsion enhance the titer and avidity of anti-aβ antibodies induced by multimeric protein antigen (1-11)E2, preserving the Igg1-skewed isotype distribution. PLoS One 2014, 9. [Google Scholar]
- Yang, H-Q, Sun, Z-K, Chen, S-D. Current advances in the treatment of Alzheimer’s disease: focused on considerations targeting Aβ and tau. Transl Neurodegener. 2012, 1, 1–12.
- Lansdall, C.J. An effective treatment for Alzheimer's disease must consider both amyloid and tau. Biosci. Horizons 2014, 7, hzu002–hzu002. [Google Scholar] [CrossRef]
- Das, S.; Basu, S. Multi-targeting Strategies for Alzheimer's Disease Therapeutics: Pros and Cons. Curr. Top. Med. Chem. 2017, 17, 1–44. [Google Scholar] [CrossRef]
- Plotkin, S.S.; Cashman, N.R. Passive immunotherapies targeting Aβ and tau in Alzheimer's disease. Neurobiol. Dis. 2020, 144, 105010–105010. [Google Scholar] [CrossRef]
- Benek, O.; Korabecny, J.; Soukup, O. A Perspective on Multi-target Drugs for Alzheimer’s Disease. Trends Pharmacol Sci 2020, 41, 434–445. [Google Scholar] [CrossRef]
- Waqas, M.; Halim, S.A.; Alsalman, A.; Khan, A.; Elkord, E.; Al-Harrasi, A. Structure-based small inhibitors search combined with molecular dynamics driven energies for human programmed cell death-1 (PD-1) protein. J. Biomol. Struct. Dyn. 2023, 41, 14771–14785. [Google Scholar] [CrossRef]
- de Ruyck, J.; Brysbaert, G.; Blossey, R.; Lensink, M. Molecular docking as a popular tool in drug design, an in silico travel. Adv. Appl. Bioinform. Chem. 2016, ume 9, 1–11. [Google Scholar] [CrossRef]
- Shaker, B.; Ahmad, S.; Lee, J.; Jung, C.; Na, D. In silico methods and tools for drug discovery. Comput. Biol. Med. 2021, 137, 104851. [Google Scholar] [CrossRef]
- Bulusu, K.C.; Guha, R.; Mason, D.J.; Lewis, R.P.; Muratov, E.; Motamedi, Y.K.; Cokol, M.; Bender, A. Modelling of compound combination effects and applications to efficacy and toxicity: state-of-the-art, challenges and perspectives. Drug Discov. Today 2016, 21, 225–238. [Google Scholar] [CrossRef]
- Sabe, V.T.; Ntombela, T.; Jhamba, L.A.; Maguire, G.E.; Govender, T.; Naicker, T.; Kruger, H.G. Current trends in computer aided drug design and a highlight of drugs discovered via computational techniques: A review. Eur. J. Med. Chem. 2021, 224, 113705. [Google Scholar] [CrossRef]
- Prieto-Martínez, F.D.; López-López, E.; Eurídice Juárez-Mercado, K.; Medina-Franco, J.L. Computational Drug Design Methods—Current and Future Perspectives. Silico Drug Des Repurposing Tech Methodol. 2019, 19–44. [Google Scholar]
- Almihyawi, R.A.H.; Naman, Z.T.; Al-Hasani, H.M.H.; Muhseen, Z.T.; Zhang, S.; Chen, G. Integrated computer-aided drug design and biophysical simulation approaches to determine natural anti-bacterial compounds for Acinetobacter baumannii. Sci. Rep. 2022, 12, 1–14. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



